Submitted:
13 February 2026
Posted:
13 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Reagents and Chemicals
2.2. Preparation of C. paliurus Leaves Sample
2.3. Extraction C. paliurus Leaves Samples
2.4. Determination of Total Phenolic Content (TPC), Total Flavonoid Content (TFC), Total Polysaccharide Content (TP) and Total Saponin Content (TSC)
2.5. Antioxidant Evaluation of C. paliurus Leaves Extracts (DPPH Test)
2.6. Antiglycative Evaluation of C. paliurus leaves Extracts in BSA Models
2.7. Gas Chromatography Mass Spectrometry (GC-MS) Analysis
2.8. Statistical Analysis
3. Results
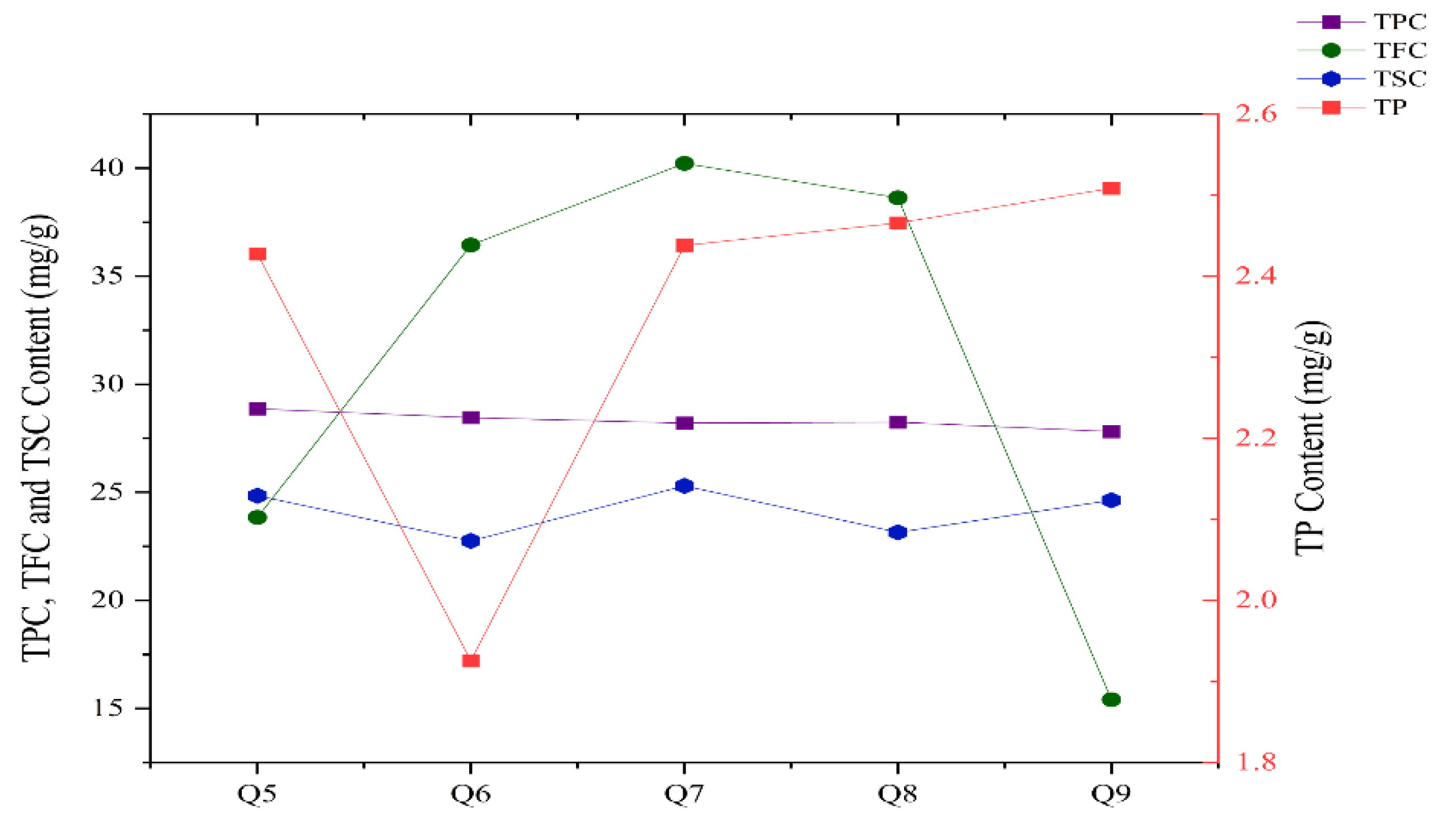
3.1. Changes in TPC, TFC, TP and TSC of C. Paliurus Leaves in Different Growth Periods
3.2. Changes of Antioxidant Activity
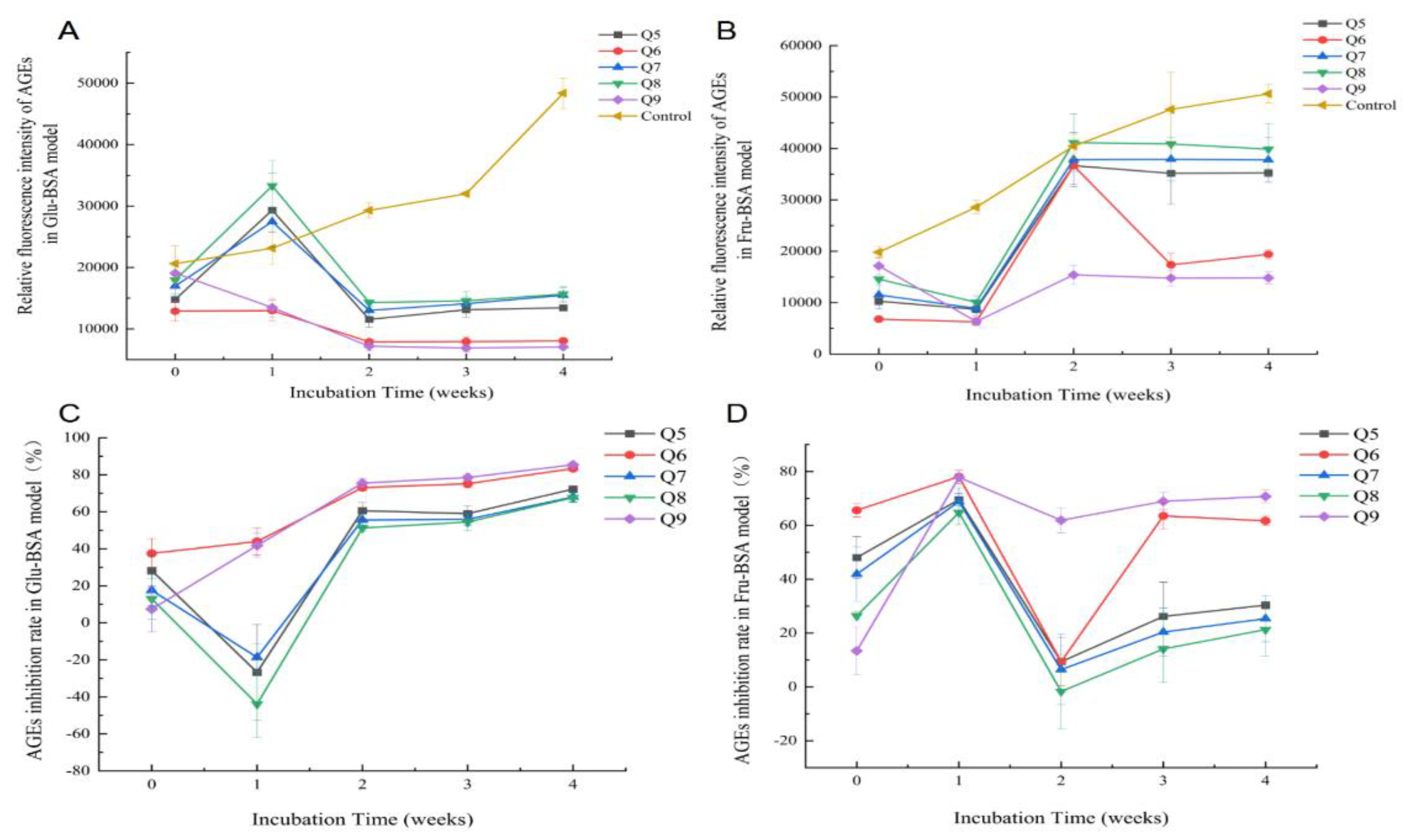
3.3. Changes of Anti-Glycation Capacity
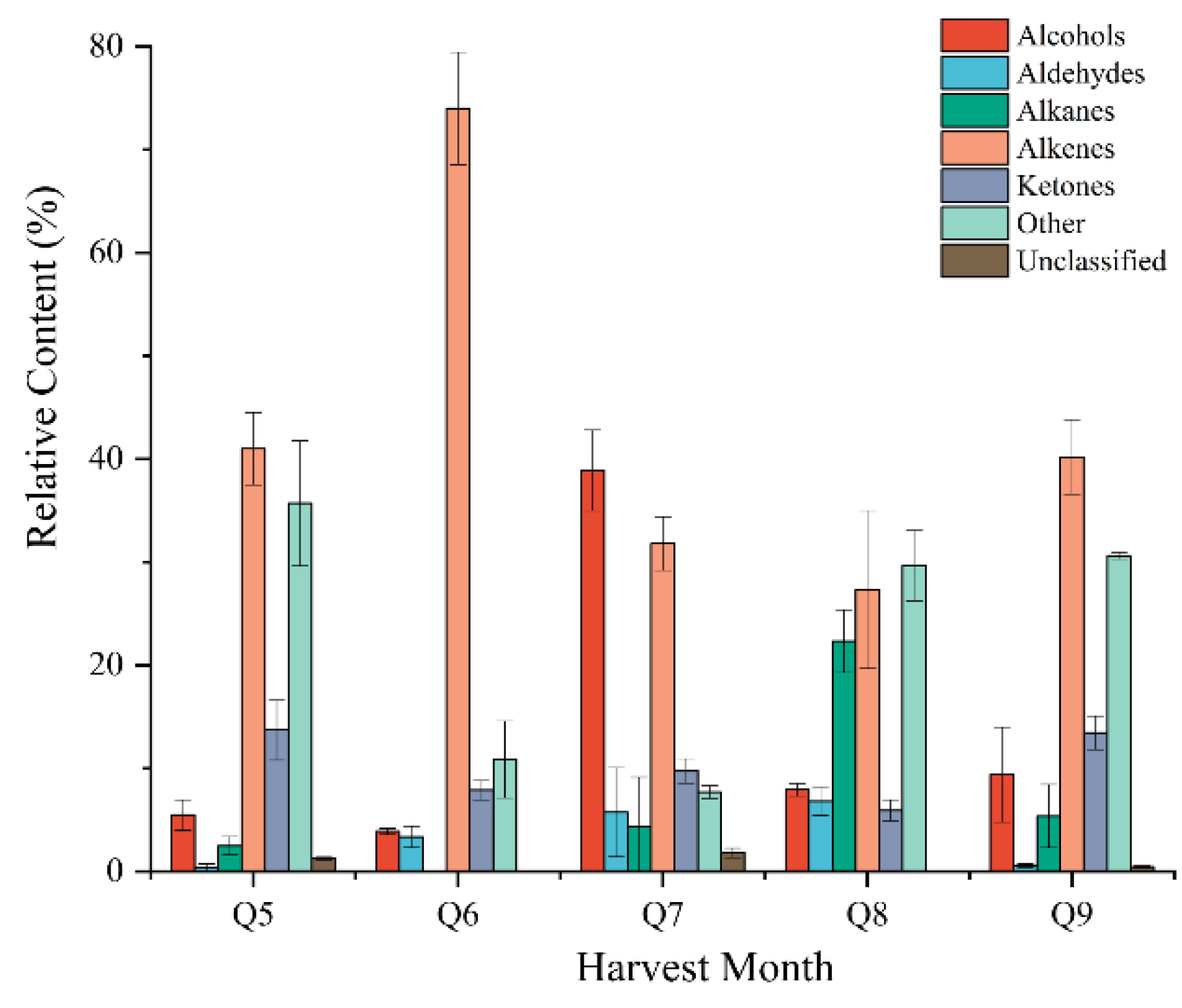
3.4. Analysis of VOCs by GC-MS
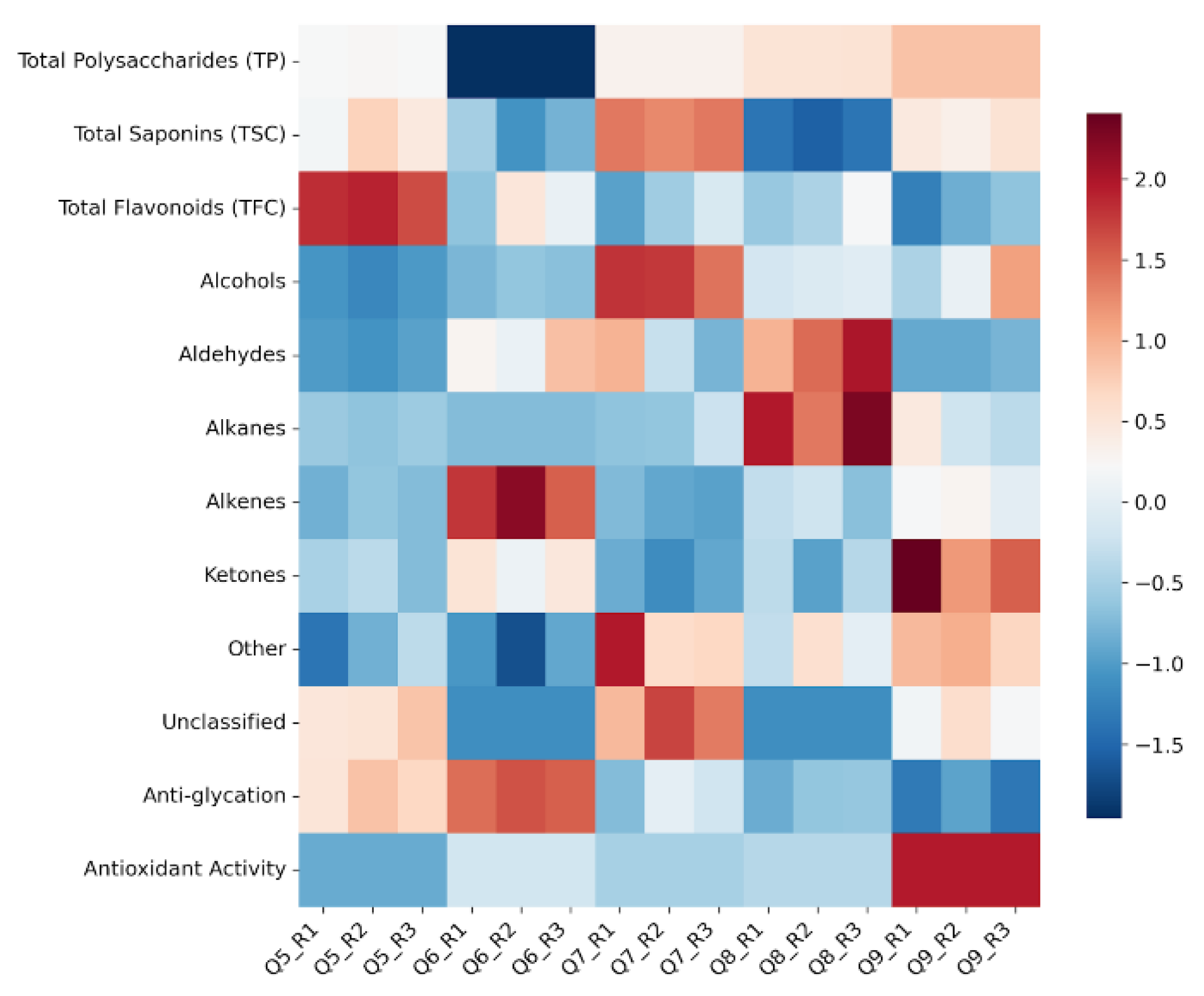
3.5. Correlation Analysis Between Volatile Organic Compounds and Antioxidant and Anti-Glycation Activities of C. paliurus Leaves Collected in Different Months
3.6. Characterization of Key Aroma-Active Compounds and Sensory Profiles
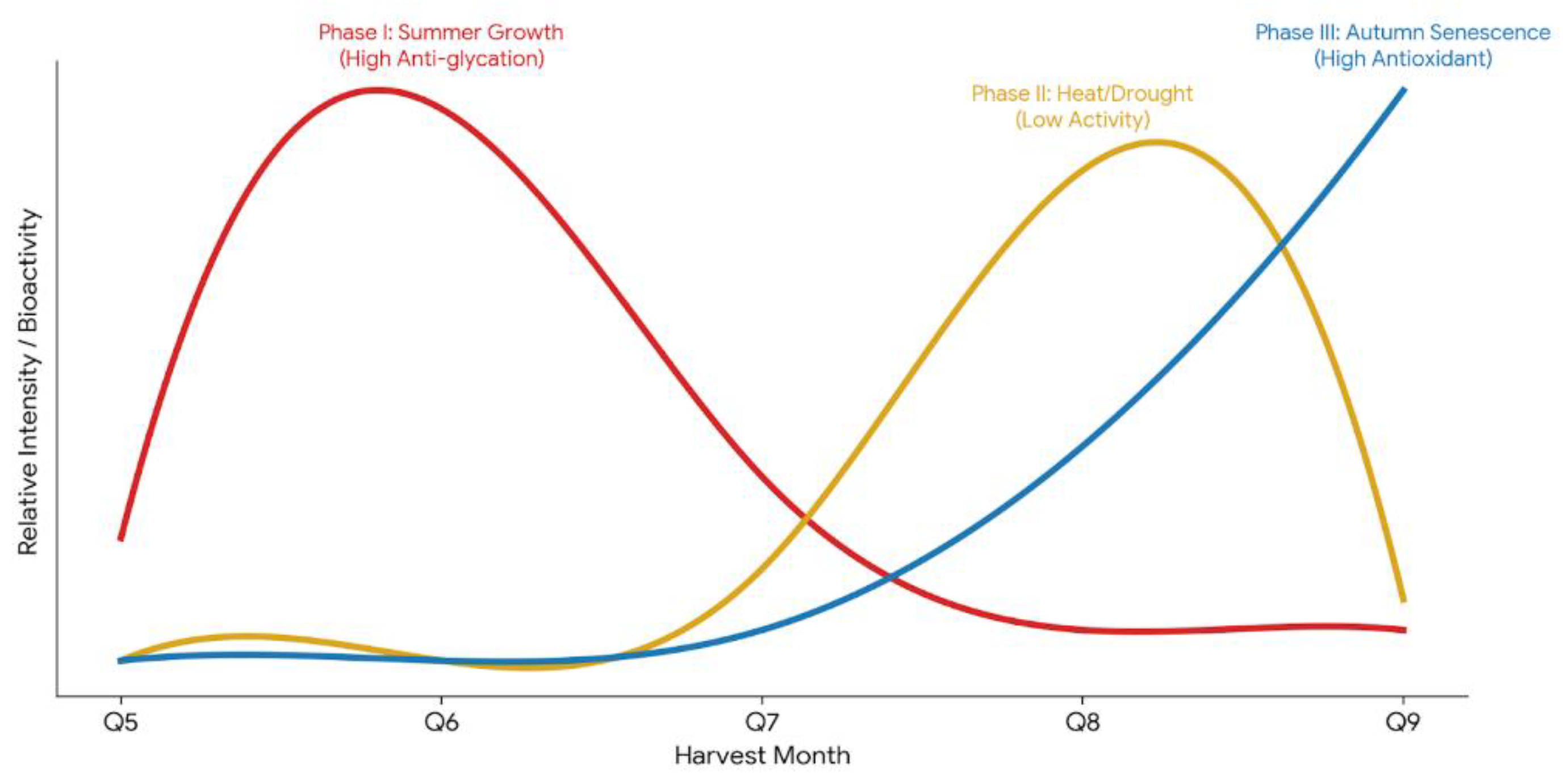
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chen, L.; Lu, D.; Wan, Y.; Zou, Y.; Zhang, R.; Zhou, T.; Long, B.; Zhu, K.; Wang, W.; Tian, X. Metabolite Profiling and Identification of Sweet/Bitter Taste Compounds in the Growth of Cyclocarya Paliurus Leaves Using Multiplatform Metabolomics. Foods 2024, 13, 3089. [Google Scholar] [CrossRef]
- Fang, S.; Sun, D.; Shang, X.; Fu, X.; Yang, W. Variation in radial growth and wood density of Cyclocarya paliurus across its natural distribution. New Forests 2020, 51, 453–467. [Google Scholar] [CrossRef]
- Xie, J.; Wang, Z.; Shen, M.; Nie, S.; Gong, B.; Li, H.; Zhao, Q.; Li, W.; Xie, M. Sulfated modification, characterization and antioxidant activities of polysaccharide from Cyclocarya paliurus. Food Hydrocolloids: A Key to Human Health Food Hydrocolloids 2016, 53, 7–15. [Google Scholar] [CrossRef]
- Wang, J.; Wang, K. Fatigue-alleviating effect of polysaccharides from Cyclocarya paliurus (Batal) Iljinskaja in mice. Afr J Microbiol Res. 2012, 6, 5243–5248. [Google Scholar]
- Wu, Z.; Gao, T.; Zhong, R.; Lin, Z.; Jiang, C.; Ouyang, S.; Zhao, M.; Che, C.; Zhang, J.; Yin, Z. Antihyperlipidaemic effect of triterpenic acid-enriched fraction from Cyclocarya paliurus leaves in hyperlipidaemic rats. Pharm Biol. 2017, 55, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Peng, Y.; Zhu, X.; Li, H.; Zhang, L.; Kong, F.; Wang, J.; Yu, D. The phytochemicals and health benefits of Cyclocarya paliurus (Batalin) Iljinskaja. Front. Nutr. 2023, 10, 1158158. [Google Scholar] [CrossRef]
- Zhang, J.; Shen, Q.; Lu, J.; Li, J.; Liu, W.; Yang, J.; Li, J.; Xiao, K. Phenolic compounds from the leaves of Cyclocarya paliurus (Batal.) Ijinskaja and their inhibitory activity against PTP1B. Food Chem. 2010, 119, 1491–1496. [Google Scholar] [CrossRef]
- Wang, Y.; Ma, J.; Tong, Y.; Li, N.; Li, J.; Qi, Z. Antidiabetic effects and mechanisms of Cyclocarya paliurus leaf flavonoids via PIK3CA. J. Funct. Foods. 2024, 113, 106031. [Google Scholar] [CrossRef]
- Zhou, M.; Lin, Y.; Fang, S.; Liu, Y.; Shang, X. Phytochemical content and antioxidant activity in aqueous extracts of Cyclocarya paliurus leaves collected from different populations. PeerJ. 2019, 7, e6492. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Wang, Z.; Xia, Q.; Chen, J.; Lv, Q.; Zhang, S.; Cheng, S.; Chen, X.; Dong, X. X. Preparation, Structural Characterization and Biological Activity Study of Selenium-Rich Polysaccharides from Cyclocarya paliurus. Foods 2025, 14, 1641. [Google Scholar] [CrossRef]
- Wang, Y.; Zheng, X.; Li, L.; Wang, H.; Wu, H.; Chen, K.; Xu, M.; Wu, Y.; Huang, X. Cyclocarya paliurus ethanol leaf extracts protect against diabetic cardiomyopathy in db/db mice via regulating PI3K/Akt/NF-κB signaling. Food Nutr. Res. 2020, 64, 4267. [Google Scholar] [CrossRef]
- Fang, S.; Liu, J.; Wu, C.; Zhang, X.; Dong, H.; Zhang, D.; Hu, C.; Zhang, J.; Pan, K.; Yin, Z.; Qin, Y.; Wang, L. Terpene and lignan glycosides from the leaves of Cyclocarya paliurus and their anti-inflammatory activity. Phytochemistry 2025, 234, 114443. [Google Scholar] [CrossRef]
- Feng, Z.; Fang, Z.; Chen, C.; Vong, C.T.; Chen, J.; Lou, R.; Hoi, M.P.M.; Gan, L.; Lin, L. Anti-hyperglycemic effects of refined fractions from Cyclocarya paliurus leaves on streptozotocin-induced diabetic mice. Molecules 2021, 26, 6886. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Wen, B.; Ning, Z.; Zhai, L.; Liao, C.; Lin, C.; Mu, H.; Bian, Z. Cyclocarya paliurus tea leaves enhances pancreatic β cell preservation through inhibition of apoptosis. Sci. Rep. 2017, 100, 51–64. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, Y.; Liu, G.; Deng, Y.; Wang, A.; Ren, F. Effect of Cyclocarya paliurus on hypoglycemic effect in type 2 diabetic mice. Med. Sci. Monit. 2019, 25, 2976–2983. [Google Scholar] [CrossRef]
- Liu, Y.; Cao, Y.; Fang, S.; Wang, T.; Yin, Z.; Shang, X.; Fu, X. Antidiabetic effect of Cyclocarya paliurus leaves depends on the contents of antihyperglycemic flavonoids and antihyperlipidemic triterpenoids. Molecules 2018, 23, 1420. [Google Scholar] [CrossRef]
- Wang, X.; Li, W.; Kong, D. Cyclocarya paliurus extract alleviates diabetic nephropathy by inhibiting oxidative stress and aldose reductase. Ren. Fail. 2016, 38, 678–685. [Google Scholar] [CrossRef]
- Liang, L.; Zhang, X.; Zhou, Y.; Liu, Y.; Gan, S.; Liu, Y.; Lu, Y.; Lam, H.; Pi, E. Untargeted metabolomics analysis based on HS-SPME-GC-MS and UPLC-Q-TOF/MS reveals the contribution of stem to the flavor of Cyclocarya paliurus herbal extract. LWT 2022, 163, 113549. [Google Scholar] [CrossRef]
- Lou, J.; Zou, Y.; Tian, X. Characterization and analysis of the volatile components of Cyclocarya paliurus tea from different origins based on GC-IMS technology. Food Mach. (In Chinese). 2024, 40, 161–167. [Google Scholar] [CrossRef]
- Chen, W.; Zhong, P.; Wang, Y. Analysis of volatile components in Cyclocarya paliurus leaves by solid-phase microextraction coupled with gas chromatography-mass spectrometry. Food Sci. (In Chinese). 2016, 37, 136–140. [Google Scholar] [CrossRef]
- Zheng, X.; Zhang, M.; Qiao, Y.; Li, R.; Alkan, N.; Chen, J.; Chen, F. Cyclocarya paliurus Reprograms the Flavonoid Biosynthesis Pathway Against Colletotrichum fructicola. Front. Plant Sci. 2022, 13, 933484. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Y.; He, C.; Chen, Y.; Ho, C.; Wu, X.; Huang, Y.; Gao, Y.; Hou, A.; Li, Z.; Wang, Y.; et al. UPLC–QQQ–MS/MS-based widely targeted metabolomic analysis reveals the effect of solid-state fermentation with Eurotium cristatum on the dynamic changes in the metabolite profile of dark tea. Food Chem. 2022, 378, 131999. [Google Scholar] [CrossRef]
- Yang, J.; Guo, J.; Yuan, J. In vitro antioxidant properties of rutin. LWT-Food Sci. Technol. 2008, 41, 1060–1066. [Google Scholar] [CrossRef]
- Wu, L.; Gao, Y.; Ren, W.; Su, Y.; Li, J.; Du, Y.; Wang, Q.; Kuang, H. Rapid determination and origin identification of total polysaccharides contents in Schisandra chinensis by near-infrared spectroscopy. Spectrochim Acta A Mol Biomol Spectrosc. 2022, 264, 120327. [Google Scholar] [CrossRef]
- Biswas, T.; Dwivedi, U.N. Plant triterpenoid saponins: biosynthesis, in vitro production, and pharmacological relevance. Protoplasma 2019, 25, 1463–1486. [Google Scholar] [CrossRef] [PubMed]
- Svečnjak, L.; Marijanović, Z.; Okińczyc, P.; Marek Kuś, P.; Jerković, I. Mediterranean Propolis from the Adriatic Sea Islands as a Source of Natural Antioxidants: Comprehensive Chemical Biodiversity Determined by GC-MS, FTIR-ATR, UHPLC-DAD-QqTOF-MS, DPPH and FRAP Assay. Antioxidants 2020, 9, 337. [Google Scholar] [CrossRef]
- Hasan, M.; Haque, M.; Hoque, M.; et al. Antioxidant activity study and GC-MS profiling of Camellia sinensis Linn. Heliyon 2024, 10, e23514. [Google Scholar] [CrossRef]
- Zhou, Q.; Liang, W.; Wan, J.; Wang, M. Spinach (Spinacia oleracea) microgreen prevents the formation of advanced glycation end products in model systems and breads. Curr. Res. Food Sci. 2023, 6, 100490. [Google Scholar] [CrossRef]
- Fu, Y.; Zou, Y.; Tian, X. Analysis of flavor characteristics and bioactive components of Cyclocarya paliurus tea from different origins based on electronic tongue and HS-SPME-GC-MS technology. Sci. Technol. Food Ind. (In Chinese). 2025, 1–21. [Google Scholar] [CrossRef]
- Cao, Y.; Fang, S.; Fu, X.; Shang, X.; Yang, W. Seasonal variation in phenolic compounds and antioxidant activity in leaves of Cyclocarya paliurus (Batal.) Iljinskaja. Forests 2019, 10, 624. [Google Scholar] [CrossRef]
- Zheng, X.; Xiao, H.; Chen, J.; Zhu, J.; Fu, Y.; Ouyang, S.; Chen, Y.; Chen, D.; Su, J.; Xue, T. Metabolome and whole-transcriptome analyses reveal the molecular mechanisms underlying hypoglycemic nutrient metabolites biosynthesis in Cyclocarya paliurus leaves during different harvest stages. Front. Nutr. 2022, 9, 851569. [Google Scholar] [CrossRef]
- Fu, X.; Zhou, X.; Deng, B.; Liu, F.; Shang, X.; Fang, S. Seasonal and genotypic variation of water-soluble polysaccharide content in leaves of Cyclocarya paliurus. South. Forests 2015, 77, 231–275. [Google Scholar] [CrossRef]
- Fang, S.; Yang, W.; Chu, X.; Shang, X.; She, C.; Fu, X. Provenance and temporal variations in selected flavonoids in leaves of Cyclocarya paliurus. Food Chem. 2011, 124, 1382–1386. [Google Scholar] [CrossRef]
- Sheng, X.; Chen, H.; Wang, J.; Zheng, Y.; Li, Y.; Jin, Z. Joint Transcriptomic and Metabolic Analysis of Flavonoids in Cyclocarya paliurus Leaves. ACS Omega 2021, 6, 8272–8282. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Li, Y.; Lu, Q.; Lu, H.; Li, J. Combined Analysis of the Metabolome and Transcriptome Identified Candidate Genes Involved in Phenolic Acid Biosynthesis in the Leaves of Cyclocarya paliurus. Int. J. Mol. Sci. 2020, 21, 1337. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Chen, H.; Wang, J.; Zheng, Y.; Lu, Q.; Zhu, Z.; Li, N.; Jin, Z.; Li, J.; Lu, H. Transcriptome analysis associated with polysaccharide synthesis and their antioxidant activity in Cyclocarya paliurus leaves of different developmental stages. PeerJ. 2021, 9, e11615. [Google Scholar] [CrossRef]
- Xia, X.; Xue, S.; Song, G.; Li, B.; Wang, H.; Qiu, J.; Wang, J.; Liu, Q.; Ma, Y.; Ouyang, J. Antioxidative and immunological role of Cyclocarya paliurus polysaccharide in diabetic rats. J. Tradit. Chin. Med. 2021, 41(6), 739–746. [Google Scholar] [CrossRef]
- Cao, Y.; Fang, S.; Yin, Z.; Fu, X.; Shang, X.; Yang, W.; Yang, H. Chemical fingerprint and multicomponent quantitative analysis for the quality evaluation of Cyclocarya paliurus leaves by HPLC-Q-TOF-MS. Molecules 2017, 22, 1927. [Google Scholar] [CrossRef]
- Bai, M.; Wang, L. Untargeted metabolomics analysis reveals the effect of fixation on the profile of volatile compounds of Cyclocarya paliurus tea. Pol. J. Food Nutr. Sci. 2022, 74, 333–345. [Google Scholar] [CrossRef]
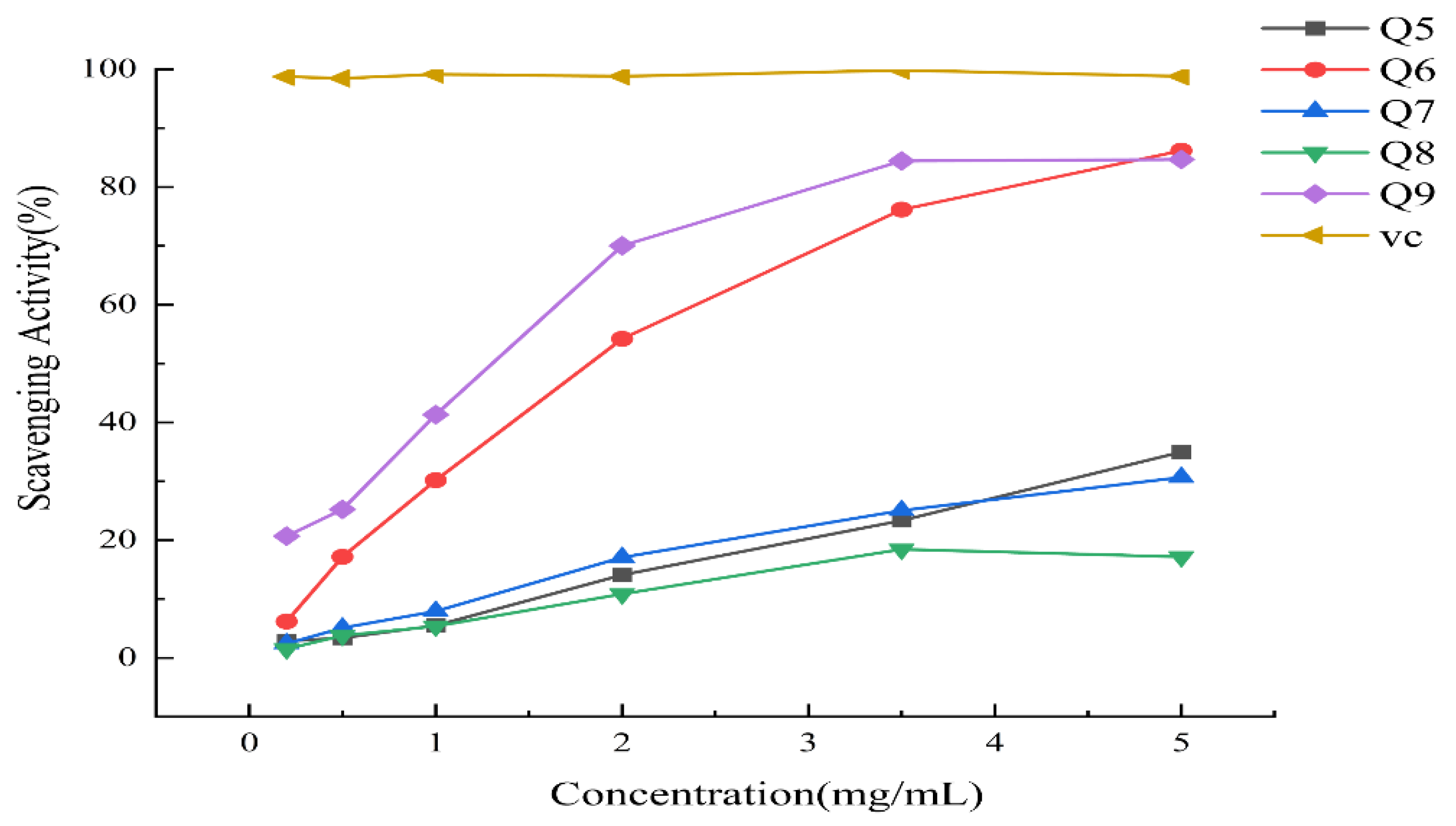
| Month | IC50( DPPH·) |
|---|---|
| Q5 | 0.297 |
| Q6 | 0.222 |
| Q7 | 0.252 |
| Q8 | 0.242 |
| Q9 | 0.119 |
| class | Compound Name | CAS No. | Q5 | Q6 | Q7 | Q8 | Q9 | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Content (%) | RT (min) | Content (%) | RT (min) | Content (%) | RT (min) | Content (%) | RT (min) | Content (%) | RT (min) | |||
| Alkanes | Cyclohexene, 4-bromo- | 3540-84-9 | - | - | 1.74 | 15.86 | - | - | 1.71 | 15.83 | 0.97 | 15.83 |
| 1-Chloroheptane | 629-06-1 | - | - | - | - | 0.39 | 14.42 | 0.32 | 14.38 | - | - | |
| 1-Chlorohexane | 544-10-5 | - | - | - | - | 1.31 | 8.97 | - | - | 0.46 | 8.97 | |
| Tridecane | 629-50-5 | - | - | - | - | - | - | 0.37 | 40.94 | 0.18 | 40.93 | |
| trans-Sabinene hydrate | 17699-16-0 | - | - | 2.52 | 22.75 | - | - | - | - | - | - | |
| (1S,2R,5S)-2,6,6-trimethylbicyclo [3.1.1]heptane | 6876-13-7 | - | - | - | - | 0.98 | 17.15 | - | - | - | - | |
| 1-Methyl-2-(1-methylpentyl)cyclopropane | 62238-06-6 | - | - | 0.38 | 14.43 | - | - | - | - | - | - | |
| 1-methylene-4-(1-methylvinyl)cyclohexane | 499-97-8 | - | - | - | - | - | - | - | - | 0.50 | 17.73 | |
| 2-methylbutan-2-ylcyclohexane | 31797-64-5 | - | - | 0.90 | 27.26 | - | - | - | - | - | - | |
| 3,9-dimethylundecane | 17301-31-4 | - | - | - | - | 0.45 | 36.25 | - | - | - | - | |
| 3-CHLOROOCTANE | 1117-79-9 | - | - | 4.75 | 18.01 | - | - | - | - | - | - | |
| 3-Methylundecane | 1002-43-3 | - | - | - | - | - | - | - | - | 0.27 | 31.29 | |
| 4-BROMO-1-CYCLOHEXENE | 3540-84-9 | 1.32 | 15.76 | - | - | - | - | - | - | - | - | |
| 4-bromocyclohexene | - | - | - | - | 2.17 | 15.80 | - | - | - | - | ||
| Bicyclo[3.1.1]heptane, 2,6,6-trimethyl-, (1R,2R,5R)-rel- | 10281-53-5 | - | - | - | - | - | - | - | - | 0.39 | 17.18 | |
| Bicyclo[3.2.0]heptane,2-ethenyl-2,6,6-trimethyl-1-(1-methylethenyl)-,(1R,2S,5S)-rel- | 871660-95-6 | - | - | - | - | - | - | - | - | 15.82 | 40.66 | |
| Bromo-1-chloro | - | - | - | - | - | - | - | - | 10.77 | 5.09 | ||
| Cyclodecane | 293-96-9 | - | - | - | - | 1.11 | 29.00 | - | - | - | - | |
| Cyclooctane, 1,4-dimethyl-, cis- | 10565-37-4 | - | - | - | - | - | - | 3.65 | 20.73 | - | - | |
| α-Isocomene | 65372-78-3 | - | - | - | - | 1.02 | 43.83 | 16.04 | 43.85 | - | - | |
| 2-Chlorooctane | 628-61-5 | 1.46 | 17.91 | - | - | - | - | - | - | - | - | |
| 4-Chlorooctane | 999-97-3 | - | - | - | - | 7.19 | 17.51 | - | - | - | - | |
| Octylcyclopropane | 1472-12-4 | - | - | - | - | 0.98 | 24.53 | - | - | - | - | |
| Alkenes | α-Cubebene | 17699-14-8 | 0.96 | 43.45 | 1.38 | 43.45 | 1.04 | 43.49 | 6.18 | 43.52 | - | - |
| 3-Bromocyclohexene | 1521-51-3 | 1.61 | 16.62 | 3.67 | 16.73 | 2.04 | 16.68 | 3.07 | 16.68 | - | - | |
| α-ylangene | 14912-44-8 | 45.35 | 44.79 | 0.92 | 44.77 | 1.88 | 44.77 | - | - | - | - | |
| Cyclosativene | 22469-52-9 | - | - | 0.39 | 44.59 | - | - | - | - | 3.41 | 44.02 | |
| (Z)-2-Undecene | 764-85-2 | - | - | 1.19 | 25.71 | 2.30 | 25.67 | - | - | - | - | |
| Hydrocarbon C12 | - | - | 0.98 | 26.87 | 3.00 | 25.92 | - | - | - | - | ||
| α-Longipinene | 5989-08-2 | - | - | 0.84 | 42.73 | 6.37 | 43.87 | - | - | - | - | |
| Menthofuran | 494-90-6 | - | - | - | - | 0.75 | 32.58 | 1.01 | 32.60 | - | - | |
| Panaxene | 871660-95-6 | - | - | - | - | 3.12 | 40.64 | 15.72 | 41.55 | - | - | |
| β-Elemene | 3388-04-3 | - | - | - | - | 0.99 | 45.82 | 1.21 | 45.82 | - | - | |
| Viridoflorene | 21747-46-6 | - | - | - | - | 2.36 | 46.63 | 0.55 | 46.63 | - | - | |
| β-Caryophyllene | 87-44-5 | 1.76 | 47.15 | - | - | 2.89 | 47.54 | - | - | - | - | |
| Longifolene | 475-20-7 | - | - | - | - | 3.08 | 46.36 | 3.42 | 46.36 | - | - | |
| β-Longipinene | 15301-20-4 | - | - | - | - | - | - | - | - | 0.67 | 46.34 | |
| α-Guaiene | 3691-12-1 | - | - | - | - | 12.93 | 47.97 | - | - | - | - | |
| Longicyclene | 1137-12-8 | - | - | - | - | - | - | - | - | 9.47 | 44.32 | |
| Alloaromadendrene | 25246-27-9 | - | - | 1.97 | 47.92 | - | - | - | - | - | - | |
| α-Calamenene | 483-77-2 | - | - | - | - | - | - | 2.30 | 44.77 | - | - | |
| Ascaridole | 512-85-6 | - | - | 0.33 | 37.84 | - | - | - | - | - | - | |
| trans-Carveol | 1197-07-5 | - | - | - | - | 3.04 | 35.77 | 0.83 | 35.78 | 0.22 | 35.75 | |
| 8-Methyl-1-decene | 13151-34-3 | - | - | - | - | 2.21 | 22.14 | - | - | - | - | |
| α-Terpinene | 99-86-5 | 1.22 | 19.10 | - | - | - | - | - | - | - | - | |
| 3-Carene | 13466-78-9 | - | - | - | - | 0.48 | 19.14 | - | - | - | - | |
| Viridoflorene | 21747-46-6 | - | - | - | - | - | - | - | - | 0.37 | 47.46 | |
| Longifolene | 475-20-7 | - | - | - | - | 1.22 | 45.58 | - | - | - | - | |
| Edulan I | 41678-25-5 | - | - | - | - | 2.05 | 41.40 | - | - | - | - | |
| Valencene | 4630-07-3 | - | - | - | - | - | - | - | - | 0.62 | 48.51 | |
| Rhodinol | 141-25-3 | - | - | - | - | - | - | - | - | 0.25 | 33.44 | |
| α-Copaene | 3856-25-5 | - | - | - | - | - | - | - | - | 2.30 | 44.76 | |
| α-Ylangene | 14912-44-8 | - | - | - | - | - | - | - | - | 0.17 | 45.01 | |
| β-Copaene | 18252-44-3 | - | - | - | - | - | - | - | - | 0.15 | 47.39 | |
| α-Patchoulene | 560-32-7 | - | - | - | - | - | - | 9.57 | 43.95 | - | - | |
| δ-Elemene | 20307-84-0 | - | - | - | - | - | - | 1.60 | 41.84 | - | - | |
| Alkynes | 1-Undecyne | 2243-98-3 | - | - | - | - | 4.86 | 23.08 | 9.19 | 23.10 | 2.71 | 23.06 |
| 2-Decyne | 2384-86-3 | 1.43 | 20.69 | - | - | - | - | - | - | - | - | |
| 2-Decyne | 2384-86-3 | - | - | 5.14 | 20.75 | - | - | - | - | - | - | |
| Aldehydes | (Z)-6-Nonenal | 2277-19-2 | - | - | - | - | 4.11 | 24.03 | - | - | - | - |
| 10-Undecenal | 112-45-8 | - | - | - | - | - | - | 0.34 | 39.69 | - | - | |
| (E)-2-Ethyl-2-butenal | 19780-25-7 | - | - | - | - | 4.35 | 8.26 | - | - | - | - | |
| 2-Ethylhexanal | 123-05-7 | - | - | - | - | 0.74 | 14.82 | - | - | - | - | |
| (E)-2-Hexenal | 6728-26-3 | - | - | - | - | - | - | - | - | 0.27 | 8.37 | |
| 2-Methyl-2-pentenal | 623-36-9 | - | - | 2.97 | 8.22 | - | - | - | - | - | - | |
| Hexanal | 66-25-1 | - | - | 1.09 | 6.38 | - | - | - | - | - | - | |
| (E)-2-Nonen-1-ol | 31502-14-4 | 0.97 | 31.76 | - | - | 1.48 | 31.33 | 1.37 | 31.79 | - | - | |
| Alcohols | trans-Isopiperitenol | 16721-39-4 | - | - | 0.95 | 34.11 | - | - | 0.44 | 34.12 | - | - |
| Ocimenol | 2270-40-8 | - | - | 3.12 | 30.24 | 1.54 | 30.24 | - | - | - | - | |
| trans-Carveol | 1197-07-5 | - | - | - | - | 3.04 | 35.77 | 0.83 | 35.78 | 0.22 | 35.75 | |
| (-)-Isopulegol | 89-79-2 | - | - | 3.32 | 29.06 | - | - | 1.65 | 29.04 | - | - | |
| cis-p-Mentha-2,8-dien-1-ol | 4017-02-1 | - | - | - | - | 1.84 | 23.65 | - | - | - | - | |
| 3-Thujanol | 546-79-2 | - | - | - | - | - | - | 0.56 | 27.80 | - | - | |
| cis-Sabinene hydrate | 15537-55-0 | 0.99 | 22.66 | - | - | - | - | - | - | - | - | |
| β-Terpineol | 138-87-4 | - | - | - | - | - | - | 5.74 | 30.36 | - | - | |
| cis-Carveol | 13429-07-7 | - | - | - | - | 2.20 | 34.10 | - | - | - | - | |
| 3,3,5-Trimethylcyclohexanol | 116-02-9 | - | - | 5.05 | 23.10 | - | - | - | - | - | - | |
| 3-Methyl-1-heptanol | 1470-24-2 | - | - | - | - | - | - | - | - | 1.05 | 20.74 | |
| 4-Methyl-1-pentanol | 626-89-1 | - | - | 2.67 | 8.93 | - | - | - | - | - | - | |
| (Z)-Ocimenol | 106064-70-0 | - | - | - | - | - | - | - | - | 4.34 | 30.23 | |
| Sulcatol | 1569-60-4 | - | - | 0.62 | 17.15 | - | - | - | - | - | - | |
| Neoisothujanol | 21653-20-3 | - | - | 0.35 | 31.35 | - | - | - | - | - | - | |
| 3-Thujanol | 546-79-2 | - | - | - | - | 0.86 | 30.91 | - | - | - | - | |
| trans-Verbenol | 1820-09-3 | - | - | - | - | - | - | 0.37 | 28.81 | - | - | |
| cis-Sabinene hydrate | 15537-55-0 | - | - | - | - | - | - | - | - | 0.52 | 22.72 | |
| (Z)-2-(3,3-Dimethylcyclohexylidene)ethanol | 55253-28-6 | - | - | - | - | 2.82 | 36.47 | - | - | - | - | |
| Isopulegol | 89-79-2 | - | - | - | - | - | - | - | - | 2.44 | 30.25 | |
| Sabinene hydrate | 546-87-2 | - | - | - | - | - | - | 1.03 | 22.74 | - | - | |
| trans-p-Mentha-2,8-dien-1-ol | 3886-78-0 | - | - | - | - | - | - | - | - | 0.26 | 26.84 | |
| 6-Methyl-6-hepten-2-one | 10408-15-8 | 2.11 | 15.22 | 5.98 | 15.30 | 3.40 | 15.28 | 1.73 | 15.33 | 2.23 | 15.33 | |
| Ketones | 2-Octyl trifluoroacetate | 13109-64-3 | - | - | 21.54 | 17.60 | - | - | - | - | - | - |
| Isolongifolene | 1135-66-6 | - | - | - | - | - | - | - | - | 1.88 | 42.37 | |
| Isobornyl acetate | 125-12-2 | - | - | - | - | 1.55 | 32.37 | - | - | - | - | |
| Dihydrojasmone | 1128-08-1 | - | - | 0.23 | 44.94 | - | - | - | - | - | - | |
| 2-Tridecanone | 593-08-8 | - | - | 0.29 | 51.08 | - | - | - | - | - | - | |
| Carbofuran phenol | 1563-38-8 | - | - | 0.40 | 39.42 | 1.89 | 39.41 | 1.32 | 39.42 | 0.82 | 39.42 | |
| Dill ether | 74410-10-9 | - | - | - | - | - | - | - | - | 0.50 | 32.57 | |
| Chloroacetyl chloride | 79-04-9 | - | - | - | - | - | - | - | - | 14.18 | 3.24 | |
| Others | Cyperene | 2387-78-2 | - | - | - | - | - | - | - | - | 0.15 | 45.16 |
| Piperitol acetate | 5258-29-7 | - | - | - | - | 0.99 | 41.83 | - | - | - | - | |
| Neoisothujyl acetate | 52673-82-4 | - | - | - | - | - | - | 4.43 | 38.60 | - | - | |
| Emylcamate | 78-28-4 | - | - | 0.59 | 24.56 | - | - | - | - | - | - | |
| Isopentylbenzene | 2049-94-7 | - | - | 0.42 | 23.75 | - | - | - | - | - | - | |
| 2,8-Dimethylquinoline | 1463-17-8 | - | - | - | - | - | - | - | - | 0.22 | 45.29 | |
| 2,5-Di-tert-butyl-1,4-benzoquinone | 2460-77-7 | - | - | - | - | - | - | - | - | 0.25 | 48.81 | |
| EdulanI | 41678-25-5 | - | - | - | - | - | - | - | - | 0.26 | 41.40 | |
| month | CAS | Compound | molecular formula | Odor description |
|---|---|---|---|---|
| Q5 | 2384-70-5 | 2-Decyne | C10H18 | Lemon, medicinal, citrus |
| 31502-14-4 | trans-2-Nonen-1-ol | C9H18O | Waxy, violet | |
| 74410-10-9 | Dill ether | C10H16O | Dill-like | |
| 17699-14-8 | α-Cubebene | C15H24 | Sweet, herbal, woody | |
| 118-65-0 | γ-Muurolene | C15H24 | Woody, spicy | |
| 87-44-5 | β-Caryophyllene | C15H24 | Sweet, woody, spicy, clove-like | |
| Q6 | 66-25-1 | Hexanal | C6H12O | Pungent |
| 623-36-9 | 2-Methyl-2-pentenal | C6H10O | Aldehydic, fruity, garlic-like, ripe cherry, earthy | |
| 626-89-1 | 4-Methyl-1-pentanol | C6H14O | Nutty | |
| 2384-70-5 | 2-Decyne | C10H18 | Lemon, medicinal, citrus | |
| 116-02-9 | 3,3,5-Trimethylcyclohexanol | C9H18O | Minty, musty, spicy | |
| 89-79-2 | (-)-Isopulegol | C10H18O | Minty, cooling, medicinal, woody | |
| 17699-14-8 | α-Cubebene | C15H24 | Sweet, herbal, woody | |
| 1128-08-1 | Dihydrojasmone | C11H18O | Jasmine, myrrh, woody | |
| Q7 | 13466-78-9 | 3-Carene | C10H16 | Citrus, terpenic, medicinal, woody |
| 31502-14-4 | trans-2-Nonen-1-ol | C9H18O | Waxy, violet | |
| 1197-07-5 | trans-Carveol | C10H16O | Spearmint | |
| 1197-06-4 | cis-Carveol | C10H16O | Caraway | |
| 17699-14-8 | α-Cubebene | C15H24 | Sweet, herbal, woody | |
| 515-13-9 | β-Elemene | C15H24 | Sweet | |
| 87-44-5 | β-Caryophyllene | C15H24 | Sweet, woody, spicy, clove-like | |
| 3691-12-1 | α-Guaiene | C15H24 | Sweet, woody, balsamic, peppery | |
| Q8 | 15537-55-0 | cis-Sabinene hydrate | C10H18O | Balsam-like |
| 89-79-2 | (-)-Isopulegol | C10H18O | Minty, cooling, medicinal, woody | |
| 31502-14-4 | trans-2-Nonen-1-ol | C9H18O | Waxy, violet | |
| 74410-10-9 | Dill ether | C10H16O | Dill-like | |
| 1197-06-4 | cis-Carveol | C10H16O | Caraway | |
| 20307-84-0 | δ-Elemene | C15H24 | Sweet, herbal, woody | |
| 17699-14-8 | α-Cubebene | C15H24 | Sweet, herbal, woody | |
| 515-13-9 | β-Elemene | C15H24 | Sweet | |
| 3691-12-1 | α-Guaiene | C15H24 | Sweet, woody, balsamic, peppery | |
| Q9 | 979-4-9 | Chloroacetyl chloride | C2H2Cl2O | Strong, pungent |
| 505-57-7 | 2-Hexenal | C6H10O | Green, leafy, fruity | |
| 3886-78-0 | trans-p-Mentha-2,8-dien-1-ol | C10H16O | Fresh, minty | |
| 89-79-2 | Isopulegol | C10H18O | Minty, cooling, medicinal, woody | |
| 1002-43-3 | 3-Methylundecane | C12H26 | Mild, waxy | |
| 74410-10-9 | Dill ether | C10H16O | Dill-like | |
| 6812-78-8 | Rhodinol | C10H20O | Floral | |
| 1197-06-4 | cis-Carveol | C10H16O | Caraway | |
| 20307-84-0 | δ-Elemene | C15H24 | Sweet, herbal, woody | |
| 17699-14-8 | α-Cubebene | C15H24 | Herbal | |
| 3856-25-5 | α-Copaene | C15H24 | Honey-like | |
| 515-13-9 | β-Elemene | C15H24 | Sweet |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




