Submitted:
10 February 2026
Posted:
11 February 2026
You are already at the latest version
Abstract
Photosensitisation is a clinically significant dermatological and systemic disorder affecting grazing livestock worldwide. The condition arises following the ingestion or dermal exposure to photodynamic compounds that, upon activation by ultraviolet (UV) or visible light, induce tissue injury. Plant‑associated photosensitisation remains one of the most important aetiological categories in veterinary toxicology and may occur via primary (direct phototoxic) or secondary (hepatogenous) mechanisms. This review synthesises current knowledge on the occurrence of photosensitising compounds in plants, their biochemical and toxicodynamic properties, and their clinical relevance in livestock species. Emphasis is placed on major primary photosensitising taxa, including Heracleum spp. and Hypericum perforatum, as well as hepatotoxic pyrrolizidine alkaloid‑containing plants such as Senecio spp. Mechanistic pathways, plant metabolite ecology, and toxicopathological outcomes are discussed alongside illustrative case material. The review aims to provide a consolidated veterinary toxicology framework for understanding plant‑associated photosensitisation in grazing systems.
Keywords:
1. Introduction
2. Role of Secondary Plant Metabolites
3. Photosensitization Induced by Secondary Metabolites
4. Mechanism of Action
5. Occurrence of Photosensitizing Compounds in Plants
6. Development of Photosensitization in Animals
7. Plant Species and Compounds Causing Primary Photosensitization
7.1. Hogweeds (Heracleum spp.)
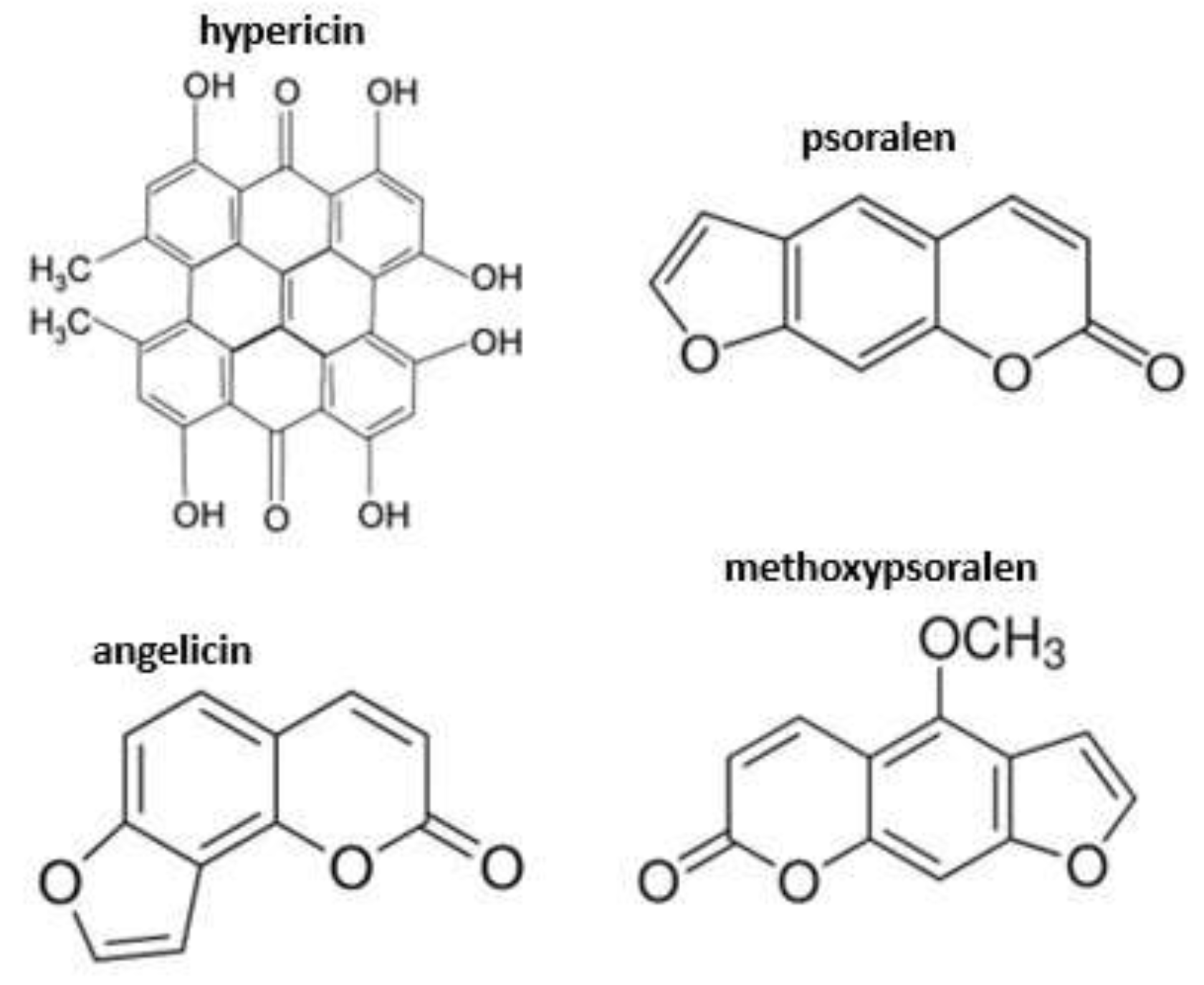
7.2. St John’s Wort (Hypericum perforatum)
8. Plant Species Causing Secondary (Hepatogenous) Photosensitization and Their Compounds
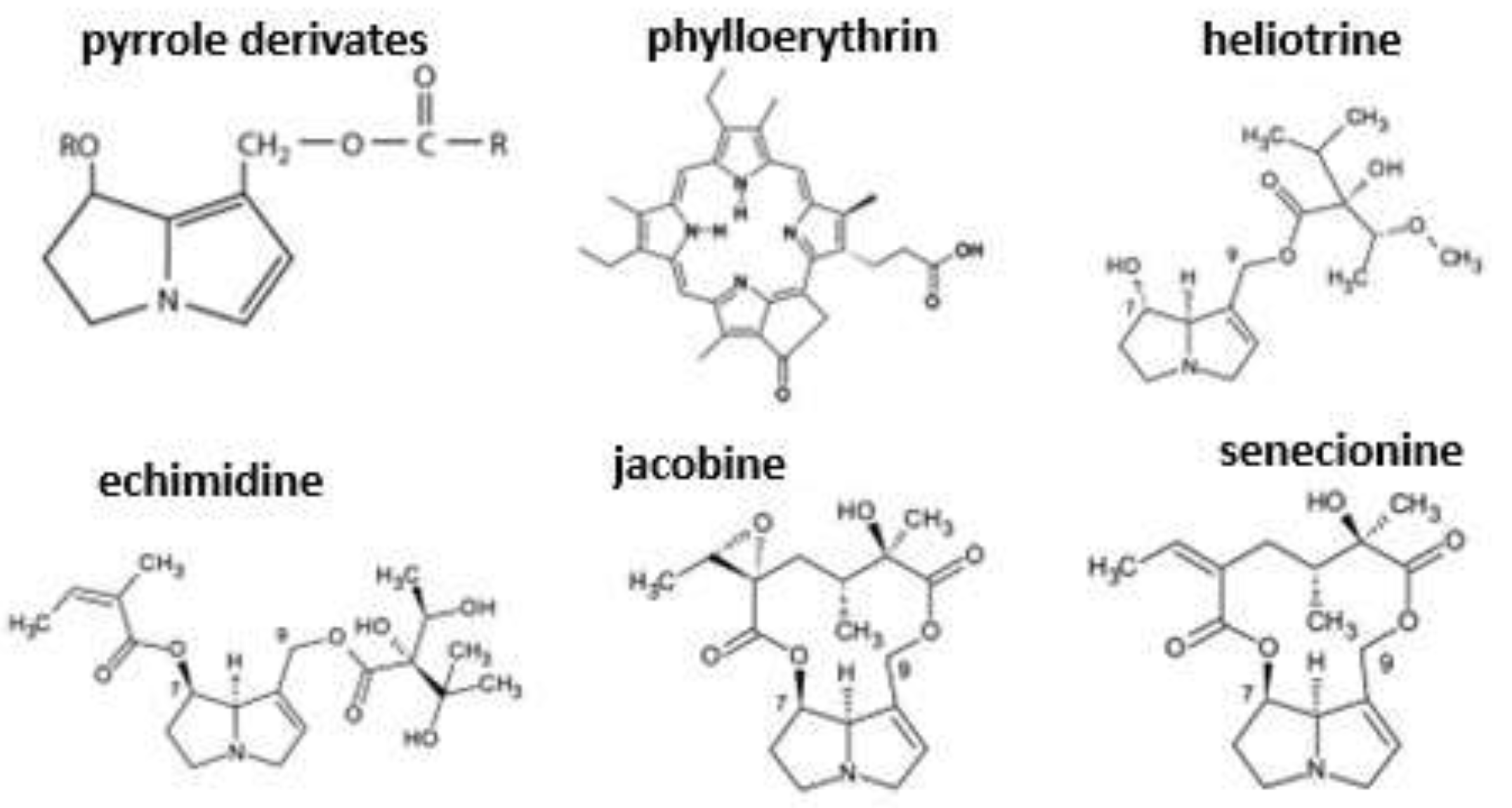
8.1. Ragworts (Senecio spp.)
9. Case Studies
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PA | pyrrolizidine alkaloids |
| ROS | reactive oxygen species |
| UV | ultraviolet |
| SC | stratum corneum |
| SB | stratum basale |
| SPP | secondary plant products/metabolites |
References
- Croteau, R.; Kutchman, T.M.; Lewis, N.G. Natural products (secondary metabolites). In Biochemistry and Molecular Biology of Plants, 2nd ed.; Buchanan, B.B., Gruissem, W., Jones, R.L., Eds.; Wiley Blackwell: New Jersey, USA, 2000; pp. 1250–1319. [Google Scholar]
- Field, B.; Jordan, F.; Osbourn, A. First encounters - Deployment of defence-related natural products by plants. New Phytol 2006, 172, 193–207. [Google Scholar] [CrossRef] [PubMed]
- Pickett, J.A. New synthesis: Chemical ecology and sustainable food production. J. Chem. Ecol. 2012, 38, 1071–1071. [Google Scholar] [CrossRef] [PubMed]
- Weston, P.A.; Weston, L.A.; Hildebrand, P.A. Metabolic profiling in Echium plantagineum: Presence of bioactive pyrrolizidine alkaloids and naphthoquinones from accessions across Australia. Phytochem. Rev. 2013, 12, 831–837. [Google Scholar] [CrossRef]
- Weston, L.A.; Mathesius, U. Flavonoids: Their structure, biosynthesis and role in the rhizosphere, including allelopathy. J. Chem. Ecol. 2013, 39, 283–297. [Google Scholar] [CrossRef]
- Wink, M. Biochemistry of plant secondary metabolism, 2nd ed.; Wiley Blackwell: New Jersey, USA, 2000; pp. 1–16. [Google Scholar]
- Launchbaugh, K.L. Biochemical Aspects of Grazing Behaviour. In The ecology and management of grazing systems, 1st ed.; Hodgson, J., Illius, A.W., Eds.; CAB International: Wallingford, Oxfordshire, UK, 1996; pp. 159–184. [Google Scholar]
- Gupta, R.C. Veterinary Toxicology, 2nd ed.; Elseiver, London, UK, 2012; p. pp. 1154. [Google Scholar]
- Rowe, L.D. Photosensitization problems in livestock. Vet. Clin. N. Am.: Food. Anim. Pract. 1989, 5, 301–323. [Google Scholar] [CrossRef]
- McKenzie, R. Australia’s Poisonous Plants, Fungi and Cyanobacteria, 1st ed.; CSIRO Publishing: Collingwood, Australia, 2012; p. pp. 950. [Google Scholar]
- Spikes, J.D. Photosensitization, 2nd ed.; Plenum Press: New York, NY, USA, 1989; pp. 79–110. [Google Scholar]
- Knight, A.P.; Walter, R.G. A Guide to Plant Poisoning of Animals in North America, 1st ed.; Teton New Media: Jackson, WY, USA, 2001; p. pp. 367. [Google Scholar]
- Smith, E.; Kiss, F.; Porter, R.M.; Anstey, A.V. A review of UVA-mediated photosensitivity disorders. Photochem. Photobiol. Sci. 2012, 11, 199–206. [Google Scholar] [CrossRef]
- Yamori, W.; Sakata, N.; Suzuki, Y.; Shikanai, T.; Makino, A. Cyclic electron flow around photosystem I via chloroplast NAD(P)H dehydrogenase (NDH) complex performs a significant physiological role during photosynthesis and plant growth at low temperature in rice. Plant J. 2011, 68, 966–976. [Google Scholar] [CrossRef]
- Yamori, W. Improving photosynthesis to increase food and fuel production by biotechnological strategies in crops. J. Plant Biochem. Physiol. 2013, 1, pp. 113. [Google Scholar]
- Quinn, J.C.; Kessell, A.; Weston, L.A. Secondary plant products causing photosensitisation in grazing herbivores: their structure, activity and regulation. Intl. J. Mol. Sci. 2014, 15, 1441–1465. [Google Scholar] [CrossRef]
- Wallaart, T.E.; Pras, N.; Quax, W.J. Isolation and identification of dihydroartemisinic acid hydroperoxide from Artemisia annua: A novel biosynthetic precursor of artemisinin. J. Nat. Prod. 1999, 62, 1160–1162. [Google Scholar] [CrossRef]
- Ramel, F.; Birtic, S.; Cuine, S.; Triantaphylides, C.; Ravanat, J.L.; Havaux, M. Chemical quenching of singlet oxygen by carotenoids in plants. Plant Physiol. 2012, 158, 1267–1278. [Google Scholar] [CrossRef] [PubMed]
- Ramel, F.; Birtic, S.; Ginies, C.; Soubigou-Taconnat, L.; Triantaphylides, C.; Havaux, M. Carotenoid oxidation products are stress signals that mediate gene responses to singlet oxygen in plants. Proc. Natl. Acad. Sci. USA 2012, 109, 5535–5540. [Google Scholar] [PubMed]
- Alscher, R.G.; Donahue, J.L.; Cramer, C.L. Reactive oxygen species and antioxidants: Relationships in green cells. Physiol. Plant. 1997, 100, 224–233. [Google Scholar] [CrossRef]
- Apel, K.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef]
- Wallaart, T.E.; Pras, N.; Quax, W.J. Seasonal variations of artemisinin and its biosynthetic precursors in tetraploid Artemisia annua plants compared with the diploid wild-type. Planta Med. 1999, 65, 723–728. [Google Scholar] [CrossRef]
- Theodossiou, T.A.; Hothersall, J.S.; De Witte, P.A.; Pantos, A.; Agostinis, P. The multifaceted photocytotoxic profile of hypericin. Mol. Pharm. 2009, 6, 1775–1789. [Google Scholar] [CrossRef]
- Brechner, M.L.; Albright, L.D.; Weston, L.A. Effects of UV-B on secondary metabolites of St. John’s Wort (Hypericum perforatum L.) grown in controlled environments. Photochem. Photobiol. 2011, 87, 680–684. [Google Scholar] [CrossRef]
- Moritz, R.; Aboling, S. Incidence of photosensitization in husbandry animals: A meta-study on the effects of feed diversity and feed choice. Agriculture 2024, 14, pp 1137. [Google Scholar] [CrossRef]
- Araya, O.S.; Ford, E.J.H. An investigation of the type of photosensitization caused by the ingestion of St John's Wort (Hypericum perforatum) by calves. J. Comp. Pathol. 1981, 91, 135–141. [Google Scholar] [CrossRef]
- Weston, L.A.; Ryan, P.R.; Watt, M. Mechanisms for cellular transport and release of allelochemicals from plant roots into the rhizosphere. J. Exp. Bot. 2012, 63, 3445–3454. [Google Scholar] [CrossRef]
- Puschner, B.; Chen, X.; Read, D.; Affolter, V.K. Alfalfa hay induced primary photosensitization in horses. Vet. J. 2016, 211, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Monteiro-Riviere, N.A.; Bristol, D.G.; Manning, T.O.; Rogers, R.A.; Riviere, J.E. Interspecies and interregional analysis of the comparative histologic thickness and laser doppler blood-flow measurements at 5 cutaneous sites in 9 species. J. Invest. Dermatol. 1990, 95, 582–586. [Google Scholar] [CrossRef] [PubMed]
- Montiero-Riveiere, N. The Integument. In Dellmann’s Textbook of Veterinary Histology, 6th ed.; Eurell, J.A., Frappier, BlL., Eds.; Blackwell Press: Ames, IA, USA, 2006; pp. 320–349. [Google Scholar]
- Monteiro-Riviere, N.A.; Inman, A.O.; Mak, V.; Wertz, P.; Riviere, J.E. Effect of selective lipid extraction from different body regions on epidermal barrier function. Pharm. Res. 2001, 18, 992–998. [Google Scholar] [CrossRef]
- Stegelmeier, B.L. Equine photosensitization. Clin. Techn. Equine Pract. 2002, 1, 81–88. [Google Scholar] [CrossRef]
- Bourke, C.A. The effect of shade, shearing and wool type in the protection of Merino sheep from Hypericum perforatum (St. John’s wort) poisoning. Aust. Vet. J. 2003, 81, 494–498. [Google Scholar] [CrossRef]
- Bourke, C.A.; White, J.G. Reassessment of the toxicity of Hypericum perforatum (St. John’s wort) for cattle. Aust. Vet. J. 2004, 82, 707–710. [Google Scholar] [CrossRef]
- Campbell, W.M.; Dombroski, G.S.; Sharma, I.; Partridge, A.C.; Collett, M.G. Photodynamic chlorophyll a metabolites, including phytoporphyrin (phylloerythrin), in the blood of photosensitive livestock: Overview and measurement. N. Z. Vet. J. 2010, 58, 146–154. [Google Scholar] [CrossRef]
- Scheie, E.; Flaoyen, A.; Moan, J.; Berg, K. Phylloerythrin: Mechanisms for cellular uptake and location, photosensitisation and spectroscopic evaluation. N. Z. Vet. J. 2002, 50, 104–110. [Google Scholar] [CrossRef]
- Richarson, P.M. Plant and Fungal Toxins. In Handbook of Natural Toxins, 1st ed.; Keeler, R.F., Tu, A.T., Eds.; Marcel Dekker: New York, NY, USA, 1983; Volume 1. p. pp. 73. [Google Scholar]
- Stegelmeier, B.L.; Davis, T.Z.; Clayton, M.J. Plant-Induced Photosensitivity and Dermatitis in Livestock. Vet. Clin. North Am. Food Anim. Pract. 2020, 36, 725–733. [Google Scholar] [CrossRef] [PubMed]
- Collett, M.G. Photosensitisation diseases of animals: Classification and a weight of evidence approach to primary causes. Toxicon: X 2019, 3, 100012. [Google Scholar] [CrossRef] [PubMed]
- Kessell, A.; Ladmore, G.; Quinn, J. An outbreak of primary photosensitisation in lambs secondary to consumption of Biserrula pelecinus (biserrula). Aust. J. Vet. Sci. 2015, 93, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Lagey, K.; Duinslaeger, L.; Vanderkelen, A. Burns induced by plants. Burns 1995, 21, 542–543. [Google Scholar] [CrossRef]
- Lehmann, P. Sun exposed skin disease. Clin. Dermatol. 2011, 29, 180–188. [Google Scholar] [CrossRef]
- Mathieu, A.; Jeanmougin, M.; Cavelierballoy, B.; Ribrioux, A.; Civatte, J. Phytodermatitis with Heracleum-Mantegazzianum. Ann. Dermatol. Vener. 1986, 113, 1005–1007. [Google Scholar]
- Tiley, G.E.D.; Dodd, F.S.; Wade, P.M. Heracleum mantegazzianum Sommier & Levier. J. Ecol. 1996, 84, 297–319. [Google Scholar] [CrossRef]
- Camm, E.; Buck, H.W.; Mitchell, J.C. Phytophotodermatitis from Heracleum mantegazzianum. Contact Dermat. 1976, 2, 68–72. [Google Scholar] [CrossRef]
- Musajo, L.; Rodighiero, G. The skin-photosensitizing furocoumarins. Experientia 1962, 18, 153–161. [Google Scholar] [CrossRef]
- Knudsen, E.A. Seasonal variations in the content of phototoxic compounds in giant hogweed. Contact Dermat. 1983, 9, 281–284. [Google Scholar] [CrossRef]
- Berenbaum, M. Patterns of Furanocoumarin distribution and insect herbivory in the Umbelliferae—Plant chemistry and community structure. Ecology 1981, 62, 1254–1266. [Google Scholar] [CrossRef]
- Berenbaum, M.; Feeny, P. Toxicity of angular furanocoumarins to swallowtail butterflies—Escalation in a co-evolutionary arms-race. Science 1981, 212, 927–929. [Google Scholar] [CrossRef] [PubMed]
- Sardari, S.; Mori, Y.; Horita, K.; Micetich, R.G.; Nishibe, S.; Daneshtalab, M. Synthesis and antifungal activity of coumarins and angular furanocoumarins. Bioorg. Med. Chem. 1999, 7, 1933–1940. [Google Scholar] [CrossRef] [PubMed]
- Bissonnette, L.; Arnason, J.T.; Smith, M.L. Real-time fluorescence-based detection of furanocoumarin photoadducts of DNA. Phytochem. Anal. 2008, 19, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, N.; Kohtani, S.; Nakagaki, R. Molecular aspects of furocoumarin reactions: Photophysics, photochemistry, photobiology, and structural analysis. J. Photochem. Photobiol. 2005, 6, 168–185. [Google Scholar] [CrossRef]
- Musajo, L.; Rodighie, G.; Breccia, A.; Dallacqu, F.; Malesani, G. Photoreaction between DNA and skin-photosensitizing furocoumarins studied using labelled bergapten. Experientia 1966, 22, pp. 75. [Google Scholar] [CrossRef] [PubMed]
- Bourke, C.A. Sunlight associated hyperthermia as a consistent and rapidly developing clinical sign in sheep intoxicated by St. John’s wort (Hypericum perforatum). Aust. Vet. J. 2000, 78, 483–488. [Google Scholar] [CrossRef]
- Cunningham, I.J. Photosensitivity disease in New Zealand. N. Z. J. Sci. Technol. 1947, 29, 207–213. [Google Scholar]
- Holzl, J.; Ostrowski, E. Analysis of the essential compounds of Hypericum perforatum. Planta Med. 1986, 52, 531–531. [Google Scholar] [CrossRef]
- Pace, N.; Mackinney, G. Hypericin, the photodynamic pigment from St. John’s wort. J. Am. Chem. Soc. 1941, 63, 2570–2574. [Google Scholar] [CrossRef]
- Bourke, C.A.; Rayward, D. Photosensitisation in dairy cattle grazing alligator weed (Alternanthera philoxeroides) infested pastures. Aust. Vet. J. 2003, 81, 361–362. [Google Scholar] [CrossRef]
- Agostinis, P.; Vantieghem, A.; Merlevede, W.; de Witte, P.A. Hypericin in cancer treatment: More light on the way. Int. J. Biochem. Cell Biol. 2002, 34, 221–241. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.M.; Chee, S.K.; Yuen, G.Y.; Olivo, M. Hypericin induced death receptor-mediated apoptosis in photoactivated tumor cells. Int. J. Mol. Med. 2002, 9, 601–616. [Google Scholar] [CrossRef]
- Chen, B.; Xu, Y.; Roskams, T.; Delaey, E.; Agostinis, P.; Vandenheede, J.R.; de Witte, P. Efficacy of antitumoral photodynamic therapy with hypericin: relationship between biodistribution and photodynamic effects in the RIF-1 mouse tumor model. Int. J. Cancer 2001, 93, 275–282. [Google Scholar] [CrossRef]
- Diwu, Z.; Lown, J.W. Photosensitization with anticancer agents. 17. EPR studies of photodynamic action of hypericin: formation of semiquinone radical and activated oxygen species on illumination. Free Radic. Biol. Med. 1993, 14, 209–215. [Google Scholar] [CrossRef]
- Roslaniec, M.; Weitman, H.; Freeman, D.; Mazur, Y.; Ehrenberg, B. Liposome binding constants and singlet oxygen quantum yields of hypericin, tetrahydroxy helianthrone and their derivatives: Studies in organic solutions and in liposomes. J. Photochem. Photobiol. 2000, 57, 149–158. [Google Scholar] [CrossRef]
- Smith, L.W.; Culvenor, C.C.J. Plant sources of hepatotoxic pyrrolizidine alkaloids. J. Nat. Prod. 1981, 44, 129–152. [Google Scholar] [CrossRef]
- Ali, S.M.; Chee, S.K.; Yuen, G.Y.; Olivo, M. Hypocrellins and hypericin induced apoptosis in human tumor cells: A possible role of hydrogen peroxide. Int. J. Mol. Med. 2002, 9, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Assefa, Z.; Vantieghem, A.; Declercq, W.; Vandenabeele, P.; Vandenheede, J.R.; Merlevede, W.; de Witte, P.; Agostinis, P. The activation of the c-Jun N-terminal kinase and p38 mitogen-activated protein kinase signaling pathways protects HeLa cells from apoptosis following photodynamic therapy with hypericin. J. Biol. Chem. 1999, 274, 8788–8796. [Google Scholar] [CrossRef]
- Buytaert, E.; Callewaert, G.; Hendrickx, N.; Scorrano, L.; Hartmann, D.; Missiaen, L.; Vandenheede, J.R.; Heirman, I..; Grooten, J.; Agostinis, P. Role of endoplasmic reticulum depletion and multidomain proapoptotic BAX and BAK proteins in shaping cell death after hypericin-mediated photodynamic therapy. FASEB J. 2006, 20, 756–758. [Google Scholar] [CrossRef] [PubMed]
- Semelakova, M.; Mikes, J.; Jendzelovsky, R.; Fedorocko, P. The pro-apoptotic and anti-invasive effects of hypericin-mediated photodynamic therapy are enhanced by hyperforin or aristoforin in HT-29 colon adenocarcinoma cells. J. Photochem. Photobiol. B 2012, 117, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Vantieghem, A.; Assefa, Z.; Vandenabeele, P.; Declercq, W.; Courtois, S.; Vandenheede, J.R.; Merlevede, W.; de Witte, P.; Agostinis, P. Hypericin-induced photosensitization of HeLa cells leads to apoptosis or necrosis. Involvement of cytochrome c and procaspase-3 activation in the mechanism of apoptosis. FEBS Lett. 1998, 440, 19–24. [Google Scholar] [CrossRef]
- Ali, S.M.; Olivo, M. Bio-distribution and subcellular localization of Hypericin and its role in PDT induced apoptosis in cancer cells. Int. J. Oncol. 2002, 21, 531–540. [Google Scholar] [CrossRef]
- Chaloupka, R.; Obsil, T.; Plasek, J.; Sureau, F. The effect of hypericin and hypocrellin-A on lipid membranes and membrane potential of 3T3 fibroblasts. Biochim. Biophys. Acta, Biomemb 1999, 1418, 39–47. [Google Scholar] [CrossRef]
- Ho, Y.F.; Wu, M.H.; Cheng, B.H.; Chen, Y.W.; Shih, M.C. Lipid-mediated preferential localization of hypericin in lipid membranes. Biochim. Biophys. Acta, Biomemb 2009, 1788, 1287–1295. [Google Scholar] [CrossRef]
- Huntosova, V.; Nadova, Z.; Dzurova, L.; Jakusova, V.; Sureau, F.; Miskovsky, P. Cell death response of U87 glioma cells on hypericin photoactivation is mediated by dynamics of hypericin subcellular distribution and its aggregation in cellular organelles. Photochem. Photobiol. Sci. 2012, 11, 1428–1436. [Google Scholar] [CrossRef]
- Ritz, R.; Roser, F.; Radomski, N.; Strauss, W.S.; Tatagiba, M.; Gharabaghi, A. Subcellular colocalization of hypericin with respect to endoplasmic reticulum and Golgi apparatus in glioblastoma cells. Anticancer Res. 2008, 28, 2033–2038. [Google Scholar]
- Stegelmeier, B.L. Pyrrolizidine alkaloid-containing toxic plants (Senecio, Crotalaria, Cynoglossum, Amsinckia, Heliotropium, and Echium spp.). Vet. Clin. N. Am. Food A 2011, 27, 419–428. [Google Scholar] [CrossRef]
- Stegelmeier, B.L.; Edgar, J.A.; Colegate, S.M.; Gardner, D.R.; Schoch, T.K.; Coulombe, R.A.; Molyneux, R.J. Pyrrolizidine alkaloid plants, metabolism and toxicity. J. Nat. Toxins 1999, 8, 95–116. [Google Scholar]
- Adams, R. Veterinary Pharmacology and Therapeutics, 8th ed.; Iowa State University Press: Ames, IA, USA, 2001; p. pp. 1201. [Google Scholar]
- Mattocks, A.R. Chemistry and Toxicology of Pyrrolizidine Alkaloids, 1st ed.; Academic Press: Orlando, FL, USA, 1986; p. pp. 393. [Google Scholar]
- Cheeke, P.R. Toxicity and metabolism of pyrrolizidine alkaloids. J. Anim. Sci. 1988, 66, 2343–2350. [Google Scholar] [CrossRef] [PubMed]
- İçen, H.; Sekin, S.; Karataș, A.; Çakmak, F.; Vural, M. E. Hypericum perforatum toxication in Awassi sheep. YYU Veteriner Fakultesi Dergisi 2012, 23, 51–53. [Google Scholar]
- Schrader, A.; Schulz, O.; Völker, H.; Puls, H. Recent plant poisoning in ruminants of northern and eastern Germany. Communication from the practice for the practice. Berl. Munch. Tierarztl. Wochenschr. 2001, 114, 218–221. [Google Scholar] [PubMed]
- Mavromati, J.; Dervishi, M. The issue of intoxication in sheep from the consumption of plants with photodynamic content. J. Multidiscip. Eng. Sci. Tech. 2024, 11, 16509–16512. [Google Scholar]
- Giaretta, P.R.; Panziera, W.; Hammerschmitt, M.E.; Bianchi, R.M.; Galiza, G.J.; Wiethan, I.S.; Bazzi, T.; Barros, C.S. Clinical and pathological aspects of chronic Senecio spp. poisoning in sheep. Pesq. Vet. Bras. 2014, 34, 967–973. [Google Scholar] [CrossRef]
- Barraza, V.; Flores, M.M. Chronic pyrrolizidine alkaloid toxicosis in a heifer. Braz. J. Vet. Pathol. 2021, 14, 206–209. [Google Scholar] [CrossRef]
- Kopecka, A.; Novotna, T.; Svobodova, Z.; Drabkova, Z. Senecio ovatus poisoning in a horse - A case report. Vet. Med. 2024, 69, pp. 329. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



