Submitted:
10 February 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
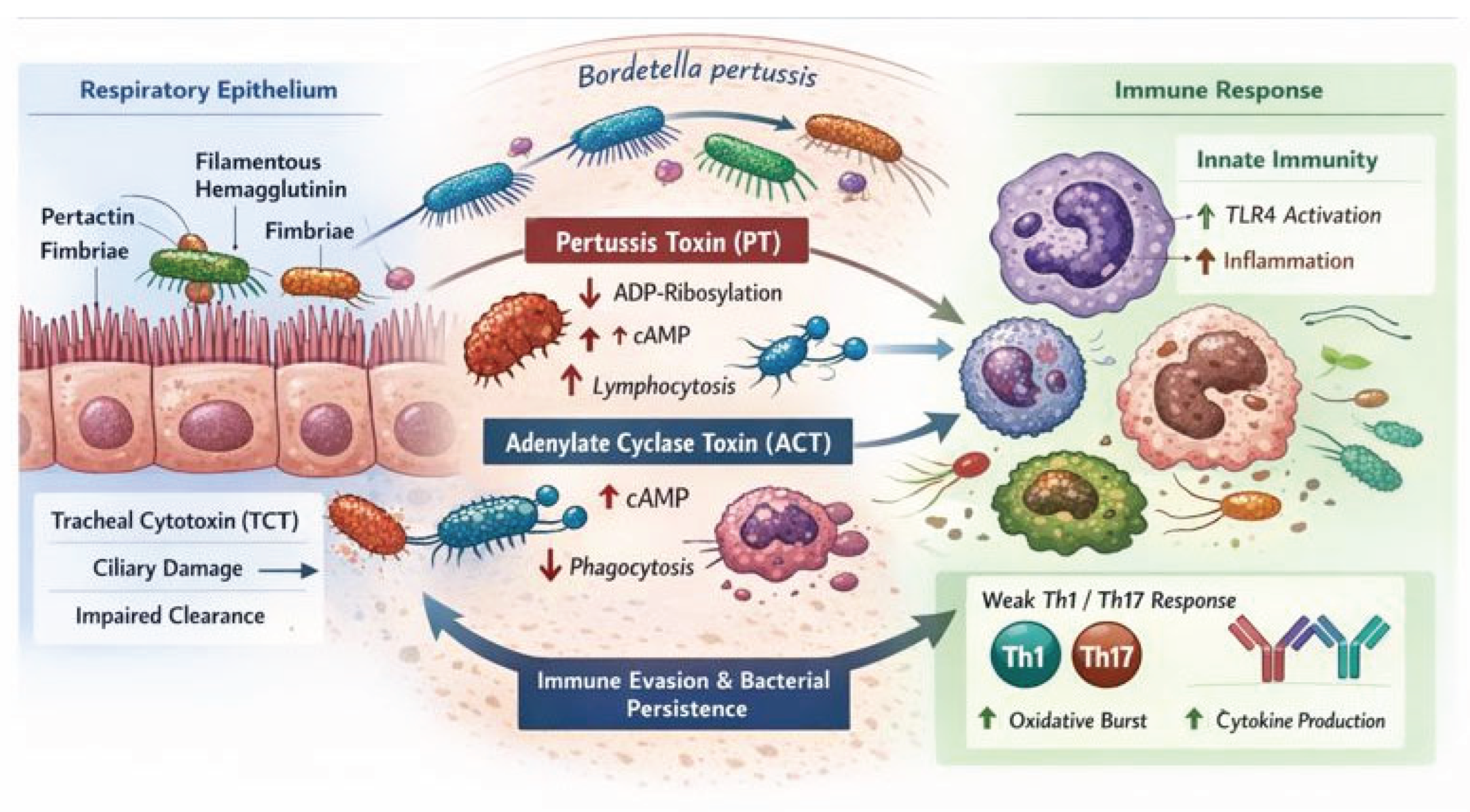
2. Bacterial Structure and Virulence Factors
3. Adhesion and Colonization Mechanisms
4. Toxins Produced by Bordetella pertussis
4.1. Pertussis Toxin (PT)
4.2. Adenylate Cyclase Toxin (ACT)
4.3. Tracheal Cytotoxin (TCT)
4.4. Human Carriage and Controlled Human Infection Models
5. Immune Response to Bordetella pertussis
6. Cellular Interactions During Infection
7. Inflammatory Response in Pertussis
8. Immune Pathogenesis of Bordetella pertussis
9. Immune Pathogenesis as a Blueprint for Vaccine Redesign
9.1. Redesign Target: Block Colonization and Transmission (Mucosal Immunity, T_RM, sIgA)
9.2. Redesign Target: Restore Th1/Th17-Skewing Through Innate-Programming Adjuvants
9.3. Redesign Target: Broaden Antigenic Breadth and Present Antigens in “Pathogen-Like” Formats (Omv, Biofilm/Clinical Isolate Relevance)
9.4. Redesign Target: Consider Live-Attenuated Nasal Vaccines to Recapitulate Infection-Like Immunity Safely
9.5. Structured Comparison of Next-Generation Pertussis Vaccine Platforms
10. Resistance to Antibiotics in Bordetella pertussis
10.1. Molecular Mechanisms of Macrolide Resistance
10.2. Global Epidemiology of Macrolide-Resistant B. pertussis
11. Clinical and Public Health Implications
12. Implications for Surveillance, Stewardship, and Prevention
13. Emerging Issues and Research Directions
14. Future research priorities
Author Contributions
Conflicts of Interest
References
- Melvin JA, Scheller EV, Miller JF, Cotter PA. B. pertussispathogenesis: current and future challenges. Nat Rev Microbiol. 2014;12(4):274-88. [CrossRef]
- Kapil P, Merkel TJ. Pertussis vaccines and protective immunity. Curr Opin Immunol. 2019;59:72-78. [CrossRef]
- Schwartz KL, Kwong JC, Deeks SL, Campitelli MA, Jamieson FB, Marchand-Austin A, Stukel TA, Rosella L, Daneman N, Bolotin S, Drews SJ, Rilkoff H, Crowcroft NS. Effectiveness of pertussis vaccination and duration of immunity. CMAJ. 2016;188(16):E399-E406. [CrossRef]
- Seppälä E, Bråthen Kristoffersen A, Bøås H, Frimann Vestrheim D, Greve-Isdahl M, Freiesleben De Blasio B, Steens A. Pertussis epidemiology including direct and indirect effects of the childhood pertussis booster vaccinations, Norway, 1998-2019. Vaccine. 2022;40(23):3142-3149. [CrossRef]
- Burdin N, Handy LK, Plotkin SA. What Is Wrong with Pertussis Vaccine Immunity? The Problem of Waning Effectiveness of Pertussis Vaccines. Cold Spring Harb Perspect Biol. 2017;9(12):a029454. [CrossRef]
- Mooi FR, Van Der Maas NA, De Melker HE. Pertussis resurgence: waning immunity and pathogen adaptation - two sides of the same coin. Epidemiol Infect. 2014;142(4):685-94. [CrossRef]
- Kardos P, Correia de Sousa J, Heininger U, Konstantopoulos A, MacIntyre CR, Middleton D, Nolan T, Papi A, Rendon A, Rizzo A, Sampson K, Sette A, Sobczyk E, Tan T, Weil-Olivier C, Weinberger B, Wilkinson T, Wirsing von König CH. Understanding the impact of adult pertussis and current approaches to vaccination: A narrative review and expert panel recommendations. Hum Vaccin Immunother. 2024;20(1):2324547. [CrossRef]
- Hodder SL, Mortimer EA Jr. Epidemiology of pertussis and reactions to pertussis vaccine. Epidemiol Rev. 1992;14:243-67. [CrossRef]
- Gregg KA, Merkel TJ. Pertussis Toxin: A Key Component in Pertussis Vaccines? Toxins (Basel). 2019 Sep 21;11(10):557. [CrossRef]
- Duda-Madej A, Łabaz J, Topola E, Bazan H, Viscardi S. Pertussis-A Re-Emerging Threat Despite Immunization: An Analysis of Vaccine Effectiveness and Antibiotic Resistance. Int J Mol Sci. 2025;26(19):9607. [CrossRef]
- Sheng Y, Ma S, Zhou Q, Xu J. Pertussis resurgence: epidemiological trends, pathogenic mechanisms, and preventive strategies. Front Immunol. 2025;16:1618883. [CrossRef]
- Macina D, Evans KE. B. pertussisin School-Age Children, Adolescents and Adults: A Systematic Review of Epidemiology and Mortality in Europe. Infect Dis Ther. 2021;10(4):2071-2118. [CrossRef]
- Zughaier SM, Shafer WM, Stephens DS. Antimicrobial peptides and endotoxin inhibit cytokine and nitric oxide release but amplify respiratory burst response in human and murine macrophages. Cell Microbiol. 2005;7(9):1251-62. [CrossRef]
- Marr N, Hajjar AM, Shah NR, Novikov A, Yam CS, Caroff M, Fernandez RC. Substitution of the B. pertussislipid A phosphate groups with glucosamine is required for robust NF-kappaB activation and release of proinflammatory cytokines in cells expressing human but not murine Toll-like receptor 4-MD-2-CD14. Infect Immun. 2010;78(5):2060-9. [CrossRef]
- Caroff M, Deprun C, Richards JC, Karibian D. Structural characterization of the lipid A of B. pertussis1414 endotoxin. J Bacteriol. 1994;176(16):5156-9. [CrossRef]
- Maeshima N, Evans-Atkinson T, Hajjar AM, Fernandez RC. B. pertussisLipid A Recognition by Toll-like Receptor 4 and MD-2 Is Dependent on Distinct Charged and Uncharged Interfaces. J Biol Chem. 2015;290(21):13440-53. [CrossRef]
- Carbonetti NH. Bordetella pertussis: new concepts in pathogenesis and treatment. Curr Opin Infect Dis. 2016;29(3):287-94. [CrossRef]
- Mattoo S, Cherry JD. Molecular pathogenesis, epidemiology, and clinical manifestations of respiratory infections due to B. pertussisand other Bordetella subspecies. Clin Microbiol Rev. 2005;18(2):326-82. [CrossRef]
- Rossi Paccani S, Benagiano M, Capitani N, Zornetta I, Ladant D, Montecucco C, D’Elios MM, Baldari CT. The adenylate cyclase toxins of Bacillus anthracis and B. pertussispromote Th2 cell development by shaping T cell antigen receptor signaling. PLoS Pathog. 2009;5(3):e1000325. [CrossRef]
- Locht C. Molecular aspects of B. pertussispathogenesis. Int Microbiol. 1999;2(3):137-44.
- Relman DA, Domenighini M, Tuomanen E, Rappuoli R, Falkow S. Filamentous hemagglutinin of Bordetella pertussis: nucleotide sequence and crucial role in adherence. Proc Natl Acad Sci U S A. 1989;86(8):2637-41. [CrossRef]
- Geuijen CA, Willems RJ, Bongaerts M, Top J, Gielen H, Mooi FR. Role of the B. pertussisminor fimbrial subunit, FimD, in colonization of the mouse respiratory tract. Infect Immun. 1997;65(10):4222-8. [CrossRef]
- Cherry JD, Gornbein J, Heininger U, Stehr K. A search for serologic correlates of immunity to B. pertussiscough illnesses. Vaccine. 1998;16(20):1901-6. [CrossRef]
- Chen Q, Stibitz S. The BvgASR virulence regulon of Bordetella pertussis. Curr Opin Microbiol. 2019;47:74-81. [CrossRef]
- Burns DL. Secretion of Pertussis Toxin from Bordetella pertussis. Toxins (Basel). 2021;13(8):574. [CrossRef]
- Cundell DR, Kanthakumar K, Taylor GW, Goldman WE, Flak T, Cole PJ, Wilson R. Effect of tracheal cytotoxin from B. pertussison human neutrophil function in vitro. Infect Immun. 1994;62(2):639-43. [CrossRef]
- Tuomanen EI, Hendley JO. Adherence of B. pertussisto human respiratory epithelial cells. J Infect Dis. 1983;148(1):125-30. [CrossRef]
- Leininger E, Roberts M, Kenimer JG, Charles IG, Fairweather N, Novotny P, Brennan MJ. Pertactin, an Arg-Gly-Asp-containing B. pertussissurface protein that promotes adherence of mammalian cells. Proc Natl Acad Sci U S A. 1991;88(2):345-9. [CrossRef]
- Carbonetti NH. Contribution of pertussis toxin to the pathogenesis of pertussis disease. Pathog Dis. 2015;73(8):ftv073. [CrossRef]
- Hewlett EL, Kim KJ, Lee SJ, Gray MC. Adenylate cyclase toxin from Bordetella pertussis: current concepts and problems in the study of toxin functions. Int J Med Microbiol. 2000;290(4-5):333-5. [CrossRef]
- Guiso N. Bordetella Adenylate Cyclase-Hemolysin Toxins. Toxins (Basel). 2017;9(9):277. [CrossRef]
- Graf R, Codina J, Birnbaumer L. Peptide inhibitors of ADP-ribosylation by pertussis toxin are substrates with affinities comparable to those of the trimeric GTP-binding proteins. Mol Pharmacol. 199242(5):760-4.
- Heiss LN, Lancaster JR Jr, Corbett JA, Goldman WE. Epithelial autotoxicity of nitric oxide: role in the respiratory cytopathology of pertussis. Proc Natl Acad Sci U S A. 1994;91(1):267-70. [CrossRef]
- Eberhardt CS, Siegrist CA. What Is Wrong with Pertussis Vaccine Immunity? Inducing and Recalling Vaccine-Specific Immunity. Cold Spring Harb Perspect Biol. 2017;9(12):a029629. [CrossRef]
- Guiso N, Koenig CHWV. Pertussis Vaccination: The Challenges Ahead. Semin Respir Crit Care Med. 2025;46(1):14-19. [CrossRef]
- Ricci S, Rappuoli R, Scarlato V. The pertussis toxin liberation genes of B. pertussisare transcriptionally linked to the pertussis toxin operon. Infect Immun. 1996;64(4):1458-60. [CrossRef]
- Lam C, Octavia S, Ricafort L, Sintchenko V, Gilbert GL, Wood N, McIntyre P, Marshall H, Guiso N, Keil AD, Lawrence A, Robson J, Hogg G, Lan R. Rapid increase in pertactin-deficient B. pertussisisolates, Australia. Emerg Infect Dis. 2014;20(4):626-33. [CrossRef]
- Bart MJ, Harris SR, Advani A, Arakawa Y, Bottero D, Bouchez V, Cassiday PK, Chiang CS, Dalby T, Fry NK, Gaillard ME, van Gent M, Guiso N, Hallander HO, Harvill ET, He Q, van der Heide HG, Heuvelman K, Hozbor DF, Kamachi K, Karataev GI, Lan R, Lutyńska A, Maharjan RP, Mertsola J, Miyamura T, Octavia S, Preston A, Quail MA, Sintchenko V, Stefanelli P, Tondella ML, Tsang RS, Xu Y, Yao SM, Zhang S, Parkhill J, Mooi FR. Global population structure and evolution of B. pertussisand their relationship with vaccination. mBio. 2014;5(2):e01074. [CrossRef]
- Park J, Zhang Y, Buboltz AM, Zhang X, Schuster SC, Ahuja U, Liu M, Miller JF, Sebaihia M, Bentley SD, Parkhill J, Harvill ET. Comparative genomics of the classical Bordetella subspecies: the evolution and exchange of virulence-associated diversity amongst closely related pathogens. BMC Genomics. 2012;13:545. [CrossRef]
- Confer DL, Eaton JW. Phagocyte impotence caused by an invasive bacterial adenylate cyclase. Science. 1982;217(4563):948-50. [CrossRef]
- Bouchez V, Douché T, Dazas M, Delaplane S, Matondo M, Chamot-Rooke J, Guiso N. Characterization of Post-Translational Modifications and Cytotoxic Properties of the Adenylate-Cyclase Hemolysin Produced by Various B. pertussisand Bordetella parapertussis Isolates. Toxins (Basel). 2017;9(10):304. [CrossRef]
- Kroes MM, Miranda-Bedate A, Jacobi RHJ, van Woudenbergh E, den Hartog G, van Putten JPM, de Wit J, Pinelli E. Bordetella pertussis-infected innate immune cells drive the anti-pertussis response of human airway epithelium. Sci Rep. 2022;12(1):3622. [CrossRef]
- Goldman WE, Klapper DG, Baseman JB. Detection, isolation, and analysis of a released B. pertussisproduct toxic to cultured tracheal cells. Infect Immun. 1982;36(2):782-94. [CrossRef]
- Rosenthal RS, Nogami W, Cookson BT, Goldman WE, Folkening WJ. Major fragment of soluble peptidoglycan released from growing B. pertussisis tracheal cytotoxin. Infect Immun. 1987;55(9):2117-20. [CrossRef]
- Cookson BT, Tyler AN, Goldman WE. Primary structure of the peptidoglycan-derived tracheal cytotoxin of Bordetella pertussis. Biochemistry. 1989;28(4):1744-9. [CrossRef]
- Heiss LN, Flak TA, Lancaster JR Jr, McDaniel ML, Goldman WE. Nitric oxide mediates B. pertussistracheal cytotoxin damage to the respiratory epithelium. Infect Agents Dis. 1993 Aug;2(4):173-7.
- Solans L, Locht C. The Role of Mucosal Immunity in Pertussis. Front Immunol. 2019 Jan 14;9:3068. [CrossRef]
- Smith AM, Guzmán CA, Walker MJ. The virulence factors of Bordetella pertussis: a matter of control. FEMS Microbiol Rev. 2001 May;25(3):309-33. [CrossRef]
- Wendelboe AM, Njamkepo E, Bourillon A, Floret DD, Gaudelus J, Gerber M, Grimprel E, Greenberg D, Halperin S, Liese J, Muñoz-Rivas F, Teyssou R, Guiso N, Van Rie A; Infant Pertussis Study Group. Transmission of B. pertussisto young infants. Pediatr Infect Dis J. 2007 Apr;26(4):293-9. [CrossRef]
- Cherry JD. The epidemiology of pertussis: a comparison of the epidemiology of the disease pertussis with the epidemiology of B. pertussisinfection. Pediatrics. 2005;115(5):1422-7. [CrossRef]
- Bartkus JM, Juni BA, Ehresmann K, Miller CA, Sanden GN, Cassiday PK, Saubolle M, Lee B, Long J, Harrison AR Jr, Besser JM. Identification of a mutation associated with erythromycin resistance in Bordetella pertussis: implications for surveillance of antimicrobial resistance. J Clin Microbiol. 2003 Mar;41(3):1167-72. [CrossRef]
- Wang Z, Han R, Liu Y, Du Q, Liu J, Ma C, Li H, He Q, Yan Y. Direct Detection of Erythromycin-Resistant B. pertussisin Clinical Specimens by PCR. J Clin Microbiol. 2015 Nov;53(11):3418-22. [CrossRef]
- Petráčková D, Farman MR, Amman F, Linhartová I, Dienstbier A, Kumar D, Držmíšek J, Hofacker I, Rodriguez ME, Večerek B. Transcriptional profiling of human macrophages during infection with Bordetella pertussis. RNA Biol. 2020 May;17(5):731-742. [CrossRef]
- Zhang Q, Li M, Wang L, Xin T, He Q. High-resolution melting analysis for the detection of two erythromycin-resistant B. pertussisstrains carried by healthy schoolchildren in China. Clin Microbiol Infect. 2013;19(6):E260-2. [CrossRef]
- Wendelboe AM, Van Rie A, Salmaso S, Englund JA. Duration of immunity against pertussis after natural infection or vaccination. Pediatr Infect Dis J. 2005;24(5 Suppl):S58-61. [CrossRef]
- Warfel JM, Edwards KM. Pertussis vaccines and the challenge of inducing durable immunity. Curr Opin Immunol. 2015;35:48-54. [CrossRef]
- Higgins SC, Jarnicki AG, Lavelle EC, Mills KH. TLR4 mediates vaccine-induced protective cellular immunity to Bordetella pertussis: role of IL-17-producing T cells. J Immunol. 2006;177(11):7980-9. [CrossRef]
- Fedele G, Nasso M, Spensieri F, Palazzo R, Frasca L, Watanabe M, Ausiello CM. Lipopolysaccharides from B. pertussisand Bordetella parapertussis differently modulate human dendritic cell functions resulting in divergent prevalence of Th17-polarized responses. J Immunol. 2008;181(1):208-16. [CrossRef]
- Higgs R, Higgins SC, Ross PJ, Mills KH. Immunity to the respiratory pathogen Bordetella pertussis. Mucosal Immunol. 2012;5(5):485-500. [CrossRef]
- Ross PJ, Sutton CE, Higgins S, Allen AC, Walsh K, Misiak A, Lavelle EC, McLoughlin RM, Mills KH. Relative contribution of Th1 and Th17 cells in adaptive immunity to Bordetella pertussis: towards the rational design of an improved acellular pertussis vaccine. PLoS Pathog. 2013;9(4):e1003264. [CrossRef]
- Warfel JM, Zimmerman LI, Merkel TJ. Acellular pertussis vaccines protect against disease but fail to prevent infection and transmission in a nonhuman primate model. Proc Natl Acad Sci U S A. 2014;111(2):787-92. [CrossRef]
- Rodríguez ME, Hellwig SM, Pérez Vidakovics ML, Berbers GA, van de Winkel JG. B. pertussisattachment to respiratory epithelial cells can be impaired by fimbriae-specific antibodies. FEMS Immunol Med Microbiol. 2006;46(1):39-47. [CrossRef]
- Guiso N, Capiau C, Carletti G, Poolman J, Hauser P. Intranasal murine model of B. pertussisinfection. I. Prediction of protection in human infants by acellular vaccines. Vaccine. 1999;17(19):2366-76. [CrossRef]
- Belcher T, Dubois V, Rivera-Millot A, Locht C, Jacob-Dubuisson F. Pathogenicity and virulence of B. pertussis and its adaptation to its strictly human host. Virulence. 2021;12(1):2608-2632. [CrossRef]
- Weiss AA, Patton AK, Millen SH, Chang SJ, Ward JI, Bernstein DI. Acellular pertussis vaccines and complement killing of Bordetella pertussis. Infect Immun. 2004;72(12):7346-51. [CrossRef]
- Mills KH. Immunity to Bordetella pertussis. Microbes Infect. 2001 Jul;3(8):655-77. [CrossRef]
- von Olleschik-Elbheim L, el Bayâ A, Schmidt MA. ADP-ribosylation of alpha-Gi proteins by pertussis toxin. Positional dissection of acceptor sites using membrane anchored synthetic peptides. Methods Mol Biol. 2000;145:203-17. [CrossRef]
- Carbonetti NH. Pertussis leukocytosis: mechanisms, clinical relevance and treatment. Pathog Dis. 2016;74(7):ftw087. [CrossRef]
- Luker KE, Tyler AN, Marshall GR, Goldman WE. Tracheal cytotoxin structural requirements for respiratory epithelial damage in pertussis. Mol Microbiol. 1995;16(4):733-43. [CrossRef]
- Allen AC, Wilk MM, Misiak A, Borkner L, Murphy D, Mills KHG. Sustained protective immunity against B. pertussisnasal colonization by intranasal immunization with a vaccine-adjuvant combination that induces IL-17-secreting TRM cells. Mucosal Immunol. 2018;11(6):1763-1776. [CrossRef]
- Cherry JD. Why do pertussis vaccines fail? Pediatrics. 2012;129(5):968-70. [CrossRef]
- Ni Chasaide C, Mills KHG. Next-Generation Pertussis Vaccines Based on the Induction of Protective T Cells in the Respiratory Tract. Vaccines (Basel). 2020;8(4):621. [CrossRef]
- Kapil P, Merkel TJ. Pertussis vaccines and protective immunity. Curr Opin Immunol. 2019;59:72-78. [CrossRef]
- Boehm DT, Wolf MA, Hall JM, Wong TY, Sen-Kilic E, Basinger HD, Dziadowicz SA, de la Paz Gutierrez M, Blackwood CB, Bradford SD, Begley KA, Witt WT, Varney ME, Barbier M, Damron FH. Intranasal acellular pertussis vaccine provides mucosal immunity and protects mice from Bordetella pertussis. NPJ Vaccines. 2019;4:40. [CrossRef]
- Misiak A, Leuzzi R, Allen AC, Galletti B, Baudner BC, D’Oro U, O’Hagan DT, Pizza M, Seubert A, Mills KHG. Addition of a TLR7 agonist to an acellular pertussis vaccine enhances Th1 and Th17 responses and protective immunity in a mouse model. Vaccine. 2017;35(39):5256-5263. [CrossRef]
- Mata-Haro V, Cekic C, Martin M, Chilton PM, Casella CR, Mitchell TC. The vaccine adjuvant monophosphoryl lipid A as a TRIF-biased agonist of TLR4. Science. 2007;316(5831):1628-1632. [CrossRef]
- Gaillard ME, Bottero D, Errea A, Ormazabal M, Zurita ME, Moreno G, Rumbo M, Castuma C, Bartel E, Flores D, van der Ley P, van der Ark A, Hozbor DF. Acellular pertussis vaccine based on outer membrane vesicles capable of conferring both long-lasting immunity and protection against different strain genotypes. Vaccine. 2014;32(8):931-937. [CrossRef]
- Raeven RHM, Rockx-Brouwer D, Kanojia G, van der Maas L, Bindels THE, Ten Have R, van Riet E, Metz B, Kersten GFA. Intranasal immunization with outer membrane vesicle pertussis vaccine confers broad protection through mucosal IgA and Th17 responses. Sci Rep. 2020;10(1):7396. [CrossRef]
- Keech C, Miller VE, Rizzardi B, Hoyle C, Pryor MJ, Ferrand J, Solovay K, Thalen M, Noviello S, Goldstein P, Gorringe A, Cavell B, He Q, Barkoff AM, Rubin K, Locht C. Immunogenicity and safety of BPZE1, an intranasal live attenuated pertussis vaccine, versus tetanus-diphtheria-acellular pertussis vaccine: a randomised, double-blind, phase 2b trial. Lancet. 2023;401(10379):843-855. [CrossRef]
- Gbesemete D, Ramasamy MN, Ibrahim M, Hill AR, Raud L, Ferreira DM, Guy J, Dale AP, Laver JR, Coutinho T, Faust SN, Reed TAN, Babbage G, Weissfeld L, Lang W, Locht C, Samal V, Goldstein P, Solovay K, Rubin K, Noviello S, Read RC. Efficacy, immunogenicity, and safety of the live attenuated nasal pertussis vaccine, BPZE1, in the UK: a randomised, placebo-controlled, phase 2b trial using a controlled human infection model with virulent Bordetella pertussis. Lancet Microbe. 2025;6(12):101211. [CrossRef]
- Cherry JD. Pertussis in young infants throughout the world. Clin Infect Dis. 2016 Dec 1;63(Suppl 4):S119-S122. [CrossRef]
- Altunaiji S, Kukuruzovic R, Curtis N, Massie J. Antibiotics for whooping cough (pertussis). Cochrane Database Syst Rev. 2007;2007(3):CD004404. [CrossRef]
- Li L, Deng J, Ma X, Zhou K, Meng Q, Yuan L, Shi W, Wang Q, Li Y, Yao K. High prevalence of macrolide-resistant B. pertussis and ptxP1 genotype, mainland China, 2014-2016. Emerg Infect Dis. 2019;25(12):2205-2214. [CrossRef]
- Wang Z, Li Y, Hou T, Liu X, Liu Y, Yu T, Chen Z, Gao Y, Li H, He Q. Appearance of macrolide-resistant B. pertussis strains in China. Antimicrob Agents Chemother. 2013t;57(10):5193-5194. [CrossRef]
- Fry NK, Duncan J, Vaghji L, George RC, Harrison TG. Antimicrobial susceptibility testing of historical and recent clinical isolates of B. pertussis in the United Kingdom using the Etest method. Eur J Clin Microbiol Infect Dis. 2010;29(9):1183-1185. [CrossRef]
- Mai Q, Wen J, Luo Y, Guo J, Qin Y, Lai W, Deng W, Ji C, Mai R, Zheng M, Chen Z, Chen Y, Gu C, Guo L, Li H, Tang Y, Huang D, Luo M. Molecular epidemiology and increasing macrolide resistance of B. pertussisisolates in Guangzhou, China. BMC Infect Dis. 2025;25(1):1152. [CrossRef]
- Iwasaki T, Koide K, Kido T, Nakagawa S, Goto M, Kenri T, Suzuki H, Otsuka N, Takada H. Fatal case of macrolide-resistant B. pertussis infection, Japan, 2024. J Infect Chemother. 2025 Jul;31(7):102727. [CrossRef] [PubMed]
- Mooi FR. B. pertussis and vaccination: the persistence of a genetically monomorphic pathogen. Infect Genet Evol. 2010;10(1):36-49. [CrossRef]
- Wardlaw AC, Parton R. B. pertussistoxins. Pharmacol Ther. 1982;19(1):1-53. [CrossRef]
- Tuomanen E, Weiss A. Filamentous hemagglutinin and pertussis toxin promote adherence of B. pertussis to cilia. Infect Immun. 1985;50(3):575-579.
- Centers for Disease Control and Prevention (CDC). Treatment of Pertussis (Whooping Cough): Clinical Care. Updated Dec 2, 2025. Available from: https://www.cdc.gov/pertussis/hcp/clinical-care/index.html.
- Tiwari T, Murphy TV, Moran J; National Immunization Program, CDC. Recommended antimicrobial agents for the treatment and postexposure prophylaxis of pertussis: 2005 CDC Guidelines. MMWR Recomm Rep. 2005;54(RR-14):1-16.
- Eberly MD, Eide MB, Thompson JL, Nylund CM. Azithromycin in early infancy and pyloric stenosis. Pediatrics. 2015 Mar;135(3):483-8. [CrossRef] [PubMed] [PubMed Central]
- Bartkus JM, Juni BA, Ehresmann K, Miller CA, Sanden GN, Cassiday PK, Saubolle M, Lee B, Long J, Harrison AR Jr, Besser JM. Identification of a mutation associated with erythromycin resistance in Bordetella pertussis: implications for surveillance of antimicrobial resistance. J Clin Microbiol. 2003 Mar;41(3):1167-72. [CrossRef]
- Ivaska L, Barkoff AM, Mertsola J, He Q. Macrolide Resistance in Bordetella pertussis: Current Situation and Future Challenges. Antibiotics (Basel). 2022;11(11):1570. [CrossRef]
- European Centre for Disease Prevention and Control (ECDC). Antimicrobial susceptibility testing of B. pertussis– Technical report. ECDC; 2022. https://www.ecdc.europa.eu/sites/default/files/documents/bordetella-pertussis-antimicrobial-susceptibility-testing-2022_0.pdf.
- Morse SI, Morse JH. Isolation and properties of the leukocytosis- and lymphocytosis-promoting factor of Bordetella pertussis. J Exp Med. 1976;143(6):1483-1502. [CrossRef]
- Weiss AA, Hewlett EL. Virulence factors of Bordetella pertussis. Annu Rev Microbiol. 1986;40:661-686. [CrossRef]
- Redhead K, Watkins J, Barnard A, Mills KH. Effective immunization against B. pertussis respiratory infection in mice is dependent on induction of cell-mediated immunity. Infect Immun. 1993;61(8):3190-3198. [CrossRef]
- Mills KH, Barnard A, Watkins J, Redhead K. Cell-mediated immunity to Bordetella pertussis: role of Th1 cells in bacterial clearance in a murine respiratory infection model. Infect Immun. 1993;61(2):399-410. [CrossRef]
- Hellwig SM, van Spriel AB, Schellekens JF, Mooi FR, van de Winkel JG. Immunoglobulin A-mediated protection against B. pertussis infection. Infect Immun. 2001;69(8):4846-4850. [CrossRef]
- Flak TA, Goldman WE. Autotoxicity of nitric oxide in airway disease. Am J Respir Crit Care Med. 1996;154(4 Pt 1):S202-S206.
| Platform (typical route) | Mucosal IgA | Th1/Th17 | Airway T_RM | Prevents colonization / transmission | Key evidence (selected) |
|---|---|---|---|---|---|
| Licensed acellular pertussis (aP; IM) |
Low | Weak (Th2-leaning with alum) | Limited | Poor (protection mainly against disease, not carriage) | Strong serum anti-toxin IgG and reduced severe disease but limited mucosal immunity and weak Th1/Th17; does not reliably block carriage/transmission [56,72,73]. |
| OMV-based pertussis vaccines (IN or IM; often IN in next-gen designs) | High with intranasal delivery | Robust Th17 (and Th1) in preclinical models | Airway-local T-cell memory (preclinical) | Improved; intranasal OMV prevented nasal and lung colonization in mice | Intranasal OMV vaccination induced mucosal IgA and Th17 responses and prevented colonization in both nasal cavity and lungs in mice [78]. |
| BPZE1 live-attenuated nasal vaccine (IN) | High (mucosal + systemic responses) | Th1/Th17-skewed, infection-like programming | Expected strong airway T_RM (supported in models) | Yes/strong signal; reduces or prevents colonization in controlled settings | Human trials show acceptable safety and immunogenicity [79]; a controlled human infection model phase 2b study showed that a single intranasal dose could prevent or substantially reduce colonization by virulent B. pertussis and markedly lower bacterial burden versus placebo [80]. |
| Intranasal subunit strategies (e.g., aP antigens reformulated for IN delivery ± PRR agonists) | Moderate-High (route-dependent) | Improved when paired with innate-programming adjuvants (e.g., TLR agonists) | Potential to seed airway memory (data emerging) | Partial; reduction in upper-airway burden in animals; clinical evidence limited | Intranasal delivery of acellular antigens can elicit mucosal IgA and IL-17A and reduce bacterial burden in upper and lower airways in mice, highlighting the importance of route and adjuvanting to restore mucosal correlates [74,75]. |
| Population / scenario | Preferred agent(s) when MRBP suspected/confirmed | Key safety constraints | Role of susceptibility testing | Notes in high-MRBP regions |
|---|---|---|---|---|
| Neonate (<1 month) | Azithromycin remains the only guideline-endorsed macrolide option; if MRBP confirmed, individualized management with specialist input | TMP–SMX contraindicated <2 months; erythromycin linked to infantile hypertrophic pyloric stenosis; limited safety/efficacy data for alternatives | Prioritize rapid genotyping (A2047G) + culture/qPCR where feasible | If MRBP is confirmed, there is no well-validated oral alternative; focus on supportive care, infection control, and expert consultation; consider investigational/locally recommended parenteral options only within protocolized care [68,91,92,93] |
| Infant (1–<2 months) | Azithromycin (standard of care); if MRBP confirmed, specialist-led individualized approach | TMP–SMX not recommended <2 months; tetracyclines/fluoroquinolones generally avoided | Genotype/culture to confirm MRBP whenever possible | Highest-risk group for severe disease; hospital monitoring often warranted; prioritize eradication of household source cases [68,91,92,93] |
| ≥2 months (children/adolescents) | TMP–SMX (alternative agent when MRBP suspected/confirmed) | Avoid in sulfonamide allergy; monitor for adverse effects; avoid in late pregnancy if adolescent is pregnant | Genotyping useful to avoid ineffective macrolides; phenotypic MIC testing may support local surveillance | If TMP–SMX cannot be used, evidence for alternatives is limited; some reports suggest in vitro/in vivo activity of certain β-lactam/β-lactamase inhibitor combinations, but these are not standard guideline therapies [54,92,94,95] |
| Adults (non-pregnant) | TMP–SMX when MRBP suspected/confirmed; macrolides only if susceptibility likely/confirmed | Contraindications as above; consider drug interactions | Genotyping/culture where available to guide therapy and prophylaxis | For non-severe disease, the main antibiotic goal is to reduce transmission; if MRBP is circulating, public-health measures and targeted prophylaxis strategies become more important [54,92,94,95] |
| Pregnancy / immediate postpartum | Macrolide (azithromycin) is typically preferred when susceptible; in high-MRBP settings, prioritize susceptibility testing and individualized risk–benefit decisions | TMP–SMX generally avoided (especially 1st trimester and near term); tetracyclines/fluoroquinolones generally avoided in pregnancy | Rapid genotyping is particularly valuable to avoid prolonged ineffective therapy | Because robust MRBP-specific pregnancy-safe alternatives are lacking, prevention (maternal vaccination) + early case identification and infection control are critical; consult obstetrics/ID for any non-standard regimens [91,92,93] |
| Immunocompromised hosts | Treat early; TMP–SMX may be used when MRBP suspected/confirmed if not contraindicated | Higher risk of complications; drug–drug interactions common; monitor closely | Lower threshold for microbiologic confirmation and follow-up sampling | Consider longer observation and follow-up to ensure clearance; integrate with infection control to reduce nosocomial transmission [92,94,95,96] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




