Submitted:
07 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results and Discussion
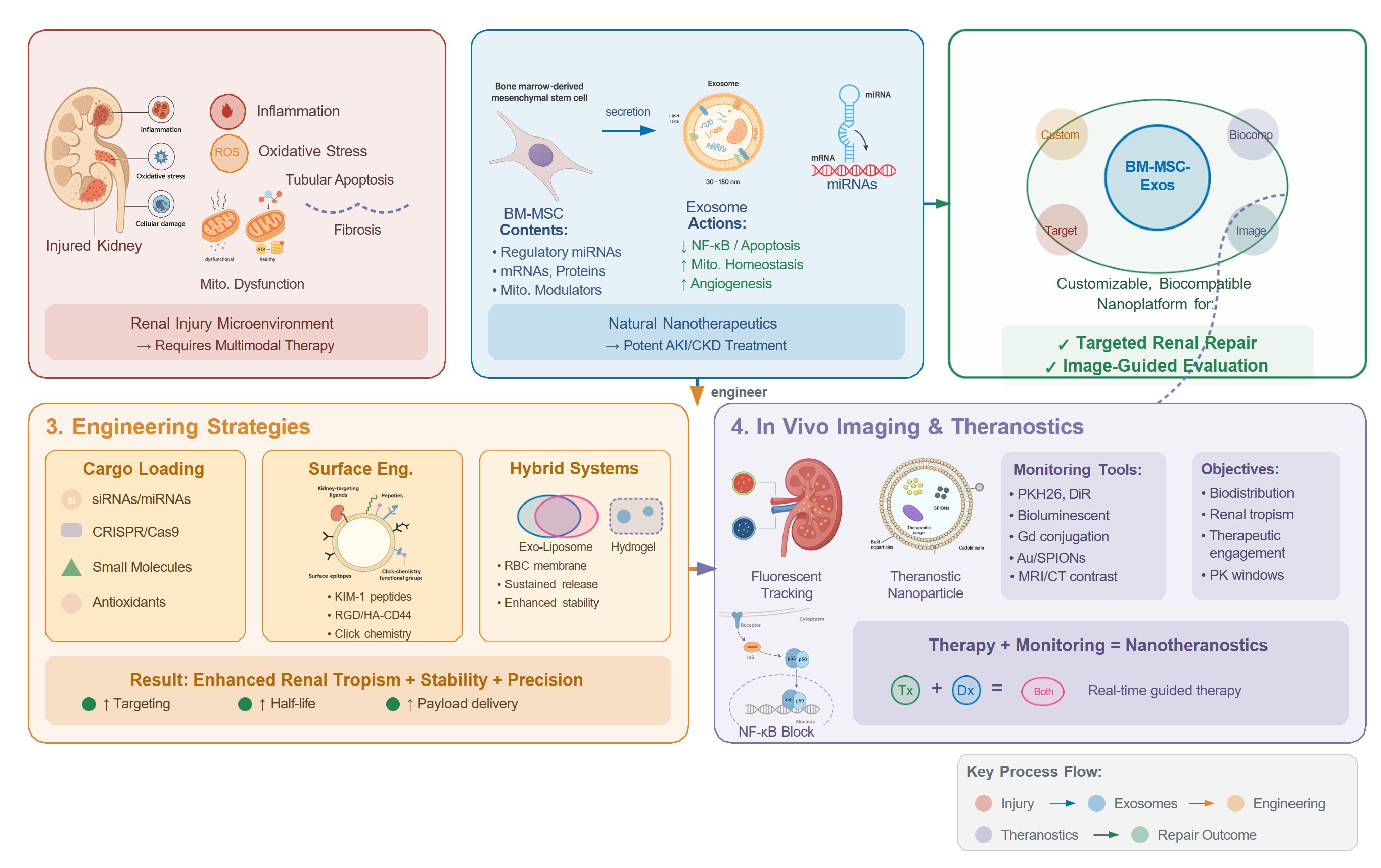
3.1. Bone Marrow Mesenchymal Stem Cell–Derived Exosomes (BM-MSC-Exos)
3.1.1. General Properties and Advantages
3.1.2. BM-MSC-Exos: Acute Kidney Injury (AKI)
3.1.3. BM-MSC-Exos: Chronic Kidney Disease (CKD)
3.1.4. Exosomes as Biosensors and Biomarkers in Kidney Disease
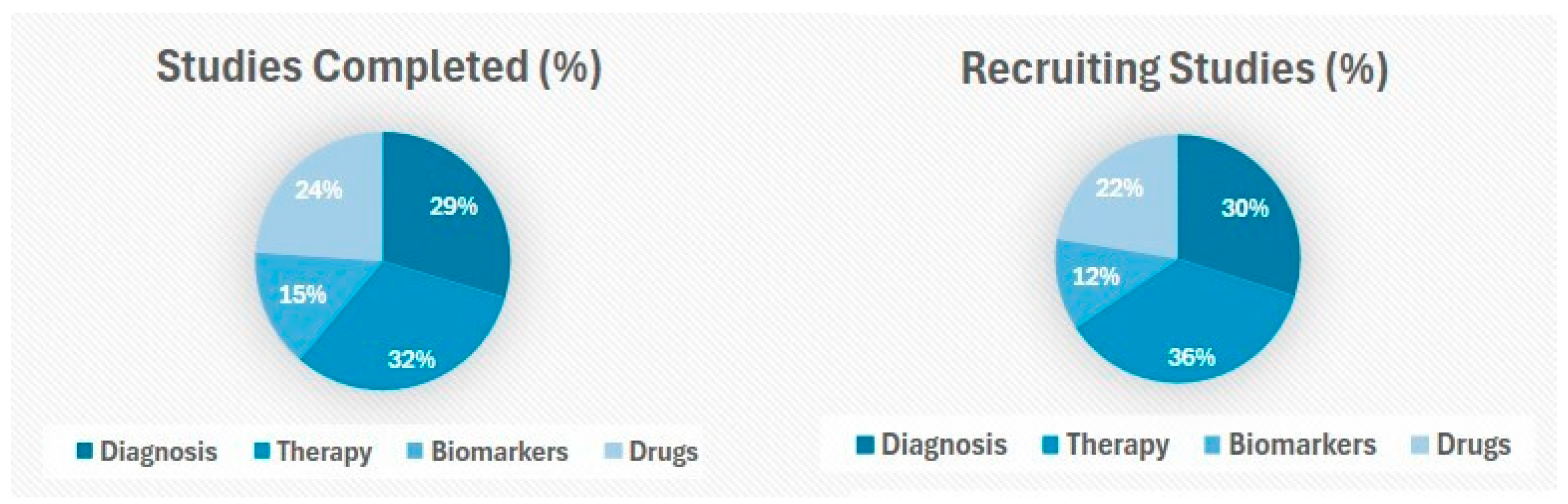
3.1.5. Exosomes: Clinical Trials
- 1. NCT03503461 (France): Urinary exosomal NCC and pendrin evaluated as biomarkers of post-transplant hypertension in calcineurin inhibitor–treated kidney transplant recipients.
- 2. NCT05695573 (Egypt): UMOD gene and uromodulin protein in urinary exosomes assessed as early biomarkers for type 2 diabetes–related nephropathy.
- 3. NCT04142138 (USA): Proteomic profiling of urinary exosomes in individuals with prehypertension following DASH diet intervention.
- 4. NCT04536688 (USA): RGLS4326 therapy in ADPKD patients monitored through urinary exosomal PC- 1 and PC-2 profiles.
- 5. NCT03227055 (Taiwan): Urinary exosomal miRNAs as early cardiovascular–renal risk markers in pediatric CKD (G1–G4).
- 6. NCT03202212 (Italy): Changes in plasma EVs-RNA content and inflammatory markers in dialysis patients undergoing mixed hemodiafiltration.
- 7. NCT03034265 (Switzerland): Urinary sodium, channel exosomal profiles, and plasma angiotensin peptides assessed in resistant hypertension.
- 8. NCT02226055 (Sri Lanka): Urinary exosomes, proteomics, genotoxic markers, and vascular stiffness evaluated in CKDu pathogenesis.
- 9. NCT02822131 (Switzerland): Urinary exosomal transporter proteins (NCC, NaPi, IIa) assessed after controlled phosphate intake.
- 10. NCT06123871 (China): Urinary exosomal miR-136-5p investigated as an early biomarker of diabetic nephropathy.
-
11. NCT06097351 (China): Diagnostic potential of urinary exosomal miR-142-3p in diabetic kidney disease.12. NCT04894695 (China): Identification of urinary exosomal biomarkers in lupus nephritis.
- 13. NCT04534647 (Latin America): Validation of exosomal signatures for lupus nephritis activity and progression.
- 14. NCT03487861 (South Korea): Exosomal transglutaminase 2 (TG2) evaluated as a biomarker of chronic allograft fibrosis after kidney transplantation.
3.2. Engineering BM-MSC and Their Exosomes for Kidney Diseases
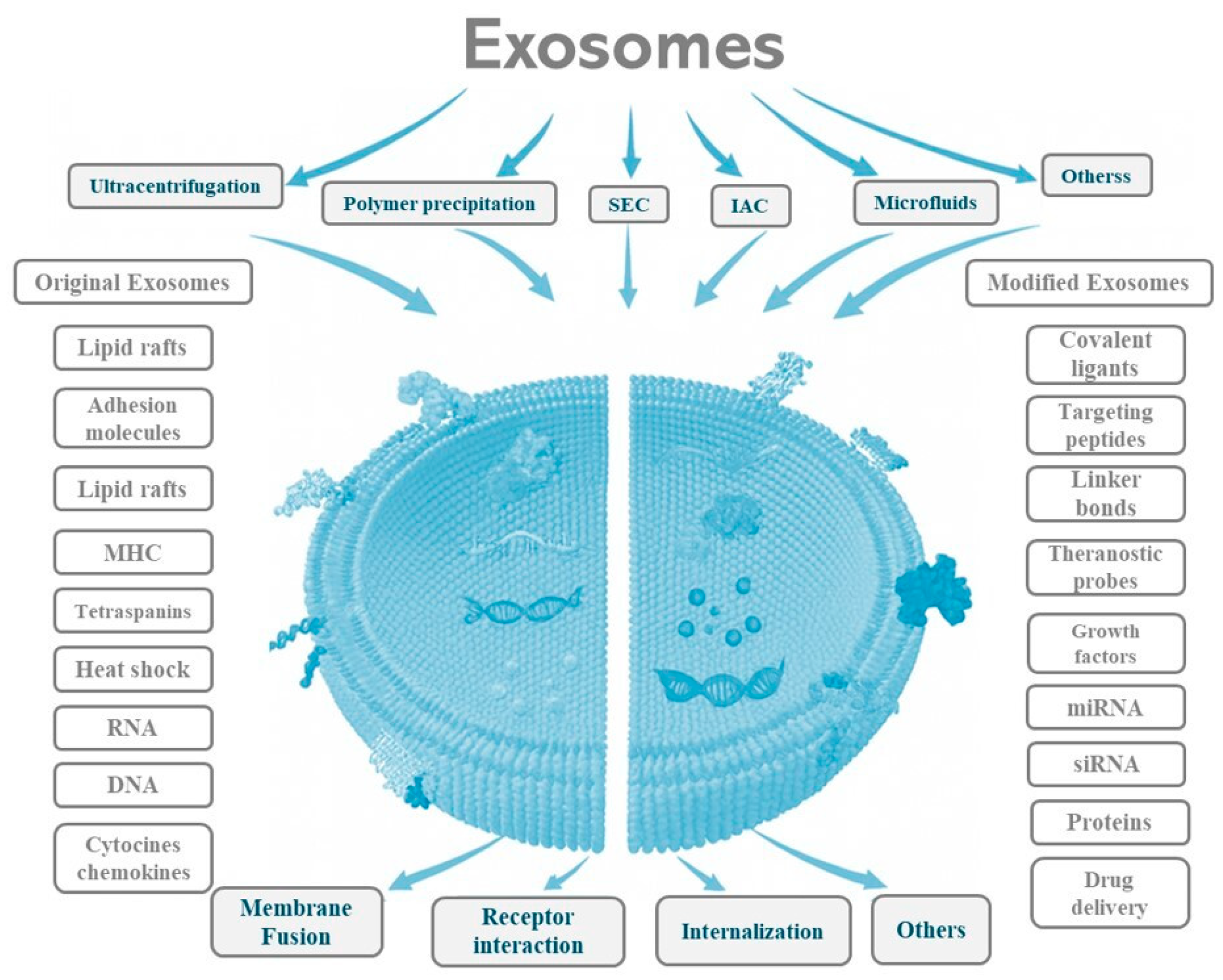
3.2.1. Surface Engineering: Membrane Modification
Non-Covalent Engineering
Covalent Surface Engineering: Click Chemistry
Protein-Based Membrane Engineering
3.2.2. Cargo EngineeringEndogenous Engineering (Genetic /Cellular Reprogramming)
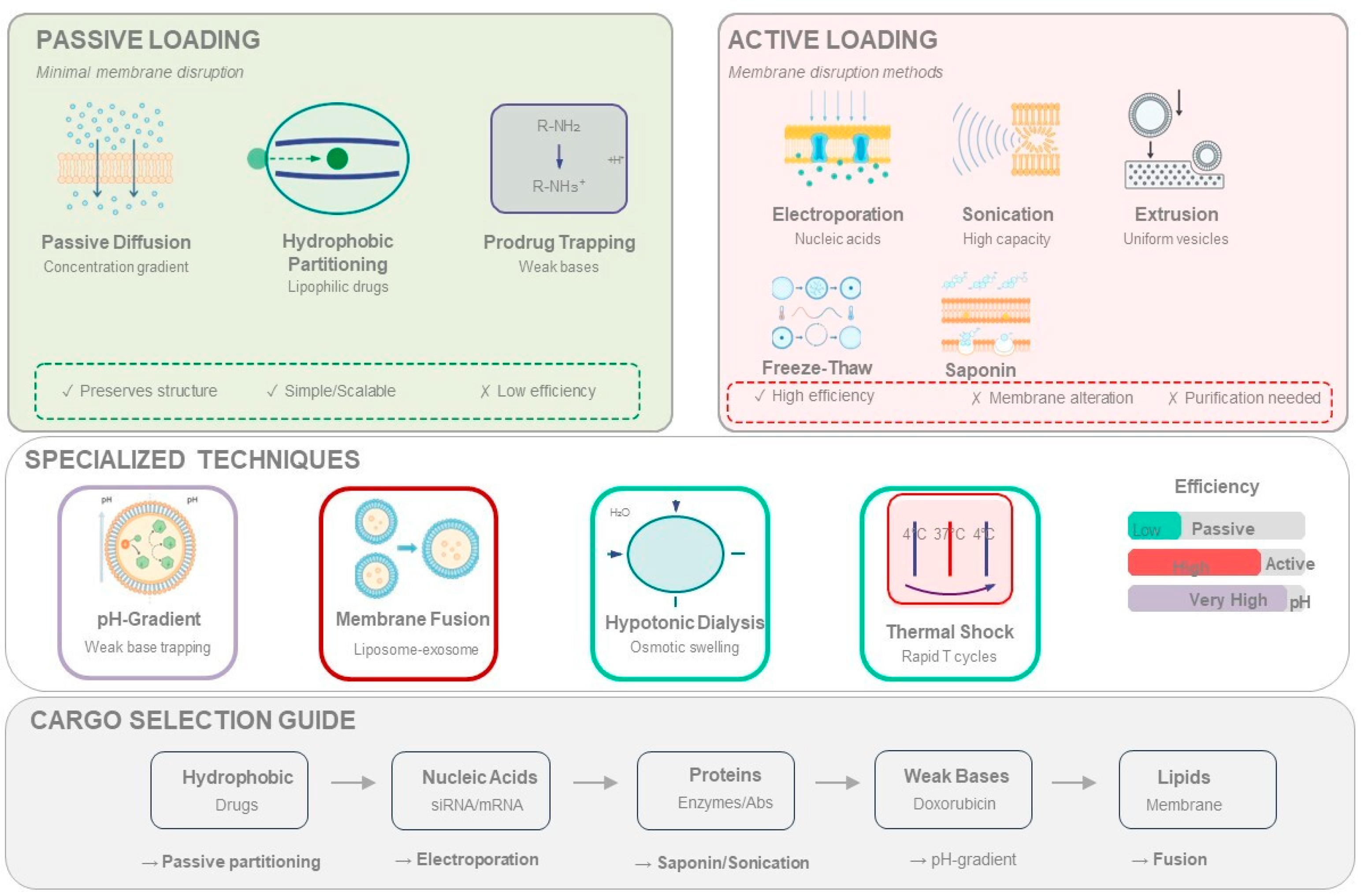
Exogenous Cargo Loading
Hybrid Systems, Biomimetic and Artificial EVs Systems (EVs-Mimetics)
3.3. Safety and Regulatory Considerations
3.4. Tracking and Imaging of Engineered Exosomes
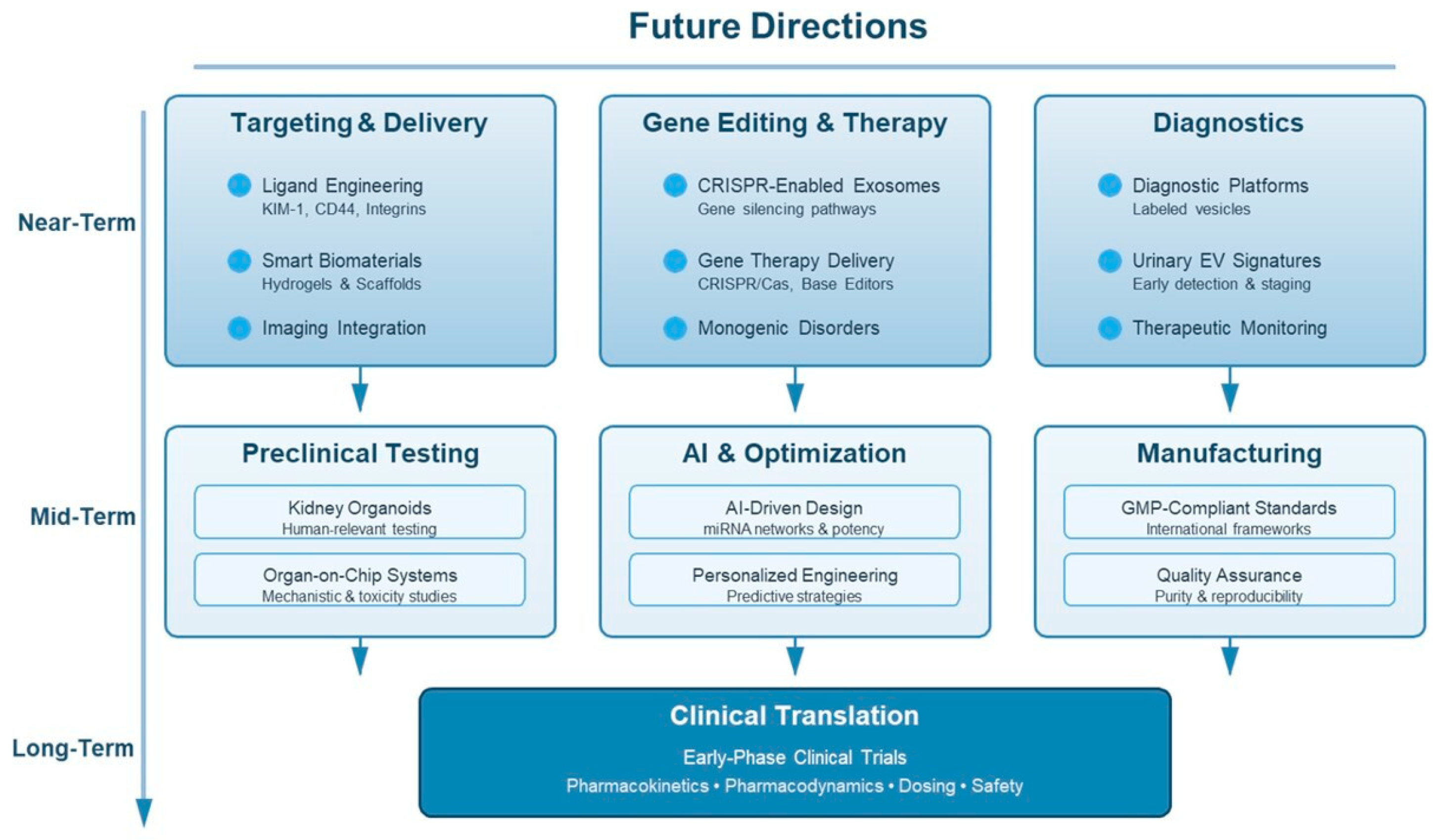
4. Future Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Convento, M.B.; Borges, F.T. Challenges and Opportunities in the Study of Bone Marrow-Derived Mesenchymal Stromal Cells: From Where We Stand to Where We Go. In Handbook of Regenerative Medicine: Stem Cell-based Approach; Haider, K.H., Ed.; CRC Press: Boca Raton, FL, USA, 2025; pp 181–198. [CrossRef]
- Welsh, J.A.; Goberdhan, D.C.I.; O’Driscoll, L.; Buzas, E.I.; Blenkiron, C.; Bussolati, B.; Cai, H.; Di Vizio, D.; Driedonks, T.A.P.; Erdbrügger, U.; et al. Minimal Information for Studies of Extracellular Vesicles (MISEV2023): From Basic to Advanced Approaches. J. Extracell. Vesicles 2024, 13, e12404. [CrossRef]
- Maguire, G. Stem Cell Therapy without the Cells. Commun. Integr. Biol. 2013, 6, e26631. [CrossRef]
- van Niel, G.; D’Angelo, G.; Raposo, G. Shedding Light on the Cell Biology of Extracellular Vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [CrossRef]
- Kowal, J.; Tkach, M.; Théry, C. Biogenesis and Secretion of Exosomes. Curr. Opin. Cell Biol. 2014, 29, 116–125. [CrossRef]
- Bianco, P.; Cao, X.; Frenette, P.S.; Mao, J.J.; Robey, P.G.; Simmons, P.J.; Wang, C.Y. The Meaning, the Sense and the Significance: Translating the Science of Mesenchymal Stem Cells into Medicine. Nat. Med. 2013, 19, 35–42. [CrossRef]
- Li, D.; Li, D.; Wang, Z.; Li, J.; Shahzad, K.A.; Wang, Y.; Tan, F. Signaling Pathways Activated and Regulated by Stem Cell-Derived Exosome Therapy. Cell Biosci. 2024, 14, 105. [CrossRef]
- Lotfy, A.; AboQuella, N.M.; Wang, H. Mesenchymal Stromal/Stem Cell (MSC)-Derived Exosomes in Clinical Trials. Stem Cell Res. Ther. 2023, 14, 66. [CrossRef]
- Butkiene, G.; Daugelaite, A.M.; Poderys, V.; Marin, R.; Steponkiene, S.; Kazlauske, E.; Uzieliene, I.; Daunoravicius, D.; Jaque, D.; Rotomskis, R.; et al. Synergistic Enhancement of Photodynamic Cancer Therapy with Mesenchymal Stem Cells and Theranostic Nanoparticles. ACS Appl. Mater. Interfaces 2024, 16, 49092–49103. [CrossRef]
- Pesce, F.; Vadalà, M.; Almeida, E.; Fernandez, B.; Fouque, D.; Malyszko, J.; Schmidt-Ott, K.; Stenvinkel, P.; Wheeler, D.C.; Seidu, S.; et al. International Nephrology Masterclass in Chronic Kidney Disease: Rationale, Summary, and Future Perspectives. Life 2024, 14, 1668. [CrossRef]
- Delrue, C.; De Bruyne, S.; Speeckaert, R.; Speeckaert, M.M. Urinary Extracellular Vesicles in Chronic Kidney Disease: From Bench to Bedside? Diagnostics 2023, 13, 443. [CrossRef]
- Himanshu; Gunjan; Pandey, R.P.; Mukherjee, R.; Chang, C.M. Meta-Analysis Study of the Therapeutic Impact of Mesenchymal Stem Cell–Derived Exosomes for Chronic Kidney Diseases. Biochem. Biophys. Rep. 2025, 43, 102072. [CrossRef]
- Bahadorani, M.; Nasiri, M.; Dellinger, K.; Aravamudhan, S.; Zadegan, R. Engineering Exosomes for Therapeutic Applications: Decoding Biogenesis, Content Modification, and Cargo Loading Strategies. Int. J. Nanomed. 2024, 19, 7137–7164. [CrossRef]
- Maldonado, V.V.; Patel, N.H.; Smith, E.E.; Barnes, C.L.; Gustafson, M.P.; Rao, R.R.; Samsonraj, R.M. Clinical Utility of Mesenchymal Stem/Stromal Cells in Regenerative Medicine and Cellular Therapy. J. Biol. Eng. 2023, 17, 44. [CrossRef]
- Ma, Z.J.; Yang, J.J.; Lu, Y.B.; Liu, Z.Y.; Wang, X.X. Mesenchymal Stem Cell-Derived Exosomes: Toward Cell-Free Therapeutic Strategies in Regenerative Medicine. World J. Stem Cells 2020, 12, 814–840. [CrossRef]
- Zhang, B.; Tian, X.; Hao, J.; Xu, G.; Zhang, W. Mesenchymal Stem Cell-Derived Extracellular Vesicles in Tissue Regeneration. Cell Transplant. 2020, 29, 963689720908500. [CrossRef]
- Lötvall, J.; Hill, A.F.; Hochberg, F.; Buzás, E.I.; Di Vizio, D.; Gardiner, C.; Gho, Y.S.; Kurochkin, I.V.; Mathivanan, S.; Quesenberry, P.; et al. Minimal Experimental Requirements for Definition of Extracellular Vesicles and Their Functions: A Position Statement from the International Society for Extracellular Vesicles. J. Extracell. Vesicles 2014, 3, 26913. [CrossRef]
- Reiner, A.T.; Witwer, K.W.; van Balkom, B.W.M.; de Beer, J.; Brodie, C.; Corteling, R.L.; Gabrielsson, S.; Gimona, M.; Ibrahim, A.G.; de Kleijn, D.; et al. Concise Review: Developing Best-Practice Models for the Therapeutic Use of Extracellular Vesicles. Stem Cells Transl. Med. 2017, 6, 1730–1773. [CrossRef]
- Barile, L.; Vassalli, G. Exosomes: Therapy Delivery Tools and Biomarkers of Diseases. Pharmacol. Ther. 2017, 174, 63–78. [CrossRef]
- Bruno, S.; Grange, C.; Deregibus, M.C.; Calogero, R.A.; Saviozzi, S.; Collino, F.; Morando, L.; Busca, A.; Falda, M.; Bussolati, B.; et al. Mesenchymal Stem Cell-Derived Microvesicles Protect against Acute Tubular Injury. J. Am. Soc. Nephrol. 2009, 20, 1053–1067. [CrossRef]
- Skotland, T.; Sandvig, K.; Llorente, A. Lipids in Exosomes: Current Knowledge and the Way Forward. Prog. Lipid Res. 2017, 66, 30–41. [CrossRef]
- Record, M.; Silvente-Poirot, S.; Poirot, M.; Wakelam, M.J.O. Extracellular Vesicles: Lipids as Key Components of Their Biogenesis and Functions. J. Lipid Res. 2018, 59, 1316–1324. [CrossRef]
- Dalal, V.; Lata, H.; Kharkwal, G.; Jotwani, G. Regulation of Clinical Research for Cellular and Gene Therapy Products in India. Adv. Exp. Med. Biol. 2023, 1430, 135–154. [CrossRef]
- Al-Jipouri, A.; Almurisi, S.H.; Al-Japairai, K.; Bakar, L.M.; Doolaanea, A.A. Liposomes or Extracellular Vesicles: A Comprehensive Comparison of Both Lipid Bilayer Vesicles for Pulmonary Drug Delivery. Polymers (Basel) 2023, 15, 318. [CrossRef]
- Pan, L.; Garcia, S.G.; Font-Morón, M.; Sanroque-Muñoz, M.; Clos-Sansalvador, M.; Garcia, G.M.; Borràs, F.E.; Franquesa, M. Beyond Preclinical Promise: Can Mesenchymal Stromal Cell-Derived Extracellular Vesicles Reliably Target Tubular Epithelial Cells? Extracell. Vesicles Circ. Nucleic Acids 2025, 6, 580–593. [CrossRef]
- Luiz, R.D.S.; Rampaso, R.R.; Dos Santos, A.A.C.; Convento, M.B.; Barbosa, D.A.; da Fonseca, C.D.; de Oliveira, A.S.; Caires, A.; Furlan, A.; Schor, N.; et al. BM-MSC–Derived Small Extracellular Vesicles (sEV) from Trained Animals Present Nephroprotective Potential in a Unilateral Ureteral Obstruction Model. J. Venom Anim. Toxins Incl. Trop. Dis. 2021, 27, e20200187. [CrossRef]
- Ahmadian, S.; Jafari, N.; Tamadon, A.; Ghaffarzadeh, A.; Rahbarghazi, R.; Mahdipour, M. Different Storage and Freezing Protocols for Extracellular Vesicles: A Systematic Review. Stem Cell Res. Ther. 2024, 15, 453. [CrossRef]
- Li, R.; Tao, H.; Pan, K.; Li, R.; Guo, Z.; Chen, X.; Li, Z. Extracellular Vesicles Derived from Mesenchymal Stem Cells Alleviate Renal Fibrosis via the miR-99b-5p/mTOR/Autophagy Axis in Diabetic Kidney Disease. Stem Cell Res. Ther. 2025, 16, 142. [CrossRef]
- Yoo, D.; Jung, S.Y.; Go, D.; Park, J.Y.; You, D.G.; Jung, W.K.; Li, Y.; Ding, J.; Park, J.H.; Um, W. Functionalized Extracellular Vesicles of Mesenchymal Stem Cells for Regenerative Medicine. J. Nanobiotechnol. 2025, 23, 219. [CrossRef]
- Zuk, A.; Bonventre, J.V. Acute Kidney Injury. Annu. Rev. Med. 2016, 67, 293–307. [CrossRef]
- Malek, M.; Nematbakhsh, M. Renal Ischemia/Reperfusion Injury: From Pathophysiology to Treatment. J. Renal Inj. Prev. 2015, 4, 20–27. [CrossRef]
- Mulcahy, L.A.; Pink, R.C.; Carter, D.R. Routes and Mechanisms of Extracellular Vesicle Uptake. J. Extracell. Vesicles 2014, 3, 24641. [CrossRef]
- Costa Verdera, H.; Gitz-Francois, J.J.; Schiffelers, R.M.; Vader, P. Cellular Uptake of Extracellular Vesicles Is Mediated by Clathrin-Independent Endocytosis and Macropinocytosis. J. Control. Release 2017, 266, 100–108. [CrossRef]
- Ji, X.; Nie, S.; Li, X.; Liu,H.; Du, X.; Fan, L. MSC-Derived Exosomal miR-127-3p Alleviates Acute Kidney Ischemia–Reperfusion Injury via Suppressing ATG5/ATG7-Mediated Autophagy. Nephrology 2025, 30, e70054. [CrossRef]
- Taghavi, S.; Keshtkar, S.; Abedanzadeh, M.; Hashemi, M.; Heidari, R.; Abolmaali, S.S.; Dara, M.; Aghdaei, M.H.; Sabegh, A.; Azarpira, N. Exosome Loaded in Microneedle Patch Ameliorates Renal Ischemia–Reperfusion Injury in a Mouse Model. Stem Cells Int. 2025, 2025, 3106634. [CrossRef]
- Kong, D.; Yang, Z.; Zhang, X.; Song, Y.; Anayyat, U.; Li, Y.; Liu, H.; Ou, Z.; Pang, S.; Wang, X. Cisplatin-Induced Acute Kidney Injury Is Alleviated by BMSC-Derived Exosomes via mmu-miR-874-3p–Mediated Activation of the Wnt/β-Catenin Signaling Pathway. Regen. Ther. 2025, 30, 719–729. [CrossRef]
- Yang, T.; Yu, H.; Xie, Z. Curcumin-Induced Exosomal FTO from Bone Marrow Stem Cells Alleviates Sepsis-Associated Acute Kidney Injury by Modulating the m6A Methylation of OXSR1. Kaohsiung J. Med. Sci. 2025, 41, e12923. [CrossRef]
- Çam, S.B.; Çiftci, E.; Gürbüz, N.; Altun, B.; Korkusuz, P. Allogeneic Bone Marrow Mesenchymal Stem Cell-Derived Exosomes Alleviate Human Hypoxic AKI-on-a-Chip within a Tight Treatment Window. Stem Cell Res. Ther. 2024, 15, 105. [CrossRef]
- Chen, Y.; Hou, S. Targeted Treatment of Rat AKI Induced by Rhabdomyolysis Using BMSC-Derived Magnetic Exosomes and Its Mechanism. Nanoscale Adv. 2024, 6, 4180–4195. [CrossRef]
- Dong, Y.; Tong, Y. The Pathogenesis of Chronic Kidney Disease (CKD) and the Preventive and Therapeutic Effects of Natural Products. Curr. Issues Mol. Biol. 2025, 47, 853. [CrossRef]
- Kandel, R.; Roy, P.; Singh, K.P. Molecular Basis of Oxidative Stress-Induced Acute Kidney Injury, Kidney Fibrosis, Chronic Kidney Disease, and Clinical Significance of Targeting Reactive Oxygen Species-Regulated Pathways to Treat Kidney Disease. Front. Biosci. (Schol. Ed.) 2025, 17 (3), 38963. [CrossRef]
- Lv, Y.; Hua, Z.; Lu, X. Protective Effects and Possible Mechanisms of Mesenchymal Stem Cells and Mesenchymal Stem Cell-Derived Extracellular Vesicles against Kidney Fibrosis in Animal Models: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2025, 15, 1511525. [CrossRef]
- Tian, J.; Wang, Z.; Tang, Z.; Zhu, H.; Sun, L. Engineered Exosomes Conferred with ROS Regulation and Immunosuppression for Ameliorating Lupus Nephritis. J. Nanobiotechnol. 2025, 23, 639. [CrossRef]
- Zhang, J.; Wang, L.; Liu, D.; Liu, Z.; Liu, Z.; Zhou, S. Bone Marrow Mesenchymal Stem Cell-Derived Exosomes Protect against β-Cell Destruction Models and Kidney Injury by Suppressing Ferroptosis. Sci. Rep. 2025, 15, 40644. [CrossRef]
- Wang, Y.; Lu, D.; Lv, S.; Liu, X.; Liu, G. Mesenchymal Stem Cell-Derived Exosomes Ameliorate Diabetic Kidney Disease through the NOD2 Signaling Pathway. Ren. Fail. 2024, 46, 2381597. [CrossRef]
- Chen, Y.; Yang, X.; Feng, M.; Yu, Y.; Hu, Y.; Jiang, W. Exosomal miR-223-3p from Bone Marrow Mesenchymal Stem Cells Targets HDAC2 to Downregulate STAT3 Phosphorylation and Alleviate HBx-Induced Ferroptosis in Podocytes. Front. Pharmacol. 2024, 15, 1327149. [CrossRef]
- Convento, M.B.; de Oliveira, A.S.; Boim, M.A.; Borges, F.T. Mesenchymal Stromal Cell Nanovesicles Carry microRNA with Nephroprotective Properties Regardless of Aging. Curr. Aging Sci. 2024, 17, 118–126. [CrossRef]
- Di Trapani, M.; Bassi, G.; Midolo, M.; Gatti, A.; Kamga, P.T.; Cassaro, A.; Carusone, R.; Adamo, A.; Krampera, M. Differential and Transferable Modulatory Effects of Mesenchymal Stromal Cell-Derived Extracellular Vesicles on T, B and NK Cell Functions. Sci. Rep. 2016, 6, 24120. [CrossRef]
- Li, J.; Ge, Z.; Ji, W.; Yuan, N.; Wang, K. The Proosteogenic and Proangiogenic Effects of Small Extracellular Vesicles Derived from Bone Marrow Mesenchymal Stem Cells Are Attenuated in Steroid-Induced Osteonecrosis of the Femoral Head. Biomed. Res. Int. 2020, 2020, 4176926. [CrossRef]
- Pisitkun, T.; Shen, R.F.; Knepper, M.A. Identification and Proteomic Profiling of Exosomes in Human Urine. Proc. Natl. Acad. Sci. U.S.A. 2004, 101, 13368–13373. [CrossRef]
- Sun, I.O.; Lerman, L.O. Urinary Extracellular Vesicles as Biomarkers of Kidney Disease: From Diagnostics to Therapeutics. Diagnostics 2020, 10, 311. [CrossRef]
- Grange, C.; Dalmasso, A.; Cortez, J.J.; Spokeviciute, B.; Bussolati, B. Exploring the Role of Urinary Extracellular Vesicles in Kidney Physiology, Aging, and Disease Progression. Am. J. Physiol. Cell Physiol. 2023, 325, C1439–C1450. [CrossRef]
- Takizawa, K.; Ueda, K.; Sekiguchi, M.; Nakano, E.; Nishimura, T.; Kajiho, Y.; Kanda, S.; Miura, K.; Hattori, M.; Hashimoto, J.; et al. Urinary Extracellular Vesicles Signature for Diagnosis of Kidney Disease. iScience 2022, 25, 105416. [CrossRef]
- Barreiro, K.; Dwivedi, O.P.; Rannikko, A.; Holthöfer, H.; Tuomi, T.; Groop, P.H.; Puhka, M. Capturing the Kidney Transcriptome by Urinary Extracellular Vesicles—From Pre-Analytical Obstacles to Biomarker Research. Genes 2023, 14, 1415. [CrossRef]
- Tutrone, R.; Donovan, M.J.; Torkler, P.; Tadigotla, V.; McLain, T.; Noerholm, M.; Skog, J.; McKiernan, J. Clinical Utility of the Exosome-Based ExoDx Prostate (IntelliScore) EPI Test in Men Presenting for Initial Biopsy with a PSA of 2–10 ng/mL. Prostate Cancer Prostatic Dis. 2020, 23, 607–614. [CrossRef]
- Chen, Y.S.; Lin, E.Y.; Chiou, T.W.; Harn, H.J. Exosomes in Clinical Trial and Their Production in Compliance with Good Manufacturing Practice. Tzu Chi Med. J. 2020, 32, 113–120. [CrossRef]
- Rezaie, J.; Feghhi, M.; Etemadi, T. A Review on Exosomes Application in Clinical Trials: Perspective, Questions, and Challenges. Cell Commun. Signal. 2022, 20, 145. [CrossRef]
- Yadav, K.; Sahu, K.K.; Sucheta; Minz, S.; Pradhan, M. Unlocking Exosome Therapeutics: The Critical Role of Pharmacokinetics in Clinical Applications. Tissue Cell 2025, 93, 102749. [CrossRef]
- Lenzini, S. Establishing a Working Range for Effective MSC–EV Dose. RoosterBio, 2021. (accessed Nov 10, 2025).
- ClinicalTrials.gov. Search Results for “Exosome OR Exosomes”. U.S. National Library of Medicine. (accessed Nov 10, 2025).
- Tan, F.; Li, X.; Wang, Z.; Li, J.; Shahzad, K.; Zheng, J. Clinical Applications of Stem Cell-Derived Exosomes. Signal Transduct. Target. Ther. 2024, 9, 17. [CrossRef]
- Ichimura, T.; Asseldonk, E.J.P.; Humphreys, B.D.; Gunaratnam, L.; Duffield, J.S.; Bonventre, J.V. Kidney Injury Molecule-1 Is a Phosphatidylserine Receptor That Confers a Phagocytic Phenotype on Epithelial Cells. J. Clin. Invest. 2008, 118, 1657–1668. [CrossRef]
- Matsushita, K.; Toyoda, T.; Akane, H.; Morikawa, T.; Ogawa, K. CD44 Expression in Renal Tubular Epithelial Cells in the Kidneys of Rats with Cyclosporine-Induced Chronic Kidney Disease. J. Toxicol. Pathol. 2024, 37, 55–67. [CrossRef]
- Ferenbach, D.A.; Bonventre, J.V. Mechanisms of Maladaptive Repair after AKI Leading to Accelerated Kidney Ageing and CKD. Nat. Rev. Nephrol. 2015, 11, 264–276. [CrossRef]
- Chao, T.; Zhao, J.; Gao, R.; Wang, H.; Guo, J.; Gao, Z.; Wang, Y. Exosome Surface Modification and Functionalization: A Narrative Review of Emerging Technologies and Their Application Potential in Precision Medicine. Adv. Technol. Neurosci. 2025, 2, 27–33. [CrossRef]
- Wang, B.; Yao, K.; Huuskes, B.M.; Shen, H.H.; Zhuang, J.; Godson, C.; Brennan, E.P.; Wilkinson-Berka, J.L.; Wise, A.F.; Ricardo, S.D. Mesenchymal Stem Cells Deliver Exogenous microRNA-let7c via Exosomes to Attenuate Renal Fibrosis. Mol. Ther. 2016, 24, 1290–1301. [CrossRef]
- Tang, T.T.; Wang, B.; Li, Z.L.; Wen, Y.; Feng, S.T.; Wu, M.; Liu, D.; Cao, J.Y.; Yin, Q.; Yin, D.; et al. Kim-1 Targeted Extracellular Vesicles: A New Therapeutic Platform for RNAi to Treat AKI. J. Am. Soc. Nephrol. 2021, 32 (10), 2467–2483. [CrossRef]
- Birtwistle, L.; Chen, X.M.; Pollock, C. Mesenchymal Stem Cell-Derived Extracellular Vesicles to the Rescue of Renal Injury. Int. J. Mol. Sci. 2021, 22 (12), 6596. [CrossRef]
- Zhou, X.; Chen, Q.; Guo, C.; Su, Y.; Guo, H.; Cao, M.; Liu, Z.; Zhang, D.; Diao, N.; Fan, H.; et al. CD44 Receptor-Targeted and Reactive Oxygen Species-Responsive H2S Donor Micelles Based on Hyaluronic Acid for the Therapy of Renal Ischemia/Reperfusion Injury. ACS Omega 2022, 7, 42339–42346. [CrossRef]
- Luo, Z.W.; Li, F.X.; Liu, Y.W.; Rao, S.S.; Yin, H.; Huang, J.; Chen, C.Y.; Hu, Y.; Zhang, Y.; Tan, Y.J.; et al. Aptamer-Functionalized Exosomes from Bone Marrow Stromal Cells Target Bone to Promote Bone Regeneration. Nanoscale 2019, 11, 20884–20892. [CrossRef]
- Gao, S.; Hein, S.; Dagnæs-Hansen, F.; Weyer, K.; Yang, C.; Nielsen, R.; Christensen, E.I.; Fenton, R.A.; Kjems, J. Megalin-Mediated Specific Uptake of Chitosan/siRNA Nanoparticles in Mouse Kidney Proximal Tubule Epithelial Cells Enables AQP1 Gene Silencing. Theranostics 2014, 4, 1039–1051. [CrossRef]
- Knudsen, K.B.; Northeved, H.; Kumar, P.E.; Permin, A.; Gjetting, T.; Andresen, T.L.; Larsen, S.; Wegener, K.M.; Lykkesfeldt, J.; Jantzen, K.; et al. In Vivo Toxicity of Cationic Micelles and Liposomes. Nanomedicine 2015, 11, 467–477. [CrossRef]
- Smyth, T.; Kullberg, M.; Malik, N.; Smith-Jones, P.; Graner, M.W.; Anchordoquy, T.J. Biodistribution and Delivery Efficiency of Unmodified Tumor-Derived Exosomes. J. Control. Release 2015, 199, 145–155. [CrossRef]
- Vader, P.; Mol, E.A.; Pasterkamp, G.; Schiffelers, R.M. Extracellular Vesicles for Drug Delivery. Adv. Drug Deliv. Rev. 2016, 106, 148–156. [CrossRef]
- Li, X.; Xiong, Y. Application of “Click” Chemistry in Biomedical Hydrogels. ACS Omega 2022, 7, 36918–36928. [CrossRef]
- Li, B.; Qi, C.; Zhang, Y.; Shi, L.; Zhang, J.; Qian, H.; Ji, C. Frontier Role of Extracellular Vesicles in Kidney Disease. J. Nanobiotechnol. 2024, 22, 583. [CrossRef]
- Ruan, H.; Li, Y.; Wang, C.; Jiang, Y.; Han, Y.; Li, Y.; Zheng, D.; Ye, J.; Chen, G.; Yang, G.Y.; et al. Click Chemistry Extracellular Vesicle/Peptide/Chemokine Nanocarriers for Treating Central Nervous System Injuries. Acta Pharm. Sin. B 2023, 13, 2202–2218. [CrossRef]
- Kim, J.; Lee, N.; Hyeon, T. Recent Development of Nanoparticles for Molecular Imaging. Philos. Trans. A Math. Phys. Eng. Sci. 2017, 375, 20170022. [CrossRef]
- Stichler, S.; Jungst, T.; Schamel, M.; Zilkowski, I.; Kuhlmann, M.; Böck, T.; Blunk, T.; Teßmar, J.; Groll, J. Thiol–Ene Clickable Poly(glycidol) Hydrogels for Biofabrication. Ann. Biomed. Eng. 2017, 45, 273–285. [CrossRef]
- Smyth, T.; Petrova, K.; Payton, N.M.; Persaud, I.; Redzic, J.S.; Graner, M.W.; Smith-Jones, P.; Anchordoquy, T.J. Surface Functionalization of Exosomes Using Click Chemistry. Bioconjugate Chem. 2014, 25, 1777–1784. [CrossRef]
- Nakase, I.; Akita, H.; Kogure, K.; Gräslund, A.; Langel, U.; Harashima, H.; Futaki, S. Efficient Intracellular Delivery of Nucleic Acid Pharmaceuticals Using Cell-Penetrating Peptides. Acc. Chem. Res. 2012, 45, 1132–1139. [CrossRef]
- Meng, W.; He, C.; Hao, Y.; Wang, L.; Li, L.; Zhu, G. Prospects and Challenges of Extracellular Vesicle-Based Drug Delivery System: Considering Cell Source. Drug Deliv. 2020, 27 (1), 585–598. [CrossRef]
- Shao, H.; Chung, J.; Balaj, L.; Charest, A.; Bigner, D.D.; Carter, B.S.; Hochberg, F.H.; Breakefield, X.O.; Weissleder, R.; Lee, H. Protein Typing of Circulating Microvesicles Allows Real-Time Monitoring of Glioblastoma Therapy. Nat. Med. 2012, 18, 1835–1840. [CrossRef]
- Mediratta, K.; Diab, M.D.; Han, P.; Hu, H.; Wang, L. Emerging Strategies for Cargo Loading and Engineering of Extracellular Vesicles for Breast Cancer Treatment. Nanomaterials 2025, 15, 1418. [CrossRef]
- Qiao, L.; Hu, J.; Qiu, X.; Wang, C.; Peng, J.; Zhang, C.; Zhang, M.; Lu, H.; Chen, W. LAMP2A, LAMP2B and LAMP2C: Similar Structures, Divergent Roles. Autophagy 2023, 19, 2837–2852. [CrossRef]
- Liu, S.; Lv, K.; Wang, Y.; Lou, P.; Zhou, P.; Wang, C.; Li, L.; Liao, G.; Zhang, Y.; Chen, Y.; et al. Improving the Circulation Time and Renal Therapeutic Potency of Extracellular Vesicles Using an Endogenous Ligand Binding Strategy. J. Control. Release 2022, 352, 1009–1023. [CrossRef]
- Kim, S.; Lee, S.A.; Yoon, H.; Kim, M.Y.; Yoo, J.K.; Ahn, S.H.; Park, C.H.; Park, J.; Nam, B.Y.; Park, J.T.; et al. Exosome-Based Delivery of Super-Repressor IκBα Ameliorates Kidney Ischemia–Reperfusion Injury. Kidney Int. 2021, 100, 570–584. [CrossRef]
- Han, G.; Zhang, Y.; Zhong, L.; Wang, B.; Qiu, S.; Song, J.; Lin, C.; Zou, F.; Wu, J.; Yu, H.; et al. Generalizable Anchor Aptamer Strategy for Loading Nucleic Acid Therapeutics on Exosomes. EMBO Mol. Med. 2024, 16, 1027–1045. [CrossRef]
- Kooijmans, S.A.A.; Gitz-François, J.J.J.M.; Schiffelers, R.M.; Vader, P. Recombinant Phosphatidylserine-Binding Nanobodies for Targeting of Extracellular Vesicles to Tumor Cells: A Plug-and-Play Approach. Nanoscale 2018, 10, 2413–2426. [CrossRef]
- Humphreys, B.D.; Xu, F.; Sabbisetti, V.; Grgic, I.; Movahedi Naini, S.; Wang, N.; Chen, G.; Xiao, S.; Patel, D.; Henderson, J.M.; et al. Chronic Epithelial Kidney Injury Molecule-1 Expression Causes Murine Kidney Fibrosis. J. Clin. Invest. 2013, 123 (9), 4023–4035. [CrossRef]
- Verweij, F.J.; Revenu, C.; Arras, G.; Dingli, F.; Loew, D.; Pegtel, D.M.; Follain, G.; Allio, G.; Goetz, J.G.; Zimmermann, P.; et al. Live Tracking of Inter-Organ Communication by Endogenous Exosomes In Vivo. Dev. Cell 2019, 48, 573–589.e4. [CrossRef]
- Kowal, J.; Arras, G.; Colombo, M.; Jouve, M.; Morath, J.P.; Primdal-Bengtson, B.; Dingli, F.; Loew, D.; Tkach, M.; Théry, C. Proteomic Comparison Defines Novel Markers to Characterize Heterogeneous Populations of Extracellular Vesicle Sub-Types. Proc. Natl. Acad. Sci. U.S.A. 2016, 113, E968–E977. [CrossRef]
- Grange, C.; Tapparo, M.; Bruno, S.; Chatterjee, D.; Quesenberry, P.J.; Tetta, C.; Camussi, G. Biodistribution of Mesenchymal Stem Cell-Derived Extracellular Vesicles in a Model of Acute Kidney Injury Monitored by Optical Imaging. Int. J. Mol. Med. 2014, 33, 1055–1063. [CrossRef]
- György, B.; Hung, M.E.; Breakefield, X.O.; Leonard, J.N. Therapeutic Applications of Extracellular Vesicles: Clinical Promise and Open Questions. Annu. Rev. Pharmacol. Toxicol. 2015, 55, 439–464. [CrossRef]
- M.; Liu, S.; Wang, C.; Wang, Y.; Wan, M.; Liu, F.; Gong, M.; Yuan, Y.; Chen, Y.; Cheng, J.; et al. Mesenchymal Stem Cell-Derived Extracellular Vesicles Attenuate Mitochondrial Damage and Inflammation by Stabilizing Mitochondrial DNA. ACS Nano 2021, 15, 1519–1538. [CrossRef]
- Grange, C.; Tritta, S.; Tapparo, M.; Cedrino, M.; Tetta, C.; Camussi, G.; Brizzi, M.F. Stem Cell-Derived Extracellular Vesicles Inhibit and Revert Fibrosis Progression in a Mouse Model of Diabetic Nephropathy. Sci. Rep. 2019, 9, 4468. [CrossRef]
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-Mediated Transfer of mRNAs and microRNAs Is a Novel Mechanism of Genetic Exchange Between Cells. Nat. Cell Biol. 2007, 9, 654–659. [CrossRef]
- Villarroya-Beltri, C.; Gutiérrez-Vázquez, C.; Sánchez-Cabo, F.; Pérez-Hernández, D.; Vázquez, J.; Martin-Cofreces, N.; Martinez-Herrera, D.J.; Pascual-Montano, A.; Mittelbrunn, M.; Sánchez-Madrid, F. Sumoylated hnRNPA2B1 Controls the Sorting of miRNAs into Exosomes through Binding to Specific Motifs. Nat. Commun. 2013, 4, 2980. [CrossRef]
- Shurtleff, M.J.; Temoche-Diaz, M.M.; Karfilis, K.V.; Ri, S.; Schekman, R. Y-Box Protein 1 Is Required to Sort microRNAs into Exosomes in Cells and in a Cell-Free Reaction. eLife 2016, 5, e19276. [CrossRef]
- Géminard, C.; De Gassart, A.; Blanc, L.; Vidal, M. Degradation of AP2 during Reticulocyte Maturation Enhances Binding of hsc70 and Alix to a Common Site on TFR for Sorting into Exosomes. Traffic 2004, 5, 181–193. [CrossRef]
- Dashti, M.; Mohammaddust Sarab, M.; Shad, F.; Dehnavi, S. CRISPR-Mediated Engineering of Mesenchymal Stromal/Stem Cells: A Summary of Recent Progress in Immunological Applications for Regenerative Medicine and Cancer Therapy. Stem Cell Res. Ther. 2025, 16, 592. [CrossRef]
- Hu, X.; Li, L.; Yu, X.; Zhang, R.; Yan, S.; Zeng, Z.; Shu, Y.; Zhao, C.; Wu, X.; Lei, J.; et al. CRISPR/Cas9-Mediated Reversibly Immortalized Mouse Bone Marrow Stromal Stem Cells (BMSCs) Retain Multipotent Features of Mesenchymal Stem Cells (MSCs). Oncotarget 2017, 8, 111847–111865. [CrossRef]
- D’Ambrosio, V.; Huimei, C.; Vo, N.; Siew, K.; Evans, R.D.R.; Freedman, B.; Pesce, F. CRISPR and Gene Editing for Kidney Diseases: Where Are We? Clin. Kidney J. 2025, 18, sfaf246. [CrossRef]
- Freedman, B.S.; Brooks, C.R.; Lam, A.Q.; Fu, H.; Morizane, R.; Agrawal, V.; Saad, A.F.; Li, M.K.; Hughes, M.R.; van Werff, R.; et al. Modelling Kidney Disease with CRISPR-Mutant Kidney Organoids Derived from Human Pluripotent Epiblast Spheroids. Nat. Commun. 2015, 6, 8715. [CrossRef]
- Vishy, C.E.; Thomas, C.; Vincent, T.; Crawford, D.K.; Goddeeris, M.M.; Freedman, B.S. Genetics of Cystogenesis in Base-Edited Human Organoids Reveal Therapeutic Strategies for Polycystic Kidney Disease. Cell Stem Cell 2024, 31, 537–553.e5. [CrossRef]
- Kim, Y.K.; Refaeli, I.; Brooks, C.R.; Jing, P.; Gulieva, R.E.; Hughes, M.R.; Cruz, N.M.; Liu, Y.; Churchill, A.J.; Wang, Y.; et al. Gene-Edited Human Kidney Organoids Reveal Mechanisms of Disease in Podocyte Development. Stem Cells 2017, 35, 2366–2378. [CrossRef]
- Gu, D.; Zou, X.; Ju, G.; Zhang, G.; Bao, E.; Zhu, Y. Mesenchymal Stromal Cells–Derived Extracellular Vesicles Ameliorate Acute Renal IRI by Inhibition of Mitochondrial Fission through miR-30. Stem Cells Int. 2016, 2016, 2093940. [CrossRef]
- Huang, L.F.; Ye, Q.R.; Chen, X.C.; Huang, X.R.; Zhang, Q.F.; Wu, C.Y.; Liu, H.F.; Yang, C. Research Progress of Drug Delivery Systems Targeting the Kidneys. Pharmaceuticals 2024, 17, 625. [CrossRef]
- Batrakova, E.V.; Kim, M.S. Using Exosomes, Naturally Equipped Nanocarriers, for Drug Delivery. J. Control. Release 2015, 219, 396–405. [CrossRef]
- Lamichhane, T.N.; Raiker, R.S.; Jay, S.M. Exogenous DNA Loading into Extracellular Vesicles via Electroporation Is Size-Dependent and Enables Limited Gene Delivery. Mol. Pharm. 2015, 12, 3650–3657. [CrossRef]
- Haney, M.J.; Klyachko, N.L.; Zhao, Y.; Gupta, R.; Plotnikova, E.G.; He, Z.; Patel, T.; Piroyan, A.; Sokolsky, M.; Kabanov, A.V.; et al. Exosomes as Drug Delivery Vehicles for Parkinson’s Disease Therapy. J. Control. Release 2015, 207, 18–30. [CrossRef]
- Sato, Y.; Umezaki, K.; Sawada, S.; Sasaki, Y.; Harada, N.; Shiku, H.; Akiyoshi, K. Engineering Hybrid Exosomes by Membrane Fusion with Liposomes. Sci. Rep. 2016, 6, 21933. [CrossRef]
- van Hoogevest, P.; Wendel, A. The Use of Natural and Synthetic Phospholipids as Pharmaceutical Excipients. Eur. J. Lipid Sci. Technol. 2014, 116, 1088–1107. [CrossRef]
- Lin, Y.; Wu, J.; Gu, W.; Huang, Y.; Tong, Z.; Huang, L.; Tan, J. Exosome–Liposome Hybrid Nanoparticles Deliver CRISPR/Cas9 System in MSCs. Adv. Sci. 2018, 5, 1700611. [CrossRef]
- Li, Y.J.; Wu, J.Y.; Liu, J.; Xu, W.; Qiu, X.; Huang, S.; Hu, X.B.; Xiang, D.X. Artificial Exosomes for Translational Nanomedicine. J. Nanobiotechnol. 2021, 19, 242. [CrossRef]
- Lener, T.; Gimona, M.; Aigner, L.; Börger, V.; Buzas, E.; Camussi, G.; Chaput, N.; Chatterjee, D.; Court, F.A.; Del Portillo, H.A.; et al. Applying Extracellular Vesicles Based Therapeutics in Clinical Trials—An ISEV Position Paper. J. Extracell. Vesicles 2015, 4, 30087. [CrossRef]
- Zhu, K.; Huang, Y.; Li, K.; Zhang, K. Biomimetic Erythrocyte-Based Drug Delivery Systems for Organ-Targeted Therapy. Front. Bioeng. Biotechnol. 2025, 13, 1663092. [CrossRef]
- Betzer, O.; Perets, N.; Angel, A.; Motiei, M.; Sadan, T.; Yadid, G.; Offen, D.; Popovtzer, R. In Vivo Neuroimaging of Exosomes Using Gold Nanoparticles. ACS Nano 2017, 11 (11), 10883–10893. [CrossRef]
- Lai, C.P.; Kim, E.Y.; Badr, C.E.; Weissleder, R.; Mempel, T.R.; Tannous, B.A.; Breakefield, X.O. Visualization and Tracking of Tumour Extracellular Vesicle Delivery and RNA Translation Using Multiplexed Reporters. Nat. Commun. 2015, 6, 7029. [CrossRef]
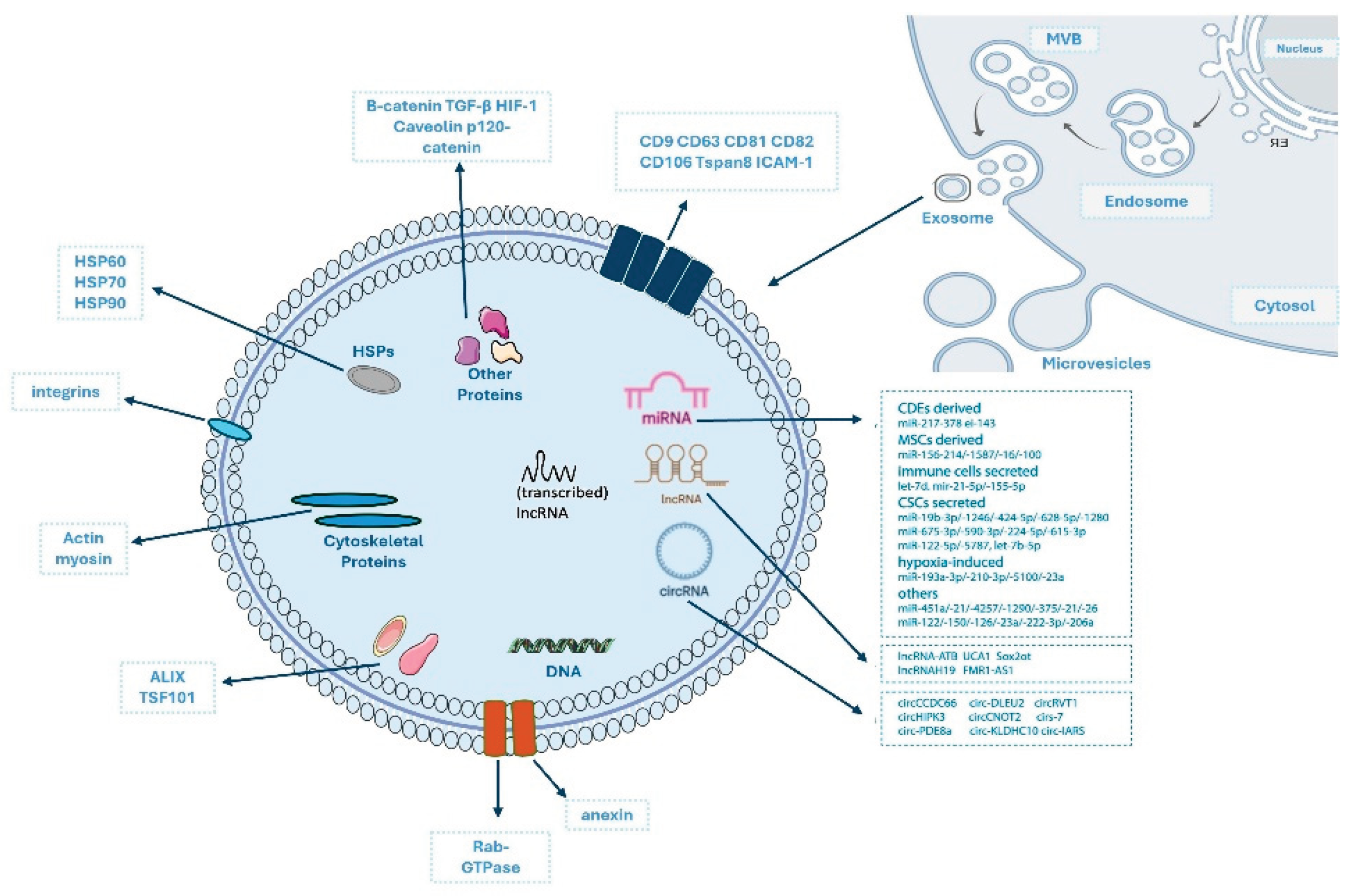
| Category | MISEV2023 Recommendations | Purpose |
|---|---|---|
|
Positive markers (Membrane-associated) |
CD63, CD9, CD81 | Confirm EVs membrane origin |
|
Positive markers (Cytosolic) |
TSG101, ALIX, HSP70, HSP90 | Validate endosome-derived or EVs-enriched cytosolic cargo |
| Negative markers | ApoA1/ApoB, Albumin, Calnexin, GM130 | Exclude non-EVs contaminants |
| Isolation approaches | Differential UC, Cushion, UC, Density gradients, SEC, TFF, Ultrafiltration, AF4, Immunocapture | Achieve selective enrichment using orthogonal strategies |
| Precipitation methods | PEG, Protamine, Ammonium sulfate, Organic solvents |
High yield but low purity, requiring validation with negative markers |
| Particle quantification | NTA, DLS, TRPS | EVs concentration and size distribution; report the particle-to-protein ratio |
| Protein quantification | BCA, Bradford, Qubit | Estimate total protein levels |
| Morphological validation | TEM, SEM, Cryo-EM, AFM, SRM | EVs structure and membrane integrity |
| Advanced phenotyping | High-sensitivity FCM, Nano-flow FCM | Assess EVs markers and heterogeneity |
| Quality controls | Negative controls, Batch reproducibility, Calibration reporting | Ensure methodological rigor and comparability |
| Category | BM-MSC-Exos | BM-MSC |
|---|---|---|
| Composition | Acellular, containing bioactive molecules | Living cells are capable of self-renewal and differentiation |
| Mechanism of Action | Paracrine communication through the delivery of RNAs, proteins, and lipids | Cell differentiation and tissue integration, in addition to paracrine effects |
| Safety | Low immunogenic risk and no tumorigenic potential | Potential for tumorigenicity, immune rejection, and embolism |
| Therapeutic Effectiveness | More predictable paracrine effects; dependent on vesicle yield and purity | Highly potent but variable due to donor heterogeneity and constraints on cell survival. |
| Production & Scalability | Less complex process, though challenged by purification and yield | Requires cell culture, expansion, differentiation, and cryopreservation |
| Stability & Transport | Storage possible at −80 °C; amenable to lyophilization | Requires cryopreservation and transport in liquid nitrogen |
| Quality Control | High potential for standardization, though batch variability exists | High donor variability; more complex manipulation |
| Regulation | Regulatory frameworks under development; lack of standardized clinical protocols | More established regulations and clinical guidelines |
| Ethical Aspects | Ethically low risk; cell-free products from adult donors | Ethically acceptable but requires invasive bone marrow collection and stringent donor oversight |
| Clinical Applications | Used as personalized therapeutic carriers with wide applicability | Broad applications, primarily in regenerative medicine |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




