Submitted:
11 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
| UPDRS III (score) | |||||
| Groups | n | Mean (SD) | Median (Min, Max) | p-value | |
| Overall | |||||
| Baseline | 16 | 38.5 (14.8) | 35.5 (9, 61) | ||
| Follow-up | 16 | 34 (11) | 36 (14, 52) | ||
| Change | 16 | -4.5 (8) | -5.5 (-18, 11) | 0.040 | |
| UPDRS | |||||
| Baseline | 1 | 9 | 9 | ||
| <25 | Follow-up | 1 | 14 | 14 | |
| Change | 1 | 5 | 5 | NA | |
| Baseline | 15 | 40.5 (12.9) | 38 (25, 61) | ||
| >25 | Follow-up | 15 | 35.3 (9.9) | 36 (18, 52) | |
| Change | 15 | -5.2 (7.8) | -7 (-18, 11) | 0.024 | |
| Age | |||||
| Baseline | 8 | 34.2 (15.3) | 31 (9, 56) | ||
| <80 | Follow-up | 8 | 30.6 (12) | 30 (14, 52) | |
| Change | 8 | -3.6 (8.3) | -6.5 (-12, 11) | 0.083 | |
| Baseline | 8 | 42.8 (13.9) | 41.5 (26, 61) | ||
| >80 | Follow-up | 8 | 37.4 (9.4) | 40.5 (18, 47) | |
| Change | 8 | -5.4 (8.1) | -5 (-18, 6) | 0.077 | |
| Dyskinesia/Dystonia/Freezing of gait/Fall | |||||
| Baseline | 8 | 40.1 (13.1) | 39 (26, 61) | ||
| No | Follow-up | 8 | 34.1 (10.6) | 36 (18, 47) | |
| Change | 8 | -6 (7.8) |
-7.5 (-18, 6) | 0.031 | |
| Baseline | 8 | 36.9 (17) | 35 (9, 58) | ||
| Yes | Follow-up | 8 | 33.9 (12.1) | 36 (14, 52) | |
| Change | 8 | -3.0 (8.4) | -3 (-14, 11) | 0.157 | |
| Length of diagnosis (years) | |||||
| Baseline | 5 | 39.6 (11.8) | 33 (30, 58) | ||
| <7 | Follow-up | 5 | 32.0 (9.9) | 33 (21, 44) | |
| Change | 5 | -7.6 (7.9) | -9 (-14, 11) | 0.098 |
|
| Baseline | 11 | 38 (16.4) | 38 (9, 61) | ||
| >7 | Follow-up | 11 | 34.9 (11.8) | 36 (14, 52) | |
| Change | 11 | -3.1 (8) | -3 (-18, 11) | 0.016 | |
| TUG (seconds) | |||||
| Groups | n | Mean (SD) |
Median (Min, Max) | p-value | |
| Overall | |||||
| Baseline | 15 | 13.8 (4.3) | 12.6 (7.5, 20.2) | ||
| Follow-up | 15 | 11.7 (3.1) | 11.8 (6.7, 16.4) | ||
| Change | 15 | -2.1 (2.2) | -1.7 (-7.8, 0.2) | 0.009 | |
| UPDRS | |||||
| Baseline | 1 | 7.5 | 7.5 | ||
| <25 | Follow-up | 1 | 7.4 | 7.4 | |
| Change | 1 | -0.1 | -0.1 | NA | |
| Baseline | 12 | 13.8 (4.2) | 12.2 (9.7, 20.2) | ||
| >25 | Follow-up | 12 | 11.6 | 11.5 (6.7, 16.4) | |
| Change | 12 | -2.2 | -1.6 (-7.8, 0.2) | 0.009 | |
| Age | |||||
| Baseline | 6 | 12.2 (4.6) | 10 (7.5, 19) | ||
| <80 | Follow-up | 6 | 9.7 (3) | 8.9 (6.7, 15) | |
| Change | 6 | -2.5 (2.8) | -1.6 (-7.8, -0.1) | 0.083 | |
| Baseline | 7 | 14.3 (4.3) | |||
| >80 | Follow-up | 7 | 12.6 (2.5) | ||
| Change | 7 | -1.7 (2.1) | |||
| Dyskinesia/Dystonia/Freezing of gait/Fall | |||||
| Baseline | 8 | 13.5 (4.4) | 11.4 (9.9, 20.2) | ||
| No | Follow-up | 8 | 11.7 (2.9) | 11.1 (8.7, 16.4) | |
| Change | 8 | -1.8 (1.9) | -1.1 (-4.4, 0.2) | 0.031 | |
| Baseline | 5 | 12.9 (4.8) | 11.8 (7.5, 19) | ||
| Yes | Follow-up | 5 | 10.4 (3.4) | 11.2 (6.7, 15) | |
| Change | 5 | -2.5 (3.2) | -1.7 (-7.8, 0.1) | 0.157 | |
| Length of diagnosis (years) | |||||
| Baseline | 4 | 12.3 (4.4) | 10.2 (9.9, 19) | ||
| <7 | Follow-up | 4 | 9.9 (1.1) | 9.8 (8.7, 11.2) | |
| Change | 4 | -2.4 (3.6) | -1.1 (-7.8, 0.1) | 0.267 | |
| Baseline | 9 | 13.7 (4.5) | 12.6 (7.5, 20.2) | ||
| >7 | Follow-up | 9 | 11.9 (3.5) | 11.9 (6.7, 16.4) | |
| Change | 9 | -1.8 (1.8) | -1.7 (-4.4, 0.2) | 0.016 | |
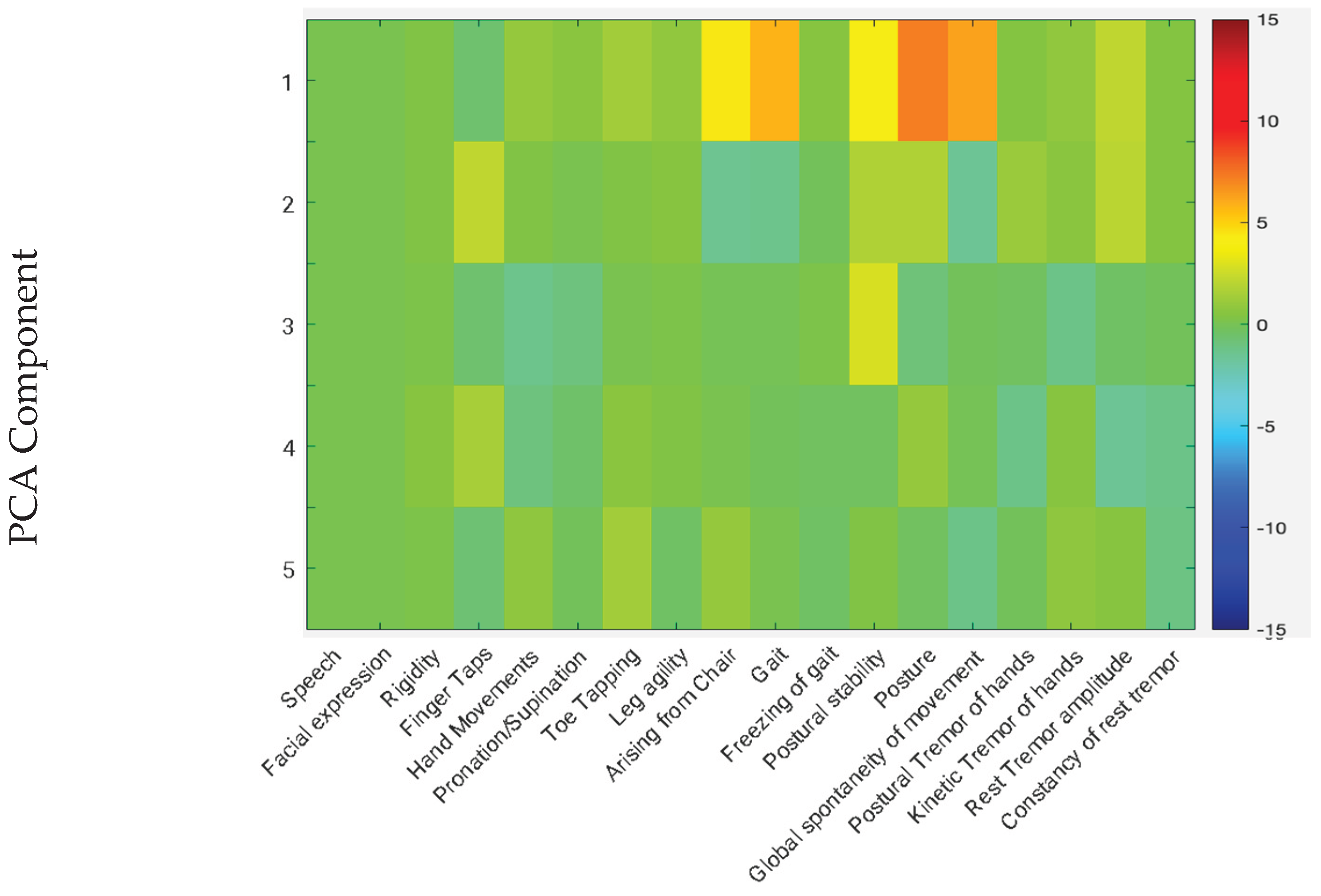
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PD | Parkinson’s Disease |
| MDS-UPDRS | Movement Disorder Society-Unified Parkinsons Disease Rating Scale |
| TUG | Timed-up-and-go |
References
- Tolosa, E. G. Challenges in the diagnosis of Parkinson's disease. The Lancet Neurology 2021, 20(5), 385–397. [Google Scholar] [CrossRef] [PubMed]
- Pringsheim, T; Jette, N; Frolkis, A; Steeves, TD. The prevalence of Parkinson's disease: a systematic review and meta-analysis. Movement disorders 2014, 29(13), 1583–90. [Google Scholar] [CrossRef] [PubMed]
- Thake, M.; Lowry, A. A systematic review of trends in the selective exclusion of older participant from randomised clinical trials. Arch. Gerontol. Geriatr. 2017, 72, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Crome, P.; Lally, F.; Cherubini, A.; Oristrell, J.; Beswick, A. D.; Clarfield, A. M.; Mills, G. Exclusion of older people from clinical trials: professional views from nine European countries participating in the PREDICT study. Drugs & aging 2011, 28(8), 667–677. [Google Scholar]
- Pirmohamed, M.; James, S.; Meakin, S.; Green, C.; Scott, A.K.; Walley, T.J.; Farrar, K.; Park, B.K.; Breckenridge, A.M. Adverse drug reactions as cause of admission to hospital: prospective analysis of 18 820 patients. BMJ 2004, 329, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Zahoor, I.; Shafi, A.; Haq, E. Pharmacological treatment of Parkinson’s disease; Exon Publications, 2018; pp. 129–144. [Google Scholar]
- Antonini, A.; Stoessl, A.J.; Kleinman, L.S.; Skalicky, A.M.; Marshall, T.S.; Sail, K.R.; Onuk, K.; Odin, P.L.A. Developing consensus among movement disorder specialists on clinical indicators for identification and management of advanced Parkinson’s disease: a multi-country Delphi-panel approach. Curr. Med Res. Opin. 2018, 34, 2063–2073. [Google Scholar] [CrossRef] [PubMed]
- Kapur, S. S. Vibration therapy for Parkinson's disease: Charcot's studies revisited. Journal of Parkinson's disease 2012, 2(1), 23–27. [Google Scholar] [CrossRef] [PubMed]
- Macerollo, A.; Palmer, C.; Foltynie, T.; Korlipara, P.; Limousin, P.; Edwards, M.; Kilner, J.M. High-frequency peripheral vibration decreases completion time on a number of motor tasks. Eur. J. Neurosci. 2018, 48, 1789–1802. [Google Scholar] [CrossRef] [PubMed]
- Cosentino, C.; Putzolu, M.; Mezzarobba, S.; Cecchella, M.; Innocenti, T.; Bonassi, G.; Botta, A.; Lagravinese, G.; Avanzino, L.; Pelosin, E. One cue does not fit all: A systematic review with meta-analysis of the effectiveness of cueing on freezing of gait in Parkinson’s disease. Neurosci. Biobehav. Rev. 2023, 150, 105189. [Google Scholar] [CrossRef] [PubMed]
- Ginis, P.; Nackaerts, E.; Nieuwboer, A.; Heremans, E. Cueing for people with Parkinson's disease with freezing of gait: A narrative review of the state-of-the-art and novel perspectives. Ann. Phys. Rehabilitation Med. 2018, 61, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Azoidou, V.; Rowsell, K.; Camboe, E.; Dey, K.C.; Zirra, A.; Quah, C.; Boyle, T.; Gallagher, D.; Noyce, A.J.; Simonet, C. A pilot interventional study on feasibility and effectiveness of the CUE1 device in Parkinson's disease. Park. Relat. Disord. 2025, 133, 107349. [Google Scholar] [CrossRef] [PubMed]
- Moes, H.R.; Henriksen, T.; Sławek, J.; Phokaewvarangkul, O.; Buskens, E.; van Laar, T. Tools and criteria to select patients with advanced Parkinson’s disease for device-aided therapies: a narrative review. J. Neural Transm. 2023, 130, 1359–1377. [Google Scholar] [CrossRef] [PubMed]
- Azoidou, V.; Bhadra, E.; Camboe, E.; Dey, K. C.; Zirra, A.; Quah, C.; Simonet, C. A Sternum-Worn, Non-Invasive Vibrotactile Device to Alleviate Symptoms in Parkinson’s: a Multi-Site Double-Blind Randomised Controlled Trial. medRxiv 2025, 2025–07. [Google Scholar]
- Horváth, K.; Aschermann, Z.; Ács, P.; Deli, G.; Janszky, J.; Komoly, S.; Balázs, É.; Takács, K.; Karádi, K.; Kovács, N. Minimal clinically important difference on the Motor Examination part of MDS-UPDRS. Parkinsonism Relat. Disord. 2015, 21, 1421–1426. [Google Scholar] [CrossRef] [PubMed]
- Taghizadeh, G.; Eissazade, N.; Fereshtehnejad, S.-M.; Sharabiani, P.T.A.; Shati, M.; Mortazavi, S.S.; Habibi, S.A.H.; SalemiJuybari, M.; Mehdizadeh, M. Minimal clinically important difference and substantial clinical benefits for single- and dual-task timed up and go test following motor-cognitive training in Parkinson’s disease. Age and Ageing 2025, 54. [Google Scholar] [CrossRef] [PubMed]
- Barry, E.; Galvin, R.; Keogh, C.; Horgan, F.; Fahey, T. Is the Timed Up and Go test a useful predictor of risk of falls in community dwelling older adults: a systematic review and meta- analysis. BMC Geriatr. 2014, 14, 14–14. [Google Scholar] [CrossRef] [PubMed]
- Curtze, C.; Nutt, J.G.; Carlson-Kuhta, P.; Mancini, M.; Horak, F.B. Levodopa Is a Double-Edged Sword for Balance and Gait in People With Parkinson's Disease. Mov. Disord. 2015, 30, 1361–1370. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente-FernánDez, R.; Ruth, T.J.; Sossi, V.; Schulzer, M.; Calne, D.B.; Stoessl, A.J. Expectation and Dopamine Release: Mechanism of the Placebo Effect in Parkinson's Disease. Science 2001, 293, 1164–1166. [Google Scholar] [CrossRef] [PubMed]
| n (%) | |
| UPRS, n (%) | |
| <25 | 1 (5.3) |
| >=25 | 15 (78.9) |
| Missing | 3 (15.8) |
| Age, n (%) | |
| <80y | 8 (42.1) |
| >80y | 8 (42.1) |
| Missing | 3 (15.8) |
| Dyskinesia/Dystonia/Freezing of gait/Falls | |
| No | 8 (42.1) |
| Yes | 8 (42.1) |
| Missing | 3 (15.8) |
| Length of diagnosis | |
| <7y | 5 (26.3) |
| >7y | 11 (57.9) |
| Missing | 3 (15.8) |
| n (%) | |
| Completion rate of diary | |
| Full, n (%) | 13 (81) |
| Partial, n (%) | 3 (19) |
| Median duration of use per day (hours) | 8 |
| Median duration of charge (hours) | 10 |
| Median comfort of device (1-5) | 5 |
| Skin irritation from adhesive (% of affected days) | 0.6 |
| Other side effects (% of affected days) | 0 |
| Device malfunction | |
| Yes n (%) | 3 (19) |
| No n (%) | 13 (81) |
| Continue to use the device | |
| Yes n (%) | 12 (75) |
| No n (%) | 1 (6) |
| Missing n (%) | 3 (19) |
| Able to independently use the device | |
| Yes n (%) | 12 (75) |
| No n (%) | 1 (6) |
| Missing n (%) | 3 (19) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




