Submitted:
13 February 2026
Posted:
13 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Toxicities Associated with CAR-T Cell Therapy
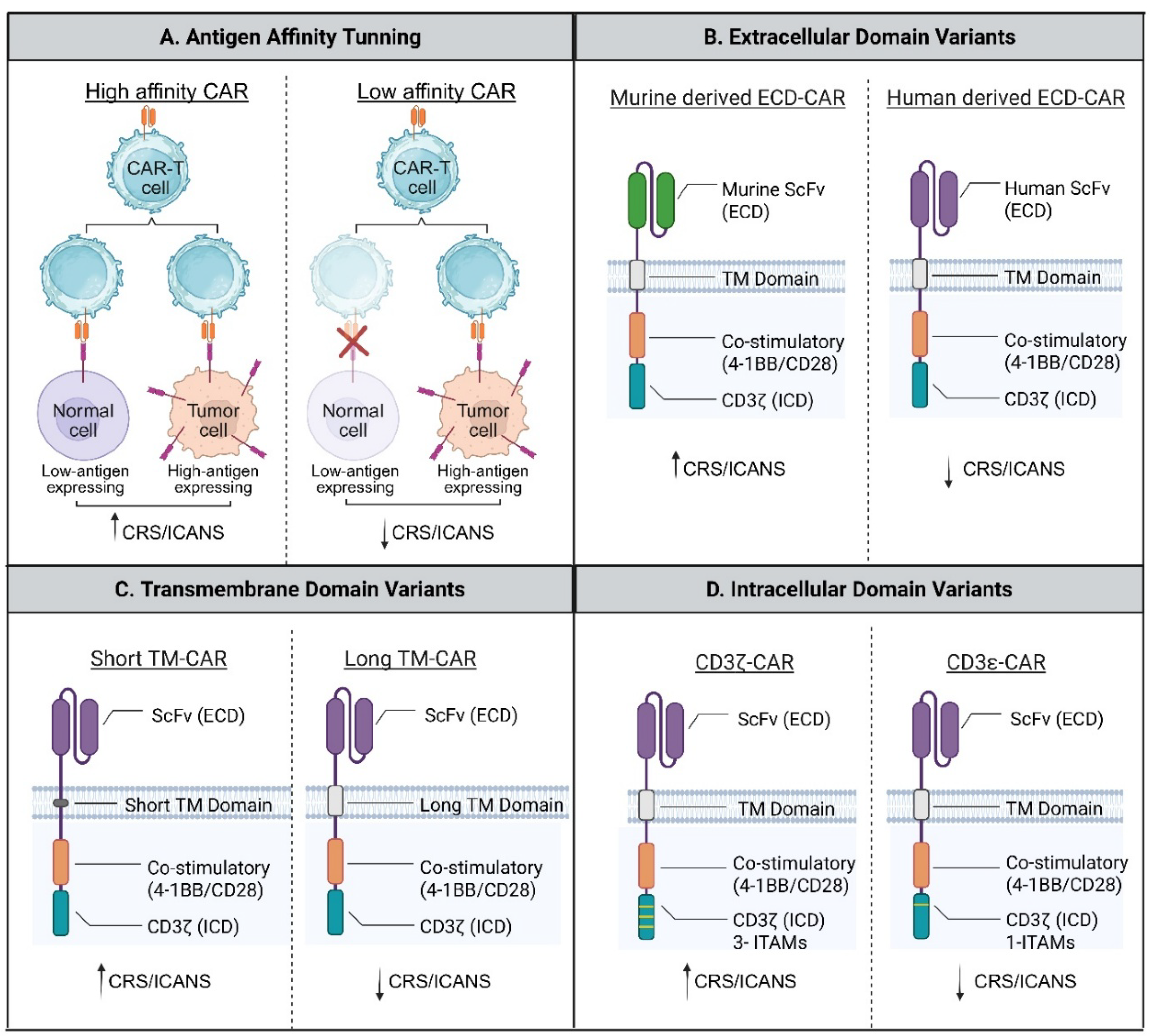
2.1. Self-Regulating CARs
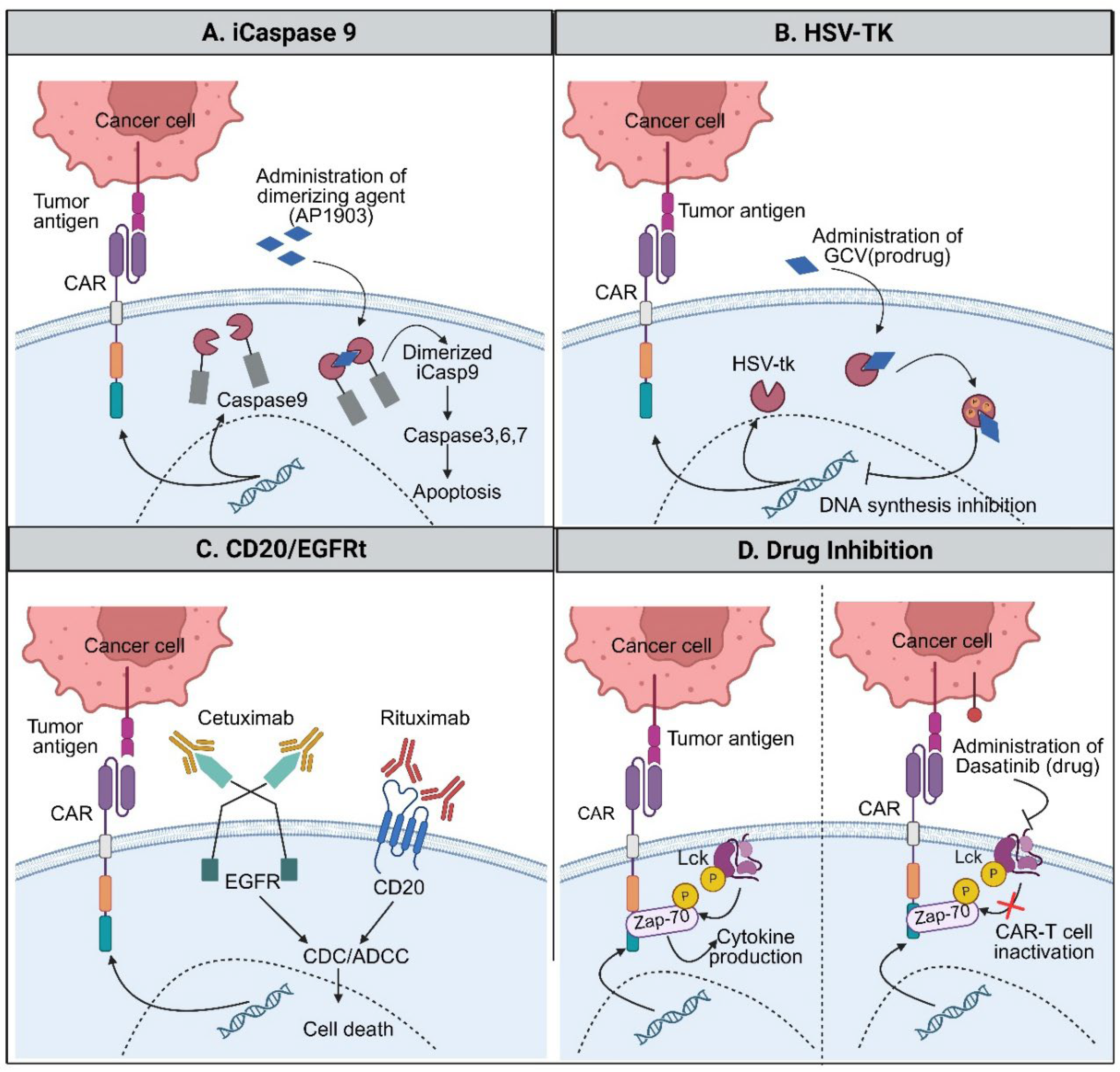
2.2. The ‘Switch Off’ CARs
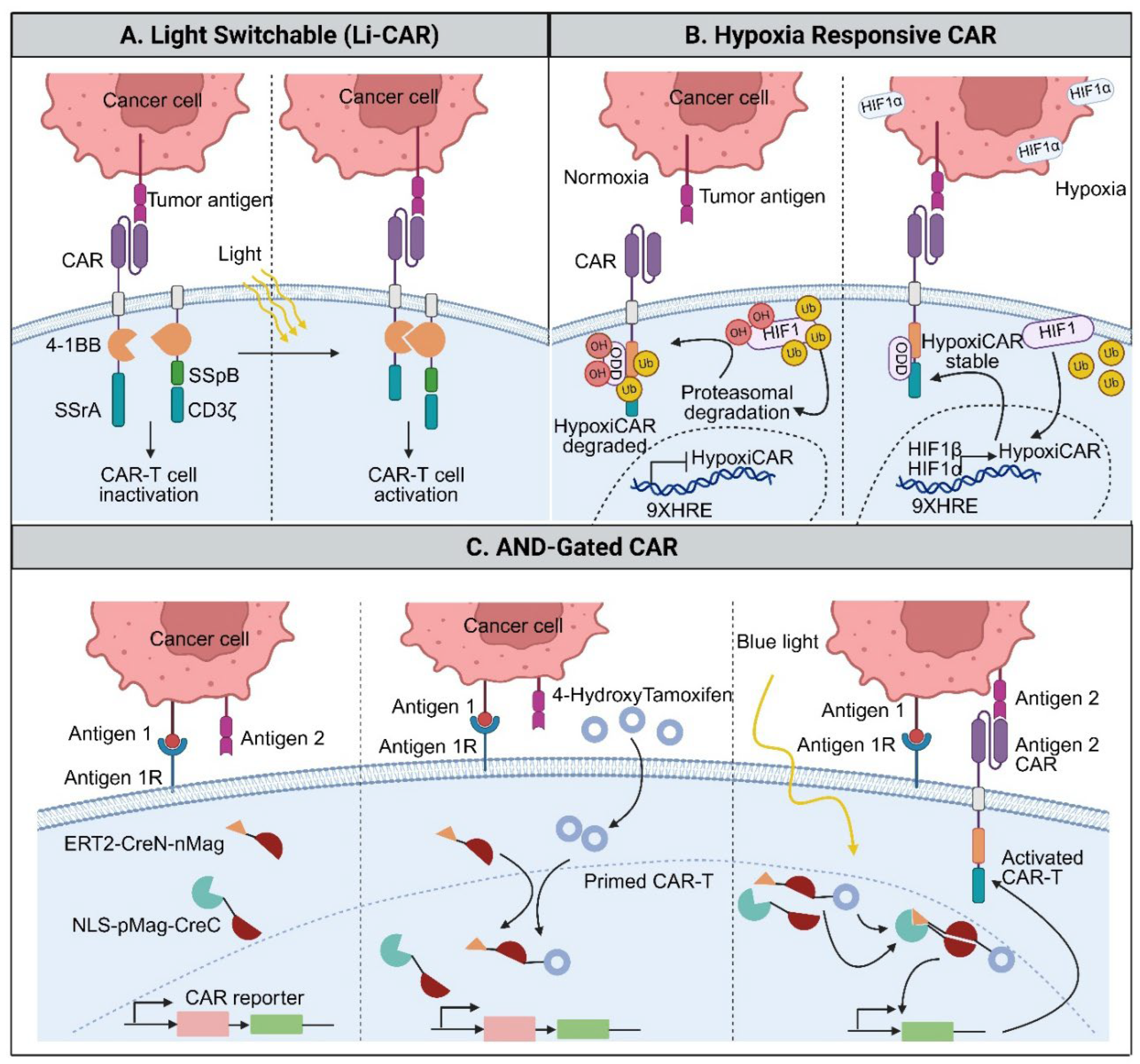
2.3. The ‘Switch On’ CARs
3. Immunosuppression in the Tumor Microenvironment
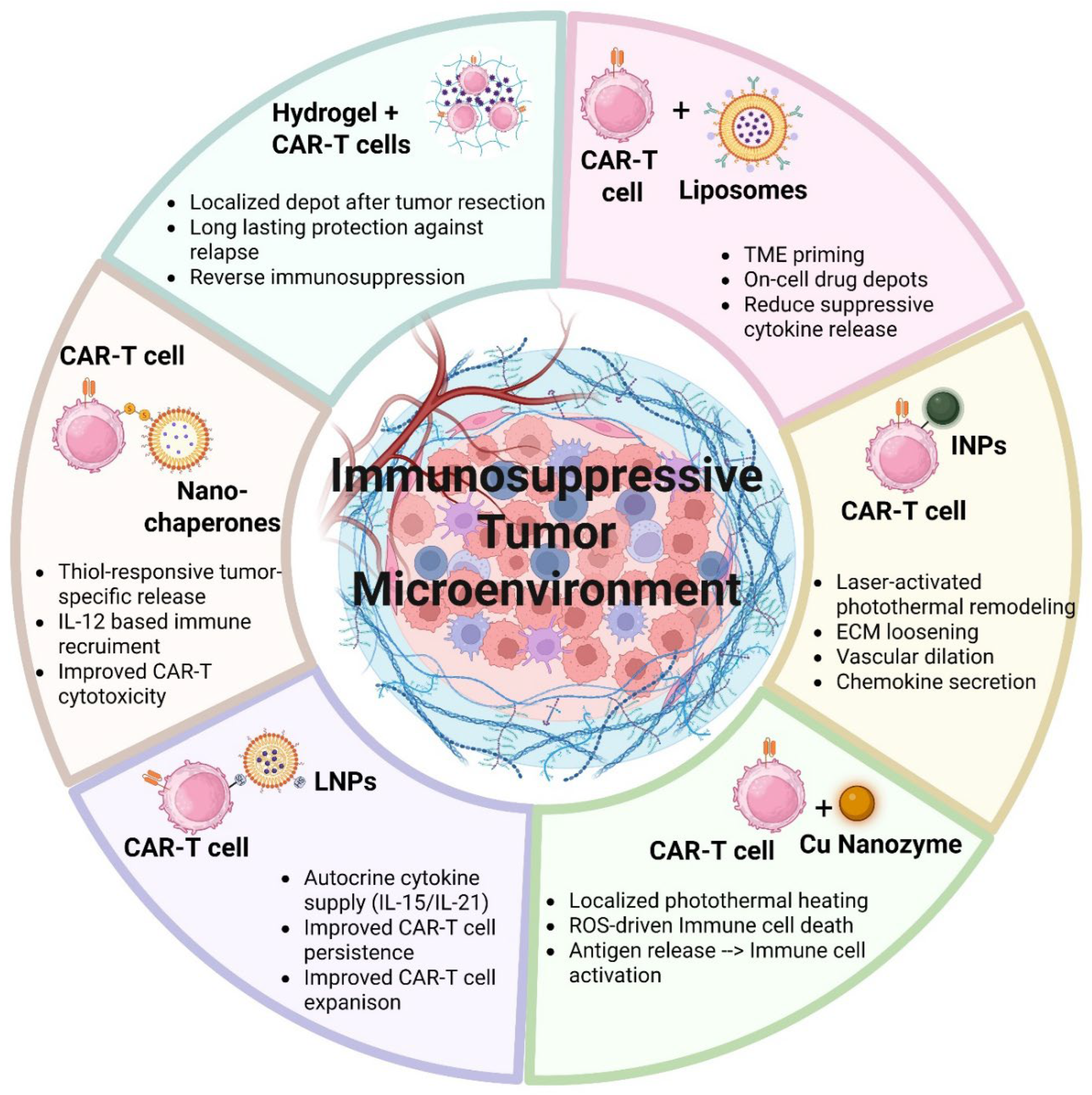
3.1. Nanomaterials
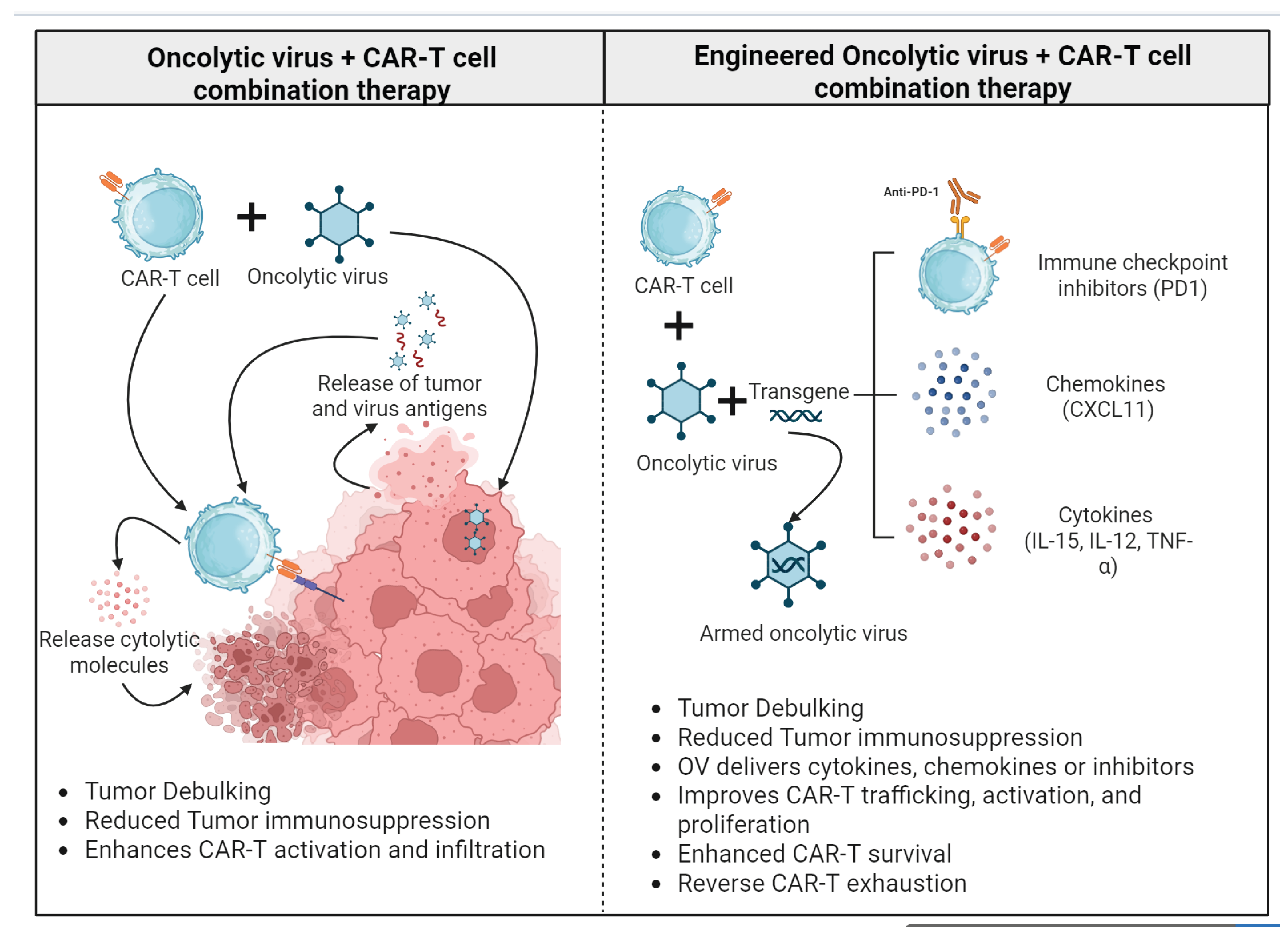
3.2. Oncolytic Viruses
4. Limited Efficacy and Longevity of CAR-T Cell Responses
4.1. CRISPR Technology
4.2. shRNA Technology
5. Trafficking Deficits and Infiltration Challenges
5.1. Chemokine-Based CAR-T Cell Advancements
6. Conclusions and Future Perspective
Author Contributions
Funding
Institutional review board statement
Informed consent statement
Data availability statement
Acknowledgments
Conflicts of interest
Abbreviation
References
- Asmamaw, DT; Tiruneh, GM; Dessie, TG; Tadele, AF; Wale, TW; Chekol, AE. Current updates on generations, approvals, and clinical trials of CAR T-cell therapy. Hum Vaccin Immunother. 2022, 18(6), 2114254. [Google Scholar] [CrossRef]
- Locke, FL; Ghobadi, A; Jacobson, CA; Miklos, DB; Lekakis, LJ; Oluwole, OO; et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1-2 trial. Lancet Oncol. 2019, 20(1), 31–42. [Google Scholar] [CrossRef]
- Maude, SL; Laetsch, TW; Buechner, J; Rives, S; Boyer, M; Bittencourt, H; et al. Tisagenlecleucel in Children and Young Adults with B-Cell Lymphoblastic Leukemia. N Eng J Med. 2018, 378(5), 439–48. [Google Scholar] [CrossRef]
- Munshi, NC; Anderson, LD; Shah, N; Madduri, D; Berdeja, J; Lonial, S; et al. Idecabtagene Vicleucel in Relapsed and Refractory Multiple Myeloma. N Eng J Med. 2021, 384(8), 705–16. [Google Scholar] [CrossRef]
- Wang, M; Munoz, J; Goy, A; Locke, FL; Jacobson, CA; Hill, BT; et al. Three-Year Follow-Up of KTE-X19 in Patients With Relapsed/Refractory Mantle Cell Lymphoma, Including High-Risk Subgroups, in the ZUMA-2 Study. J Clin Oncol. 2023, 41(3), 555–67. [Google Scholar] [CrossRef]
- Kuwana, Y; Asakura, Y; Utsunomiya, N; Nakanishi, M; Arata, Y; Itoh, S; et al. Expression of chimeric receptor composed of immunoglobulin-derived V resions and T-cell receptor-derived C regions. Biochem Biophys Res Commun. 1987, 149(3), 960–8. [Google Scholar] [CrossRef] [PubMed]
- Shirasu, N; Kuroki, M. Functional Design of Chimeric T-Cell Antigen Receptors for Adoptive Immunotherapy of Cancer: Architecture and Outcomes. Anticancer Res. 2012, 32(6), 2377–83. [Google Scholar]
- Honikel, MM; Olejniczak, SH. Co-Stimulatory Receptor Signaling in CAR-T Cells. Biomolecules 2022, 12(9), 1303. [Google Scholar] [CrossRef] [PubMed]
- Sadelain, M; Brentjens, R; Rivière, I. The promise and potential pitfalls of chimeric antigen receptors. Curr Opin Immunol. 2009, 21(2), 215–23. [Google Scholar] [CrossRef]
- Park, JH; Geyer, MB; Brentjens, RJ. CD19-targeted CAR T-cell therapeutics for hematologic malignancies: interpreting clinical outcomes to date. Blood 2016, 127(26), 3312–20. [Google Scholar] [CrossRef] [PubMed]
- Mitra, A; Barua, A; Huang, L; Ganguly, S; Feng, Q; He, B. From bench to bedside: the history and progress of CAR T cell therapy. Front Immunol. 2023, 14, 1188049. [Google Scholar] [CrossRef]
- Leyfman, Y. Chimeric antigen receptors: unleashing a new age of anti-cancer therapy. Cancer Cell Int. 2018, 18, 182. [Google Scholar] [CrossRef]
- Cappell, KM; Kochenderfer, JN. A comparison of chimeric antigen receptors containing CD28 versus 4-1BB costimulatory domains. Nat Rev Clin Oncol. 2021, 18(11), 715–27. [Google Scholar] [CrossRef]
- Smith, R; Shen, R. Complexities in comparing the impact of costimulatory domains on approved CD19 CAR functionality. J Transl Med. 2023, 21(1), 515. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S; Gu, C; Huang, L; Wu, H; Shi, J; Zhang, Z; et al. The third-generation anti-CD30 CAR T-cells specifically homing to the tumor and mediating powerful antitumor activity. Sci Rep. 2022, 12(1), 10488. [Google Scholar] [CrossRef] [PubMed]
- Tang, L; Pan, S; Wei, X; Xu, X; Wei, Q. Arming CAR-T cells with cytokines and more: Innovations in the fourth-generation CAR-T development. Mol Ther. 2023, 31(11), 3146–62. [Google Scholar] [CrossRef] [PubMed]
- Minguet, S; Maus, MV; Schamel, WW. From TCR fundamental research to innovative chimeric antigen receptor design. Nat Rev Immunol. 2025, 25(3), 212–24. [Google Scholar] [CrossRef]
- Mehrabadi, AZ; Ranjbar, R; Farzanehpour, M; Shahriary, A; Dorostkar, R; Hamidinejad, MA; et al. Therapeutic potential of CAR T cell in malignancies: A scoping review. Biomed Pharmacother. 2022, 146, 112512. [Google Scholar] [CrossRef]
- Lin, M-Y; Nam, E; Shih, RM; Shafer, A; Bouren, A; Ayala Ceja, M; et al. Self-regulating CAR-T cells modulate cytokine release syndrome in adoptive T-cell therapy. J Exp Med. 2024, 221(6), e20221988. [Google Scholar] [CrossRef]
- Dong, J; Wu, J; Jin, Y; Zheng, Z; Su, T; Shao, L; et al. In-depth analysis of the safety of CAR-T cell therapy for solid tumors. Front Immunol. 2025, 16, 1548979. [Google Scholar] [CrossRef]
- Xiao, X; Huang, S; Chen, S; Wang, Y; Sun, Q; Xu, X; et al. Mechanisms of cytokine release syndrome and neurotoxicity of CAR T-cell therapy and associated prevention and management strategies. J Exp Clin Cancer Res. 2021, 40(1), 367. [Google Scholar] [CrossRef]
- Hughes, AD; Teachey, DT; Diorio, C. Riding the storm: managing cytokine-related toxicities in CAR-T cell therapy. Semin Immunopathol 2024, 46(3-4), 5. [Google Scholar] [CrossRef]
- Ventin, M; Cattaneo, G; Maggs, L; Arya, S; Wang, X; Ferrone, CR. Implications of High Tumor Burden on Chimeric Antigen Receptor T-Cell Immunotherapy: A Review. JAMA Oncol. 2024, 10(1), 115. [Google Scholar] [CrossRef]
- Frigault, M; Rotte, A; Ansari, A; Gliner, B; Heery, C; Shah, B. Dose fractionation of CAR-T cells. A systematic review of clinical outcomes. J Exp Clin Cancer Res. 2023, 42(1), 11. [Google Scholar] [CrossRef]
- Rotte, A; Frigault, MJ; Ansari, A; Gliner, B; Heery, C; Shah, B. Dose-response correlation for CAR-T cells: a systematic review of clinical studies. J Immunother Cancer 2022, 10(12), e005678. [Google Scholar] [CrossRef]
- Wang, Z; Li, P; Zeng, X; Guo, J; Zhang, C; Fan, Z; et al. CAR-T therapy dilemma and innovative design strategies for next generation. Cell Death Dis. 2025, 16(1), 211. [Google Scholar] [CrossRef] [PubMed]
- Drent, E; Poels, R; Ruiter, R; van de Donk, NWCJ; Zweegman, S; Yuan, H; et al. Combined CD28 and 4-1BB Costimulation Potentiates Affinity-tuned Chimeric Antigen Receptor-engineered T Cells. Clin Cancer Res. 2019, 25(13), 4014–25. [Google Scholar] [CrossRef]
- Ghorashian, S; Kramer, AM; Onuoha, S; Wright, G; Bartram, J; Richardson, R; et al. Enhanced CAR T cell expansion and prolonged persistence in pediatric patients with ALL treated with a low-affinity CD19 CAR. Nat Med. 2019, 25(9), 1408–14. [Google Scholar] [CrossRef] [PubMed]
- Myers, RM; Li, Y; Barz Leahy, A; Barrett, DM; Teachey, DT; Callahan, C; et al. Humanized CD19-Targeted Chimeric Antigen Receptor (CAR) T Cells in CAR-Naive and CAR-Exposed Children and Young Adults With Relapsed or Refractory Acute Lymphoblastic Leukemia. J Clin Oncol. 2021, 39(27), 3044–55. [Google Scholar] [CrossRef] [PubMed]
- Wu, S; Luo, Q; Li, F; Zhang, S; Zhang, C; Liu, J; et al. Development of novel humanized CD19/BAFFR bicistronic chimeric antigen receptor T cells with potent antitumor activity against B-cell lineage neoplasms. Br J Haematol. 2024, 205(4), 1361–73. [Google Scholar] [CrossRef]
- Brudno, JN; Lam, N; Vanasse, D; Shen, Y; Rose, JJ; Rossi, J; et al. Safety and feasibility of anti-CD19 CAR T cells with fully human binding domains in patients with B-cell lymphoma. Nat Med. 2020, 26(2), 270–80. [Google Scholar] [CrossRef] [PubMed]
- Zenere, G; Wu, C; Midkiff, CC; Johnson, NM; Grice, CP; Wimley, WC; et al. Extracellular domain, hinge, and transmembrane determinants affecting surface CD4 expression of a novel anti-HIV chimeric antigen receptor (CAR) construct. PLos One 2024, 19(8), e0293990. [Google Scholar] [CrossRef]
- Gu, T; Hu, K; Si, X; Hu, Y; Huang, H. Mechanisms of immune effector cell-associated neurotoxicity syndrome after CAR-T treatment. WIREs Mech Dis. 2022, 14(6), e1576. [Google Scholar] [CrossRef]
- Fujiwara, K; Tsunei, A; Kusabuka, H; Ogaki, E; Tachibana, M; Okada, N; et al. Hinge and Transmembrane Domains of Chimeric Antigen Receptor Regulate Receptor Expression and Signaling Threshold. Cells 2020, 9(5), 1182. [Google Scholar] [CrossRef]
- Jayaraman, J; Mellody, MP; Hou, AJ; Desai, RP; Fung, AW; Pham, AHT; et al. CAR-T design: Elements and their synergistic function. eBioMedicine 2020, 58, 102931. [Google Scholar] [CrossRef] [PubMed]
- Ferreras, C; Fernández, L; Clares-Villa, L; Ibáñez-Navarro, M; Martín-Cortázar, C; Esteban-Rodríguez, I; et al. Facing CAR T Cell Challenges on the Deadliest Paediatric Brain Tumours. Cells 2021, 10(11), 2940. [Google Scholar] [CrossRef] [PubMed]
- Wu, L; Chen, J; Cai, R; Wang, X; Liu, Y; Zheng, Q; et al. Difference in Efficacy and Safety of Anti-CD19 Chimeric Antigen Receptor T-Cell Therapy Containing 4-1BB and CD28 Co-Stimulatory Domains for B-Cell Acute Lymphoblastic Leukemia. Cancers (Basel) 2023, 15(10), 2767. [Google Scholar] [CrossRef]
- Sun, C; Shou, P; Du, H; Hirabayashi, K; Chen, Y; Herring, LE; et al. THEMIS-SHP1 Recruitment by 4-1BB Tunes LCK-Mediated Priming of Chimeric Antigen Receptor-Redirected T Cells. Cancer Cell. 2020, 37(2), 216–25.e6. [Google Scholar] [CrossRef]
- Chuan, Tong; Yao, Wang; Weidong, Han. Structural optimization and prospect of chimeric antigen receptor T cells. Zhonghua Xueyexue Zazhi 2021, 42(9), 771–7. [Google Scholar] [CrossRef]
- Feucht, J; Sun, J; Eyquem, J; Ho, Y-J; Zhao, Z; Leibold, J; et al. Calibration of CAR activation potential directs alternative T cell fates and therapeutic potency. Nat Med. 2019, 25(1), 82–8. [Google Scholar] [CrossRef]
- Moghanloo, E; Mollanoori, H; Talebi, M; Pashangzadeh, S; Faraji, F; Hadjilooei, F; et al. Remote controlling of CAR-T cells and toxicity management: Molecular switches and next generation CARs. Translational Oncology 2021, 14(6), 101070. [Google Scholar] [CrossRef]
- Koehne, G; Doubrovin, M; Doubrovina, E; Zanzonico, P; Gallardo, HF; Ivanova, A; et al. Serial in vivo imaging of the targeted migration of human HSV-TK-transduced antigen-specific lymphocytes. Nat Biotechnol. 2003, 21(4), 405–13. [Google Scholar] [CrossRef] [PubMed]
- Greco, R; Oliveira, G; Stanghellini, MTL; Vago, L; Bondanza, A; Peccatori, J; et al. Improving the safety of cell therapy with the TK-suicide gene. Front Pharmacol. 2015, 6, 95. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T; Sakoda, Y; Adachi, K; Tokunaga, Y; Tamada, K. Therapeutic effects of anti-GM2 CAR-T cells expressing IL-7 and CCL19 for GM2-positive solid cancer in xenograft model. Cancer Med. 2023, 12(11), 12569–80. [Google Scholar] [CrossRef] [PubMed]
- Murty, S; Labanieh, L; Murty, T; Gowrishankar, G; Haywood, T; Alam, IS; et al. PET Reporter Gene Imaging and Ganciclovir-Mediated Ablation of Chimeric Antigen Receptor T Cells in Solid Tumors. Cancer Res. 2020, 80(21), 4731–40. [Google Scholar] [CrossRef]
- Haist, C; Poschinski, Z; Bister, A; Hoffmann, MJ; Grunewald, CM; Hamacher, A; et al. Engineering a single-chain variable fragment of cetuximab for CAR T-cell therapy against head and neck squamous cell carcinomas. Oral Oncol. 2022, 129, 105867. [Google Scholar] [CrossRef]
- Shabaneh, TB; Moffett, HF; Stull, SM; Derezes, T; Tait, LJ; Park, S; et al. Safety switch optimization enhances antibody-mediated elimination of CAR T cells. Front Mol Med. 2022, 2, 1026474. [Google Scholar] [CrossRef]
- Maus, MV. Designing CAR T cells for glioblastoma. OncoImmunology 2015, 4(12), e1048956. [Google Scholar] [CrossRef]
- Lin, H; Cheng, J; Zhu, L; Zeng, Y; Dai, Z; Zhang, Y; et al. Anti-CD5 CAR-T cells with a tEGFR safety switch exhibit potent toxicity control. Blood Cancer J 2024, 14(1), 98. [Google Scholar] [CrossRef]
- Lungova, K; Putman, M. Barriers to CAR T-cell therapy in rheumatology. Lancet Rheumatol. 2025, 7(3), e212-6. [Google Scholar] [CrossRef]
- Philip, B; Kokalaki, E; Mekkaoui, L; Thomas, S; Straathof, K; Flutter, B; et al. A highly compact epitope-based marker/suicide gene for easier and safer T-cell therapy. Blood 2014, 124(8), 1277–87. [Google Scholar] [CrossRef]
- McKee, B; Liu, S; Cai, PX; Yang, Z; Lan, T-H; Zhou, Y. Optogenetic control of T cells for immunomodulation. Essays Biochem. 2025, 69(2), 33–46. [Google Scholar] [CrossRef]
- Flugel, CL; Majzner, RG; Krenciute, G; Dotti, G; Riddell, SR; Wagner, DL; et al. Overcoming on-target, off-tumour toxicity of CAR T cell therapy for solid tumours. Nat Rev Clin Oncol. 2023, 20(1), 49–62. [Google Scholar] [CrossRef]
- Russell, GC; Hamzaoui, Y; Rho, D; Sutrave, G; Choi, JS; Missan, DS; et al. Synthetic biology approaches for enhancing safety and specificity of CAR-T cell therapies for solid cancers. Cytotherapy 2024, 26(8), 842–57. [Google Scholar] [CrossRef]
- Chandrasekar, S; Beach, JR; Oakes, PW. Shining a light on RhoA: Optical control of cell contractility. Int J Biochem Cell Biol. 2023, 161, 106442. [Google Scholar] [CrossRef]
- Wang, W; Wildes, CP; Pattarabanjird, T; Sanchez, MI; Glober, GF; Matthews, GA; et al. A light- and calcium-gated transcription factor for imaging and manipulating activated neurons. Nat Biotechnol. 2017, 35(9), 864–71. [Google Scholar] [CrossRef]
- Zhu, C; Wu, Q; Sheng, T; Shi, J; Shen, X; Yu, J; et al. Rationally designed approaches to augment CAR-T therapy for solid tumor treatment. Bioact Mater. 2024, 33, 377–95. [Google Scholar] [CrossRef]
- Allen, ME; Zhou, W; Thangaraj, J; Kyriakakis, P; Wu, Y; Huang, Z; et al. An AND-Gated Drug and Photoactivatable Cre-loxP System for Spatiotemporal Control in Cell-Based Therapeutics. ACS Synth Biol. 2019, 8(10), 2359–71. [Google Scholar] [CrossRef]
- Zhou, Y; Wei, Y; Yin, J; Kong, D; Li, W; Wang, X; et al. A rapid and efficient red-light-activated Cre recombinase system for genome engineering in mammalian cells and transgenic mice. Nucleic Acids Res. 2025, 53(15), gkaf758. [Google Scholar] [CrossRef]
- Juillerat, A; Marechal, A; Filhol, JM; Valogne, Y; Valton, J; Duclert, A; et al. An oxygen sensitive self-decision making engineered CAR T-cell. Sci Rep. 2017, 7, 39833. [Google Scholar] [CrossRef]
- Liao, Q; He, H; Mao, Y; Ding, X; Zhang, X; Xu, J. Engineering T cells with hypoxia-inducible chimeric antigen receptor (HiCAR) for selective tumor killing. Biomark Res. 2020, 8(1), 56. [Google Scholar] [CrossRef]
- Basheeruddin, M; Qausain, S. Hypoxia-Inducible Factor 1-Alpha (HIF-1α): An Essential Regulator in Cellular Metabolic Control. Cureus 2024, 16(7), e63852. [Google Scholar] [CrossRef]
- Kankeu Fonkoua, LA; Sirpilla, O; Sakemura, R; Siegler, EL; Kenderian, SS. CAR T cell therapy and the tumor microenvironment: Current challenges and opportunities. MolTher Oncolytics 2022, 25, 69–77. [Google Scholar] [CrossRef]
- Zhu, X; Chen, J; Li, W; Xu, Y; Shan, J; Hong, J; et al. Hypoxia-Responsive CAR-T Cells Exhibit Reduced Exhaustion and Enhanced Efficacy in Solid Tumors. Cancer Res. 2024, 84(1), 84–100. [Google Scholar] [CrossRef]
- Zheng, S; Che, X; Zhang, K; Bai, Y; Deng, H. Potentiating CAR-T cell function in the immunosuppressive tumor microenvironment by inverting the TGF-β signal. Mol Ther. 2025, 33(2), 688–702. [Google Scholar] [CrossRef]
- Farooq, MA; Ajmal, I; Hui, X; Chen, Y; Ren, Y; Jiang, W; et al. β2-Adrenergic Receptor Mediated Inhibition of T Cell Function and Its Implications for CAR-T Cell Therapy. Int J Mol Sci. 2023, 24(16), 12837. [Google Scholar] [CrossRef]
- Balakrishnan, PB; Sweeney, EE. Nanoparticles for Enhanced Adoptive T Cell Therapies and Future Perspectives for CNS Tumors. Front Immunol. 2021, 12, 600659. [Google Scholar] [CrossRef]
- Ponterio, E; Haas, TL; De Maria, R. Oncolytic virus and CAR-T cell therapy in solid tumors. Front Immunol. 2024, 15, 1455163. [Google Scholar] [CrossRef]
- Lafuente-Gómez, N; Kang, S; Mooney, DJ. Nanotechnology for CAR T cells and tumour-infiltrating lymphocyte therapies. Nat Nanotechnol. 2025, 20(9), 1186–98. [Google Scholar] [CrossRef]
- Zhang, F; Stephan, SB; Ene, CI; Smith, TT; Holland, EC; Stephan, MT. Nanoparticles That Reshape the Tumor Milieu Create a Therapeutic Window for Effective T-cell Therapy in Solid Malignancies. Cancer Res. 2018, 78(13), 3718–30. [Google Scholar] [CrossRef]
- Zheng, Y; Tang, L; Mabardi, L; Kumari, S; Irvine, DJ. Enhancing Adoptive Cell Therapy of Cancer through Targeted Delivery of Small-Molecule Immunomodulators to Internalizing or Noninternalizing Receptors. ACS Nano 2017, 11(3), 3089–100. [Google Scholar] [CrossRef]
- Wang, Z; Li, Z; Sun, Z; Wang, S; Ali, Z; Zhu, S; et al. Visualization nanozyme based on tumor microenvironment “unlocking” for intensive combination therapy of breast cancer. Sci Adv. 2020, 6(48), eabc8733. [Google Scholar] [CrossRef]
- Zeng, Q; Liu, Z; Niu, T; He, C; Qu, Y; Qian, Z. Application of nanotechnology in CAR-T-cell immunotherapy. Chinese Chemical Letters 2023, 34(3), 107747. [Google Scholar] [CrossRef]
- Chen, Z; Pan, H; Luo, Y; Yin, T; Zhang, B; Liao, J; et al. Nanoengineered CAR-T Biohybrids for Solid Tumor Immunotherapy with Microenvironment Photothermal-Remodeling Strategy. Small 2021, 17(14), 2007494. [Google Scholar] [CrossRef]
- Phan, NM; Nguyen, TL; Kim, J. Nanozyme-Based Enhanced Cancer Immunotherapy. Tissue Eng Regen Med. 2022, 19(2), 237–52. [Google Scholar] [CrossRef]
- Zhu, L; Liu, J; Zhou, G; Liu, T-M; Dai, Y; Nie, G; et al. Remodeling of Tumor Microenvironment by Tumor-Targeting Nanozymes Enhances Immune Activation of CAR T Cells for Combination Therapy. Small 2021, 17(43), 2102624. [Google Scholar] [CrossRef]
- Wu, W; Li, H; Chen, W; Hu, Y; Wang, Z; She, W; et al. CAR T Cell Membrane Camouflaged Nanocatalyst Augments CAR T Cell Therapy Efficacy Against Solid Tumor. Small 2024, 20(37), 2401299. [Google Scholar] [CrossRef]
- Huang, S; Zhao, Y; Shen, J. Advances in integrating biomaterials with CAR-T cells for enhancing solid tumor therapy. Chin J Cancer Res. 2025, 37(5), 742–58. [Google Scholar] [CrossRef]
- Bhagwat, AS; Torres, L; Shestova, O; Shestov, M; Mellors, PW; Fisher, HR; et al. Cytokine-mediated CAR T therapy resistance in AML. Nat Med. 2024, 30(12), 3697–708. [Google Scholar] [CrossRef]
- Biery, DN; Turicek, DP; Diorio, C; Schroeder, BA; Shah, NN. Need for standardization of cytokine profiling in CAR T cell therapy. Mol Ther. 2024, 32(9), 2979–83. [Google Scholar] [CrossRef]
- Stephan, MT; Moon, JJ; Um, SH; Bershteyn, A; Irvine, DJ. Therapeutic cell engineering with surface-conjugated synthetic nanoparticles. Nat Med. 2010, 16(9), 1035–41. [Google Scholar] [CrossRef]
- Luo, Y; Chen, Z; Sun, M; Li, B; Pan, F; Ma, A; et al. IL-12 nanochaperone-engineered CAR T cell for robust tumor-immunotherapy. Biomaterials 2022, 281, 121341. [Google Scholar] [CrossRef]
- Hu, Q; Li, H; Archibong, E; Chen, Q; Ruan, H; Ahn, S; et al. Inhibition of post-surgery tumour recurrence via a hydrogel releasing CAR-T cells and anti-PDL1-conjugated platelets. Nat Biomed Eng. 2021, 5(9), 1038–47. [Google Scholar] [CrossRef]
- Fukuhara, H; Ino, Y; Todo, T. Oncolytic virus therapy: A new era of cancer treatment at dawn. Cancer Sci. 2016, 107(10), 1373–9. [Google Scholar] [CrossRef]
- Lawler, SE; Speranza, M-C; Cho, C-F; Chiocca, EA. Oncolytic Viruses in Cancer Treatment: A Review. JAMA Oncol. 2017, 3(6), 841–9. [Google Scholar] [CrossRef]
- Shalhout, SZ; Miller, DM; Emerick, KS; Kaufman, HL. Therapy with oncolytic viruses: progress and challenges. Nat Rev Clin Oncol. 2023, 20(3), 160–77. [Google Scholar] [CrossRef]
- Ajina, A; Maher, J. Prospects for combined use of oncolytic viruses and CAR T-cells. J Immunother Cancer 2017, 5(1), 90. [Google Scholar] [CrossRef]
- Evgin, L; Kottke, T; Tonne, J; Thompson, J; Huff, AL; van Vloten, J; et al. Oncolytic virus–mediated expansion of dual-specific CAR T cells improves efficacy against solid tumors in mice. Sci Transl Med. 2022, 14(640), eabn2231. [Google Scholar] [CrossRef]
- Wang, S; Li, Y; Xu, C; Dong, J; Wei, J. An oncolytic vaccinia virus encoding hyaluronidase reshapes the extracellular matrix to enhance cancer chemotherapy and immunotherapy. J Immunother Cancer 2024, 12(3), e008431. [Google Scholar] [CrossRef]
- McGrath, K; Dotti, G. Combining Oncolytic Viruses with Chimeric Antigen Receptor T Cell Therapy. Human Gene Ther. 2021, 32(3-4), 150–7. [Google Scholar] [CrossRef]
- Munn, DH; Bronte, V. Immune suppressive mechanisms in the tumor microenvironment. Curr Opin Immunol. 2016, 39, 1–6. [Google Scholar] [CrossRef]
- Watanabe, K; Luo, Y; Da, T; Guedan, S; Ruella, M; Scholler, J; et al. Pancreatic cancer therapy with combined mesothelin-redirected chimeric antigen receptor T cells and cytokine-armed oncolytic adenoviruses. JCI Insight 2018, 3(7), e99573. [Google Scholar] [CrossRef]
- Shaw, AR; Porter, CE; Watanabe, N; Tanoue, K; Sikora, A; Gottschalk, S; et al. Adenovirotherapy Delivering Cytokine and Checkpoint Inhibitor Augments CAR T Cells against Metastatic Head and Neck Cancer. Mol Ther. 2017, 25(11), 2440–51. [Google Scholar] [CrossRef]
- Zarezadeh Mehrabadi, A; Roozbahani, F; Ranjbar, R; Farzanehpour, M; Shahriary, A; Dorostkar, R; et al. Overview of the pre-clinical and clinical studies about the use of CAR-T cell therapy of cancer combined with oncolytic viruses. World J Surg Onc 2022, 20(1), 16. [Google Scholar] [CrossRef]
- Li, C; Mei, H; Hu, Y. Applications and explorations of CRISPR/Cas9 in CAR T-cell therapy. Brief Funct Genomics 2020, 19(3), 175–82. [Google Scholar] [CrossRef] [PubMed]
- Razeghian, E; Nasution, MKM; Rahman, HS; Gardanova, ZR; Abdelbasset, WK; Aravindhan, S; et al. A deep insight into CRISPR/Cas9 application in CAR-T cell-based tumor immunotherapies. Stem Cell Res Ther. 2021, 12(1), 428. [Google Scholar] [CrossRef] [PubMed]
- Tao, R; Han, X; Bai, X; Yu, J; Ma, Y; Chen, W; et al. Revolutionizing cancer treatment: enhancing CAR-T cell therapy with CRISPR/Cas9 gene editing technology. Front Immunol. 2024, 15, 1354825. [Google Scholar] [CrossRef] [PubMed]
- Abdoli Shadbad, M; Hemmat, N; Khaze Shahgoli, V; Derakhshani, A; Baradaran, F; Brunetti, O; et al. A Systematic Review on PD-1 Blockade and PD-1 Gene-Editing of CAR-T Cells for Glioma Therapy: From Deciphering to Personalized Medicine. Front Immunol. 2022, 12, 788211. [Google Scholar] [CrossRef]
- McGowan, E; Lin, Q; Ma, G; Yin, H; Chen, S; Lin, Y. PD-1 disrupted CAR-T cells in the treatment of solid tumors: Promises and challenges. Biomed Pharmacother. 2020, 121, 109625. [Google Scholar] [CrossRef]
- Wang, Z; Li, N; Feng, K; Chen, M; Zhang, Y; Liu, Y; et al. Phase I study of CAR-T cells with PD-1 and TCR disruption in mesothelin-positive solid tumors. Cell Mol Immunol. 2021, 18(9), 2188–98. [Google Scholar] [CrossRef]
- Shi, L; Meng, T; Zhao, Z; Han, J; Zhang, W; Gao, F; et al. CRISPR knock out CTLA-4 enhances the anti-tumor activity of cytotoxic T lymphocytes. Gene 2017, 636, 36–41. [Google Scholar] [CrossRef]
- Cianciotti, BC; Magnani, ZI; Ugolini, A; Camisa, B; Merelli, I; Vavassori, V; et al. TIM-3, LAG-3, or 2B4 gene disruptions increase the anti-tumor response of engineered T cells. Front Immunol. 2024, 15, 1315283. [Google Scholar] [CrossRef]
- Ren, J; Liu, X; Fang, C; Jiang, S; June, CH; Zhao, Y. Multiplex Genome Editing to Generate Universal CAR T Cells Resistant to PD1 Inhibition. Clin Cancer Res. 2017, 23(9), 2255–66. [Google Scholar] [CrossRef]
- Jenkins, E; Whitehead, T; Fellermeyer, M; Davis, SJ; Sharma, S. The current state and future of T-cell exhaustion research. Oxf Open Immunol 2023, 4(1), iqad006. [Google Scholar] [CrossRef]
- Nakagawara, K; Ando, M; Srirat, T; Mise-Omata, S; Hayakawa, T; Ito, M; et al. NR4A ablation improves mitochondrial fitness for long persistence in human CAR-T cells against solid tumors. J Immunother Cancer 2024, 12(8), e008665. [Google Scholar] [CrossRef] [PubMed]
- Xiang, M; Li, H; Zhan, Y; Ma, D; Gao, Q; Fang, Y. Functional CRISPR screens in T cells reveal new opportunities for cancer immunotherapies. Mol Cancer 2024, 23(1), 73. [Google Scholar] [CrossRef] [PubMed]
- Tang, N; Cheng, C; Zhang, X; Qiao, M; Li, N; Mu, W; et al. TGF-β inhibition via CRISPR promotes the long-term efficacy of CAR T cells against solid tumors. JCI Insight 2020, 5(4), e133977. [Google Scholar] [CrossRef]
- Merlin, JPJ; Abrahamse, H. Optimizing CRISPR/Cas9 precision: Mitigating off-target effects for safe integration with photodynamic and stem cell therapies in cancer treatment. Biomed Pharmacother. 2024, 180, 117516. [Google Scholar] [CrossRef] [PubMed]
- Dimitri, A; Herbst, F; Fraietta, JA. Engineering the next-generation of CAR T-cells with CRISPR-Cas9 gene editing. Mol Cancer 2022, 21(1), 78. [Google Scholar] [CrossRef]
- Su, Q; Yao, J; Farooq, MA; Ajmal, I; Duan, Y; He, C; et al. Modulating Cholesterol Metabolism via ACAT1 Knockdown Enhances Anti-B-Cell Lymphoma Activities of CD19-Specific Chimeric Antigen Receptor T Cells by Improving the Cell Activation and Proliferation. Cells 2024, 13(6), 555. [Google Scholar] [CrossRef]
- Grosser, R; Cherkassky, L; Chintala, N; Adusumilli, PS. Combination Immunotherapy with CAR T Cells and Checkpoint Blockade for the Treatment of Solid Tumors. Cancer Cell. 2019, 36(5), 471–82. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H; Yao, J; Ajmal, I; Farooq, MA; Jiang, W. shRNA-mediated gene silencing of HDAC11 empowers CAR-T cells against prostate cancer. Front Immunol. 2024, 15, 1369406. [Google Scholar] [CrossRef]
- Ajmal, I; Farooq, MA; Duan, Y; Yao, J; Gao, Y; Hui, X; et al. Intrinsic ADRB2 inhibition improves CAR-T cell therapy efficacy against prostate cancer. Mol Ther. 2024, 32(10), 3539–57. [Google Scholar] [CrossRef]
- Gao, Y; Lin, H; Guo, D; Cheng, S; Zhou, Y; Zhang, L; et al. Suppression of 4.1R enhances the potency of NKG2D-CAR T cells against pancreatic carcinoma via activating ERK signaling pathway. Oncogenesis 2021, 10(9), 62. [Google Scholar] [CrossRef]
- Ajmal, I; Farooq, MA; Abbas, SQ; Shah, J; Majid, M; Jiang, W. Isoprenaline and salbutamol inhibit pyroptosis and promote mitochondrial biogenesis in arthritic chondrocytes by downregulating β-arrestin and GRK2. Front Pharmacol. 2022, 13, 996321. [Google Scholar] [CrossRef]
- Zhou, J; Yu, J; Wang, Y; Wang, H; Wang, J; Wang, Y; et al. ShRNA-mediated silencing of PD-1 augments the efficacy of chimeric antigen receptor T cells on subcutaneous prostate and leukemia xenograft. Biomed Pharmacother. 2021, 137, 111339. [Google Scholar] [CrossRef]
- Jafarzadeh, L; Masoumi, E; Mirzaei, HR; Alishah, K; Fallah-Mehrjardi, K; Khakpoor-Koosheh, M; et al. Targeted knockdown of Tim3 by short hairpin RNAs improves the function of anti-mesothelin CAR T cells. Mol Immunol 2021, 139, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Koyama, S; Akbay, EA; Li, YY; Herter-Sprie, GS; Buczkowski, KA; Richards, WG; et al. Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints. Nat Commun. 2016, 7, 10501. [Google Scholar] [CrossRef]
- Park, Y; Jeon, D-H; Yi, H; Lee, S-H; Kim, HC. Characteristics of CRC01, an investigational CD19 CAR-T product with PD-1 and TIGIT genes silenced by shRNAs. J Clin Oncol. 2022, 40, e14514–e14514. [Google Scholar] [CrossRef]
- Steklov, M; Lecalve, B; Marijse, J; Huberty, F; Ramelot, N; Jacques-Hespel, C; et al. 146 Evolving Mutliplexed shRNA to generate tailored CAR T cell therapy. J Immunother Cancer 2021, 9, A154–154. [Google Scholar] [CrossRef]
- Foeng, J; Comerford, I; McColl, SR. Harnessing the chemokine system to home CAR-T cells into solid tumors. Cell Rep Med. 2022, 3(3), 100543. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P-F; Wang, C; Zhang, L; Li, Q. Reversing Chemokine/Chemokine Receptor Mismatch to Enhance the Antitumor Efficacy of CAR-T Cells. Immunotherapy 2022, 14(6), 459–73. [Google Scholar] [CrossRef]
- Emami-Shahri, N; Foster, J; Kashani, R; Gazinska, P; Cook, C; Sosabowski, J; et al. Clinically compliant spatial and temporal imaging of chimeric antigen receptor T-cells. Nat Commun. 2018, 9(1), 1081. [Google Scholar] [CrossRef] [PubMed]
- Chen, T; Wang, M; Chen, Y; Liu, Y. Current challenges and therapeutic advances of CAR-T cell therapy for solid tumors. Cancer Cell Int. 2024, 24(1), 133. [Google Scholar] [CrossRef]
- Zhang, Y; Guan, X; Jiang, P. Cytokine and Chemokine Signals of T-Cell Exclusion in Tumors. Front Immunol. 2020, 11, 594609. [Google Scholar] [CrossRef]
- Liu, G; Rui, W; Zhao, X; Lin, X. Enhancing CAR-T cell efficacy in solid tumors by targeting the tumor microenvironment. Cell Mol Immunol. 2021, 18(5), 1085–95. [Google Scholar] [CrossRef]
- Jin, L; Tao, H; Karachi, A; Long, Y; Hou, AY; Na, M; et al. CXCR1- or CXCR2-modified CAR T cells co-opt IL-8 for maximal antitumor efficacy in solid tumors. Nat Commun. 2019, 10(1), 4016. [Google Scholar] [CrossRef]
- Dai, Z; Lin, X; Wang, X; Zou, X; Yan, Y; Wang, R; et al. Ectopic CXCR2 expression cells improve the anti-tumor efficiency of CAR-T cells and remodel the immune microenvironment of pancreatic ductal adenocarcinoma. Cancer Immunol Immunother. 2024, 73(4), 61. [Google Scholar] [CrossRef]
- Girbl, T; Lenn, T; Perez, L; Rolas, L; Barkaway, A; Thiriot, A; et al. Distinct Compartmentalization of the Chemokines CXCL1 and CXCL2 and the Atypical Receptor ACKR1 Determine Discrete Stages of Neutrophil Diapedesis. Immunity 2018, 49(6), 1062–1076.e6. [Google Scholar] [CrossRef] [PubMed]
- Idorn, M; Skadborg, SK; Kellermann, L; Halldórsdóttir, HR; Holmen Olofsson, G; Met, Ö; et al. Chemokine receptor engineering of T cells with CXCR2 improves homing towards subcutaneous human melanomas in xenograft mouse model. OncoImmunology 2018, 7(8), e1450715. [Google Scholar] [CrossRef]
- Whilding, LM; Halim, L; Draper, B; Parente-Pereira, AC; Zabinski, T; Davies, DM; et al. CAR T-Cells Targeting the Integrin αvβ6 and Co-Expressing the Chemokine Receptor CXCR2 Demonstrate Enhanced Homing and Efficacy against Several Solid Malignancies. Cancers. (Basel) 2019, 11(5), 674. [Google Scholar] [CrossRef]
- Gu, X; Li, D; Wu, P; Zhang, C; Cui, X; Shang, D; et al. Revisiting the CXCL13/CXCR5 axis in the tumor microenvironment in the era of single-cell omics: Implications for immunotherapy. Cancer Lett. 2024, 605, 217278. [Google Scholar] [CrossRef]
- Han, D; Jeong, B-K; Hong, JM; Seo, J-H; Lee, G; Kim, K; et al. Optimal chemokine receptors for enhancing immune cell trafficking in adoptive cell therapy. Immunol Res. 2025, 73(1), 36. [Google Scholar] [CrossRef]
- Hui, X; Farooq, MA; Chen, Y; Ajmal, I; Ren, Y; Xue, M; et al. A novel strategy of co-expressing CXCR5 and IL-7 enhances CAR-T cell effectiveness in osteosarcoma. Front Immunol. 2024, 15, 1462076. [Google Scholar] [CrossRef]
- Li, G; Guo, J; Zheng, Y; Ding, W; Han, Z; Qin, L; et al. CXCR5 guides migration and tumor eradication of anti-EGFR chimeric antigen receptor T cells. Mol Ther Oncolytics 2021, 22, 507–17. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A; Dagur, PK; Biancotto, A. Multiparametric Flow Cytometry Analysis of Naïve, Memory, and Effector T Cells. Methods Mol Biol. 2019, 2032, 129–40. [Google Scholar] [CrossRef] [PubMed]
- Goto, S; Sakoda, Y; Adachi, K; Sekido, Y; Yano, S; Eto, M; et al. Enhanced anti-tumor efficacy of IL-7/CCL19-producing human CAR-T cells in orthotopic and patient-derived xenograft tumor models. Cancer Immunol Immunother. 2021, 70(9), 2503–15. [Google Scholar] [CrossRef]
- Hu, J; Wang, Z; Liao, C; Chen, Z; Kang, F; Lin, C; et al. Induced expression of CCL19 promotes the anti-tumor ability of CAR-T cells by increasing their infiltration ability. Front Immunol. 2022, 13, 958960. [Google Scholar] [CrossRef]
- Ohta, K; Sakoda, Y; Adachi, K; Shinozaki, T; Nakajima, M; Yasuda, H; et al. Therapeutic Efficacy of IL7/CCL19-Expressing CAR-T Cells in Intractable Solid Tumor Models of Glioblastoma and Pancreatic Cancer. Cancer Res Commun. 2024, 4(9), 2514–24. [Google Scholar] [CrossRef]
- Qian, W; Liu, H; Lei, W; Wang, L; Chen, P. Long-Term Efficacy and Safety of CD19-Specific CAR-T Cells Armed with IL-7 and CCL19 in Patients with Relapsed or Refractory Large B-Cell Lymphoma: A Multi-Center Prospective Study. Blood 2024, 144, 2087. [Google Scholar] [CrossRef]
- Bhutani, B; Sharma, V; Ganguly, NK; Rana, R. Unravelling the modified T cell receptor through Gen-Next CAR T cell therapy in Glioblastoma: Current status and future challenges. Biomed Pharmacother. 2025, 186, 117987. [Google Scholar] [CrossRef] [PubMed]
- Chen, W; Hong, L; Lin, S; Xian, N; Yan, C; Zhao, N; et al. Enhanced anti−tumor efficacy of “IL−15 and CCL19” −secreting CAR−T cells in human glioblastoma orthotopic xenograft model. Front Oncol. 2025, 15, 1539055. [Google Scholar] [CrossRef]
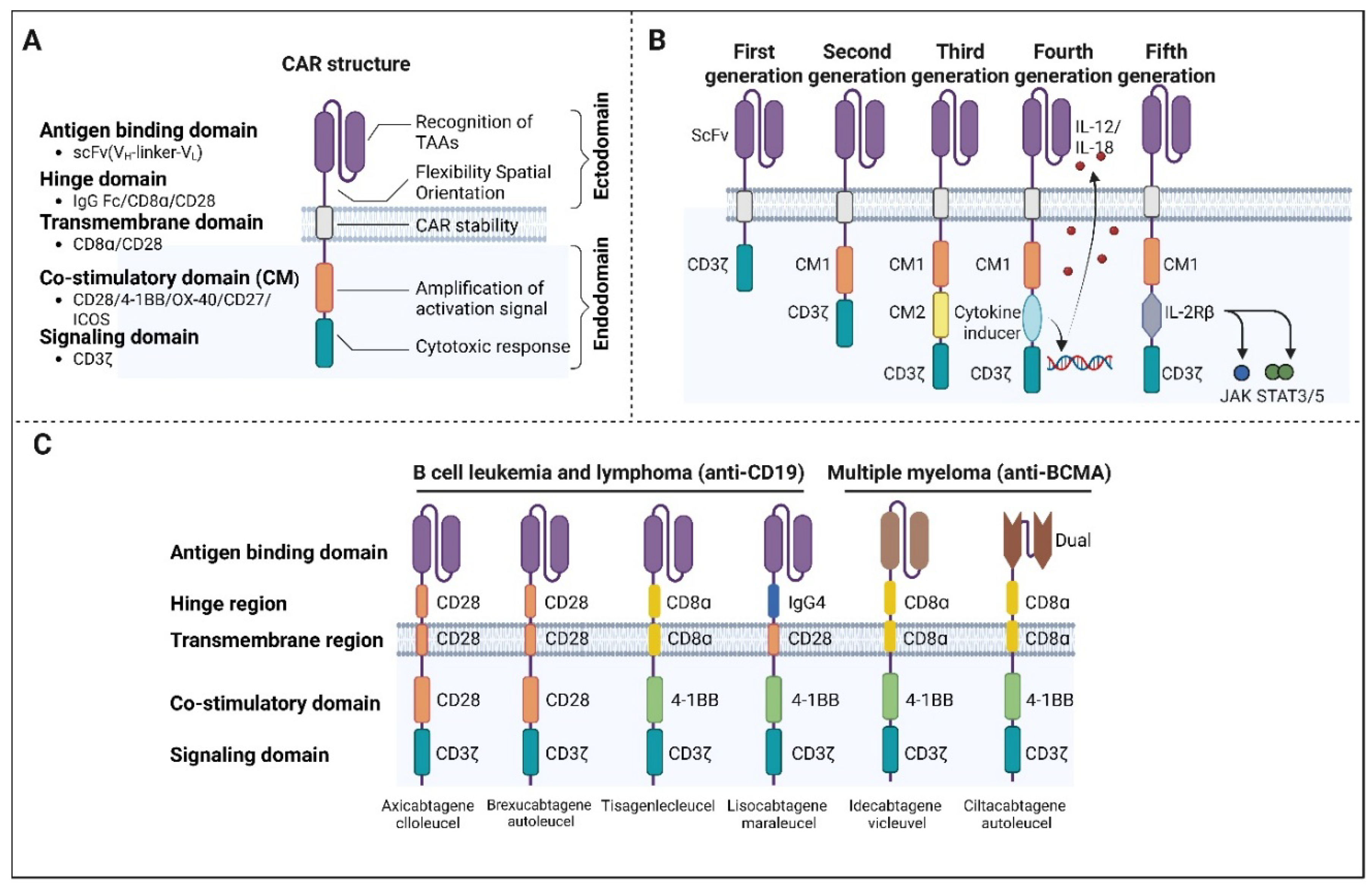
| CAR Generation | Key Structural Components | Primary Signaling Domains | Functional Advantages | Major Limitations | Clinical Status |
| 1st generation | scFv + hinge/spacer + transmembrane domain | CD3ζ | Antigen-specific T cell activation | Poor proliferation and persistence; limited cytokine production; weak antitumor efficacy | Early clinical trials; no FDA-approved products |
| 2nd generation | scFv + hinge/spacer + transmembrane domain | CD3ζ + one co-stimulatory CD28 or 4-1BB | Enhanced activation, expansion, cytokine secretion, and in vivo persistence | Risk of CRS and neurotoxicity; limited efficacy in solid tumors | FDA-approved products for B-ALL, DLBCL, MM |
| 3rd generation | scFv + hinge/spacer + transmembrane domain | CD3ζ + two co-stimulatory domains (e.g., CD28 + 4-1BB) | Strong signaling; improved resistance to exhaustion in preclinical models | No consistent clinical superiority over the second generation; increased signaling complexity | Clinical trials ongoing; limited adoption |
| 4th generation | scFv + hinge/spacer + transmembrane domain + inducible cytokine cassette | CD3ζ + co-stimulatory molecule + cytokine gene (e.g. IL-12) | Local cytokine release (e.g., IL-12) remodels TME; recruits innate immunity | Risk of systemic inflammation; safety concerns | Primarily preclinical and early clinical development |
| 5th generation | scFv + hinge/spacer + transmembrane domain + cytokine receptor motifs | CD3ζ + co-stimulatory molecule + JAK/STAT signaling domain | Mimics physiological T cell activation; enhanced persistence and antitumor activity | Early-stage development; safety and durability under evaluation | Preclinical and early-phase clinical studies |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




