Submitted:
09 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
Study Design and Protocol
Study Population
Objectives
Variables
Addressing Potential Sources of Bias
Statistical Analysis
Results
Baseline Characteristics
Outcomes
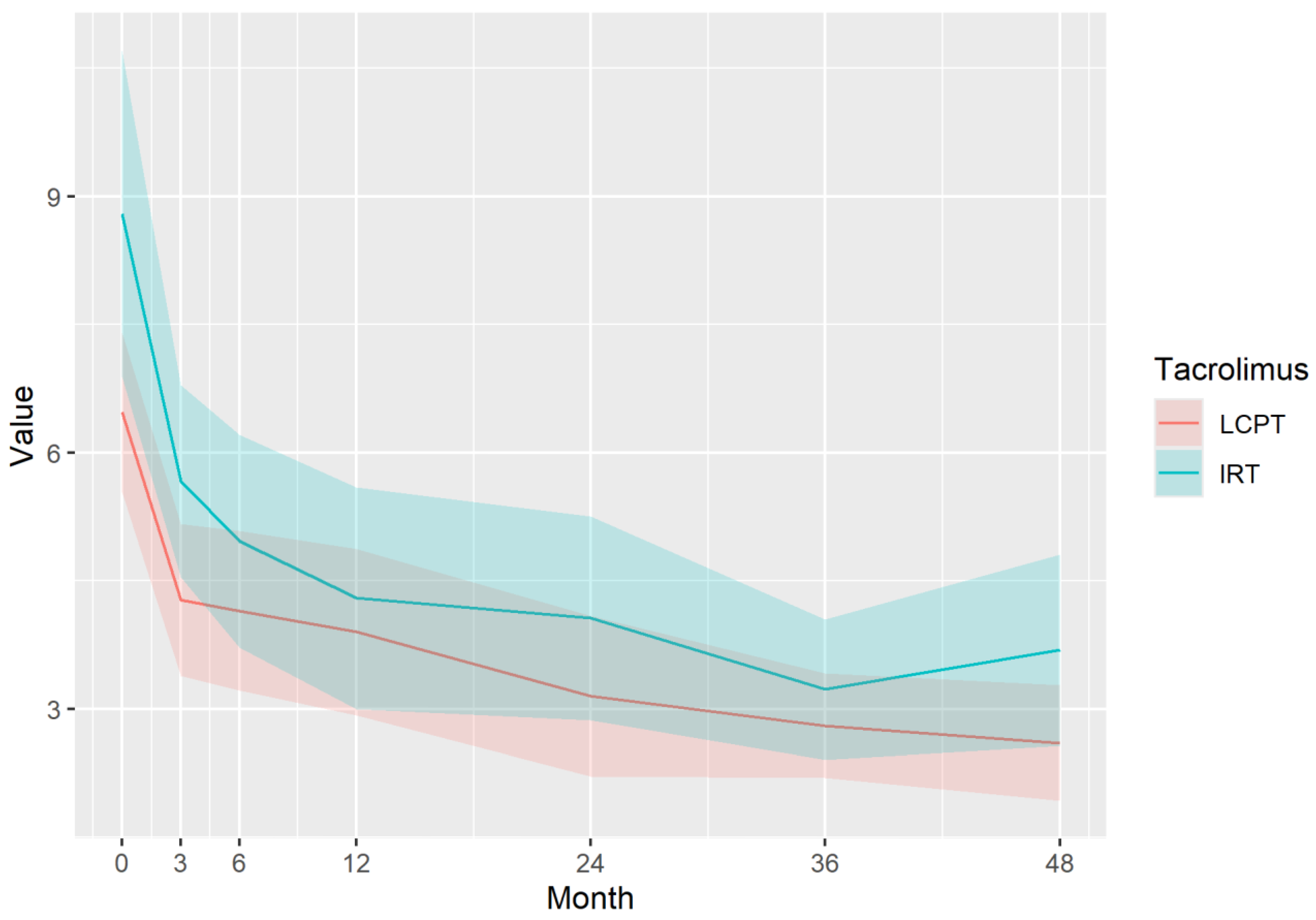
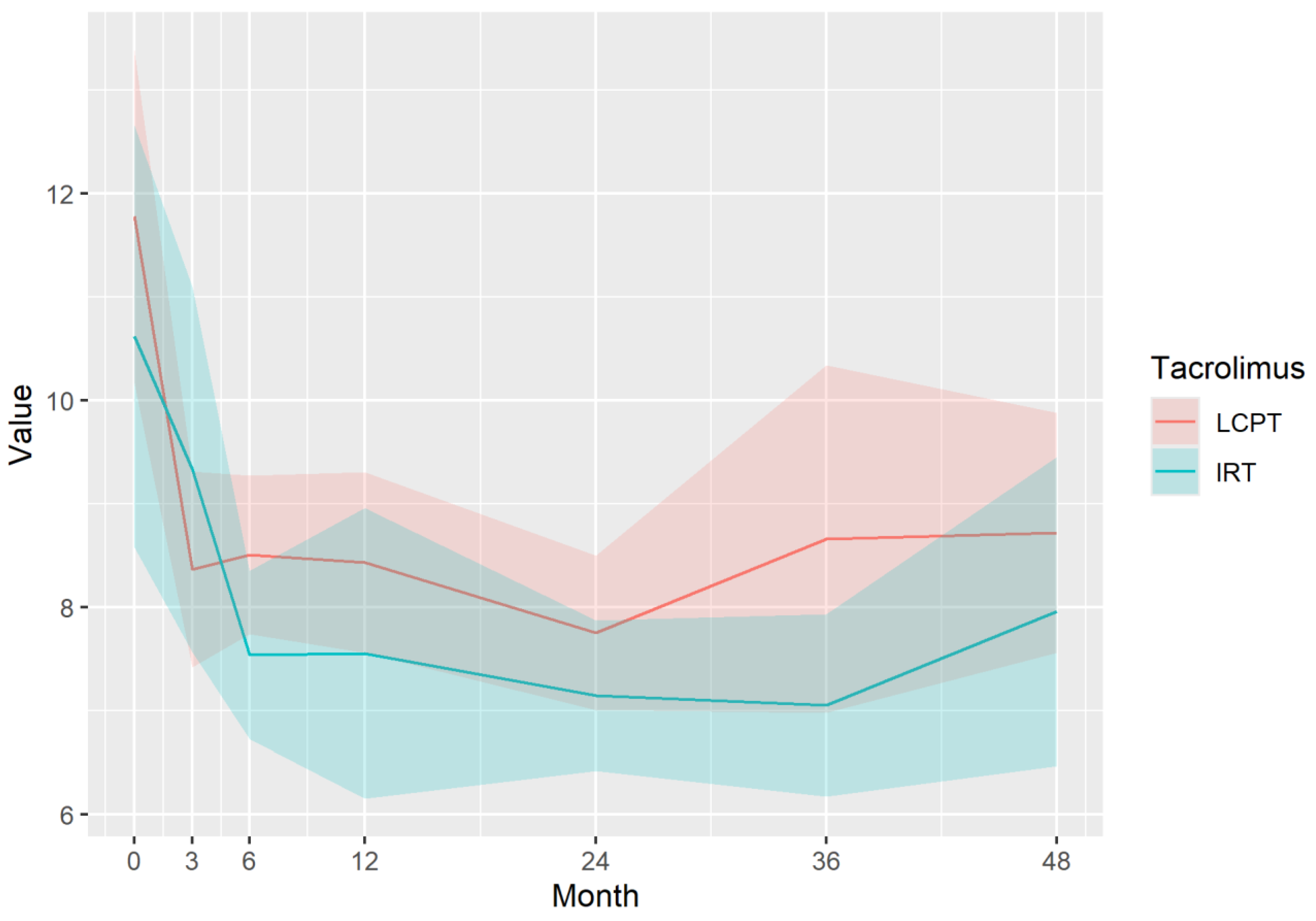
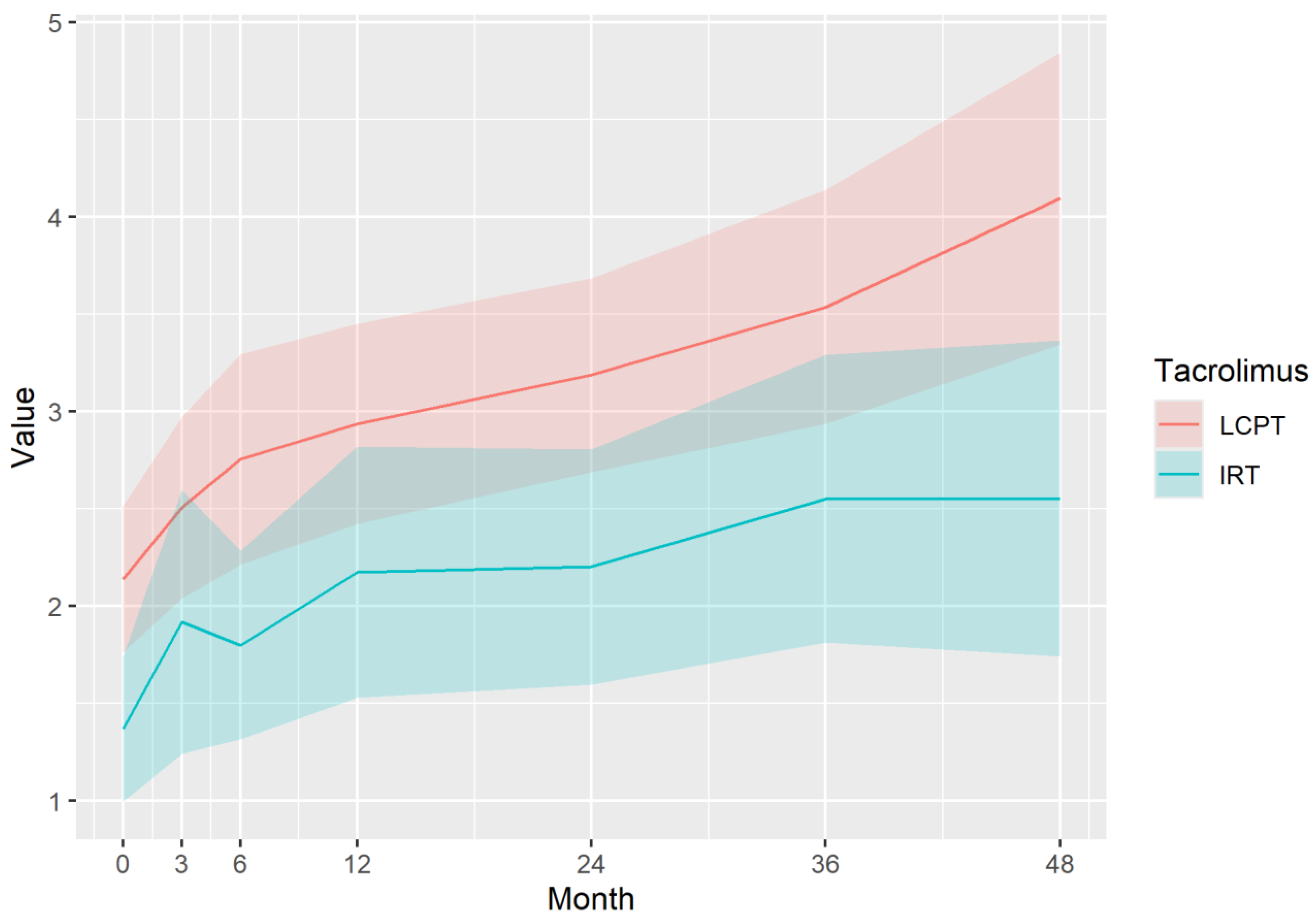
Immunosuppressive Profile of Tacrolimus
Safety
Discussion
Supplementary Materials
Funding
Data Availability Statement
Acknowledgements
Institutional Review Board Statement
Informed Consent Statement and Data Protection
Author’s contributions
Conflict of Interest
References
- Ekberg, H; Tedesco-Silva, H; Demirbas, A; et al. Reduced exposure to calcineurin inhibitors in renal transplantation. N Engl J Med 2007, 357, 2562–75. [Google Scholar] [CrossRef]
- Azzi, JR; Sayegh, MH; Mallat, SG. Calcineurin inhibitors: 40 years later, can’t live without. J Immunol 2013, 191, 5785–91. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) Transplant Work Group. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am J Transplant. 2009, 9 Suppl 3, S1–155. [CrossRef] [PubMed]
- Ruiz, R; Kirk, AD. Long-Term Toxicity of Immunosuppressive Therapy. Transplantation of the Liver 2015, 1354–63, Epub 2015 Apr 3. [Google Scholar] [CrossRef] [PubMed Central]
- Wallemacq, P; Armstrong, VW; Brunet, M; Haufroid, V; Holt, DW; Johnston, A; Kuypers, D; Le Meur, Y; Marquet, P; Oellerich, M; Thervet, E; Toenshoff, B; Undre, N; Weber, LT; Westley, IS; Mourad, M. Opportunities to optimize tacrolimus therapy in solid organ transplantation: report of the European consensus conference. Ther Drug Monit 2009, 31(2), 139–52. [Google Scholar] [CrossRef] [PubMed]
- Sapir-Pichhadze, R; Wang, Y; Famure, O; Li, Y; Kim, SJ. Time-dependent variability in tacrolimus trough blood levels is a risk factor for late kidney transplant failure. Kidney Int. 2014, 85(6), 1404–11, Epub 2013 Dec 11. Erratum in: Kidney Int. 2016 Jan;89(1):248. doi: 10.1016/j.kint.2015.10.003. [Google Scholar] [CrossRef] [PubMed]
- Bentata, Y. Tacrolimus: 20 years of use in adult kidney transplantation. What we should know about its nephrotoxicity. Artif Organs 2020, 44(2), 140–152, Epub 2019 Sep 4. [Google Scholar] [CrossRef] [PubMed]
- Ekberg, H; Mamelok, RD; Pearson, TC; Vincenti, F; Tedesco-Silva, H; Daloze, P. The challenge of achieving target drug concentrations in clinical trials: experience from the Symphony study. Transplantation 2009, 87(9), 1360–6. [Google Scholar] [CrossRef] [PubMed]
- Grinyó, JM; Ekberg, H; Mamelok, RD; et al. The pharmacokinetics of mycophenolate mofetil in renal transplant recipients receiving standard-dose or low-dose cyclosporine, low-dose tacrolimus or low-dose sirolimus: the Symphony pharmacokinetic substudy. Nephrol Dial Transplant 2009, 24, 2269–76. [Google Scholar] [CrossRef]
- Nowicka, M; Górska, M; Nowicka, Z; Edyko, K; Edyko, P; Wiślicki, S; Zawiasa-Bryszewska, A; Strzelczyk, J; Matych, J; Kurnatowska, I. Tacrolimus: Influence of the Posttransplant Concentration/Dose Ratio on Kidney Graft Function in a Two-Year Follow-Up. Kidney Blood Press Res. 2019, 44(5), 1075–1088, Epub 2019 Sep 13. [Google Scholar] [CrossRef] [PubMed]
- Schütte-Nütgen, K; Thölking, G; Steinke, J; Pavenstädt, H; Schmidt, R; Suwelack, B; Reuter, S. Fast Tac Metabolizers at Risk ⁻ It is Time for a C/D Ratio Calculation. J Clin Med. 2019, 8(5), 587, Erratum in: J Clin Med. 2019 Nov 04;8(11):E1870. doi: 10.3390/jcm8111870. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Thölking, G; Schütte-Nütgen, K; Schmitz, J; et al. A Low Tacrolimus Concentration/Dose Ratio Increases the Risk for the Development of Acute Calcineurin Inhibitor-Induced Nephrotoxicity. J Clin Med 2019, 8. [Google Scholar] [CrossRef]
- Wiseman, A; Alhamad, T; Alloway, RR; et al. Use of LCP-Tacrolimus (LCPT) in Kidney Transplantation: A Delphi Consensus Survey of Expert Clinicians. Ann Transplant 2024, 29, e943498. [Google Scholar] [CrossRef] [PubMed]
- Bunnapradist, S; Ciechanowski, K; West-Thielke, P; et al. Conversion from twice-daily tacrolimus to once-daily extended-release tacrolimus (LCPT): the phase III randomized MELT trial. Am J Transplant 2013, 13, 760. [Google Scholar] [CrossRef]
- Thölking, G; Tosun-Koç, F; Jehn, U; et al. Improved Kidney Allograft Function after Early Conversion of Fast IR-Tac Metabolizers to LCP-Tac. J Clin Med 2022, 11. [Google Scholar] [CrossRef]
- Oberbauer, R; Bestard, O; Furian, L; et al. Optimization of tacrolimus in kidney transplantation: New pharmacokinetic perspectives. Transplant Rev (Orlando) 2020, 34, 100531. [Google Scholar] [CrossRef]
- Berger, SP; Sommerer, C; Witzke, O; et al. Two-year outcomes in de novo renal transplant recipients receiving everolimus-facilitated calcineurin inhibitor reduction regimen from the TRANSFORM study. Am J Transplant 2019, 19, 3018–34. [Google Scholar] [CrossRef]
- Nunes Ficher K, Dreige Y, Gessolo Lins PR, Nicolau Ferreira A, Toniato de Rezende Freschi J, Linhares K, Stopa Martins S, Custodio L, Cristelli M, Viana L, Wagner Santos D, de Marco R, Gerbase-DeLima M, Proença H, Aguiar W, Nakamura M, Rosso Felipe C, Medina Pestana J, Tedesco Silva H Jr. Long-term Efficacy and Safety of Everolimus Versus Mycophenolate in Kidney Transplant Recipients Receiving Tacrolimus. Transplantation. 2022 Feb 1;106(2):381-390. [CrossRef] [PubMed]
- Available online: https://baasis.nursing.unibas.ch/.
- Meier-Kriesche, HU; Kaplan, B. Waiting time on dialysis as the strongest modifiable risk factor for renal transplant outcomes: a paired donor kidney analysis. Transplantation 2002, 74(10), 1377–81. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kriesche, HU.; Port, FK.; Ojo, AO.; Rudich, SM.; Hanson, JA.; Cibrik, DM.; Leichtman, AB.; Kaplan, B. Effect of waiting time on renal transplant outcome. Kidney Int. 2000, 58(3), 1311–7. [Google Scholar] [CrossRef] [PubMed]
- Loucaidou, M.1; McLean, A. G.; Cairns, T. D.H.; Griffith, M.; Hakim, N.; Palmer, A.; Papalois, V.; Van Tromp, J.; Loucaides, C.; Welsh, K. I.; Taube, D.. Five-year results of kidney transplantation under tacrolimus-based regimes: the persisting significance of vascular rejection. Transplantation 76(7):p 1120-1122, October 15, 2003. [CrossRef]
- Tremblay, S; Nigro, V; Weinberg, J; Woodle, ES; Alloway, RR. A Steady-State Head-to-Head Pharmacokinetic Comparison of All FK-506 (Tacrolimus) Formulations (ASTCOFF): An Open-Label, Prospective, Randomized, Two-Arm, Three-Period Crossover Study. Am J Transplant. 2017, 17(2), 432–442, Epub 2016 Aug 2. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Masuda, S; Inui, K. An up-to-date review on individualized dosage adjustment of calcineurin inhibitors in organ transplant patients. Pharmacol Ther 2006, 112(1), 184–98. [Google Scholar] [CrossRef] [PubMed]
- Naesens, M; Kuypers, DR; Sarwal, M. Calcineurin inhibitor nephrotoxicity. Clin J Am Soc Nephrol 2009, 4(2), 481–508. [Google Scholar] [CrossRef]
- von Einsiedel, J; Thölking, G; Wilms, C; Vorona, E; Bokemeyer, A; Schmidt, HH; Kabar, I; Hüsing-Kabar, A. Conversion from Standard-Release Tacrolimus to MeltDose® Tacrolimus (LCPT) Improves Renal Function after Liver Transplantation. J Clin Med. 2020, 9(6), 1654. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Baraldo, M. Meltdose Tacrolimus Pharmacokinetics. Transplant. Proc. 2016, 48, 420–423. [Google Scholar] [CrossRef]
- Bunnapradist, S.; Rostaing, L.; Alloway, R.R.; West-Thielke, P.; Denny, J.; Mulgaonkar, S.; Budde, K. LCPT once-daily extended-release tacrolimus tablets versus twice-daily capsules: A pooled analysis of two phase 3 trials in important de novo and stable kidney transplant recipient subgroups. Transpl. Int. 2016, 29, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Suwelack, B; Bunnapradist, S; Meier-Kriesche, U; Stevens, DR; Procaccianti, C; Morganti, R; Budde, K. Effect of Concentration/Dose Ratio in De Novo Kidney Transplant Recipients Receiving LCP-Tacrolimus or Immediate-Release Tacrolimus: Post Hoc Analysis of a Phase 3 Clinical Trial. Ann Transplant. 2020, 25, e923278. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
| Visit number | 1 - Baseline | 2 | 3 | 4 | 5 | 6 | 7 – End of Study |
|---|---|---|---|---|---|---|---|
| Schedule since Baseline (months) | 0 | 3 | 6 | 12 | 24 | 36 | 48 |
| Simple visit | X | X | |||||
| Complex visit | Baseline | X | X | X | X |
| Variables | All | LCPT | IRT | p-value |
|---|---|---|---|---|
| Recipient specific factors | ||||
| n (%) | 63 (100%) | 40 (63.49%) | 23 (36.51%) | NA |
| Age (year) | 53.35 (±11.85) | 54.05 (±12.25) | 52.13 (±11.28) | 0.5315 |
| Gender (males, %) | 41 (65.08%) | 26 (65.0%) | 15 (65.22%) | 1.000 |
| Height (cm) | 170.29 (±8.82) | 169.88 (±8.22) | 171.00 (±9.92) | 0.6476 |
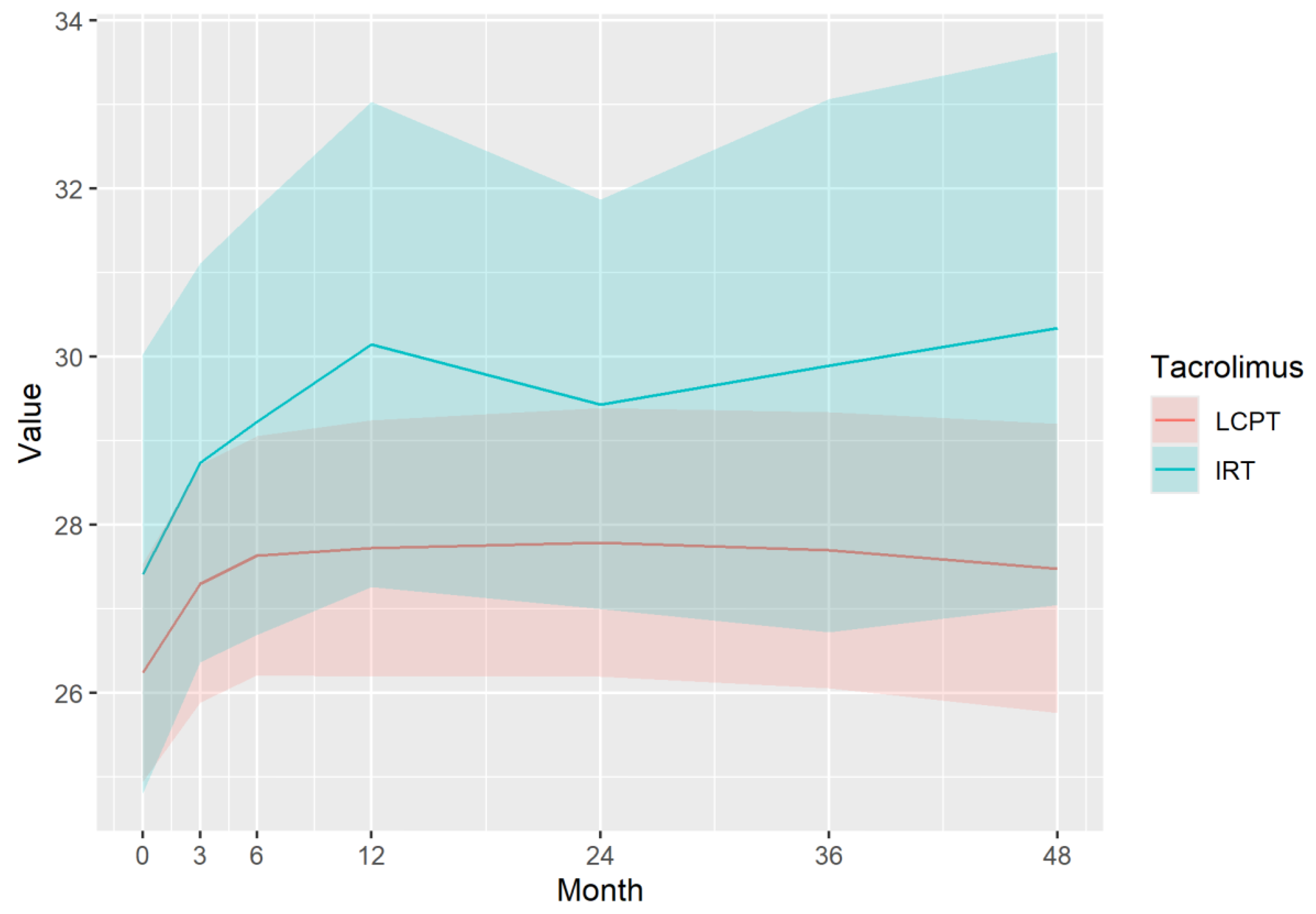
| Systolic blood pressure (Hgmm) | 143.46 (±18.58) | 146.95 (±19.09) | 137.39 (±16.30) | 0.0404 |
| Diastolic blood pressure (Hgmm) | 83.33 (±12.18) | 83.45 (±11.33) | 83.13 (±13.80) | 0.9254 |
| Pulse (/min) | 86.33 (±17.15) | 84.70 (±15.03) | 89.17 (±20.37) | 0.3641 |
| Weight (kg) | 76.76 (±13.6) | 75.73 (±12.17) | 78.55 (±15.91) | 0.4774 |
| BMI (kg/m2) | 26.52 (±3.88) | 26.28 (±3.70) | 26.93 (±4.23) | 0.5541 |
| Education level (n, %) | 0.3038 | |||
| Elementary school | 11 (17.46%) | 8 (20%) | 3 (13.04%) | |
| Apprenticeship | 23 (36.51%) | 11 (27.50%) | 12 (52.17%) | |
| High school | 18 (28.57%) | 13 (32.5%) | 5 (21.74%) | |
| University | 11 (17.46%) | 8 (20%) | 3 (13.04%) | |
| Work circumstances (n, %) | 0.1869 | |||
| Full time | 19 (30.16%) | 15 (37.5%) | 4 (17.39%) | |
| Part-time | 6 (9.52%) | 3 (7.5%) | 3 (13.04%) | |
| Unemployed | 4 (6.35%) | 2 (5%) | 2 (8.7%) | |
| Disabled | 16 (25.40%) | 7 (17.5%) | 9 (39.13%) | |
| Retired | 18 (28.57%) | 13 (32.5%) | 5 (21.74%) | |
| Social environment (n, %) In family |
55 (87.3%) | 34 (85%) | 21 (91.3%) | 0.6977 |
| Distance between home and the Transplant Centre (km) | 81.71 (±45.08) | 86 (±44.01) | 74.24 (±46.49) | 0.3328 |
| Blood parameters | ||||
| Na (mmol/l) | 138.46 (±3.2) | 138.32 (±3.38) | 138.70 (±2.90) | 0.6481 |
| K (mmol/l) | 4.69 (±0.61) | 4.70 (±0.65) | 4.68 (±0.56) | 0.9145 |
| Cl (mmol/l) | 104.21 (±4.02) | 103.95 (±4.11) | 104.65 (±3.93) | 0.5049 |
| Glucose (mmol/l) | 6.27 (±2.95) | 6.2 (±2.63) | 6.39 (±3.50) | 0.8235 |
| HbA1C (%) | 5.61 (±0.86) | 5.67(±0.68) | 5.52 (±1.09) | 0.6512 |
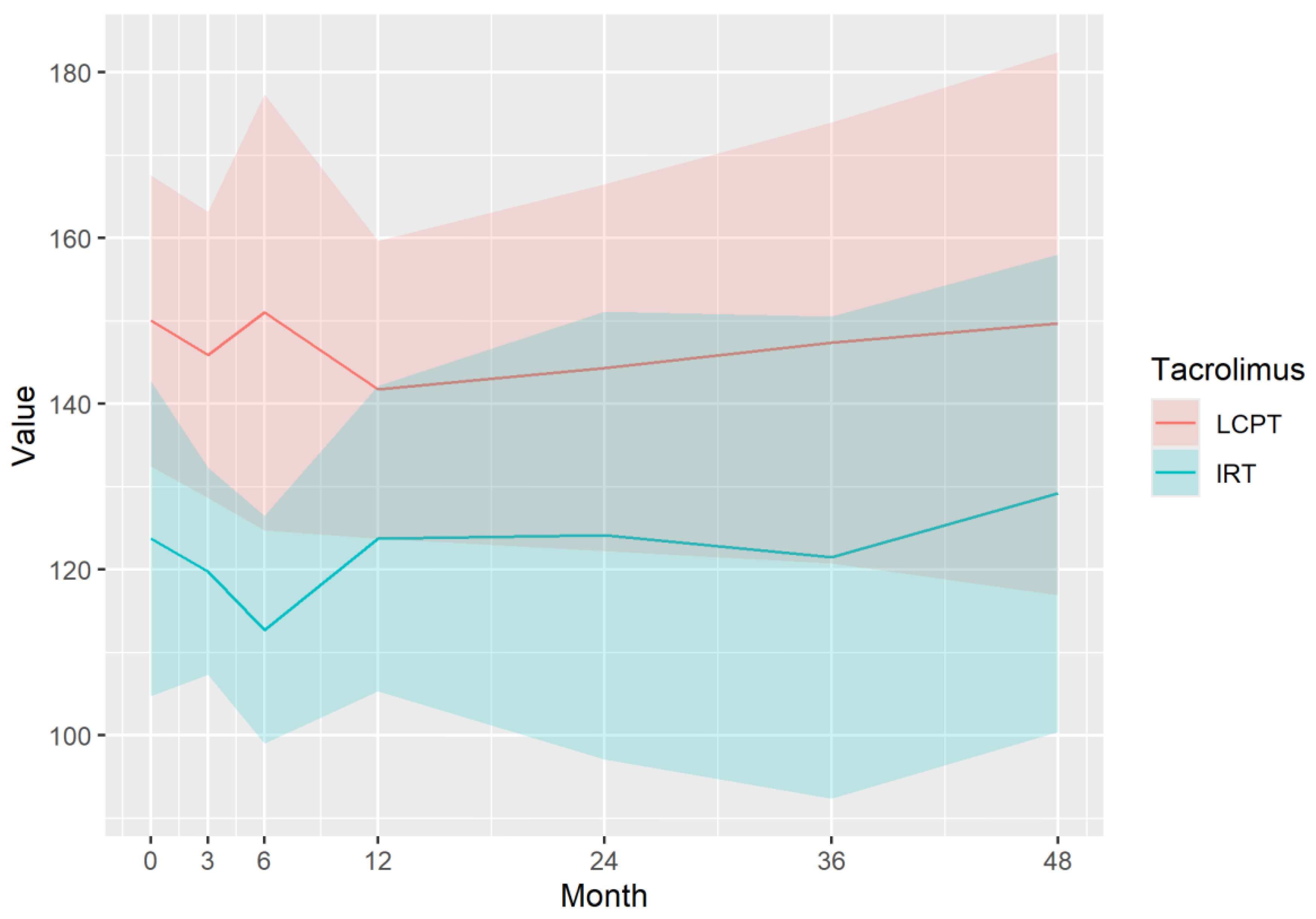
| Creatinine (µmol/l) | 138.11 (±47.46) | 148.97 (±52.24) | 119.22 (±30.34) | 0.0058 |
| Urea (mmol/l) | 9.08 (±3.3) | 9.62 (±3.58) | 8.14 (±2.55) | 0.0614 |
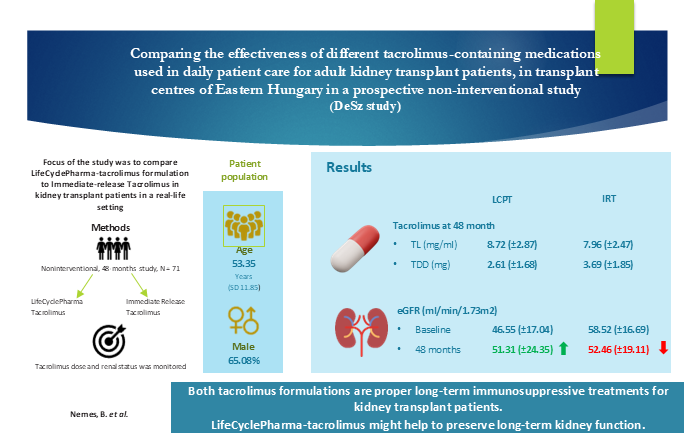
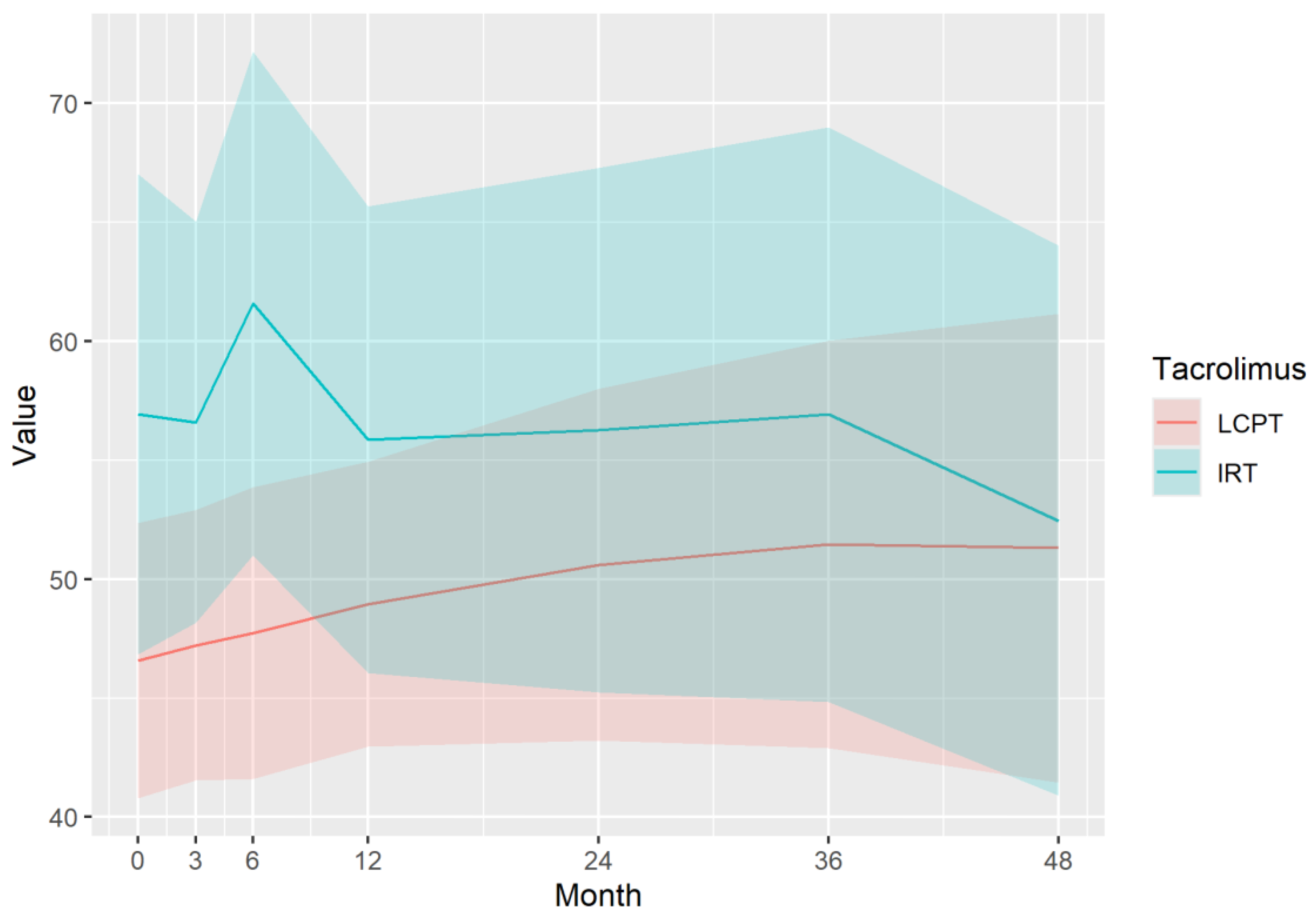
| GFR (ml/min/1.73m2) | 50.92 (±17.75) | 46.55 (±17.04) | 58.52 (±16.69) | 0.0091 |
| GOT (U/l) | 13.9 (±6.07) | 13.77 (±6.64) | 14.14 (±4.97) | 0.8067 |
| GPT (U/l) | 26.07 (±19.3) | 24.33 (±15.42) | 29.29 (±25.11) | 0.4168 |
| GGT (U/l) | 46.55 (±41.97) | 49.38 (±50.12) | 41.29 (±19.43) | 0.3763 |
| Total bilirubin (µmol/l) | 7.68 (±3.35) | 7.41 (±3.41) | 8.22 (±3.26) | 0.3796 |
| ALP (U/l) | 94.65 (±51.22) | 87.59 (±29.95) | 107.76 (±75.88) | 0.2537 |
| LDH (U/l) | 244.89 (±53.47) | 244.88 (±52.58) | 244.90 (±55.81) | 0.9988 |
| Triglyceride (mmol/l) | 2.1 (±1.14) | 2.1 (±1.25) | 2.1 (±0.93) | 0.9989 |
| Total cholesterol (mmol/l) | 5.32 (±1.28) | 5.27 (±1.31) | 5.42 (±1.25) | 0.6861 |
| LDL cholesterol (mmol/l) | 3.05 (±1.12) | 2.94 (±1.12) | 3.24 (±1.12) | 0.3478 |
| HDL cholesterol (mmol/l) | 1.63 (±0.44) | 1.64 (±0.42) | 1.60 (±0.50) | 0.7595 |
| Hgb (g/l) | 120.79 (±15.39) | 116.38 (±12.54) | 128.48 (±17.08) | 0.0053 |
| RBC (T/l) | 4.04 (±0.59) | 3.89 (±0.45) | 4.29 (±0.72) | 0.024 |
| RDV (%) | 14.86 (±1.02) | 14.84 (±0.94) | 14.91 (±1.16) | 0.8033 |
| WBC (Giga/l) | 8.22 (±2.56) | 8.04 (±2.40) | 8.53 (±2.84) | 0.4986 |
| Platelets (Giga/l) | 221.7 (±71.93) | 224.95 (±75.72) | 216.04 (±66.04) | 0.6267 |
| CRP (mg/l) | 4.2 (±8.22) | 3.08 (±3.74) | 6.15 (±12.62) | 0.2655 |
| Urine parameters | ||||
| pH | 5.7 (±0.55) | 5.7 (±0.55) | 5.7 (±0.57) | 0.9759 |
| Protein-quantitative (g/l) | 0.13 (±0.22) | 0.13 (±0.18) | 0.14 (±0.26) | 0.8338 |
| Albumin-creatine ratio (mg/mmol) | 10.89 (±21.35) | 10.38 (±18.74) | 12.17 (±27.75) | 0.8403 |
| Protein-creatinine ratio (mg/mmol) | 43.31 (±45.11) | 51.88 (±49.35) | 32.77 (±38.55) | 0.252 |
| Urine culture (positive) | 11 (17.46%) | 2 (5%) | 9 (39.13%) | 0.0005 |
| Comorbidities (yes n, %) | ||||
| Previous heart attack | 1 (1.59%) | 0 (0%) | 1 (4.35%) | 0.3503 |
| Congestive heart failure | 1 (1.59%) | 0 (0%) | 1 (4.35%) | 0.3503 |
| Peripheral vascular disease | 6 (9.52%) | 5 (12.50%) | 1 (4.35%) | 0.3913 |
| Cerebrovascular disease without residual symptoms | 8 (12.7%) | 6 (15%) | 2 (8.70%) | 0.6977 |
| Cerebrovascular disease with hemiplegia | 2 (3.17%) | 1 (2.50%) | 1 (4.35%) | 1 |
| Chronic lung disease | 8 (12.7%) | 3 (7.50%) | 5 (21.74%) | 0.1209 |
| Connective tissue disease | 1 (1.59%) | 0 (0%) | 1 (4.35%) | 0.3503 |
| Peptic ulcer | 3 (4.76%) | 2 (5.00%) | 1 (4.35%) | 1 |
| Mild liver disease | 3 (4.76%) | 1 (2.50%) | 2 (8.70%) | 0.5427 |
| Moderate or severe liver disease | 1 (1.59%) | 1 (2.50%) | 0 (0%) | 1 |
| Diabetes - no complications | 2 (3.17%) | 1 (2.50%) | 1 (4.35%) | 1 |
| Diabetes with complications | 8 (12.7%) | 4 (10%) | 4 (17.39%) | 0.4393 |
| Tumour. leucaemia or lymphoma | 1 (1.59%) | 1 (2.50%) | 0 (0%) | 1 |
| Charlson comorbidity index (points) | 2.92 (± 1.25) | 2.83 (± 1.32) | 3.09 (± 1.12) | 0.4076 |
| Transplant specific factors | ||||
| Number of kidney transplants (n, %) | 0.8186 | |||
| First kidney transplant | 55 (87.3%) | 34 (85%) | 21 (91.3%) | |
| Second kidney transplant | 7 (11.11%) | 5 (12.5%) | 2 (8.7%) | |
| Third kidney transplant | 1 (1.59%) | 1 (2.5%) | 0 (0%) | |
| Origin of transplanted organ (cadaver, %) | 62 (98.41%) | 39 (97.5%) | 23 (100%) | 1 |
| Previous member of chronic dialysis program (yes, %) | 59 (93.65%) | 38 (95.00%) | 21 (91.3%) | 0.6222 |
| Previously time spent on dialysis (months) | 44.53 (±31.84) | 34.92 (±25.44) | 61.90 (±35.35) | 0.0042 |
| Previous dialysis type (peritoneal dialysis, %) | 17 (28.81%) | 14 (36.84%) | 3 (14.29%) | 0.1257 |
| HLA-A mismatch (n, %) | ||||
| HLA-A | 0.084 | |||
| HLA-A 0 | 11 (17.46%) | 4 (10%) | 7 (30.43%) | |
| HLA-A 1 | 43 (68.25%) | 31 (77.5%) | 12 (52.17%) | |
| HLA-A 2 | 9 (14.29) | 5 (12.5%) | 4 (17.39%) | |
| HLA-B | 0.3533 | |||
| HLA-B 0 | 20 (31.75%) | 13 (32.5%) | 7 (30.43%) | |
| HLA-B 1 | 30 (47.62%) | 21 (52.5%) | 9 (39.13%) | |
| HLA-B 2 | 13 (20.63%) | 6 (15%) | 7 (30.43%) | |
| HLA-C | 0.1769 | |||
| HLA-C 0 | 24 (38.10%) | 13 (32.5%) | 11 (47.83%) | |
| HLA-C 1 | 29 (46.03%) | 22 (55%) | 7 (30.43%) | |
| HLA-C 2 | 10 (15.87%) | 5 (12.5%) | 5 (21.74%) | |
| HLA-DR | 0.1624 | |||
| HLA-DR 0 | 12 (19.05%) | 7 (17.5%) | 5 (21.74%) | |
| HLA-DR 1 | 26 (41.27%) | 20 (50%) | 6 (26.09%) | |
| HLA-DR 2 | 25 (39.68%) | 13 (32.5%) | 12 (52.17%) | |
| HLA-DQ | 0.1059 | |||
| HLA-DQ 1 | 11 (17.46%) | 7 (17.5%) | 4 (17.39%) | |
| HLA-DQ 1 | 21 (33.33%) | 17 (42.5%) | 4 (17.39%) | |
| HLA-DQ 2 | 31 (49.21%) | 16 (40%) | 15 (65.22%) | |
| Previous circulating antigen before present transplant (yes, %) | 10 (15.87%) | 7 (17.5%) | 3 (13.04%) | 0.7341 |
| Pretransplant DSA (yes, %) | 4 (7.02%) | 1 (2.7%) | 3 (15%) | 0.1184 |
| Delayed Graft Function (yes, %) | 12 (19.05%) | 8 (20%) | 4 (17.39%) | 1 |
| Baseline treatment data | ||||
| Received induction therapy for this implantation (yes, %) | 61 (96.83%) | 39 (97.5%) | 22 (95.65%) | 1 |
| Induction therapy | 0.0357 | |||
| Basiliximab | 19 (31.15%) | 8 (20.51%) | 11 (50%) | |
| ATG | 42 (68.85%) | 31 (79.49%) | 11 (50%) | |
| Tacrolimus TDD (mg) | 7.23 (± 3.3) | 6.24 (± 2.83) | 8.96 (± 3.40) | 0.0024 |
| Tacrolimus TL (ng/ml) | 11.2 (± 4.25) | 11.76 (± 4.69) | 10.22 (±3.22) | 0.1299 |
| CD: TL/TDD ratio (ng/ml)/mg | 1.91 (±1.11) | 2.25 (±1.18) | 1.30 (±0.63) | 0.0001 |
| Time between the last dose of tacrolimus and blood draw (min) | 1277.14 (±348.07) | 1520.62 (±116.22) | 853.70 (±149.11) | 0 |
| second immunosuppressant | 0.0917 | |||
| mycophenolate mofetil | 23 (36.51%) | 11 (27.5%) | 12 (52.17%) | |
| mycophenol acid | 40 (63.49%) | 29 (72.5%) | 11 (47.83%) | |
| daily dose of mycophenol (mg) | 1360.95 (±536.75) | 1253 (±444.58) | 1548.7 (±635.20) | 0.0566 |
| transplanted kidney US-RI value | 0.69 (±0.12) | 0.70 (±0.09) | 0.67 (±0.16) | 0.4688 |
| transplanted kidney abnormality on US (yes, %) | 34 (57.63%) | 19 (51.35%) | 15 (68.18%) | 0.3209 |
| transplanted kidney biopsy (yes, %) | 6 (9.52%) | 5 (12.5%) | 1 (4.35%) | 0.3988 |
| First biopsy type (indicated) | 3 (50%) | 3 (60%) | 0 (0%) | 1 |
| Adherence | ||||
| Patient agrees to complete the BAASIS questionnaire (yes, %) | 59 (93.65%) | 36 (90%) | 23 (100%) | 0.2744 |
| Missed any dose during the last 4 weeks (yes, %) | 1 (1.72%) | 0 (0%) | 1 (4.35%) | 0.3833 |
| Patient adherence self-assessment (of 100) | 99.12 (±3.26) | 99.69 (±1.18) | 98.26 (±4.91) | 0.1848 |
| Centre/arm Visits |
Timing | Debrecen centre | Szeged centre | Total | ||
|---|---|---|---|---|---|---|
| LCPT | IRT | LCPT | IRT | |||
| Visit 1 | 0 months (4-6 weeks after KT) |
26 | 23 | 14 | 0 | 63 |
| Visit 2 | 3 months | 24 | 23 | 14 | 0 | 61 |
| Visit 3 | 6 months | 23 | 21 | 14 | 0 | 58 |
| Visit 4 | 12 months | 22 | 21 | 14 | 0 | 57 |
| Visit 5 | 24 months | 20 | 18 | 12 | 0 | 50 |
| Visit 6 | 36 months | 19 | 13 | 12 | 0 | 44 |
| Visit 7 | 48 months | 14 | 13 | 12 | 0 | 39 |
| Months | 0 | 3 | 6 | 12 | 24 | 36 | 48 | |
| TDD | LCPT | 5.42 | 3.94 | 3.46 | 3.00 | 2.59 | 2.36 | 2.21 |
| IRT | 7.64 | 5.56 | 4.88 | 4.24 | 3.64 | 3.33 | 3.12 | |
| CD* | LCPT | 2.54 | 2.63 | 2.72 | 2.9 | 3.26 | 3.62 | 3.98 |
| IRT | 1.58 | 1.67 | 1.76 | 1.94 | 2.3 | 2.66 | 3.02 | |
| eGFR | LCPT | 46.99 | 47.20 | 47.41 | 47.83 | 48.67 | 49.51 | 50.35 |
| IRT | 57.89 | 57.47 | 57.05 | 56.21 | 54.53 | 52.85 | 51.17 | |
| * for patients who were not treated by mTORi | ||||||||
| In each category yellow is the lowest, and green is the highest value | ||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




