Submitted:
11 February 2026
Posted:
12 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
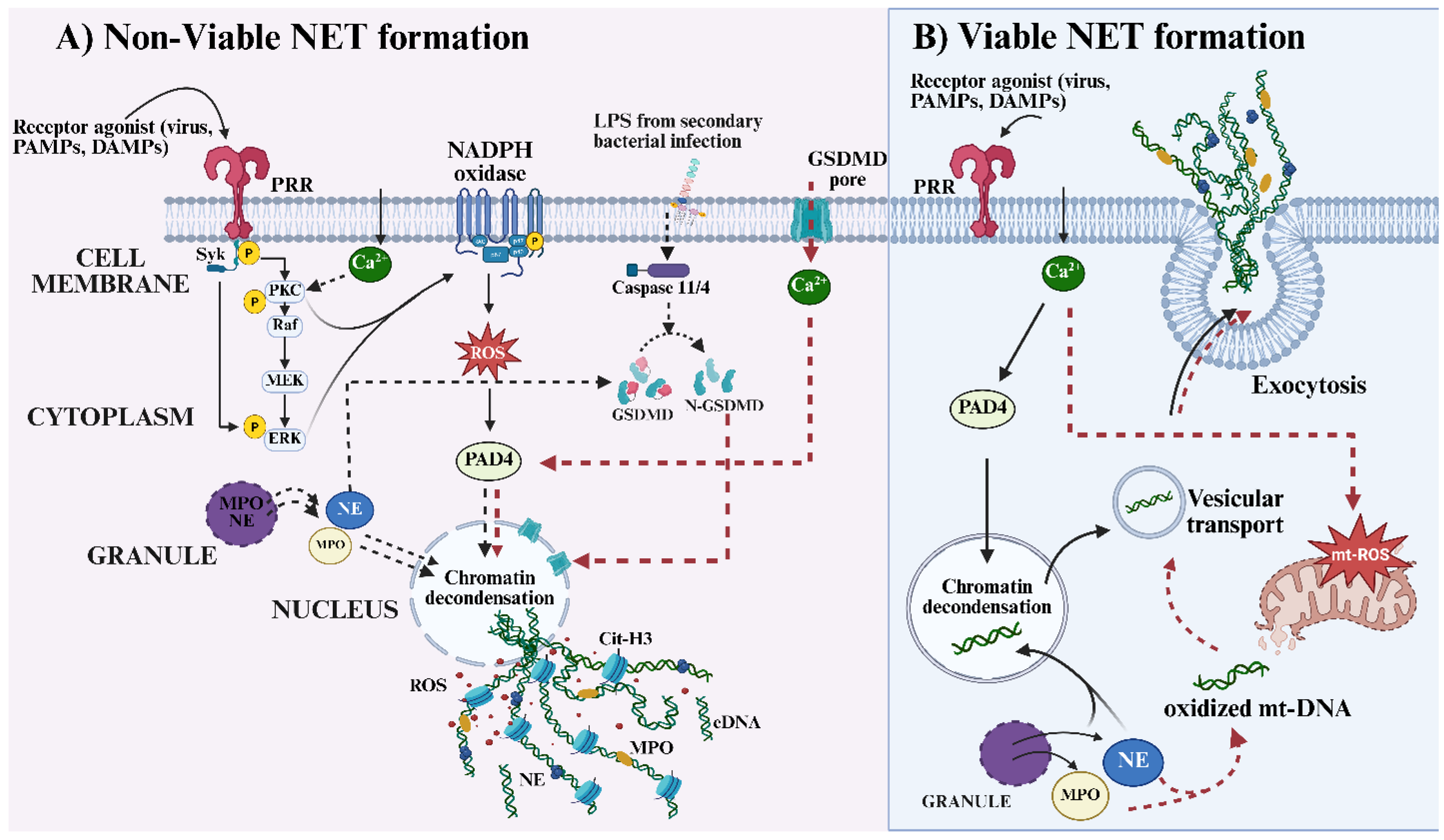
2. Viral Induction and Modulation of Neutrophil Extracellular Trap (NET) Formation
2.1. Viral Evasion and Exploitation of Neutrophil Extracellular Traps
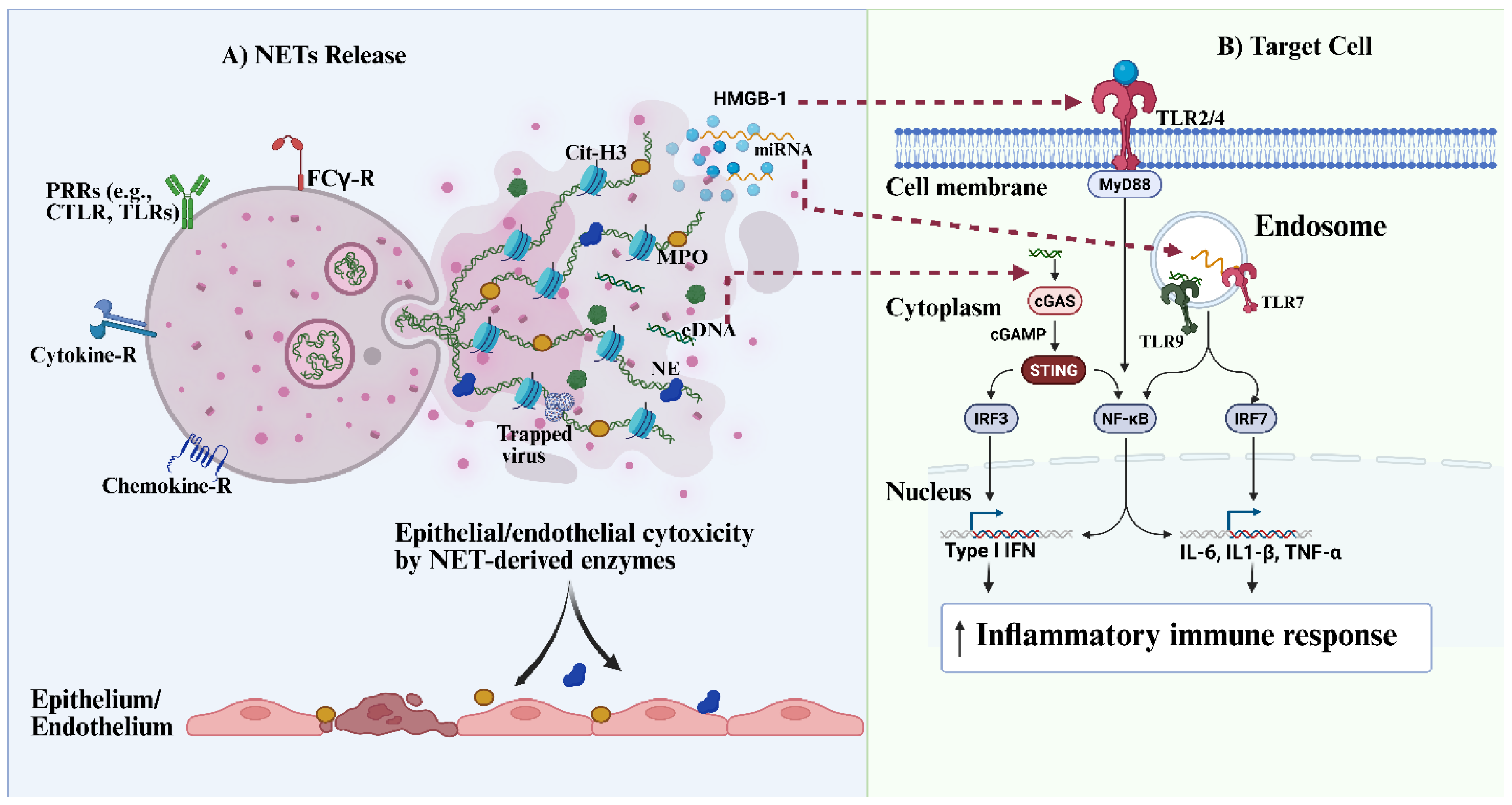
3. Consequences of Dysregulated NET Formation and Persistent NET Components During Viral Infection
3.1. Mechanistic Insights into NETs-Induced Systemic and Organ Dysfunction
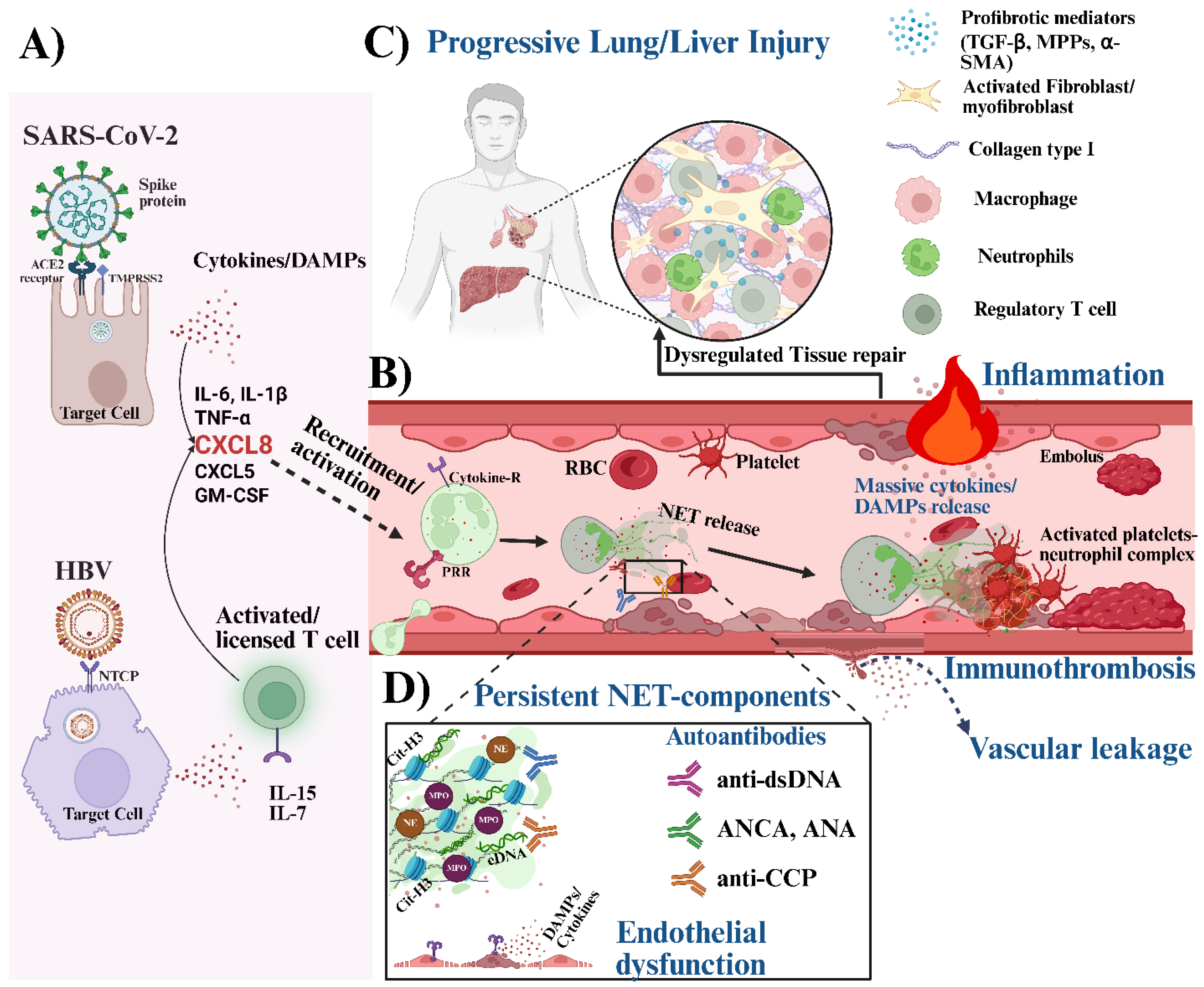
4. NETs as Drivers of Viral Immunopathology and Post-Virus Syndromes
4.1. Immunothrombosis, Endothelial Injury, and Vascular Leakage
4.2. Neutrophils in Hyperinflammation and Progressive Lung/Liver Injury
4.3. Autoinflammatory and Autoimmune Sequelae
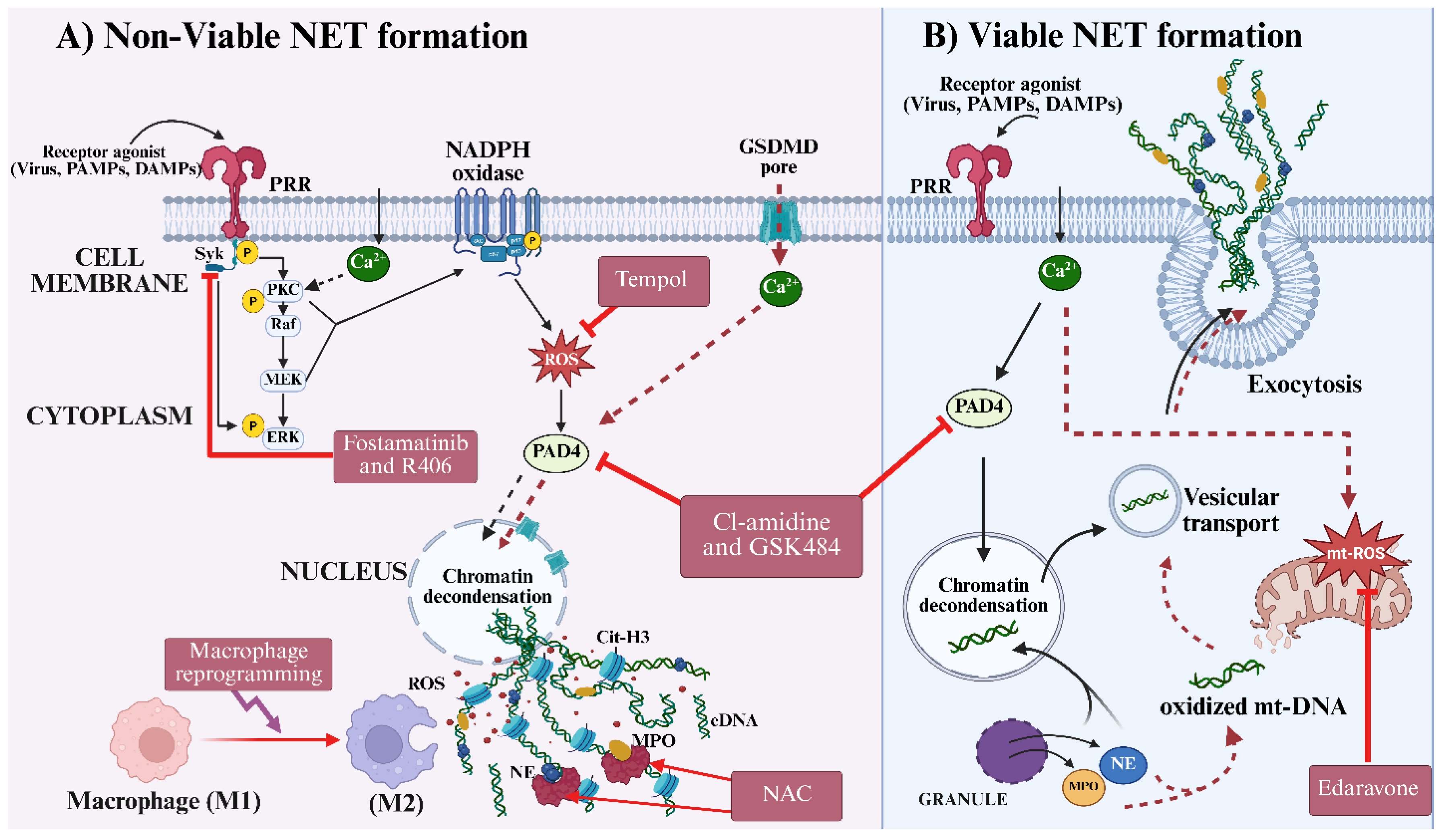
5. Therapeutic Strategies Targeting NET Dysregulation in Viral Infections
5.1. Targeting NET Formation
5.1.1. Peptidylarginine Deiminase 4 (PAD4)
5.1.2. Reactive Oxygen Species (ROS) Scavengers
5.1.3. Spleen Tyrosine Kinase (SYK) and Mitogen-Activated Protein Kinase (MAPK) Inhibitors
5.2. Enhancing NET Clearance
5.2.1. Deoxyribonuclease I (DNase I) Therapy
5.2.2. Macrophage Reprogramming
5.3. Lessons from COVID-19 and Hepatitis B Virus Clinical Trials
6. Conclusion and Future Perspectives
Author Contributions
Funding Statement
Data Sharing Statement
Disclosure
Abbreviations
References
- Vorobjeva, N.V.; Chernyak, B.V. NETosis: Molecular Mechanisms, Role in Physiology and Pathology. Biochemistry (Mosc) 2020, 85, 1178–1190. [Google Scholar] [CrossRef]
- He, Y.; Yang, F.Y.; Sun, E.W. Neutrophil Extracellular Traps in Autoimmune Diseases. Chin Med J (Engl) 2018, 131, 1513–1519. [Google Scholar] [CrossRef]
- Krinsky, N.; Sizikov, S.; Nissim, S.; Dror, A.; Sas, A.; Prinz, H.; Pri-Or, E.; Perek, S.; Raz-Pasteur, A.; Lejbkowicz, I.; et al. NETosis induction reflects COVID-19 severity and long COVID: insights from a 2-center patient cohort study in Israel. J. Thromb. Haemost. 2023, 21, 2569–2584. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, V.; Reichard, U.; Goosmann, C.; Fauler, B.; Uhlemann, Y.; Weiss, D.S.; Weinrauch, Y.; Zychlinsky, A. Neutrophil extracellular traps kill bacteria. Science 2004, 303, 1532–1535. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Kim, S.J.; Lei, Y.; Wang, S.; Wang, H.; Huang, H.; Zhang, H.; Tsung, A. Neutrophil extracellular traps in homeostasis and disease. Signal Transduct. Target. Ther. 2024, 9, 235. [Google Scholar] [CrossRef]
- Lebourgeois, S.; David, A.; Chenane, H.R.; Granger, V.; Menidjel, R.; Fidouh, N.; Noël, B.; Delelis, O.; Richetta, C.; Charpentier, C.; et al. Differential activation of human neutrophils by SARS-CoV-2 variants of concern. Front. Immunol. 2022, 13, 1010140. [Google Scholar] [CrossRef]
- Zhan, X.; Wu, R.; Kong, X.H.; You, Y.; He, K.; Sun, X.Y.; Huang, Y.; Chen, W.X.; Duan, L. Elevated neutrophil extracellular traps by HBV-mediated S100A9-TLR4/RAGE-ROS cascade facilitate the growth and metastasis of hepatocellular carcinoma. Cancer Commun (Lond) 2023, 43, 225–245. [Google Scholar] [CrossRef]
- Wei, W.C.; Liaw, C.C.; Tsai, K.C.; Chiou, C.T.; Tseng, Y.H.; Chiou, W.F.; Lin, Y.C.; Tsai, C.I.; Lin, C.S.; Lin, C.S.; et al. Targeting spike protein-induced TLR/NET axis by COVID-19 therapeutic NRICM102 ameliorates pulmonary embolism and fibrosis. Pharmacol. Res. 2022, 184, 106424. [Google Scholar] [CrossRef]
- Youn, Y.J.; Lee, Y.B.; Kim, S.H.; Jin, H.K.; Bae, J.S.; Hong, C.W. Nucleocapsid and Spike Proteins of SARS-CoV-2 Drive Neutrophil Extracellular Trap Formation. Immune Netw. 2021, 21, e16. [Google Scholar] [CrossRef]
- Chan, L.L.Y.; Nicholls, J.M.; Peiris, J.S.M.; Lau, Y.L.; Chan, M.C.W.; Chan, R.W.Y. Host DNA released by NETosis in neutrophils exposed to seasonal H1N1 and highly pathogenic H5N1 influenza viruses. Respir. Res. 2020, 21, 160. [Google Scholar] [CrossRef] [PubMed]
- Muraro, S.P.; De Souza, G.F.; Gallo, S.W.; Da Silva, B.K.; De Oliveira, S.D.; Vinolo, M.A.R.; Saraiva, E.M.; Porto, B.N. Respiratory Syncytial Virus induces the classical ROS-dependent NETosis through PAD-4 and necroptosis pathways activation. Sci. Rep. 2018, 8, 14166. [Google Scholar] [CrossRef]
- Backman, E.; Gröning, R.; Lind, A.; Granvik, C.; Eilers, H.; Lange, A.; Ahlm, C.; Cajander, S.; Forsell, M.N.E.; Normark, J.; et al. Elevated Plasma Levels of NET Components in Men with Severe COVID-19 Correlates to Increased Amounts of IL-18. Eur. J. Immunol. 2025, 55, e202451546. [Google Scholar] [CrossRef]
- Tanaka, A.; Wakayama, K.; Fukuda, Y.; Ohta, S.; Homma, T.; Ando, K.; Nishihara, Y.; Nakano, R.; Zhao, J.; Suzuki, Y.; et al. Increased levels of circulating cell-free DNA in COVID-19 patients with respiratory failure. Sci. Rep. 2024, 14, 17399. [Google Scholar] [CrossRef]
- Mahmoodpoor, A.; Mohammadzadeh, M.; Asghari, R.; Tagizadeh, M.; Iranpour, A.; Rezayi, M.; Pahnvar, A.J.; Emamalizadeh, B.; Sohrabifar, N.; Kazeminasab, S. Prognostic potential of circulating cell free mitochondrial DNA levels in COVID-19 patients. Mol. Biol. Rep. 2023, 50, 10249–10255. [Google Scholar] [CrossRef]
- Asaba, C.N.; Bitazar, R.; Labonté, P.; Bukong, T.N. Bronchoalveolar lavage single-cell transcriptomics reveals immune dysregulations driving COVID-19 severity. PLoS One 2025, 20, e0309880. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, C.; Wang, H.; Zhou, Y.; Yu, Y.; Liu, H.; Gu, C. Research progress on neutrophil extracellular traps and hepatitis-to-hepatocellular carcinoma transformation (Review). Oncol. Lett. 2026, 31, 33. [Google Scholar] [CrossRef]
- Chen, J.; He, R.; Luo, J.; Yan, S.; Zhu, W.; Liu, S. Neutrophil Extracellular Traps in Viral Infections. Pathogens 2025, 14. [Google Scholar] [CrossRef]
- Monsalve, D.M.; Acosta-Ampudia, Y.; Acosta, N.G.; Celis-Andrade, M.; Şahin, A.; Yilmaz, A.M.; Shoenfeld, Y.; Ramírez-Santana, C. NETosis: A key player in autoimmunity, COVID-19, and long COVID. J. Transl. Autoimmun. 2025, 10, 100280. [Google Scholar] [CrossRef] [PubMed]
- Heneghan, M.A.; Lohse, A.W. Update in clinical science: Autoimmune hepatitis. J. Hepatol. 2025, 82, 926–937. [Google Scholar] [CrossRef] [PubMed]
- Domerecka, W.; Homa-Mlak, I.; Mlak, R.; Michalak, A.; Wilińska, A.; Kowalska-Kępczyńska, A.; Dreher, P.; Cichoż-Lach, H.; Małecka-Massalska, T. Indicator of Inflammation and NETosis-Low-Density Granulocytes as a Biomarker of Autoimmune Hepatitis. J. Clin. Med. 2022, 11. [Google Scholar] [CrossRef]
- Morrissey, S.M.; Geller, A.E.; Hu, X.; Tieri, D.; Ding, C.; Klaes, C.K.; Cooke, E.A.; Woeste, M.R.; Martin, Z.C.; Chen, O.; et al. A specific low-density neutrophil population correlates with hypercoagulation and disease severity in hospitalized COVID-19 patients. JCI Insight 2021, 6. [Google Scholar] [CrossRef]
- Dwivedi, A.; Ui Mhaonaigh, A.; Carroll, M.; Khosravi, B.; Batten, I.; Ballantine, R.S.; Hendricken Phelan, S.; O'Doherty, L.; George, A.M.; Sui, J.; et al. Emergence of dysfunctional neutrophils with a defect in arginase-1 release in severe COVID-19. JCI Insight 2024, 9. [Google Scholar] [CrossRef]
- Tay, S.H.; Celhar, T.; Fairhurst, A.M. Low-Density Neutrophils in Systemic Lupus Erythematosus. Arthritis Rheumatol. 2020, 72, 1587–1595. [Google Scholar] [CrossRef]
- Bukong, T.N.; Cho, Y.; Iracheta-Vellve, A.; Saha, B.; Lowe, P.; Adejumo, A.; Furi, I.; Ambade, A.; Gyongyosi, B.; Catalano, D.; et al. Abnormal neutrophil traps and impaired efferocytosis contribute to liver injury and sepsis severity after binge alcohol use. J. Hepatol. 2018, 69, 1145–1154. [Google Scholar] [CrossRef]
- Wu, W.; Sun, S.; Wang, Y.; Zhao, R.; Ren, H.; Li, Z.; Zhao, H.; Zhang, Y.; Sheng, J.; Chen, Z.; et al. Circulating Neutrophil Dysfunction in HBV-Related Acute-on-Chronic Liver Failure. Front. Immunol. 2021, 12, 620365. [Google Scholar] [CrossRef]
- Xue, J.; Dong, P.; Pida, M.; Ma, K.; Zhao, H.; Chen, J.; Wu, X. Evaluation of peripheral blood neutrophil extracellular traps as fibrotic markers in patients with chronic hepatitis B. Arab. J. Gastroenterol. 2025. [Google Scholar] [CrossRef]
- Dhawan, U.K.; Vartak, T.; Englert, H.; Russo, S.; Vasconcellos, L.R.C.; Singhal, A.; Chakraborty, R.; Bhagat, K.K.; McDonnell, C.; Connolly, M.; et al. Macrophage DNases Limit Neutrophil Extracellular Trap-Mediated Defective Efferocytosis in Atherosclerosis. Circ. Res. 2025, 137, 1255–1275. [Google Scholar] [CrossRef]
- Shahzad, A.; Ni, Y.; Yang, Y.; Liu, W.; Teng, Z.; Bai, H.; Liu, X.; Sun, Y.; Xia, J.; Cui, K.; et al. Neutrophil Extracellular Traps (NETs) in health and disease. Mol. Biomed. 2025, 6, 130. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Liu, J.; Yan, N. Endothelial Dysfunction Induced by Extracellular Neutrophil Traps Plays Important Role in the Occurrence and Treatment of Extracellular Neutrophil Traps-Related Disease. Int. J. Mol. Sci. 2022, 23. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ye, Y.; Peng, K.; Zeng, Z.; Chen, L.; Zeng, Y. Histones: The critical players in innate immunity. Front. Immunol. 2022, 13, 1030610. [Google Scholar] [CrossRef]
- Apel, F.; Andreeva, L.; Knackstedt, L.S.; Streeck, R.; Frese, C.K.; Goosmann, C.; Hopfner, K.P.; Zychlinsky, A. The cytosolic DNA sensor cGAS recognizes neutrophil extracellular traps. Sci. Signal 2021, 14. [Google Scholar] [CrossRef] [PubMed]
- Hong, W.; Yang, J.; Zou, J.; Bi, Z.; He, C.; Lei, H.; He, X.; Li, X.; Alu, A.; Ren, W.; et al. Histones released by NETosis enhance the infectivity of SARS-CoV-2 by bridging the spike protein subunit 2 and sialic acid on host cells. Cell Mol. Immunol. 2022, 19, 577–587. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Wang, Y.; Xiong, Q.; He, J.; Wang, H.; Islam, M.; Zhou, X.; Kim, A.; Zhang, H.; Huang, H.; et al. Neutrophil extracellular traps promote MASH fibrosis by metabolic reprogramming of HSC. Hepatology 2025, 81, 947–961. [Google Scholar] [CrossRef]
- Shen, J.; Huang, S.; Wang, Y.; Wang, Q.; Lin, S.; Guan, W.; Gong, Y.; Si, Y.; Zhao, M.; Zhou, H.; et al. PAD4+ neutrophils promote hepatic stellate cell activation and accelerate MASH fibrosis progression viaNET-DNA/TAOK1/MAPK pathways. JCI Insight 2026, 11. [Google Scholar] [CrossRef] [PubMed]
- Mak, K.M.; Shekhar, A.C.; Ding, S.Y. Neutrophil extracellular traps mediate pathophysiology of hepatic cells during liver injury. Anat Rec (Hoboken) 2026, 309, 214–234. [Google Scholar] [CrossRef]
- Deinhardt-Emmer, S.; Böttcher, S.; Häring, C.; Giebeler, L.; Henke, A.; Zell, R.; Jungwirth, J.; Jordan, P.M.; Werz, O.; Hornung, F.; et al. SARS-CoV-2 causes severe epithelial inflammation and barrier dysfunction. J. Virol. 2021, 95. [Google Scholar] [CrossRef]
- Cai, J.; Ma, W.; Wang, X.; Chang, H.; Wei, Z.; Li, J.; Zeng, M. The spike protein of SARS-CoV-2 induces inflammation and EMT of lung epithelial cells and fibroblasts through the upregulation of GADD45A. Open Med (Wars) 2023, 18, 20230779. [Google Scholar] [CrossRef]
- Chen, H.; Liu, W.; Wang, Y.; Liu, D.; Zhao, L.; Yu, J. SARS-CoV-2 activates lung epithelial cell proinflammatory signaling and leads to immune dysregulation in COVID-19 patients. EBioMedicine 2021, 70, 103500. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, H.K. Re-analysis of Single Cell Transcriptome Reveals That the NR3C1-CXCL8-Neutrophil Axis Determines the Severity of COVID-19. Front. Immunol. 2020, 11, 2145. [Google Scholar] [CrossRef]
- Melero, I.; Villalba-Esparza, M.; Recalde-Zamacona, B.; Jiménez-Sánchez, D.; Teijeira, Á.; Argueta, A.; García-Tobar, L.; Álvarez-Gigli, L.; Sainz, C.; Garcia-Ros, D.; et al. Neutrophil Extracellular Traps, Local IL-8 Expression, and Cytotoxic T-Lymphocyte Response in the Lungs of Patients With Fatal COVID-19. Chest 2022, 162, 1006–1016. [Google Scholar] [CrossRef]
- Gehring, A.J.; Koh, S.; Chia, A.; Paramasivam, K.; Chew, V.S.; Ho, Z.Z.; Lee, K.H.; Maini, M.K.; Madhavan, K.; Lim, S.G.; et al. Licensing virus-specific T cells to secrete the neutrophil attracting chemokine CXCL-8 during hepatitis B virus infection. PLoS One 2011, 6, e23330. [Google Scholar] [CrossRef]
- Iwaniuk, A.; Jablonska, E. Neutrophils in Health and Disease: From Receptor Sensing to Inflammasome Activation. Int. J. Mol. Sci. 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Stegelmeier, A.A.; Darzianiazizi, M.; Hanada, K.; Sharif, S.; Wootton, S.K.; Bridle, B.W.; Karimi, K. Type I Interferon-Mediated Regulation of Antiviral Capabilities of Neutrophils. Int. J. Mol. Sci. 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Funchal, G.A.; Jaeger, N.; Czepielewski, R.S.; Machado, M.S.; Muraro, S.P.; Stein, R.T.; Bonorino, C.B.; Porto, B.N. Respiratory syncytial virus fusion protein promotes TLR-4-dependent neutrophil extracellular trap formation by human neutrophils. PLoS One 2015, 10, e0124082. [Google Scholar] [CrossRef]
- Sung, P.S.; Yang, S.P.; Peng, Y.C.; Sun, C.P.; Tao, M.H.; Hsieh, S.L. CLEC5A and TLR2 are critical in SARS-CoV-2-induced NET formation and lung inflammation. J. Biomed. Sci. 2022, 29, 52. [Google Scholar] [CrossRef] [PubMed]
- Shahrakyvahed, A.; Sanchooli, J.; Sanadgol, N.; Arababadi, M.K.; Kennedy, D. TLR9: an important molecule in the fight against hepatitis B virus. Postgrad. Med. J. 2014, 90, 396–401. [Google Scholar] [CrossRef]
- Rehwinkel, J.; Gack, M.U. RIG-I-like receptors: their regulation and roles in RNA sensing. Nat. Rev. Immunol. 2020, 20, 537–551. [Google Scholar] [CrossRef]
- Vieira, S.M.; Lemos, H.P.; Grespan, R.; Napimoga, M.H.; Dal-Secco, D.; Freitas, A.; Cunha, T.M.; Verri, W.A., Jr.; Souza-Junior, D.A.; Jamur, M.C.; et al. A crucial role for TNF-alpha in mediating neutrophil influx induced by endogenously generated or exogenous chemokines, KC/CXCL1 and LIX/CXCL5. Br. J. Pharmacol. 2009, 158, 779–789. [Google Scholar] [CrossRef]
- Worley, M.J.; Fei, K.; Lopez-Denman, A.J.; Kelleher, A.D.; Kent, S.J.; Chung, A.W. Neutrophils mediate HIV-specific antibody-dependent phagocytosis and ADCC. J. Immunol. Methods 2018, 457, 41–52. [Google Scholar] [CrossRef]
- Allen, K.C.; Warner, S.; Teague, H.L.; Ramos-Benitez, M.J.; Miao, R.; Tian, X.; Reger, R.; Burbelo, P.D.; Pang, C.W.J.; Kanthi, Y.; et al. SARS-CoV-2 Immune Complex-Mediated Neutrophil Activation. Open Forum Infect. Dis. 2025, 12, ofaf199. [Google Scholar] [CrossRef]
- Zhang, A.; Stacey, H.D.; D'Agostino, M.R.; Tugg, Y.; Marzok, A.; Miller, M.S. Beyond neutralization: Fc-dependent antibody effector functions in SARS-CoV-2 infection. Nat. Rev. Immunol. 2023, 23, 381–396. [Google Scholar] [CrossRef]
- Strich, J.R.; Ramos-Benitez, M.J.; Randazzo, D.; Stein, S.R.; Babyak, A.; Davey, R.T.; Suffredini, A.F.; Childs, R.W.; Chertow, D.S. Fostamatinib Inhibits Neutrophils Extracellular Traps Induced by COVID-19 Patient Plasma: A Potential Therapeutic. J. Infect. Dis. 2021, 223, 981–984. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, L.; Lu, Y.; Guo, C.; Zhang, T.; Zhang, L. Targeting spleen tyrosine kinase (SYK): structure, mechanisms and drug discovery. Drug Discov. Today 2025, 30, 104257. [Google Scholar] [CrossRef]
- Chen, T.; Li, Y.; Sun, R.; Hu, H.; Liu, Y.; Herrmann, M.; Zhao, Y.; Muñoz, L.E. Receptor-Mediated NETosis on Neutrophils. Front. Immunol. 2021, 12, 775267. [Google Scholar] [CrossRef]
- Tatsiy, O.; McDonald, P.P. Physiological Stimuli Induce PAD4-Dependent, ROS-Independent NETosis, With Early and Late Events Controlled by Discrete Signaling Pathways. Front. Immunol. 2018, 9, 2036. [Google Scholar] [CrossRef]
- Li, Y.; Li, M.; Weigel, B.; Mall, M.; Werth, V.P.; Liu, M.L. Nuclear envelope rupture and NET formation is driven by PKCα-mediated lamin B disassembly. EMBO Rep. 2020, 21, e48779. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.W.; Monteleone, M.; Boucher, D.; Sollberger, G.; Ramnath, D.; Condon, N.D.; von Pein, J.B.; Broz, P.; Sweet, M.J.; Schroder, K. Noncanonical inflammasome signaling elicits gasdermin D-dependent neutrophil extracellular traps. Sci. Immunol. 2018, 3. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Liu, X.; Gao, Y.; Zhou, R.; Wei, M.; Dong, J.; Yan, H.; Zhao, Y. Hepatitis B Virus Inhibits Neutrophil Extracellular Trap Release by Modulating Reactive Oxygen Species Production and Autophagy. J. Immunol. 2019, 202, 805–815. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Gonzalo, I.; Menéndez-Jandula, B.; Franco-García, E.; Arévalo-Vargas, I.; Lahoz-Gil, C.; Latre, P.; Roca-Esteve, S.; Köhler, R.; López de Frutos, L.; Giraldo, P. Neutrophil extracellular traps and macrophage activation contibute to thrombosis and post-covid syndrome in SARS-CoV-2 infection. Front. Immunol. 2025, 16, 1507167. [Google Scholar] [CrossRef]
- Cho, Y.; Bukong, T.N.; Tornai, D.; Babuta, M.; Vlachos, I.S.; Kanata, E.; Catalano, D.; Szabo, G. Neutrophil extracellular traps contribute to liver damage and increase defective low-density neutrophils in alcohol-associated hepatitis. J. Hepatol. 2023, 78, 28–44. [Google Scholar] [CrossRef]
- Eddins, D.J.; Yang, J.; Kosters, A.; Giacalone, V.D.; Pechuan-Jorge, X.; Chandler, J.D.; Eum, J.; Babcock, B.R.; Dobosh, B.S.; Hernández, M.R.; et al. Transcriptional reprogramming of infiltrating neutrophils drives lung pathology in severe COVID-19 despite low viral load. Blood Adv. 2023, 7, 778–799. [Google Scholar] [CrossRef] [PubMed]
- Creusat, F.; Jouan, Y.; Gonzalez, L.; Barsac, E.; Ilango, G.; Lemoine, R.; Soulard, D.; Hankard, A.; Boisseau, C.; Guillon, A.; et al. IFN-γ primes bone marrow neutrophils to acquire regulatory functions in severe viral respiratory infections. Sci. Adv. 2024, 10, eadn3257. [Google Scholar] [CrossRef]
- Belchamber, K.B.R.; Thein, O.S.; Hazeldine, J.; Grudzinska, F.S.; Faniyi, A.A.; Hughes, M.J.; Jasper, A.E.; Yip, K.P.; Crowley, L.E.; Lugg, S.T.; et al. Dysregulated Neutrophil Phenotype and Function in Hospitalised Non-ICU COVID-19 Pneumonia. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Aubé, F.A.; Bidias, A.; Pépin, G. Who and how, DNA sensors in NETs-driven inflammation. Front. Immunol. 2023, 14, 1190177. [Google Scholar] [CrossRef]
- Zhou, Z.; Ou-Yang, C.; Chen, Q.; Ren, Z.; Guo, X.; Lei, M.; Liu, C.; Yang, X. Trafficking and effect of released DNA on cGAS-STING signaling pathway and cardiovascular disease. Front. Immunol. 2023, 14, 1287130. [Google Scholar] [CrossRef]
- Blanco, L.P.; Wang, X.; Carlucci, P.M.; Torres-Ruiz, J.J.; Romo-Tena, J.; Sun, H.W.; Hafner, M.; Kaplan, M.J. RNA Externalized by Neutrophil Extracellular Traps Promotes Inflammatory Pathways in Endothelial Cells. Arthritis Rheumatol. 2021, 73, 2282–2292. [Google Scholar] [CrossRef]
- Tsourouktsoglou, T.D.; Warnatsch, A.; Ioannou, M.; Hoving, D.; Wang, Q.; Papayannopoulos, V. Histones, DNA, and Citrullination Promote Neutrophil Extracellular Trap Inflammation by Regulating the Localization and Activation of TLR4. Cell Rep. 2020, 31, 107602. [Google Scholar] [CrossRef]
- Wang, X.; Mayorga-Flores, M.; Bien, K.G.; Bailey, A.O.; Iwahara, J. DNA-mediated proteolysis by neutrophil elastase enhances binding activities of the HMGB1 protein. J. Biol. Chem. 2022, 298, 102577. [Google Scholar] [CrossRef]
- Szturmowicz, M.; Demkow, U. Neutrophil Extracellular Traps (NETs) in Severe SARS-CoV-2 Lung Disease. Int. J. Mol. Sci. 2021, 22. [Google Scholar] [CrossRef] [PubMed]
- Giryes, S.; Bragazzi, N.L.; Bridgewood, C.; De Marco, G.; McGonagle, D. COVID-19 Vasculitis and vasculopathy-Distinct immunopathology emerging from the close juxtaposition of Type II Pneumocytes and Pulmonary Endothelial Cells. Semin. Immunopathol. 2022, 44, 375–390. [Google Scholar] [CrossRef]
- Ackermann, M.; Anders, H.J.; Bilyy, R.; Bowlin, G.L.; Daniel, C.; De Lorenzo, R.; Egeblad, M.; Henneck, T.; Hidalgo, A.; Hoffmann, M.; et al. Patients with COVID-19: in the dark-NETs of neutrophils. Cell Death Differ. 2021, 28, 3125–3139. [Google Scholar] [CrossRef]
- Caillon, A.; Trimaille, A.; Favre, J.; Jesel, L.; Morel, O.; Kauffenstein, G. Role of neutrophils, platelets, and extracellular vesicles and their interactions in COVID-19-associated thrombopathy. J. Thromb. Haemost. 2022, 20, 17–31. [Google Scholar] [CrossRef]
- Hirsch, J.; Uzun, G.; Zlamal, J.; Singh, A.; Bakchoul, T. Platelet-neutrophil interaction in COVID-19 and vaccine-induced thrombotic thrombocytopenia. Front. Immunol. 2023, 14, 1186000. [Google Scholar] [CrossRef]
- Klenk, C.; Erber, J.; Fresacher, D.; Röhrl, S.; Lengl, M.; Heim, D.; Irl, H.; Schlegel, M.; Haller, B.; Lahmer, T.; et al. Platelet aggregates detected using quantitative phase imaging associate with COVID-19 severity. Commun Med (Lond) 2023, 3, 161. [Google Scholar] [CrossRef] [PubMed]
- Hottz, E.D.; Bozza, P.T. Platelet-leukocyte interactions in COVID-19: Contributions to hypercoagulability, inflammation, and disease severity. Res. Pract. Thromb. Haemost. 2022, 6, e12709. [Google Scholar] [CrossRef]
- Lopez-Castaneda, S.; García-Larragoiti, N.; Cano-Mendez, A.; Blancas-Ayala, K.; Damian-Vázquez, G.; Perez-Medina, A.I.; Chora-Hernández, L.D.; Arean-Martínez, C.; Viveros-Sandoval, M.E. Inflammatory and Prothrombotic Biomarkers Associated With the Severity of COVID-19 Infection. Clin. Appl. Thromb. Hemost. 2021, 27, 1076029621999099. [Google Scholar] [CrossRef] [PubMed]
- Obermayer, A.; Jakob, L.M.; Haslbauer, J.D.; Matter, M.S.; Tzankov, A.; Stoiber, W. Neutrophil Extracellular Traps in Fatal COVID-19-Associated Lung Injury. Dis. Markers 2021, 2021, 5566826. [Google Scholar] [CrossRef]
- Middleton, E.A.; He, X.Y.; Denorme, F.; Campbell, R.A.; Ng, D.; Salvatore, S.P.; Mostyka, M.; Baxter-Stoltzfus, A.; Borczuk, A.C.; Loda, M.; et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood 2020, 136, 1169–1179. [Google Scholar] [CrossRef]
- Li, Z.; Jiang, Y.; Wang, S.; Qiao, Y.; Kong, X.; Jing, X. Correlation between virological response and portal vein thrombosis in patients with chronic hepatitis B. Sci. Rep. 2025, 15, 20856. [Google Scholar] [CrossRef] [PubMed]
- Lertpipopmetha, K.; Auewarakul, C.U. High incidence of hepatitis B infection-associated cirrhosis and hepatocellular carcinoma in the Southeast Asian patients with portal vein thrombosis. BMC Gastroenterol. 2011, 11, 66. [Google Scholar] [CrossRef]
- Han, M.; Liu, Y.; Cao, Y.; Zhang, Y.; Yan, Y.; Deng, S.; Yuan, X.; Xing, H.; Huang, Y.; Zhu, L. The Imbalance of Homeostasis in Neutrophil Extracellular Traps is Associated with Portal Vein Thrombosis in Patients with Decompensated Cirrhosis. J. Clin. Transl. Hepatol. 2024, 12, 1009–1019. [Google Scholar] [CrossRef]
- Muhammad, I.; Contes, K.; Bility, M.T.; Tang, Q. Chasing Virus Replication and Infection: PAMP-PRR Interaction Drives Type I Interferon Production, Which in Turn Activates ISG Expression and ISGylation. Viruses 2025, 17. [Google Scholar] [CrossRef]
- Asaba, C.N.; Ekabe, C.J.; Ayuk, H.S.; Gwanyama, B.N.; Bitazar, R.; Bukong, T.N. Interplay of TLR4 and SARS-CoV-2: Unveiling the Complex Mechanisms of Inflammation and Severity in COVID-19 Infections. J. Inflamm. Res. 2024, 17, 5077–5091. [Google Scholar] [CrossRef]
- Lupfer, C.; Malik, A.; Kanneganti, T.D. Inflammasome control of viral infection. Curr. Opin. Virol. 2015, 12, 38–46. [Google Scholar] [CrossRef]
- Narasaraju, T.; Tang, B.M.; Herrmann, M.; Muller, S.; Chow, V.T.K.; Radic, M. Neutrophilia and NETopathy as Key Pathologic Drivers of Progressive Lung Impairment in Patients With COVID-19. Front. Pharmacol. 2020, 11, 870. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Shan, R.; Miller, H.; Filatov, A.; Byazrova, M.G.; Yang, L.; Liu, C. The roles of macrophages and monocytes in COVID-19 Severe Respiratory Syndrome. Cell Insight 2025, 4, 100250. [Google Scholar] [CrossRef] [PubMed]
- Khalil, B.A.; Elemam, N.M.; Maghazachi, A.A. Chemokines and chemokine receptors during COVID-19 infection. Comput. Struct. Biotechnol. J. 2021, 19, 976–988. [Google Scholar] [CrossRef]
- Zuo, Y.; Yalavarthi, S.; Shi, H.; Gockman, K.; Zuo, M.; Madison, J.A.; Blair, C.; Weber, A.; Barnes, B.J.; Egeblad, M.; et al. Neutrophil extracellular traps in COVID-19. JCI Insight 2020, 5. [Google Scholar] [CrossRef]
- Veras, F.P.; Pontelli, M.C.; Silva, C.M.; Toller-Kawahisa, J.E.; de Lima, M.; Nascimento, D.C.; Schneider, A.H.; Caetité, D.; Tavares, L.A.; Paiva, I.M.; et al. SARS-CoV-2-triggered neutrophil extracellular traps mediate COVID-19 pathology. J. Exp. Med. 2020, 217. [Google Scholar] [CrossRef] [PubMed]
- Wynn, T.A.; Vannella, K.M. Macrophages in Tissue Repair, Regeneration, and Fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef]
- Radermecker, C.; Detrembleur, N.; Guiot, J.; Cavalier, E.; Henket, M.; d'Emal, C.; Vanwinge, C.; Cataldo, D.; Oury, C.; Delvenne, P.; et al. Neutrophil extracellular traps infiltrate the lung airway, interstitial, and vascular compartments in severe COVID-19. J. Exp. Med. 2020, 217. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, D.M.; Post, C.E.; Yao, W.; Wahl, A.; Gralinski, L.E.; Liu, H.; Dang, H.; Madden, V.J.; White, K.K.; Leist, S.R.; et al. SARS-CoV-2 infection induces pro-fibrotic and pro-thrombotic foam cell formation. Nat. Microbiol. 2025, 10, 2616–2630. [Google Scholar] [CrossRef]
- Esendağli, D.; Yilmaz, A.; Akçay, Ş.; Özlü, T. Post-COVID syndrome: pulmonary complications. Turk. J. Med. Sci. 2021, 51, 3359–3371. [Google Scholar] [CrossRef]
- Andersson, U.; Ottestad, W.; Tracey, K.J. Extracellular HMGB1: a therapeutic target in severe pulmonary inflammation including COVID-19? Mol. Med. 2020, 26, 42. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, J.; Wang, J.; Hong, S.; Han, F.; Chen, J.; Chen, G. HMGB1 induces lung fibroblast to myofibroblast differentiation through NF-κB-mediated TGF-β1 release. Mol. Med. Rep. 2017, 15, 3062–3068. [Google Scholar] [CrossRef]
- Torres-Ruiz, J.; Absalón-Aguilar, A.; Nuñez-Aguirre, M.; Pérez-Fragoso, A.; Carrillo-Vázquez, D.A.; Maravillas-Montero, J.L.; Mejía-Domínguez, N.R.; Llorente, L.; Alcalá-Carmona, B.; Lira-Luna, J.; et al. Neutrophil Extracellular Traps Contribute to COVID-19 Hyperinflammation and Humoral Autoimmunity. Cells 2021, 10. [Google Scholar] [CrossRef]
- Yan, S.; Li, M.; Liu, B.; Ma, Z.; Yang, Q. Neutrophil extracellular traps and pulmonary fibrosis: an update. J Inflamm (Lond) 2023, 20, 2. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Peng, F.; Zhou, Y. Pulmonary fibrosis: A short- or long-term sequelae of severe COVID-19? Chin. Med. J. Pulm. Crit. Care Med. 2023, 1, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Kamiya, M.; Carter, H.; Espindola, M.S.; Doyle, T.J.; Lee, J.S.; Merriam, L.T.; Zhang, F.; Kawano-Dourado, L.; Sparks, J.A.; Hogaboam, C.M.; et al. Immune mechanisms in fibrotic interstitial lung disease. Cell 2024, 187, 3506–3530. [Google Scholar] [CrossRef]
- Gustine, J.N.; Jones, D. Immunopathology of Hyperinflammation in COVID-19. Am. J. Pathol. 2021, 191, 4–17. [Google Scholar] [CrossRef]
- MacDonald, L.; Alivernini, S.; Tolusso, B.; Elmesmari, A.; Somma, D.; Perniola, S.; Paglionico, A.; Petricca, L.; Bosello, S.L.; Carfì, A.; et al. COVID-19 and RA share an SPP1 myeloid pathway that drives PD-L1+ neutrophils and CD14+ monocytes. JCI Insight 2021, 6. [Google Scholar] [CrossRef] [PubMed]
- Bility, M.T.; Cheng, L.; Zhang, Z.; Luan, Y.; Li, F.; Chi, L.; Zhang, L.; Tu, Z.; Gao, Y.; Fu, Y.; et al. Hepatitis B virus infection and immunopathogenesis in a humanized mouse model: induction of human-specific liver fibrosis and M2-like macrophages. PLoS Pathog. 2014, 10, e1004032. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Guo, H.; Zhang, C.; Jin, H.; Guo, X.; Li, T. Elevated matrix metalloproteinase-9 expression is associated with COVID-19 severity: A meta-analysis. Exp. Ther. Med. 2023, 26, 545. [Google Scholar] [CrossRef]
- Castanheira, F.V.S.; Kubes, P. Neutrophils during SARS-CoV-2 infection: Friend or foe? Immunol. Rev. 2023, 314, 399–412. [Google Scholar] [CrossRef]
- Fajgenbaum, D.C.; June, C.H. Cytokine Storm. N. Engl. J. Med. 2020, 383, 2255–2273. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, P.R.S.; Alrubayyi, A.; Pring, E.; Bart, V.M.T.; Jones, R.; Coveney, C.; Lu, F.; Tellier, M.; Maleki-Toyserkani, S.; Richter, F.C.; et al. Innate immunology in COVID-19-a living review. Part II: dysregulated inflammation drives immunopathology. Oxf. Open Immunol. 2020, 1, iqaa005. [Google Scholar] [CrossRef]
- Shaath, H.; Vishnubalaji, R.; Elkord, E.; Alajez, N.M. Single-Cell Transcriptome Analysis Highlights a Role for Neutrophils and Inflammatory Macrophages in the Pathogenesis of Severe COVID-19. Cells 2020, 9. [Google Scholar] [CrossRef]
- Spengler, J.; Lugonja, B.; Ytterberg, A.J.; Zubarev, R.A.; Creese, A.J.; Pearson, M.J.; Grant, M.M.; Milward, M.; Lundberg, K.; Buckley, C.D.; et al. Release of Active Peptidyl Arginine Deiminases by Neutrophils Can Explain Production of Extracellular Citrullinated Autoantigens in Rheumatoid Arthritis Synovial Fluid. Arthritis Rheumatol. 2015, 67, 3135–3145. [Google Scholar] [CrossRef]
- Cheung, C.C.L.; Mok, C.C. Long COVID in patients with systemic lupus erythematosus: A case-control study. Lupus 2025, 34, 452–459. [Google Scholar] [CrossRef]
- Yadav, S.; Bonnes, S.L.; Gilman, E.A.; Mueller, M.R.; Collins, N.M.; Hurt, R.T.; Ganesh, R. Inflammatory Arthritis After COVID-19: A Case Series. Am. J. Case Rep. 2023, 24, e939870. [Google Scholar] [CrossRef]
- Fernandes de Souza, W.D.; Fonseca, D.M.D.; Sartori, A. COVID-19 and Multiple Sclerosis: A Complex Relationship Possibly Aggravated by Low Vitamin D Levels. Cells 2023, 12. [Google Scholar] [CrossRef]
- Du, S.N.; Chen, J.W.; Li, W.; Wang, M.C.; Mao, Y.S. Development of autoimmune thyroid disease after COVID-19 infection: case report. Front Med (Lausanne) 2024, 11, 1303855. [Google Scholar] [CrossRef]
- Heald, A.H.; Williams, R.; Jenkins, D.A.; Stewart, S.; Bakerly, N.D.; McCay, K.; Ollier, W. The prevalence of long COVID in people with diabetes mellitus-evidence from a UK cohort. EClinicalMedicine 2024, 71, 102607. [Google Scholar] [CrossRef] [PubMed]
- Rojas, M.; Rodríguez, Y.; Acosta-Ampudia, Y.; Monsalve, D.M.; Zhu, C.; Li, Q.Z.; Ramírez-Santana, C.; Anaya, J.M. Autoimmunity is a hallmark of post-COVID syndrome. J. Transl. Med. 2022, 20, 129. [Google Scholar] [CrossRef]
- Sadeghi, M.; Dehnavi, S.; Jamialahmadi, T.; Johnston, T.P.; Sahebkar, A. Neutrophil extracellular trap: A key player in the pathogenesis of autoimmune diseases. Int. Immunopharmacol. 2023, 116, 109843. [Google Scholar] [CrossRef]
- Gregorio, G.V.; Jones, H.; Choudhuri, K.; Vegnente, A.; Bortolotti, F.; Mieli-Vergani, G.; Vergani, D. Autoantibody prevalence in chronic hepatitis B virus infection: effect in interferon alfa. Hepatology 1996, 24, 520–523. [Google Scholar] [CrossRef] [PubMed]
- Mazzaro, C.; Adinolfi, L.E.; Pozzato, G.; Nevola, R.; Zanier, A.; Serraino, D.; Andreone, P.; Fenoglio, R.; Sciascia, S.; Gattei, V.; et al. Extrahepatic Manifestations of Chronic HBV Infection and the Role of Antiviral Therapy. J. Clin. Med. 2022, 11. [Google Scholar] [CrossRef]
- Maya, R.; Gershwin, M.E.; Shoenfeld, Y. Hepatitis B virus (HBV) and autoimmune disease. Clin. Rev. Allergy Immunol. 2008, 34, 85–102. [Google Scholar] [CrossRef]
- Bhagoowani, S.; Devi, U.; Munir, A.; Hasnain, U.; Iqbal, J. Antineutrophil cytoplasmic antibodies (ANCA)-associated vasculitis in chronic Hepatitis B: Unraveling the immune puzzle - a rare case report with review of literature. IDCases 2024, 38, e02100. [Google Scholar] [CrossRef] [PubMed]
- Rohrbach, A.S.; Slade, D.J.; Thompson, P.R.; Mowen, K.A. Activation of PAD4 in NET formation. Front. Immunol. 2012, 3, 360. [Google Scholar] [CrossRef]
- Liu, X.; Arfman, T.; Wichapong, K.; Reutelingsperger, C.P.M.; Voorberg, J.; Nicolaes, G.A.F. PAD4 takes charge during neutrophil activation: Impact of PAD4 mediated NET formation on immune-mediated disease. J. Thromb. Haemost. 2021, 19, 1607–1617. [Google Scholar] [CrossRef]
- Lewis, H.D.; Liddle, J.; Coote, J.E.; Atkinson, S.J.; Barker, M.D.; Bax, B.D.; Bicker, K.L.; Bingham, R.P.; Campbell, M.; Chen, Y.H.; et al. Inhibition of PAD4 activity is sufficient to disrupt mouse and human NET formation. Nat. Chem. Biol. 2015, 11, 189–191. [Google Scholar] [CrossRef]
- Katsumata, M.; Ikari, J.; Urano, A.; Suzuki, E.; Kugou, K.; Hasegawa, Y.; Tatsumi, K.; Suzuki, T. Peptidylarginine Deiminase 4 Deficiency Suppresses Neutrophil Extracellular Trap Formation and Ameliorates Elastase-Induced Emphysema in Mouse Lung. Int. J. Mol. Sci. 2025, 26. [Google Scholar] [CrossRef]
- Suzuki, M.; Ikari, J.; Anazawa, R.; Tanaka, N.; Katsumata, Y.; Shimada, A.; Suzuki, E.; Tatsumi, K. PAD4 Deficiency Improves Bleomycin-induced Neutrophil Extracellular Traps and Fibrosis in Mouse Lung. Am. J. Respir. Cell Mol. Biol. 2020, 63, 806–818. [Google Scholar] [CrossRef]
- Kijak-Boćkowska, M.; Czerwińska, J.; Owczarczyk-Saczonek, A. Peptidylarginine Deiminases: An Overview of Recent Advances in Citrullination Research. Int. J. Mol. Sci. 2025, 26. [Google Scholar] [CrossRef]
- Knight, J.S.; Subramanian, V.; O'Dell, A.A.; Yalavarthi, S.; Zhao, W.; Smith, C.K.; Hodgin, J.B.; Thompson, P.R.; Kaplan, M.J. Peptidylarginine deiminase inhibition disrupts NET formation and protects against kidney, skin and vascular disease in lupus-prone MRL/lpr mice. Ann. Rheum. Dis. 2015, 74, 2199–2206. [Google Scholar] [CrossRef]
- Zhu, D.; Lu, Y.; Wang, Y.; Wang, Y. PAD4 and Its Inhibitors in Cancer Progression and Prognosis. Pharmaceutics 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Jia, R.; Taledaohan, A.; Wang, Y.; Wang, Y. Structure-Activity Relationship of PAD4 Inhibitors and Their Role in Tumor Immunotherapy. Pharmaceutics 2024, 16. [Google Scholar] [CrossRef] [PubMed]
- Urban, C.F.; Reichard, U.; Brinkmann, V.; Zychlinsky, A. Neutrophil extracellular traps capture and kill Candida albicans yeast and hyphal forms. Cell Microbiol. 2006, 8, 668–676. [Google Scholar] [CrossRef]
- Abi Abdallah, D.S.; Lin, C.; Ball, C.J.; King, M.R.; Duhamel, G.E.; Denkers, E.Y. Toxoplasma gondii triggers release of human and mouse neutrophil extracellular traps. Infect. Immun. 2012, 80, 768–777. [Google Scholar] [CrossRef] [PubMed]
- Lopes, B.R.P.; da Silva, G.S.; de Lima Menezes, G.; de Oliveira, J.; Watanabe, A.S.A.; Porto, B.N.; da Silva, R.A.; Toledo, K.A. Serine proteases in neutrophil extracellular traps exhibit anti-Respiratory Syncytial Virus activity. Int. Immunopharmacol. 2022, 106, 108573. [Google Scholar] [CrossRef]
- Meng, W.; Paunel-Görgülü, A.; Flohé, S.; Hoffmann, A.; Witte, I.; MacKenzie, C.; Baldus, S.E.; Windolf, J.; Lögters, T.T. Depletion of neutrophil extracellular traps in vivo results in hypersusceptibility to polymicrobial sepsis in mice. Crit. Care 2012, 16, R137. [Google Scholar] [CrossRef]
- King, P.T.; Dousha, L. Neutrophil Extracellular Traps and Respiratory Disease. J. Clin. Med. 2024, 13. [Google Scholar] [CrossRef]
- Manoharan, R.R.; Zachová, K.; Buzáš, M.; Pospíšil, P.; Křupka, M.; Prasad, A. NADPH oxidase-dependent free radical generation and protein adduct formation in neutrophils. RSC Adv. 2024, 14, 24765–24780. [Google Scholar] [CrossRef]
- Azzouz, D.; Palaniyar, N. How Do ROS Induce NETosis? Oxidative DNA Damage, DNA Repair, and Chromatin Decondensation. Biomolecules 2024, 14. [Google Scholar] [CrossRef]
- Kalyanaraman, B. NAC, NAC, Knockin' on Heaven's door: Interpreting the mechanism of action of N-acetylcysteine in tumor and immune cells. Redox Biol. 2022, 57, 102497. [Google Scholar] [CrossRef] [PubMed]
- Tenório, M.; Graciliano, N.G.; Moura, F.A.; Oliveira, A.C.M.; Goulart, M.O.F. N-Acetylcysteine (NAC): Impacts on Human Health. Antioxidants 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Aldini, G.; Altomare, A.; Baron, G.; Vistoli, G.; Carini, M.; Borsani, L.; Sergio, F. N-Acetylcysteine as an antioxidant and disulphide breaking agent: the reasons why. Free Radic. Res. 2018, 52, 751–762. [Google Scholar] [CrossRef]
- Metzler, K.D.; Goosmann, C.; Lubojemska, A.; Zychlinsky, A.; Papayannopoulos, V. A myeloperoxidase-containing complex regulates neutrophil elastase release and actin dynamics during NETosis. Cell Rep. 2014, 8, 883–896. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Ren, K.; Xiang, L.; Yao, H.; Huang, Y.; Gao, Y. Efficacy of N-Acetylcysteine as an Adjuvant Therapy for Rheumatoid Arthritis: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Br. J. Hosp. Med. 2024, 85, 1–16. [Google Scholar] [CrossRef]
- Di Paola, R.; Mazzon, E.; Zito, D.; Maiere, D.; Britti, D.; Genovese, T.; Cuzzocrea, S. Effects of Tempol, a membrane-permeable radical scavenger, in a rodent model periodontitis. J. Clin. Periodontol. 2005, 32, 1062–1068. [Google Scholar] [CrossRef]
- Wilcox, C.S. Effects of tempol and redox-cycling nitroxides in models of oxidative stress. Pharmacol. Ther. 2010, 126, 119–145. [Google Scholar] [CrossRef]
- Reddan, J.; Sevilla, M.; Giblin, F.; Padgaonkar, V.; Dziedzic, D.; Leverenz, V. Tempol and deferoxamine protect cultured rabbit lens epithelial cells from H2O2 insult: insight into the mechanism of H2O2-induced injury. Lens Eye Toxic. Res. 1992, 9, 385–393. [Google Scholar]
- Laight, D.W.; Andrews, T.J.; Haj-Yehia, A.I.; Carrier, M.J.; Anggård, E.E. Microassay of superoxide anion scavenging activity in vitro. Environ. Toxicol. Pharmacol. 1997, 3, 65–68. [Google Scholar] [CrossRef] [PubMed]
- Abouzied, M.M.; Eltahir, H.M.; Taye, A.; Abdelrahman, M.S. Experimental evidence for the therapeutic potential of tempol in the treatment of acute liver injury. Mol. Cell Biochem. 2016, 411, 107–115. [Google Scholar] [CrossRef]
- Toyoda, K.; Fujii, K.; Kamouchi, M.; Nakane, H.; Arihiro, S.; Okada, Y.; Ibayashi, S.; Iida, M. Free radical scavenger, edaravone, in stroke with internal carotid artery occlusion. J. Neurol. Sci. 2004, 221, 11–17. [Google Scholar] [CrossRef]
- Cha, S.J.; Kim, K. Effects of the Edaravone, a Drug Approved for the Treatment of Amyotrophic Lateral Sclerosis, on Mitochondrial Function and Neuroprotection. Antioxidants 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, H.; Yanai, H.; Namiki, Y.; Fukatsu-Sasaki, K.; Furutani, N.; Tada, N. Neuroprotective effects of edaravone: a novel free radical scavenger in cerebrovascular injury. CNS Drug Rev. 2006, 12, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, A.; Yamamoto, Y. Edaravone, a potent free radical scavenger, reacts with peroxynitrite to produce predominantly 4-NO-edaravone. Redox Rep. 2016, 21, 98–103. [Google Scholar] [CrossRef]
- Dupré-Crochet, S.; Erard, M.; Nüβe, O. ROS production in phagocytes: why, when, and where? J. Leukoc. Biol. 2013, 94, 657–670. [Google Scholar] [CrossRef]
- Riva, D.A.; de Molina, M.C.; Rocchetta, I.; Gerhardt, E.; Coulombié, F.C.; Mersich, S.E. Oxidative stress in vero cells infected with vesicular stomatitis virus. Intervirology 2006, 49, 294–298. [Google Scholar] [CrossRef]
- Sada, K.; Takano, T.; Yanagi, S.; Yamamura, H. Structure and function of Syk protein-tyrosine kinase. J. Biochem. 2001, 130, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Y.; Huang, J.H.; Chen, W.Y.; Chan, Y.C.; Lin, C.H.; Chen, Y.C.; Liu, F.T.; Wu-Hsieh, B.A. Cell Intrinsic Galectin-3 Attenuates Neutrophil ROS-Dependent Killing of Candida by Modulating CR3 Downstream Syk Activation. Front. Immunol. 2017, 8, 48. [Google Scholar] [CrossRef] [PubMed]
- Moens, U.; Kostenko, S.; Sveinbjørnsson, B. The Role of Mitogen-Activated Protein Kinase-Activated Protein Kinases (MAPKAPKs) in Inflammation. Genes. 2013, 4, 101–133. [Google Scholar] [CrossRef]
- Cooper, N.; Ghanima, W.; Hill, Q.A.; Nicolson, P.L.; Markovtsov, V.; Kessler, C. Recent advances in understanding spleen tyrosine kinase (SYK) in human biology and disease, with a focus on fostamatinib. Platelets 2023, 34, 2131751. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Liu, J.; Sun, Y.; Hu, Y.; Cong, C.; Chen, Y.; Fang, Y. Targeting p38 MAPK signaling pathway and neutrophil extracellular traps: An important anti-inflammatory mechanism of Huangqin Qingre Chubi Capsule in rheumatoid arthritis. Int. Immunopharmacol. 2025, 148, 114112. [Google Scholar] [CrossRef]
- Jones, L.P.; Bergeron, H.C.; Martin, D.E.; Murray, J.; Sancilio, F.D.; Tripp, R.A. Probenecid Inhibits Extracellular Signal-Regulated Kinase and c-Jun N-Terminal Kinase Mitogen-Activated Protein Kinase Pathways in Regulating Respiratory Syncytial Virus Response. Int. J. Mol. Sci. 2024, 25. [Google Scholar] [CrossRef]
- Borges, L.; Pithon-Curi, T.C.; Curi, R.; Hatanaka, E. COVID-19 and Neutrophils: The Relationship between Hyperinflammation and Neutrophil Extracellular Traps. Mediators Inflamm. 2020, 2020, 8829674. [Google Scholar] [CrossRef]
- Ackermann, J.A.; Nys, J.; Schweighoffer, E.; McCleary, S.; Smithers, N.; Tybulewicz, V.L. Syk tyrosine kinase is critical for B cell antibody responses and memory B cell survival. J. Immunol. 2015, 194, 4650–4656. [Google Scholar] [CrossRef]
- Marko, A.J.; Miller, R.A.; Kelman, A.; Frauwirth, K.A. Induction of glucose metabolism in stimulated T lymphocytes is regulated by mitogen-activated protein kinase signaling. PLoS One 2010, 5, e15425. [Google Scholar] [CrossRef]
- Vendel, A.C.; Calemine-Fenaux, J.; Izrael-Tomasevic, A.; Chauhan, V.; Arnott, D.; Eaton, D.L. B and T lymphocyte attenuator regulates B cell receptor signaling by targeting Syk and BLNK. J. Immunol. 2009, 182, 1509–1517. [Google Scholar] [CrossRef]
- da Cunha, A.A.; Nuñez, N.K.; de Souza, R.G.; Moraes Vargas, M.H.; Silveira, J.S.; Antunes, G.L.; Durante Lda, S.; Porto, B.N.; Marczak, E.S.; Jones, M.H.; et al. Recombinant human deoxyribonuclease therapy improves airway resistance and reduces DNA extracellular traps in a murine acute asthma model. Exp. Lung Res. 2016, 42, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Tsokos, G.C.; Lo, M.S.; Costa Reis, P.; Sullivan, K.E. New insights into the immunopathogenesis of systemic lupus erythematosus. Nat. Rev. Rheumatol. 2016, 12, 716–730. [Google Scholar] [CrossRef]
- Tang, R.; Yin, J.; Qin, Z.; Zhang, M.; Jia, X. NETs: a new target for autoimmune disease. Front. Immunol. 2025, 16, 1646527. [Google Scholar] [CrossRef]
- Chen, X.Q.; Tu, L.; Tang, Q.; Zou, J.S.; Yun, X.; Qin, Y.H. DNase I targeted degradation of neutrophil extracellular traps to reduce the damage on IgAV rat. PLoS One 2023, 18, e0291592. [Google Scholar] [CrossRef]
- Shak, S.; Capon, D.J.; Hellmiss, R.; Marsters, S.A.; Baker, C.L. Recombinant human DNase I reduces the viscosity of cystic fibrosis sputum. Proc. Natl. Acad. Sci. U S A 1990, 87, 9188–9192. [Google Scholar] [CrossRef] [PubMed]
- Roesch, E.A.; Rahmaoui, A.; Lazarus, R.A.; Konstan, M.W. The continuing need for dornase alfa for extracellular airway DNA hydrolysis in the era of CFTR modulators. Expert. Rev. Respir. Med. 2024, 18, 677–691. [Google Scholar] [CrossRef] [PubMed]
- Toma, A.; Darwish, C.; Taylor, M.; Harlacher, J.; Darwish, R. The Use of Dornase Alfa in the Management of COVID-19-Associated Adult Respiratory Distress Syndrome. Crit. Care Res. Pract. 2021, 2021, 8881115. [Google Scholar] [CrossRef]
- Holliday, Z.M.; Earhart, A.P.; Alnijoumi, M.M.; Krvavac, A.; Allen, L.H.; Schrum, A.G. Non-Randomized Trial of Dornase Alfa for Acute Respiratory Distress Syndrome Secondary to Covid-19. Front. Immunol. 2021, 12, 714833. [Google Scholar] [CrossRef]
- Davis, J.C., Jr.; Manzi, S.; Yarboro, C.; Rairie, J.; McInnes, I.; Averthelyi, D.; Sinicropi, D.; Hale, V.G.; Balow, J.; Austin, H.; et al. Recombinant human Dnase I (rhDNase) in patients with lupus nephritis. Lupus 1999, 8, 68–76. [Google Scholar] [CrossRef]
- Yong, J.; Abrams, S.T.; Wang, G.; Toh, C.H. Cell-free histones and the cell-based model of coagulation. J. Thromb. Haemost. 2023, 21, 1724–1736. [Google Scholar] [CrossRef]
- Strasser, D.; Neumann, K.; Bergmann, H.; Marakalala, M.J.; Guler, R.; Rojowska, A.; Hopfner, K.P.; Brombacher, F.; Urlaub, H.; Baier, G.; et al. Syk kinase-coupled C-type lectin receptors engage protein kinase C-δ to elicit Card9 adaptor-mediated innate immunity. Immunity 2012, 36, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Li, K.; Chen, L.; Wang, A.; Xi, Y.; Xiao, H.; Yuan, L. Metabolic reprogramming in efferocytosis. Front. Cell Dev. Biol. 2025, 13, 1677028. [Google Scholar] [CrossRef]
- Yu, S.; Ge, H.; Li, S.; Qiu, H.J. Modulation of Macrophage Polarization by Viruses: Turning Off/On Host Antiviral Responses. Front. Microbiol. 2022, 13, 839585. [Google Scholar] [CrossRef] [PubMed]
- Smyth, T.R.; Brocke, S.; Kim, Y.H.; Christianson, C.; Kovalcik, K.D.; Pancras, J.P.; Hays, M.D.; Wu, W.; An, Z.; Jaspers, I. Human Monocyte-Derived Macrophages Demonstrate Distinct Responses to Ambient Particulate Matter in a Polarization State- and Particle Seasonality-Specific Manner. Chem. Res. Toxicol. 2025, 38, 73–90. [Google Scholar] [CrossRef]
- Li, F.; Piattini, F.; Pohlmeier, L.; Feng, Q.; Rehrauer, H.; Kopf, M. Monocyte-derived alveolar macrophages autonomously determine severe outcome of respiratory viral infection. Sci. Immunol. 2022, 7, eabj5761. [Google Scholar] [CrossRef] [PubMed]
- Valente, M.; Dentoni, M.; Bellizzi, F.; Kuris, F.; Gigli, G.L. Specialized Pro-Resolving Mediators in Neuroinflammation: Overview of Studies and Perspectives of Clinical Applications. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Kvietys, P.R.; Fakhoury, H.M.A.; Kadan, S.; Yaqinuddin, A.; Al-Mutairy, E.; Al-Kattan, K. COVID-19: Lung-Centric Immunothrombosis. Front. Cell Infect. Microbiol. 2021, 11, 679878. [Google Scholar] [CrossRef]
- Lax, S.F.; Skok, K.; Zechner, P.; Kessler, H.H.; Kaufmann, N.; Koelblinger, C.; Vander, K.; Bargfrieder, U.; Trauner, M. Pulmonary Arterial Thrombosis in COVID-19 With Fatal Outcome: Results From a Prospective, Single-Center, Clinicopathologic Case Series. Ann. Intern. Med. 2020, 173, 350–361. [Google Scholar] [CrossRef]
- Ackermann, M.; Verleden, S.E.; Kuehnel, M.; Haverich, A.; Welte, T.; Laenger, F.; Vanstapel, A.; Werlein, C.; Stark, H.; Tzankov, A.; et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. 2020, 383, 120–128. [Google Scholar] [CrossRef]
- Jing, H.; Chen, X.; Zhang, S.; Liu, H.; Zhang, C.; Du, J.; Li, Y.; Wu, X.; Li, M.; Xiang, M.; et al. Neutrophil extracellular traps (NETs): the role of inflammation and coagulation in COVID-19. Am. J. Transl. Res. 2021, 13, 8575–8588. [Google Scholar]
- Zhu, Y.; Chen, X.; Liu, X. NETosis and Neutrophil Extracellular Traps in COVID-19: Immunothrombosis and Beyond. Front. Immunol. 2022, 13, 838011. [Google Scholar] [CrossRef] [PubMed]
- Porter, J.C.; Inshaw, J.; Solis, V.J.; Denneny, E.; Evans, R.; Temkin, M.I.; De Vasconcelos, N.; Aramburu, I.V.; Hoving, D.; Basire, D.; et al. Anti-inflammatory therapy with nebulized dornase alfa for severe COVID-19 pneumonia: a randomized unblinded trial. Elife 2024, 12. [Google Scholar] [CrossRef]
- Gregoire, C.; Di Meglio, L.; Le Cossec, C.; Ho-Tin-Noé, B.; Nomenjanahary, M.S.; Guillaume, J.; Hamdani, M.; Losser, M.R.; Lambiotte, F.; Le Tacon, S.; et al. Multicenter randomized trial assessing efficacy and safety of aerosolized dornase Alfa in COVID-19 ARDS. Sci. Rep. 2025, 15, 36876. [Google Scholar] [CrossRef]
- Åkesson, P.; Mellhammar, L.; Rasmussen, M.; Inghammar, M.; Jesperson, S.; Månsson, F.; Economou Lundeberg, E.; Walles, J.; Wallberg, M.; Frigyesi, A.; et al. Aerosolized Dornase Alfa (DNase I) for the Treatment of Severe Respiratory Failure in COVID-19: A Randomized Controlled Trial. Open Forum Infect. Dis. 2025, 12, ofaf246. [Google Scholar] [CrossRef]
- Ntinopoulou, M.; Konstantinidis, T.; Chalkidou, A.; Papagianni, E.; Skeva, A.; Panopoulou, M.; Chrysanthopoulou, A. IL-1b-Bearing NETs: Bridging Inflammation to Early Cirrhosis in Hepatitis B. Int. J. Mol. Sci. 2025, 26. [Google Scholar] [CrossRef]
- Li, X.; Gao, Q.; Wu, W.; Hai, S.; Hu, J.; You, J.; Huang, D.; Wang, H.; Wu, D.; Han, M.; et al. FGL2-MCOLN3-Autophagy Axis-Triggered Neutrophil Extracellular Traps Exacerbate Liver Injury in Fulminant Viral Hepatitis. Cell Mol. Gastroenterol. Hepatol. 2022, 14, 1077–1101. [Google Scholar] [CrossRef] [PubMed]
- EASL Clinical Practice Guidelines on the management of hepatitis B virus infection. J. Hepatol. 2025, 83, 502–583. [CrossRef]
- Pol, S.; Lampertico, P. First-line treatment of chronic hepatitis B with entecavir or tenofovir in 'real-life' settings: from clinical trials to clinical practice. J. Viral Hepat. 2012, 19, 377–386. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



