Submitted:
10 February 2026
Posted:
11 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Epidemiology
3. Aetiology and Pathogenesis
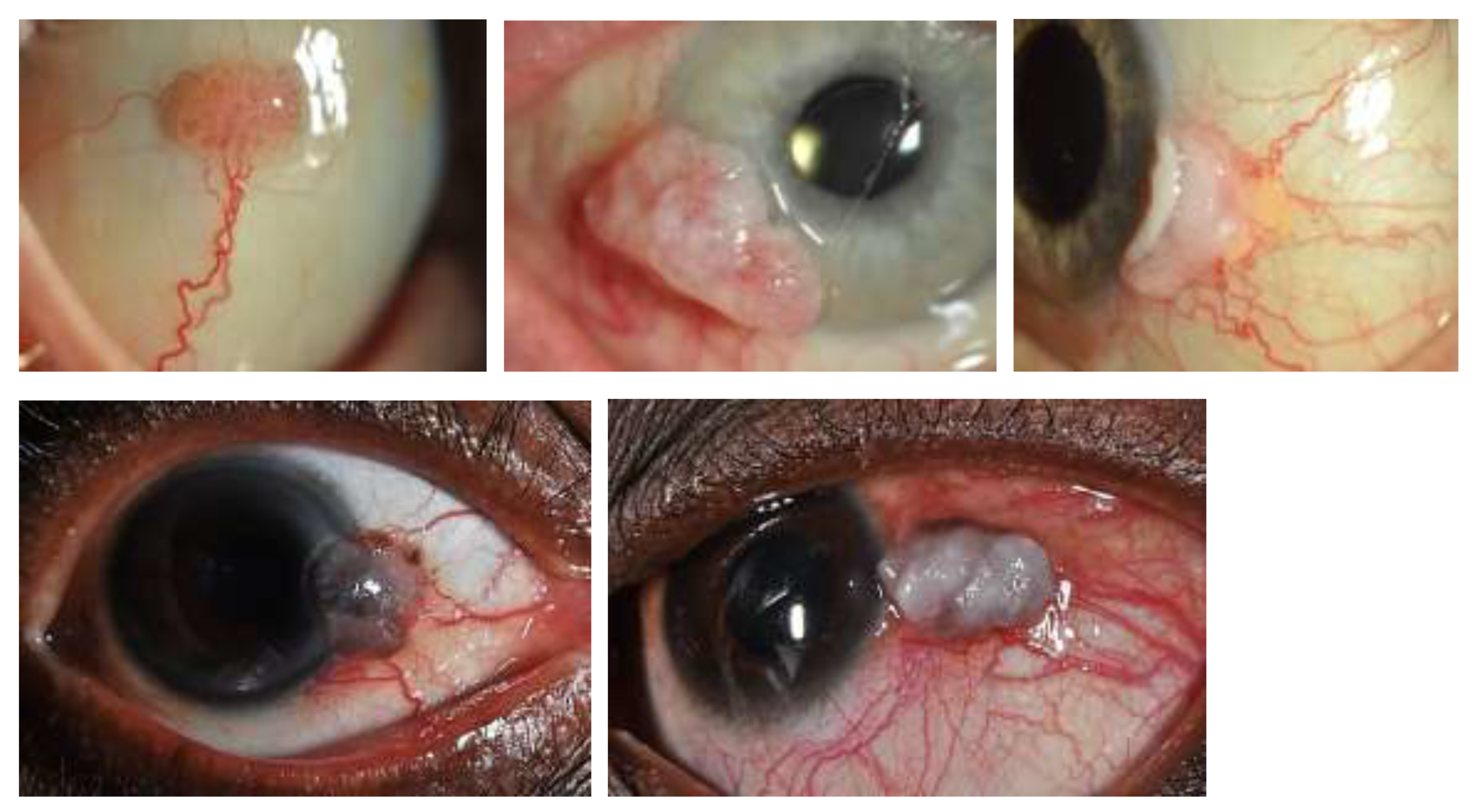
4. Clinical Presentation and Assessment
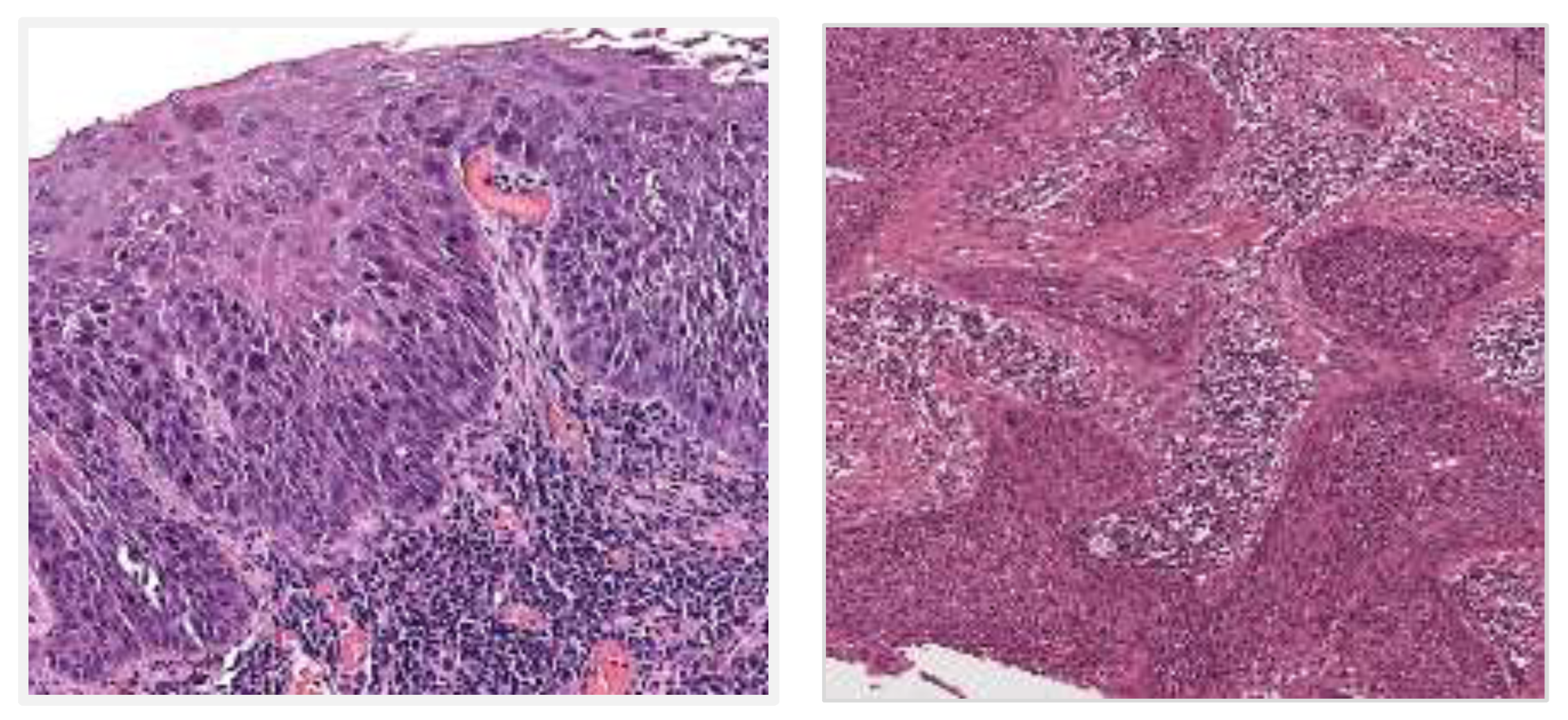
5. Histomorphological Features
6. Treatment and Prognosis
6. Future Direction and Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yang, J; Foster, CS. Squamous Cell Carcinoma of the Conjunctiva. International Ophthalmology Clinics 1997, 37(4), 73–85. [Google Scholar] [CrossRef] [PubMed]
- Gichuhi, S; Sagoo, MS; Weiss, HA; Burton, MJ. Epidemiology of ocular surface squamous neoplasia in Africa. Trop Med Int Health 2013, 18(12), 1424–43. [Google Scholar] [CrossRef]
- Newton, R; Ferlay, J; Reeves, G; Beral, V; Parkin, DM. Effect of ambient solar ultraviolet radiation on incidence of squamous-cell carcinoma of the eye. Lancet 1996, 347(9013), 1450–1. [Google Scholar] [CrossRef] [PubMed]
- Sun, EC; Fears, TR; Goedert, JJ. Epidemiology of squamous cell conjunctival cancer. Cancer Epidemiology, Biomarkers & Prevention 1997, 6(2), 73–7. [Google Scholar]
- Darwich, R; Ghazawi, FM; Le, M; Rahme, E; Alghazawi, N; Zubarev, A; et al. Epidemiology of invasive ocular surface squamous neoplasia in Canada during 1992-2010. Br J Ophthalmol. 2020, 104(10), 1368–72. [Google Scholar] [CrossRef]
- Yin, VT; Merritt, HA; Sniegowski, M; Esmaeli, B. Eyelid and ocular surface carcinoma: diagnosis and management. Clin Dermatol 2015, 33(2), 159–69. [Google Scholar] [CrossRef]
- Emmanuel, B; Ruder, E; Lin, SW; Abnet, C; Hollenbeck, A; Mbulaiteye, S. Incidence of squamous-cell carcinoma of the conjunctiva and other eye cancers in the NIH-AARP Diet and Health Study. Ecancermedicalscience 2012, 6, 254. [Google Scholar]
- McKelvie, PA; Daniell, M; McNab, A; Loughnan, M; Santamaria, JD. Squamous cell carcinoma of the conjunctiva: a series of 26 cases. Br J Ophthalmol. 2002, 86(2), 168–73. [Google Scholar] [CrossRef]
- Margo, CE; White, AA. Ocular surface squamous neoplasia: terminology that is conceptually friendly but clinically perilous. Eye (Lond) 2014, 28(5), 507–9. [Google Scholar] [CrossRef]
- Donaldson, MJ; Sullivan, TJ; Whitehead, KJ; Williamson, RM. Squamous cell carcinoma of the eyelids. Br J Ophthalmol. 2002, 86(10), 1161–5. [Google Scholar] [CrossRef]
- Gichuhi, S; Macharia, E; Kabiru, J; Zindamoyen, AM; Rono, H; Ollando, E; et al. Clinical Presentation of Ocular Surface Squamous Neoplasia in Kenya. JAMA Ophthalmol. 2015, 133(11), 1305–13. [Google Scholar] [CrossRef] [PubMed]
- Gichuhi, S; Sagoo, MS. Squamous cell carcinoma of the conjunctiva. Community Eye Health 2016, 29(95), 52–3. [Google Scholar]
- Gichuhi, S; Ohnuma, S; Sagoo, MS; Burton, MJ. Pathophysiology of ocular surface squamous neoplasia. Exp Eye Res. 2014, 129, 172–82. [Google Scholar] [CrossRef] [PubMed]
- Kaliki, S; Vempuluru, VS; Ghose, N; Gunda, S; Vithalani, NM; Sultana, S; et al. Ocular surface squamous neoplasia in India: a study of 438 patients. Int Ophthalmol. 2022, 42(6), 1915–26. [Google Scholar] [CrossRef] [PubMed]
- Choi, CJ; Jakobiec, FA; Zakka, FR; Foster, CS; Chodosh, J; Freitag, SK. Conjunctival Squamous Cell Neoplasia Associated With Ocular Cicatricial Pemphigoid. Ophthalmic Plast Reconstr Surg 2017, 33(6), e157–e60. [Google Scholar] [CrossRef]
- Galati, L; Combes, JD; Gupta, P; Sen, R; Robitaille, A; Brancaccio, RN; et al. Detection of a large spectrum of viral infections in conjunctival premalignant and malignant lesions. Int J Cancer 2020, 147(10), 2862–70. [Google Scholar] [CrossRef]
- Hämmerl, L; Ferlay, J; Borok, M; Carrilho, C; Parkin, DM. The burden of squamous cell carcinoma of the conjunctiva in Africa. Cancer Epidemiol. 2019, 61, 150–3. [Google Scholar] [CrossRef]
- Djulbegovic, MB; Uversky, VN; Karp, CL; Harbour, JW. Functional impact of titin (TTN) mutations in ocular surface squamous neoplasia. Int J Biol Macromol. 2022, 195, 93–101. [Google Scholar] [CrossRef]
- Sakai, A; Tagami, M; Kakehashi, A; Katsuyama-Yoshikawa, A; Misawa, N; Wanibuchi, H; et al. Expression, intracellular localization, and mutation of EGFR in conjunctival squamous cell carcinoma and the association with prognosis and treatment. PLoS One 2020, 15(8), e0238120. [Google Scholar] [CrossRef]
- Scholz, SL; Thomasen, H; Reis, H; Möller, I; Darawsha, R; Müller, B; et al. Frequent TERT Promoter Mutations in Ocular Surface Squamous Neoplasia. Invest Ophthalmol Vis Sci. 2015, 56(10), 5854–61. [Google Scholar] [CrossRef]
- Ng, J; Coroneo, MT; Wakefield, D; Di Girolamo, N. Ultraviolet Radiation and the Role of Matrix Metalloproteinases in the Pathogenesis of Ocular Surface Squamous Neoplasia. Investigative Ophthalmology & Visual Science 2008, 49(12), 5295–306. [Google Scholar] [CrossRef]
- Ateenyi-Agaba, C; Dai, M; Le Calvez, F; Katongole-Mbidde, E; Smet, A; Tommasino, M; et al. TP53 mutations in squamous-cell carcinomas of the conjunctiva: evidence for UV-induced mutagenesis. Mutagenesis 2004, 19(5), 399–401. [Google Scholar] [CrossRef]
- Mahale, A; Alkatan, H; Alwadani, S; Othman, M; Suarez, MJ; Price, A; et al. Altered gene expression in conjunctival squamous cell carcinoma. Modern Pathology 2016, 29(5), 452–60. [Google Scholar] [CrossRef]
- Paulina García de Alba Graue EE, Pablo Zoroquiain, Ana Beatriz, Toledo Dias, Miguel Burnier. HER2 as a possible therapeutic target in squamous cell carcinoma of the conjunctiva. Annals of Eye Science 2018, 3. [CrossRef]
- Asnaghi, L; Alkatan, H; Mahale, A; Othman, M; Alwadani, S; Al-Hussain, H; et al. Identification of multiple DNA copy number alterations including frequent 8p11.22 amplification in conjunctival squamous cell carcinoma. Invest Ophthalmol Vis Sci. 2014, 55(12), 8604–13. [Google Scholar] [CrossRef]
- Starita, N; Buonaguro, L; Buonaguro, FM; Tornesello, ML. Telomerase promoter mutations in human immunodeficiency virus-related conjunctiva neoplasia. J Transl Med. 2018, 16(1), 77. [Google Scholar] [CrossRef]
- Lin, SY; Liao, SL; Hong, JB; Chu, CY; Sheen, YS; Jhuang, JY; et al. TERT promoter mutations in periocular carcinomas: implications of ultraviolet light in pathogenesis. Br J Ophthalmol. 2016, 100(2), 274–7. [Google Scholar] [CrossRef]
- Lazo de la Vega, L; Bick, N; Hu, K; Rahrig, SE; Silva, CD; Matayoshi, S; et al. Invasive squamous cell carcinomas and precursor lesions on UV-exposed epithelia demonstrate concordant genomic complexity in driver genes. Mod Pathol. 2020, 33(11), 2280–94. [Google Scholar] [CrossRef]
- Di Girolamo, N; Atik, A; McCluskey, PJ; Wakefield, D. Matrix metalloproteinases and their inhibitors in squamous cell carcinoma of the conjunctiva. Ocul Surf. 2013, 11(3), 193–205. [Google Scholar] [CrossRef]
- Kenawy, N; Garrick, A; Heimann, H; Coupland, SE; Damato, BE. Conjunctival squamous cell neoplasia: the Liverpool Ocular Oncology Centre experience. Graefes Arch Clin Exp Ophthalmol. 2015, 253(1), 143–50. [Google Scholar] [CrossRef]
- Yousef, YA; Finger, PT. Squamous carcinoma and dysplasia of the conjunctiva and cornea: an analysis of 101 cases. Ophthalmology 2012, 119(2), 233–40. [Google Scholar] [CrossRef]
- Miller, CV; Wolf, A; Klingenstein, A; Decker, C; Garip, A; Kampik, A; et al. Clinical outcome of advanced squamous cell carcinoma of the conjunctiva. Eye (Lond) 2014, 28(8), 962–7. [Google Scholar] [CrossRef]
- Faustina, M; Diba, R; Ahmadi, MA; Esmaeli, B. Patterns of regional and distant metastasis in patients with eyelid and periocular squamous cell carcinoma. Ophthalmology 2004, 111(10), 1930–2. [Google Scholar] [CrossRef]
- Chauhan, S; Sen, S; Sharma, A; Tandon, R; Kashyap, S; Pushker, N; et al. American Joint Committee on Cancer Staging and clinicopathological high-risk predictors of ocular surface squamous neoplasia: a study from a tertiary eye center in India. Arch Pathol Lab Med. 2014, 138(11), 1488–94. [Google Scholar] [CrossRef]
- Yamamoto H, Mudhar HS, Cherepanoff S, Eberhart CG, Heindl LM, Ferreira T. Conjunctival squamous cell carcinoma. In: Board WCoTE, editor. WHO Classification of Tumours: Eye and Orbit Tumours. WHO Classification of Tumours series. 13. 5 ed. Lyon, France: International Agency for Research on Cancer; 2025.
- Heegaard S, Alkatan H, Eberhart CG, M. HL. Conjunctival squamous intraepithelial neoplasia. In: Board WCoTE, editor. WHO Classification of Tumours: Eye and Orbit Tumours. WHO Classification of Tumours series. 13. 5 ed. Lyon, France: International Agency for Research on Cancer; 2025.
- Demirci, H; Elner, VM; Demirci, FY; Robinson, DR; Chinnaiyan, A; Schlachter, D; et al. Immunotherapy for Conjunctival Squamous Cell Carcinoma with Orbital Extension. Ophthalmology 2021, 128(5), 801–4. [Google Scholar] [CrossRef]
- Demirci, H; Vo, JN; Wu, YM; Elner, V; Chinnaiyan, AM; Robinson, D; et al. Next-Generation Sequencing-Based Molecular Profiling of Conjunctival Squamous Cell Carcinoma and Its Potential Application for Therapy. Ophthalmol Sci. 2025, 5(5), 100801. [Google Scholar] [CrossRef]
- Kanda, M; Jayawickrema, N; Davagnanam, I; Samia-Aly, E; Gupta, T; Daniel, C. Cemiplimab in advanced periocular conjunctival and cutaneous squamous cell carcinoma: a United Kingdom case series. Orbit 2025, 1–11. [Google Scholar] [CrossRef]
- Esmaeli, B; Fan, J; Goldberg, H; Lu, T; Gross, ND; Akhave, N; et al. Immune checkpoint inhibitors with or without chemotherapy for orbital, conjunctival, and ocular adnexal squamous cell carcinoma. Canadian Journal of Ophthalmology 2025, 60(1), e38–e51. [Google Scholar] [CrossRef]
- Azad, AD; Zhang, JJ; Emerick, KS; Shalhout, SZ; Kaufman, HL; Miller, DM; et al. Immunotherapy for Advanced Conjunctival Squamous Cell Carcinoma: Treatment Failures. Ophthalmic Plast Reconstr Surg. 2025, 41(6), 646–50. [Google Scholar] [CrossRef]
- Ceylanoglu, KS; Eisenberg, A; Worden, FP; Elner, VM; Demirci, H. Use of Cemiplimab, an Immune Checkpoint Inhibitor for Conjunctival Intraepithelial Neoplasia. Cornea 2024, 43(4), 528–30. [Google Scholar] [CrossRef]
- Steren, B; Burtness, B; Bhatia, A; Demirci, H; Shinder, R; Yoo, D; et al. Cemiplimab for Orbital Squamous Cell Carcinoma in 11 Cases. Ophthalmic Plastic & Reconstructive Surgery 2022, 38(5), 496–502. [Google Scholar] [CrossRef]
- Hoffmann, M; Schardt, J; Ionescu, C; Wolf-Schnurrbusch, UE; Wagner, F; Dettmer, MS; et al. Long lasting response to anti-PD-1 therapy of recurrent conjunctival squamous cell carcinoma-A case report and systematic review of the literature. Current Problems in Cancer: Case Reports 2021, 4, 100088. [Google Scholar] [CrossRef]
- El-Sawy, T; Sabichi, AL; Myers, JN; Kies, MS; William, WN; Glisson, BS; et al. Epidermal Growth Factor Receptor Inhibitors for Treatment of Orbital Squamous Cell Carcinoma. Archives of Ophthalmology 2012, 130(12), 1608–11. [Google Scholar] [CrossRef]
- Youn, GM; Case, AG; Jarin, T; Li, B; Swarup, A; Naranjo, A; et al. The Use of Panitumumab-IRDye800CW in a Novel Murine Model for Conjunctival Squamous Cell Carcinoma. Translational Vision Science & Technology 2022, 11(7), 23. [Google Scholar]
- Finger, PT; Chin, KJ. Refractory Squamous Cell Carcinoma of the Conjunctiva Treated With Subconjunctival Ranibizumab (Lucentis): A Two-Year Study. Ophthalmic Plastic & Reconstructive Surgery 2012, 28(2), 85–9. [Google Scholar]
- Pe'er, J. Ocular surface squamous neoplasia. Ophthalmol Clin North Am. 2005, 18(1), 1–13, vii. [Google Scholar] [CrossRef]
- Turner, HC; Budak, MT; Akinci, MA; Wolosin, JM. Comparative analysis of human conjunctival and corneal epithelial gene expression with oligonucleotide microarrays. Invest Ophthalmol Vis Sci. 2007, 48(5), 2050–61. [Google Scholar] [CrossRef]
- Mehta, M; Fay, A. Squamous cell carcinoma of the eyelid and conjunctiva. Int Ophthalmol Clin. 2009, 49(1), 111–21. [Google Scholar] [CrossRef]
- Shields, CL; Demirci, H; Karatza, E; Shields, JA. Clinical survey of 1643 melanocytic and nonmelanocytic conjunctival tumors. Ophthalmology 2004, 111(9), 1747–54. [Google Scholar] [CrossRef]
- Ateenyi-Agaba, C. Conjunctival squamous-cell carcinoma associated with HIV infection in Kampala, Uganda. Lancet 1995, 345(8951), 695–6. [Google Scholar] [CrossRef]
- Daya-Grosjean, L; Dumaz, N; Sarasin, A. The specificity of p53 mutation spectra in sunlight induced human cancers. J Photochem Photobiol B 1995, 28(2), 115–24. [Google Scholar] [CrossRef]
- Dérijard, B; Hibi, M; Wu, IH; Barrett, T; Su, B; Deng, T; et al. JNK1: a protein kinase stimulated by UV light and Ha-Ras that binds and phosphorylates the c-Jun activation domain. Cell. 1994, 76(6), 1025–37. [Google Scholar] [CrossRef]
- Blackburn, EH. Telomeres and telomerase: their mechanisms of action and the effects of altering their functions. FEBS Lett. 2005, 579(4), 859–62. [Google Scholar] [CrossRef]
- Chauhan, S; Sen, S; Chauhan, SS; Pushker, N; Tandon, R; Kashyap, S; et al. Stratifin in ocular surface squamous neoplasia and its association with p53. Acta Ophthalmol. 2021, 99(8), e1483–e91. [Google Scholar] [CrossRef]
- Dong, Z; Kase, S; Ando, R; Fukuhara, J; Kinoshita, S; Dong, Y; et al. Expression of αB-crystallin and vascular endothelial growth factor in conjunctival squamous cell carcinoma. Anticancer Res. 2013, 33(9), 3745–51. [Google Scholar]
- Nagarajan, P; El-Hadad, C; Gruschkus, SK; Ning, J; Hudgens, CW; Sagiv, O; et al. PD-L1/PD1 Expression, Composition of Tumor-Associated Immune Infiltrate, and HPV Status in Conjunctival Squamous Cell Carcinoma. Invest Ophthalmol Vis Sci. 2019, 60(6), 2388–98. [Google Scholar] [CrossRef]
- Slater, NA; Googe, PB. PD-L1 expression in cutaneous squamous cell carcinoma correlates with risk of metastasis. J Cutan Pathol. 2016, 43(8), 663–70. [Google Scholar] [CrossRef]
- Muchengeti, M; Bohlius, J; Dhokotera, TG. Conjunctival cancer in people living with HIV. Curr Opin Infect Dis. 2021, 34(1), 1–7. [Google Scholar] [CrossRef]
- Ramberg, I; Møller-Hansen, M; Toft, PB; Funding, M; Heegaard, S. Human papillomavirus infection plays a role in conjunctival squamous cell carcinoma: a systematic review and meta-analysis of observational studies. Acta Ophthalmologica 2021, 99(5), 478–88. [Google Scholar] [CrossRef]
- Griffin, H; Mudhar, HS; Rundle, P; Shiraz, A; Mahmood, R; Egawa, N; et al. Human papillomavirus type 16 causes a defined subset of conjunctival in situ squamous cell carcinomas. Mod Pathol. 2020, 33(1), 74–90. [Google Scholar] [CrossRef]
- Ramberg, I; Toft, PB; Georgsen, JB; Siersma, VD; Funding, M; Jensen, DH; et al. Conjunctival intraepithelial neoplasia and carcinoma: distinct clinical and histological features in relation to human papilloma virus status. Br J Ophthalmol. 2021, 105(6), 878–83. [Google Scholar] [CrossRef]
- Cervantes, G; Rodríguez, AA, Jr.; Leal, AG. Squamous cell carcinoma of the conjunctiva: clinicopathological features in 287 cases. Can J Ophthalmol. 2002, 37(1), 14-9; discussion 9-20. [Google Scholar] [CrossRef]
- Krachmer, JH; Mannis, MJ; Holland, EJ. Cornea: Fundamentals, Diagnosis, and Management. American Orthoptic Journal. 2011, 61, 147. [Google Scholar]
- Nelson, KD; McSoley, JJ. Clinical findings and management of conjunctival intraepithelial neoplasia. Optometry 2011, 82(1), 15–21. [Google Scholar] [CrossRef]
- Shields, CL; Shields, JA. Tumors of the conjunctiva and cornea. Surv Ophthalmol. 2004, 49(1), 3–24. [Google Scholar] [CrossRef]
- KWITKO, ML; BONIUK, M; ZIMMERMAN, LE. Eyelid Tumors With Reference to Lesions Confused With Squamous Cell Carcinoma: I. Incidence and Errors in Diagnosis. Archives of Ophthalmology 1963, 69(6), 693–7. [Google Scholar] [CrossRef]
- Gichuhi, S; Macharia, E; Kabiru, J; Zindamoyen, AMb; Rono, H; Ollando, E; et al. Toluidine blue 0.05% vital staining for the diagnosis of ocular surface squamous neoplasia in Kenya. JAMA ophthalmology 2015, 133(11), 1314–21. [Google Scholar] [CrossRef]
- Thomas, BJ; Galor, A; Nanji, AA; El Sayyad, F; Wang, J; Dubovy, SR; et al. Ultra high-resolution anterior segment optical coherence tomography in the diagnosis and management of ocular surface squamous neoplasia. The ocular surface 2014, 12(1), 46–58. [Google Scholar] [CrossRef]
- Stevens, SM; Reyes-Capo, DP; Patel, U; Choudhary, A; Khzam, RA; Tang, V; et al. Clinical and Optical Coherence Tomography Comparison Between Ocular Surface Squamous Neoplasia and Squamous Metaplasia. Cornea 2023, 42(4), 429–34. [Google Scholar] [CrossRef]
- Xu, Y; Zhou, Z; Wang, M; Liu, F; Qu, H; Hong, J. The clinical value of in vivo confocal microscopy for diagnosis of ocular surface squamous neoplasia. Eye 2012, 26(6), 781–7. [Google Scholar] [CrossRef]
- Liu, Z; Karp, CL; Galor, A; Al Bayyat, GJ; Jiang, H; Wang, J. Role of optical coherence tomography angiography in the characterization of vascular network patterns of ocular surface squamous neoplasia. Ocul Surf. 2020, 18(4), 926–35. [Google Scholar] [CrossRef]
- Karp, CL; Mercado, C; Venkateswaran, N; Ruggeri, M; Galor, A; Garcia, A; et al. Use of High-Resolution Optical Coherence Tomography in the Surgical Management of Ocular Surface Squamous Neoplasia: A Pilot Study. Am J Ophthalmol. 2019, 206, 17–31. [Google Scholar] [CrossRef] [PubMed]
- Jaworski, A; Wolffsohn, JS; Napper, GA. Detection, aetiology and management of conjunctival intraepithelial neoplasia. Ophthalmic Physiol Opt. 2000, 20(5), 371–80. [Google Scholar] [CrossRef] [PubMed]
- Lee, GA; Hirst, LW. Ocular surface squamous neoplasia. Surv Ophthalmol. 1995, 39(6), 429–50. [Google Scholar] [CrossRef] [PubMed]
- Erie, JC; Campbell, RJ; Liesegang, TJ. Conjunctival and corneal intraepithelial and invasive neoplasia. Ophthalmology 1986, 93(2), 176–83. [Google Scholar] [CrossRef]
- Grossniklaus, HE; Green, WR; Luckenbach, M; Chan, CC. Conjunctival lesions in adults. A clinical and histopathologic review. Cornea 1987, 6(2), 78–116. [Google Scholar] [CrossRef]
- Mauriello, JA, Jr.; Napolitano, J; McLean, I. Actinic keratosis and dysplasia of the conjunctiva: a clinicopathological study of 45 cases. Can J Ophthalmol. 1995, 30(6), 312–6. [Google Scholar]
- Kao, AA; Galor, A; Karp, CL; Abdelaziz, A; Feuer, WJ; Dubovy, SR. Clinicopathologic correlation of ocular surface squamous neoplasms at Bascom Palmer Eye Institute: 2001 to 2010. Ophthalmology 2012, 119(9), 1773–6. [Google Scholar] [CrossRef]
- Reese, A; Margo, CE. Conjunctival squamous intraepithelial neoplasia and its differential diagnosis. J Clin Pathol. 2022, 75(5), 354–8. [Google Scholar] [CrossRef]
- Mishra, DK; Taneja, S; Singh, V; Veena, U; Kaliki, S. Histopathological spectrum of ocular surface squamous neoplasia: A retrospective study of 776 lesions. Indian J Pathol Microbiol. 2022, 65(1), 3–7. [Google Scholar] [CrossRef]
- Hamam, R; Bhat, P; Foster, CS. Conjunctival/corneal intraepithelial neoplasia. Int Ophthalmol Clin. 2009, 49(1), 63–70. [Google Scholar] [CrossRef]
- Pizzarello, LD JF. Bowen's disease of the conjunctiva: a misnomer Ocular and adnexal tumors 1978, 553–71.
- Iliff, WJ; Marback, R; Green, WR. Invasive Squamous Cell Carcinoma of the Conjunctiva. Archives of Ophthalmology 1975, 93(2), 119–22. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z; Li, B; Shi, J; Xu, X; Li, L; Gao, F. Intraocular Extension of Conjunctival Squamous Cell Carcinoma. Ophthalmologica 2007, 221(3), 200–3. [Google Scholar] [CrossRef] [PubMed]
- Cottel, WI. Perineural invasion by squamous-cell carcinoma. J Dermatol Surg Oncol. 1982, 8(7), 589–600. [Google Scholar] [CrossRef]
- Albert, DM; Jakobiec, FA. Principles and practice of ophthalmology; Saunders, 2000. [Google Scholar]
- Vempuluru, VS; Kapoor, AG; Kaliki, S; Jajapuram, SD; Mohamed, A; Mishra, DK. Comparative evaluation of clinical characteristics of biopsy-proven conjunctival intraepithelial neoplasia and invasive squamous cell carcinoma using image processing software programs. International Ophthalmology 2021, 41(4), 1301–7. [Google Scholar] [CrossRef] [PubMed]
- Nanji, AA; Mercado, C; Galor, A; Dubovy, S; Karp, CL. Updates in Ocular Surface Tumor Diagnostics. Int Ophthalmol Clin. 2017, 57(3), 47–62. [Google Scholar] [CrossRef]
- Mittal, R; Rath, S; Vemuganti, GK. Ocular surface squamous neoplasia - Review of etio-pathogenesis and an update on clinico-pathological diagnosis. Saudi J Ophthalmol. 2013, 27(3), 177–86. [Google Scholar] [CrossRef]
- Singh, S; Mohamed, A; Kaliki, S. Ocular surface squamous neoplasia: analysis based on the 8th American Joint Committee on Cancer classification. International Ophthalmology 2019, 39(6), 1283–91. [Google Scholar] [CrossRef]
- Brierley, JD; Gospodarowicz, M; Wittekind, C. TNM Classification of Malignant Tumours, 8 ed.; Wiley-Blackwell: Oxford, UK, 2017. [Google Scholar]
- Adler, E; Turner, JR; Stone, DU. Ocular surface squamous neoplasia: a survey of changes in the standard of care from 2003 to 2012. Cornea 2013, 32(12), 1558–61. [Google Scholar] [CrossRef]
- Shields, JA; Shields, CL; De Potter, P. Surgical management of conjunctival tumors. The 1994 Lynn B. McMahan Lecture. Arch Ophthalmol. 1997, 115(6), 808–15. [Google Scholar] [CrossRef]
- Al Bayyat, G; Arreaza-Kaufman, D; Venkateswaran, N; Galor, A; Karp, CL. Update on pharmacotherapy for ocular surface squamous neoplasia. Eye and Vision. 2019, 6(1), 24. [Google Scholar] [CrossRef]
- Ali, MJ; Pujari, A; Dave, TV; Kaliki, S; Naik, MN. Clinicopathological profile of orbital exenteration: 14 years of experience from a tertiary eye care center in South India. International Ophthalmology 2016, 36(2), 253–8. [Google Scholar] [CrossRef]
- Karp, CL; Galor, A; Chhabra, S; Barnes, SD; Alfonso, EC. Subconjunctival/perilesional recombinant interferon α2b for ocular surface squamous neoplasia: a 10-year review. Ophthalmology 2010, 117(12), 2241–6. [Google Scholar] [CrossRef]
- Nanji, AA; Moon, CS; Galor, A; Sein, J; Oellers, P; Karp, CL. Surgical versus medical treatment of ocular surface squamous neoplasia: a comparison of recurrences and complications. Ophthalmology 2014, 121(5), 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Joag, MG; Sise, A; Murillo, JC; Sayed-Ahmed, IO; Wong, JR; Mercado, C; et al. Topical 5-Fluorouracil 1% as Primary Treatment for Ocular Surface Squamous Neoplasia. Ophthalmology 2016, 123(7), 1442–8. [Google Scholar] [CrossRef]
- Gichuhi, S; Macharia, E; Kabiru, J; Zindamoyen, AM; Rono, H; Ollando, E; et al. Topical fluorouracil after surgery for ocular surface squamous neoplasia in Kenya: a randomised, double-blind, placebo-controlled trial. Lancet Glob Health 2016, 4(6), e378-85. [Google Scholar] [CrossRef] [PubMed]
- Zein, M; De Arrigunaga, S; Amer, MM; Galor, A; Nichols, AJ; Ioannides, T; et al. Therapeutic Response to Treatment of a Papillomatous Ocular Surface Squamous Neoplasia With Intramuscular Human Papillomavirus Vaccine. Cornea 2024, 43(8), 1049–52. [Google Scholar] [CrossRef] [PubMed]
- Sripawadkul, W; AlBayyat, G; Galor, A; Wylegala, A; Nichols, AJ; Ioannides, T; et al. Resolution of a Presumed Conjunctival Papilloma After Therapeutic Treatment With the Human Papillomavirus Vaccine. JAMA Ophthalmol. 2022, 140(4), 434–5. [Google Scholar] [CrossRef]
- Seth, I; Roberts, P; Cadieux, D. Gardasil-9 Vaccine as a Possible Treatment Option for Human Papillomavirus Conjunctival Papilloma. Ophthalmology 2023, 130(5), 532. [Google Scholar] [CrossRef]
- Cekiç, O; Bardak, Y; Kapucuoğlu, N. Photodynamic therapy for conjunctival ocular surface squamous neoplasia. J Ocul Pharmacol Ther. 2011, 27(2), 205–7. [Google Scholar] [CrossRef]
- Arepalli, S; Kaliki, S; Shields, CL; Emrich, J; Komarnicky, L; Shields, JA. Plaque Radiotherapy in the Management of Scleral-Invasive Conjunctival Squamous Cell Carcinoma: An Analysis of 15 Eyes. JAMA Ophthalmology 2014, 132(6), 691–6. [Google Scholar] [CrossRef]
- Panda, BB; Dasmajumdar, SK; Sadangi, D; Tripathy, SR. Radiation therapy in conjunctival squamous cell carcinoma. BMJ Case Rep. 2023, 16(7). [Google Scholar] [CrossRef] [PubMed]
- Zheng, T; Wang, D; Miao, Y; Dong, M; Liu, Q; Zhang, Q; et al. Clinical efficacy and safety of proton radiotherapy for ocular conjunctival malignancies: a systematic review and meta-analysis. Front Public Health 2025, 13, 1486988. [Google Scholar] [CrossRef] [PubMed]
- Graue, GF; Tena, LB; Finger, PT. Electron beam radiation for conjunctival squamous carcinoma. Ophthalmic Plast Reconstr Surg. 2011, 27(4), 277–81. [Google Scholar] [CrossRef]
- Abt, NB; Zhao, J; Huang, Y; Eghrari, AO. Prognostic factors and survival for malignant conjunctival melanoma and squamous cell carcinoma over four decades. Am J Otolaryngol. 2019, 40(4), 577–82. [Google Scholar] [CrossRef]
- Vempuluru, VS; Indusekhar, H; Mohamed, A; Kaliki, S. Metastatic conjunctival squamous cell carcinoma: a study of 5 patients. Int Ophthalmol. 2022, 42(10), 3097–108. [Google Scholar] [CrossRef] [PubMed]
- Bellerive, C; Berry, JL; Polski, A; Singh, AD. Conjunctival Squamous Neoplasia: Staging and Initial Treatment. Cornea 2018, 37(10), 1287–91. [Google Scholar] [CrossRef]
- Alkatan, HM; Al-Motlak, MA; Al-Shedoukhy, AA. Metastatic Squamous Spindle Cell Carcinoma of the conjunctiva. Saudi J Ophthalmol. 2010, 24(4), 155–8. [Google Scholar] [CrossRef]
- Asena, L; Dursun Altınörs, D. Topical Bevacizumab for the Treatment of Ocular Surface Squamous Neoplasia. Journal of Ocular Pharmacology and Therapeutics 2015, 31(8), 487–90. [Google Scholar] [CrossRef]
- Faramarzi, A; Feizi, S. Subconjunctival bevacizumab injection for ocular surface squamous neoplasia. Cornea 2013, 32(7), 998–1001. [Google Scholar] [CrossRef] [PubMed]
| Study | Patient (s) | Stage | Primary treatment | Agent used | Dosage | Outcome | Adverse reactions |
|---|---|---|---|---|---|---|---|
| Esmaeli et al, 2025 [40] | 17 (5 CSCC) | Advanced periocular SCC (orbital, conjunctival, lacrimal) | Neoadjuvant or definitive systemic ICI therapy | Cemiplimab or Pembrolizumab | Not specified | 5 complete responses, 8 partial responses, 4 stable disease. At least 1 CSCC with nodal metastasis achieved complete response and avoided surgery | Not specified |
| Azad et al, 2025 [41] | 5 | Advanced CSCC | Systemic ICI therapy | Not specified | Not specified | All showed progression, 3 required exenteration | |
| Kanda et al, 2025 [39] | 9 | Advanced CSCC | Systemic ICI therapy | Cemiplimab | Not specified | 22% complete response, 33% partial response, 44% progressed | Hypothyroidism, hepatitis |
| Ceylanoglu et al, 2024 [42] | 70Y, Male | CSIN lesion on ocular surface | Excisional biopsy with cryotherapy | Cemiplimab | 350 mg IV every 3 weeks | Response shown in metastatic lesions. CSIN continued to grow. | Not specified |
| Ceylanoglu et al, 2024 [42] | 84Y, Female | CSIN of inferior bulbar and palpebral conjunctiva | Excisional biopsy with cryotherapy | Cemiplimab | 350 mg IV every 3 weeks | Progression of CSIN | Not specified |
| Steren et al, 2022 [43] | 11 | CSCC with orbital involvement | Excisional biopsy | Cemiplimab | 350 mg IV every 3 weeks | Complete response in 9/11 patients | Not specified |
| Hoffmann et al, 2021 [44] | 59Y, Female | Recurrent CSCC | Radiotherapy | Cemiplimab | 3mg/kg IV every 2 weeks | Resolution after 19 months of treatment | None reported |
| Demirci at al, 2020 [37] | 5 (82M, 59M, 86F, 71M, 61M) |
Advanced CSCC with orbital involvement | Excisional biopsy | Pembrolizumab and Cemiplimab | Pembrolizumab – 350 mg every 3 weeks Cemiplimab – 200mg every 3 weeks |
4/5 patients complete response. 1 showed progression | 86M – Colitis, 86F – Rash, pain |
| Study | Patient (s) | Stage | Primary treatment | Agent used | Dosage | Outcome | Adverse reactions |
|---|---|---|---|---|---|---|---|
| El Sawy et al, 2022 (EGFR inhibitor) [45] | 2 | Advanced CSCC | Topical/systemic therapy with surgery | Erlotinib, Cetuximab | Not specified | Regression of ocular tumour size after 7.5 months | None reported |
| Asena et al, 2015 (anti-VEGF) [114] | 6 | CSCC | Topical targeted therapy | Bevacizumab | 5 mg/ml eye drops, 4 times daily for 8 weeks | 2/6 complete tumour resolutions, 4 patients required excisional surgery | None reported |
| Faramarzi et al, 2013 (anti-VEGF) [115] | 10 | Combination of CSIN and CSCC | Perilesional/subconjunctival injection with excision/cryotherapy if needed | Bevacizumab | 2 injections at 2 week interval | Mean tumour area reduced by 25% after 1st and 42% after 2nd injection, complete disappearance in 2 eyes with CSIN only | None reported |
| Finger et al, 2012 (anti-VEGF) [47] | 5 | Recurrent CSCC | Excision, cryotherapy or topical therapy | Ranibizumab | 0.5 mg monthly or bimonthly | 3/5 complete responses, 2 failed treatment | None reported |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




