1. Introduction
Limb salvage surgery (LSS) with reconstruction is nowadays the preferred surgical treatment for bone sarcomas of the lower extremity when free margins can be achieved and the adjacent joints can be preserved. It is often combined with (neo)adjuvant therapies, and offers better functional outcomes, while survival rates maintain comparable to amputation [
1,
2].
Reconstructions of segmental bone defects remain challenging in pediatric patients, especially when the defect exceeds 6 cm [
3]. Moreover, in the pediatric population it is important, when oncologically safe, to preserve the adjacent joint and epiphyseal plate of the affected limb to allow for remaining bone growth. Currently, there is a lack of literature analysing the surgical outcomes of biological reconstructions after intercalary resection in pediatric patients who had a segmental resection due to a lower extremity malignancy, particularly the impact the length of the defect has on the surgical outcomes. Possible biological methods for restoring intercalary bone defects are a massive allograft, a free vascularized fibula graft (FVFG), or the Capanna technique, in which an allograft is combined with an FVFG. An allograft is favourable for its strength and convenience as an “off-the-shelf product”. However, the avascular nature of the allograft is associated with complications such as non-union, graft fracture, and infections. Alternatively, reconstruction with an autologous vascularized fibula has the advantage of an intrinsic blood supply that promotes bone formation. These enhanced vascular and osteogenic capabilities can facilitate primary bone healing, promote bone union, and improve structural integrity of the bone. Drawbacks of using a vascularized fibula graft include the risk of flap failure (failure of the microsurgical anastomosis), donor-site morbidity (including risk of peroneal nerve palsy), and autograft fractures since a fibula shaft alone usually does not provide enough structural stability, when compared to massive allografts, especially in larger children and adolescents [
4]. In clinical practice, local preferences differ regarding when to combine techniques. A commonly applied rule of thumb among surgeons is to use a 10 cm defect length cutoff to add an FVFG to a massive allograft, whereas in smaller defects FVFG or allograft is more often selected as a standalone reconstruction. The Capanna technique combines the advantages of both techniques as it combines a structural allograft with an FVFG, providing the benefits of both stability and biology. This method involves placing a vascularized fibula graft within the cortical bone allograft, possibly leading to lower rates of non-union, fracture, and infection [
3,
5].
Current literature on the Capanna technique after oncological resection is limited in both size and scope. Most studies describe its application in larger defects. However, it remains unclear whether these benefits might also extend to smaller segmental defects, for which FVFG or allograft alone are more commonly used. This systematic review describes outcomes of allografts, FVFG, and the Capanna technique in pediatric and young adults following intercalary resection for lower extremity sarcoma. In this review, we specifically assess differences in non-union rates in relation to defect length, and evaluate union rates, the proportion of complications, and time to full weight-bearing (FWB). These insights may contribute to patient-specific decision-making and to explore whether current defect-size thresholds for selecting reconstructive strategies should be reconsidered. In addition, we explore whether the Capanna technique may provide advantages in shorter defects and questions the eligibility of using a fixed 10 cm threshold to guide surgical decision-making.
2. Materials and Methods
2.1. Literature Search
Prior to the literature search, a review protocol was developed based on the Preferred Reporting Items for Systematic Reviews (PRISMA) guidelines [
6]. A literature search was conducted in PubMed, Scopus, Embase, and the Cochrane Library, up to November 2025. The search strategy, developed in collaboration with the institutional medical library, included the terms and synonymous variations in phrasing and spelling: “Lower extremity” AND “length defect” AND “oncologic” AND (“allograft” OR “vascularized fibula” OR “Capanna”).
2.2. Study Identification and Selection
Studies were included if they reported on the reconstruction of oncologic bone defects in the long bones of the lower extremities using an allograft, an FVFG, or the Capanna technique. Only studies addressing intercalary bone defects with a clearly described defect length were considered eligible. Additional inclusion criteria were a minimum follow-up period of 12 months and a study population exceeding five patients. This review primarily focused on pediatric patients, as most studies reporting intercalary reconstructions after oncologic resection are performed in children. However, studies including young adult patients were not excluded. Eligible study designs included randomized controlled trials, cohort studies, case-control studies, and case series. Articles were included if published in English or translated into English, with no restriction on publication date.
Studies were excluded if they focused on anatomical regions other than the long bones of the lower extremities or employed other reconstructive methods such as arthrodesis, autograft, or prosthetic replacement. Further exclusions included pilot or feasibility studies, animal studies, conference abstracts, and studies lacking explicit reporting of defect length. The full inclusion and exclusion criteria are summarized in
Table 1.
2.3. Screening
All studies were imported into the Covidence platform for screening [
7], with duplicates automatically removed. Two independent researchers (W.K., L.S.) screened titles, abstracts, and full texts of all articles according to predefined eligibility criteria. Reasons for exclusion were documented. Conflicting interpretations were resolved by consulting a third reviewer (D.K.) to reach a consensus.
2.4. Data Extraction
Data were extracted on author, year, patient demographics (age and sex), tumor histology, reconstruction site (femur or tibia), follow-up duration, and outcomes including defect length, time to FWB, time to union, and rates of non-union, fracture, and infection. All outcomes were predefined and extracted in Microsoft Excel (Microsoft Corporation, Redmond, WA, USA, 2023) using an extraction format developed by the review team. Defect length was defined as the segment of bone resected due to tumor involvement. Functional outcomes were evaluated using the Musculoskeletal Tumor Society (MSTS) score, with ranges from 0 (poor function) to 30 (optimal limb function) [
8].
For studies involving both lower extremity and non-lower extremity reconstructions, only femoral and tibial data were extracted and analyzed.
2.5. Quality Assessment
Study quality was assessed using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Case Series [
9], which evaluates risk of bias related to confounding, selection, information bias, and reporting quality. Bias assessments were performed in Microsoft Excel (Microsoft Corporation, Redmond, WA, USA, 2023).
3. Results
3.1. Selection and Inclusion of Studies
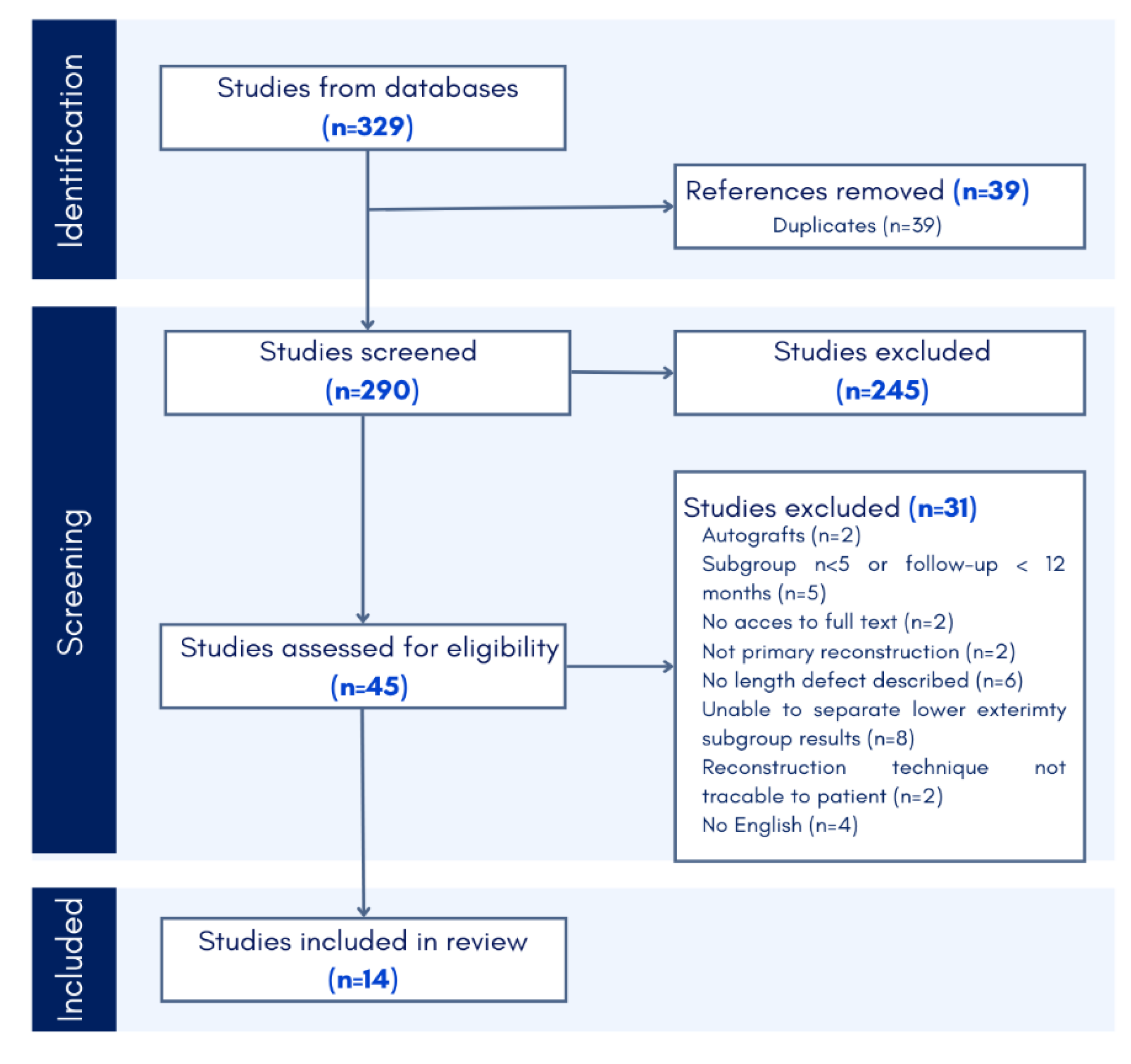
In total, 329 articles were identified through the initial search across PubMed, Scopus, Embase, and the Cochrane Library. After removal of duplicates, title and abstract screening, and full text screening, 14 articles were deemed suitable for descriptive analysis (
Figure 1).
3.2. Study Characteristics and Description of Participants
The final selection included 13 case series and one cohort study, published between July 2005 and July 2024. Of these, two were focused on allograft alone, two on FVFG alone, and ten on the Capanna technique. The total sample size comprised: 17 patients in the allograft group, 16 patients in the FVFG group, and 148 patients in the Capanna group. Mean patient ages were 12.5, 9.6, and 13.0 years, respectively, with female representation of 58.8%, 62.5%, and 41.2%.
Reconstructions were performed in the femur or tibia region, with two studies focusing exclusively on tibial reconstructions [
10,
11]. Osteosarcoma (OS, 48%) and Ewing sarcoma (ES, 35%) were the most common diagnoses, followed by adamantinoma and chondrosarcoma. Mean follow-up was 70.9 months (range: 12 – 288 months). Functional outcomes were most often assessed using the MSTS score, which was analyzed when reported. Full study characteristics are summarized in
Table 2 and
Table 3.
3.3. Operative Features and Functional Outcomes
3.3.1. Defect Length
The allograft group showed a mean defect length of 15.5 cm, with a tibial average defect length of 14 cm [
10], and femoral defect lengths were not further specified. In the FVFG group, the overall mean defect length was 11.4 cm, with tibial and femoral defect lengths of 11.6 cm and 11.9 cm, respectively. In the Capanna group, the overall mean defect length was 15.0 cm, with tibial and femoral defect lengths of 16.3 cm and 18.3 cm, respectively.
3.3.2. Graft Union and Full Weight Bearing (MSTS)
Graft union times varied widely depending on the reconstruction technique.
In the allograft group, the mean union time was 15.5 months. Misaghi et al. [
10] reported 11 ± 2 months, and Houdek et al. [
4] reported 17 months (range: 5 – 20 months), both defining union as bridging callus in three of the four cortices. The FWB for this group was not reported, and the mean MSTS score was 24.
In the FVFG group, the mean union time was 11.4 months. Ruiz-Moya et al. [
12] observed the shortest union time of 4.8 months (range: 4 – 8 months), while Adam et al. [
13] reported union time of 16.3 months (range: 2 – 55 months). FWB was achieved after 12 months, and the mean MSTS score was reported in a single study and was 23.
In the Capanna group, the mean union time was 11.6 months. Xu et al. [
14] reported union times from 5 ± 1.6 months, and Adam et al. [
13] union times of 23.7 months (range: 14 – 36 months). FWB was achieved after a mean time of 10.1 months. Adam et al. [
13] reported FWB after 7.3 months (range: 4 – 19 months), and Ruiz-Moya et al. [
12] up to 15 months (range: 9 – 33 months). The mean MSTS score was 26.8. Eight studies reported MSTS scores ≥ 26; Halim et al. [
15] reported an MSTS score of 24.
3.4. Surgical Complications and Postoperative Sequelae
3.4.1. Graft Fracture, and Non-Union.
In the allograft group, the mean fracture rate was 35.3%. Houdek et al. [
4] reported a rate of 45% (n=5), with three cases requiring revision surgery, whereas Misaghi et al. [
10] reported a much lower rate of 6% (n=1). Non-union data in this group were not consistently reported, but delayed union was observed in 36% of patients in the allograft group.
In the FVFG group, the mean fracture rate was 56.3%. Adam et al. [
13] observed a 50% rate (n=5), with one patient requiring operative fixation. Ruiz-Moya et al. [
12] reported a higher fracture rate of 67% (n=4), with three cases requiring operative fixation. Non-union rates were 30% (Adam et al. [
13]) and 17% (Ruiz-Moya et al. [
12]).
In the Capanna group, the mean fracture rate was 23.6% (35/148 patients), with reported rates ranging from 0% to 67%. Management varied and included casting (n=5), open reduction and internal fixation (ORIF) (n=12), plate fixation (n=1), construct removal (n=3); in 12 cases, management was not specified. Several studies, including those by Li et al. [
16], Lu et al. [
17], and Opyrcal et al. [
11], reported no fractures. Moran et al. [
18] documented two late fractures (> 2 years postoperative). Rabitsch et al. [
19] described two trauma-related fractures treated with ORIF and bone grafting. Although delayed and non-union rates were sparsely reported, most studies noted successful primary union. Where reported, non-union rates ranged from 0% (Adam et al. [
13]) to 33% (Rabitsch et al. [
19]), with a mean rate of 11.0%. The majority of series reported values below 20%.
3.4.2. Disease Recurrence and Limb Salvage
In the allograft group, eight secondary amputations were reported, three due to deep infection and five due to recurrence. Most patients received chemotherapy. In the FVFG group, one secondary amputation occurred due to local recurrence. In the Capanna group, seven secondary amputations were reported, all attributed to local recurrence. Full surgical complications and postoperative sequelae are summarized in
Table 4.
3.5. Quality Assessment
The risk of bias assessment, based on the JBI Checklist for Case Series, is summarized in Supplementary 1. Four studies had a low risk of bias [
4,
14,
17,
20], while the remaining eight showed moderate risk [
10,
11,
12,
13,
15,
16,
18,
19,
21].
4. Discussion
This systematic review evaluates three biological reconstruction techniques after intercalary resection in pediatric and young adult patients: allograft, FVFG, and the Capanna technique. By stratifying outcomes according to defect length, this review explored whether graft choice should be guided by defect size and whether the Capanna technique may also be justified in shorter defects.
Across the included studies, FVFG and Capanna reconstructions achieved the shortest mean union times (11.4 and 11.6 months), while allograft reconstructions showed a longer time to union (15.5 months) and more delayed unions. These findings are consistent with the avascular nature of allografts and their limited biological integration [
22,
23]. Although FVFG and Capanna showed similar union times, Capanna reconstructions were associated with a lower non-union rate (11.0% vs 25% for FVFG) and fewer reoperations. Functional outcomes reflected these differences: MSTS scores were highest for Capanna (≥26) and lower for FVFG and allograft (approximately 23 and 24). Importantly, patients reconstructed with the Capanna technique achieved FWB earlier (mean 10 months) compared with FVFG (mean 12 months), suggesting that mechanical stability, rather than radiographic union alone, determines safe mobilization [
12,
13].
Complication profiles differed across techniques; FVFG was strongly associated with fractures (56%), reflecting the limited initial load-bearing capacity of the fibula despite its osteogenic potential [
13]. Allografts carried the highest complication rate overall, with frequent delayed unions, fractures, and infections. By contrast, Capanna reconstructions demonstrated lower fracture and infection rates, consistent with previous reports by Bakri et al. and Feltri et al. [
24,
27] and with long-term results described by Dolen et al. [
28] The synergistic combination of mechanical stability from the allograft and biological activity from the fibula likely underpins these advantages. These findings support the Capanna technique as the most reliable option, particularly in load-bearing bones where mechanical insufficiency is a major concern.
Traditionally, the Capanna reconstruction is reserved for large segmental defects (>10 cm). However, several included cases reported successful outcomes in defects <10 cm, with lower complication rates compared to FVFG or allograft alone. Although promising, these data remain very limited (n=6, only two <8 cm). No firm conclusion can therefore be drawn. Nevertheless, the trend suggests that combined mechanical and biological reconstruction may be beneficial even in smaller gaps when risk factors such as chemotherapy, poor host bone quality, or high mechanical load are present. Current evidence does not justify the routine use of the Capanna technique in small defects, but it may be considered selectively in high-risk patients.
Systemic treatments such as chemotherapy and radiotherapy further affect graft performance by impairing osteogenesis, angiogenesis, and immune defence [
30,
31]. These effects may disproportionally compromise avascular allografts. Some studies in this review suggested shorter union times in patients not receiving chemotherapy [
14,
16], but reporting was inconsistent. However, the biological plausibility of systemic treatment impairing bone healing is well established. Surgeons should therefore consider systemic risk factors when selecting a reconstruction strategy, as biologically active techniques may be more resilient under these conditions.
4.1. Strengths, Limitations, and Reporting Gaps
This review has several methodological strengths. It is, to our knowledge, the first to systematically compare allograft, FVFG, and Capanna reconstructions in relation to defect length in pediatric and young adult patients. The focus on biological techniques, strict inclusion of studies reporting defect size, and adherence to the PRISMA guidelines enhance robustness and clinical relevance.
Several limitations, however, must be acknowledged. Study heterogeneity was considerable, particularly in the reporting of union time, functional scores, and complications. Follow-up periods were often limited, potentially underestimating late complications such as graft failure. Details on reoperations, adjuvant therapy, and complication severity were inconsistently reported, introducing reporting bias. The small overall sample size, especially for patients with shorter defects, precludes definitive conclusions. Consequently, while the trends observed are consistent and clinically relevant, the strength of evidence remains limited.
4.2. Clinical Implications
The findings of this review reaffirm the Capanna techniques as a preferred strategy for large segmental reconstructions, particularly in load-bearing bones of the lower extremity. Its combination of biological and mechanical advantages enables faster and more reliable union, earlier weight-bearing, and lower complication rates than either FVFG or allograft alone.
Allograft reconstructions remain associated with the highest complication burden and should be used cautiously [
12,
29]. FVFG provides biological potential but suffers from mechanical vulnerability, leading to frequent fractures and delayed mobilisation [
13,
25]. Capanna reconstruction mitigates these limitations and appears most favourable when both union and mechanical integrity are critical. Although this review focused on pediatric and young adult populations, previous studies in adult cohorts have reported higher rates of complications and non-union following biological reconstruction, particularly when using allografts alone [
22,
24]. The lower rates observed in this review may therefore reflect the enhanced bone regenerative capacity of younger patients, which is related to greater vascularity and osteogenic potential. However, direct comparative analyses between pediatric and adult populations are scarce and would provide valuable insight into the role of skeletal maturity in graft incorporation and complication risk.
For smaller defects, evidence is currently insufficient to recommend Capanna as a standard approach [
12]. Nonetheless, selective use may be justified in patients with additional risk factors, where improved healing potential and earlier mobilisation could outweigh surgical complexity. In such cases, the decision should be tailored to the individual patient, taking into account defect size, systemic therapy, host bone quality, and anticipated mechanical demands.
Practical considerations must also inform surgical choice. The Capanna technique entails donor-site morbidity, longer operative time, and requires microsurgical expertise, which may not be available in all centres. Complications such as ankle instability, donor-site pain, and growth disturbances have been reported in younger patients [
33]. Cost-effectiveness and resource availability should therefore also guide decision-making.
4.3. Recommendations and Future Directions
Further research is needed to refine patient selection for biological reconstructions. Large, multicentre prospective studies are essential to overcome the small sample size and reporting heterogeneity that currently limit the evidence base. Standardised documentation of outcomes, including union times, complications, severity, reoperation rates, and functional scores, would improve comparability across studies. Moreover, systematic reporting of adjuvant therapies such as chemotherapy and radiotherapy is critical, given their established influence on bone healing and graft integration.
In addition, defect length should be reported not only in absolute terms (cm) but also relative to the overall bone length, as this provides greater clinical relevance in pediatric patients, where the same absolute defect can represent a markedly different anatomical and functional burden. Sanders et al. have shown that relative defect length is a meaningful predictor of long-term outcomes, and adopting this approach should improve the accuracy of treatment planning [
34].
Future studies should also aim to define clinical thresholds at which the addition of an FVFG to an allograft becomes beneficial, whether this lies at 8, 10, or 12 cm. Alongside structural outcomes, comparative analyses should evaluate cost-effectiveness and donor-site morbidity, particularly in the pediatric population, where complications such as ankle instability, donor-site pain, and growth disturbance are concerns [
33]. These considerations are crucial to determine whether the biological and mechanical benefits of the Capanna technique justify its greater surgical complexity and resource requirements.
5. Conclusions
This review demonstrates that the Capanna technique is the most reliable reconstructive option for large segmental bone defects in pediatric and young adult patients, combining the lowest non-union rates, favourable union times, the highest functional outcomes, and lower complication rates. Limited but consistent evidence suggests potential benefits in selected smaller defects, though current data are insufficient to recommend routine use. Until stronger evidence emerges, reconstruction choice should be based on patient-specific factors rather than defect length alone. Future multicenter studies with standardized reporting are essential to establish clear thresholds and optimize patient-tailored reconstruction strategies.
Supplementary Materials
The following supporting information can be downloaded at the website of this paper posted on Preprints.org, Table S1 providing the risk of bias assessment using the JBI Critical Appraisal Checklist for Case Series.
Author Contributions
Conceptualization, W.K., L.H.M.v.S., L.v.d.H., and D.K.; methodology, W.K. and L.H.M.v.S.; software, W.K. and L.H.M.v.S.; validation, W.K, L.H.M.v.S., H.C.N., M.A.J.v.d.S., L.v.d.H., and D.K.; formal analysis, W.K., L.H.M.v.S. and D.K.; investigation, W.K. and L.H.M.v.S.; resources, W.K, L.H.M.v.S., M.A.J.v.d.S., and L.v.d.H.; data curation, W.K, L.H.M.v.S., H.C.N., M.A.J.v.d.S., L.v.d.H., and D.K.; writing—original draft preparation, W.K and L.H.M.v.S.; writing—review and editing, W.K, L.H.M.v.S., H.C.N., M.A.J.v.d.S., L.v.d.H., and D.K.; visualization, W.K. and L.H.M.v.S.; supervision, H.C.N., M.A.J.v.d.S., L.v.d.H., and D.K.; project administration, W.K and L.H.M.v.S.; funding acquisition, M.A.J.v.d.S., L.v.d.H., and D.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
This article is a systematic review and does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. Some of the data may not be publicly available due to confidentially and in accordance with the Dutch Personal Data Protection Act.
Acknowledgments
The authors thank the Utrecht University Library for assistance in identifying and accessing key literature that supported the preparation of this review.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| FVFG |
Free vascularized fibular graft |
| LSS |
Limb salvage surgery |
| FWB |
Full weight-bearing |
| PRISMA |
Preferred reporting items for systematic reviews |
| JBI |
The Joanna Briggs Institute |
| ES |
Ewing’s sarcoma |
| OS |
Osteosarcoma |
| MSTS |
Musculoskeletal Tumor Society |
| ORIF |
Open reduction and internal fixation |
References
- Robert RS, Ottaviani G, Huh WW, Palla S, Jaffe N. Psychosocial and functional outcomes in long-term survivors of osteosarcoma: a comparison of limb-salvage surgery and amputation. Pediatr Blood Cancer. 2010;54(7):990–9.
- Evans DR, Lazarides AL, Visgauss JD, Somarelli JA, Blazer DG 3rd, Brigman BE, et al. Limb salvage versus amputation in patients with osteosarcoma of the extremities: an update in the modern era using the National Cancer Database. BMC Cancer. 2020;20(1):995.
- Houben RH, Rots M, van den Heuvel SCM, Winters HAH. Combined Massive Allograft and Intramedullary Vascularized Fibula as the Primary Reconstruction Method for Segmental Bone Loss in the Lower Extremity: A Systematic Review and Meta-Analysis. JBJS Rev. 2019;7(8):e2.
- Houdek MT, Rose PS, Milbrandt TA, Stans AA, Moran SL, Sim FH. Comparison of Pediatric Intercalary Allograft Reconstructions with and without a Free Vascularized Fibula. Plast Reconstr Surg. 2018;142(4):1065–71.
- McCullough MC, Arkader A, Ariani R, Lightdale-Miric N, Tolo V, Stevanovic M. Surgical Outcomes, Complications, and Long-Term Functionality for Free Vascularized Fibula Grafts in the Pediatric Population: A 17-Year Experience and Systematic Review of the Literature. J Reconstr Microsurg. 2020;36(5):386–96.
- Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). Available from: https://www.prisma-statement.org.
- Covidence systematic review software. Melbourne (Australia): Veritas Health Innovation; 2024. Available from: https://www.covidence.org.
- Enneking WF, Dunham W, Gebhardt MC, Malawar M, Pritchard DJ. A system for the functional evaluation of reconstructive procedures after surgical treatment of tumors of the musculoskeletal system. Clin Orthop Relat Res. 1993(286):241–6.
- Munn Z BT, Barker TH, Moola S, Tufanaru C, Stern C, McArthur A, et al. Methodological quality of case series studies: an introduction to the JBI critical appraisal tool. JBI Evid Synth. 2020;18(10):2127–2133.
- Misaghi A, Jackson TJ, Stans AA, Shaughnessy WJ, Rose PS, Moran SL, Houdek MT. Intercalary Allograft Reconstruction of the Proximal Tibia With and Without a Free Fibula Flap in Pediatric Patients. J Pediatr Orthop. 2020;40(9):e833–e8.
- Opyrchał J, Bula D, Dowgierd K, Pachuta B, Krakowczyk D, Raciborska A, et al. Case Series: Fibula Free Flap with Bone Allograft as the Gold Standard in Lower Limb-Salvage Surgery for Adolescent Patients with Primary Bone Tumors Located within Tibial Diaphysis: Technical Modifications and Short-Term Follow-Up. J Clin Med. 2024;13(14).
- Ruiz-Moya A, Lagares-Borrego A, Sicilia-Castro D, Barrera-Pulido FJ, Gallo-Ayala JM, Santos-Rodas A, et al. Pediatric extremity bone sarcoma reconstruction with the vascularized fibula flap: Observational study assessing long-term functional outcomes, complications, and survival. J Plast Reconstr Aesthetic Surg. 2019;72(12):1887–99.
- Adam D, Hamel A, Perrot P, Duteille F. Long-term behavior of the vascularized fibular free flap for reconstruction of bony defects in children. Ann Chir Plast Esthet. 2020;65(3):219–27.
- Xu L, Wen L, Qiao J, Zhu Z, Qiu Y, Xiong J, et al. Clinical Outcome of Free Vascularized Fibula Graft in the Surgical Treatment of Extremity Osteosarcoma. Orthop Surg. 2020;12(3):727–33.
- Halim AS, Chai SC, Wan Ismail WF, Wan Azman WS, Mat Saad AZ, Wan Z. Long-term outcome of free fibula osteocutaneous flap and massive allograft in the reconstruction of long bone defect. J Plast Reconstr Aesthetic Surg. 2015;68(12):1755–62.
- Li J, Wang Z, Guo Z, Chen GJ, Fu J, Pei GX. The use of allograft shell with intramedullary vascularized fibula graft for intercalary reconstruction after diaphyseal resection for lower extremity bony malignancy. J Surg Oncol. 2010;102(5):368–74.
- Lu X, Li J, Zhang Y, Zhang Y, Liu Y, Wen J, et al. Reconstruction strategy in limb salvage surgery for primary malignant tumor of femoral shaft. Chin J Orthop. 2020;40(18):1282–90.
- Moran SL, Shin AY, Bishop AT. The use of massive bone allograft with intramedullary free fibular flap for limb salvage in a pediatric and adolescent population. Plast Reconstr Surg. 2006;118(2):413–9.
- Rabitsch K, Maurer-Ertl W, Pirker-Fruhauf U, Wibmer C, Leithner A. Intercalary reconstructions with vascularised fibula and allograft after tumour resection in the lower limb. Sarcoma. 2013;2013:160295.
- Luca D, Sara T, Marco I, Andrea CD. The use of vascularized fibula flap with allograft in post-oncologic microsurgical bone reconstruction of lower limbs in pediatric patients. Microsurgery. 2024;44(4):e31172.
- Weichman KE, Dec W, Morris CD, Mehrara BJ, Disa JJ. Lower extremity osseous oncologic reconstruction with composite microsurgical free fibula inside massive bony allograft. Plast Reconstr Surg. 2015;136(2):396–403.
- Othman S, Bricker JT, Azoury SC, Elfanagely O, Weber KL, Kovach SJ. Allograft Alone vs. Allograft with Intramedullary Vascularized Fibular Graft for Lower Extremity Bone Cancer: A Systematic Review and Meta-Analysis. J Plast Reconstr Aesthetic Surg. 2020;73(7):1221–31.
- Jover Carbonell P, Zarzuela Sánchez VM, Marín Bertolín S, Martínez Bovaira G, Castillo Ruipérez L, Ferrando LH. Biological reconstruction of large bone defects with vascularized fibular autograft. Rev Asoc Argent Ortop Traumatol. 2026;88(6):620–9.
- Feltri P, Solaro L, Di Martino A, Candrian C, Errani C, Filardo G. Union, complication, reintervention and failure rates of surgical techniques for large diaphyseal defects: a systematic review and meta-analysis. Sci Rep. 2022;12(1):9098.
- Shi LL, Garg R, Jawa A, Wang Q, Chai Y, Zeng B, Jupiter JB. Bony Hypertrophy in Vascularized Fibular Grafts. Hand (N Y). 2022;17(1):106–13.
- Liu Q, He H, Duan Z, Zeng H, Yuan Y, Wang Z, Luo W. Intercalary Allograft to Reconstruct Large-Segment Diaphysis Defects After Resection of Lower Extremity Malignant Bone Tumor. Cancer Manag Res. 2020;12:4299–308.
- Bakri K, Stans AA, Mardini S, Moran SL. Combined massive allograft and intramedullary vascularized fibula transfer: the capanna technique for lower-limb reconstruction. Semin Plast Surg. 2008;22(3):234–41.
- Dolen UC, Moran SL. Long Term Functional Outcomes of the Capanna Technique for Pediatric Limb Salvage. Plast Reconstr Surg. 2012;130(5S-1):90–1.
- Campanacci DA, Scanferla R, Marsico M, Scolari F, Scoccianti G, Beltrami G, et al. Intercalary Resection of the Tibia for Primary Bone Tumors: Are Vascularized Fibula Autografts with or Without Allografts a Durable Reconstruction? Clin Orthop Relat Res. 2024;482(6):960–75.
- Granberg HA, de Paulo Martins Coelho V Jr, Palmer JD, Grossbach A, Khalsa SS, Viljoen S, et al. The effect of radiotherapy, chemotherapy, and immunotherapy on fusion rate in spinal surgery using osteobiologics for patients with metastatic spinal disease: a systematic review. Neurosurg Rev. 2024;47(1):796.
- Wisanuyotin T, Paholpak P, Sirichativapee W, Kosuwon W. Allograft versus autograft for reconstruction after resection of primary bone tumors: a comparative study of long-term clinical outcomes and risk factors for failure of reconstruction. Sci Rep. 2022;12(1):14346.
- Arif F, Mirza A, Yasmeen S, Rahman MF, Shaikh SA. Vascularized Free Fibula Flap for Limb Salvage After Long Bone Tumor Resection in Pediatric Patients: A Single-Center Seven-Year Experience From a Developing Country. Cureus. 2025;17(3):e80187.
- Goldin AN, Johnson SR, Hajdu KS, Kowalski BL, Volkmar AJ, Moran CP, et al. Surgical Fixation Method in Lower Extremity Intercalary Allograft Reconstruction After Oncologic Resection: A Comparison of Plates and Nails. J Am Acad Orthop Surg. 2023;31(16):881–92.
- Sanders PTJ, Spierings JF, Albergo JI, Bus MPA, Fiocco M, Farfalli GL, et al. Long-Term Clinical Outcomes of Intercalary Allograft Reconstruction for Lower-Extremity Bone Tumors. J Bone Jt Surg. 2020;102(12):1042–9.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).