Submitted:
10 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
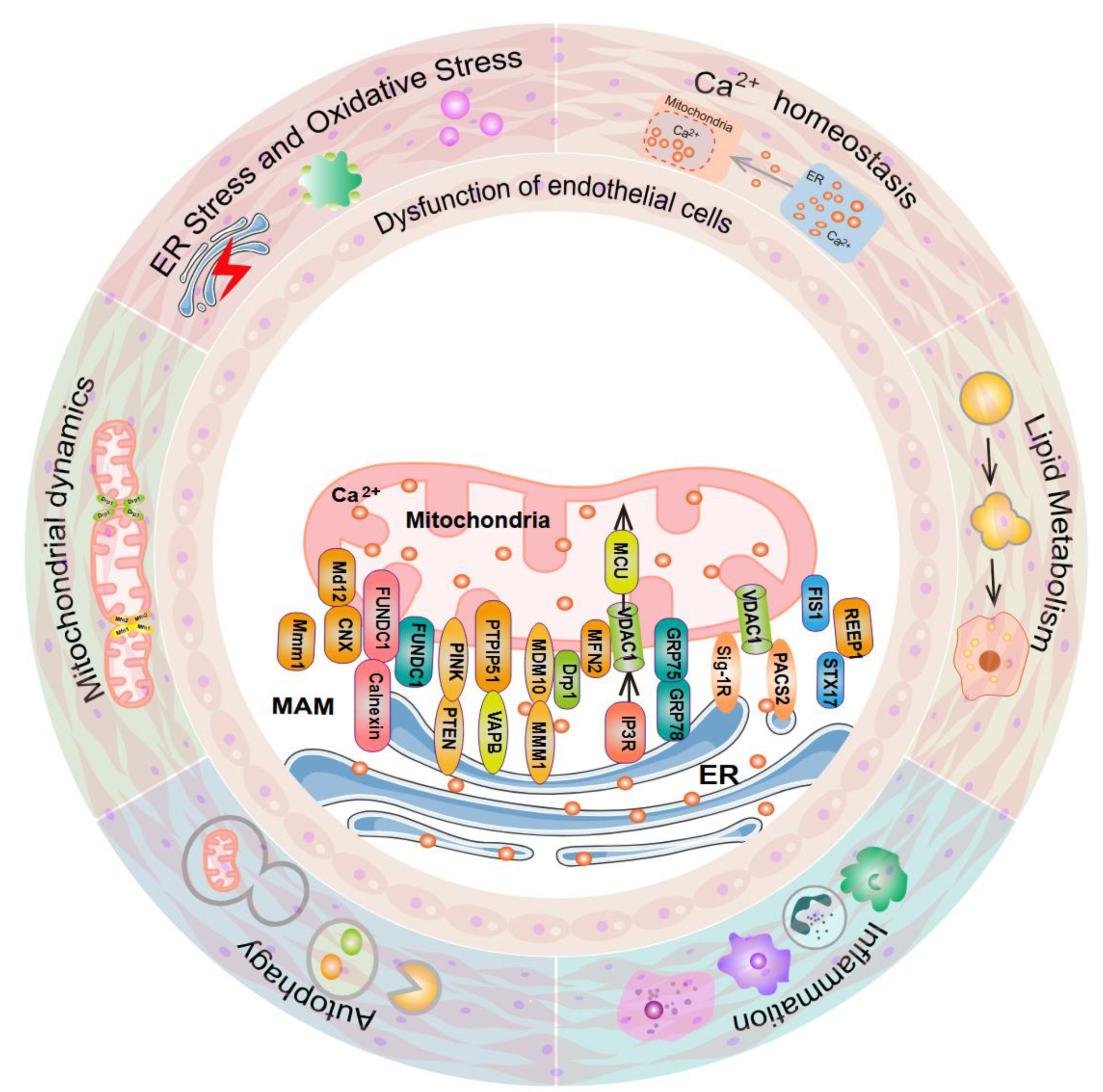
2. Composition and Structural Characteristics of MAMs
3. The Pathology of PH
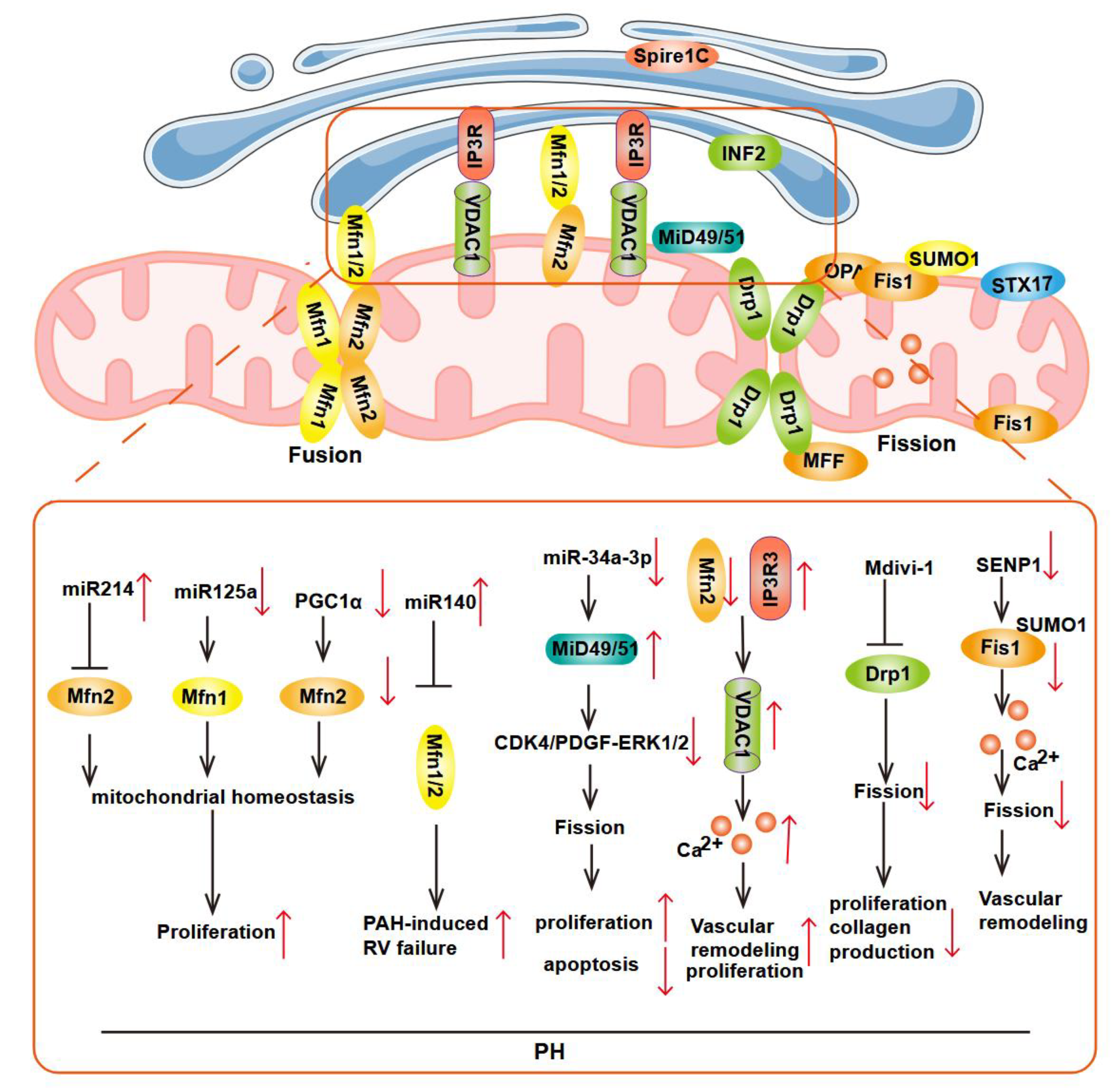
3.1. Regulatory Mechanism of MAM in Mitochondrial Fission and Fusion
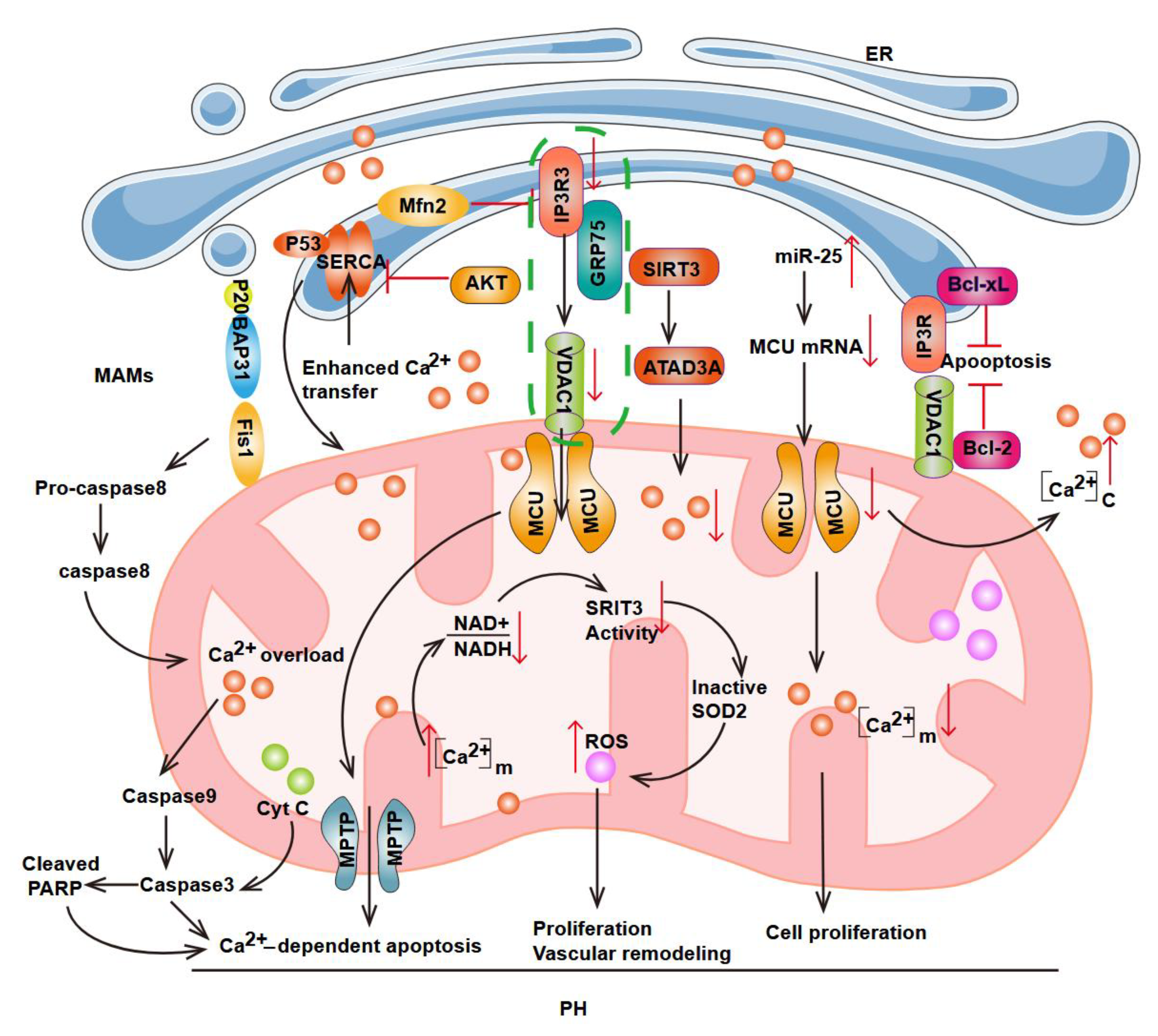
3.2. Mechanisms of Ca²⁺ Homeostasis Imbalance
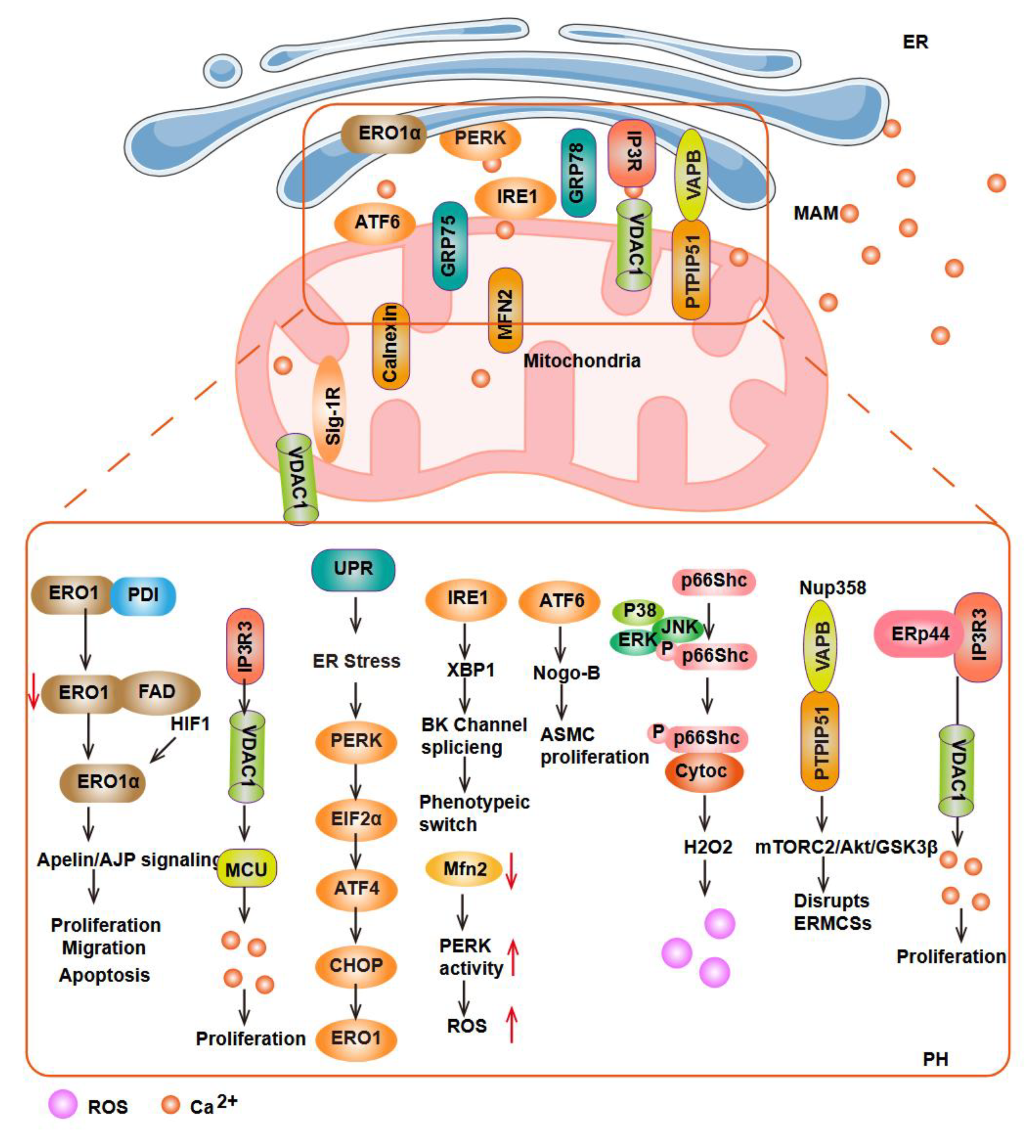
3.3. ER Stress and Oxidative Stress in PH
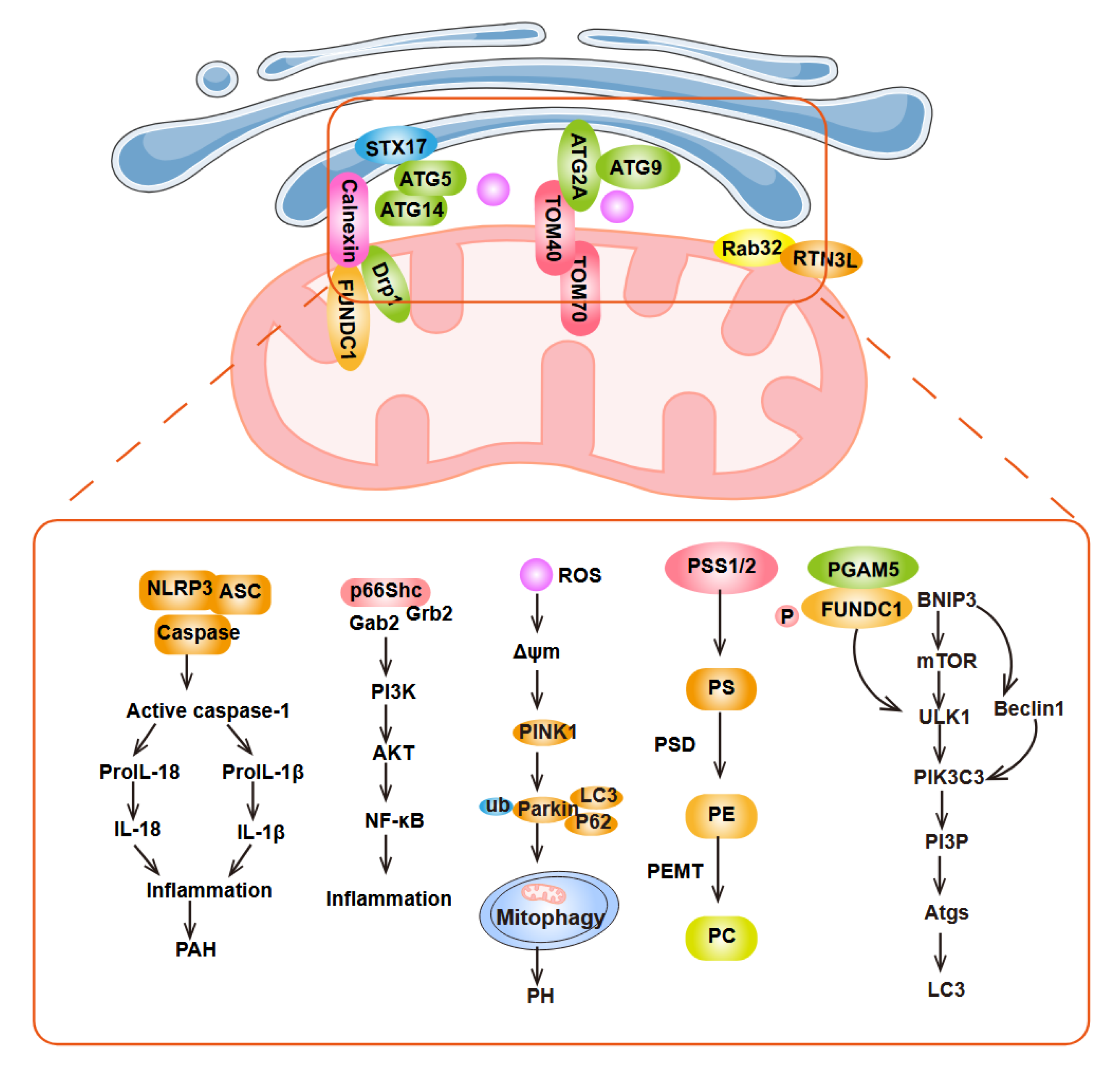
3.4. Involvement of MAM in Inflammation, Autophagy and Lipid Metabolism in PH
3.5. MAM-Mediated Apoptosis in PH
4. Therapeutic Strategies Targeting Mitochondria-ER Axis
4.1. Mitochondrial Dynamics as A Potential Therapeutic Target
4.2. Antioxidant and Metabolic Regulating Drugs
4.3. Ca²⁺ Signaling Regulation-Related Functional Roles and Their Therapeutic Targets
4.4. Autophagy Regulation-Related Functional Roles and Their Therapeutic Targets
5. Conclusions
Funding Statement
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ER | Endoplasmic reticulum |
| ERS | Endoplasmic Reticulum Stress |
| IP3Rs | Inositol triphosphate receptors |
| MAMs | Mitochondria-associated endoplasmic reticulum membranes |
| MCU | mitochondrial Ca²⁺ uniporter |
| NO | Nitric oxide |
| PAH | Pulmonary arterial hypertension |
| PASMCs | pulmonary artery smooth muscle cells |
| PAECs | Pulmonary artery endothelial cells |
| ROS | Reactive Oxygen Species |
| VDAC1 | Voltage-dependent anion channel 1 |
References
- Simonneau, G.; Gatzoulis, M.A.; Adatia, I.; Celermajer, D.; Denton, C.; Ghofrani, A.; Gomez Sanchez, M.A.; Krishna Kumar, R.; Landzberg, M.; Machado, R.F.; et al. Updated Clinical Classification of Pulmonary Hypertension. Journal of the American College of Cardiology 2013, 62, D34–D41. [Google Scholar] [CrossRef] [PubMed]
- Hoeper, M.M.; Humbert, M.; Souza, R.; Idrees, M.; Kawut, S.M.; Sliwa-Hahnle, K.; Jing, Z.C.; Gibbs, J.S. A global view of pulmonary hypertension. Lancet Respir Med 2016, 4, 306–322. [Google Scholar] [CrossRef] [PubMed]
- Humbert, M.; Kovacs, G.; Hoeper, M.M.; Badagliacca, R.; Berger, R.M.F.; Brida, M.; Carlsen, J.; Coats, A.J.S.; Escribano-Subias, P.; Ferrari, P.; et al. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J 2022, 43, 3618–3731. [Google Scholar] [CrossRef] [PubMed]
- Rabinovitch, M. Pathobiology of Pulmonary Hypertension. Annual Review of Pathology: Mechanisms of Disease 2007, 2, 369–399. [Google Scholar] [CrossRef]
- Rabinovitch, M. Molecular pathogenesis of pulmonary arterial hypertension. Journal of Clinical Investigation 2012, 122, 4306–4313. [Google Scholar] [CrossRef]
- Stacher, E.; Graham, B.B.; Hunt, J.M.; Gandjeva, A.; Groshong, S.D.; McLaughlin, V.V.; Jessup, M.; Grizzle, W.E.; Aldred, M.A.; Cool, C.D.; Tuder, R.M. Modern Age Pathology of Pulmonary Arterial Hypertension. American Journal of Respiratory and Critical Care Medicine 2012, 186, 261–272. [Google Scholar] [CrossRef]
- Thenappan, T.; Ormiston, M.L.; Ryan, J.J.; Archer, S.L. Pulmonary arterial hypertension: pathogenesis and clinical management. BMJ 2018, 360, j5492. [Google Scholar] [CrossRef]
- Gao, P.; Yan, Z.; Zhu, Z. Mitochondria-Associated Endoplasmic Reticulum Membranes in Cardiovascular Diseases. Frontiers in Cell and Developmental Biology 2020, 8. [Google Scholar] [CrossRef]
- Thenappan, T.; Al-Naamani, N.; Ghio, S.; Ghofrani, H.A.; Hassoun, P.M.; Pritzker, M.; Torbicki, A.; Nikkho, S.; Busse, D.; Preston, I.R. Effect of riociguat on pulmonary arterial compliance in the PATENT and CHEST studies. Pulmonary Circulation 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Huang, L.; Li, W.; Yang, T.; Xiong, C.; Ni, X.; Gu, Q.; He, J. Association between splenectomy and portal hypertension in the development of pulmonary hypertension. Pulmonary Circulation 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Sahay, S.; Tsang, Y.; Flynn, M.; Agron, P.; Dufour, R. Burden of pulmonary hypertension in patients with portal hypertension in the United States: a retrospective database study. Pulmonary Circulation 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Nathan, S.D.; Barnett, S.D.; King, C.S.; Provencher, S.; Barbera, J.A.; Pastre, J.; Shlobin, O.A.; Seeger, W. Impact of the new definition for pulmonary hypertension in patients with lung disease: an analysis of the United Network for Organ Sharing database. Pulmonary Circulation 2021, 11, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Chen, L.; Ye, F.; Tang, J.; Liu, B.; Lin, J.; Zhou, P.-H.; Lu, B.; Wu, M.; Lu, J.-H.; et al. Mic19 depletion impairs endoplasmic reticulum-mitochondrial contacts and mitochondrial lipid metabolism and triggers liver disease. Nature Communications 2024, 15. [Google Scholar] [CrossRef] [PubMed]
- Xiao, P.; Wu, S.; Wang, Z.; Shen, G.; Shi, X. Biotoxicity of paraquat to lung cells mediated by endoplasmic reticulum-mitochondria interaction. Journal of Molecular Histology 2024, 55, 1063–1077. [Google Scholar] [CrossRef]
- Rajagopal, S; Ghadimi, R.K.; Horn, K; Kelava, EM; Kudelko, M; Moreno-Duarte, KT; Preston, I; Rose Bovino, I; Smilowitz, LL; Vaidya, NR; A; American Heart Association Council on Cardiopulmonary; Critical Care; Perioperative and Resuscitation; and the Council on Cardiovascular and Stroke Nursing. Evaluation and Management of Pulmonary Hypertension in Noncardiac Surgery_ A Scientific Statement From the American Heart Association. Circulation 2023, 147, 1317–134. [CrossRef]
- Friedman, S.H.; Harley, R.A.; Williams, J.; Forcucci, J.A.; Ramakrishnan, V.; Strange, C.; Silver, R.M.; Feghali-Bostwick, C.; Argula, R.G. Pathologic differences between systemic sclerosis–associated and idiopathic pulmonary arterial hypertension. Journal of Scleroderma and Related Disorders 2024. [Google Scholar] [CrossRef]
- Mocumbi, A.; Humbert, M.; Saxena, A.; Jing, Z.-C.; Sliwa, K.; Thienemann, F.; Archer, S.L.; Stewart, S. Pulmonary hypertension. Nature Reviews Disease Primers 2024, 10. [Google Scholar] [CrossRef]
- Garry, J.D.; Kundu, S.; Annis, J.; Alcorn, C.; Eden, S.; Smith, E.; Greevy, R.; Maron, B.A.; Freiberg, M.S.; Brittain, E.L. Incidence of Pulmonary Hypertension in the Echocardiography Referral Population. Ann Am Thorac Soc 2025, 22. [Google Scholar] [CrossRef]
- Bonnet, D.; Szezepanski, I.; Delacourt, C.; Malkezadeh-Milani, S.; Lévy, M. Multifactorial pulmonary hypertension in infantile scimitar syndrome. Archives of Cardiovascular Diseases 2022, 115, 142–150. [Google Scholar] [CrossRef]
- Takano, R.; Aoki, T.; Asano, R.; Ueda, J.; Tsuji, A.; Omae, K.; Ogo, T. Recurrent pulmonary hypertension after balloon pulmonary angioplasty for inoperable chronic thromboembolic pulmonary hypertension. The Journal of Heart and Lung Transplantation 2024, 43, 737–744. [Google Scholar] [CrossRef]
- Yang, Y.D.; Li, M.M.; Xu, G.; Zhang, E.L.; Chen, J.; Sun, B.; Chen, D.W.; Gao, Y.Q. Targeting mitochondria-associated membranes as a potential therapy against endothelial injury induced by hypoxia. Journal of Cellular Biochemistry 2019, 120, 18967–18978. [Google Scholar] [CrossRef]
- Muñoz, E.; Valero, R.A.; Quintana, A.; Hoth, M.; Núñez, L.; Villalobos, C. Nonsteroidal Anti-inflammatory Drugs Inhibit Vascular Smooth Muscle Cell Proliferation by Enabling the Ca2+-dependent Inactivation of Calcium Release-activated Calcium/Orai Channels Normally Prevented by Mitochondria. Journal of Biological Chemistry 2011, 286, 16186–16196. [Google Scholar] [CrossRef] [PubMed]
- V, T. (2021 Mar 13). Life (Basel) 11. [CrossRef]
- V, T. Oligodendroglial Energy Metabolism and (re)Myelination. Life (Basel) 2021, 11, 238. [Google Scholar] [CrossRef]
- Luo, L.; Yin, H.; Gou, D. Gut Microbiota and Metabolome Changes in Three Pulmonary Hypertension Rat Models. Microorganisms 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Missiroli, S.; Patergnani, S.; Caroccia, N.; Pedriali, G.; Perrone, M.; Previati, M.; Wieckowski, M.R.; Giorgi, C. Mitochondria-associated membranes (MAMs) and inflammation. Cell Death & Disease 2018, 9. [Google Scholar] [CrossRef]
- Dorn, G.W. Mitofusins as mitochondrial anchors and tethers. Journal of Molecular and Cellular Cardiology 2020, 142, 146–153. [Google Scholar] [CrossRef]
- Szabadkai, G.r.; Bianchi, K.; Várnai, P.t.; De Stefani, D.; Wieckowski, M.R.; Cavagna, D.; Nagy, A.I.; Balla, T.s.; Rizzuto, R. Chaperone-mediated coupling of endoplasmic reticulum and mitochondrial Ca2+ channels. The Journal of Cell Biology 2006, 175, 901–911. [Google Scholar] [CrossRef]
- Peng, Y.; Zhou, L.; Jin, Y.; Wu, D.; Chen, N.; Zhang, C.; Liu, H.; Li, C.; Ning, R.; Yang, X.; et al. Calcium bridges built by mitochondria-associated endoplasmic reticulum membranes: potential targets for neural repair in neurological diseases. Neural Regeneration Research 2025, 20, 3349–3369. [Google Scholar] [CrossRef]
- Hayashi, T.; Su, T.-P. Sigma-1 Receptor Chaperones at the ER- Mitochondrion Interface Regulate Ca2+ Signaling and Cell Survival. Cell 2007, 131, 596–610. [Google Scholar] [CrossRef]
- Gomez-Suaga, P.; Paillusson, S.; Stoica, R.; Noble, W.; Hanger, D.P.; Miller, C.C.J. The ER-Mitochondria Tethering Complex VAPB-PTPIP51 Regulates Autophagy. Current Biology 2017, 27, 371–385. [Google Scholar] [CrossRef]
- Lim, Y.; Cho, I.T.; Schoel, L.J.; Cho, G.; Golden, J.A. Hereditary spastic paraplegia-linked REEP1 modulates endoplasmic reticulum/mitochondria contacts. Annals of Neurology 2015, 78, 679–696. [Google Scholar] [CrossRef]
- Gelmetti, V.; De Rosa, P.; Torosantucci, L.; Marini, E.S.; Romagnoli, A.; Di Rienzo, M.; Arena, G.; Vignone, D.; Fimia, G.M.; Valente, E.M. PINK1 and BECN1 relocalize at mitochondria-associated membranes during mitophagy and promote ER-mitochondria tethering and autophagosome formation. Autophagy 2017, 13, 654–669. [Google Scholar] [CrossRef]
- Gutiérrez, T.; Simmen, T. Endoplasmic reticulum chaperones tweak the mitochondrial calcium rheostat to control metabolism and cell death. Cell Calcium 2018, 70, 64–75. [Google Scholar] [CrossRef]
- Betz, C.; Stracka, D.; Prescianotto-Baschong, C.; Frieden, M.; Demaurex, N.; Hall, M.N. mTOR complex 2-Akt signaling at mitochondria-associated endoplasmic reticulum membranes (MAM) regulates mitochondrial physiology. Proceedings of the National Academy of Sciences 2013, 110, 12526–12534. [Google Scholar] [CrossRef] [PubMed]
- Ding, W.-X.; Yin, X.-M. Mitophagy: mechanisms, pathophysiological roles, and analysis. bchm 2012, 393, 547–564. [Google Scholar] [CrossRef] [PubMed]
- Hamasaki, M.; Furuta, N.; Matsuda, A.; Nezu, A.; Yamamoto, A.; Fujita, N.; Oomori, H.; Noda, T.; Haraguchi, T.; Hiraoka, Y.; et al. Autophagosomes form at ER–mitochondria contact sites. Nature 2013, 495, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Shuda, M.; Kondoh, N.; Imazeki, N.; Tanaka, K.; Okada, T.; Mori, K.; Hada, A.; Arai, M.; Wakatsuki, T.; Matsubara, O.; et al. Activation of the ATF6, XBP1 and grp78 genes in human hepatocellular carcinoma: a possible involvement of the ER stress pathway in hepatocarcinogenesis. Journal of Hepatology 2003, 38, 605–614. [Google Scholar] [CrossRef]
- Zhang, S.-S.; Zhou, S.; Crowley-McHattan, Z.J.; Wang, R.-Y.; Li, J.-P. A Review of the Role of Endo/Sarcoplasmic Reticulum-Mitochondria Ca2+ Transport in Diseases and Skeletal Muscle Function. International Journal of Environmental Research and Public Health 2021, 18. [Google Scholar] [CrossRef]
- Pokharel, M.D.; Garcia-Flores, A.; Marciano, D.; Franco, M.C.; Fineman, J.R.; Aggarwal, S.; Wang, T.; Black, S.M. Mitochondrial network dynamics in pulmonary disease: Bridging the gap between inflammation, oxidative stress, and bioenergetics. Redox Biology 2024, 70. [Google Scholar] [CrossRef]
- Wu, D.; Jansen-van Vuuren, R.D.; Dasgupta, A.; Al-Qazazi, R.; Chen, K.-H.; Martin, A.Y.; Mewburn, J.D.; Alizadeh, E.; Lima, P.D.A.; Jones, O.; et al. Novel Drp1 GTPase Inhibitor, Drpitor1a: Efficacy in Pulmonary Hypertension. Hypertension 2024, 81, 2189–2201. [Google Scholar] [CrossRef]
- Colpman, P.; Dasgupta, A.; Archer, S.L. The Role of Mitochondrial Dynamics and Mitotic Fission in Regulating the Cell Cycle in Cancer and Pulmonary Arterial Hypertension: Implications for Dynamin-Related Protein 1 and Mitofusin2 in Hyperproliferative Diseases. Cells 2023, 12. [Google Scholar] [CrossRef]
- Luo, F.; Fu, M.; Wang, T.; Qi, Y.; Zhong, X.; Li, D.; Liu, B. Down-regulation of the mitochondrial fusion protein Opa1/Mfn2 promotes cardiomyocyte hypertrophy in Su5416/hypoxia-induced pulmonary hypertension rats. Archives of Biochemistry and Biophysics 2023, 747. [Google Scholar] [CrossRef]
- Heresi, G.A.; Mey, J.T.; Bartholomew, J.R.; Haddadin, I.S.; Tonelli, A.R.; Dweik, R.A.; Kirwan, J.P.; Kalhan, S.C. Plasma metabolomic profile in chronic thromboembolic pulmonary hypertension. Pulmonary Circulation 2020, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Zhang, C.; Ma, M.; Zhang, L.; Zhang, L.; Zhang, F.; Chen, Y.; Cao, F.; Li, M.; Wang, G.; et al. MiR-125a regulates mitochondrial homeostasis through targeting mitofusin 1 to control hypoxic pulmonary vascular remodeling. Journal of Molecular Medicine 2017, 95, 977–993. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Potus, F.; Wu, D.; Dasgupta, A.; Chen, K.-H.; Mewburn, J.; Lima, P.; Archer, S.L. Increased Drp1-Mediated Mitochondrial Fission Promotes Proliferation and Collagen Production by Right Ventricular Fibroblasts in Experimental Pulmonary Arterial Hypertension. Frontiers in Physiology 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.-H.; Dasgupta, A.; Lin, J.; Potus, F.; Bonnet, S.; Iremonger, J.; Fu, J.; Mewburn, J.; Wu, D.; Dunham-Snary, K.; et al. Epigenetic Dysregulation of the Dynamin-Related Protein 1 Binding Partners MiD49 and MiD51 Increases Mitotic Mitochondrial Fission and Promotes Pulmonary Arterial Hypertension. Circulation 2018, 138, 287–304. [Google Scholar] [CrossRef]
- Ryan, J.J.; Marsboom, G.; Fang, Y.-H.; Toth, P.T.; Morrow, E.; Luo, N.; Piao, L.; Hong, Z.; Ericson, K.; Zhang, H.J.; et al. PGC1α-mediated Mitofusin-2 Deficiency in Female Rats and Humans with Pulmonary Arterial Hypertension. American Journal of Respiratory and Critical Care Medicine 2013, 187, 865–878. [Google Scholar] [CrossRef]
- Marsboom, G.; Toth, P.T.; Ryan, J.J.; Hong, Z.; Wu, X.; Fang, Y.-H.; Thenappan, T.; Piao, L.; Zhang, H.J.; Pogoriler, J.; et al. Dynamin-Related Protein 1–Mediated Mitochondrial Mitotic Fission Permits Hyperproliferation of Vascular Smooth Muscle Cells and Offers a Novel Therapeutic Target in Pulmonary Hypertension. Circulation Research 2012, 110, 1484–1497. [Google Scholar] [CrossRef]
- Zhou, X.; Jiang, Y.; Wang, Y.; Fan, L.; Zhu, Y.; Chen, Y.; Wang, Y.; Zhu, Y.; Wang, H.; Pan, Z.; et al. Endothelial FIS1 DeSUMOylation Protects Against Hypoxic Pulmonary Hypertension. Circulation Research 2023, 133, 508–531. [Google Scholar] [CrossRef]
- Friedman, J.R.; Lackner, L.L.; West, M.; DiBenedetto, J.R.; Nunnari, J.; Voeltz, G.K. ER Tubules Mark Sites of Mitochondrial Division. Science 2011, 334, 358–362. [Google Scholar] [CrossRef]
- Ma, L.; Chen, C.; Hai, S.; Wang, C.; Rahman, S.U.; Huang, W.; Zhao, C.; Feng, S.; Wang, X. Inhibition of Mitochondrial Fission Alleviates Zearalenone-Induced Mitochondria-Associated Endoplasmic Reticulum Membrane Dysfunction in Piglet Sertoli Cells. Toxins 2023, 15. [Google Scholar] [CrossRef]
- Li, X.; Yang, Q.; Liu, S.; Song, S.; Wang, C. Mitochondria-associated endoplasmic reticulum membranes promote mitochondrial fission through AKAP1-Drp1 pathway in podocytes under high glucose conditions. Experimental Cell Research 2023, 424. [Google Scholar] [CrossRef]
- Dunham-Snary, K.J.; Wu, D.; Sykes, E.A.; Thakrar, A.; Parlow, L.R.G.; Mewburn, J.D.; Parlow, J.L.; Archer, S.L. Hypoxic Pulmonary Vasoconstriction: From Molecular Mechanisms to Medicine. Chest 2017, 151, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Masson, B.; Saint-Martin Willer, A.; Dutheil, M.; Penalva, L.; Le Ribeuz, H.; El Jekmek, K.; Ruchon, Y.; Cohen-Kaminsky, S.; Sabourin, J.; Humbert, M.; et al. Contribution of transient receptor potential canonical channels in human and experimental pulmonary arterial hypertension. American Journal of Physiology-Lung Cellular and Molecular Physiology 2023, 325, L246–L261. [Google Scholar] [CrossRef] [PubMed]
- Masson, B.; Montani, D.; Humbert, M.; Capuano, V.; Antigny, F. Role of Store-Operated Ca2+ Entry in the Pulmonary Vascular Remodeling Occurring in Pulmonary Arterial Hypertension. Biomolecules 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Chen, J.; Wang, T.; Chen, B.; Yang, L.; Liao, J.; Chen, Y.; Wang, J; Tang, H.; Yi, J.; et al. PDGF_MEK_ERK axis represses Ca2+ clearance via decreasing the abundance of plasma membrane Ca2+ pump PMCA4 in pulmonary arterial smooth muscle cells. Am J Physiol Cell Physiol 2021, 320, C66–C79. [Google Scholar] [CrossRef]
- Neumann, J.; Van Nieuwenhove, E.; Terry, L.E.; Staels, F.; Knebel, T.R.; Welkenhuyzen, K.; Ahmadzadeh, K.; Baker, M.R.; Gerbaux, M.; Willemsen, M.; et al. Disrupted Ca2+ homeostasis and immunodeficiency in patients with functional IP3 receptor subtype 3 defects. Cellular & Molecular Immunology 2022, 20, 11–25. [Google Scholar] [CrossRef]
- Shoshan-Barmatz, V.; Krelin, Y.; Shteinfer-Kuzmine, A. VDAC1 functions in Ca2+ homeostasis and cell life and death in health and disease. Cell Calcium 2018, 69, 81–100. [Google Scholar] [CrossRef]
- Chen, Y.; Csordás, G.; Jowdy, C.; Schneider, T.G.; Csordás, N.; Wang, W.; Liu, Y.; Kohlhaas, M.; Meiser, M.; Bergem, S.; et al. Mitofusin 2-Containing Mitochondrial-Reticular Microdomains Direct Rapid Cardiomyocyte Bioenergetic Responses Via Interorganelle Ca2+Crosstalk. Circulation Research 2012, 111, 863–875. [Google Scholar] [CrossRef]
- Han, C.; Kim, J. Transcriptome profiling reveals novel insights into the regulation of calcium ion and detoxification genes driving chlorantraniliprole resistance in Spodoptera exigua. Heliyon 2024. [Google Scholar] [CrossRef]
- Wang, Q.-C.; Zheng, Q.; Tan, H.; Zhang, B.; Li, X.; Yang, Y.; Yu, J.; Liu, Y.; Chai, H.; Wang, X.; et al. TMCO1 Is an ER Ca 2+ Load-Activated Ca 2+ Channel. Cell 2016, 165, 1454–1466. [Google Scholar] [CrossRef]
- Breckenridge, D.G.; Stojanovic, M.; Marcellus, R.C.; Shore, G.C. Caspase cleavage product of BAP31 induces mitochondrial fission through endoplasmic reticulum calcium signals, enhancing cytochrome c release to the cytosol. The Journal of Cell Biology 2003, 160, 1115–1127. [Google Scholar] [CrossRef] [PubMed]
- Chandra, D.; Choy, G.; Deng, X.; Bhatia, B.; Daniel, P.; Tang, D.G. Association of Active Caspase 8 with the Mitochondrial Membrane during Apoptosis: Potential Roles in Cleaving BAP31 and Caspase 3 and Mediating Mitochondrion-Endoplasmic Reticulum Cross Talk in Etoposide-Induced Cell Death. Molecular and Cellular Biology 2023, 24, 6592–6607. [Google Scholar] [CrossRef] [PubMed]
- Iwasawa, R.; Mahul-Mellier, A.-L.; Datler, C.; Pazarentzos, E.; Grimm, S. Fis1 and Bap31 bridge the mitochondria-ER interface to establish a platform for apoptosis induction. The EMBO Journal 2011, 30, 556–568. [Google Scholar] [CrossRef] [PubMed]
- Simmen, T.; Aslan, J.E.; Blagoveshchenskaya, A.D.; Thomas, L.; Wan, L.; Xiang, Y.; Feliciangeli, S.F.; Hung, C.-H.; Crump, C.M.; Thomas, G. PACS-2 controls endoplasmic reticulum–mitochondria communication and Bid-mediated apoptosis. The EMBO Journal 2005, 24, 717–729. [Google Scholar] [CrossRef]
- Hong, Z.; Chen, K.-H.; DasGupta, A.; Potus, F.; Dunham-Snary, K.; Bonnet, S.; Tian, L.; Fu, J.; Breuils-Bonnet, S.; Provencher, S.; et al. MicroRNA-138 and MicroRNA-25 Down-regulate Mitochondrial Calcium Uniporter, Causing the Pulmonary Arterial Hypertension Cancer Phenotype. American Journal of Respiratory and Critical Care Medicine 2017, 195, 515–529. [Google Scholar] [CrossRef]
- Monaco, G.; Decrock, E.; Arbel, N.; van Vliet, A.R.; La Rovere, R.M.; De Smedt, H.; Parys, J.B.; Agostinis, P.; Leybaert, L.; Shoshan-Barmatz, V.; Bultynck, G. The BH4 Domain of Anti-apoptotic Bcl-XL, but Not That of the Related Bcl-2, Limits the Voltage-dependent Anion Channel 1 (VDAC1)-mediated Transfer of Pro-apoptotic Ca2+ Signals to Mitochondria. Journal of Biological Chemistry 2015, 290, 9150–9161. [Google Scholar] [CrossRef]
- Chen, J.; Rodriguez, M.; Miao, J.; Liao, J.; Jain, P.P.; Zhao, M.; Zhao, T.; Babicheva, A.; Wang, Z.; Parmisano, S.; et al. Mechanosensitive channel Piezo1 is required for pulmonary artery smooth muscle cell proliferation. American Journal of Physiology-Lung Cellular and Molecular Physiology 2022, 322, L737–L760. [Google Scholar] [CrossRef]
- Romito, O.; Guéguinou, M.; Raoul, W.; Champion, O.; Robert, A.; Trebak, M.; Goupille, C.; Potier-Cartereau, M. Calcium signaling: A therapeutic target to overcome resistance to therapies in cancer. Cell Calcium 2022, 108. [Google Scholar] [CrossRef]
- Tang, H.; Yamamura, A.; Yamamura, H.; Song, S.; Fraidenburg, D.R.; Chen, J.; Gu, Y.; Pohl, N.M.; Zhou, T.; Jiménez-Pérez, L.; et al. Pathogenic role of calcium-sensing receptors in the development and progression of pulmonary hypertension. American Journal of Physiology-Lung Cellular and Molecular Physiology 2016, 310, L846–L859. [Google Scholar] [CrossRef]
- Wang, R.; Wang, J.; Yu, J.; Li, Z.; Zhang, M.; Chen, Y.; Liu, F.; Jiang, D.; Guo, J.; Li, X.; Wu, Y. Mfn2 regulates calcium homeostasis and suppresses PASMCs proliferation via interaction with IP3R3 to mitigate pulmonary arterial hypertension. Journal of Translational Medicine 2025, 23. [Google Scholar] [CrossRef]
- Guo, X.; Meng, Y.; Wang, Y.; Nan, S.; Lu, Y.; Lu, D.; Yin, Y. Mice lacking 1,4,5-triphosphate inositol type III receptor demonstrate inhibition of hypoxic pulmonary hypertension. Biochemical and Biophysical Research Communications 2022, 629, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.-L.; Yi, J.; Cao, X.-Y.; Wang, F.-Y.; Xie, S.-L.; Zhou, L.-L.; Qin, L.; Dai, A.-G. Celastrol: The new dawn in the treatment of vascular remodeling diseases. Biomedicine & Pharmacotherapy 2023, 158. [Google Scholar] [CrossRef] [PubMed]
- Sutendra, G.; Dromparis, P.; Wright, P.; Bonnet, S.; Haromy, A.; Hao, Z.; McMurtry, M.S.; Michalak, M.; Vance, J.E.; Sessa, W.C.; Michelakis, E.D. The Role of Nogo and the Mitochondria–Endoplasmic Reticulum Unit in Pulmonary Hypertension. Science Translational Medicine 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, J.P.; Zorzano, A. Endoplasmic reticulum stress enters a Nogo zone. Sci Transl Med 2011, 3, 88ps26. [Google Scholar] [CrossRef]
- Zhang, C.F.; Zhao, F.Y.; Xu, S.L.; Liu, J.; Xing, X.Q.; Yang, J. Autophagy in pulmonary hypertension: Emerging roles and therapeutic implications. Journal of Cellular Physiology 2019, 234, 16755–16767. [Google Scholar] [CrossRef]
- Li, Y.; Li, H.-Y.; Shao, J.; Zhu, L.; Xie, T.-H.; Cai, J.; Wang, W.; Cai, M.-X.; Wang, Z.-L.; Yao, Y.; Wei, T.-T. GRP75 Modulates Endoplasmic Reticulum–Mitochondria Coupling and Accelerates Ca2+-Dependent Endothelial Cell Apoptosis in Diabetic Retinopathy. Biomolecules 2022, 12. [Google Scholar] [CrossRef]
- Koyama, M.; Furuhashi, M.; Ishimura, S.; Mita, T.; Fuseya, T.; Okazaki, Y.; Yoshida, H.; Tsuchihashi, K.; Miura, T. Reduction of endoplasmic reticulum stress by 4-phenylbutyric acid prevents the development of hypoxia-induced pulmonary arterial hypertension. American Journal of Physiology-Heart and Circulatory Physiology 2014, 306, H1314–H1323. [Google Scholar] [CrossRef]
- Victor, P.; Umapathy, D.; George, L.; Juttada, U.; Ganesh, G.V.; Amin, K.N.; Viswanathan, V.; Ramkumar, K.M. Crosstalk between endoplasmic reticulum stress and oxidative stress in the progression of diabetic nephropathy. Cell Stress and Chaperones 2021, 26, 311–321. [Google Scholar] [CrossRef]
- Ding, W.; Zhang, X.; Zhang, Q.; Dong, Y.; Wang, W.; Ding, N. Adiponectin ameliorates lung injury induced by intermittent hypoxia through inhibition of ROS-associated pulmonary cell apoptosis. Sleep and Breathing 2020, 25, 459–470. [Google Scholar] [CrossRef]
- Rabinovitch, M.; Guignabert, C.; Humbert, M.; Nicolls, M.R. Inflammation and Immunity in the Pathogenesis of Pulmonary Arterial Hypertension. Circulation Research 2014, 115, 165–175. [Google Scholar] [CrossRef]
- Golembeski, S.M.; West, J.; Tada, Y.; Fagan, K.A. Interleukin-6 Causes Mild Pulmonary Hypertension and Augments Hypoxia-Induced Pulmonary Hypertension in Mice. Chest 2005, 128, 572S–573S. [Google Scholar] [CrossRef]
- Rieusset, J. Mitochondria-associated membranes (MAMs): An emerging platform connecting energy and immune sensing to metabolic flexibility. Biochemical and Biophysical Research Communications 2018, 500, 35–44. [Google Scholar] [CrossRef]
- Liu, Helene M.; Loo, Y.-M.; Horner, Stacy M.; Zornetzer, Gregory A.; Katze, Michael G.; Gale, M. The Mitochondrial Targeting Chaperone 14-3-3ε Regulates a RIG-I Translocon that Mediates Membrane Association and Innate Antiviral Immunity. Cell Host & Microbe 2012, 11, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Callaway, J.B.; Ting, J.P.Y. Inflammasomes: mechanism of action, role in disease, and therapeutics. Nature Medicine 2015, 21, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Strowig, T.; Henao-Mejia, J.; Elinav, E.; Flavell, R. Inflammasomes in health and disease. Nature 2012, 481, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. International Journal of Molecular Sciences 2019, 20. [Google Scholar] [CrossRef]
- Zhao, J.; Li, J.; Li, G.; Chen, M. The role of mitochondria-associated membranes mediated ROS on NLRP3 inflammasome in cardiovascular diseases. Frontiers in Cardiovascular Medicine 2022, 9. [Google Scholar] [CrossRef]
- Zhou, R.; Yazdi, A.S.; Menu, P.; Tschopp, J. A role for mitochondria in NLRP3 inflammasome activation. Nature 2010, 469, 221–225. [Google Scholar] [CrossRef]
- Li, W.; Cao, T.; Luo, C.; Cai, J.; Zhou, X.; Xiao, X.; Liu, S. Crosstalk between ER stress, NLRP3 inflammasome, and inflammation. Applied Microbiology and Biotechnology 2020, 104, 6129–6140. [Google Scholar] [CrossRef]
- Humbert M, M.G., Brenot F, Sitbon O, Portier A, Grangeot-Keros L, Duroux P, Galanaud P, Simonneau G, Emilie D. (1995). Increased interleukin-1 and interleukin-6 serum concentrations in severe primary pulmonary hypertension. Am J Respir Crit Care Med 151, 1628-1631. [CrossRef]
- Itoh T, N.N., Ishibashi-Ueda H, Kyotani S, Oya H, Sakamaki F, Kimura H, Nakanishi N. (2006). Increased plasma monocyte chemoattractant protein-1 level in idiopathic pulmonary arterial hypertension. Respirology 11, 158-163. [CrossRef] [PubMed]
- Steiner, M.K.; Syrkina, O.L.; Kolliputi, N.; Mark, E.J.; Hales, C.A.; Waxman, A.B. Interleukin-6 Overexpression Induces Pulmonary Hypertension. Circulation Research 2009, 104, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Zhao, Y.; Li, S.; Wu, H.; Ma, D.; Wan, C. TNF-α and IL-8 levels are positively correlated with hypobaric hypoxic pulmonary hypertension and pulmonary vascular remodeling in rats. Open Life Sciences 2023, 18. [Google Scholar] [CrossRef] [PubMed]
- Vringer, E.; Tait, S.W.G. Mitochondria and cell death-associated inflammation. Cell Death & Differentiation 2022, 30, 304–312. [Google Scholar] [CrossRef]
- Mittal, M.; Siddiqui, M.R.; Tran, K.; Reddy, S.P.; Malik, A.B. Reactive Oxygen Species in Inflammation and Tissue Injury. Antioxidants & Redox Signaling 2014, 20, 1126–1167. [Google Scholar] [CrossRef]
- De Gaetano, A.; Solodka, K.; Zanini, G.; Selleri, V.; Mattioli, A.V.; Nasi, M.; Pinti, M. Molecular Mechanisms of mtDNA-Mediated Inflammation. Cells 2021, 10. [Google Scholar] [CrossRef]
- Kazmirczak, F.; Vogel, N.T.; Prisco, S.Z.; Patterson, M.T.; Annis, J.; Moon, R.T.; Hartweck, L.M.; Mendelson, J.B.; Kim, M.; Calixto Mancipe, N.; et al. Ferroptosis-Mediated Inflammation Promotes Pulmonary Hypertension. Circulation Research 2024, 135, 1067–1083. [Google Scholar] [CrossRef]
- Wang, Y.; Cai, J.; Tang, C.; Dong, Z. Mitophagy in Acute Kidney Injury and Kidney Repair. Cells 2020, 9. [Google Scholar] [CrossRef]
- Saraji, A.; Sydykov, A.; Schafer, K.; Garcia-Castro, C.F.; Henneke, I.; Alebrahimdehkordi, N.; Kosanovic, D.; Hadzic, S.; Guenther, A.; Hecker, M.; et al. PINK1-mediated Mitophagy Contributes to Pulmonary Vascular Remodeling in Pulmonary Hypertension. Am J Respir Cell Mol Biol 2021, 65, 226–228. [Google Scholar] [CrossRef]
- Calì, T.; Ottolini, D.; Negro, A.; Brini, M. Enhanced parkin levels favor ER-mitochondria crosstalk and guarantee Ca2+ transfer to sustain cell bioenergetics. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease 2013, 1832, 495–508. [Google Scholar] [CrossRef]
- Wu, W.; Li, W.; Chen, H.; Jiang, L.; Zhu, R.; Feng, D. FUNDC1 is a novel mitochondrial-associated-membrane (MAM) protein required for hypoxia-induced mitochondrial fission and mitophagy. Autophagy 2016, 12, 1675–1676. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Chen, H.; Li, J.; Xin, Y.; Li, J. Phosphatidylethanolamine N-methyltransferase: from Functions to Diseases. Aging and disease 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Zhao, H.; Kalionis, B.; Huai, X.; Hu, X.; Wu, W.; Jiang, R.; Gong, S.; Wang, L.; Liu, J.; et al. The Impact of Abnormal Lipid Metabolism on the Occurrence Risk of Idiopathic Pulmonary Arterial Hypertension. International Journal of Molecular Sciences 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Wang, X.; Zhang, L.; Zhu, X.; Bai, J.; He, S.; Mei, J.; Jiang, J.; Guan, X.; Zheng, X.; et al. Super Enhancer-Associated Circular RNA-CircKrt4 Regulates Hypoxic Pulmonary Artery Endothelial Cell Dysfunction in Mice. Arterioscler Thromb Vasc Biol 2023, 43, 1179–1198. [Google Scholar] [CrossRef]
- Newton, D.A.; Lottes, R.G.; Ryan, R.M.; Spyropoulos, D.D.; Baatz, J.E. Dysfunctional lactate metabolism in human alveolar type II cells from idiopathic pulmonary fibrosis lung explant tissue. Respiratory Research 2021, 22. [Google Scholar] [CrossRef]
- Leopold, J.; Maron, B. Molecular Mechanisms of Pulmonary Vascular Remodeling in Pulmonary Arterial Hypertension. International Journal of Molecular Sciences 2016, 17. [Google Scholar] [CrossRef]
- Tang, H.; Babicheva, A.; McDermott, K.M.; Gu, Y.; Ayon, R.J.; Song, S.; Wang, Z.; Gupta, A.; Zhou, T.; Sun, X.; et al. Endothelial HIF-2α Contributes to Severe Pulmonary Hypertension by Inducing Endothelial-to-Mesenchymal Transition. American Journal of Physiology-Lung Cellular and Molecular Physiology 2017. [Google Scholar] [CrossRef]
- Plecitá-Hlavatá, L.; Tauber, J.; Li, M.; Zhang, H.; Flockton, A.R.; Pullamsetti, S.S.; Chelladurai, P.; D’Alessandro, A.; El Kasmi, K.C.; Ježek, P.; Stenmark, K.R. Constitutive Reprogramming of Fibroblast Mitochondrial Metabolism in Pulmonary Hypertension. American Journal of Respiratory Cell and Molecular Biology 2016, 55, 47–57. [Google Scholar] [CrossRef]
- McMurtry, M.S.; Bonnet, S.; Wu, X.; Dyck, J.R.B.; Haromy, A.; Hashimoto, K.; Michelakis, E.D. Dichloroacetate Prevents and Reverses Pulmonary Hypertension by Inducing Pulmonary Artery Smooth Muscle Cell Apoptosis. Circulation Research 2004, 95, 830–840. [Google Scholar] [CrossRef]
- Guignabert, C.; Tu, L.; Izikki, M.; Dewachter, L.; Zadigue, P.; Humbert, M.; Adnot, S.; Fadel, E.; Eddahibi, S. Dichloroacetate treatment partially regresses established pulmonary hypertension in mice with SM22α-targeted overexpression of the serotonin transporter. The FASEB Journal 2009, 23, 4135–4147. [Google Scholar] [CrossRef]
- Zeng, W.-J.; Xiong, C.-M.; Zhao, L.; Shan, G.-L.; Liu, Z.-H.; Xue, F.; Gu, Q.; Ni, X.-H.; Zhao, Z.-H.; Cheng, X.-S.; et al. Atorvastatin in Pulmonary Arterial Hypertension (APATH) study. European Respiratory Journal 2012, 40, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Haslem, L.; Hays, J.M.; Hays, F.A. p66Shc in Cardiovascular Pathology. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S. P66Shc and vascular endothelial function. Bioscience Reports 2019, 39. [Google Scholar] [CrossRef] [PubMed]
- Freund-Michel, V.; Guibert, C.; Dubois, M.; Courtois, A.; Marthan, R.; Savineau, J.-P.; Muller, B. Reactive oxygen species as therapeutic targets in pulmonary hypertension. Therapeutic Advances in Respiratory Disease 2013, 7, 175–200. [Google Scholar] [CrossRef]
- Liu, J.; Wang, L.; Ge, L.; Sun, W.; Song, Z.; Lu, X.; Jin, C.; Wu, S.; Yang, J. Lanthanum decreased VAPB-PTPP51, BAP31-FIS1, and MFN2-MFN1 expression of mitochondria-associated membranes and induced abnormal autophagy in rat hippocampus. Food and Chemical Toxicology 2022, 161. [Google Scholar] [CrossRef]
- Bononi, A.; Bonora, M.; Marchi, S.; Missiroli, S.; Poletti, F.; Giorgi, C.; Pandolfi, P.P.; Pinton, P. Identification of PTEN at the ER and MAMs and its regulation of Ca2+ signaling and apoptosis in a protein phosphatase-dependent manner. Cell Death & Differentiation 2013, 20, 1631–1643. [Google Scholar] [CrossRef]
- Williams, A.; Hayashi, T.; Wolozny, D.; Yin, B.; Su, T.-C.; Betenbaugh, M.J.; Su, T.-P. The non-apoptotic action of Bcl-xL: regulating Ca2+ signaling and bioenergetics at the ER-mitochondrion interface. Journal of Bioenergetics and Biomembranes 2016, 48, 211–225. [Google Scholar] [CrossRef]
- Lalier, L.; Mignard, V.; Joalland, M.-P.; Lanoé, D.; Cartron, P.-F.; Manon, S.; Vallette, F.M. TOM20-mediated transfer of Bcl2 from ER to MAM and mitochondria upon induction of apoptosis. Cell Death & Disease 2021, 12. [Google Scholar] [CrossRef]
- Lee, G.-H.; Lee, H.-Y.; Li, B.; Kim, H.-R.; Chae, H.-J. Bax Inhibitor-1-Mediated Inhibition of Mitochondrial Ca2+ Intake Regulates Mitochondrial Permeability Transition Pore Opening and Cell Death. Scientific Reports 2014, 4. [Google Scholar] [CrossRef]
- Takeda, K.; Nagashima, S.; Shiiba, I.; Uda, A.; Tokuyama, T.; Ito, N.; Fukuda, T.; Matsushita, N.; Ishido, S.; Iwawaki, T.; et al. MITOL prevents ER stress-induced apoptosis by IRE 1α ubiquitylation at ER–mitochondria contact sites. The EMBO Journal 2019, 38. [Google Scholar] [CrossRef]
- Yang, H.; Niemeijer, M.; van de Water, B.; Beltman, J.B. ATF6 Is a Critical Determinant of CHOP Dynamics during the Unfolded Protein Response. iScience 2020, 23. [Google Scholar] [CrossRef] [PubMed]
- Misaka, T.; Murakawa, T.; Nishida, K.; Omori, Y.; Taneike, M.; Omiya, S.; Molenaar, C.; Uno, Y.; Yamaguchi, O.; Takeda, J.; et al. FKBP8 protects the heart from hemodynamic stress by preventing the accumulation of misfolded proteins and endoplasmic reticulum-associated apoptosis in mice. Journal of Molecular and Cellular Cardiology 2018, 114, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Giorgi, C.; Wieckowski, M.R.; Paolo Pandolfi, P.; Pinton, P. Mitochondria associated membranes (MAMs) as critical hubs for apoptosis. Communicative & Integrative Biology 2014, 4, 334–335. [Google Scholar] [CrossRef]
- Patergnani, S.; Missiroli, S.; Marchi, S.; Giorgi, C. Mitochondria-Associated Endoplasmic Reticulum Membranes Microenvironment: Targeting Autophagic and Apoptotic Pathways in Cancer Therapy. Frontiers in Oncology 2015, 5. [Google Scholar] [CrossRef]
- Kurakula, K.; Smolders, V.F.E.D.; Tura-Ceide, O.; Jukema, J.W.; Quax, P.H.A.; Goumans, M.-J. Endothelial Dysfunction in Pulmonary Hypertension: Cause or Consequence? Biomedicines 2021, 9. [Google Scholar] [CrossRef]
- Wang, C.; Dai, X.; Wu, S.; Xu, W.; Song, P.; Huang, K.; Zou, M.-H. FUNDC1-dependent mitochondria-associated endoplasmic reticulum membranes are involved in angiogenesis and neoangiogenesis. Nature Communications 2021, 12. [Google Scholar] [CrossRef]
- Li, T.; Li, S.; Feng, Y.; Zeng, X.; Dong, S.; Li, J.; Zha, L.; Luo, H.; Zhao, L.; Liu, B.; et al. Combination of Dichloroacetate and Atorvastatin Regulates Excessive Proliferation and Oxidative Stress in Pulmonary Arterial Hypertension Development via p38 Signaling. Oxidative Medicine and Cellular Longevity 2020, 2020, 1–15. [Google Scholar] [CrossRef]
- Pokharel, M.D.; Marciano, D.P.; Fu, P.; Franco, M.C.; Unwalla, H.; Tieu, K.; Fineman, J.R.; Wang, T.; Black, S.M. Metabolic reprogramming, oxidative stress, and pulmonary hypertension. Redox Biology 2023, 64. [Google Scholar] [CrossRef]
- Xu, D.; Hu, Y.-H.; Gou, X.; Li, F.-Y.; Yang, X.-Y.-C.; Li, Y.-M.; Chen, F. Oxidative Stress and Antioxidative Therapy in Pulmonary Arterial Hypertension. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Paulin, R.; Dromparis, P.; Sutendra, G.; Gurtu, V.; Zervopoulos, S.; Bowers, L.; Haromy, A.; Webster, L.; Provencher, S.; Bonnet, S.; Michelakis, Evangelos D. Sirtuin 3 Deficiency Is Associated with Inhibited Mitochondrial Function and Pulmonary Arterial Hypertension in Rodents and Humans. Cell Metabolism 2014, 20, 827–839. [Google Scholar] [CrossRef]
- Meng, M.; Jiang, Y.; Wang, Y.; Huo, R.; Ma, N.; Shen, X.; Chang, G. β-carotene targets IP3R/GRP75/VDAC1-MCU axis to renovate LPS-induced mitochondrial oxidative damage by regulating STIM1. Free Radical Biology and Medicine 2023, 205, 25–46. [Google Scholar] [CrossRef]
- An, P.; Wan, S.; Luo, Y.; Luo, J.; Zhang, X.; Zhou, S.; Xu, T.; He, J.; Mechanick, J.I.; Wu, W.-C.; et al. Micronutrient Supplementation to Reduce Cardiovascular Risk. Journal of the American College of Cardiology 2022, 80, 2269–2285. [Google Scholar] [CrossRef]
- Carbone, E.A.; Menculini, G.; de Filippis, R.; D’Angelo, M.; De Fazio, P.; Tortorella, A.; Steardo, L. Sleep Disturbances in Generalized Anxiety Disorder: The Role of Calcium Homeostasis Imbalance. International Journal of Environmental Research and Public Health 2023, 20. [Google Scholar] [CrossRef] [PubMed]
- Amaravadi, R.K.; Kimmelman, A.C.; Debnath, J. Targeting Autophagy in Cancer: Recent Advances and Future Directions. Cancer Discovery 2019, 9, 1167–1181. [Google Scholar] [CrossRef] [PubMed]
- Kocaturk, N.M.; Akkoc, Y.; Kig, C.; Bayraktar, O.; Gozuacik, D.; Kutlu, O. Autophagy as a molecular target for cancer treatment. European Journal of Pharmaceutical Sciences 2019, 134, 116–137. [Google Scholar] [CrossRef] [PubMed]
- Bao, C.; Liang, S.; Han, Y.; Yang, Z.; Liu, S.; Sun, Y.; Zheng, S.; Li, Y.; Wang, T.; Gu, Y.; et al. The Novel Lysosomal Autophagy Inhibitor (ROC-325) Ameliorates Experimental Pulmonary Hypertension. Hypertension 2023, 80, 70–83. [Google Scholar] [CrossRef]
- Houssaini A, A.S. mTOR_ A Key to Both Pulmonary Vessel Remodeling and Right Ventricular Function in Pulmonary Arterial Hypertension. Am J Respir Cell Mol Biol 2017, 57, 509–511. [Google Scholar] [CrossRef]
- Zhao, Q.; Song, P.; Zou, M.-H. AMPK and Pulmonary Hypertension: Crossroads Between Vasoconstriction and Vascular Remodeling. Frontiers in Cell and Developmental Biology 2021, 9. [Google Scholar] [CrossRef]
- McNair, B.D.; Polson, S.M.; Shorthill, S.K.; Yusifov, A.; Walker, L.A.; Weiser-Evans, M.C.M.; Kovacs, E.J.; Bruns, D.R. Metformin protects against pulmonary hypertension-induced right ventricular dysfunction in an age- and sex-specific manner independent of cardiac AMPK. American Journal of Physiology-Heart and Circulatory Physiology 2023, 325, H278–H292. [Google Scholar] [CrossRef]
- Zhang, J.; Tao, J.; Zhou, Z.; Pei, W.; Xiao, Y.; Guo, Y.; Gao, J.; Jiang, C.; Dai, L.; Zhang, G.; Tan, C. Current research on mitochondria-associated membranes in cardiovascular diseases (Review). Molecular Medicine Reports 2025, 31, 1–17. [Google Scholar] [CrossRef]
- Li, Z.; Hu, O.; Xu, S.; Lin, C.; Yu, W.; Ma, D.; Lu, J.; Liu, P. The SIRT3-ATAD3A axis regulates MAM dynamics and mitochondrial calcium homeostasis in cardiac hypertrophy. International Journal of Biological Sciences 2024, 20, 831–847. [Google Scholar] [CrossRef]
- Wang, H.; Yi, X.; Guo, S.; Wang, S.; Ma, J.; Zhao, T.; Shi, Q.; Tian, Y.; Wang, H.; Jia, L.; et al. The XBP1‒MARCH5‒MFN2 Axis Confers Endoplasmic Reticulum Stress Resistance by Coordinating Mitochondrial Fission and Mitophagy in Melanoma. Journal of Investigative Dermatology 2021, 141, 2932–2943.e2912. [Google Scholar] [CrossRef]
- Kalarikkal, M.; Saikia, R.; Oliveira, L.; Bhorkar, Y.; Lonare, A.; Varshney, P.; Dhamale, P.; Majumdar, A.; Joseph, J. Nup358 restricts ER-mitochondria connectivity by modulating mTORC2/Akt/GSK3β signalling. EMBO Reports 2024, 25, 4226–4251. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




