Submitted:
13 February 2026
Posted:
14 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Population and Design
2.2. Statistical Analyses
3. Results
3.1. Baseline Characteristics
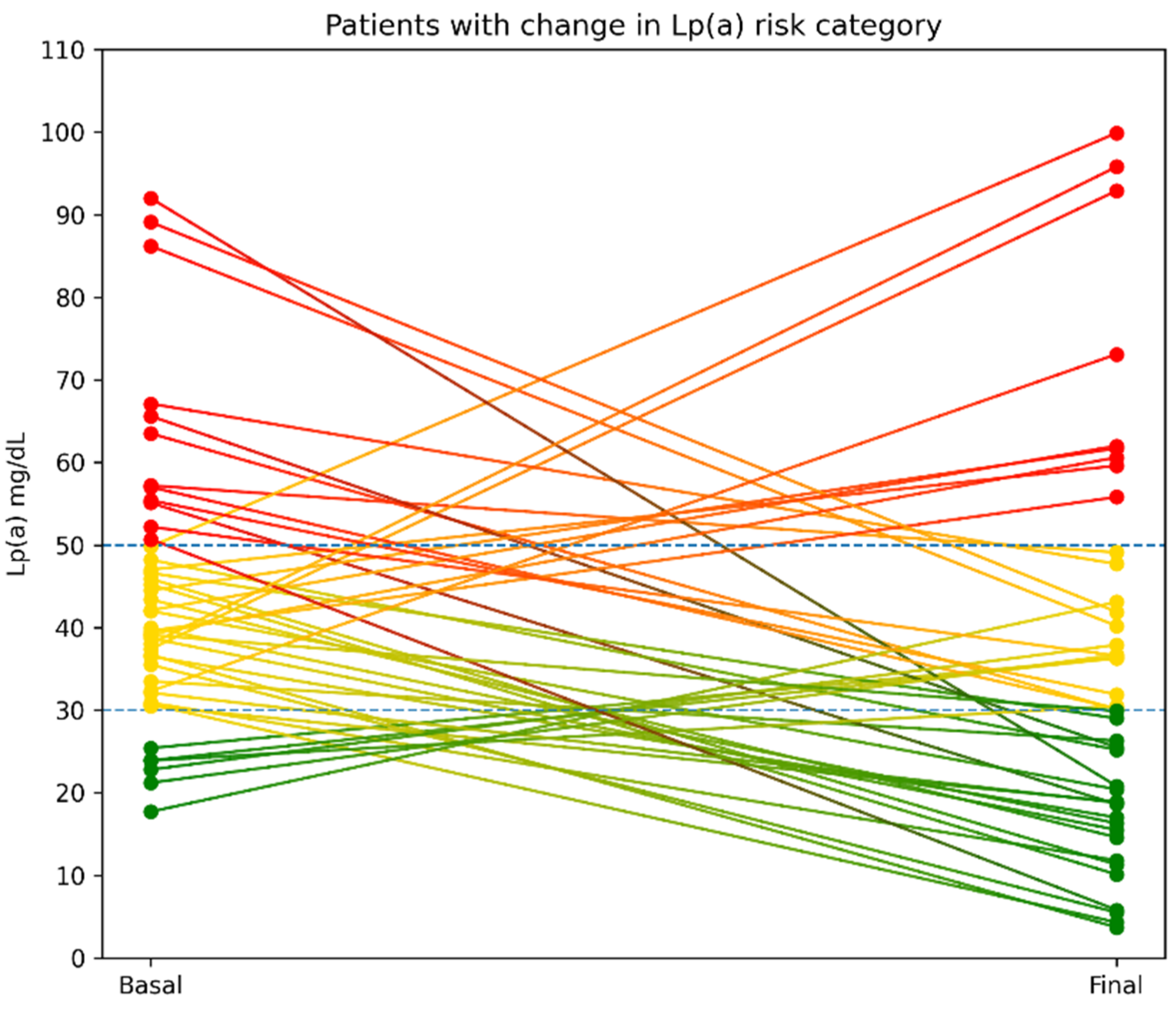
3.2. Intra-Individual Lipoprotein(a) Variability and Clinical Characteristics Stratified by Lp(a) Variability
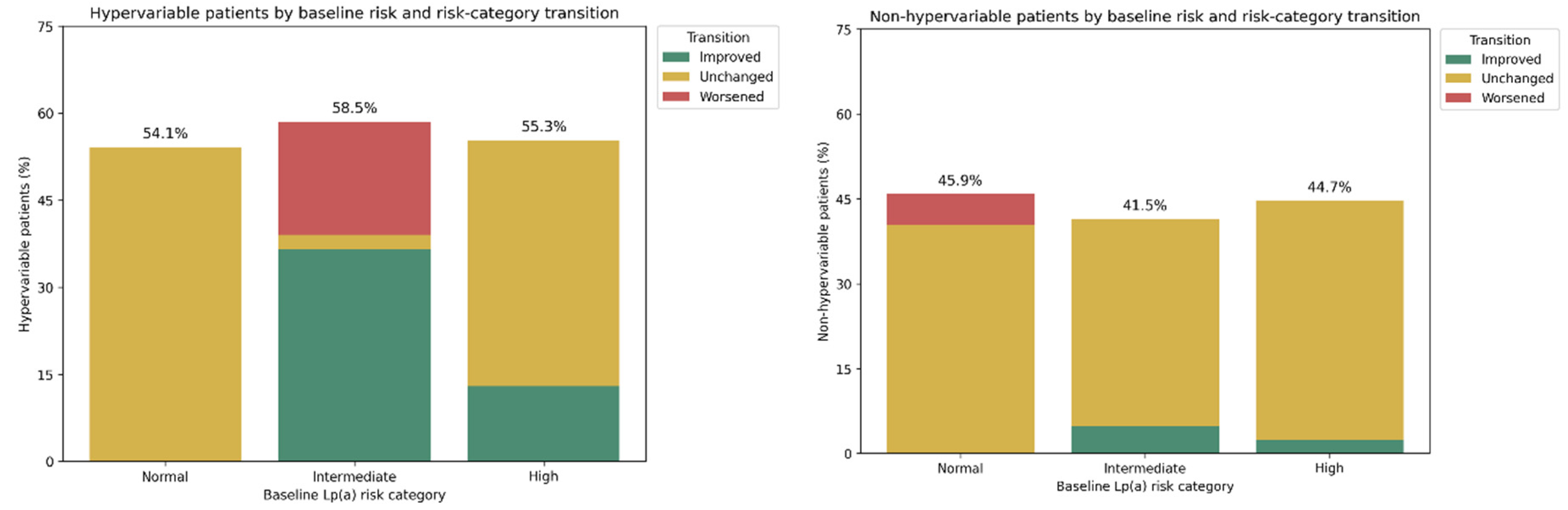
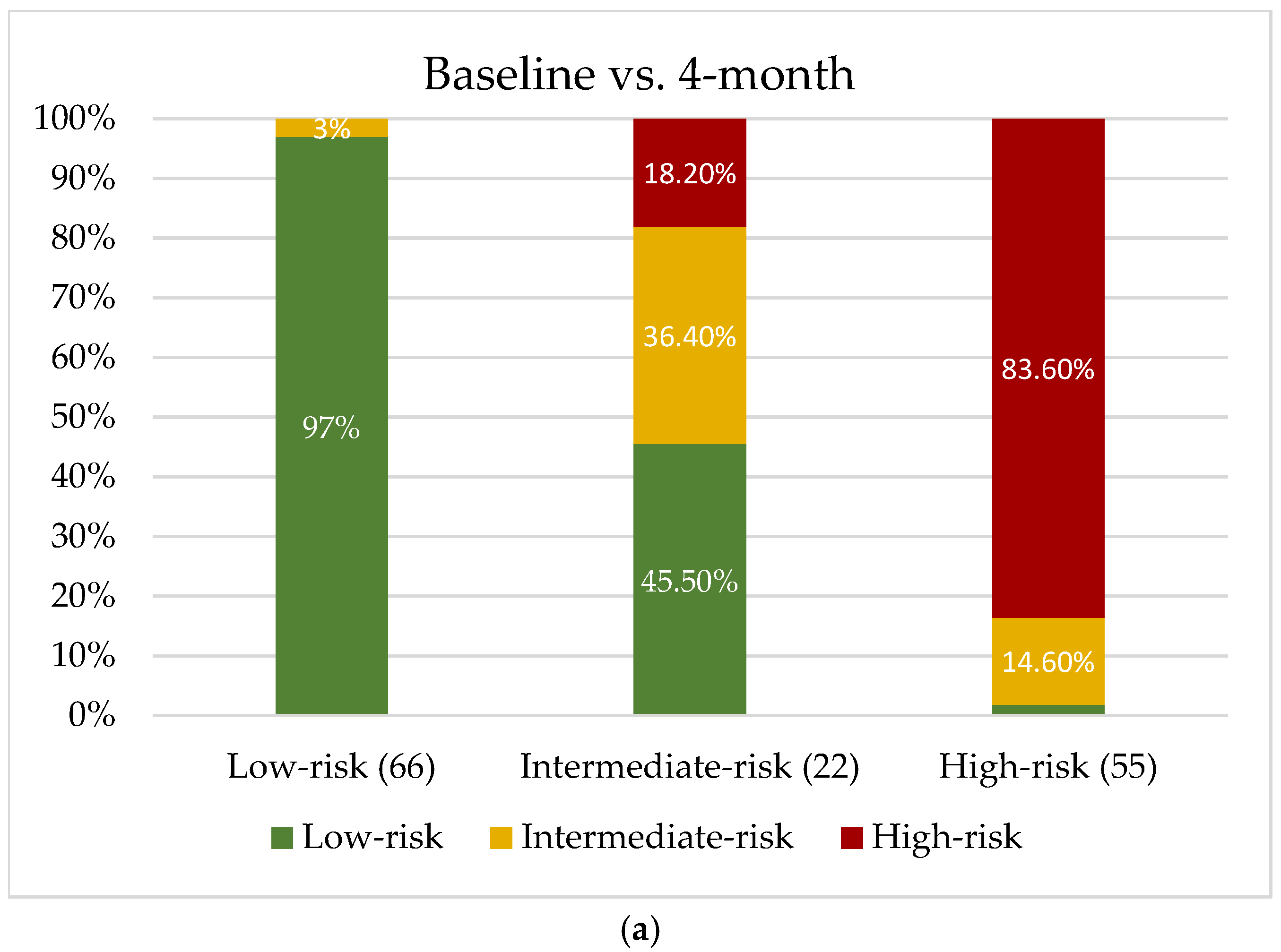
3.3. Transition Between Lp(a) Categories
3.4. Multivariable Logistic Regression Analysis of Factors Independently Associated with High Lp(a) Variability
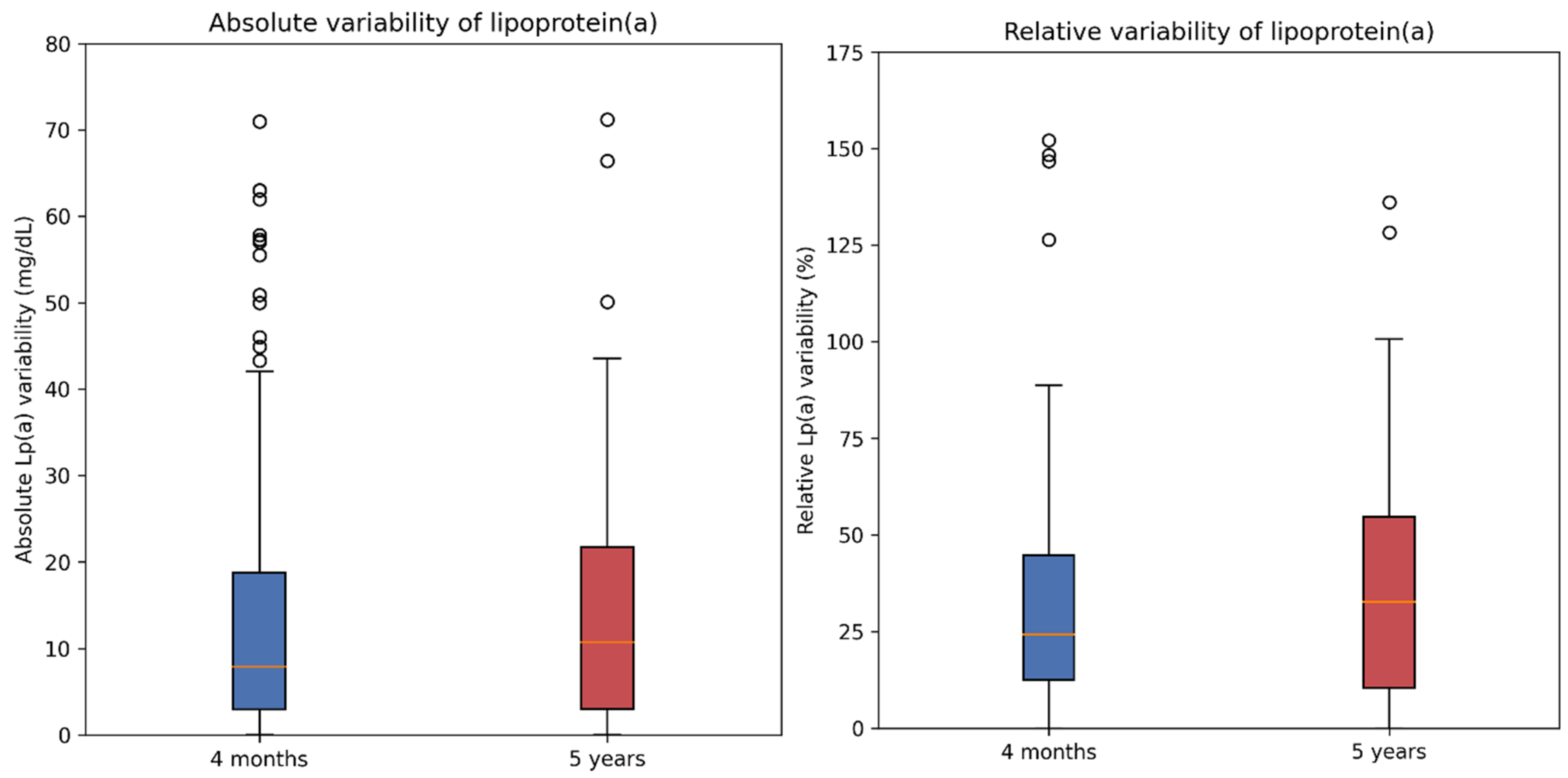
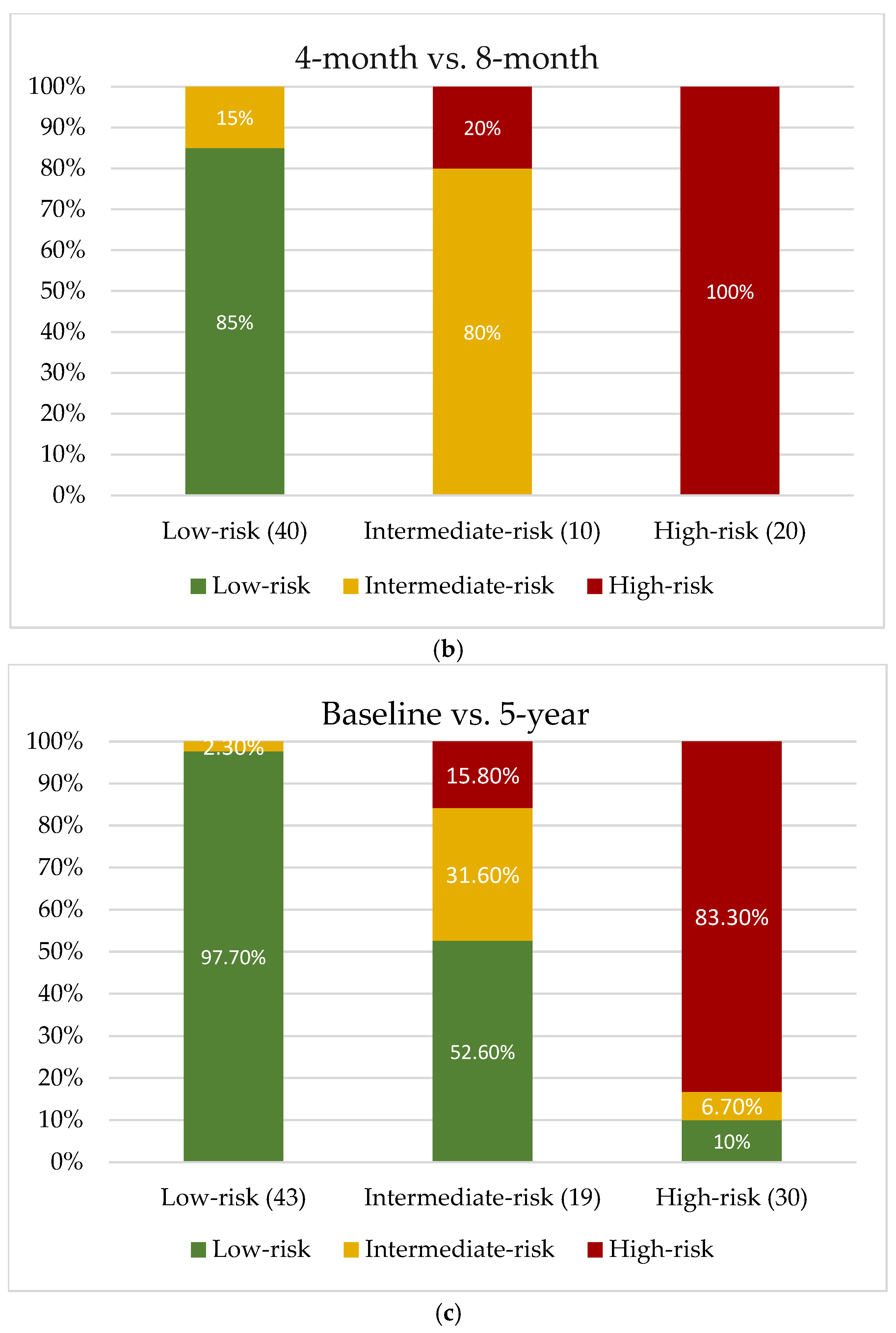
3.5. Variability of Lipoprotein(a) According to the Timing of Determination
4. Discussion
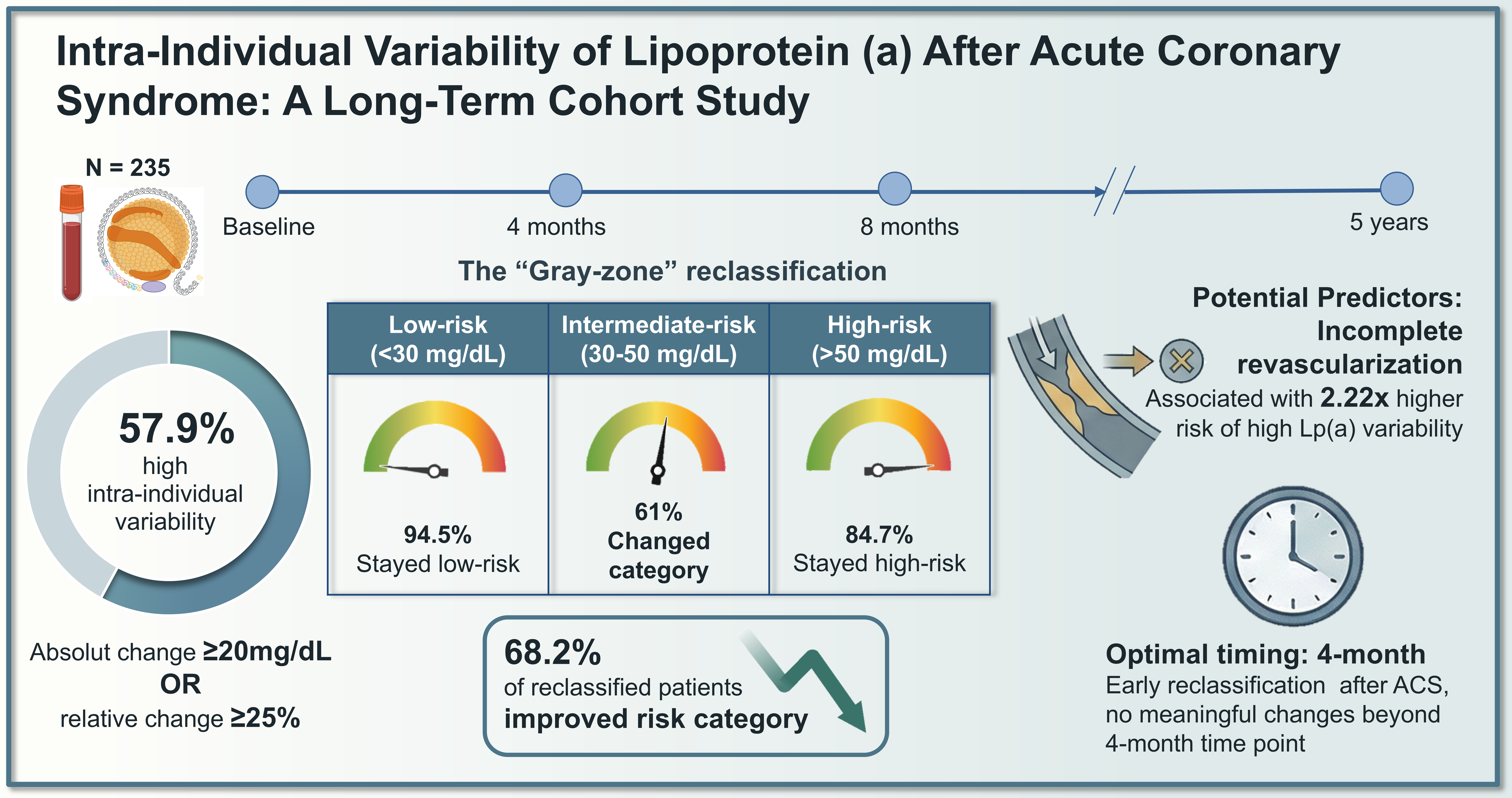
- Substantial intra-individual variability in Lp(a) is common after ACS, affecting nearly six in ten patients when using clinically relevant variability thresholds. Incomplete revascularization, female sex and sex-adjusted menopause were observed as potential predictors of high Lp(a) variability.
- Despite this variability, most patients remain within the same Lp(a) risk category over time, with reclassification concentrated in those with intermediate baseline Lp(a) levels, whereas patients starting in the low-risk range were essentially never reclassified at high risk.
- Reclassification patterns were broadly comparable across reassessment timepoints after ACS, with similarly high category concordance and similar proportions of patients changing risk category whether Lp(a) was remeasured early or at longer-term follow-up.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Lp(a) | Lipoprotein(a) |
| LDL-C | Low-Density Lipoprotein Cholesterol |
| Apo(a) | Apolipoprotein(a) |
| ApoB-100 | Apolipoprotein B-100 |
| ASCVD | Atherosclerotic Cardiovascular Disease |
| ACS | Acute Coronary Syndrome |
| PCI | Percutaneous Coronary Intervention |
| MACE | Major Adverse Cardiovascular Events |
| MI | Myocardial Infarction |
| OR | Odds Ratio |
| CI | Confidence Interval |
| IQR | Interquartile Range |
| hsCRP | High-Sensitivity C-Reactive Protein |
| HR | Hazard Ratio |
| CAD | Coronary Artery Disease |
Appendix A
Appendix A.1 Inclusion and Exclusion Criteria
Appendix B
| Cohort A (N = 143) | Cohort B (N = 92) | |
| Age, years | 67.0 (58.4-74.9) | 56.0 (52.0-60.0) |
| Male sex | 111 (77.6) | 75 (81.5) |
| Diabetes mellitus | 62 (43.7) | 22 (23.9) |
| Hypertension | 99 (69.2) | 49 (53.3) |
| Dyslipidemia | 108 (75.5) | 66 (71.7) |
| Menopausal status | 31 (32) | 13 (17) |
| Body mass index, kg/m² | 27.6 (25.3-30.9) | 28.4 (25.5-31.8) |
| Stroke | 7 (4.9) | 1 (1.1) |
| Transient ischemic attack | 2 (1.4) | 1 (1.1) |
| Peripheral artery disease | 12 (8.4) | 1 (1.1) |
| Prior MI | 40 (28.0) | 10 (10.9) |
| Prior PCI | 39 (27.3) | 4 (4.3) |
| Chronic kidney disease | 106 (74.1) | 12 (13.0) |
| Smoking status (Former or Current) | 103 (73) | 76 (82.6) |
| Alcohol consumption (Former or Current) | 14 (9.8) | 8 (8.7) |
| STEMI presentation | 76 (53.1) | 50 (54.3) |
| Number of treated lesions | 2.0 (1.1) | 1.8 (1.0) |
| Left main treated | 12 (8.4) | 3 (3.3) |
| Number of treated vessels | 1.7 (0.8) | 1.7 (0.9) |
| Incomplete revascularization | 24 (16.8) | 37 (40.2) |
| High-intensity statin | 141 (98.6) | 92 (100) |
| Ezetimibe | 85 (59.4) | 27 (29.3) |
| Total cholesterol, mg/dL | 145.0 (113.5-179.0) | 180.0 (150.5-211.5) |
| HDL-C, mg/dL | 34.0 (31.0-39.0) | 37.5 (32.0-42.5) |
| LDL-C, mg/dL | 83.0 (55.5-114.5) | 112.5 (89.5-138.0) |
| Non–HDL-C, mg/dL | 109.0 (83.0-143.0) | 142.5 (114.0-164.5) |
| Remnant-C, mg/dL | 25.0 (20.0-33.0) | 28.0 (22.0-37.0) |
| TG, mg/dL | 140.0 (110.0-185.5) | 139.0 (108.0-178.0) |
| VLD, mg/dL | 28.0 (22.0-37.1) | 27.8 (21.6-35.6) |
| TC/HDL ratio | 4.3 (3.3-5.1) | 4.7 (4.2-5.5) |
| LDL/ApoB-100 ratio | 1.0 (0.8-1.1) | 1.1 (0.9-1.2) |
| LDL/HDL ratio | 2.4 (1.6-3.2) | 2.9 (2.3-3.6) |
| TG/HDL ratio | 4.3 (2.9-5.5) | 3.7 (2.9-4.9) |
| ApoB-100, mg/dL | 83.0 (67.0-111.5) | 103.5 (83.0-123.0) |
| Baseline Lp(a) | 36.5 (15.6-83.1) | 30.6 (11.3-62.4) |
| Follow-up Lp(a) | 31.8 (10.0-75.0) | 18.2 (5.4-62.1) |
| Glucose, mg/dL | 111.0 (91.5-155.0) | 111.5 (91.0-154.5) |
| HbA1c, % | 6.0 (5.6-6.8) | 5.9 (5.5-6.3) |
| Creatinine, mg/dL | 1.0 (0.9-1.2) | 0.9 (0.8-1.0) |
| eGFR, mL/min/1.73 m² | 75.0 (58.5-85.0) | 89.0 (80.0-90.0) |
| Urea, mg/dL | 43.0 (37.0-56.0) | 36.5 (31.5-45.0) |
| Uric acid, mg/dL | 5.9 (4.8-7.4) | 6.6 (5.2-7.8) |
| MACE | 7 (4.9) | 20 (21.7) |
| Baseline Lp(a) risk category | Normal (< 30 mg/dL) | Intermediate (30–49 mg/dL) | High (≥ 50 mg/dL) | p-value | ||||||
| N (%) | 109 (46.4) | 41 (17.4) | 85 (36.2) | |||||||
| Follow-up Lp(a) risk category | Normal | Intermediate | High | Normal | Intermediate | High | Normal | Intermediate | High | |
| N (%) | 103 (94.5) | 6 (5.5) | 0 | 17 (41.5) | 16 (39.0) | 8 (19.5) |
4 (4.7) | 9 (10.6) | 72 (84.7) |
|
| Age, years, median (IQR) | 59.8 (12.8) | 68.0 (12.4) | - | 64.5 (15.9) | 61.6 (20.9) | 62.0 (9.3) | 56.5 (10.8) | 67.4 (16.9) | 61.0 (22.9) | 0.59 |
| Male sex, n (%) | 82 (79.6) | 4 (66.7) | - | 14 (82.4) | 15 (93.8) | 6 (75.0) | 3 (75.0) | 6 (66.7) | 56 (77.8) | 0.80 |
| Diabetes mellitus, n (%) | 36 (35.0) | 2 (33.3) | - | 10 (58.8) | 7 (46.7) | 3 (37.5) | 0 | 4 (44.4) | 22 (30.6) | 0.32 |
| Hypertension, n (%) | 59 (57.3) | 5 (83.3) | - | 11 (64.7) | 10 (62.5) | 6 (75.0) | 1 (25.0) | 5 (55.6) | 51 (70.8) | 0.37 |
| Dyslipidemia, n (%) | 73 (70.9) | 5 (83.3) | - | 11 (64.8) | 11 (68.8) | 8 (100) | 3 (75.0) | 7 (77.8) | 56 (77.8) | 0.64 |
| Menopausal status, n (%) | 20 (95.2) | 2 (100) | - | 3 (100) | 1 (100) | 2 (100) | 1 (100) | 3 (100) | 12 (75.0) | 0.56 |
| Body mass index, kg/m2, median (IQR) | 28.1 (5.6) | 29.1 (8.7) | - | 28.4 (6.2) | 28.1 (4.2) | 29.2 (5.9) | 25.5 (3.6) | 26.7 (8.0) | 27.6 (7.2) | 0.74 |
| Stroke, n (%) | 0 | 0 | - | 1 (5.9) | 1 (6.3) | 1 (12.5) | 0 | 0 | 5 (6.9) | 0.20 |
| Transient ischemic attack, n (%) | 1 (1.0) | 0 | - | 1 (5.9) | 0 | 0 | 0 | 0 | 1 (1.4) | 0.84 |
| Peripheral artery disease, n (%) | 4 (3.9) | 0 | - | 0 | 0 | 0 | 0 | 8 (11.1) | 0.30 | |
| Previous MI, n (%) | 22 (21.4) | 1 (16.7) | - | 3 (17.6) | 3 (18.8) | 2 (25.0) | 1 (25.0) | 2 (22.2) | 16 (22.2) | 0.99 |
| Previous PCI, n (%) | 19 (18.4) | 0 | - | 3 (17.6) | 2 (12.5) | 2 (25.0) | 1 (25.0) | 2 (22.2) | 14 (19.4) | 0.95 |
| Chronic kidney disease, n (%) | 50 (48.5) | 5 (83.3) | - | 5 (29.4) | 6 (37.5) | 5 (62.5) | 1 (25.0) | 6 (66.7) | 40 (55.6) | 0.19 |
| Previous smoking, n (%) | 79 (77.5) | 3 (50.0) | - | 14 (82.4) | 14 (93.3) | 6 (75.0) | 4 (100) | 8 (88.9) | 51 (70.8) | 0.3 |
| Previous alcoholism, n (%) | 12 (11.7) | 0 | - | 3 (17.6) | 0 | 1 (12.5) | 0 | 1 (11.1) | 5 (6.9) | 0.6 |
| STEMI, n (%) | 55 (53.4) | 2 (33.3) | - | 14 (82.4) | 8 (50.0) | 6 (75.0) | 2 (50.0) | 4 (44.4) | 35 (48.6) | 0.24 |
| Number of treated lesions, mean (SD) | 1.83 (1.0) | 1.83 (1.0) | - | 1.88 (0.9) | 2.13 (1.3) | 3.13 (1.6) | 2.0 (1.2) | 1.67 (0.7) | 2.03 (1.0) | 0.08 |
| Left main trated, n (%) | 3 (2.9) | 0 | - | 1 (5.9) | 1 (6.3) | 1 (12.5) | 0 | 1 (11.1) | 8 (11.1) | 0.51 |
| Number of treated vessels, mean (SD) | 1.61 (0.8) | 1.5 (0.6) | - | 1.65 (0.9) | 1.63 (0.9) | 2.25 (0.9) | 1.5 (1.0) | 1.56 (0.5) | 1.75 (0.8) | 0.52 |
| Incomplete revacularization, n (%) | 26 (25.2) | 2 (33.3) | - | 4 (23.5) | 1 (6.3) | 6 (75.0) | 4 (100.0) | 2 (22.2) | 16 (22.2) | <0.001 |
| Statin, n (%) | 102 (99.0) | 6 (100.0) | - | 17 (100.0) | 16 (100.0) | 8 (100.0) | 4 (100.0) | 8 (88.9) | 72 (100.0) | 0.09 |
| Ezetimibe, n (%) | 42 (40.8) | 3 (50.0) | - | 8 (47.1) | 10 (62.5) | 4 (50.0) | 2 (50.0) | 4 (44.4) | 39 (54.2) | 0.7 |
| Total cholesterol, median (IQR) | 156.0 (57.0) | 166.5 (62.0) | - | 133.0 (38.0) | 160 (108.0) | 179.0 (50.0) | 207.5 (27.0) | 137.5 (90.0) | 159 (82.0) | 0.58 |
| HDL-C, median (IQR) | 34 (7.0) | 41 (11.0) | - | 31.5 (12.0) | 33 (13.0) | 32.5 (7.0) | 39.0 (11.0) | 32.0 (16.0) | 36.0 (12.0) | 0.24 |
| LDL-C, median (IQR) | 97 (58.0) | 105.5 (66.0) | - | 74.5 (44.0) | 91.0 (93.0) | 113.0 (40.0) | 134.5 (14.0) | 81.0 (84.0) | 92.5 (60.0) | 0.54 |
| Non-HDL-C, median (IQR) | 122.0 (57.0) | 130.0 (66.0) | - | 98.0 (54.0) | 125.0 (101.0) | 141.0 (54.0) | 164.5 (24.0) | 109.5 (85.0) | 118.5 (71.0) | 0.51 |
| Remnant-C, median (IQR) | 25 (12.0) | 31.5 (12.0) | - | 23.5 (8.0) | 30.0 (15.0) | 26.5 (57.0) | 26.0 (17.0) | 27.0 (17.0) | 27.0 (14.0) | 0.99 |
| TG, median (IQR) | 141.0 (78.0) | 118.0 (115.0) | - | 118.5 (37) | 139.0 (81.0) | 153.5 (262.0) | 146.5 (54.0) | 133.5 (43.0) | 146.0 (83.0) | 0.84 |
| VLDL, median (IQR) | 28.2 (15.6) | 23.6 (22.9) | - | 23.7 (7.4) | 27.8 (16.2) | 30.7 (52.4) | 29.3 (10.8) | 26.7 (8.6) | 29.2 (16.5) | 0.84 |
| TC/HDL ratio, median (IQR) | 4.4 (0.9) | 4.2 (1.8) | - | 4.0 (1.5) | 4.6 (2.8) | 4.8 (2.5) | 5.3 (0.7) | 4.5 (2.7) | 4.3 (1.7) | 0.46 |
| LDL/ApoB-100 ratio, median (IQR) | 1.0 (0.2) | 1.1 (0.4) | - | 0.9 (0.1) | 1.0 (0.3) | 1.0 (0.3) | 1.1 (0.2) | 0.9 (0.2) | 1.0 (0.3) | 0.31 |
| LDL/HDL ratio, median (IQR) | 2.6 (1.7) | 2.7 (1.7) | - | 2.2 (1.1) | 2.8 (2.7) | 3.1 (1.5) | 3.5 (0.8) | 2.5 (2.8) | 2.6 (1.4) | 0.64 |
| TG/HDL ratio, median (IQR) | 3.9 (2.7) | 3.2 (3.7) | - | 3.2 (2.1) | 3.7 (2.3) | 4.7 (9.4) | 4.2 (2.0) | 4.4 (2.4) | 4.3 (2.1) | 0.82 |
| ApoB-100, median (IQR) | 87.0 (42.0) | 88.0 (38.0) | - | 75.0 (37.0) | 98.0 (51.0) | 114.5 (33.0) | 120.0 (25.0) | 85.0 (58.0) | 90.5 (49.0) | 0.45 |
| Baseline Lp(a), median (IQR) | 12.3 (13.8) | 23.4 (3.9) | - | 36.6 (9.9) | 39.7 (6.8) | 39.4 (6.4) | 60.4 (33.6) | 60.4 (28.4) | 102.5 (44.4) | <0.001 |
| Follow-up Lp(a), median (IQR) | 8.7 (10.5) | 36.5 (4.4) | - | 16.3 (12.1) | 42.4 (13.3) | 61.8 (28.1) | 19.7 (15.3) | 37.7 (13.8) | 105.5 (55.3) | <0.001 |
| Glucose, median (IQR) | 115.0 (64.0) | 100.0 (37.0) | - | 129.0 (67.0) | 110.0 (74.0) | 159.0 (87.0) | 88.5 (69.0) | 101.0 (124.0) | 111.0 (58) | 0.78 |
| HbA1c, median (IQR) | 6.0 (1.3) | 5.7 (0.9) | - | 6.6 (2.2) | 6.3 (0.7) | 5.9 (1.5) | 5.7 (0.7) | 6.2 (1.5) | 5.9 (0.9) | 0.45 |
| Creatinine, median (IQR) | 0.91 (0.3) | 0.98 (0.3) | - | 0.98 (0.3) | 0.85 (0.3) | 1.0 (0.5) | 0.9 (0.2) | 1.0 (0.3) | 1.0 (0.4) | 0.10 |
| eGFR, median (IQR) | 82.0 (17.0) | 77.0 (16.0) | - | 78.5 (25.0) | 90.0 (18.0) | 78.5 (29.0) | 90.0 (11.0) | 77.0 (30.0) | 77.0 (33.0) | 0.02 |
| Urea, median (IQR) | 41.0 (13.0) | 36.0 (11.0) | - | 40.5 (22.0) | 36.0 (13) | 42.0 (43.0) | 43.5 (32.0) | 47.5 (26.0) | 43.5 (15) | 0.15 |
| Uric acid, median (IQR) | 6.3 (2.9) | 6.3 (1.9) | - | 5.7 (2.8) | 4.8 (2.7) | 7.5 (2.7) | 4.6 (5.4) | 5.7 (2.4) | 6.3 (2.5) | 0.14 |
| MACE, n (%) | 8 (18.2) | 0 | - | 4 (33.3) | 2 (33.3) | 0 | 0 | 0 | 13 (46.4) | 0.15 |
| High Lp(a) variability, n (%) | 59 (57.3) | 6 (100.0) | - | 15 (88.2) | 1 (6.3) | 8 (100.0) | 4 (100.0) | 7 (77.8) | 36 (50.0) | <0.01 |
| Baseline Lp(a) risk category | Follow-up Lp(a) Risk, N (%) | Lp(a) variability | N (%) |
|---|---|---|---|
| Low-risk (109), Low | Low-risk, 103 (94.5) | High | 59 (57.3) |
| variability 50 (45.9%), High | Low | 44 (42.7) | |
| variability 59 (54.1%) | Intermediate-risk, 6 (5.5) | High | 0 (0.0) |
| Low | 6 (100.0) | ||
| High-risk, 0 (0%) | High | 0 (0.0) | |
| Low | 0 (0.0) | ||
| Intermediate-risk (41), Low | Low-risk, 17 (41.5) | High | 15 (88.2) |
| variability 17 (41.5%), High | Low | 2 (11.8) | |
| variability 24 (58.5%) | Intermediate-risk, 16 (39) | High | 1 (6.3) |
| Low | 15 (93.7) | ||
| High-risk, 8 (19.1) | High | 8 (100) | |
| Low | 0 (0.0) | ||
| High-risk (85), Low | Low-risk, 4 (4.7) | High | 4 (100.0) |
| variability 38 (44.7%), High | Low | 0 (0.0) | |
| variability 47 (55.3%) | Intermediate-risk, 9 (10.6) | High | 7 (77.8) |
| Low | 2 (22.2) | ||
| High-risk, 72 (84.7) | High | 36 (50.0) | |
| Low | 36 (50.0) |
References
- Nordestgaard, B.G.; Langsted, A. Lipoprotein(a) and cardiovascular disease. Lancet 2024, 404, 1255–1264. [Google Scholar] [CrossRef] [PubMed]
- Volgman, A.S.; Koschinsky, M.L.; Mehta, A.; Rosenson, R.S. Genetics and Pathophysiological Mechanisms of Lipoprotein(a)-Associated Cardiovascular Risk. J. Am. Heart Assoc. 2024, 13, e033654. [Google Scholar] [CrossRef] [PubMed]
- Greco, A.; Finocchiaro, S.; Spagnolo, M.; Faro, D.C.; Mauro, M.S.; Raffo, C.; Sangiorgio, G.; Imbesi, A.; Laudani, C.; Mazzone, P.M.; et al. Lipoprotein(a) as a Pharmacological Target: Premises, Promises, and Prospects. Circulation 2025, 151, 400–415. [Google Scholar] [CrossRef]
- Patel, A.P.; Wang, M.; Pirruccello, J.P.; Ellinor, P.T.; Ng, K.; Kathiresan, S.; Khera, A.V. Lp(a) (Lipoprotein[a]) concentrations and incident atherosclerotic cardiovascular disease: New insights from a large national biobank. Arterioscler. Thromb. Vasc. Biol. 2021, 41, 465–474. [Google Scholar] [CrossRef]
- Kim, A.-R.; Ahn, J.-M.; Kang, D.-Y.; Jun, T.J.; Sun, B.J.; Kim, H.J.; Kim, J.B.; Kim, D.-H.; Park, D.-W.; Kim, Y.-H.; et al. Association of Lipoprotein(a) With Severe Degenerative Aortic Valve Stenosis. JACC Asia 2024, 4, 751–760. [Google Scholar] [CrossRef]
- Arsenault, B.J.; Loganath, K.; Girard, A.; Botezatu, S.; Zheng, K.H.; Tzolos, E.; Abdoun, K.; Tastet, L.; Capoulade, R.; Côté, N.; et al. Lipoprotein(a) and calcific aortic valve stenosis progression: A systematic review and meta-analysis. JAMA Cardiol. 2024, 9, 835–842. [Google Scholar] [CrossRef]
- Thomas, P.E.; Vedel-Krogh, S.; Nielsen, S.F.; Nordestgaard, B.G.; Kamstrup, P.R. Lipoprotein(a) and risks of peripheral artery disease, abdominal aortic aneurysm, and major adverse limb events. J. Am. Coll. Cardiol. 2023, 82, 2265–2276. [Google Scholar] [CrossRef]
- Romero-Trevejo, J.L.; Fernández-Romero, L.; Delgado, J.; Muñoz-García, E.; Sánchez-Pérez, A.; Murri, M.; Gutiérrez-Bedmar, M.; Jiménez-Navarro, M.F. Choroidal thickness and granulocyte colony-stimulating factor in tears improve the prediction model for coronary artery disease. Cardiovasc. Diabetol. 2022, 21, 103. [Google Scholar] [CrossRef]
- Langsted, A.; Kamstrup, P.R.; Nordestgaard, B.G. High lipoprotein(a) and high risk of mortality. Eur. Heart J. 2019, 40, 2760–2770. [Google Scholar] [CrossRef]
- Amiri, M.; Raeisi-Dehkordi, H.; Verkaar, A.J.C.F.; Wu, Y.; van Westing, A.C.; Berk, K.A.; Bramer, W.M.; Aune, D.; Voortman, T. Circulating lipoprotein(a) and all-cause and cause-specific mortality: A systematic review and dose–response meta-analysis. Eur. J. Epidemiol. 2023, 38, 485–499. [Google Scholar] [CrossRef]
- Bhatia, H.S.; Wandel, S.; Willeit, P.; Lesogor, A.; Bailey, K.; Ridker, P.M.; Nestel, P.; Simes, J.; Tonkin, A.; Schwartz, G.G.; et al. Independence of lipoprotein(a) and low-density lipoprotein cholesterol–mediated cardiovascular risk: A participant-level meta-analysis. Circulation 2025, 151, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Mach, F.; Koskinas, K.C.; Roeters van Lennep, J.E.; Tokgözoğlu, L.; Badimon, L.; Baigent, C.; Binder, C.J.; Catapano, A.L.; De Backer, G.; Delgado, V.; et al. 2025 focused update of the 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur. Heart J. 2025, 46, 4359–4378. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Stone, N.J.; Bailey, A.L.; Beam, C.; Birtcher, K.K.; Blumenthal, R.S.; Braun, L.T.; de Ferranti, S.; Faiella-Tommasino, J.; Forman, D.E.; et al. 2018 AHA/ACC Guideline on the management of blood cholesterol. Circulation 2019, 139, e1082–e1143. [Google Scholar] [CrossRef] [PubMed]
- Greco, A.; Finocchiaro, S.; Spagnolo, M.; Faro, D.C.; Mauro, M.S.; Raffo, C.; Sangiorgio, G.; Imbesi, A.; Laudani, C.; Mazzone, P.M.; et al. Lipoprotein(a) as a Pharmacological Target: Premises, Promises, and Prospects. Circulation 2025, 151, 400–415. [Google Scholar] [CrossRef]
- OCEAN(a) Investigators. Olpasiran Trials of Cardiovascular Events and Lipoprotein(a) Reduction (OCEAN(a)) - Outcomes Trial. ClinicalTrials.gov 2025, NCT05581303. Sponsor: Amgen. Available online: https://clinicaltrials.gov/ct2/show/NCT05581303 (accessed on 27 January 2026).
- Cho, L.; Nicholls, S.J.; Nordestgaard, B.G.; Landmesser, U.; Tsimikas, S.; Blaha, M.J.; Leitersdorf, E.; Lincoff, A.M.; Lesogor, A.; Manning, B.; et al. Design and rationale of Lp(a)HORIZON trial: assessing the effect of lipoprotein(a) lowering with pelacarsen on major cardiovascular events in patients with CVD and elevated Lp(a). Am. Heart J. 2025, 287, 1–9. [Google Scholar] [CrossRef]
- Volgman, A.S.; Navar, A.M.; Virani, S.S.; Michos, E.D.; Blaha, M.J.; Martin, S.S.; Ballantyne, C.M.; Virani, S.; Khera, A.V.; Orkaby, A.R.; et al. Genetics and Pathophysiological Mechanisms of Lipoprotein(a)-Associated Cardiovascular Risk. J. Am. Heart Assoc. 2024, 13, e033654. [Google Scholar] [CrossRef]
- Jiménez-Salva, M.; Carmona-Segovia, A.; Molina-Ramos, A.I.; García-Rodríguez, L.; Puyol-Ruiz, F.; Doncel-Abad, V.; Romero-Cuevas, M.; Serrano, A.; Muñoz-García, A.; Flores-López, M.; et al. Older adults with acute coronary syndrome: The impact of frailty and nutritional status on in-hospital complications. Eur. J. Cardiovasc. Nurs. 2025, zvaf006. [Google Scholar] [CrossRef]
- Anagnostis, P.; Antza, C.; Trakatelli, C.; Lambrinoudaki, I.; Goulis, D.G.; Kotsis, V. The effect of menopause on lipoprotein(a) concentrations: A systematic review and meta-analysis. Maturitas 2022, 164, 1–11. [Google Scholar] [CrossRef]
- Jenner, J.L.; Ordovas, J.M.; Lamon-Fava, S.; Schaefer, M.M.; Wilson, P.W.; Castelli, W.P.; Schaefer, E.J. Effects of age, sex, and menopausal status on plasma lipoprotein(a) levels: The Framingham Offspring Study. Circulation 1993, 87, 1135–1141. [Google Scholar] [CrossRef]
- Shlipak, M.G.; Kooperberg, C.; Psaty, B.M.; Furberg, C.D.; Siscovick, D.S.; Rutan, G.H.; Cornoni-Huntley, J.; Robbins, J.A.; Harris, T.B.; Newman, A.B.; et al. Estrogen and progestin, lipoprotein(a), and the risk of recurrent coronary heart disease events after menopause. JAMA 2000, 283, 1845–1852. [Google Scholar] [CrossRef]
- Nakajima, K.; Hata, Y. Intraindividual Variations in Lipoprotein(a) Levels and Factors Related to These Changes. J. Atheroscler. Thromb. 1996, 2, 96–106. [Google Scholar] [CrossRef]
- Trinder, M.; Paruchuri, K.; Haidermota, S.; Bernardo, R.; Zekavat, S.M.; Gilliland, T.; Januzzi, J.L., Jr.; Natarajan, P. Repeat Measures of Lipoprotein(a) Molar Concentration and Cardiovascular Risk. J. Am. Coll. Cardiol. 2022, 79, 617–628. [Google Scholar] [CrossRef]
- Deshotels, M.R.; Sun, C.; Nambi, V.; Virani, S.S.; Matsushita, K.; Yu, B.; Ballantyne, C.M.; Hoogeveen, R.C. Temporal Trends in Lipoprotein(a) Concentrations: The Atherosclerosis Risk in Communities Study. J. Am. Heart Assoc. 2022, 11, e026762. [Google Scholar] [CrossRef]
- Awad, K.; Mahmoud, A.K.; Abbas, M.T.; Alsidawi, S.; Ayoub, C.; Arsanjani, R.; Farina, J.M. Intra-individual variability in lipoprotein(a) levels: findings from a large academic health system population. Eur. J. Prev. Cardiol. 2025, 32, 716–721. [Google Scholar] [CrossRef]
- Joo, H.J.; Yun, S.G.; Park, J.H.; Hong, S.J.; Yu, C.W.; Shin, S.Y.; Kim, E.J. Predictors of lipoprotein(a) variability in clinical practice and their impact on cardiovascular risk. Lipids Health Dis. 2025, 24, 250. [Google Scholar] [CrossRef]
- Sung, D.E.; Lee, M.Y.; Kwon, M.J.; Sung, K.C. Longitudinal changes and borderline reclassification of Lipoprotein(a) compared with conventional lipids in over 230,000 adults. Atherosclerosis. 2025, 410, 120524. [Google Scholar] [CrossRef] [PubMed]
- Harb, T.; Ziogos, E.; Blumenthal, R.S.; Gerstenblith, G.; Leucker, T.M. Intra-individual variability in lipoprotein(a): the value of a repeat measure for reclassifying individuals at intermediate risk. Eur. Heart J. Open. 2024, 4, oeae064. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.S.; Hu, W.Y.; Li, Y.J.; Yu, J.; Sang, S.; Alsalman, Z.M.; Xie, D.Q. Lipoprotein(a) variability is associated with mean follow-up C-reactive protein in patients with coronary artery disease following percutaneous coronary intervention. World J. Clin. Cases. 2022, 10, 12909–12919. [Google Scholar] [CrossRef]
- Matta, M.G.; Schreier, L.; Lavalle-Cobo, A.; Garcia-Zamora, S.; Ferraresi, A.; Madsen, A.; Bellini, S.; Ramos, G.; Roubicek, P.; Corral, P. Temporal variability of Lp(a) in clinically stable patients: implications for cardiovascular risk assessment. Med. Clin. (Barc). 2024, 163, 436–441. [Google Scholar] [CrossRef] [PubMed]
- Gaba, P.; Rosenson, R.S.; López, J.A.G.; Watts, G.F.; Leucker, T.M.; Kuder, J.F.; Im, K.; Kassahun, H.; Wang, H.; Wu, Y.; et al. Intraindividual variability in serial lipoprotein(a) concentrations among placebo-treated patients in the OCEAN(a)-DOSE trial. J. Am. Coll. Cardiol. 2025, 85, 550–553. [Google Scholar] [CrossRef]
- Ziogos, E.; Vavuranakis, M.A.; Harb, T.; Foran, P.L.; Blaha, M.J.; Jones, S.R.; Lai, S.; Gerstenblith, G.; Leucker, T.M. Lipoprotein(a) concentrations in acute myocardial infarction patients are not indicative of levels at six month follow-up. Eur. Heart J. Open 2023, 3, oead035. [Google Scholar] [CrossRef]
- Saeki, Y.; Sawaguchi, J.; Akita, S.; Takamura, T.A.; Fujibayashi, K.; Wakasa, M.; Akao, H.; Kitayama, M.; Kawai, Y.; Kajinami, K. Initial decrease in the lipoprotein(a) level is a novel prognostic biomarker in patients with acute coronary syndrome. World J. Cardiol. 2024, 16, 329–338. [Google Scholar] [CrossRef]
- Vavuranakis, M.A.; Jones, S.R.; Ziogos, E.; Blaha, M.J.; Williams, M.S.; Foran, P.; Schindler, T.H.; Lai, S.; Schulman, S.P.; Gerstenblith, G.; et al. The trajectory of lipoprotein(a) during the peri- and early postinfarction period and the impact of proprotein convertase subtilisin/kexin type 9 inhibition. Am. J. Cardiol. 2022, 171, 1–6. [Google Scholar] [CrossRef]
- Sourij, C.; Aziz, F.; Krappinger, S.; Praschk, A.; Metzner, T.; Kojzar, H.; Zirlik, A.; Stojakovic, T.; Pätzold, D.; von Lewinski, D.; et al. Changes in lipoprotein(a) levels in people after ST elevation myocardial infarction—The STEMI-Lipids Study. Int. J. Mol. Sci. 2023, 24, 15531. [Google Scholar] [CrossRef] [PubMed]
- Kronenberg, F.; Mora, S.; Stroes, E.S.G.; Ference, B.A.; Arsenault, B.J.; Berglund, L.; Dweck, M.R.; Koschinsky, M.; Lambert, G.; Mach, F.; et al. Lipoprotein(a) in atherosclerotic cardiovascular disease and aortic stenosis: A European Atherosclerosis Society consensus statement. Eur. Heart J. 2022, 43, 3925–3946. [Google Scholar] [CrossRef] [PubMed]
- Tsimikas, S.; Marcovina, S.M. Ancestry, lipoprotein(a), and cardiovascular risk thresholds: JACC Review Topic of the Week. J. Am. Coll. Cardiol. 2022, 80, 934–946. [Google Scholar] [CrossRef]
- Dzobo, K.E.; Kraaijenhof, J.M.; Stroes, E.S.G.; Nurmohamed, N.S.; Kroon, J. Lipoprotein(a): An underestimated inflammatory mastermind. Atherosclerosis 2022, 349, 101–109. [Google Scholar] [CrossRef]
- Mooser, V.; Berger, M.M.; Tappy, L.; Cayeux, C.; Marcovina, S.M.; Darioli, R.; Nicod, P.; Chioléro, R. Major reduction in plasma Lp(a) levels during sepsis and burns. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1137–1142. [Google Scholar] [CrossRef]
- Willeit, P.; Ridker, P.M.; Nestel, P.J.; Simes, J.; Tonkin, A.M.; Pedersen, T.R.; Schwartz, G.G.; Olsson, A.G.; Colhoun, H.M.; Kronenberg, F.; et al. Baseline and on-statin treatment lipoprotein(a) levels for prediction of cardiovascular events: Individual patient-data meta-analysis of statin outcome trials. Lancet. 2018, 392, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Xiao, S.; Liu, N. Association of lipoprotein(a) with coronary severity in patients with new-onset acute myocardial infarction: A large cross-sectional study. Clin. Chim. Acta 2023, 540, 117220. [Google Scholar] [CrossRef]
| Characteristic | N = 235 |
|---|---|
| Age, years | 61.1 (55.0-70.6) |
| Male sex | 186 (79.1) |
| Diabetes mellitus | 84 (35.7) |
| Hypertension | 148 (63.0) |
| Dyslipidemia | 174 (74.0) |
| Menopausal status | 44 (89.8) |
| Body mass index, kg/m² | 27.9 (25.3-31.0) |
| Stroke | 8 (3.4) |
| Transient ischemic attack | 3 (1.3) |
| Peripheral artery disease | 13 (5.5) |
| Prior MI | 50 (21.3) |
| Prior PCI | 43 (18.3) |
| Chronic kidney disease | 118 (50.2) |
| Smoking status (Never / Former / Current) | 54 (23) / 90 (38.3) / 89 (37.9) |
| Alcohol consumption (Never / Former / Current) | 213 (90.6) / 17 (7.2) / 5 (2.1) |
| STEMI presentation | 126 (53.6) |
| Number of treated lesions | 2.0 (1.1) |
| Left main treated | 15 (6.4) |
| Number of treated vessels | 1.7 (0.8) |
| Incomplete revascularization | 61 (26) |
| High-intensity statin | 233 (99.1) |
| Ezetimibe | 112 (47.7) |
| Total cholesterol, mg/dL | 158.0 (124.5-191.0) |
| HDL-C, mg/dL | 34.5 (31.0-40.0) |
| LDL-C, mg/dL | 93.0 (61.8-123.3) |
| Non–HDL-C, mg/dL | 121.0 (91.8-156.0) |
| Remnant-C, mg/dL | 26.0 (21.0-33.0) |
| TG, mg/dL | 139.0 (108.0-183.0) |
| VLDL, mg/dL | 27.8 (21.6-36.6) |
| TC/HDL ratio | 4.4 (3.5-5.4) |
| LDL/ApoB-100 ratio | 1.0 (0.9-1.1) |
| LDL/HDL ratio | 2.7 (1.8-3.5) |
| TG/HDL ratio | 4.1 (2.9-5.3) |
| ApoB-100, mg/dL | 90.0 (69.8-115.0) |
| Baseline Lp(a) | 34.7 (14.8-79.0) |
| Glucose, mg/dL | 111.0 (91.0-155.3) |
| HbA1c, % | 6.0 (5.6-6.7) |
| Creatinine, mg/dL | 0.9 (0.8-1.1) |
| eGFR, mL/min/1.73 m² | 80.0 (67.5-90.0) |
| Urea, mg/dL | 41.0 (35.0-50.0) |
| Uric acid, mg/dL | 6.1 (4.8-7.5) |
| MACE | 27 (11.5) |
| Characteristic | High Lp(a) Variability (N = 136) | Low Lp(a) Variability (N = 99) | Adjusted P Value |
|---|---|---|---|
| Age, years | 61.4 (11.1) | 61.9 (11.5) | 0.720 |
| Male sex | 103 (75.7) | 83 (83.8) | 0.131 |
| Diabetes mellitus | 53 (39.0) | 31 (31.6) | 0.248 |
| Hypertension | 85 (62.5) | 63 (63.6) | 0.859 |
| Dyslipidemia | 104 (76.5) | 70 (70.7) | 0.320 |
| Menopausal status* | 32 (23.5) | 12 (12.1) | 0.034 |
| Body mass index, kg/m² | 28.1 (5.8) | 27.8 (6.2) | 0.795 |
| Stroke | 4 (2.9) | 4 (4.0) | 0.646 |
| Transient ischemic attack | 1 (0.7) | 2 (2.0) | 0.386 |
| Peripheral artery disease | 5 (3.7) | 8 (8.1) | 0.145 |
| Prior MI | 30 (22.1) | 20 (20.2) | 0.731 |
| Prior PCI | 25 (18.4) | 18 (18.2) | 0.969 |
| Chronic kidney disease | 69 (50.7) | 49 (49.5) | 0.851 |
| Smoking status (Never / Former / Current) | 34 / 45 / 56 | 20 / 45 / 33 | 0.150 |
| Alcohol consumption (Never / Former / Current) | 124 / 10 / 2 | 89 / 7 / 3 | 0.715 |
| STEMI presentation | 76 (55.9) | 50 (50.5) | 0.414 |
| Number of treated lesions | 2 (2) | 2 (2) | 0.628 |
| Left main treated | 8 (5.9) | 7 (7.1) | 0.713 |
| Number of treated vessels | 1.5 (1) | 1 (1) | 0.596 |
| Incomplete revascularization | 43 (31.6) | 18 (18.2) | 0.020 |
| High-intensity statin | 135 (99.3) | 98 (99.0) | 0.821 |
| Ezetimibe | 67 (49.3) | 45 (45.5) | 0.564 |
| Total cholesterol, mg/dL | 160.0 (61.0) | 155.5 (73.0) | 0.236 |
| HDL-C, mg/dL | 35.0 (11.0) | 34.5 (9.0) | 0.955 |
| LDL-C, mg/dL | 100.0 (57.0) | 90.0 (64.0) | 0.279 |
| Non–HDL-C, mg/dL | 128.0 (45.8) | 120.3 (44.9) | 0.203 |
| Remnant-C, mg/dL | 27 (13) | 26 (12) | 0.113 |
| TG, mg/dL | 147 (80) | 134 (58) | 0.087 |
| VLDL, mg/dL | 29.4 (15.9) | 26.8 (11.6) | 0.087 |
| TC/HDL ratio | 4.6 (2.2) | 4.3 (1.4) | 0.110 |
| LDL/ApoB-100 ratio | 0.98 (0.3) | 0.99 (0.2) | 0.501 |
| LDL/HDL ratio | 2.8 (1.9) | 2.5 (1.3) | 0.135 |
| TG/HDL ratio | 4.4 (2.4) | 3.7 (2.5) | 0.085 |
| ApoB-100, mg/dL | 94 (45) | 86.5 (44) | 0.078 |
| Baseline Lp(a) | 30.95 (51.4) | 38.1 (65.1) | 0.930 |
| Follow-up Lp(a) | 18.7 (64.0) | 32.9 (61.7) | 0.180 |
| Glucose, mg/dL | 116 (73) | 109.5 (53) | 0.302 |
| HbA1c, % | 5.9 (1.4) | 6.0 (0.9) | 0.895 |
| Creatinine, mg/dL | 0.96 (0.3) | 0.95 (0.3) | 0.969 |
| eGFR, mL/min/1.73 m² | 80 (23) | 82 (22) | 0.719 |
| Urea, mg/dL | 42 (18) | 40 (14) | 0.215 |
| Uric acid, mg/dL | 6.1 (2.9) | 6.2 (2.9) | 0.375 |
| MACE | 15 (11.0) | 12 (12.1) | 0.616 |
| Univariable | Multivariable | |||
|---|---|---|---|---|
| OR (CI 95%) | P | OR (CI 95%) | P | |
| Age, years | 0.99 (0.97-1.02) | 0.72 | 1.00 (0.98-1.03) | 0.97 |
| Female sex | 1.67 (0.85-3.22) | 0.13 | 1.92 (0.93-4.00) | 0.08 |
| Diabetes mellitus | 1.38 (0.80-2.39) | 0.25 | ||
| Hypertension | 0.95 (0.56-1.63) | 0.86 | ||
| Dyslipidemia | 1.35 (0.75-2.42) | 0.32 | ||
| Menopausal status* | 10.67 (1.10-105.30) | 0.04 | 11.18 (0.79-157.58) | 0.07 |
| Body mass index | 1.01 (0.95-1.07) | 0.87 | ||
| Stroke | 0.72 (0.18-2.95) | 0.65 | ||
| Transient ischemic attack | 0.36 (0.03-4.12) | 0.41 | ||
| Peripheral artery disease | 0.43 (0.14-1.37) | 0.15 | 0.44 (0.12-1.61) | 0.21 |
| Prior MI | 1.12 (0.59-2.11) | 0.73 | ||
| Prior PCI | 1.01 (0.52-1.98) | 0.97 | ||
| Chronic kidney disease | 1.05 (0.63-1.76) | 0.85 | ||
| Previous smoking | 1.05 (0.75-1.48) | 0.77 | ||
| Previous alcoholism | 0.82 (0.42-1.62) | 0.57 | ||
| STEMI presentation | 1.24 (0.74-2.09) | 0.41 | ||
| Number of treated lesions | 1.06 (0.83-1.36) | 0.64 | ||
| Left main treated | 0.82 (0.29-2.35) | 0.71 | ||
| Number of treated vessels | 1.07 (0.78-1.49) | 0.67 | ||
| Incomplete revascularization | 2.08 (1.11-3.89) | 0.02 | 2.22 (1.14-4.31) | 0.02 |
| Statin | 1.38 (0.08-22.29) | 0.82 | ||
| Ezetimibe | 1.16 (0.69-1.96) | 0.56 | ||
| Total cholesterol | 1.00 (0.99-1.01) | 0.22 | ||
| HDL-C | 0.99 (0.97-1.03) | 0.89 | ||
| LDL-C | 1.00 (0.99-1.01) | 0.35 | ||
| Non-HDL-C | 1.00 (0.99-1.01) | 0.20 | ||
| Remnant-C | 1.01 (0.99-1.03) | 0.31 | ||
| TG | 1.00 (0.99-1.01) | 0.21 | ||
| VLDL | 1.01 (0.99-1.03) | 0.21 | ||
| TC/HDL ratio | 1.13 (0.94-1.36) | 0.18 | ||
| LDL/ApoB-100 ratio | 0.55 (0.17-1.73) | 0.30 | ||
| LDL/HDL ratio | 1.18 (0.94-1.48) | 0.15 | 0.98 (0.66-1.45) | 0.98 |
| TG/HDL ratio | 1.08 (0.97-1.20) | 0.17 | ||
| ApoB-100 | 1.01 (0.99-1.02) | 0.07 | 1.01 (0.99-1.02) | 0.25 |
| Baseline Lp(a) | 1.00 (0.993-1.005) | 0.80 | ||
| Follow-up Lp(a) | 1.00 (0.994-1.004) | 0.63 | ||
| Glucose | 1.00 (0.99-1.01) | 0.15 | 1.00 (0.99-1.01) | 0.36 |
| HbA1c | 1.02 (0.83-1.24) | 0.89 | ||
| Creatinine | 1.11 (0.43-2.87) | 0.82 | ||
| eGFR | 0.99 (0.98-1.01) | 0.72 | ||
| Urea | 0.99 (0.99-1.01) | 0.65 | ||
| Uric acid | 1.05 (0.94-1.18) | 0.38 | ||
| MACE | 0.79 (0.33-2.95) | 0.62 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




