Submitted:
13 February 2026
Posted:
14 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Background and Related Work
3. Materials and Methods
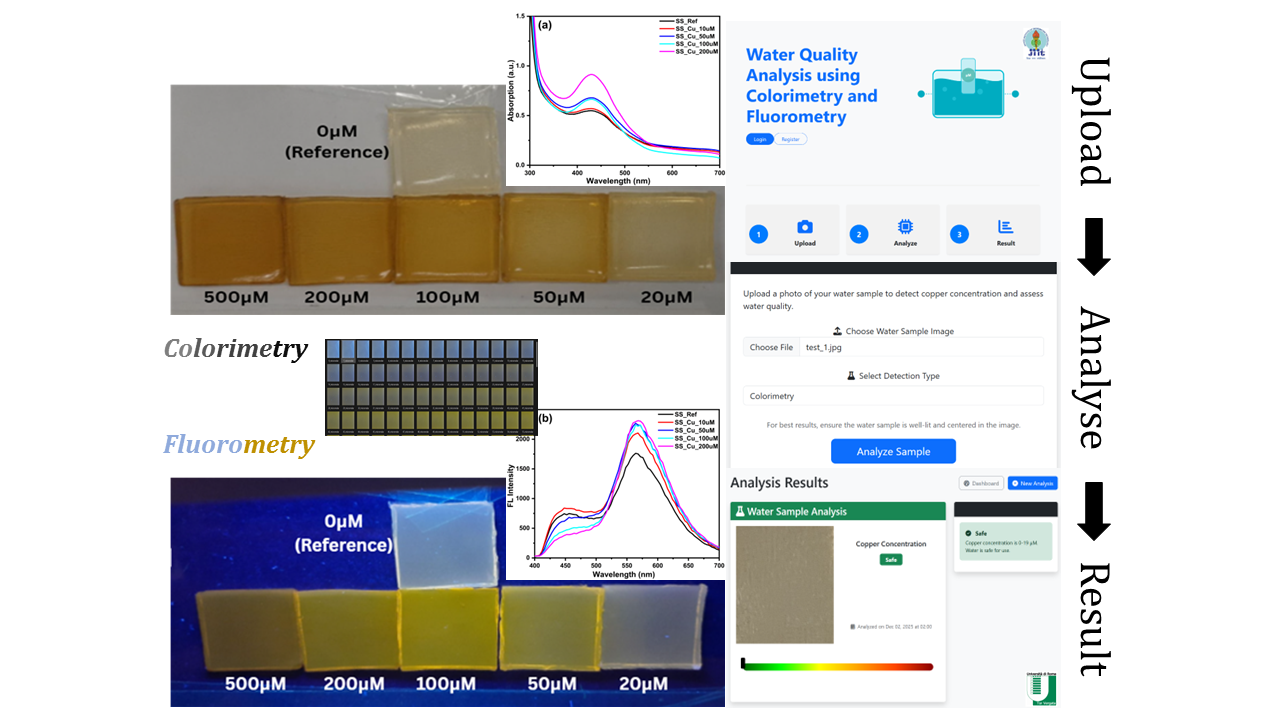
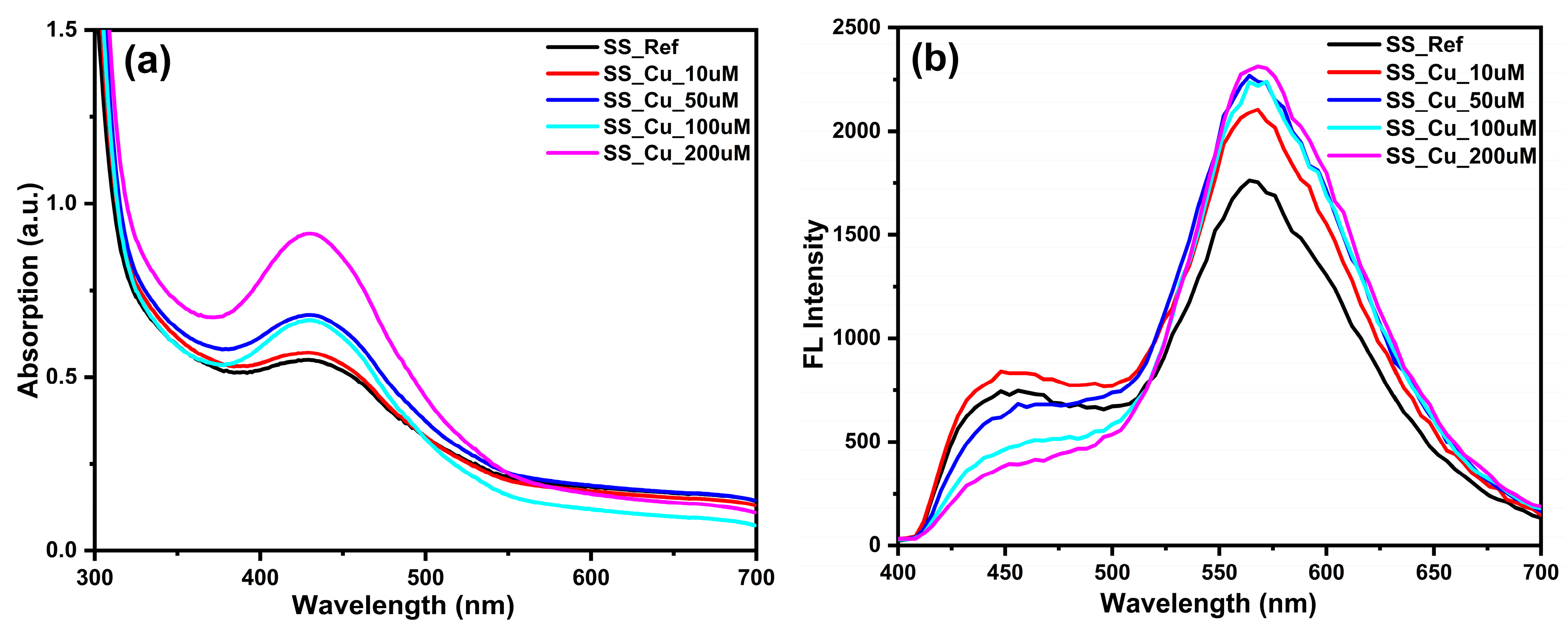
3.1. NSCDs Incorporated Hydrogel Films (Sensing System) Fabrication and Imaging
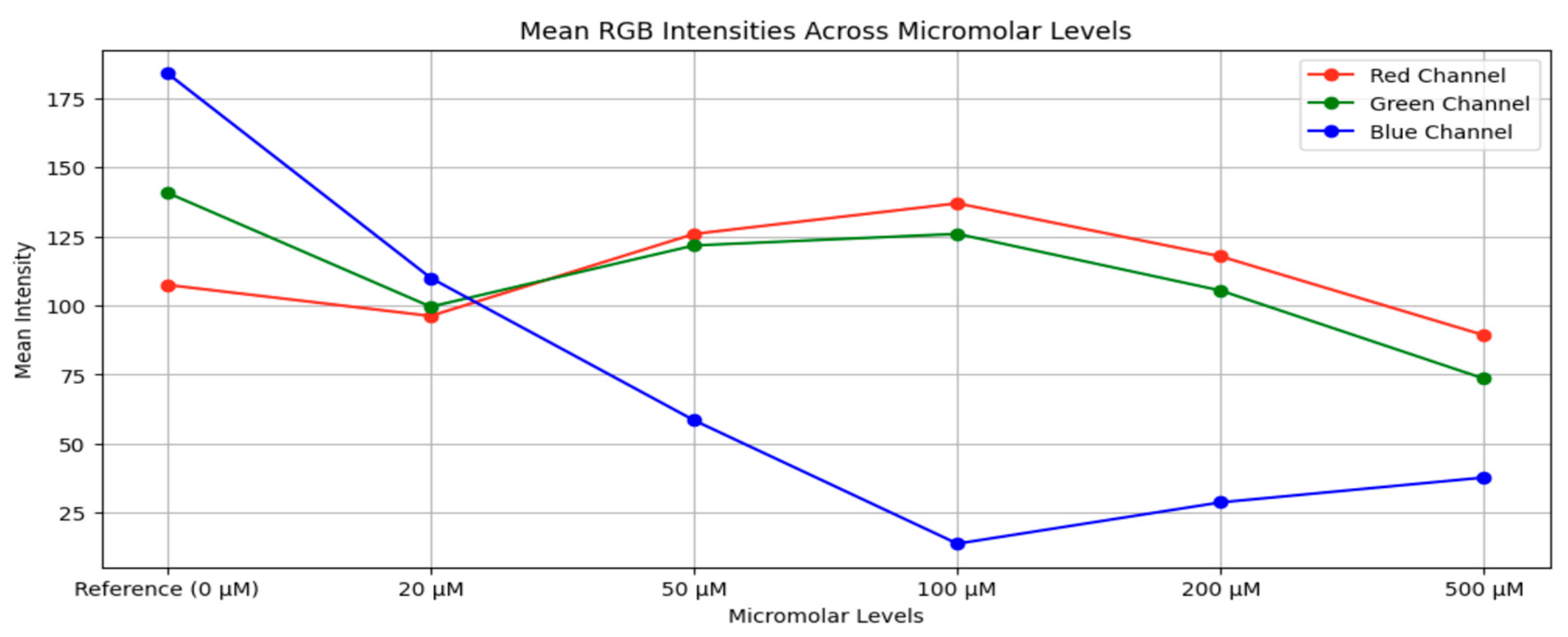
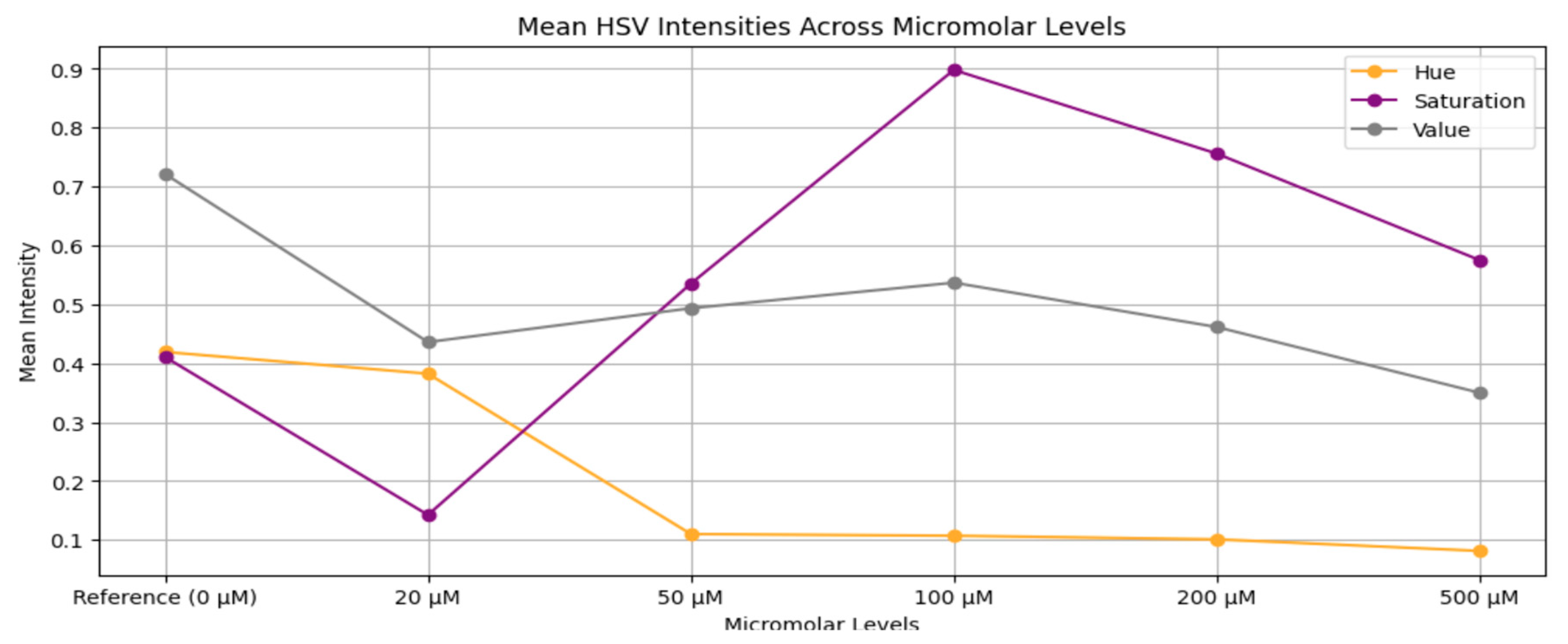
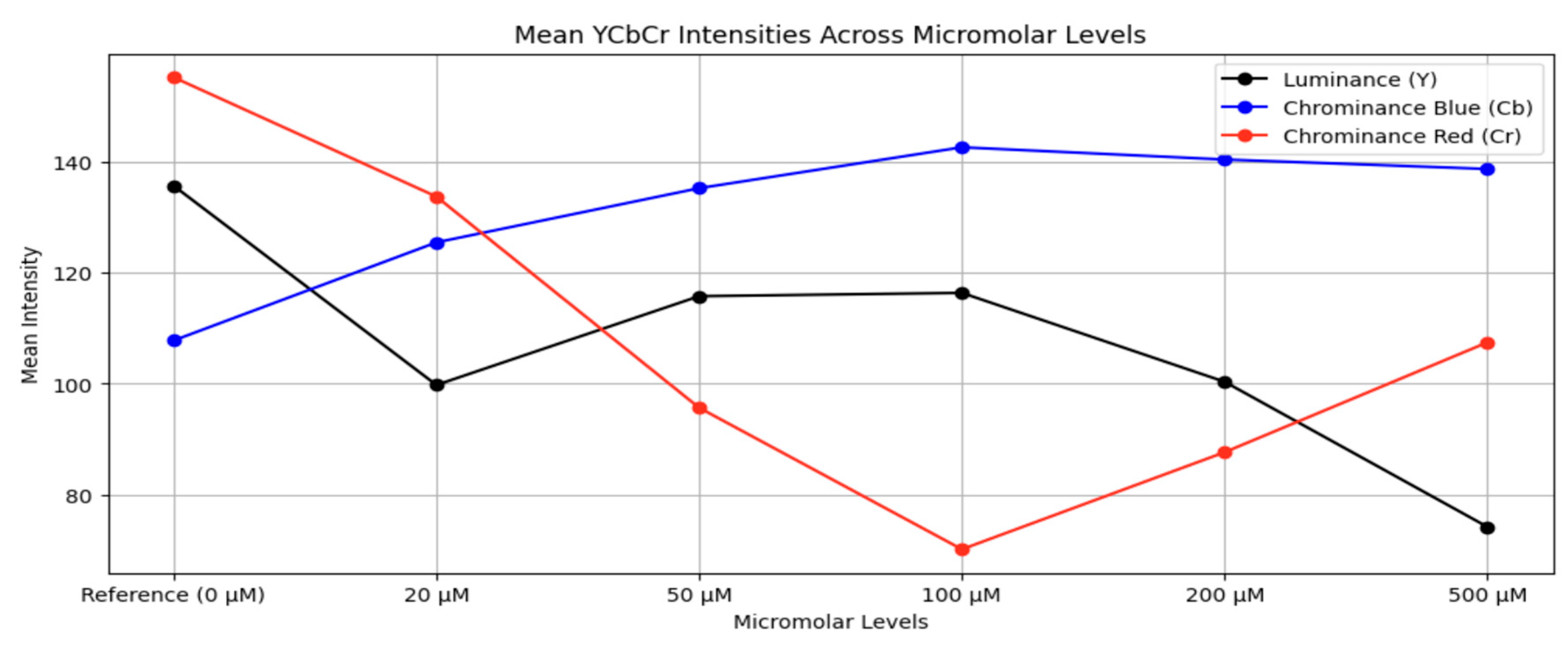
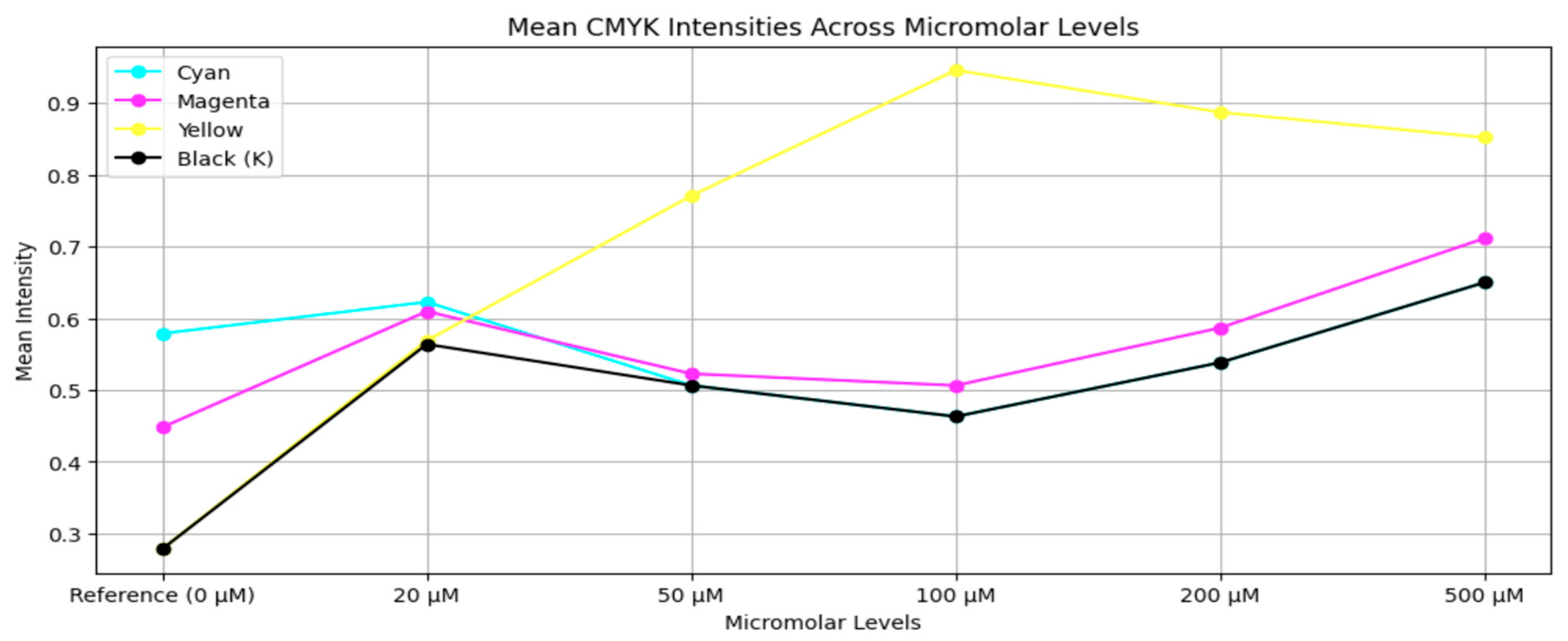
3.2. Dataset Preparation
- Interpolation: Equation (1) illustrates how digital interpolation techniques were used to create intermediate images for each integer micromolar value between 0 and 500 µM. A smooth and fine-grained optical transition between known sensor responses was produced by this method, which produced 501 distinct concentration levels.
-
Augmentation: To replicate real-world variability in imaging configurations, nine augmentation changes were applied to each interpolated image. These augmentations included:I. Rotation by –5°,II. Rotation by –10°,III. Rotation by +5°,IV. Rotation by +10°,V. Horizontal flipping,VI. Vertical flipping,VII. Brightness increase,VIII. Brightness decrease,IX. Geometric scaling (cropping followed by resizing).
3.3. Machine Learning Models
3.3.1. Evaluation Metrics
3.3.2. Practical Considerations and Justification
4. Experimental Results
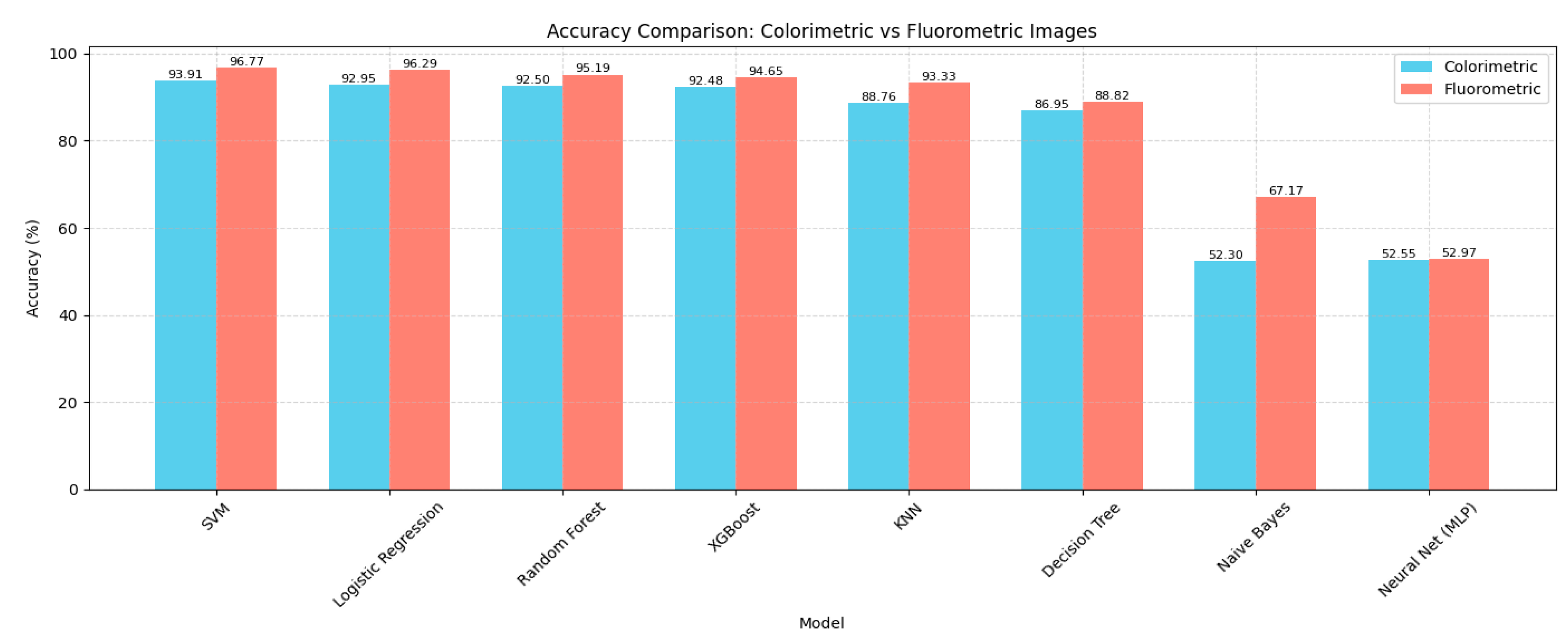
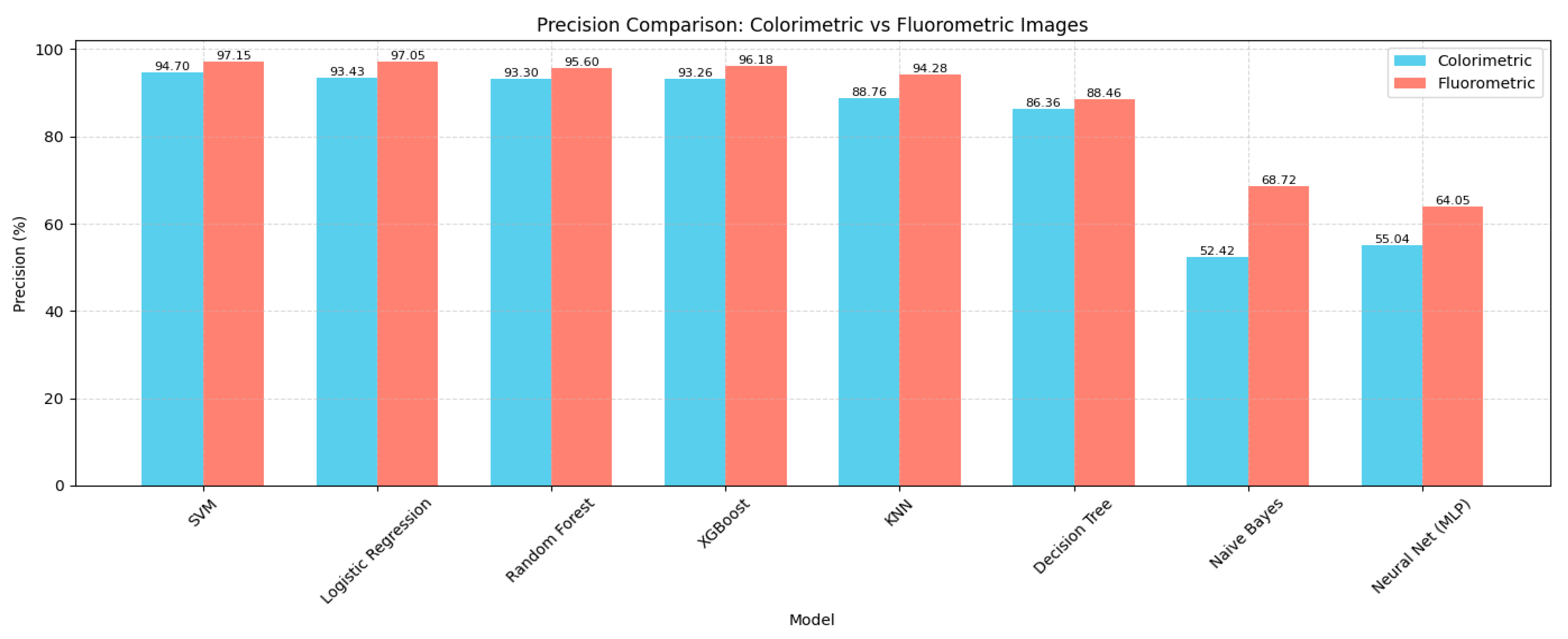
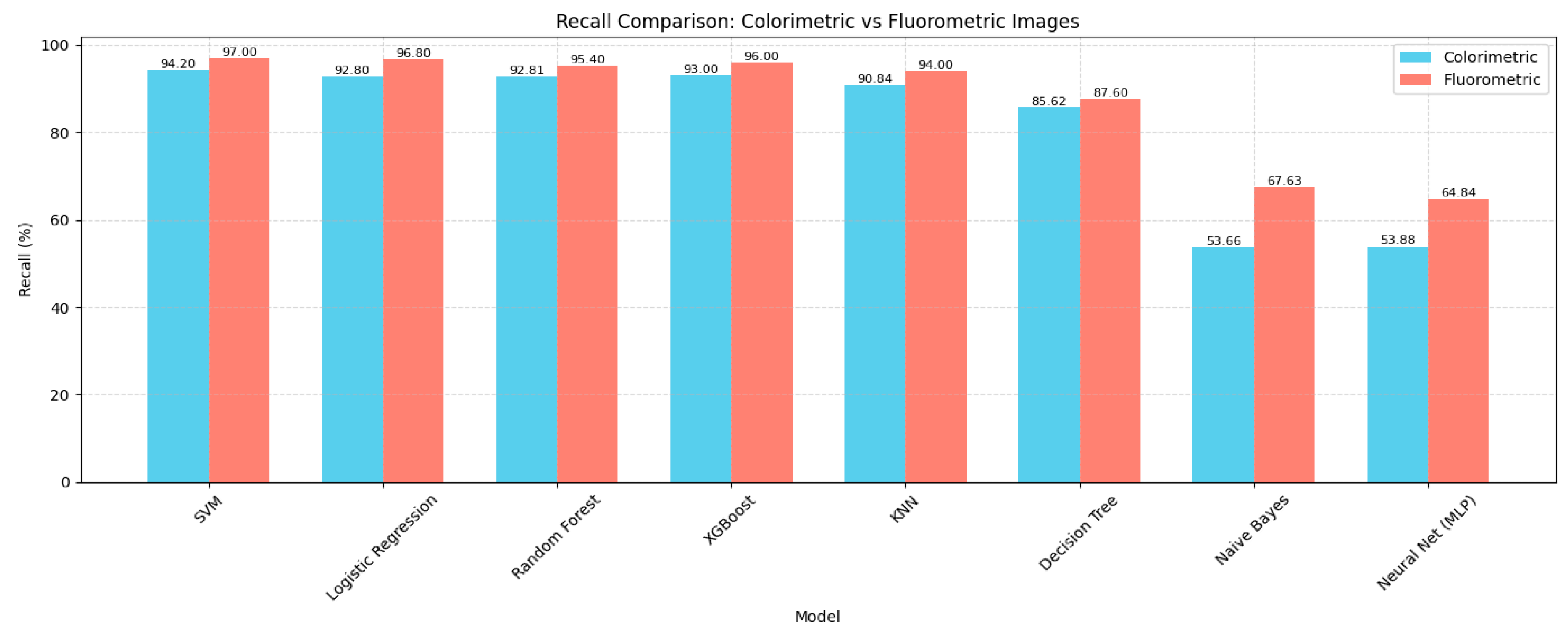
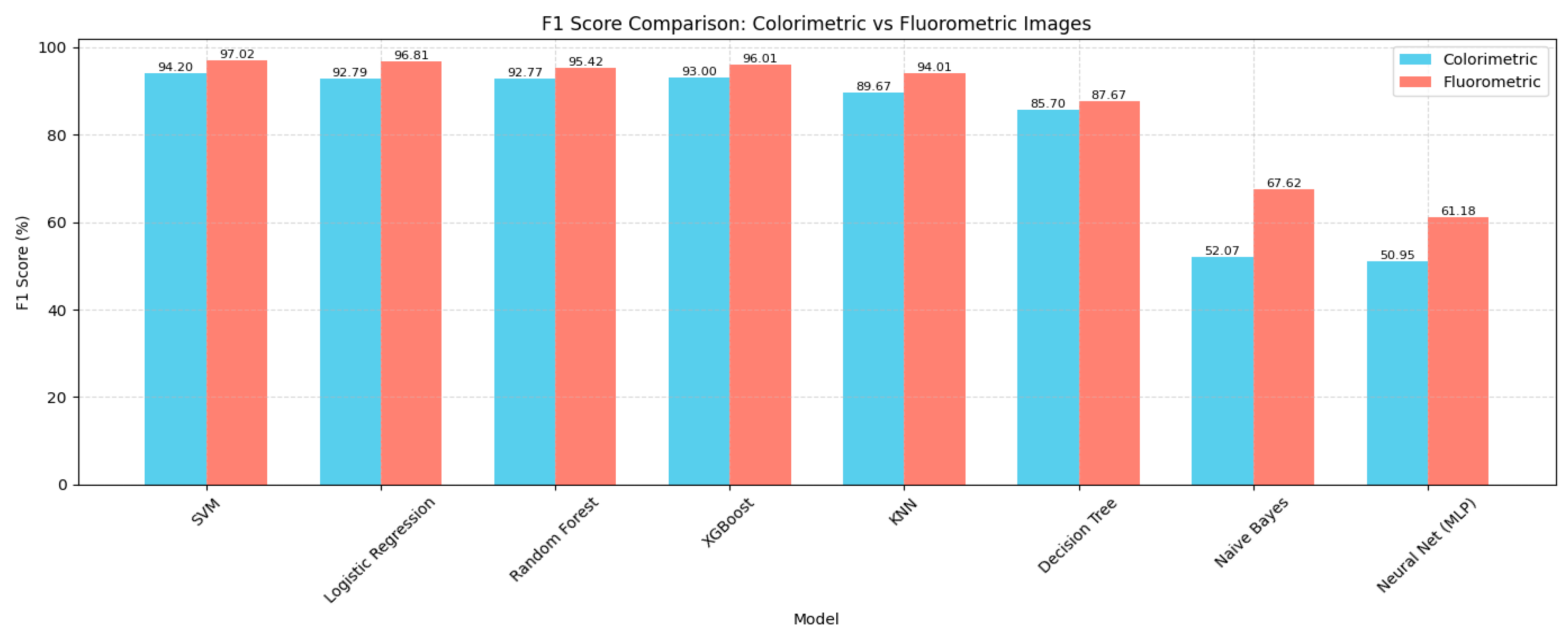
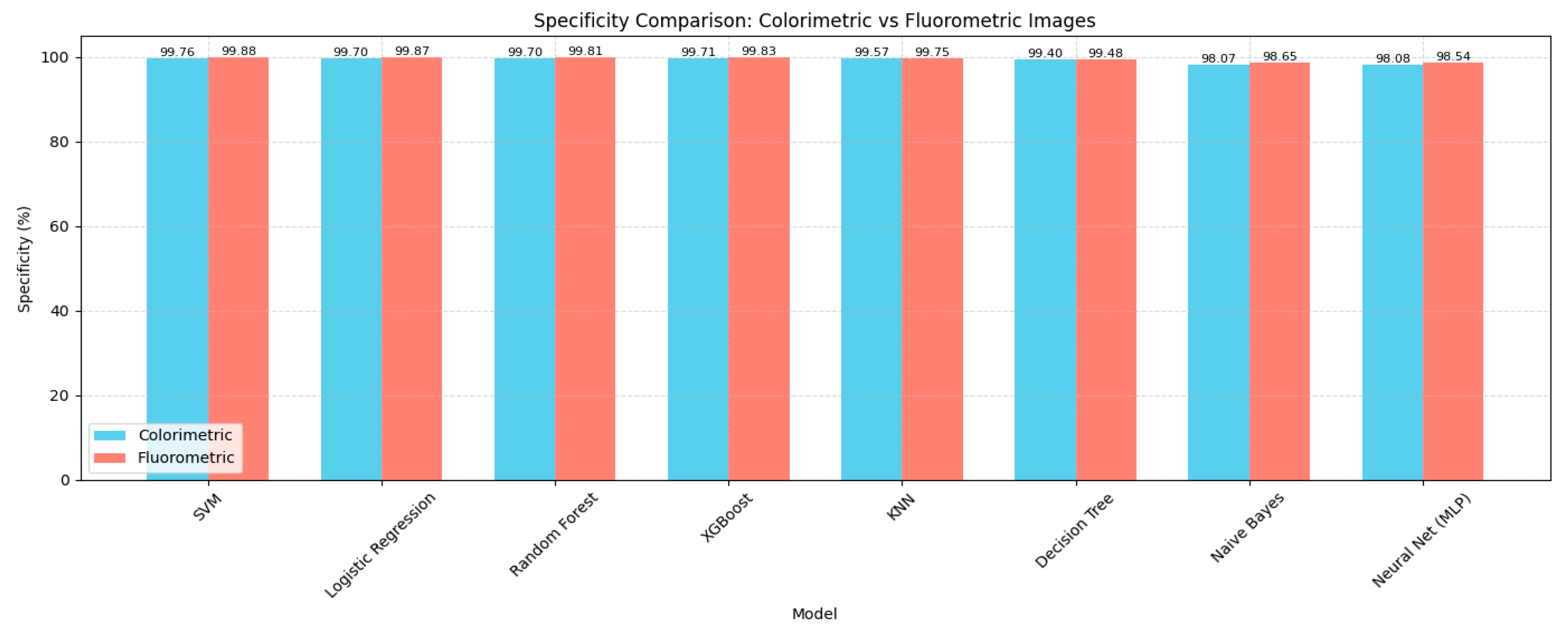
4.1. Model Performance Comparison
| Model | Accuracy (%) | Precision (%) | Recall (%) | F1 Score (%) | Specificity (%) |
|---|---|---|---|---|---|
| SVM | 93.91 | 94.7 | 94.2 | 94.2 | 99.76 |
| Logistic Regression | 92.95 | 93.43 | 92.8 | 92.79 | 99.7 |
| Random Forest | 92.5 | 93.3 | 92.81 | 92.77 | 99.7 |
| XGBoost | 92.48 | 93.26 | 93.0 | 93.0 | 99.71 |
| KNN | 88.76 | 88.76 | 90.84 | 89.67 | 99.57 |
| Decision Tree | 86.95 | 86.36 | 85.62 | 85.7 | 99.4 |
| Neural Network (MLP) | 52.55 | 55.04 | 53.88 | 50.95 | 98.08 |
| Naive Bayes | 52.3 | 52.42 | 53.66 | 52.07 | 98.07 |
| Model | Accuracy (%) | Precision (%) | Recall (%) | F1 Score (%) | Specificity (%) |
|---|---|---|---|---|---|
| SVM | 96.77 | 97.15 | 97.00 | 97.02 | 99.88 |
| Logistic Regression | 96.29 | 97.05 | 96.80 | 96.81 | 99.87 |
| Random Forest | 95.19 | 95.60 | 95.40 | 95.42 | 99.81 |
| XGBoost | 94.65 | 96.18 | 96.00 | 96.01 | 99.83 |
| KNN | 93.33 | 94.28 | 94.00 | 94.01 | 99.75 |
| Decision Tree | 88.82 | 88.46 | 87.60 | 87.67 | 99.48 |
| Naive Bayes | 67.17 | 68.72 | 67.63 | 67.62 | 98.65 |
| Neural Network (MLP) | 52.97 | 64.05 | 64.84 | 61.18 | 98.54 |
4.2. Cross-Validation Insights
5. Discussion
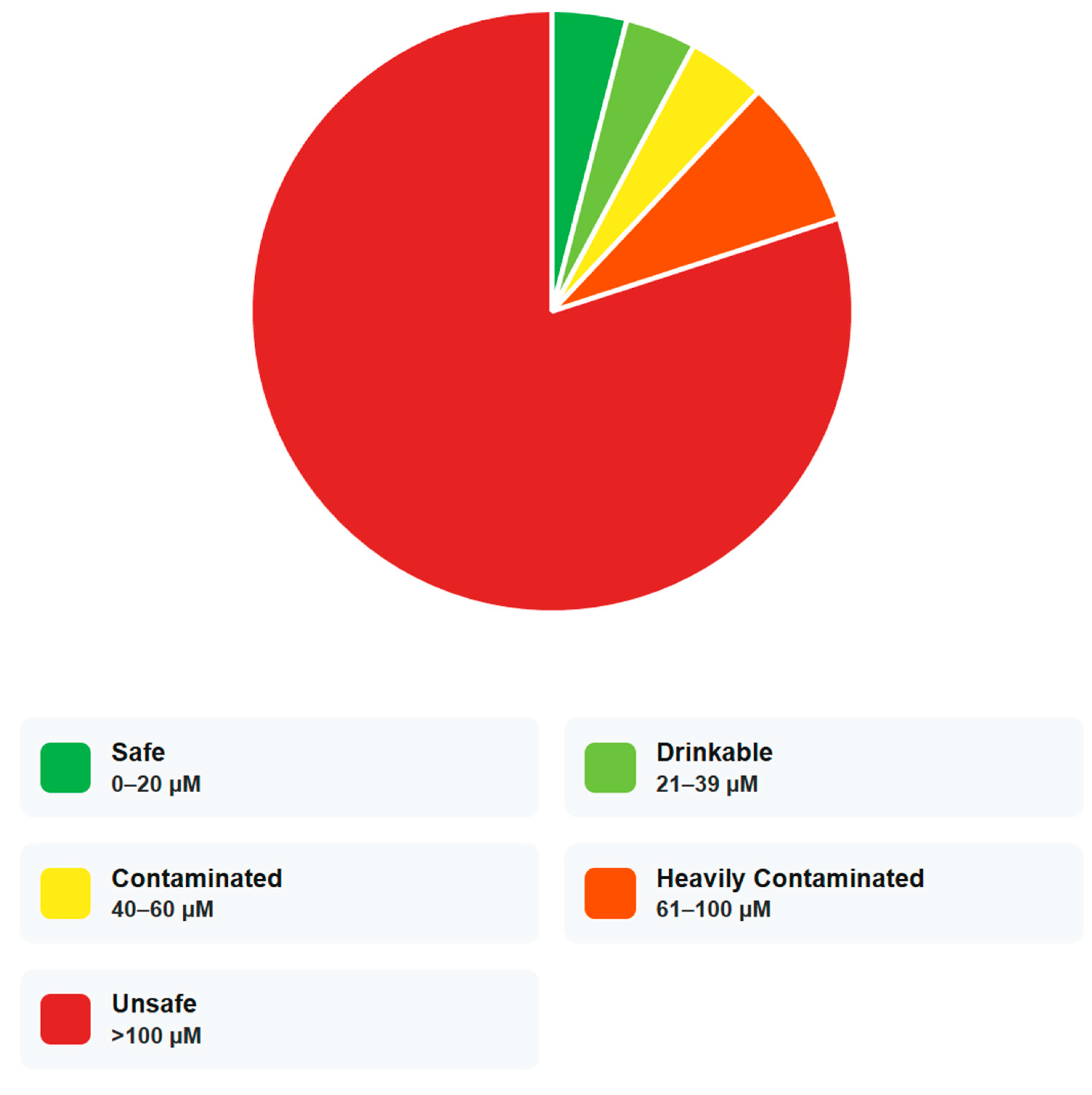
5.1. Decision Thresholds and Operational Risk Banding
| Copper Concentration (µM) | Category |
|---|---|
| 0–20 | Safe |
| 21–39 | Drinkable |
| 40–60 | Contaminated |
| 61–100 | Heavily Contaminated |
| >100 | Unsafe |
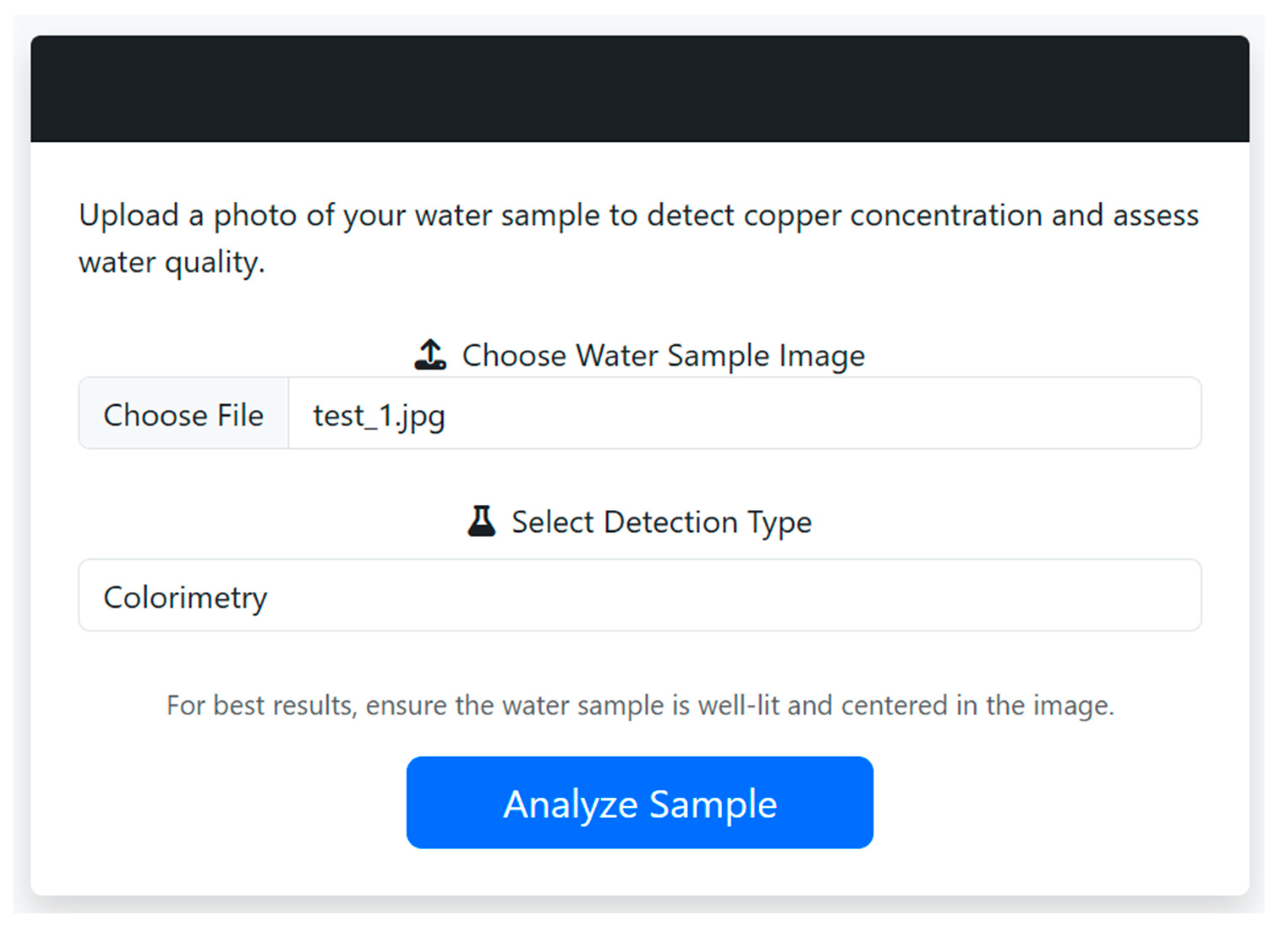
5.2. User Interface
5.2.1. CuLens Mobile Edition - Interface Overview


5.2.2. Upload and Analysis Workflow
5.2.3. Analysis Results and Interpretation
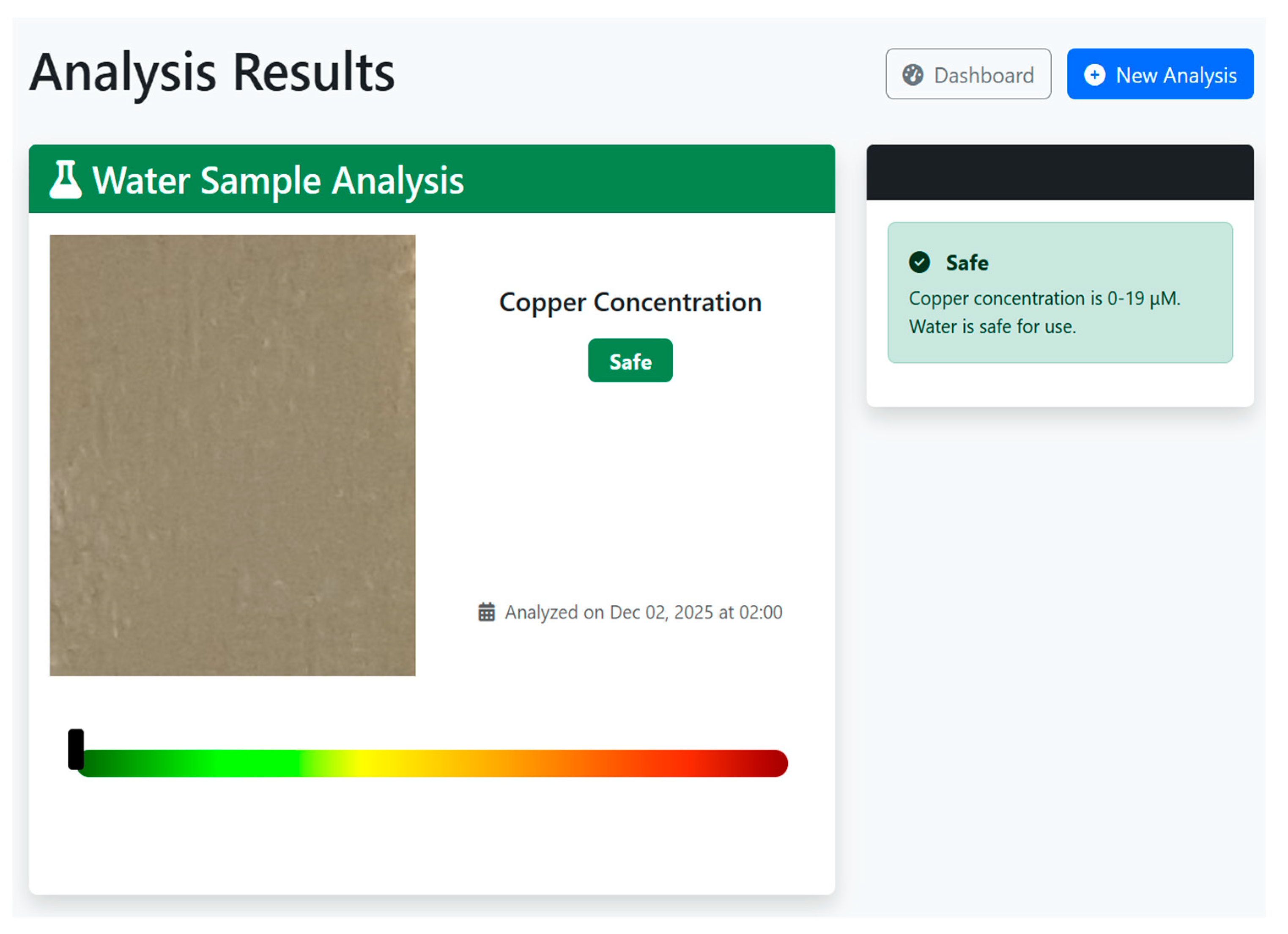
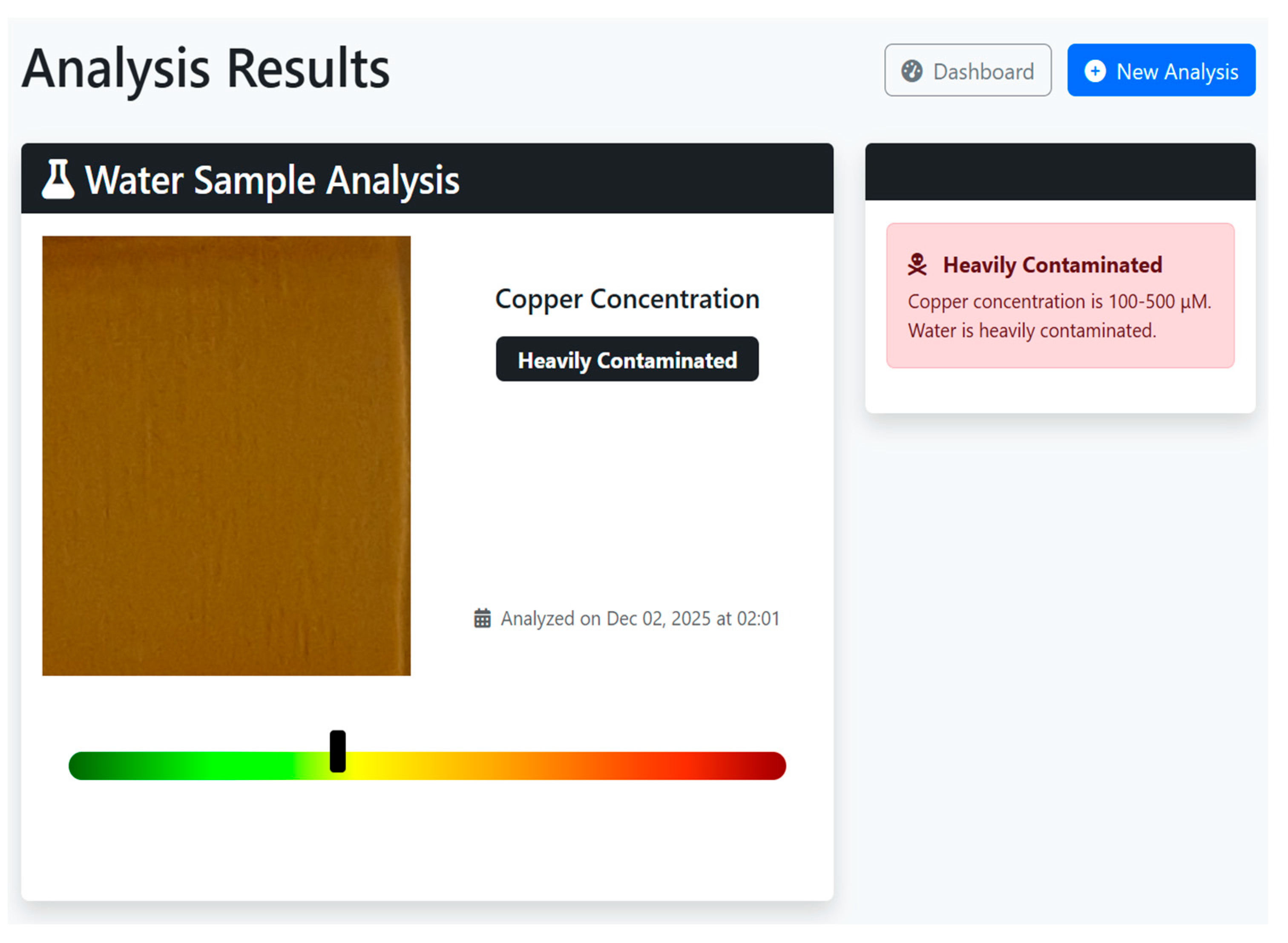
6. Conclusions
References
- J. J. Gordon and J. H. Quastel, ‘Effects of organic arsenicals on enzyme systems’, Biochem. J., vol. 42, no. 3, pp. 337–350, Jan. 1948.
- M. Jaishankar, T. Tseten, N. Anbalagan, B. B. Mathew, and K. N. Beeregowda, ‘Toxicity, mechanism and health effects of some heavy metals’, Interdiscip. Toxicol., vol. 7, no. 2, pp. 60–72, Jun. 2014.
- Z. Gao, N. Wu, X. Du, H. Li, X. Mei, and Y. Song, ‘Toxic Nephropathy Secondary to Chronic Mercury Poisoning: Clinical Characteristics and Outcomes’, Kidney Int. Reports, vol. 7, no. 6, pp. 1189–1197, 2022.
- M. S. Collin et al., ‘Bioaccumulation of lead (Pb) and its effects on human: A review’, Journal of Hazardous Materials Advances, vol. 7, no. May. Elsevier B.V., p. 100094, Aug-2022.
- M.-N. Georgaki et al., ‘Chromium in Water and Carcinogenic Human Health Risk’, Environments, vol. 10, no. 2, p. 33, Feb. 2023.
- M. Wallin, G. Sallsten, E. Fabricius-Lagging, C. Öhrn, T. Lundh, and L. Barregard, ‘Kidney cadmium levels and associations with urinary calcium and bone mineral density: a cross-sectional study in Sweden.’, Environ. Health, vol. 12, no. 1, p. 22, Mar. 2013.
- D. E. M. Camarena et al., ‘Differential impacts of nickel toxicity: NiO and NiSO4 on skin health and barrier function.’, Ecotoxicol. Environ. Saf., vol. 302, no. February, p. 118626, Sep. 2025.
- S. Krupanidhi, A. Sreekumar, and C. B. Sanjeevi, ‘Copper & biological health’, Indian J. Med. Res., vol. 128, no. 4, pp. 448–61, Oct. 2008.
- J. Sailer et al., ‘Deadly excess copper.’, Redox Biol., vol. 75, no. June, p. 103256, Sep. 2024.
- National Research Council, Copper in Drinking Water. Washington, D.C.: National Academies Press, 2000.
- A. Ayub and S. S. Ahmad, ‘Seasonal Assessment of Groundwater Contamination in Coal Mining Areas of Balochistan’, Sustainability, vol. 12, no. 17, p. 6889, Aug. 2020.
- R. S. Jaswant Sharma, ‘Geochemical and Hydrological Assessment Of Water Quality In Copper Mine Of Khetri Nagar Rajasthan’, Int. J. Creat. Res. Thoughts, vol. 1, no. 1, pp. 605–617, 2013.
- T. E. Gammons, Christopher H.; Duaime, ‘The Berkeley Pit and Surrounding Mine Waters of Butte’, Mont. Bur. Mines Geol., vol. 2, no. Geology of Montana, pp. 1–17, 2019.
- J. Donohue, ‘Copper in Drinking-water Background document for development of WHO Guidelines for Drinking-water Quality’, 2011.
- J. Dalmieda and P. Kruse, ‘Metal Cation Detection in Drinking Water’, Sensors, vol. 19, no. 23, p. 5134, Nov. 2019.
- T. Samanta and R. Shunmugam, ‘Colorimetric and fluorometric probes for the optical detection of environmental Hg(II) and As(III) ions’, Mater. Adv., vol. 2, no. 1, pp. 64–95, 2021.
- R. Pizzoferrato, R. Bisauriya, S. Antonaroli, M. Cabibbo, and A. J. Moro, ‘Colorimetric and Fluorescent Sensing of Copper Ions in Water through o-Phenylenediamine-Derived Carbon Dots’, Sensors, vol. 23, no. 6, p. 3029, Mar. 2023.
- J. Morell, A. Escobet, A. D. Dorado, and T. Escobet, ‘Design of a RGB-Arduino Device for Monitoring Copper Recovery from PCBs’, Processes, vol. 11, no. 5, p. 1319, Apr. 2023.
- J. L. D. Nelis et al., ‘The Efficiency of Color Space Channels to Quantify Color and Color Intensity Change in Liquids, pH Strips, and Lateral Flow Assays with Smartphones’, Sensors, vol. 19, no. 23, p. 5104, Nov. 2019.
- T. Hao et al., ‘Deep learning-assisted single-atom detection of copper ions by combining click chemistry and fast scan voltammetry’, Nat. Commun., vol. 15, no. 1, p. 10292, Nov. 2024.
- K. Yin, Y. Wu, S. Wang, and L. Chen, ‘A sensitive fluorescent biosensor for the detection of copper ion inspired by biological recognition element pyoverdine’, Sensors Actuators B Chem., vol. 232, pp. 257–263, Sep. 2016.
- M. K. Chattopadhyay et al., ‘Smartphone enabled machine learning approach assisted copper (II) quantification and opto-electrochemical explosive recognition by Aldazine-functionalized chemobiosensor’, Sensors and Actuators Reports, vol. 8, no. March, p. 100215, Dec. 2024.
- K. Kaewket, T. C. R. Outrequin, S. Deepaisarn, J. Wijitsak, P. Sunon, and K. Ngamchuea, ‘Machine Learning-Guided Cobalt@Copper Dual-Metal Electrochemical Sensor for Urinary Creatinine Detection’, ACS Sensors, vol. 10, no. 5, pp. 3471–3483, May 2025.
- R. Bisauriya, S. Antonaroli, M. Ardini, F. Angelucci, A. Ricci, and R. Pizzoferrato, ‘Tuning the Sensing Properties of N and S Co-Doped Carbon Dots for Colorimetric Detection of Copper and Cobalt in Water’, Sensors, vol. 22, no. 7, p. 2487, Mar. 2022.
- C. M. Bishop, Pattern Recognition and Machine Learning (Information Science and Statistics). 2006.
- R. Kohavi, ‘A Study of Cross-Validation and Bootstrap for Accuracy Estimation and Model Selection’, in IJCAI International Joint Conference on Artificial Intelligence, 1995, vol. 2, no. June, pp. 1137–1143.
- C. Cortes and V. Vapnik, ‘Support-vector networks’, Mach. Learn., vol. 20, no. 3, pp. 273–297, Sep. 1995.
- J. R. Quinlan, ‘Induction of decision trees’, Mach. Learn., vol. 1, no. 1, pp. 81–106, Mar. 1986.
- L. Breiman, ‘Random Forests’, Mach. Learn., vol. 45, no. 1, pp. 5–32, Oct. 2001.
- T. Chen and C. Guestrin, ‘XGBoost: A Scalable Tree Boosting System’, in Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, 2016, pp. 785–794.
- T. Cover and P. Hart, ‘Nearest neighbor pattern classification’, IEEE Trans. Inf. Theory, vol. 13, no. 1, pp. 21–27, Jan. 1967.
- D. M. W. Powers, ‘Evaluation: from precision, recall and F-measure to ROC, informedness, markedness and correlation’, Int. J. Mach. Learn. Technol., vol. 2, no. 1, pp. 37–63, Oct. 2020.
- J. Heaton, ‘Ian Goodfellow, Yoshua Bengio, and Aaron Courville: Deep learning’, Genet. Program. Evolvable Mach., vol. 19, no. 1–2, pp. 305–307, Jun. 2018.
- M. Sokolova and G. Lapalme, ‘A systematic analysis of performance measures for classification tasks’, Inf. Process. Manag., vol. 45, no. 4, pp. 427–437, Jul. 2009.
- K. P. Murphy, Machine Learning A Probabilistic Perspective, 2012th ed. The MIT Press, 2012.



| Detection approach | ML / data analysis | LOD, linear range (μM) | Automation & usability | Shortcomings | Year & References |
|---|---|---|---|---|---|
| FSV with click-chemistry amplification (lab bench) | Deep CNN (FSVNet) | Single-atom Cu2+ detection (reported in the 10-16 μM regime; specialized ultra-low range) | Automated voltammogram analysis with very high sensitivity | Requires specialized electrochemical setup and controlled lab conditions; not field-portable | 2024 - [1] |
| Smartphone colorimetric chemo-biosensor | SVM, RF, LR on HSV image features | LOD: 0.09 ppm Cu2+ (low-μM regime); linear working range reported across low-ppm Cu2+ concentrations | Smartphone-based, portable platform; ML improves reproducibility and enables rapid on-site screening | Sensitive to ambient lighting and camera variability; lower sensitivity than lab-grade electrochemistry | 2024 - [2] |
| Fluorometric pyoverdine-based probe | Conventional analytical calibration (non-ML) | LOD: 50 nM (0.05 μM); linear fluorescence response in the low-μM Cu2+ region (≈0.2-10 μM) | Simple probe preparation with established Cu2+ selectivity | Requires a fluorimeter; manual, instrument-dependent readout; no ML component | * 2016 - [3] |
| Co@Cu dual-metal electrochemical sensor (non-Cu target) | RF, Extra Trees, XGBoost | LOD and linear range defined for urinary creatinine (non-Cu analyte); high regression performance (R2 ≈0.98 - 0.99) | Low-cost printed electrodes; ML-assisted calibration and feature selection | Not a Cu2+ sensor; included only as an example of ML-guided electrochemical sensing | ** 2025 - [4] |
| Dual-mode RGB image sensor (colorimetric + fluorescent) | LR, SVM, RF, XGBoost | Five Cu2+ classes spanning 0- 500 μM (studied concentration window) | Fully portable, Smart Phone-dual-mode imaging; direct image-to-class ML decision | Discrete band-wise classification rather than continuous concentration; affected by optical noise and imaging conditions | Present work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




