1. Introduction
Since 2010, there has been a sharp increase in the number of clinical trials conducted in sub-Saharan Africa (sSA) to address unmet public health gaps in the region, with over 80% of the trials being randomised controlled trials [
1]. Some of these trials have made better vaccine and treatment options available for diseases of high burden among children, including infectious diseases such as malaria, typhoid fever, bacterial and viral pneumonia and meningitis, and other poverty-related diseases [
2,
3,
4].
Compared to the general paediatric population, there are known groups of children at increased risk of contracting these diseases and adverse outcomes due to their fragile immune systems and other factors. These groups include malnourished children, children living with HIV (CLHIV) and children with sickle cell disease (SCD), all relatively prevalent in sSA.
Malnutrition
Malnutrition refers to deficiencies or excesses in nutrient intake, imbalance of essential nutrients or impaired nutrient utilisation. The double burden of malnutrition consists of both undernutrition and overweight and obesity, as well as diet-related noncommunicable diseases. Undernutrition manifests in four broad forms: wasting, stunting, underweight, and micronutrient deficiencies [
5]. In this study, we focused mainly on the inclusion of children with undernutrition. The burden of undernutrition remains high in Africa, particularly in sSA. In 2024, wasting, a low weight-for-length or height, affected 11.7 [95% confidence interval: 10.0-13.5] million children under 5 years in Africa, with 9.7 million (83%) living in sSA(6). Stunting, a low length or height-for-age resulting from chronic and recurrent malnutrition, had a very high burden, affecting 64.8 [63.5-66.1] million children under 5 years in Africa, with close to 60 million (92%) living in sSA [
6].
HIV
Though sSA is showing the strongest decline in the numbers of new HIV infections, the region is still disproportionately affected by HIV, accounting in 2024 for half of all new HIV infections and for two-thirds of the global population of people living with HIV [
7]. It is estimated that in 2024, there were 98,000 new infections among children aged 0-14 years in this region, and a total of 26.3 million persons were living with HIV [
8].
Sickle Cell Disease (SCD)
SCD is one of the most common life-threatening genetic disorders among people of African heritage. Globally, around ¾ of those affected live in sSA, where more than 300,000 babies are born with SCD yearly. Estimates predict an exponential increase in SCD’s prevalence, with more than 14 million births affected from 2010 to 2050 [
9]. Persons living with SCD usually develop chronic health issues, including anaemia, and are at increased risk of infection. Many of the known cost-efficient lifesaving healthcare interventions for SCD are not routinely implemented in most sSA countries, and longitudinal cohort studies are largely missing for this population group [
10]. Despite this alarming picture, research funding for this condition has only recently begun to grow in some settings, and remains modest [
11].
Both the ICH E8(R1) guideline on General Considerations for Clinical Studies and the ICH E6(R3) Good Clinical Practice guideline emphasise that unnecessary exclusion of specific populations should be avoided when designing clinical trials. Trials should aim for inclusivity to ensure that study participants appropriately reflect those who will ultimately use the medical product in routine care. This principle is further supported by the WHO’s Guidance for Good Clinical Practice, which advocates for equitable access to research participation, particularly for high-risk and underserved populations [
12,
13,
14].
The rising number of clinical trials, including those targeting infectious diseases of high burden in sSA presents an opportunity to assess the specific efficacy of new lifesaving interventions on these vulnerable children and to provide data and recommendations which will inform the routine care provided to these children.
2. Materials and Methods
Protocol and Guidelines
The protocol of this study was registered prospectively in PROSPERO with the ID CRD42023478142.
Eligibility criteria
Inclusion: Clinical trials registered in 2010–2023 in ClinicalTrials.gov (CTG), Pan African Clinical Trials Registry (PACTR), or International Standard Randomized Controlled Trials Number (ISRCTN), assessing vaccines, drugs, nutritional supplements, or medical devices, enrolling participants aged 1–18 years in sSA. We also included trials that specifically targeted the three conditions to provide a comprehensive view of trial inclusion.
Exclusion: Diagnostic trials, phase 1 trials, non-interventional studies, behavioural trials and trials evaluating procedures.
Study records
Record extraction: Data were extracted from the three registries and combined into a single database, including study ID, title, disease area, intervention type, study design, phase, age group, gender, and inclusion/exclusion criteria.
Identification and removal of duplicates: Duplicates were removed by comparing study IDs, and cross-registry duplicates were identified using standardised titles and approximate string matching. Two independent reviewers confirmed and flagged duplicates.
Disease categorisation followed the International Classification of Diseases, 11th revision ( ICD-11).
Eligibility Classification and Algorithm
An automated text-search algorithm was developed to classify studies into
excluded, probably excluded, included, or probably included based on trial registry information. Explicit references to one of the three conditions in the inclusion or exclusion criteria were coded as “included” and “excluded”, respectively. When no explicit mention was made but broad terms were used, trials were coded as “probably included” or “probably excluded”. Key terms were standardised, and synonyms and variations were tested to improve classification. Two independent reviewers validated the classifications made by the algorithm by manually reviewing a subset of trials, with iterative refinements made to ensure consistency between algorithmic and manual coding. Detailed search terms and the classification algorithm used are provided in
Supplementary Table S1.
Statistical Analysis
Proportion of exclusion was calculated by combining excluded and probably excluded trials; corresponding 95% confidence intervals (CIs) were computed using the Wilson method [
17]. A chi-square test assessed factors associated with exclusion (trial phase, period of trial registration, study design, and intervention type). A multivariable logistic regression model including all characteristics was then used to evaluate whether these associations persisted after adjustment. Sensitivity analysis was conducted on trials with confirmed inclusion/exclusion status. Analyses were performed using R version 4.4.1 [
18].
Patient and Public Involvement
For this study, it was not possible to involve patients or the public in the design, or conduct, or reporting, or dissemination plans.
3. Results
This section may be divided by subheadings. It should provide a concise and precise description of the experimental results, their interpretation as well as the experimental conclusions that can be drawn.
3.1. Study Selection
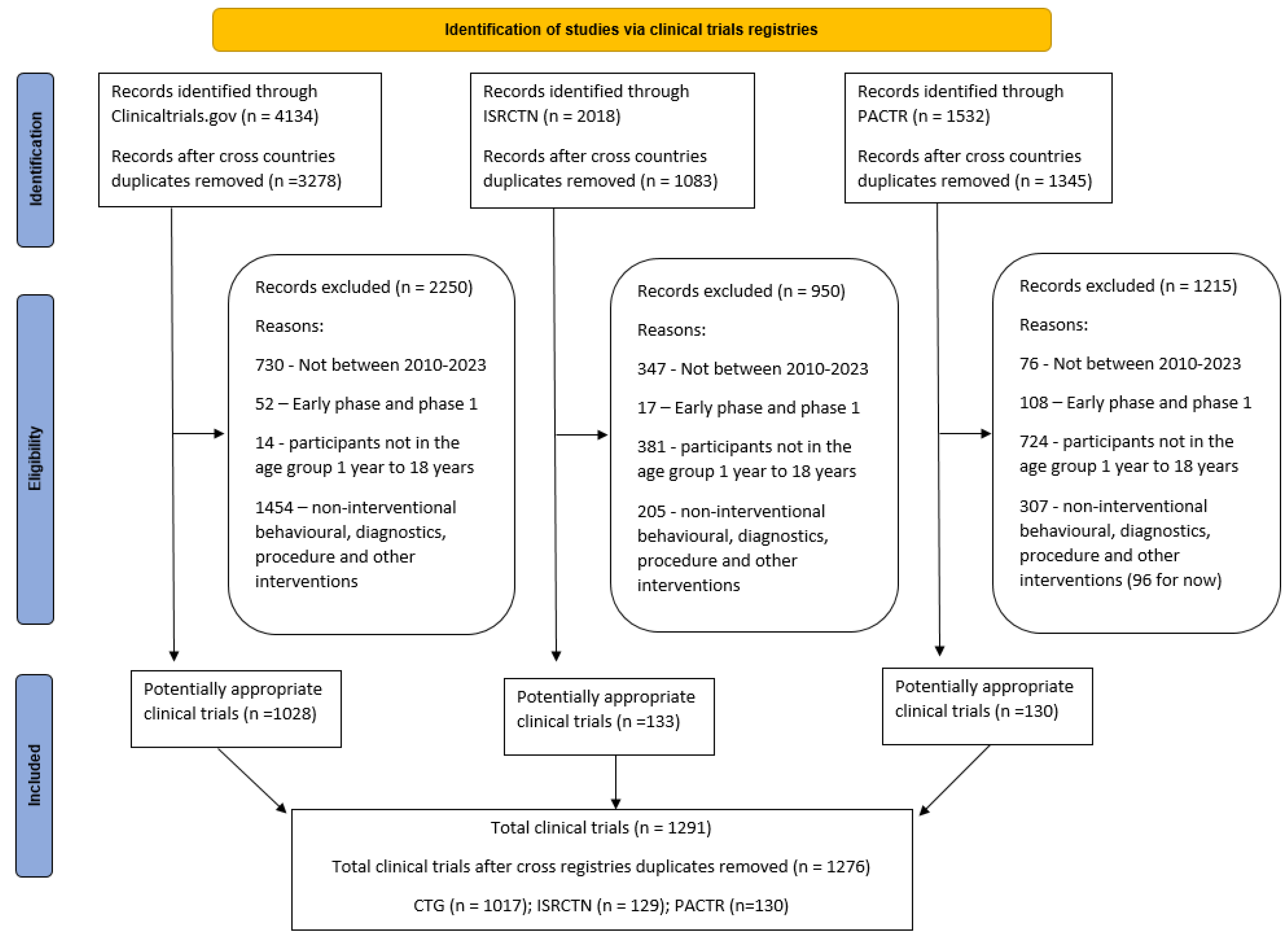
A total of 7,684 records were retrieved from ClinicalTrials.gov, PACTR, and ISRCTN registries. Following the removal of 1,978 duplicates, the exclusion of 4,415 records after applying filters for study period, intervention type, and age range, and further removal of 15 cross-registry duplicates, 1,276 unique clinical trials were retained for this study. The steps showing identification, screening, eligibility, and inclusion of records are presented in
Figure 1 (PRISMA flowchart).
3.2. Study Characteristics
The study characteristics are presented in
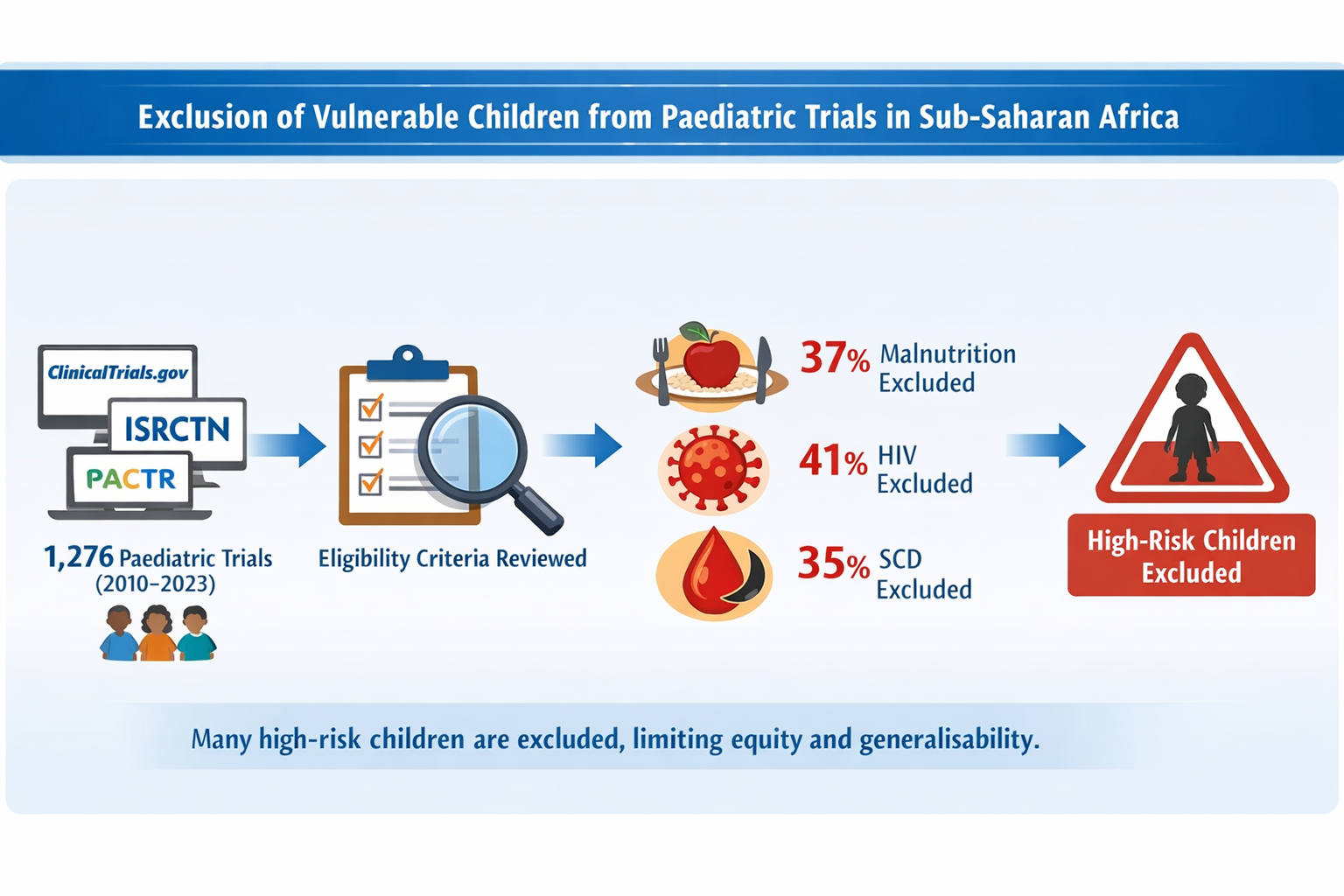
Table 1. The most common trial characteristics included phase 3 trials (41.9% [535/1276]), non-cluster randomised designs (94.9% [1211/1276]), drug trials (62.8% [801/1276]), and targeting infectious and parasitic diseases (58.3% [744/1276])- See Supplementary Table 2. The proportions of trials targeting primarily malnutrition, HIV and SCD were 5.3% (68/1276), 8.9% (114/1276) and 3.4% (44/1276), respectively.
3.3. Exclusion of the Target Population Groups
Among the trials that did not target primarily malnutrition, HIV and SCD, the proportions excluding or probably excluding the three populations are presented in
Table 2.
3.4. Factors Associated with Exclusion
Trial characteristics associated with the exclusion of the vulnerable groups are shown in
Table 3. For all three diseases, exclusion varied significantly according to trial phase, intervention type and disease area, and was most common in phase 2 trials, vaccine trials and in trials targeting infectious diseases. Non-cluster design trials excluded more children with malnutrition and HIV than cluster design trials. Notably, exclusion rates have not fallen in more recent trials.
After adjustment in the logistic regression (
Table 4), several of these associations persisted but with notable shifts. Phase 2 trials remained significantly more likely to exclude children for malnutrition and HIV, although the association attenuated for SCD. The lower exclusion observed in cluster-randomised trials in crude analyses was no longer significant after adjustment, suggesting this pattern was confounded by other trial characteristics. Vaccine trials continued to show higher odds of exclusion—significantly so for HIV—while device trials consistently showed much lower exclusion across all three diseases. Importantly, trials in non-infectious disease areas had substantially lower odds of exclusion even after adjustment, confirming disease area as an independent predictor. Consistent with the crude analysis, exclusion practices did not change over time.
4. Discussion
Key Findings
This analysis reveals a significant exclusion of malnutrition, CLHIV, and SCD from clinical trials conducted in sSA between 2010 and 2023, despite the high burden of these conditions in the region. Among trials that did not primarily target these populations, approximately one-third excluded or probably excluded children with each condition, with markedly higher exclusion rates observed in sensitivity analyses restricted to trials with fully known eligibility criteria.
These findings highlight a persistent gap between clinical research and the real-world disease burden among vulnerable paediatric populations in sSA. Exclusion limits the generalisability of trial findings and reduces the evidence base for prevention and treatment in routine care. The observed exclusion in 96.1% trials of malnourished children, 73.9% of CLHIV, and 68.0% of children with SCD in the sensitivity analysis indicates a missed opportunity to generate data that reflect the diverse health needs of children in the region. More specifically, nearly 60% of trials focused on infectious and parasitic diseases, conditions that disproportionately affect CLHIV, malnourished children, and children with SCD; the exclusion of these groups undermines external validity.
Comparison with Previous Research
Our findings align with prior critiques of LMIC trials, where high-risk children are systematically excluded. Pneumonia RCTs in Africa and Asia commonly omitted those with HIV infection or exposure, severe malnutrition, or chronic illnesses such as sickle cell disease [
20,
21]. Reviews confirm routine exclusion of malnourished children, despite evidence that malnutrition predicts treatment failure in infectious diseases such as pneumonia [
22,
23]. Observational cohorts of excluded populations show divergent outcomes [
24]. SCD is well recognised as conferring elevated infectious risk, yet remains underrepresented in interventional studies [
25,
26].
Potential Explanations for Exclusion
Exclusion may partly result from safety concerns, confounding comorbidities, or regulatory and ethical complexities. However, the frequent use of broad, non-specific exclusion criteria (e.g., "chronic illness," "immunocompromised status," "systemic disease") suggests that common conditions such as malnutrition, HIV and SCD were not deliberately considered during trial planning. This practice runs counter to the WHO guidance for best practices for clinical trials, ICH E8(R1), and ICH E6(R3) [
12,
13,
14], which emphasise the inclusion of diverse populations to ensure the applicability of results to those who will ultimately use the intervention.
Sensitivity Analysis
The sensitivity analysis, restricted to trials with clearly reported eligibility criteria, revealed substantially higher exclusion rates than the primary analysis, particularly for malnutrition, where nearly all affected children (96.1% of trials) were excluded. This increase was partly driven by the smaller denominator in the sensitivity analysis, as only a limited number of trials explicitly reported inclusion of these conditions. This striking divergence between the “definitely excluded” and “possibly excluded” groups suggests that reliance on broad or ambiguously phrased criteria (e.g., “chronic illness” or “immunocompromised status”) may have masked the true extent of exclusion in the primary estimates. The findings underscore the importance of explicit, transparent exclusion criteria in trial protocols and registries. Greater precision in defining eligibility not only improves reproducibility and interpretability but also prevents the inadvertent marginalisation of high-risk populations whose inclusion is critical for generating generalisable evidence.
Emerging Trends in African Clinical Research
Encouragingly, 2010 to 2023 saw a notable increase in the number of clinical trials conducted in Africa(1), reflecting a growing commitment to address the continent's public health challenges. This rise is partly driven by increased global investment in epidemic preparedness, expanded donor funding for neglected tropical diseases, and the growth of local research infrastructure. Regional initiatives and regulatory harmonisation efforts, notably the African Vaccine Regulatory Forum (AVAREF)-established by WHO in 2006, have facilitated multi-country trials and fostered collaboration across borders [
27,
28].
However, despite the increasing number of trials conducted during this period, our findings indicate that the systematic exclusion of high-risk paediatric populations has not improved over time. Specifically, the proportion of trials excluding children with malnutrition, HIV and SCD remained largely stable across the decade with no significant change in exclusion patterns before and after 2015, indicating that the observed growth in trial activity has not been matched by increased inclusivity in trial design.
Dominant Therapeutic Areas in African Trials
The thematic focus of trials in sSA remains heavily skewed toward infectious diseases, particularly malaria, HIV, tuberculosis, and, more recently, emerging viral pathogens such as Ebola and COVID-19. These conditions remain high-priority given their ongoing burden, especially among children. However, non-communicable diseases (NCDs) and chronic conditions, including SCD and malnutrition, receive comparatively limited attention despite their rising prevalence and clinical significance. Our findings corroborate this trend, showing that only a minority of paediatric trials primarily targeted malnutrition or SCD. A more balanced research agenda that includes chronic and comorbid conditions is critical to building a comprehensive evidence base for paediatric care in sSA.
Influence of Trial Design and Intervention Type on Exclusion
Our findings also highlight notable patterns in exclusion based on trial design and type of intervention. The vast majority of included trials were randomised trials (85.0 %), and a small proportion employed cluster randomisation. Randomised controlled trials (RCTs) remain the gold standard for evaluating the efficacy of interventions and are understandably dominant. In our analysis, non-cluster trials appeared more likely than cluster-randomised designs to exclude high-risk paediatric populations in the crude comparisons. However, this association weakened and was no longer significant after adjustment in the logistic regression, suggesting that the initial pattern was partly explained by other trial characteristics such as intervention type or disease area. This implies that while concerns about internal validity and the desire to limit heterogeneity may influence exclusion decisions in individually randomised RCTs, these factors do not operate independently of broader design and contextual features. This may reflect concerns around internal validity and the perceived need to minimise heterogeneity in RCTs, often at the expense of representativeness.
Similarly, the type of intervention was strongly associated with exclusion. Drug trials, the most common intervention category in our sample (61.4%), were more likely to exclude children with HIV, or SCD than trials evaluating dietary supplements or medical devices or mixed-intervention trials (e.g. drug and dietary intervention combined). Vaccine interventions, while less frequent, were associated with higher exclusion odds, possibly due to safety or immunogenicity concerns in immunocompromised or chronically ill children. These findings suggest that both methodological and pharmacological risk perceptions may drive exclusionary decisions, reinforcing the need for trial sponsors and investigators to adopt more inclusive and adaptive designs, particularly when evaluating interventions intended for use in high-burden, heterogeneous paediatric populations.
Gaps in Disease Classification and Registry Data
Another key limitation in the current clinical trials landscape is the inconsistency in disease classification across registries. While we attempted to categorise trials using ICD-11 codes, most clinical trial registries do not require or enforce the use of standardised disease coding such as the ICD. As noted in WHO’s updated guidance on reporting summary results in trial registries, there is substantial variability in the types of information captured across registries, with no harmonised requirement for standardised disease classification [
29]. As a result, identifying trials by therapeutic area often relies on manual review or keyword-based classification, which introduces subjectivity and potential misclassification. The lack of mandatory ICD tagging hampers large-scale analyses across conditions and regions, and limits the ability to systematically track research gaps or overlaps. Harmonising trial registration practices, including the adoption of ICD codes and structured metadata, would greatly enhance the transparency, comparability, and policy relevance of clinical trial data in LMIC settings.
Strengths and Limitations
This review is strengthened by its large sample size, robust duplicate detection, and use of an automated classification algorithm, which was independently reviewed by members of the research team. However, some limitations must be acknowledged. First, exclusion status was inferred from registry-reported eligibility criteria, which may not capture all protocol amendments or nuanced decisions made during trial implementation. Second, some trials lacked clear information on inclusion/exclusion criteria, leading to potential misclassification despite sensitivity analyses. Lastly, our analysis did not include unpublished or unregistered trials, which may differ in their inclusion practices.
Implications for Research and Policy
These findings underscore the urgent need to make paediatric trials in sSA more inclusive. Sponsors, regulators, and ethics committees should advocate for context-specific eligibility criteria and ensure that trials reflect the diversity of the population at risk. Furthermore, funders should prioritise studies that focus on or deliberately include CLHIV, malnourished children, and those with SCD to close existing evidence gaps. Improving representation will not only enhance the relevance of trial findings but also promote equity in access to emerging health technologies. In parallel, trial registries and global platforms should consider the mandatory adoption of structured disease classification systems, such as the WHO-ICD, to facilitate future landscape analyses and guide strategic investment in research.
5. Conclusions
CLHIV, malnutrition, or SCD remain underrepresented in paediatric clinical trials in sSA, despite these conditions being common and with vulnerability to poor outcomes. There is a clear need for more inclusive research practices that align with international guidance and ethical principles, thereby ensuring that the most vulnerable populations are not left behind in the advancement of global child health.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on
Preprints.org.
Author Contributions
Conceptualization, Armel Zemsi, Ed Clarke and Stephen Allen; methodology, Armel Zemsi, Nuredin Mohammed, Isatou Cham and Eric Youm.; validation, Armanda Nangmo, Lorraine Guedem, Mariama Telly Diallo, Yahya Njie .; formal analysis, Isatou Cham and Nuredin Mohammed.; data curation, Armanda Nangmo, Lorraine Guedem, Mariama Telly Diallo, Yahya Njie and Isatou Cham; writing—original draft preparation, Armel Zemsi. And Isatou Cham.; writing—review and editing, Ikechukwu Adigweme, and Stephen Allen.; visualization, Armel Zemsi & Stephen Allen.; supervision, Ed Clarke and Stephen Allen. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors on request.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CLHIV |
Children Living with HIV |
| GCP |
Good Clinical Practice |
| HIV |
Human Immunodeficiency Virus |
| ICH |
International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use |
| ICTRP |
International Clinical Trials Registry Platform |
| ISRCTN |
International Standard Randomised Controlled Trial Number |
| LMICs |
Low and Middle Income Countries |
| PACTR |
Pan African Clinical Trials Registry |
| SCD |
Sickle Cell Disease |
| sSA |
sub-Saharan Africa |
| WHO |
World Health Organization |
References
- Edem, B.; Onwuchekwa, C.; Wariri, O.; Nkereuwem, E.; Nkereuwem, O.O.; Williams, V. Trends in clinical trial registration in sub-Saharan Africa between 2010 and 2020: a cross-sectional review of three clinical trial registries. Trials 2021, 22, 1–11. [CrossRef]
- Pfizer. Pfizer. 2022 [cited 2025 Sep 3]. Pfizer Announces Positive Top-Line Data of Phase 3 Global Maternal Immunization Trial for its Bivalent Respiratory Syncytial Virus (RSV) Vaccine Candidate. Available from: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-announces-positive-top-line-data-phase-3-global.
- Clarke, E.; Bashorun, A.O.; Okoye, M.; Umesi, A.; Hydara, M.B.; Adigweme, I.; Dhere, R.; Sethna, V.; Kampmann, B.; Goldblatt, D.; et al. Safety and immunogenicity of a novel 10-valent pneumococcal conjugate vaccine candidate in adults, toddlers, and infants in The Gambia—Results of a phase 1/2 randomized, double-blinded, controlled trial. Vaccine 2020, 38, 399–410. [CrossRef]
- Adigweme, I.; Yisa, M.; Ooko, M.; Akpalu, E.; Bruce, A.; Donkor, S.; Jarju, L.B.; Danso, B.; Mendy, A.; Jeffries, D.; et al. A measles and rubella vaccine microneedle patch in The Gambia: a phase 1/2, double-blind, double-dummy, randomised, active-controlled, age de-escalation trial. Lancet 2024, 403, 1879–1892. [CrossRef]
- World Health Organization. Malnutrition [Internet]. [cited 2025 Sep 23]. Available from: https://www.who.int/health-topics/malnutrition#tab=tab_1.
- World Health Organization. Levels and trends in child malnutrition: UNICEF/WHO/The World Bank Group joint child malnutrition estimates: key findings of the 2025 edition. World Health Organization. 2025.
- Joint United Nations Programme on HIV/AIDS. AIDS, crisis and power to transform: UNAIDS global AIDS update 2025 [Internet]. LMW; 2024 [cited 2025 Sep 23]. Available from: https://www.unaids.org/sites/default/files/2025-07/2025-global-aids-update-JC3153_en.pdf.
- United Nations Joint Programme on HIV/AIDS (UNAIDS). Fact Sheet—Global AIDS Updatate. 2019; United Nations Joint Programme on HIV/AIDS (UNAIDS): Geneva, Switzerland, 2019. Available online: https://www.unaids.org/sites/default/files/media_asset/UNAIDS_FactSheet_en.pdf (accessed on 20 November 2019).
- Piel, F.B.; I Hay, S.; Gupta, S.; Weatherall, D.J.; Williams, T.N. Global Burden of Sickle Cell Anaemia in Children under Five, 2010–2050: Modelling Based on Demographics, Excess Mortality, and Interventions. PLOS Med. 2013, 10, e1001484. [CrossRef]
- Wonkam, A.; Makani, J. Sickle cell disease in Africa: an urgent need for longitudinal cohort studies. Lancet Glob. Heal. 2019, 7, e1310–e1311. [CrossRef]
- Farooq, F.; Mogayzel, P.J.; Lanzkron, S.; Haywood, C.; Strouse, J.J. Comparison of US Federal and Foundation Funding of Research for Sickle Cell Disease and Cystic Fibrosis and Factors Associated With Research Productivity. JAMA Netw. Open 2020, 3, e201737–e201737. [CrossRef]
- ICH. ICH Guideline E8: General Considerations for Clinical Trials. International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use. 1997;(July).
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). ICH Harmonised Guideline: Integrated Addendum to ICH E6(R3) – Guideline for Good Clinical Practice [Internet]. 2025 [cited 2025 Jun 6]. Available from: https://database.ich.org/sites/default/files/ICH_E6%28R3%29_Step4_FinalGuideline_2025_0106.pdf.
- World Health Organization. Guidance for best practices for clinical trials [Internet]. Geneva: World Health Organization; 2024. Available from: https://www.who.int/publications/i/item/9789240097711.
- Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Revista Espanola de Nutricion Humana y Dietetica. 2016;20(2).
- Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ WV. Handbook. 2019. Cochrane Handbook for Systematic Reviews of Interventions version 6.0 (updated July 2019). Cochrane, 2019.
- Newcombe RG. Two-sided confidence intervals for the single proportion: Comparison of seven methods. Stat Med. 1998;17(8).
- R Core Team. R: A Language and Environment for Statistical Computing_. R Foundation for Statistical Computing. at https://www.r-project.org/ (2024).
- ClinicalTrials.gov. ClinicalTrials.gov Glossary Terms [Internet]. [cited 2025 Sep 25]. Available from: https://clinicaltrials.gov/study-basics/glossary.
- Addo-Yobo, E.; Chisaka, N.; Hassan, M.; Hibberd, P.; Lozano, J.M.; Jeena, P.; MacLeod, W.B.; Maulen, I.; Patel, A.; Qazi, S.; et al. Oral amoxicillin versus injectable penicillin for severe pneumonia in children aged 3 to 59 months: a randomised multicentre equivalency study. Lancet 2004, 364, 1141–1148. [CrossRef]
- Ginsburg, A.-S.; Mvalo, T.; Nkwopara, E.; McCollum, E.D.; Phiri, M.; Schmicker, R.; Hwang, J.; Ndamala, C.B.; Phiri, A.; Lufesi, N.; et al. Amoxicillin for 3 or 5 Days for Chest-Indrawing Pneumonia in Malawian Children. New Engl. J. Med. 2020, 383, 13–23. [CrossRef]
- Lodha, R.; Kabra, S.K.; Pandey, R.M. Antibiotics for community-acquired pneumonia in children. 2013, 2013, CD004874. [CrossRef]
- Mvalo, T.; Smith, A.G.; Eckerle, M.; Hosseinipour, M.C.; Kondowe, D.; Vaidya, D.; Liu, Y.; Corbett, K.; Nansongole, D.; Mtimaukanena, T.A.; et al. Antibiotic treatment failure in children aged 1 to 59 months with World Health Organization-defined severe pneumonia in Malawi: A CPAP IMPACT trial secondary analysis. PLOS ONE 2022, 17, e0278938. [CrossRef]
- Ginsburg AS, May S, Nkwopara E, Ambler G, McCollum ED, Mvalo T, et al. Clinical outcomes of pneumonia and other comorbidities in children aged 2-59 months in Lilongwe, Malawi: Protocol for the prospective observational study “innovative treatments for pneumonia.” JMIR Res Protoc. 2019;8(7).
- Ochocinski, D.; Dalal, M.; Black, L.V.; Carr, S.; Lew, J.; Sullivan, K.; Kissoon, N. Life-Threatening Infectious Complications in Sickle Cell Disease: A Concise Narrative Review. Front. Pediatr. 2020, 8, 38. [CrossRef]
- Bencheva, V.; Mann, N.-K.; Rombey, T.; Pieper, D.; Schmiedl, S. Barriers and facilitators to enrollment in pediatric clinical trials: an overview of systematic reviews. Syst. Rev. 2024, 13, 1–12. [CrossRef]
- World Health Organization Regional Office for Africa. AVAREF [Internet]. [cited 2025 Sep 3]. Available from: https://www.afro.who.int/health-topics/immunization/avaref.
- Akanmori, B.; Mukanga, D.; Bellah, A.; Traore, T.; Ward, M.; Mihigo, R. The Role of The African Vaccine Regulatory Forum (AVAREF) in The Accelerated Clinical Evaluation of Ebola Vaccine Candidates During the Large West Africa Epidemic. J. Immunol. Sci. 2018, 2, 75–79. [CrossRef]
- Chan, A.-W.; Karam, G.; Pymento, J.; Askie, L.M.; da Silva, L.R.; Aymé, S.; Taylor, C.M.; Hooft, L.; Ross, A.L.; Moorthy, V. Reporting summary results in clinical trial registries: updated guidance from WHO. Lancet Glob. Heal. 2025, 13, e759–e768. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |