Submitted:
10 February 2026
Posted:
11 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Physiological and Pathological Alpha-Synuclein
2.1. Physiological role of α-Synuclein in Neurons
2.2. Endogenous α-Synuclein in Glial Cells Under Physiological Conditions
2.3. Subcellular Localization and Organellar Biology
2.3.1. Endogenous α-Synuclein at Presynaptic Membranes
2.3.2. Endogenous α-Synuclein in the Nucleus
2.3.3. Endogenous α-Synuclein at Other Organellar Membranes
2.3.4. Endogenous α-Synuclein in Membraneless Condensates
2.4. Alpha-Synuclein in RNA Biology
2.5. Physiological Strains and Post-Translational Modifications
2.5.1. Physiological Multimers vs Pathological Oligomers
2.5.2. The Physiological Post-Translational Modifications Landscape of α-Synuclein
2.5.3. The Post-Translational Modifications and Aggregation-Prone Conformers
2.5.4. Ubiquitination and SUMOylation
3. Unified and Divergent Pathways of Alpha-Synuclein-Driven Neurodegeneration
3.1. Lewy Pathology as a Network-Embedded Synucleinopathy
3.1.1. Selective Neuronal Vulnerability: Intrinsic Properties That Amplify Risk
3.1.2. Propagation Dynamics: Prion-Like Spread Across Connected Circuits
3.1.3. Divergent Clinical Phenotypes Shaped by Network Context
3.1.4. Preclinical Network Context
3.2. Co-Pathology in Lewy Body Disorders
3.2.1. Molecular Crosstalk Between α-Synuclein, Aβ, and Tau in Lewy Body Disorders
3.2.2. Network-Level and Clinical Consequences of αSyn–Aβ–tau Synergy
3.3. Oligodendroglial α-Synuclein Pathology in Network-Defined Neuronal Systems
3.3.1. Mechanistic Questions Surrounding Oligodendrocyte α-Synuclein Pathology
3.3.2. Hypotheses on the Origins of α-Synuclein Pathology in MSA
- Neuron-to-glia transfer hypothesis
- The oligodendrogliopathy first hypothesis
- A membraneless organelle hypothesis of MSA
3.3.3. Molecular Specificity: MSA-Associated α-Synuclein Strains
4. Targeting Alpha-Synuclein: Biomarkers
4.1. Current Biomarker Modalities
4.1.1. Diagnostic Challenges Across Synucleinopathies
4.1.2. αSyn Species: Total, Oligomeric, and Phosphorylated Forms
4.1.3. Seed Amplification Assays (SAAs)
4.1.4. Other Assays and Combined Application
4.2. Strain-, Cell Type-, and Conformation-Specific Assays
5. Summary and Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| αSyn | Alpha-synuclein |
| AD | Alzheimer’s disease |
| Aβ | Amyloid-β |
| BBB | Blood–brain barrier |
| CSF | Cerebrospinal fluid |
| DLB | Dementia with Lewy bodies |
| ER | Endoplasmic reticulum |
| ESM | Epidemic Spreading Model |
| GCIs | Glial cytoplasmic inclusions |
| GA | Golgi apparatus |
| GNIs | Glial nuclear inclusions |
| iLBD | Incidental Lewy body disease |
| iRBD | Incidental REM sleep behavior disorder |
| IHC | Immunohistochemistry |
| LLPS | liquid–liquid phase separation |
| MSA | Multiple system atrophy |
| MAMs | Mitochondria-associated ER membranes |
| NAC | Non-amyloid-β component |
| NDM | Network Diffusion Model |
| NTA | N-terminally acetylated |
| NNIs | Neuronal nuclear inclusions |
| NSD-ISS | Neuronal αSyn disease integrated staging system |
| pS129 | Phosphorylation at serine 129 |
| P-bodies | Processing bodies |
| PD | Parkinson’s disease |
| PDD | Parkinson’s disease dementia |
| PTM | Post-translational modification |
| PLA | Proximity ligation assay |
| PM | Plasma membrane |
| RT-QuIC | Real-time quaking-induced conversion |
| SAA | seed amplification assay |
| SV | Synaptic vesicle |
| TH | Tyrosine hydroxylase |
References
- Park, H.; Kam, T.I.; Dawson, V.L.; Dawson, T.M. α-Synuclein pathology as a target in neurodegenerative diseases. Nat Rev Neurol 2025, 21, 32–47. [Google Scholar] [CrossRef]
- Fu, Y.; Halliday, G.M. Dementia with Lewy bodies and Parkinson disease dementia - the same or different and is it important? Nat Rev Neurol 2025, 21, 394–403. [Google Scholar] [CrossRef]
- Bellomo, G.; De Luca, C.M.G.; Paoletti, F.P.; Gaetani, L.; Moda, F.; Parnetti, L. α-Synuclein Seed Amplification Assays for Diagnosing Synucleinopathies. Neurology 2022, 99, 195–205. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Farris, C.M.; Weber, S.; Schade, S.; Nguyen, H.; Pérez-Soriano, A.; Giraldo, D.M.; Fernández, M.; Soto, M.; Cámara, A.; et al. Sensitivity and specificity of a seed amplification assay for diagnosis of multiple system atrophy: a multicentre cohort study. Lancet Neurol 2024, 23, 1225–1237. [Google Scholar] [CrossRef]
- Orrú, C.D.; Vaughan, D.P.; Vijiaratnam, N.; Real, R.; Martinez-Carrasco, A.; Fumi, R.; Jensen, M.T.; Hodgson, M.; Girges, C.; Gil-Martinez, A.-L. Diagnostic and prognostic value of α-synuclein seed amplification assay kinetic measures in Parkinson’s disease: a longitudinal cohort study. The Lancet Neurology 2025, 24, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Simuni, T.; Chahine, L.M.; Poston, K.; Brumm, M.; Buracchio, T.; Campbell, M.; Chowdhury, S.; Coffey, C.; Concha-Marambio, L.; Dam, T.; et al. A biological definition of neuronal α-synuclein disease: towards an integrated staging system for research. Lancet Neurol 2024, 23, 178–190. [Google Scholar] [CrossRef] [PubMed]
- Dam, T.; Pagano, G.; Brumm, M.C.; Gochanour, C.; Poston, K.L.; Weintraub, D.; Chahine, L.M.; Coffey, C.; Tanner, C.M.; Kopil, C.M.; et al. Neuronal alpha-Synuclein Disease integrated staging system performance in PPMI, PASADENA, and SPARK baseline cohorts. npj Parkinson’s Disease 2024, 10, 178. [Google Scholar] [CrossRef]
- Höglinger, G.U.; Adler, C.H.; Berg, D.; Klein, C.; Outeiro, T.F.; Poewe, W.; Postuma, R.; Stoessl, A.J.; Lang, A.E. A biological classification of Parkinson’s disease: the SynNeurGe research diagnostic criteria. Lancet Neurol 2024, 23, 191–204. [Google Scholar] [CrossRef]
- Jack, C.R., Jr.; Bennett, D.A.; Blennow, K.; Carrillo, M.C.; Dunn, B.; Haeberlein, S.B.; Holtzman, D.M.; Jagust, W.; Jessen, F.; Karlawish, J.; et al. NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimers Dement 2018, 14, 535–562. [Google Scholar] [CrossRef]
- Braak, H.; Del Tredici, K.; Rüb, U.; De Vos, R.A.; Steur, E.N.J.; Braak, E. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiology of aging 2003, 24, 197–211. [Google Scholar] [CrossRef]
- Beach, T.G.; Adler, C.H.; Lue, L.; Sue, L.I.; Bachalakuri, J.; Henry-Watson, J.; Sasse, J.; Boyer, S.; Shirohi, S.; Brooks, R.; et al. Unified staging system for Lewy body disorders: correlation with nigrostriatal degeneration, cognitive impairment and motor dysfunction. Acta Neuropathol 2009, 117, 613–634. [Google Scholar] [CrossRef]
- McKeith, I.G.; Dickson, D.W.; Lowe, J.; Emre, M.; O’brien, J.; Feldman, H.; Cummings, J.; Duda, J.; Lippa, C.; Perry, E. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology 2005, 65, 1863–1872. [Google Scholar] [CrossRef]
- Leverenz, J.B.; Hamilton, R.; Tsuang, D.W.; Schantz, A.; Vavrek, D.; Larson, E.B.; Kukull, W.A.; Lopez, O.; Galasko, D.; Masliah, E.; et al. Empiric refinement of the pathologic assessment of Lewy-related pathology in the dementia patient. Brain Pathol 2008, 18, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Attems, J.; Toledo, J.B.; Walker, L.; Gelpi, E.; Gentleman, S.; Halliday, G.; Hortobagyi, T.; Jellinger, K.; Kovacs, G.G.; Lee, E.B.; et al. Neuropathological consensus criteria for the evaluation of Lewy pathology in post-mortem brains: a multi-centre study. Acta Neuropathol 2021, 141, 159–172. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Tanglay, O.; Li, H.; Halliday, G.M. The Role of Alpha-Synuclein Pathology. In Translational Methods for Parkinson’s Disease and Atypical Parkinsonism Research; Groppa, S., Schneider, S.A., Eds.; Springer US: New York, NY, 2025; pp. 21–48. [Google Scholar]
- Barnes, L.L.; Leurgans, S.; Aggarwal, N.T.; Shah, R.C.; Arvanitakis, Z.; James, B.D.; Buchman, A.S.; Bennett, D.A.; Schneider, J.A. Mixed pathology is more likely in black than white decedents with Alzheimer dementia. Neurology 2015, 85, 528–534. [Google Scholar] [CrossRef]
- Frigerio, R.; Fujishiro, H.; Ahn, T.B.; Josephs, K.A.; Maraganore, D.M.; DelleDonne, A.; Parisi, J.E.; Klos, K.J.; Boeve, B.F.; Dickson, D.W.; et al. Incidental Lewy body disease: do some cases represent a preclinical stage of dementia with Lewy bodies? Neurobiol Aging 2011, 32, 857–863. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, K.A. Lewy body-related alpha-synucleinopathy in the aged human brain. J Neural Transm (Vienna) 2004, 111, 1219–1235. [Google Scholar] [CrossRef]
- Iranzo, A.; Tolosa, E.; Gelpi, E.; Molinuevo, J.L.; Valldeoriola, F.; Serradell, M.; Sanchez-Valle, R.; Vilaseca, I.; Lomeña, F.; Vilas, D.; et al. Neurodegenerative disease status and post-mortem pathology in idiopathic rapid-eye-movement sleep behaviour disorder: an observational cohort study. Lancet Neurol 2013, 12, 443–453. [Google Scholar] [CrossRef]
- George, J.M. The synucleins. Genome biology 2001, 3, 1–6. [Google Scholar] [CrossRef]
- Sharma, M.; Burré, J. α-Synuclein in synaptic function and dysfunction. Trends Neurosci 2023, 46, 153–166. [Google Scholar] [CrossRef]
- Maroteaux, L.; Campanelli, J.T.; Scheller, R.H. Synuclein: a neuron-specific protein localized to the nucleus and presynaptic nerve terminal. J Neurosci 1988, 8, 2804–2815. [Google Scholar] [CrossRef]
- Iwai, A.; Masliah, E.; Yoshimoto, M.; Ge, N.; Flanagan, L.; de Silva, H.A.; Kittel, A.; Saitoh, T. The precursor protein of non-A beta component of Alzheimer’s disease amyloid is a presynaptic protein of the central nervous system. Neuron 1995, 14, 467–475. [Google Scholar] [CrossRef]
- Lourenco, G.F.; Torres-Pacheco, M.E.; Fu, Y.; Li, H.; McCann, H.; Shepherd, C.E.; Kril, J.J.; Halliday, G.M. Nucleolar aggregation of key neuropathological proteins in the postmortem neurodegenerative brain. Acta Neuropathologica 2025, 150, 60. [Google Scholar] [CrossRef]
- Gao, V.; Briano, J.A.; Komer, L.E.; Burré, J. Functional and Pathological Effects of α-Synuclein on Synaptic SNARE Complexes. J Mol Biol 2023, 435, 167714. [Google Scholar] [CrossRef]
- Burré, J.; Sharma, M.; Südhof, T.C. α-Synuclein assembles into higher-order multimers upon membrane binding to promote SNARE complex formation. Proceedings of the National Academy of Sciences 2014, 111, E4274–E4283. [Google Scholar] [CrossRef]
- Wang, L.; Das, U.; Scott, D.A.; Tang, Y.; McLean, P.J.; Roy, S. α-synuclein multimers cluster synaptic vesicles and attenuate recycling. Current Biology 2014, 24, 2319–2326. [Google Scholar] [CrossRef]
- Diao, J.; Burré, J.; Vivona, S.; Cipriano, D.J.; Sharma, M.; Kyoung, M.; Südhof, T.C.; Brunger, A.T. Native α-synuclein induces clustering of synaptic-vesicle mimics via binding to phospholipids and synaptobrevin-2/VAMP2. elife 2013, 2, e00592. [Google Scholar] [CrossRef]
- Murphy, DD; Rueter, R.S.; Trojanowski, JQ; Lee, VM. Synucleins Are Developmentally Expressed, and a-Synuclein Regulates the Size of the Presynaptic Vesicular Pool in Primary Hippocampal Neurons.pdf. J Neurosci 2000. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Gallardo, G.; Fernandez-Chacon, R.; Schluter, O.M.; Sudhof, T.C. Alpha-synuclein cooperates with CSPalpha in preventing neurodegeneration. Cell 2005, 123, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Lieknina, I.; Reimer, L.; Pantelejevs, T.; Lends, A.; Jaudzems, K.; El-Turabi, A.; Gram, H.; Hammi, A.; Jensen, P.H.; Tars, K. Structural basis of epitope recognition by anti-alpha-synuclein antibodies MJFR14-6-4-2. NPJ Parkinsons Dis 2024, 10, 206. [Google Scholar] [CrossRef] [PubMed]
- Man, W.K.; Tahirbegi, B.; Vrettas, M.D.; Preet, S.; Ying, L.; Vendruscolo, M.; De Simone, A.; Fusco, G. The docking of synaptic vesicles on the presynaptic membrane induced by α-synuclein is modulated by lipid composition. Nature Communications 2021, 12, 927. [Google Scholar] [CrossRef] [PubMed]
- Murphy, D.D.; Rueter, S.M.; Trojanowski, J.Q.; Lee, V.M. Synucleins are developmentally expressed, and alpha-synuclein regulates the size of the presynaptic vesicular pool in primary hippocampal neurons. J Neurosci 2000, 20, 3214–3220. [Google Scholar] [CrossRef]
- Sun, J.; Wang, L.; Bao, H.; Premi, S.; Das, U.; Chapman, E.R.; Roy, S. Functional cooperation of α-synuclein and VAMP2 in synaptic vesicle recycling. Proceedings of the National Academy of Sciences 2019, 116, 11113–11115. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Liu, K.; Li, D.; Brunger, A.; Li, C.; Burré, J.; Diao, J. α-Synuclein condensation in synaptic vesicle function and synucleinopathies. Trends in Cell Biology 2025. [Google Scholar] [CrossRef] [PubMed]
- Man, W.K.; Tahirbegi, B.; Vrettas, M.D.; Preet, S.; Ying, L.; Vendruscolo, M.; De Simone, A.; Fusco, G. The docking of synaptic vesicles on the presynaptic membrane induced by α-synuclein is modulated by lipid composition. Nat Commun 2021, 12, 927. [Google Scholar] [CrossRef]
- Snead, D.; Eliezer, D. Alpha-synuclein function and dysfunction on cellular membranes. Exp Neurobiol 2014, 23, 292–313. [Google Scholar] [CrossRef]
- Vargas, K.J.; Makani, S.; Davis, T.; Westphal, C.H.; Castillo, P.E.; Chandra, S.S. Synucleins regulate the kinetics of synaptic vesicle endocytosis. Journal of Neuroscience 2014, 34, 9364–9376. [Google Scholar] [CrossRef]
- Perez, R.G.; Waymire, J.C.; Lin, E.; Liu, J.J.; Guo, F.; Zigmond, M.J. A role for α-synuclein in the regulation of dopamine biosynthesis. Journal of Neuroscience 2002, 22, 3090–3099. [Google Scholar] [CrossRef]
- Liu, D.; Jin, L.; Wang, H.; Zhao, H.; Zhao, C.; Duan, C.; Lu, L.; Wu, B.; Yu, S.; Chan, P. Silencing α-synuclein gene expression enhances tyrosine hydroxylase activity in MN9D cells. Neurochemical research 2008, 33, 1401–1409. [Google Scholar] [CrossRef]
- Liu, S.; Ninan, I.; Antonova, I.; Battaglia, F.; Trinchese, F.; Narasanna, A.; Kolodilov, N.; Dauer, W.; Hawkins, R.D.; Arancio, O. α-Synuclein produces a long-lasting increase in neurotransmitter release. The EMBO journal 2004, 23, 4506–4516. [Google Scholar] [CrossRef]
- Somayaji, M.; Cataldi, S.; Choi, S.J.; Edwards, R.H.; Mosharov, E.V.; Sulzer, D. A dual role for α-synuclein in facilitation and depression of dopamine release from substantia nigra neurons in vivo. Proceedings of the National Academy of Sciences 2020, 117, 32701–32710. [Google Scholar] [CrossRef]
- Sagredo, G.T.; Tanglay, O.; Shahdadpuri, S.; Fu, Y.; Halliday, G.M. ⍺-Synuclein levels in Parkinson’s disease–Cell types and forms that contribute to pathogenesis. Experimental Neurology 2024, 379, 114887. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Tanglay, O.; Su, F.; Li, H.; Liu, J.; Kim, W.S.; Halliday, G.M.; Fu, Y. Distinct AQP4 Alterations in Movement Disorders with Primary Synucleinopathy. Movement Disorders 2025, 40, 2804–2810. [Google Scholar] [CrossRef]
- Tanji, K.; Imaizumi, T.; Yoshida, H.; Mori, F.; Yoshimoto, M.; Satoh, K.; Wakabayashi, K. Expression of α-synuclein in a human glioma cell line and its up-regulation by interleukin-1β. Neuroreport 2001, 12, 1909–1912. [Google Scholar] [CrossRef]
- Batiuk, M.Y.; Martirosyan, A.; Wahis, J.; de Vin, F.; Marneffe, C.; Kusserow, C.; Koeppen, J.; Viana, J.F.; Oliveira, J.F.; Voet, T. Identification of region-specific astrocyte subtypes at single cell resolution. Nature communications 2020, 11, 1220. [Google Scholar] [CrossRef]
- Mori, F.; Tanji, K.; Yoshimoto, M.; Takahashi, H.; Wakabayashi, K. Demonstration of α-synuclein immunoreactivity in neuronal and glial cytoplasm in normal human brain tissue using proteinase K and formic acid pretreatment. Experimental neurology 2002, 176, 98–104. [Google Scholar] [CrossRef]
- Rostami, J.; Mothes, T.; Kolahdouzan, M.; Eriksson, O.; Moslem, M.; Bergström, J.; Ingelsson, M.; O’Callaghan, P.; Healy, L.M.; Falk, A. Crosstalk between astrocytes and microglia results in increased degradation of α-synuclein and amyloid-β aggregates. Journal of neuroinflammation 2021, 18, 124. [Google Scholar] [CrossRef]
- Hsiao, J.-H.T.; Tanglay, O.; Li, A.A.; Strobbe, A.Y.; Kim, W.S.; Halliday, G.M.; Fu, Y. Role of oligodendrocyte lineage cells in multiple system atrophy. Cells 2023, 12, 739. [Google Scholar] [CrossRef] [PubMed]
- Djelloul, M.; Holmqvist, S.; Boza-Serrano, A.; Azevedo, C.; Yeung, M.S.; Goldwurm, S.; Frisén, J.; Deierborg, T.; Roybon, L. Alpha-synuclein expression in the oligodendrocyte lineage: an in vitro and in vivo study using rodent and human models. Stem cell reports 2015, 5, 174–184. [Google Scholar] [CrossRef]
- Kaji, S.; Maki, T.; Kinoshita, H.; Uemura, N.; Ayaki, T.; Kawamoto, Y.; Furuta, T.; Urushitani, M.; Hasegawa, M.; Kinoshita, Y. Pathological endogenous α-synuclein accumulation in oligodendrocyte precursor cells potentially induces inclusions in multiple system atrophy. Stem cell reports 2018, 10, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Wenning, G.K.; Stefanova, N.; Jellinger, K.A.; Poewe, W.; Schlossmacher, M.G. Multiple system atrophy: a primary oligodendrogliopathy. Annals of Neurology: Official Journal of the American Neurological Association and the Child Neurology Society 2008, 64, 239–246. [Google Scholar] [CrossRef]
- Cho, M.K.; Nodet, G.; Kim, H.Y.; Jensen, M.R.; Bernado, P.; Fernandez, C.O.; Becker, S.; Blackledge, M.; Zweckstetter, M. Structural characterization of α-synuclein in an aggregation prone state. Protein Sci 2009, 18, 1840–1846. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, C.O.; Hoyer, W.; Zweckstetter, M.; Jares-Erijman, E.A.; Subramaniam, V.; Griesinger, C.; Jovin, T. NMR of alpha-synuclein-polyamine complexes elucidates the mechanism and kinetics of induced aggregation. EMBO J 2004, 23, 2039–2046. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, J.A.; Ivanova, M.I.; Sawaya, M.R.; Cascio, D.; Reyes, F.E.; Shi, D.; Sangwan, S.; Guenther, E.L.; Johnson, L.M.; Zhang, M. Structure of the toxic core of α-synuclein from invisible crystals. Nature 2015, 525, 486–490. [Google Scholar] [CrossRef]
- Zhang, C.; Pei, Y.; Zhang, Z.; Xu, L.; Liu, X.; Jiang, L.; Pielak, G.J.; Zhou, X.; Liu, M.; Li, C. C-terminal truncation modulates α-Synuclein’s cytotoxicity and aggregation by promoting the interactions with membrane and chaperone. Communications biology 2022, 5, 798. [Google Scholar] [CrossRef] [PubMed]
- Bartels, T.; Ahlstrom, L.S.; Leftin, A.; Kamp, F.; Haass, C.; Brown, M.F.; Beyer, K. The N-terminus of the intrinsically disordered protein α-synuclein triggers membrane binding and helix folding. Biophysical journal 2010, 99, 2116–2124. [Google Scholar] [CrossRef]
- Bussell, R., Jr.; Eliezer, D. A structural and functional role for 11-mer repeats in α-synuclein and other exchangeable lipid binding proteins. J Mol Biol 2003, 329, 763–778. [Google Scholar] [CrossRef]
- Davidson, W.S.; Jonas, A.; Clayton, D.F.; George, J.M. Stabilization of alpha-synuclein secondary structure upon binding to synthetic membranes. J Biol Chem 1998, 273, 9443–9449. [Google Scholar] [CrossRef]
- Eliezer, D.; Kutluay, E.; Bussell, R., Jr.; Browne, G. Conformational properties of α-synuclein in its free and lipid-associated states. Journal of molecular biology 2001, 307, 1061–1073. [Google Scholar] [CrossRef]
- Jo, E.; McLaurin, J.; Yip, C.M.; George-Hyslop, P.S.; Fraser, P.E. alpha-Synuclein membrane interactions and lipid specificity. J Biol Chem 2000, 275, 34328–34334. [Google Scholar] [CrossRef]
- Bussell, R.; Eliezer, D. Effects of Parkinson’s disease-linked mutations on the structure of lipid-associated α-synuclein. Biochemistry 2004, 43, 4810–4818. [Google Scholar] [CrossRef]
- Pfefferkorn, C.M.; Jiang, Z.; Lee, J. Biophysics of α-synuclein membrane interactions. Biochim Biophys Acta 2012, 1818, 162–171. [Google Scholar] [CrossRef]
- Fortin, D.L.; Troyer, M.D.; Nakamura, K.; Kubo, S.; Anthony, M.D.; Edwards, R.H. Lipid rafts mediate the synaptic localization of alpha-synuclein. J Neurosci 2004, 24, 6715–6723. [Google Scholar] [CrossRef]
- Pranke, I.M.; Morello, V.; Bigay, J.; Gibson, K.; Verbavatz, J.-M.; Antonny, B.; Jackson, C.L. α-Synuclein and ALPS motifs are membrane curvature sensors whose contrasting chemistry mediates selective vesicle binding. Journal of Cell Biology 2011, 194, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Drin, G.; Antonny, B. Amphipathic helices and membrane curvature. FEBS letters 2010, 584, 1840–1847. [Google Scholar] [CrossRef] [PubMed]
- Takamori, S.; Holt, M.; Stenius, K.; Lemke, E.A.; Grønborg, M.; Riedel, D.; Urlaub, H.; Schenck, S.; Brügger, B.; Ringler, P. Molecular anatomy of a trafficking organelle. Cell 2006, 127, 831–846. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Gallardo, G.; Fernández-Chacón, R.; Schlüter, O.M.; Südhof, T.C. α-Synuclein cooperates with CSPα in preventing neurodegeneration. Cell 2005, 123, 383–396. [Google Scholar] [CrossRef]
- Wang, C.; Tu, J.; Zhang, S.; Cai, B.; Liu, Z.; Hou, S.; Zhong, Q.; Hu, X.; Liu, W.; Li, G.; et al. Different regions of synaptic vesicle membrane regulate VAMP2 conformation for the SNARE assembly. Nature Communications 2020, 11, 1531. [Google Scholar] [CrossRef]
- Ulmer, T.S.; Bax, A.; Cole, N.B.; Nussbaum, R.L. Structure and dynamics of micelle-bound human α-synuclein. Journal of Biological Chemistry 2005, 280, 9595–9603. [Google Scholar] [CrossRef]
- Yu, S.; Li, X.; Liu, G.; Han, J.; Zhang, C.; Li, Y.; Xu, S.; Liu, C.; Gao, Y.; Yang, H. Extensive nuclear localization of α-synuclein in normal rat brain neurons revealed by a novel monoclonal antibody. Neuroscience 2007, 145, 539–555. [Google Scholar] [CrossRef]
- Mori, F.; Tanji, K.; Yoshimoto, M.; Takahashi, H.; Wakabayashi, K. Immunohistochemical comparison of α-and β-synuclein in adult rat central nervous system. Brain research 2002, 941, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Schell, H.; Hasegawa, T.; Neumann, M.; Kahle, P. Nuclear and neuritic distribution of serine-129 phosphorylated α-synuclein in transgenic mice. Neuroscience 2009, 160, 796–804. [Google Scholar] [CrossRef]
- Koss, D.J.; Erskine, D.; Porter, A.; Palmoski, P.; Menon, H.; Todd, O.G.; Leite, M.; Attems, J.; Outeiro, T.F. Nuclear alpha-synuclein is present in the human brain and is modified in dementia with Lewy bodies. Acta Neuropathologica Communications 2022, 10, 98. [Google Scholar] [CrossRef]
- Cherny, D.; Hoyer, W.; Subramaniam, V.; Jovin, T.M. Double-stranded DNA stimulates the fibrillation of α-synuclein in vitro and is associated with the mature fibrils: an electron microscopy study. Journal of molecular biology 2004, 344, 929–938. [Google Scholar] [CrossRef] [PubMed]
- Schaser, A.J.; Osterberg, V.R.; Dent, S.E.; Stackhouse, T.L.; Wakeham, C.M.; Boutros, S.W.; Weston, L.J.; Owen, N.; Weissman, T.A.; Luna, E. Alpha-synuclein is a DNA binding protein that modulates DNA repair with implications for Lewy body disorders. Scientific reports 2019, 9, 10919. [Google Scholar] [CrossRef]
- Goers, J.; Manning-Bog, A.B.; McCormack, A.L.; Millett, I.S.; Doniach, S.; Di Monte, D.A.; Uversky, V.N.; Fink, A.L. Nuclear localization of α-synuclein and its interaction with histones. Biochemistry 2003, 42, 8465–8471. [Google Scholar] [CrossRef]
- Paiva, I.; Pinho, R.; Pavlou, M.A.; Hennion, M.; Wales, P.; Schütz, A.-L.; Rajput, A.; Szegő, É.M.; Kerimoglu, C.; Gerhardt, E. Sodium butyrate rescues dopaminergic cells from alpha-synuclein-induced transcriptional deregulation and DNA damage. Human molecular genetics 2017, 26, 2231–2246. [Google Scholar] [CrossRef] [PubMed]
- Poewe, W.; Stankovic, I.; Halliday, G.; Meissner, W.G.; Wenning, G.K.; Pellecchia, M.T.; Seppi, K.; Palma, J.A.; Kaufmann, H. Multiple system atrophy. Nat Rev Dis Primers 2022, 8, 56. [Google Scholar] [CrossRef]
- Spillantini, M.G.; Schmidt, M.L.; Lee, V.M.Y.; Trojanowski, J.Q.; Jakes, R.; Goedert, M. α-Synuclein in Lewy bodies. Nature 1997, 388, 839–840. [Google Scholar] [CrossRef]
- Spillantini, M.G.; Crowther, R.A.; Jakes, R.; Hasegawa, M.; Goedert, M. alpha-Synuclein in filamentous inclusions of Lewy bodies from Parkinson’s disease and dementia with lewy bodies. Proc Natl Acad Sci U S A 1998, 95, 6469–6473. [Google Scholar] [CrossRef]
- Tanaka, M.T.; Miki, Y.; Kon, T.; Mori, F.; Wakabayashi, K. Pathological and Molecular Insights into the Early Stage of Multiple System Atrophy. Cells 2025, 14, 1966. [Google Scholar] [CrossRef]
- Song, Y.J.; Lundvig, D.M.; Huang, Y.; Gai, W.P.; Blumbergs, P.C.; Hojrup, P.; Otzen, D.; Halliday, G.M.; Jensen, P.H. p25alpha relocalizes in oligodendroglia from myelin to cytoplasmic inclusions in multiple system atrophy. Am J Pathol 2007, 171, 1291–1303. [Google Scholar] [CrossRef]
- Kon, T.; Mori, F.; Tanji, K.; Miki, Y.; Wakabayashi, K. An autopsy case of preclinical multiple system atrophy (MSA-C). Neuropathology 2013, 33, 667–672. [Google Scholar] [CrossRef]
- Lin, W.-L.; DeLucia, M.W.; Dickson, D.W. α-Synuclein immunoreactivity in neuronal nuclear inclusions and neurites in multiple system atrophy. Neuroscience letters 2004, 354, 99–102. [Google Scholar] [CrossRef]
- Wiseman, J.A.; Halliday, G.M.; Dieriks, B.V. Neuronal α-synuclein toxicity is the key driver of neurodegeneration in multiple system atrophy. Brain 2025, awaf030. [Google Scholar] [CrossRef]
- Li, W.W.; Yang, R.; Guo, J.C.; Ren, H.M.; Zha, X.L.; Cheng, J.S.; Cai, D.F. Localization of alpha-synuclein to mitochondria within midbrain of mice. Neuroreport 2007, 18, 1543–1546. [Google Scholar] [CrossRef]
- Ludtmann, M.H.; Angelova, P.R.; Ninkina, N.N.; Gandhi, S.; Buchman, V.L.; Abramov, A.Y. Monomeric alpha-synuclein exerts a physiological role on brain ATP synthase. Journal of Neuroscience 2016, 36, 10510–10521. [Google Scholar] [CrossRef]
- Robotta, M.; Gerding, H.R.; Vogel, A.; Hauser, K.; Schildknecht, S.; Karreman, C.; Leist, M.; Subramaniam, V.; Drescher, M. Alpha-Synuclein binds to the inner membrane of mitochondria in an α-helical conformation. Chembiochem 2014, 15, 2499–2502. [Google Scholar] [CrossRef] [PubMed]
- Devi, L.; Raghavendran, V.; Prabhu, B.M.; Avadhani, N.G.; Anandatheerthavarada, H.K. Mitochondrial import and accumulation of α-synuclein impair complex I in human dopaminergic neuronal cultures and Parkinson disease brain. Journal of Biological Chemistry 2008, 283, 9089–9100. [Google Scholar] [CrossRef] [PubMed]
- Ghio, S.; Kamp, F.; Cauchi, R.; Giese, A.; Vassallo, N. Interaction of α-synuclein with biomembranes in Parkinson’s disease—role of cardiolipin. Progress in lipid research 2016, 61, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Nemani, V.M.; Wallender, E.K.; Kaehlcke, K.; Ott, M.; Edwards, R.H. Optical reporters for the conformation of α-synuclein reveal a specific interaction with mitochondria. Journal of Neuroscience 2008, 28, 12305–12317. [Google Scholar] [CrossRef] [PubMed]
- Brown, H.J.; Fan, R.Z.; Bell, R.; Salehe, S.S.; Martínez, C.M.; Lai, Y.; Tieu, K. Imbalanced mitochondrial dynamics in human PD and α-synuclein mouse brains. Neurobiology of Disease 2025, 106976. [CrossRef]
- Vicario, M.; Cieri, D.; Brini, M.; Calì, T. The close encounter between alpha-synuclein and mitochondria. Frontiers in Neuroscience 2018, 12, 388. [Google Scholar] [CrossRef] [PubMed]
- Lurette, O.; Martín-Jiménez, R.; Khan, M.; Sheta, R.; Jean, S.; Schofield, M.; Teixeira, M.; Rodriguez-Aller, R.; Perron, I.; Oueslati, A. Aggregation of alpha-synuclein disrupts mitochondrial metabolism and induce mitophagy via cardiolipin externalization. Cell death & disease 2023, 14, 729. [Google Scholar]
- Ramezani, M.; Wagenknecht-Wiesner, A.; Wang, T.; Holowka, D.A.; Eliezer, D.; Baird, B.A. Alpha synuclein modulates mitochondrial Ca2+ uptake from ER during cell stimulation and under stress conditions. npj Parkinson’s Disease 2023, 9, 137. [Google Scholar] [CrossRef]
- Guardia-Laguarta, C.; Area-Gomez, E.; Rüb, C.; Liu, Y.; Magrané, J.; Becker, D.; Voos, W.; Schon, E.A.; Przedborski, S. α-Synuclein is localized to mitochondria-associated ER membranes. Journal of Neuroscience 2014, 34, 249–259. [Google Scholar] [CrossRef]
- Zeng, H.; Liu, Y.; Liu, X.; Li, J.; Lu, L.; Xue, C.; Wu, X.; Zhang, X.; Zheng, Z.; Lu, G. Interplay of α-Synuclein Oligomers and Endoplasmic Reticulum Stress in Parkinson’S Disease: Insights into Cellular Dysfunctions. Inflammation 2025, 48, 1590–1606. [Google Scholar] [CrossRef]
- Cooper, A.A.; Gitler, A.D.; Cashikar, A.; Haynes, C.M.; Hill, K.J.; Bhullar, B.; Liu, K.; Xu, K.; Strathearn, K.E.; Liu, F. α-Synuclein blocks ER-Golgi traffic and Rab1 rescues neuron loss in Parkinson’s models. Science 2006, 313, 324–328. [Google Scholar] [CrossRef]
- Thayanidhi, N.; Helm, J.R.; Nycz, D.C.; Bentley, M.; Liang, Y.; Hay, J.C. α-Synuclein delays endoplasmic reticulum (ER)-to-Golgi transport in mammalian cells by antagonizing ER/Golgi SNAREs. Molecular biology of the cell 2010, 21, 1850–1863. [Google Scholar] [CrossRef]
- Gitler, A.D.; Bevis, B.J.; Shorter, J.; Strathearn, K.E.; Hamamichi, S.; Su, L.J.; Caldwell, K.A.; Caldwell, G.A.; Rochet, J.-C.; McCaffery, J.M. The Parkinson’s disease protein α-synuclein disrupts cellular Rab homeostasis. Proceedings of the National Academy of Sciences 2008, 105, 145–150. [Google Scholar] [CrossRef]
- Mazzulli, J.R.; Zunke, F.; Tsunemi, T.; Toker, N.J.; Jeon, S.; Burbulla, L.F.; Patnaik, S.; Sidransky, E.; Marugan, J.J.; Sue, C.M.; et al. Activation of β-Glucocerebrosidase Reduces Pathological α-Synuclein and Restores Lysosomal Function in Parkinson’s Patient Midbrain Neurons. J Neurosci 2016, 36, 7693–7706. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-J.; Khoshaghideh, F.; Patel, S.; Lee, S.-J. Clearance of α-synuclein oligomeric intermediates via the lysosomal degradation pathway. Journal of Neuroscience 2004, 24, 1888–1896. [Google Scholar] [CrossRef]
- Sanyal, A.; Scanavachi, G.; Somerville, E.; Saminathan, A.; Nair, A.; Bango Da Cunha Correia, R.F.; Aylan, B.; Sitarska, E.; Oikonomou, A.; Hatzakis, N.S. Neuronal constitutive endolysosomal perforations enable α-synuclein aggregation by internalized PFFs. Journal of Cell Biology 2024, 224, e202401136. [Google Scholar] [CrossRef]
- Masaracchia, C.; Hnida, M.; Gerhardt, E.; Lopes da Fonseca, T.; Villar-Pique, A.; Branco, T.; Stahlberg, M.A.; Dean, C.; Fernández, C.O.; Milosevic, I. Membrane binding, internalization, and sorting of alpha-synuclein in the cell. Acta neuropathologica communications 2018, 6, 79. [Google Scholar] [CrossRef]
- Breda, C.; Nugent, M.L.; Estranero, J.G.; Kyriacou, C.P.; Outeiro, T.F.; Steinert, J.R.; Giorgini, F. Rab11 modulates α-synuclein-mediated defects in synaptic transmission and behaviour. Human molecular genetics 2015, 24, 1077–1091. [Google Scholar] [CrossRef]
- Keable, R.; Hu, S.; Pfundstein, G.; Kozlova, I.; Su, F.; Du, X.; Yang, H.; Gunnersen, J.; Schachner, M.; Leshchyns’ ka, I. The BACE1-generated C-terminal fragment of the neural cell adhesion molecule 2 (NCAM2) promotes BACE1 targeting to Rab11-positive endosomes. Cellular and Molecular Life Sciences 2022, 79, 555. [Google Scholar] [CrossRef] [PubMed]
- Su, F.; Pfundstein, G.; Sah, S.; Zhang, S.; Keable, R.; Hagan, D.W.; Sharpe, L.J.; Clemens, K.J.; Begg, D.; Phelps, E.A. Neuronal growth regulator 1 (NEGR1) promotes the synaptic targeting of glutamic acid decarboxylase 65 (GAD65). Journal of Neurochemistry 2025, 169, e16279. [Google Scholar] [CrossRef]
- Yuan, L.; Mao, L.-H.; Huang, Y.-Y.; Outeiro, T.F.; Li, W.; Vieira, T.C.; Li, J.-Y. Stress granules: emerging players in neurodegenerative diseases. Translational neurodegeneration 2025, 14, 22. [Google Scholar] [CrossRef]
- Hallacli, E.; Kayatekin, C.; Nazeen, S.; Wang, X.H.; Sheinkopf, Z.; Sathyakumar, S.; Sarkar, S.; Jiang, X.; Dong, X.; Di Maio, R. The Parkinson’s disease protein alpha-synuclein is a modulator of processing bodies and mRNA stability. Cell 2022, 185, 2035–2056. e2033. [Google Scholar] [CrossRef]
- Asamitsu, S.; Yabuki, Y.; Matsuo, K.; Kawasaki, M.; Hirose, Y.; Kashiwazaki, G.; Chandran, A.; Bando, T.; Wang, D.O.; Sugiyama, H. RNA G-quadruplex organizes stress granule assembly through DNAPTP6 in neurons. Science Advances 2023, 9, eade2035. [Google Scholar] [CrossRef] [PubMed]
- Brangwynne, C.P.; Eckmann, C.R.; Courson, D.S.; Rybarska, A.; Hoege, C.; Gharakhani, J.; Jülicher, F.; Hyman, A.A. Germline P granules are liquid droplets that localize by controlled dissolution/condensation. Science 2009, 324, 1729–1732. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.; Brangwynne, C.P. Liquid phase condensation in cell physiology and disease. Science 2017, 357, eaaf4382. [Google Scholar] [CrossRef] [PubMed]
- Wolozin, B.; Ivanov, P. Stress granules and neurodegeneration. Nature Reviews Neuroscience 2019, 20, 649–666. [Google Scholar] [CrossRef]
- Matsuo, K.; Asamitsu, S.; Maeda, K.; Suzuki, H.; Kawakubo, K.; Komiya, G.; Kudo, K.; Sakai, Y.; Hori, K.; Ikenoshita, S. RNA G-quadruplexes form scaffolds that promote neuropathological α-synuclein aggregation. Cell 2024, 187, 6835–6848. e6820. [Google Scholar] [CrossRef]
- Rupert, J.; Monti, M.; Zacco, E.; Tartaglia, G.G. RNA sequestration driven by amyloid formation: the alpha synuclein case. Nucleic Acids Research 2023, 51, 11466–11478. [Google Scholar] [CrossRef] [PubMed]
- Chandra, S.; Chen, X.; Rizo, J.; Jahn, R.; Sudhof, T.C. A broken α-helix in folded α-synuclein. Journal of Biological Chemistry 2003, 278, 15313–15318. [Google Scholar] [CrossRef]
- Gao, V.; Briano, J.A.; Komer, L.E.; Burré, J. Functional and pathological effects of α-synuclein on synaptic SNARE complexes. Journal of molecular biology 2023, 435, 167714. [Google Scholar] [CrossRef]
- Ferreon, A.C.M.; Gambin, Y.; Lemke, E.A.; Deniz, A.A. Interplay of α-synuclein binding and conformational switching probed by single-molecule fluorescence. Proceedings of the National Academy of Sciences 2009, 106, 5645–5650. [Google Scholar] [CrossRef]
- Fusco, G.; Pape, T.; Stephens, A.D.; Mahou, P.; Costa, A.R.; Kaminski, C.F.; Kaminski Schierle, G.S.; Vendruscolo, M.; Veglia, G.; Dobson, C.M. Structural basis of synaptic vesicle assembly promoted by α-synuclein. Nature communications 2016, 7, 12563. [Google Scholar] [CrossRef]
- Bartels, T.; Choi, J.G.; Selkoe, D.J. α-Synuclein occurs physiologically as a helically folded tetramer that resists aggregation. Nature 2011, 477, 107–110. [Google Scholar] [CrossRef]
- De Boni, L.; Watson, A.H.; Zaccagnini, L.; Wallis, A.; Zhelcheska, K.; Kim, N.; Sanderson, J.; Jiang, H.; Martin, E.; Cantlon, A. Brain region-specific susceptibility of Lewy body pathology in synucleinopathies is governed by α-synuclein conformations. Acta Neuropathologica 2022, 143, 453–469. [Google Scholar] [CrossRef]
- Román-Vendrell, C.; Medeiros, A.T.; Sanderson, J.B.; Jiang, H.; Bartels, T.; Morgan, J.R. Effects of excess brain-derived human α-synuclein on synaptic vesicle trafficking. Frontiers in neuroscience 2021, 15, 639414. [Google Scholar] [CrossRef]
- Hardenberg, M.C.; Sinnige, T.; Casford, S.; Dada, S.T.; Poudel, C.; Robinson, E.A.; Fuxreiter, M.; Kaminksi, C.F.; Kaminski Schierle, G.S.; Nollen, E.A. Observation of an α-synuclein liquid droplet state and its maturation into Lewy body-like assemblies. Journal of molecular cell biology 2021, 13, 282–294. [Google Scholar] [CrossRef]
- Piroska, L.; Fenyi, A.; Thomas, S.; Plamont, M.-A.; Redeker, V.; Melki, R.; Gueroui, Z. α-Synuclein liquid condensates fuel fibrillar α-synuclein growth. Science Advances 2023, 9, eadg5663. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.; Singh, N.; Kumar, R.; Patel, K.; Pandey, S.; Datta, D.; Mahato, J.; Panigrahi, R.; Navalkar, A.; Mehra, S. α-Synuclein aggregation nucleates through liquid–liquid phase separation. Nature chemistry 2020, 12, 705–716. [Google Scholar] [CrossRef]
- Ma, D.-F.; Zhang, S.; Xu, S.-Y.; Huang, Z.; Tao, Y.; Chen, F.; Zhang, S.; Li, D.; Chen, T.; Liu, C. Self-limiting multimerization of α-synuclein on membrane and its implication in Parkinson’s diseases. Science Advances 2024, 10, eado4893. [Google Scholar] [CrossRef] [PubMed]
- Gautier, R.; Bacle, A.; Tiberti, M.L.; Fuchs, P.F.; Vanni, S.; Antonny, B. PackMem: a versatile tool to compute and visualize interfacial packing defects in lipid bilayers. Biophysical journal 2018, 115, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.H.; Kou, O.H.; White, J.M.; Ramirez, S.Y.; Margaritakis, A.; Chung, P.J.; Jaeger, V.W.; Zeno, W.F. Lipid Packing Defects are Necessary and Sufficient for Membrane Binding of α-Synuclein. bioRxiv 2014, 2025, 2024. [Google Scholar] [CrossRef]
- Vanni, S.; Hirose, H.; Barelli, H.; Antonny, B.; Gautier, R. A sub-nanometre view of how membrane curvature and composition modulate lipid packing and protein recruitment. Nature communications 2014, 5, 4916. [Google Scholar] [CrossRef]
- Cremades, N.; Cohen, S.I.; Deas, E.; Abramov, A.Y.; Chen, A.Y.; Orte, A.; Sandal, M.; Clarke, R.W.; Dunne, P.; Aprile, F.A. Direct observation of the interconversion of normal and toxic forms of α-synuclein. Cell 2012, 149, 1048–1059. [Google Scholar] [CrossRef]
- Forloni, G. Alpha synuclein: neurodegeneration and inflammation. International journal of molecular sciences 2023, 24, 5914. [Google Scholar] [CrossRef]
- Sonustun, B.; Altay, M.F.; Strand, C.; Ebanks, K.; Hondhamuni, G.; Warner, T.T.; Lashuel, H.A.; Bandopadhyay, R. Pathological Relevance of Post-Translationally Modified Alpha-Synuclein (pSer87, pSer129, nTyr39) in Idiopathic Parkinson’s Disease and Multiple System Atrophy. Cells 2022, 11. [Google Scholar] [CrossRef] [PubMed]
- Altay, M.F.; Kumar, S.T.; Burtscher, J.; Jagannath, S.; Strand, C.; Miki, Y.; Parkkinen, L.; Holton, J.L.; Lashuel, H.A. Development and validation of an expanded antibody toolset that captures alpha-synuclein pathological diversity in Lewy body diseases. npj Parkinson’s Disease 2023, 9, 161. [Google Scholar] [CrossRef] [PubMed]
- Paleologou, K.E.; Oueslati, A.; Shakked, G.; Rospigliosi, C.C.; Kim, H.-Y.; Lamberto, G.R.; Fernandez, C.O.; Schmid, A.; Chegini, F.; Gai, W.P. Phosphorylation at S87 is enhanced in synucleinopathies, inhibits α-synuclein oligomerization, and influences synuclein-membrane interactions. Journal of Neuroscience 2010, 30, 3184–3198. [Google Scholar] [CrossRef] [PubMed]
- Hassanzadeh, K.; Liu, J.; Maddila, S.; Mouradian, M.M. Posttranslational modifications of α-Synuclein, their therapeutic potential, and crosstalk in health and neurodegenerative diseases. Pharmacological Reviews 2024, 76, 1254–1290. [Google Scholar] [CrossRef]
- Zhang, S.; Zhu, R.; Pan, B.; Xu, H.; Olufemi, M.F.; Gathagan, R.J.; Li, Y.; Zhang, L.; Zhang, J.; Xiang, W. Post-translational modifications of soluble α-synuclein regulate the amplification of pathological α-synuclein. Nature neuroscience 2023, 26, 213–225. [Google Scholar] [CrossRef]
- Dikiy, I.; Eliezer, D. N-terminal acetylation stabilizes N-terminal helicity in lipid-and micelle-bound α-synuclein and increases its affinity for physiological membranes. Journal of Biological Chemistry 2014, 289, 3652–3665. [Google Scholar] [CrossRef]
- Theillet, F.-X.; Binolfi, A.; Bekei, B.; Martorana, A.; Rose, H.M.; Stuiver, M.; Verzini, S.; Lorenz, D.; Van Rossum, M.; Goldfarb, D. Structural disorder of monomeric α-synuclein persists in mammalian cells. Nature 2016, 530, 45–50. [Google Scholar] [CrossRef]
- Bartels, T.; Choi, J.G.; Selkoe, D.J. α-Synuclein occurs physiologically as a helically folded tetramer that resists aggregation. Nature 2011, 477, 107–110. [Google Scholar] [CrossRef]
- Huang, F.; Yan, J.; Xu, H.; Wang, Y.; Zhang, X.; Zou, Y.; Lian, J.; Ding, F.; Sun, Y. Exploring the Impact of Physiological C-Terminal Truncation on α-Synuclein Conformations to Unveil Mechanisms Regulating Pathological Aggregation. J Chem Inf Model 2024, 64, 8616–8627. [Google Scholar] [CrossRef]
- Sorrentino, Z.A.; Giasson, B.I. The emerging role of α-synuclein truncation in aggregation and disease. Journal of Biological Chemistry 2020, 295, 10224–10244. [Google Scholar] [CrossRef]
- Anderson, J.P.; Walker, D.E.; Goldstein, J.M.; de Laat, R.; Banducci, K.; Caccavello, R.J.; Barbour, R.; Huang, J.; Kling, K.; Lee, M.; et al. Phosphorylation of Ser-129 is the dominant pathological modification of alpha-synuclein in familial and sporadic Lewy body disease. J Biol Chem 2006, 281, 29739–29752. [Google Scholar] [CrossRef]
- Zhang, S.; Zhu, R.; Pan, B.; Xu, H.; Olufemi, M.F.; Gathagan, R.J.; Li, Y.; Zhang, L.; Zhang, J.; Xiang, W.; et al. Post-translational modifications of soluble α-synuclein regulate the amplification of pathological α-synuclein. Nat Neurosci 2023, 26, 213–225. [Google Scholar] [CrossRef]
- Mahul-Mellier, A.-L.; Fauvet, B.; Gysbers, A.; Dikiy, I.; Oueslati, A.; Georgeon, S.; Lamontanara, A.J.; Bisquertt, A.; Eliezer, D.; Masliah, E. c-Abl phosphorylates α-synuclein and regulates its degradation: implication for α-synuclein clearance and contribution to the pathogenesis of Parkinson’s disease. Human molecular genetics 2014, 23, 2858–2879. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Vaidya, B.; McInnes, J.; Zoghbi, H.Y. Alpha-Synuclein Phosphomimetic Y39E and S129D Knock-In Mice Show Cytosolic Alpha-Synuclein Localization without Developing Neurodegeneration or Motor Deficits. eneuro 2025, 12. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Lim, Y.-J.; Liu, Z.; Long, H.; Sun, Y.; Hu, J.-J.; Zhao, C.; Tao, Y.; Zhang, X.; Li, D. Parkinson’s disease-related phosphorylation at Tyr39 rearranges α-synuclein amyloid fibril structure revealed by cryo-EM. Proceedings of the National Academy of Sciences 2020, 117, 20305–20315. [Google Scholar] [CrossRef]
- Dikiy, I.; Fauvet, B.; Jovičić, A.; Mahul-Mellier, A.L.; Desobry, C.; El-Turk, F.; Gitler, A.D.; Lashuel, H.A.; Eliezer, D. Semisynthetic and in Vitro Phosphorylation of Alpha-Synuclein at Y39 Promotes Functional Partly Helical Membrane-Bound States Resembling Those Induced by PD Mutations. ACS Chem Biol 2016, 11, 2428–2437. [Google Scholar] [CrossRef]
- Das, D.; Mattaparthi, V.S.K. Conformational Dynamics of Post-Translational-Modified α-Synuclein (pY39 and pS87) and its Interaction with Lipid Membrane. Current Biotechnology 2024, 13, 119–130. [Google Scholar] [CrossRef]
- Sevcsik, E.; Trexler, A.J.; Dunn, J.M.; Rhoades, E. Allostery in a disordered protein: oxidative modifications to α-synuclein act distally to regulate membrane binding. Journal of the American Chemical Society 2011, 133, 7152–7158. [Google Scholar] [CrossRef] [PubMed]
- Burai, R.; Ait-Bouziad, N.; Chiki, A.; Lashuel, H.A. Elucidating the role of site-specific nitration of α-synuclein in the pathogenesis of Parkinson’s disease via protein semisynthesis and mutagenesis. Journal of the American Chemical Society 2015, 137, 5041–5052. [Google Scholar] [CrossRef]
- Otzen, D.E.; Gamon, L.F.; Hägglund, P.; Nielsen, J.; Pedersen, J.N.; Nybo, T.; Nowak, J.S.; Amstrup, S.K.; Pirhaghi, M.; Davies, M.J. Nitrated products formed on α-synuclein are preferentially incorporated into oligomers but excluded from fibrils: A mechanism for accumulation of neurotoxic species. Biochimica et Biophysica Acta (BBA)-Proteins and Proteomics 2025, 141118. [CrossRef]
- Paleologou, K.E.; Oueslati, A.; Shakked, G.; Rospigliosi, C.C.; Kim, H.Y.; Lamberto, G.R.; Fernandez, C.O.; Schmid, A.; Chegini, F.; Gai, W.P.; et al. Phosphorylation at S87 is enhanced in synucleinopathies, inhibits alpha-synuclein oligomerization, and influences synuclein-membrane interactions. J Neurosci 2010, 30, 3184–3198. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Schilderink, N.; Subramaniam, V.; Huber, M. Membrane Binding of Parkinson’s Protein α-Synuclein: Effect of Phosphorylation at Positions 87 and 129 by the S to D Mutation Approach. Isr J Chem 2017, 57, 762–770. [Google Scholar] [CrossRef]
- Ramalingam, N.; Jin, S.-X.; Moors, T.E.; Fonseca-Ornelas, L.; Shimanaka, K.; Lei, S.; Cam, H.P.; Watson, A.H.; Brontesi, L.; Ding, L. Dynamic physiological α-synuclein S129 phosphorylation is driven by neuronal activity. npj Parkinson’s Disease 2023, 9, 4. [Google Scholar] [CrossRef]
- Paleologou, K.E.; Schmid, A.W.; Rospigliosi, C.C.; Kim, H.Y.; Lamberto, G.R.; Fredenburg, R.A.; Lansbury, P.T., Jr.; Fernandez, C.O.; Eliezer, D.; Zweckstetter, M.; et al. Phosphorylation at Ser-129 but not the phosphomimics S129E/D inhibits the fibrillation of alpha-synuclein. J Biol Chem 2008, 283, 16895–16905. [Google Scholar] [CrossRef]
- Sorrentino, Z.A.; Vijayaraghavan, N.; Gorion, K.M.; Riffe, C.J.; Strang, K.H.; Caldwell, J.; Giasson, B.I. Physiological C-terminal truncation of α-synuclein potentiates the prion-like formation of pathological inclusions. J Biol Chem 2018, 293, 18914–18932. [Google Scholar] [CrossRef]
- Mahul-Mellier, A.-L.; Altay, M.F.; Maharjan, N.; Ait-Bouziad, N.; Chiki, A.; Jagannath, S.; Limorenko, G.; Novello, S.; Ricci, J.; Jasiqi, Y. Differential role of C-terminal truncations on alpha-synuclein pathology and Lewy body formation. npj Parkinson’s Disease 2025, 11, 261. [Google Scholar] [CrossRef]
- Zenko, D.; Marsh, J.; Castle, A.R.; Lewin, R.; Fischer, R.; Tofaris, G.K. Monitoring α-synuclein ubiquitination dynamics reveals key endosomal effectors mediating its trafficking and degradation. Science Advances 2023, 9, eadd8910. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.; Chan, H.Y.E.; Lee, M.M.; Chan, M.K. A SUMO1-derived peptide targeting SUMO-interacting motif inhibits α-Synuclein aggregation. Cell chemical biology 2021, 28, 180–190. e186. [Google Scholar] [CrossRef] [PubMed]
- Burke, R.E.; Dauer, W.T.; Vonsattel, J.P.G. A critical evaluation of the Braak staging scheme for Parkinson’s disease. Annals of Neurology: Official Journal of the American Neurological Association and the Child Neurology Society 2008, 64, 485–491. [Google Scholar] [CrossRef]
- Beach, T.G.; Adler, C.H.; Sue, L.I.; Shill, H.A.; Driver-Dunckley, E.; Mehta, S.H.; Intorcia, A.J.; Glass, M.J.; Walker, J.E.; Arce, R. Vagus nerve and stomach synucleinopathy in Parkinson’s disease, incidental Lewy body disease, and normal elderly subjects: evidence against the “body-first” hypothesis. Journal of Parkinson’s Disease 2021, 11, 1833–1843. [Google Scholar] [CrossRef]
- Borghammer, P.; Van Den Berge, N. Brain-first versus gut-first Parkinson’s disease: a hypothesis. Journal of Parkinson’s disease 2019, 9, S281–S295. [Google Scholar] [CrossRef]
- Borghammer, P. The α-synuclein origin and connectome model (SOC Model) of Parkinson’s disease: explaining motor asymmetry, non-motor phenotypes, and cognitive decline. Journal of Parkinson’s Disease 2021, 11, 455–474. [Google Scholar] [CrossRef]
- Pluta, S.R.; Telian, G.I.; Naka, A.; Adesnik, H. Superficial Layers Suppress the Deep Layers to Fine-tune Cortical Coding. J Neurosci 2019, 39, 2052–2064. [Google Scholar] [CrossRef]
- Hooks, B.M.; Mao, T.; Gutnisky, D.A.; Yamawaki, N.; Svoboda, K.; Shepherd, G.M. Organization of cortical and thalamic input to pyramidal neurons in mouse motor cortex. Journal of Neuroscience 2013, 33, 748–760. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.F.; Dutta, S.; Scott, A.N.; Xiao, S.; Yadav, S.; Chen, X.; Aryal, U.K.; Kinzer-Ursem, T.L.; Rochet, J.-C.; Jayant, K. Site-specific seeding of Lewy pathology induces distinct pre-motor cellular and dendritic vulnerabilities in the cortex. Nature Communications 2024, 15, 10775. [Google Scholar] [CrossRef] [PubMed]
- Bloch, A.; Probst, A.; Bissig, H.; Adams, H.; Tolnay, M. α-Synuclein pathology of the spinal and peripheral autonomic nervous system in neurologically unimpaired elderly subjects. Neuropathology and applied neurobiology 2006, 32, 284–295. [Google Scholar] [CrossRef]
- Blandini, F.; Nappi, G.; Tassorelli, C.; Martignoni, E. Functional changes of the basal ganglia circuitry in Parkinson’s disease. Prog Neurobiol 2000, 62, 63–88. [Google Scholar] [CrossRef]
- Wang, Y.; Jiang, Z.; Chu, C.; Zhang, Z.; Wang, J.; Li, D.; He, N.; Fietkiewicz, C.; Zhou, C.; Kaiser, M. Push-pull effects of basal ganglia network in Parkinson’s disease inferred by functional MRI. npj Parkinson’s Disease 2024, 10, 224. [Google Scholar] [CrossRef] [PubMed]
- Del Tredici, K.; Braak, H. Dysfunction of the locus coeruleus–norepinephrine system and related circuitry in Parkinson’s disease-related dementia. Journal of Neurology, Neurosurgery & Psychiatry 2013, 84, 774–783. [Google Scholar]
- Cykowski, M.D.; Masdeu, J.C. The cholinergic basal forebrain and its role in neurodegeneration. Journal of Neuropathology & Experimental Neurology 2025, 84, 1073–1093. [Google Scholar] [CrossRef] [PubMed]
- Rau, A.; Philipsen, L.; Frings, L.; Müller-Glaw, P.; Reisert, M.; Mast, H.; Sajonz, B.E.; Jost, W.H.; Urbach, H.; Weiller, C. Hippocampus and basal forebrain degeneration differentially impact cognition in Lewy body spectrum disorders. Brain 2025, awaf070. [Google Scholar] [CrossRef] [PubMed]
- Pagonabarraga, J.; Tinazzi, M.; Caccia, C.; Jost, W.H. The role of glutamatergic neurotransmission in the motor and non-motor symptoms in Parkinson’s disease: clinical cases and a review of the literature. Journal of Clinical Neuroscience 2021, 90, 178–183. [Google Scholar] [CrossRef]
- Nandhu, M.; Paul, J.; Kuruvila, K.P.; Abraham, P.M.; Antony, S.; Paulose, C. Glutamate and NMDA receptors activation leads to cerebellar dysfunction and impaired motor coordination in unilateral 6-hydroxydopamine lesioned Parkinson’s rat: functional recovery with bone marrow cells, serotonin and GABA. Molecular and cellular biochemistry 2011, 353, 47–57. [Google Scholar] [CrossRef]
- Oertel, W.H.; Henrich, M.T.; Janzen, A.; Geibl, F.F. The locus coeruleus: Another vulnerability target in Parkinson’s disease. Movement Disorders 2019, 34, 1423–1429. [Google Scholar] [CrossRef]
- Wong, Y.C.; Luk, K.; Purtell, K.; Burke Nanni, S.; Stoessl, A.J.; Trudeau, L.E.; Yue, Z.; Krainc, D.; Oertel, W.; Obeso, J.A.; et al. Neuronal vulnerability in Parkinson disease: Should the focus be on axons and synaptic terminals? Mov Disord 2019, 34, 1406–1422. [Google Scholar] [CrossRef]
- Matsuda, W.; Furuta, T.; Nakamura, K.C.; Hioki, H.; Fujiyama, F.; Arai, R.; Kaneko, T. Single nigrostriatal dopaminergic neurons form widely spread and highly dense axonal arborizations in the neostriatum. J Neurosci 2009, 29, 444–453. [Google Scholar] [CrossRef]
- Surmeier, D.J.; Obeso, J.A.; Halliday, G.M. Selective neuronal vulnerability in Parkinson disease. Nature Reviews Neuroscience 2017, 18, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Bulgari, D.; Deitcher, D.L.; Levitan, E.S. Limited distal organelles and synaptic function in extensive monoaminergic innervation. J Cell Sci 2017, 130, 2520–2529. [Google Scholar] [CrossRef]
- Calabresi, P.; Mechelli, A.; Natale, G.; Volpicelli-Daley, L.; Di Lazzaro, G.; Ghiglieri, V. Alpha-synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction. Cell Death & Disease 2023, 14, 176. [Google Scholar] [CrossRef]
- Ren, P.; Cui, X.; Liang, X. Connectome-based biophysical models of pathological protein spreading in neurodegenerative diseases. PLOS Computational Biology 2025, 21, e1012743. [Google Scholar] [CrossRef]
- Raj, A.; Kuceyeski, A.; Weiner, M. A network diffusion model of disease progression in dementia. Neuron 2012, 73, 1204–1215. [Google Scholar] [CrossRef]
- Iturria-Medina, Y.; Sotero, R.C.; Toussaint, P.J.; Evans, A.C.; Initiative, A.s.D.N. Epidemic spreading model to characterize misfolded proteins propagation in aging and associated neurodegenerative disorders. PLoS computational biology 2014, 10, e1003956. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.-Q.; Zhang, Y.; Yau, Y.; Zeighami, Y.; Larcher, K.; Misic, B.; Dagher, A. Local vulnerability and global connectivity jointly shape neurodegenerative disease propagation. PLoS biology 2019, 17, e3000495. [Google Scholar] [CrossRef]
- Tremblay, C.; Rahayel, S.; Vo, A.; Morys, F.; Shafiei, G.; Abbasi, N.; Markello, R.D.; Gan-Or, Z.; Misic, B.; Dagher, A. Brain atrophy progression in Parkinson’s disease is shaped by connectivity and local vulnerability. Brain Communications 2021, 3, fcab269. [Google Scholar] [CrossRef]
- Mastenbroek, S.E.; Vogel, J.W.; Collij, L.E.; Serrano, G.E.; Tremblay, C.; Young, A.L.; Arce, R.A.; Shill, H.A.; Driver-Dunckley, E.D.; Mehta, S.H. Disease progression modelling reveals heterogeneity in trajectories of Lewy-type α-synuclein pathology. Nature communications 2024, 15, 5133. [Google Scholar] [CrossRef]
- Braak, H.; Rüb, U.; Gai, W.; Del Tredici, K. Idiopathic Parkinson’s disease: possible routes by which vulnerable neuronal types may be subject to neuroinvasion by an unknown pathogen. Journal of neural transmission 2003, 110, 517–536. [Google Scholar] [CrossRef] [PubMed]
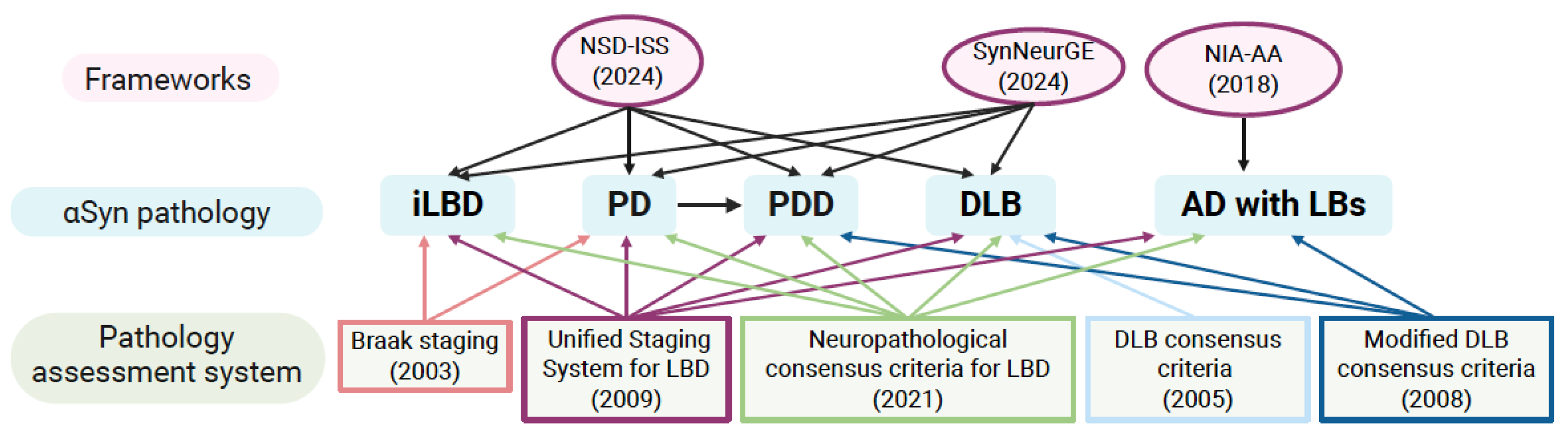
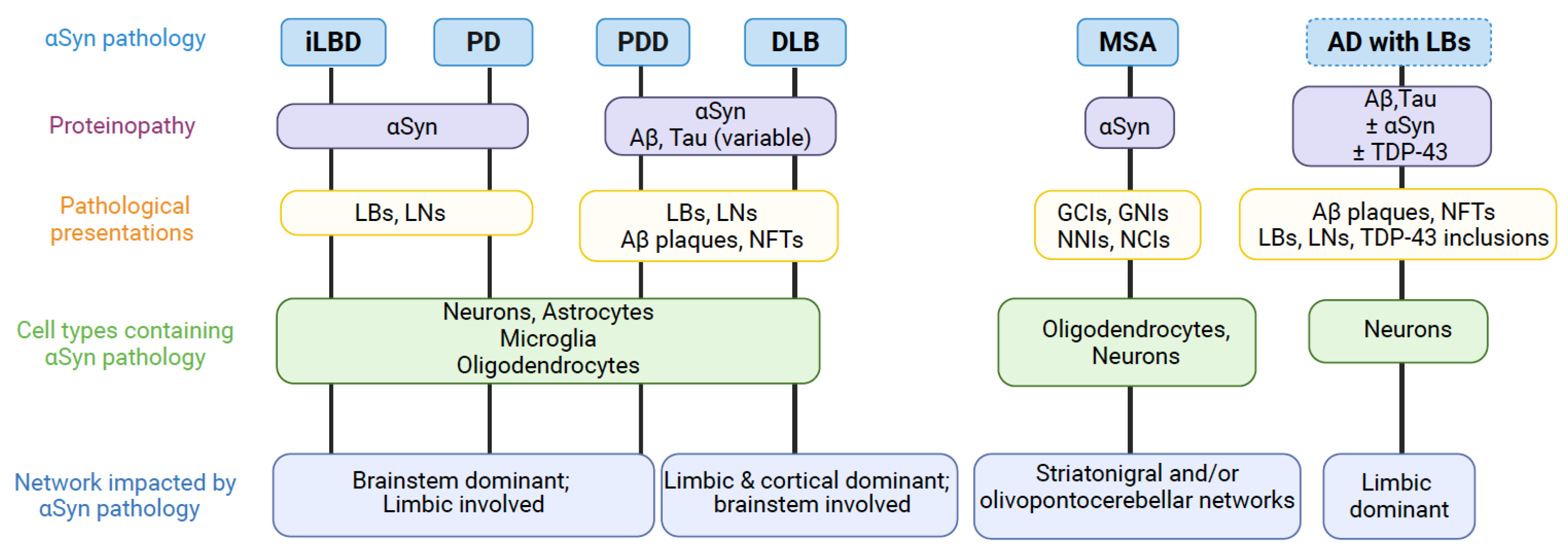
- Lippa, C.; Duda, J.; Grossman, M.; Hurtig, H.; Aarsland, D.; Boeve, B.; Brooks, D.; Dickson, D.; Dubois, B.; Emre, M. DLB and PDD boundary issues: diagnosis, treatment, molecular pathology, and biomarkers. Neurology 2007, 68, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, K.A.; Korczyn, A.D. Are dementia with Lewy bodies and Parkinson’s disease dementia the same disease? BMC Med 2018, 16, 34. [Google Scholar] [CrossRef]
- Frigerio, R.; Fujishiro, H.; Ahn, T.-B.; Josephs, K.A.; Maraganore, D.M.; DelleDonne, A.; Parisi, J.E.; Klos, K.J.; Boeve, B.F.; Dickson, D.W. Incidental Lewy body disease: do some cases represent a preclinical stage of dementia with Lewy bodies? Neurobiology of aging 2011, 32, 857–863. [Google Scholar] [CrossRef]
- Beach, T.G.; White, C.L., III; Hladik, C.L.; Sabbagh, M.N.; Connor, D.J.; Shill, H.A.; Sue, L.I.; Sasse, J.; Bachalakuri, J.; Henry-Watson, J. Olfactory bulb α-synucleinopathy has high specificity and sensitivity for Lewy body disorders. Acta neuropathologica 2009, 117, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Dickson, D.W.; Fujishiro, H.; DelleDonne, A.; Menke, J.; Ahmed, Z.; Klos, K.J.; Josephs, K.A.; Frigerio, R.; Burnett, M.; Parisi, J.E. Evidence that incidental Lewy body disease is pre-symptomatic Parkinson’s disease. Acta neuropathologica 2008, 115, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Pifl, C.; Reither, H.; Attems, J.; Zecca, L. Dopamine and vesicular monoamine transport loss supports incidental Lewy body disease as preclinical idiopathic Parkinson. npj Parkinson’s Disease 2023, 9, 89. [Google Scholar] [CrossRef] [PubMed]
- Beach, T.G.; Adler, C.H.; Sue, L.I.; Peirce, J.B.; Bachalakuri, J.; Dalsing-Hernandez, J.E.; Lue, L.F.; Caviness, J.N.; Connor, D.J.; Sabbagh, M.N. Reduced striatal tyrosine hydroxylase in incidental Lewy body disease. Acta neuropathologica 2008, 115, 445–451. [Google Scholar] [CrossRef]
- Caviness, J.N.; Adler, C.H.; Hentz, J.G.; Shill, H.A.; Evidente, V.G.; Driver-Dunckley, E.D.; Sabbagh, M.N.; Sue, L.; Beach, T.G. Incidental Lewy body disease: electrophysiological findings suggesting pre-clinical Lewy body disorders. Clinical neurophysiology 2011, 122, 2426–2432. [Google Scholar] [CrossRef]
- Keo, A.; Mahfouz, A.; Ingrassia, A.M.; Meneboo, J.-P.; Villenet, C.; Mutez, E.; Comptdaer, T.; Lelieveldt, B.P.; Figeac, M.; Chartier-Harlin, M.-C. Transcriptomic signatures of brain regional vulnerability to Parkinson’s disease. Communications biology 2020, 3, 101. [Google Scholar] [CrossRef]
- Irwin, D.J.; Grossman, M.; Weintraub, D.; Hurtig, H.I.; Duda, J.E.; Xie, S.X.; Lee, E.B.; Van Deerlin, V.M.; Lopez, O.L.; Kofler, J.K. Neuropathological and genetic correlates of survival and dementia onset in synucleinopathies: a retrospective analysis. The Lancet Neurology 2017, 16, 55–65. [Google Scholar] [CrossRef]
- Sim, J.; Li, H.; Hameed, S.; Ting, S.K.S. Clinical Manifestations of Early-Onset Dementia With Lewy Bodies Compared With Late-Onset Dementia With Lewy Bodies and Early-Onset Alzheimer Disease. JAMA Neurology 2022, 79, 702–709. [Google Scholar] [CrossRef]
- Merdes, A.; Hansen, L.; Jeste, D.; Galasko, D.; Hofstetter, C.; Ho, G.; Thal, L.; Corey-Bloom, J. Influence of Alzheimer pathology on clinical diagnostic accuracy in dementia with Lewy bodies. Neurology 2003, 60, 1586–1590. [Google Scholar] [CrossRef]
- Rongve, A.; Vossius, C.; Nore, S.; Testad, I.; Aarsland, D. Time until nursing home admission in people with mild dementia: comparison of dementia with Lewy bodies and Alzheimer’s dementia. International journal of geriatric psychiatry 2014, 29, 392–398. [Google Scholar] [CrossRef]
- Neubauer, A.; Weissenbrunner, D.; Pekrun, S.; Roeber, S.; Ruf, V.; Feyen, P.; Strübing, F.L.; Herms, J. Alpha-synuclein deposition patterns in Alzheimer’s disease: association with cortical amyloid beta and variable tau load. Acta Neuropathologica 2025, 150, 1–20. [Google Scholar] [CrossRef]
- Ishizawa, T.; Mattila, P.; Davies, P.; Wang, D.; Dickson, D.W. Colocalization of tau and alpha-synuclein epitopes in Lewy bodies. Journal of Neuropathology & Experimental Neurology 2003, 62, 389–397. [Google Scholar] [CrossRef]
- Fujishiro, H.; Tsuboi, Y.; Lin, W.-L.; Uchikado, H.; Dickson, D.W. Co-localization of tau and α-synuclein in the olfactory bulb in Alzheimer’s disease with amygdala Lewy bodies. Acta neuropathologica 2008, 116, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Sorrentino, Z.A.; Goodwin, M.S.; Riffe, C.J.; Dhillon, J.S.; Xia, Y.; Gorion, K.M.; Vijayaraghavan, N.; McFarland, K.N.; Golbe, L.I.; Yachnis, A.T.; et al. Unique α-synuclein pathology within the amygdala in Lewy body dementia: implications for disease initiation and progression. Acta Neuropathol Commun 2019, 7, 142. [Google Scholar] [CrossRef]
- Hamilton, R.L. Lewy bodies in Alzheimer’s disease: a neuropathological review of 145 cases using α-synuclein immunohistochemistry. Brain pathology 2000, 10, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Silva-Rodríguez, J.; Labrador-Espinosa, M.A.; Zhang, L.; Castro-Labrador, S.; López-González, F.J.; Moscoso, A.; Sánchez-Juan, P.; Schöll, M.; Grothe, M.J. The effect of Lewy body (co-) pathology on the clinical and imaging phenotype of amnestic patients. Brain 2025, awaf037. [Google Scholar] [CrossRef]
- Clinton, L.K.; Blurton-Jones, M.; Myczek, K.; Trojanowski, J.Q.; LaFerla, F.M. Synergistic interactions between Aβ, tau, and α-synuclein: acceleration of neuropathology and cognitive decline. Journal of Neuroscience 2010, 30, 7281–7289. [Google Scholar] [CrossRef]
- Jeyabalan, J.B.; Pandi A, V.; Veintramuthu, S.; Sivasamy, R.; Dhanasekaran, M.; Justin, A. Converging pathologies in neurodegeneration: the mechanistic interplay between α-Synuclein and Tau in Alzheimer’s and Parkinson’s. Neurological Sciences 2025, 46, 4779–4789. [Google Scholar] [CrossRef]
- Shim, K.H.; Kang, M.J.; Youn, Y.C.; An, S.S.A.; Kim, S. Alpha-synuclein: a pathological factor with Aβ and tau and biomarker in Alzheimer’s disease. Alzheimer’s research & therapy 2022, 14, 201. [Google Scholar]
- Malek-Ahmadi, M.; Beach, T.G.; Zamrini, E.; Adler, C.H.; Sabbagh, M.N.; Shill, H.A.; Jacobson, S.A.; Belden, C.M.; Caselli, R.J.; Woodruff, B.K. Faster cognitive decline in dementia due to Alzheimer disease with clinically undiagnosed Lewy body disease. PloS one 2019, 14, e0217566. [Google Scholar] [CrossRef]
- Olichney, J.M.; Galasko, D.; Salmon, D.; Hofstetter, C.; Hansen, L.; Katzman, R.; Thal, L. Cognitive decline is faster in Lewy body variant than in Alzheimer’s disease. Neurology 1998, 51, 351–357. [Google Scholar] [CrossRef]
- Ferreira, D.; Przybelski, S.A.; Lesnick, T.G.; Lemstra, A.W.; Londos, E.; Blanc, F.; Nedelska, Z.; Schwarz, C.G.; Graff-Radford, J.; Senjem, M.L. β-Amyloid and tau biomarkers and clinical phenotype in dementia with Lewy bodies. Neurology 2020, 95, e3257–e3268. [Google Scholar] [CrossRef]
- May, V.E.; Ettle, B.; Poehler, A.-M.; Nuber, S.; Ubhi, K.; Rockenstein, E.; Winner, B.; Wegner, M.; Masliah, E.; Winkler, J. α-Synuclein impairs oligodendrocyte progenitor maturation in multiple system atrophy. Neurobiology of aging 2014, 35, 2357–2368. [Google Scholar] [CrossRef] [PubMed]
- Andersen, A.M.; Kaalund, S.S.; Marner, L.; Salvesen, L.; Pakkenberg, B.; Olesen, M.V. Quantitative cellular changes in multiple system atrophy brains. Neuropathology and Applied Neurobiology 2023, 49, e12941. [Google Scholar] [CrossRef]
- Nykjær, C.H.; Brudek, T.; Salvesen, L.; Pakkenberg, B. Changes in the cell population in brain white matter in multiple system atrophy. Movement Disorders 2017, 32, 1074–1082. [Google Scholar] [CrossRef]
- Raghavan, S.; Lesnick, T.G.; Castillo, A.M.; Reid, R.I.; Fought, A.J.; Thostenson, K.B.; Johnson Sparrman, K.L.; Gehrking, T.L.; Gehrking, J.A.; Sletten, D.M.; et al. White Matter Abnormalities Track Disease Progression in Multiple System Atrophy. Mov Disord Clin Pract 2024, 11, 1085–1094. [Google Scholar] [CrossRef] [PubMed]
- Reyes, J.F.; Rey, N.L.; Bousset, L.; Melki, R.; Brundin, P.; Angot, E. Alpha-synuclein transfers from neurons to oligodendrocytes. Glia 2014, 62, 387–398. [Google Scholar] [CrossRef]
- Al-Chalabi, A.; Dürr, A.; Wood, N.W.; Parkinson, M.H.; Camuzat, A.; Hulot, J.-S.; Morrison, K.E.; Renton, A.; Sussmuth, S.D.; Landwehrmeyer, B.G. Genetic variants of the α-synuclein gene SNCA are associated with multiple system atrophy. PloS one 2009, 4, e7114. [Google Scholar] [CrossRef] [PubMed]
- Scholz, S.W.; Houlden, H.; Schulte, C.; Sharma, M.; Li, A.; Berg, D.; Melchers, A.; Paudel, R.; Gibbs, J.R.; Simon-Sanchez, J. SNCA variants are associated with increased risk for multiple system atrophy. Annals of Neurology: Official Journal of the American Neurological Association and the Child Neurology Society 2009, 65, 610–614. [Google Scholar] [CrossRef]
- Stefanova, N.; Wenning, G. Multiple system atrophy: emerging targets for interventional therapies. Neuropathology and applied neurobiology 2016, 42, 20–32. [Google Scholar] [CrossRef]
- Asi, Y.T.; Simpson, J.E.; Heath, P.R.; Wharton, S.B.; Lees, A.J.; Revesz, T.; Houlden, H.; Holton, J.L. Alpha-synuclein mRNA expression in oligodendrocytes in MSA. Glia 2014, 62, 964–970. [Google Scholar] [CrossRef] [PubMed]
- Mavroeidi, P.; Arvanitaki, F.; Karakitsou, A.-K.; Vetsi, M.; Kloukina, I.; Zweckstetter, M.; Giller, K.; Becker, S.; Sorrentino, Z.A.; Giasson, B.I. Endogenous oligodendroglial alpha-synuclein and TPPP/p25α orchestrate alpha-synuclein pathology in experimental multiple system atrophy models. Acta neuropathologica 2019, 138, 415–441. [Google Scholar] [CrossRef]
- Ozawa, T.; Paviour, D.; Quinn, N.P.; Josephs, K.A.; Sangha, H.; Kilford, L.; Healy, D.G.; Wood, N.W.; Lees, A.J.; Holton, J.L. The spectrum of pathological involvement of the striatonigral and olivopontocerebellar systems in multiple system atrophy: clinicopathological correlations. Brain 2004, 127, 2657–2671. [Google Scholar] [CrossRef]
- Staerz, S.D.; Anamoah, C.; Tepe, J.J. 20S proteasome enhancers prevent cytotoxic tubulin polymerization-promoting protein induced α-synuclein aggregation. Iscience 2024, 27. [Google Scholar] [CrossRef]
- Nave, K.-A.; Asadollahi, E.; Sasmita, A. Expanding the function of oligodendrocytes to brain energy metabolism. Current Opinion in Neurobiology 2023, 83, 102782. [Google Scholar] [CrossRef]
- Isik, F.I.; Fu, Y.; Pickford, R.; Cheng, Q.; Yang, Y.; Lewis, S.J.; Dzamko, N.; Halliday, G.M.; Kim, W.S. Dysregulation of Monounsaturated Fatty Acids is Related to α-Synuclein in Multiple System Atrophy. Movement Disorders 2025. [Google Scholar] [CrossRef] [PubMed]
- Don, A.S.; Hsiao, J.-H.T.; Bleasel, J.M.; Couttas, T.A.; Halliday, G.M.; Kim, W.S. Altered lipid levels provide evidence for myelin dysfunction in multiple system atrophy. Acta neuropathologica communications 2014, 2, 150. [Google Scholar] [CrossRef]
- Hoch-Kraft, P.; Trotter, J.; Gonsior, C. Missing in action: dysfunctional RNA metabolism in oligodendroglial cells as a contributor to neurodegenerative diseases? Neurochemical research 2020, 45, 566–579. [Google Scholar] [CrossRef] [PubMed]
- Shahnawaz, M.; Mukherjee, A.; Pritzkow, S.; Mendez, N.; Rabadia, P.; Liu, X.; Hu, B.; Schmeichel, A.; Singer, W.; Wu, G. Discriminating α-synuclein strains in Parkinson’s disease and multiple system atrophy. Nature 2020, 578, 273–277. [Google Scholar] [CrossRef]
- Schweighauser, M.; Shi, Y.; Tarutani, A.; Kametani, F.; Murzin, A.G.; Ghetti, B.; Matsubara, T.; Tomita, T.; Ando, T.; Hasegawa, K. Structures of α-synuclein filaments from multiple system atrophy. Nature 2020, 585, 464–469. [Google Scholar] [CrossRef]
- Wiseman, J.A.; Fu, Y.; Faull, R.L.; Turner, C.P.; Curtis, M.A.; Halliday, G.M.; Dieriks, B.V. N-terminus α-synuclein detection reveals new and more diverse aggregate morphologies in multiple system atrophy and Parkinson’s disease. Translational Neurodegeneration 2024, 13, 1–16. [Google Scholar] [CrossRef]
- Magalhães, P.; Lashuel, H.A. Opportunities and challenges of alpha-synuclein as a potential biomarker for Parkinson’s disease and other synucleinopathies. npj Parkinson’s Disease 2022, 8, 93. [Google Scholar] [CrossRef]
- Donadio, V.; Ingelsson, M.; Rizzo, G.; Furia, A.; Incensi, A.; Delprete, C.; Pinho, M.; Liguori, R.; Pritzkow, S. Diagnostic biomarkers for α-synucleinopathies-state of the art and future developments: a systematic review. Molecular Neurodegeneration 2025. [Google Scholar] [CrossRef]
- Mollenhauer, B.; Locascio, J.J.; Schulz-Schaeffer, W.; Sixel-Döring, F.; Trenkwalder, C.; Schlossmacher, M.G. α-Synuclein and tau concentrations in cerebrospinal fluid of patients presenting with parkinsonism: a cohort study. The Lancet Neurology 2011, 10, 230–240. [Google Scholar] [CrossRef]
- Winchester, L.; Lawton, M.; Barber, I.; Ash, J.; Liu, B.; Evetts, S.; Hopkins-Jones, L.; Lewis, S.; Bresner, C.; Vingill, S. Identification of a novel proteomic Biomarker in Parkinson’s Disease: Discovery and Replication in Blood, brain and CSF. medRxiv 2021, 2021.2012. 2026.21268282.
- Van Dijk, K.; Bidinosti, M.; Weiss, A.; Raijmakers, P.; Berendse, H.; Van De Berg, W. Reduced α-synuclein levels in cerebrospinal fluid in P arkinson’s disease are unrelated to clinical and imaging measures of disease severity. European Journal of Neurology 2014, 21, 388–394. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Tang, H.; Nie, K.; Wang, L.; Zhao, J.; Gan, R.; Huang, J.; Zhu, R.; Feng, S.; Duan, Z. Cerebrospinal fluid alpha-synuclein as a biomarker for Parkinson’s disease diagnosis: a systematic review and meta-analysis. International journal of neuroscience 2015, 125, 645–654. [Google Scholar] [CrossRef]
- Kang, J.-H.; Irwin, D.J.; Chen-Plotkin, A.S.; Siderowf, A.; Caspell, C.; Coffey, C.S.; Waligórska, T.; Taylor, P.; Pan, S.; Frasier, M. Association of cerebrospinal fluid β-amyloid 1-42, T-tau, P-tau181, and α-synuclein levels with clinical features of drug-naive patients with early Parkinson disease. JAMA neurology 2013, 70. [Google Scholar] [CrossRef] [PubMed]
- Tokuda, T.; Qureshi, M.; Ardah, M.; Varghese, S.; Shehab, S.; Kasai, T.; Ishigami, N.; Tamaoka, A.; Nakagawa, M.; El-Agnaf, O. Detection of elevated levels of α-synuclein oligomers in CSF from patients with Parkinson disease. Neurology 2010, 75, 1766–1770. [Google Scholar] [CrossRef]
- Hansson, O.; Hall, S.; Öhrfelt, A.; Zetterberg, H.; Blennow, K.; Minthon, L.; Nägga, K.; Londos, E.; Varghese, S.; Majbour, N.K. Levels of cerebrospinal fluid α-synuclein oligomers are increased in Parkinson’s disease with dementia and dementia with Lewy bodies compared to Alzheimer’s disease. Alzheimer’s research & therapy 2014, 6, 25. [Google Scholar]
- Wang, Y.; Shi, M.; Chung, K.A.; Zabetian, C.P.; Leverenz, J.B.; Berg, D.; Srulijes, K.; Trojanowski, J.Q.; Lee, V.M.-Y.; Siderowf, A.D. Phosphorylated α-synuclein in Parkinson’s disease. Science translational medicine 2012, 4, 121ra120–121ra120. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, C.C.; Alves, G.; Tysnes, O.B.; Maple-Grødem, J.; Lange, J. Cerebrospinal Fluid Phosphorylated Alpha-Synuclein in Newly Diagnosed Parkinson’s Disease. European Journal of Neurology 2025, 32, e70167. [Google Scholar] [CrossRef]
- Bellomo, G.; Stoops, E.; Vanbrabant, J.; Demeyer, L.; Francois, C.; Vanhooren, M.; Ma, Y.; Farris, C.M.; Concha-Marambio, L.; Paolini Paoletti, F. Phosphorylated α-synuclein in CSF and plasma does not reflect synucleinopathy. npj Parkinson’s Disease 2025, 11, 232. [Google Scholar] [CrossRef]
- Parnetti, L.; Gaetani, L.; Eusebi, P.; Paciotti, S.; Hansson, O.; El-Agnaf, O.; Mollenhauer, B.; Blennow, K.; Calabresi, P. CSF and blood biomarkers for Parkinson’s disease. The Lancet Neurology 2019, 18, 573–586. [Google Scholar] [CrossRef]
- Gomes, B.F.; Farris, C.M.; Ma, Y.; Concha-Marambio, L.; Lebovitz, R.; Nellgård, B.; Dalla, K.; Constantinescu, J.; Constantinescu, R.; Gobom, J. α-Synuclein seed amplification assay as a diagnostic tool for parkinsonian disorders. Parkinsonism & Related Disorders 2023, 117, 105807. [Google Scholar] [CrossRef]
- Duong, M.T.; Das, S.R.; Khandelwal, P.; Vizcarra, J.A.; Li, Y.; Xie, L.; Yushkevich, P.A.; Shaw, L.M.; Dubroff, J.G.; Siderowf, A.; et al. Discordance of Dopaminergic Dysfunction and Subcortical Atrophy by α-Synuclein Status in Sporadic and Genetic Parkinson’s Disease. Mov Disord 2026. [Google Scholar] [CrossRef]
- Rossi, M.; Candelise, N.; Baiardi, S.; Capellari, S.; Giannini, G.; Orrù, C.D.; Antelmi, E.; Mammana, A.; Hughson, A.G.; Calandra-Buonaura, G. Ultrasensitive RT-QuIC assay with high sensitivity and specificity for Lewy body-associated synucleinopathies. Acta neuropathologica 2020, 140, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Banerjee, V.; Barria, C.; Ramirez, S.; Allison, T.; Gorski, D.; Evans, H.; Nguyen, Q.; Harrison, D.; Al-Lahham, R.; et al. Seed amplification of MSA alpha-synuclein aggregates preserves the biological and structural properties of brain-derived aggregates. Nat Commun 2025, 16, 11266. [Google Scholar] [CrossRef]
- Jiménez-Jiménez, F.J.; Alonso-Navarro, H.; García-Martín, E.; Santos-García, D.; Martínez-Valbuena, I.; Agúndez, J.A. Alpha-synuclein in peripheral tissues as a possible marker for neurological diseases and other medical conditions. Biomolecules 2023, 13, 1263. [Google Scholar] [CrossRef]
- Agin-Liebes, J.; Lodge, A.; Reddy, H.; Vacchi, E.; Usseglio, J.; Honig, L.S.; Melli, G.; Noble, J.M.; Przedborski, S. α-synuclein biomarker assays: bridging research and patient care. The Lancet Neurology 2025, 24, 681–697. [Google Scholar] [CrossRef] [PubMed]
- Klingelhoefer, L.; Reichmann, H. Parkinson’s disease as a multisystem disorder. Journal of Neural Transmission 2017, 124, 709–713. [Google Scholar] [CrossRef]
- Peng, H.; Chen, S.; Wu, S.; Shi, X.; Ma, J.; Yang, H.; Li, X. Alpha-synuclein in skin as a high-quality biomarker for Parkinson’s disease. Journal of the neurological sciences 2023, 451, 120730. [Google Scholar] [CrossRef] [PubMed]
- Waqar, S.; Khan, H.; Zulfiqar, S.K.; Ahmad, A. Skin biopsy as a diagnostic tool for synucleinopathies. Cureus 2023, 15. [Google Scholar] [CrossRef]
- Donadio, V.; Incensi, A.; Leta, V.; Giannoccaro, M.P.; Scaglione, C.; Martinelli, P.; Capellari, S.; Avoni, P.; Baruzzi, A.; Liguori, R. Skin nerve α-synuclein deposits: a biomarker for idiopathic Parkinson disease. Neurology 2014, 82, 1362–1369. [Google Scholar] [CrossRef]
- Kuzkina, A.; Schulmeyer, L.; Monoranu, C.-M.; Volkmann, J.; Sommer, C.; Doppler, K. The aggregation state of α-synuclein deposits in dermal nerve fibers of patients with Parkinson’s disease resembles that in the brain. Parkinsonism & related disorders 2019, 64, 66–72. [Google Scholar]
- Han, Y.; Wu, D.; Wang, Y.; Xie, J.; Zhang, Z. Skin alpha-synuclein deposit patterns: A predictor of Parkinson’s disease subtypes. EBioMedicine 2022, 80. [Google Scholar] [CrossRef]
- Vacchi, E.; Senese, C.; Chiaro, G.; Disanto, G.; Pinton, S.; Morandi, S.; Bertaina, I.; Bianco, G.; Staedler, C.; Galati, S. Alpha-synuclein oligomers and small nerve fiber pathology in skin are potential biomarkers of Parkinson’s disease. NPJ Parkinson’s disease 2021, 7, 119. [Google Scholar] [CrossRef] [PubMed]
- Mazzetti, S.; Basellini, M.J.; Ferri, V.; Cassani, E.; Cereda, E.; Paolini, M.; Calogero, A.M.; Bolliri, C.; De Leonardis, M.; Sacilotto, G. α-Synuclein oligomers in skin biopsy of idiopathic and monozygotic twin patients with Parkinson’s disease. Brain 2020, 143, 920–931. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Becker, K.; Donadio, V.; Siedlak, S.; Yuan, J.; Rezaee, M.; Incensi, A.; Kuzkina, A.; Orrú, C.D.; Tatsuoka, C. Skin α-synuclein aggregation seeding activity as a novel biomarker for Parkinson disease. JAMA neurology 2021, 78, 30–40. [Google Scholar] [CrossRef]
- Iranzo, A.; Mammana, A.; Muñoz-Lopetegi, A.; Dellavalle, S.; Mayà, G.; Rossi, M.; Serradell, M.; Baiardi, S.; Arqueros, A.; Quadalti, C. Misfolded α-synuclein assessment in the skin and CSF by RT-QuIC in isolated REM sleep behavior disorder. Neurology 2023, 100, e1944–e1954. [Google Scholar] [CrossRef]
- McWilliam, O.H.; Bsoul, R.; Lund, E.L.; Waldemar, G.; Hasselbalch, S.G.; Simonsen, A.H.; Bruun, M.; von Buchwald, C.; Aanæs, K.; Pedersen, C.K. α-Synuclein seed amplification assay in Lewy body dementia versus Alzheimer’s disease. Alzheimer’s & Dementia: Diagnosis, Assessment & Disease Monitoring 2025, 17, e70203. [Google Scholar]
- Donadio, V.; Incensi, A.; Rizzo, G.; Westermark, G.T.; Devigili, G.; De Micco, R.; Tessitore, A.; Nyholm, D.; Parisini, S.; Nyman, D. Phosphorylated α-synuclein in skin Schwann cells: a new biomarker for multiple system atrophy. Brain 2023, 146, 1065–1074. [Google Scholar] [CrossRef]
- Ma, Y.; Farris, C.M.; Weber, S.; Schade, S.; Nguyen, H.; Pérez-Soriano, A.; Giraldo, D.M.; Fernández, M.; Soto, M.; Cámara, A. Sensitivity and specificity of a seed amplification assay for diagnosis of multiple system atrophy: a multicentre cohort study. The Lancet Neurology 2024, 23, 1225–1237. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.; Dieriks, B.V. Multiple system atrophy: α-Synuclein strains at the neuron-oligodendrocyte crossroad. Molecular Neurodegeneration 2022, 17, 77. [Google Scholar] [CrossRef]
- Baranová, S.; Matěj, R.; Soukup, J.; Dušek, P.; Holada, K. Alpha-synuclein seeding activity in postmortem tissues from patients with diffuse and isolated Lewy bodies. Acta Neuropathologica Communications 2026, 14, 12. [Google Scholar] [CrossRef]
- Ingelsson, M. Alpha-synuclein oligomers—neurotoxic molecules in Parkinson’s disease and other Lewy body disorders. Frontiers in neuroscience 2016, 10, 408. [Google Scholar] [CrossRef]
- Roberts, R.F.; Wade-Martins, R.; Alegre-Abarrategui, J. Direct visualization of alpha-synuclein oligomers reveals previously undetected pathology in Parkinson’s disease brain. Brain 2015, 138, 1642–1657. [Google Scholar] [CrossRef] [PubMed]
- Jensen, N.M.; Fu, Y.; Betzer, C.; Li, H.; Elfarrash, S.; Shaib, A.H.; Krah, D.; Vitic, Z.; Reimer, L.; Gram, H. MJF-14 proximity ligation assay detects early non-inclusion alpha-synuclein pathology with enhanced specificity and sensitivity. npj Parkinson’s Disease 2024, 10, 227. [Google Scholar] [CrossRef] [PubMed]
- Estaun-Panzano, J.; Arotcarena, M.-L.; Bezard, E. Monitoring α-synuclein aggregation. Neurobiology of disease 2023, 176, 105966. [Google Scholar] [CrossRef]
- Samson, E.; Noseworthy, M. A review of diagnostic imaging approaches to assessing Parkinson’s disease. Brain Disord 2022, 6, 100037. [Google Scholar] [CrossRef]
- van Oostveen, W.M.; de Lange, E.C. Imaging techniques in Alzheimer’s disease: a review of applications in early diagnosis and longitudinal monitoring. International journal of molecular sciences 2021, 22, 2110. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



