Submitted:
15 February 2026
Posted:
16 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
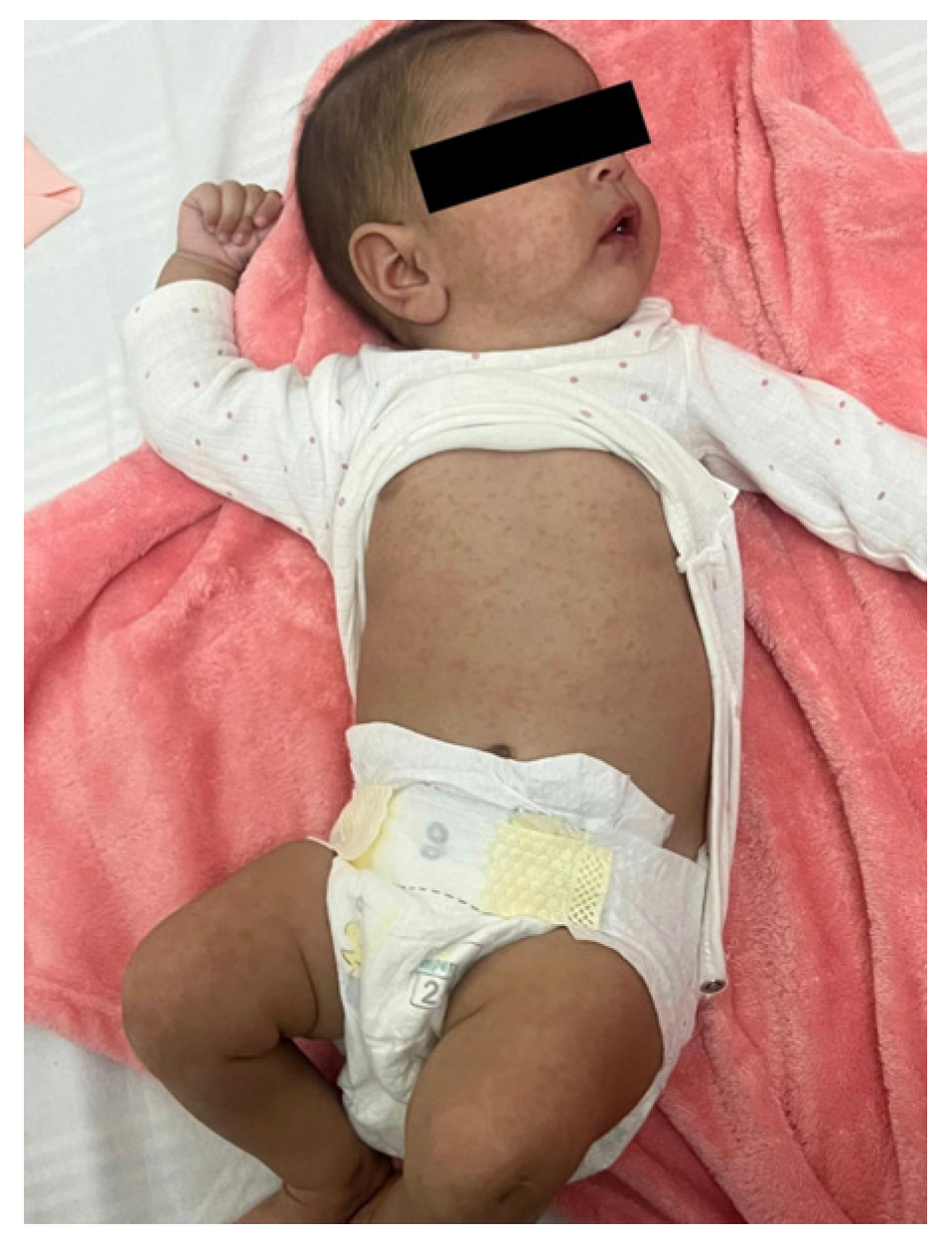
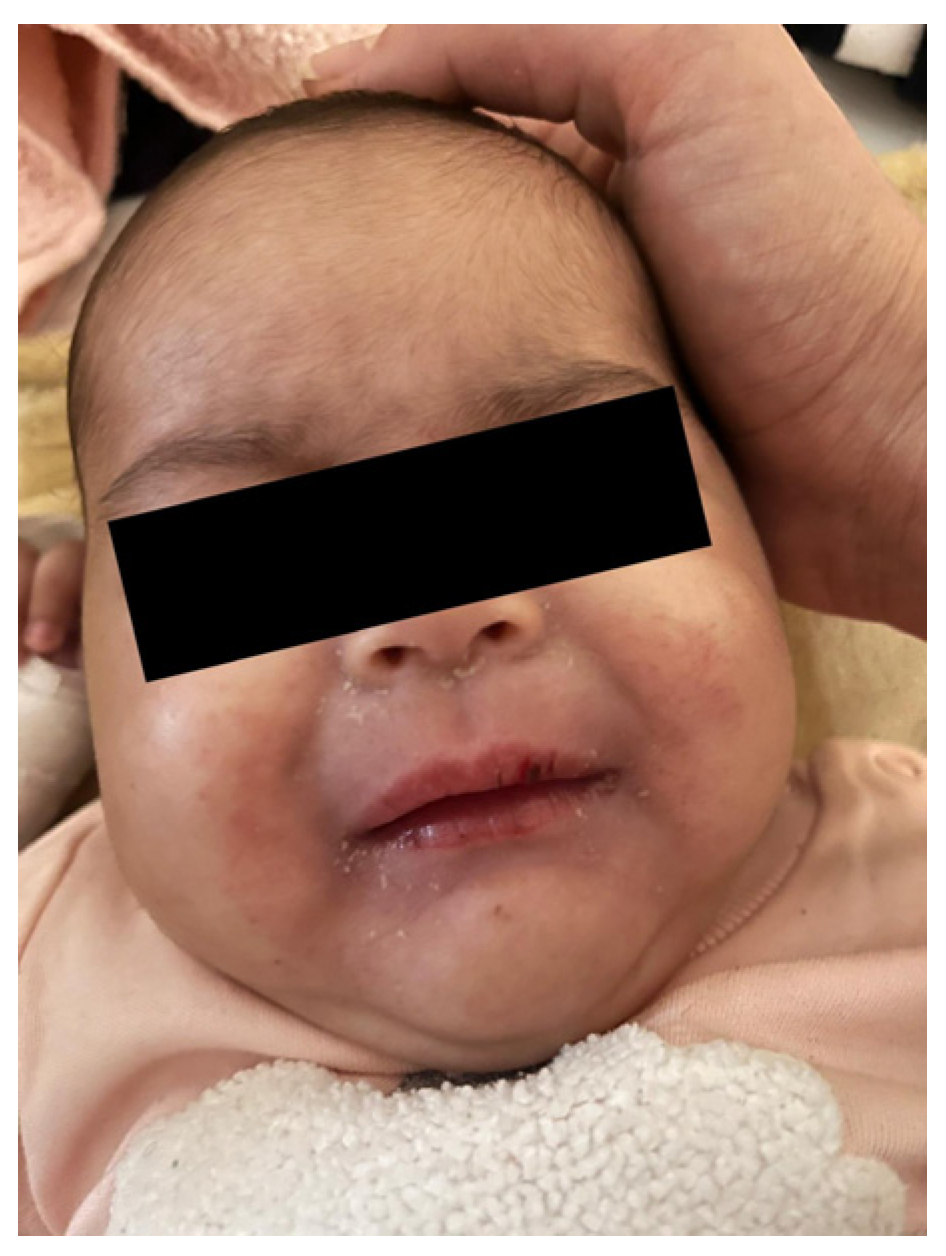
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Tsujioka, Y.; Handa, A.; Nishimura, G.; Miura, M.; Yokoyama, K.; Sato, K.; et al. Multisystem Imaging Manifestations of Kawasaki Disease. RadioGraphics 2022, 42(1), 268–288. [Google Scholar] [CrossRef]
- Kuo, H.C. Diagnosis, Progress, and Treatment Update of Kawasaki Disease. International Journal of Molecular Sciences 2023, 24(18), 13948. [Google Scholar] [CrossRef]
- Weiss, P.F. Pediatric Vasculitis. Pediatr Clin North Am. 2012, 59(2), 407–423. [Google Scholar] [CrossRef]
- Amorim-Figueiredo, R.; Pereira Lemos, A.; Rito, T.; Conde, M.; Brito, M.J.; Pinto, F. Multiresistant Kawasaki Disease in a Young Infant with Giant Aneurysms Growing Fast. J Cardiovasc Dev Dis. 2024, 11(5), 149. [Google Scholar] [CrossRef]
- Zhang, N.; Li, B.; Yan, Y.; Shao, S.; Hua, Y.; Duan, H.; et al. Infant Kawasaki disease complicated with supraventricular tachycardia: a case report and literature review. Pediatric Rheumatology 2025, 23(1), 16. [Google Scholar] [CrossRef]
- Yajima, D.; Shimizu, K.; Oka, K.; Asari, M.; Maseda, C.; Okuda, K.; et al. A Case of Sudden Infant Death Due to Incomplete Kawasaki Disease. J Forensic Sci. 2016, 61(S1), S259–64. [Google Scholar] [CrossRef] [PubMed]
- Jone, P.N.; Tremoulet, A.; Choueiter, N.; Dominguez, S.R.; Harahsheh, A.S.; Mitani, Y.; et al. Update on Diagnosis and Management of Kawasaki Disease: A Scientific Statement From the American Heart Association. Circulation 2024, 150(23), e481–e500. [Google Scholar] [CrossRef]
- Robinson, C.; Chanchlani, R.; Gayowsky, A.; Brar, S.; Darling, E.; Demers, C.; et al. Cardiovascular outcomes in children with Kawasaki disease: a population-based cohort study. Pediatr Res. 2023, 93(5), 1267–75. [Google Scholar] [CrossRef] [PubMed]
- Butters, C.; Curtis, N.; Burgner, D.P. Kawasaki disease fact check: Myths, misconceptions and mysteries. J Paediatr Child Health 2020, 56(9), 1343–1345. [Google Scholar] [CrossRef] [PubMed]
- Fukazawa, R.; Kobayashi, J.; Ayusawa, M.; Hamada, H.; Miura, M.; Mitani, Y.; et al. JCS/JSCS 2020 Guideline on Diagnosis and Management of Cardiovascular Sequelae in Kawasaki Disease. Circulation Journal 2020, 84(8), 1348–1407. [Google Scholar] [CrossRef]
- Tahir, M.; Mehmood, R.; Ahmad, M.S. Complete Kawasaki disease after COVID-19 infection in an infant. Pak J Med Sci. 2023, 39(2), 616–618. [Google Scholar] [CrossRef]
- Onouchi, Y. The genetics of Kawasaki disease. Int J Rheum Dis. 2018, 21(1), 26−30. [Google Scholar] [CrossRef]
- Noval Rivas, M.; Arditi, M. Kawasaki disease: pathophysiology and insights from mouse models. Nat Rev Rheumatol. 2020, 16(7), 391−405. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Feng, J.; Li, N.; Liu, T. Correct identification of incomplete Kawasaki disease. J Int Med Res. 2021, 49(3), 3000605211001712. [Google Scholar] [CrossRef] [PubMed]
- McCrindle, B.W.; Rowley, A.H.; Newburger, J.W.; Burns, J.C.; Bolger, A.F.; Gewitz, M.; et al. Diagnosis, treatment, and long-term management of Kawasaki disease: A scientific statement for health professionals from the American Heart Association. Circulation 2017, 135(17), e927–e999. [Google Scholar] [CrossRef]
- Rădulescu, C.R.; Drăgănescu, A.C.; Băncilă, D.M.; Bilaşco, A.; Bădescu, M.R.; Pleşca, D.A. Contemporary Diagnosis, Management, and Early Outcomes in Children with Kawasaki Disease in Romania: A Single-Center Experience. Diagnostics 2025, 15(6), 656. [Google Scholar] [CrossRef]
- Yeom, J.S.; Woo, H.O.; Park, J.S.; Park, E.S.; Seo, J.H.; Youn, H.S. Kawasaki disease in infants. Korean J Pediatr. 2013, 56(9), 377. [Google Scholar] [CrossRef] [PubMed]
- Lam, J.Y.; Song, M.S.; Kim, G.B.; Shimizu, C.; Bainto, E.; Tremoulet, A.H.; et al. Intravenous immunoglobulin resistance in Kawasaki disease patients: prediction using clinical data. Pediatr Res. 2024, 95(3), 692–697. [Google Scholar] [CrossRef]
- Idris, I.; Awadelkarim, A.M.; Saad, E.; Dayco, J.; Beker, S. Incomplete Kawasaki Disease in an Infant: A Case Report and Literature Review. Cureus 2022, 14(2), e22122. [Google Scholar] [CrossRef]
- Shen, M.; Liu, D.; Ye, F.; Zhang, J.; Wang, J. Kawasaki disease in neonates: a case report and literature review. Pediatric Rheumatology 2024, 22(1), 23. [Google Scholar] [CrossRef]
- Raza, A.A.; Khan, W.; Khan, A.A.; Mahrukh, S.K.; Balasubramanian, K. Atypical Kawasaki Disease Presentation in a Previously Healthy Infant: A Diagnostic Challenge. Cureus 2023, 15(10), e46748. [Google Scholar] [CrossRef]
- Ganeva, M.; Vasilev, T.; Temelkova, K.; Hristova, D.; Dimitrova, A.; Stefanov, S. Kawasaki disease with giant coronary aneurysms formation in a 3-month old infant: a case report and review of the literature. Biotechnology & Biotechnological Equipment 2024, 38(1), 2386071. [Google Scholar] [CrossRef]
- Marchesi, A.; Rigante, D.; Cimaz, R.; Ravelli, A.; Tarissi de Jacobis, I.; Rimini, A.; et al. Revised recommendations of the Italian Society of Pediatrics about the general management of Kawasaki disease. Ital J Pediatr. 2021, 47(1), 16. [Google Scholar] [CrossRef]
- Ren, Y. An Infant With Kawasaki Disease: A Case Report and Literature Review. Cureus 2024, 16(7), e65221. [Google Scholar] [CrossRef] [PubMed]
- Rife, E.; Gedalia, A. Kawasaki Disease: an Update. Curr Rheumatol Rep. 2020, 22(10), 75. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.; Lee, S.E.; Hong, Y.M.; Sohn, S. Is High-Dose Aspirin Necessary in the Acute Phase of Kawasaki Disease? Korean Circ J. 2013, 43(3), 182. [Google Scholar] [CrossRef] [PubMed]

| At admission | Day 7 | Day 9 | Day 11 | Day 14 | Day 16 | Day 18 | At discharge | 2 weeks after discharge | |
| Hb (g/dL) | 10 | 9.2 | 7.8 | 9.1 | 9.4 | 9.9 | 10 | 9.7 | 11.3 |
| PLT (/mm3) | 320,000 | 454,000 | 517,000 | 789,000 | 910,000 | 985,000 | 712,000 | 619,000 | 413,000 |
| CRP (mg/L) | 28 | 116.6 | 124.5 | 22.9 | 3.1 | - | - | - | < 1.0 |
| ESR (mm/h) | - | 130 | - | 110 | - | - | - | - | - |
| Albumin (g/L) | - | 28.8 | - | 29.7 | - | - | - | - | 43.8 |
| D-dimer | - | - | positive | - | positive | - | - | - | - |
| NT-proBNP (pg/mL) | - | - | 2597 | - | - | 155 | - | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




