Submitted:
07 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Autophagy Dysfunction on Pathophysiology in Kidney Diseases
3.1. Autophagy and Oxidative Stress
3.2. Autophagy and Inflammation
3.3. Autophagy and Fibrosis
3.4. Autophagy and ER Stress
3.5. Molecular Mechanisms Involved in Autophagy Dysfunction in Kidney Disease
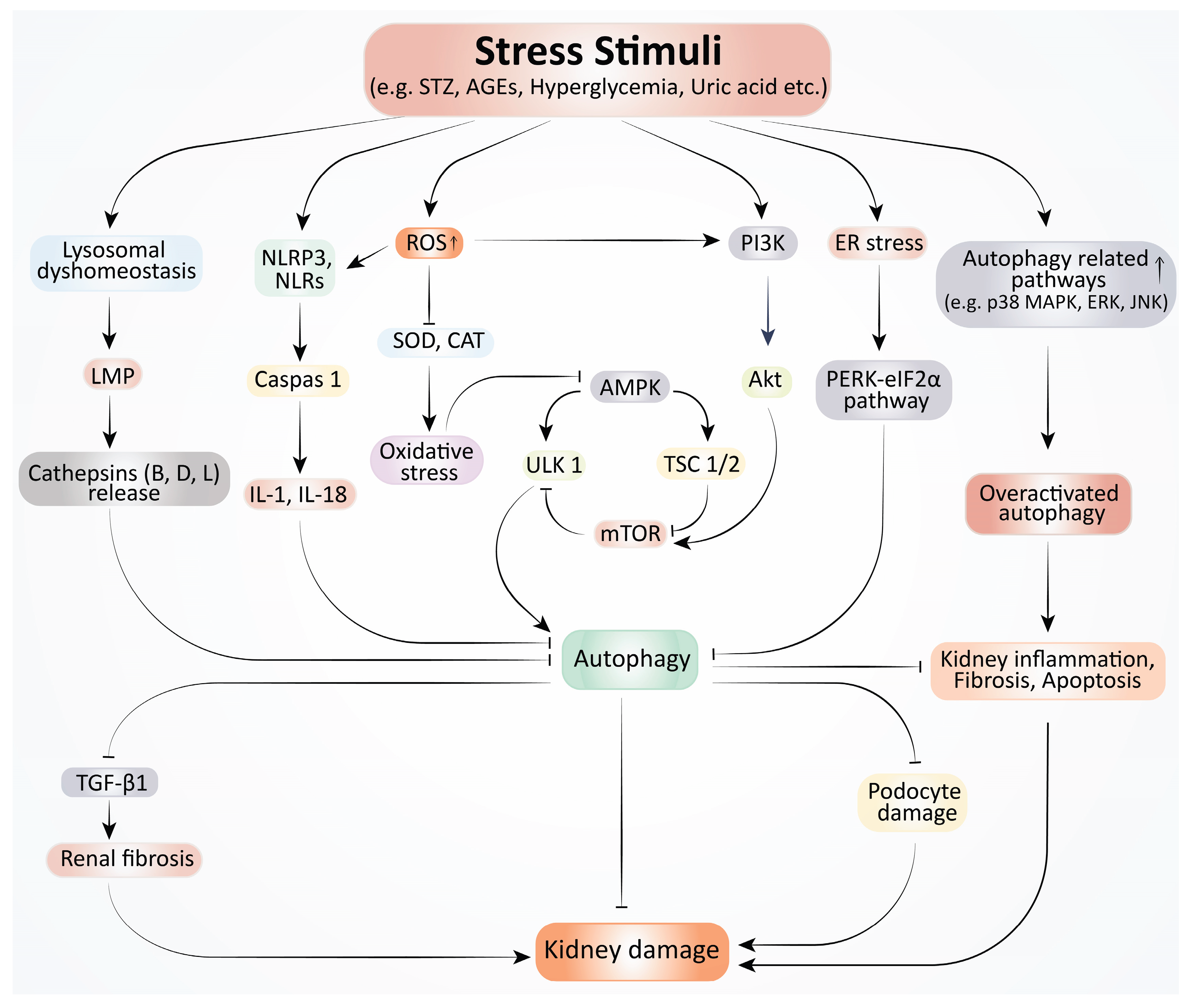
4. Therapeutic Effects of Phytochemicals against Autophagy Dysfunction in Kidney Diseases
4.1. Acute kidney Injury
4.2. Chronic Kidney Disease
4.3. Obesity-Related Nephropathy
4.4. Diabetic Nephropathy
4.5. Hypertensive Nephropathy
4.6. Obstructed Nephropathy
5. Recent Updates on Bioactive Compounds for Autophagy in Kidney Diseases
6. Prospects and Limitations
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Global, regional, and national burden of chronic kidney disease, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [CrossRef] [PubMed]
- Jager, K.J.; Kovesdy, C.; Langham, R.; Rosenberg, M.; Jha, V.; Zoccali, C. A single number for advocacy and communication-worldwide more than 850 million individuals have kidney diseases. Nephrol Dial Transplant 2019, 34, 1803–1805. [Google Scholar] [CrossRef]
- Francis, A.; Harhay, M.N.; Ong, A.C.M.; Tummalapalli, S.L.; Ortiz, A.; Fogo, A.B.; Fliser, D.; Roy-Chaudhury, P.; Fontana, M.; Nangaku, M.; et al. Chronic kidney disease and the global public health agenda: an international consensus. Nat Rev Nephrol 2024, 20, 473–485. [Google Scholar] [CrossRef]
- Sureshbabu, A.; Ryter, S.W.; Choi, M.E. Oxidative stress and autophagy: crucial modulators of kidney injury. Redox Biol 2015, 4, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, N. A brief history of autophagy from cell biology to physiology and disease. Nat Cell Biol 2018, 20, 521–527. [Google Scholar] [CrossRef]
- Levine, B.; Klionsky, D.J. Autophagy wins the 2016 Nobel Prize in Physiology or Medicine: Breakthroughs in baker's yeast fuel advances in biomedical research. Proc Natl Acad Sci U S A 2017, 114, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Levine, B.; Kroemer, G. Biological Functions of Autophagy Genes: A Disease Perspective. Cell 2019, 176, 11–42. [Google Scholar] [CrossRef] [PubMed]
- Leidal, A.M.; Levine, B.; Debnath, J. Autophagy and the cell biology of age-related disease. Nat Cell Biol 2018, 20, 1338–1348. [Google Scholar] [CrossRef] [PubMed]
- Ravikumar, B.; Sarkar, S.; Davies, J.E.; Futter, M.; Garcia-Arencibia, M.; Green-Thompson, Z.W.; Jimenez-Sanchez, M.; Korolchuk, V.I.; Lichtenberg, M.; Luo, S.; et al. Regulation of mammalian autophagy in physiology and pathophysiology. Physiol Rev 2010, 90, 1383–1435. [Google Scholar] [CrossRef] [PubMed]
- Levine, B.; Kroemer, G. Autophagy in the pathogenesis of disease. Cell 2008, 132, 27–42. [Google Scholar] [CrossRef]
- Tang, C.; Livingston, M.J.; Liu, Z.; Dong, Z. Autophagy in kidney homeostasis and disease. Nat Rev Nephrol 2020, 16, 489–508. [Google Scholar] [CrossRef]
- Sohn, M.; Kim, K.; Uddin, M.J.; Lee, G.; Hwang, I.; Kang, H.; Kim, H.; Lee, J.H.; Ha, H. Delayed treatment with fenofibrate protects against high-fat diet-induced kidney injury in mice: the possible role of AMPK autophagy. American journal of physiology. Renal physiology 2017, 312, F323–f334. [Google Scholar] [CrossRef]
- Pandiri, I.; Biotechnology, A.M.J.J.o.A.; Therapeutics, E. Ocimum herb species: a potential treatment strategy for diabetic kidney disease. 2018. [Google Scholar] [CrossRef]
- Rahman, M.A.; Hannan, M.A.; Dash, R.; Rahman, M.H.; Islam, R.; Uddin, M.J.; Sohag, A.A.M.; Rahman, M.H.; Rhim, H. Phytochemicals as a Complement to Cancer Chemotherapy: Pharmacological Modulation of the Autophagy-Apoptosis Pathway. 2021, 12–2021. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.; Ghosh, S.; Das, A.K.; Sil, P.C. Ferulic Acid Protects Hyperglycemia-Induced Kidney Damage by Regulating Oxidative Insult, Inflammation and Autophagy. Front Pharmacol 2019, 10, 27. [Google Scholar] [CrossRef]
- Liu, P.; Zhu, W.; Wang, Y.; Ma, G.; Zhao, H.; Li, P. Chinese herbal medicine and its active compounds in attenuating renal injury via regulating autophagy in diabetic kidney disease. Front Endocrinol (Lausanne) 2023, 14, 1142805. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, Q.; Wang, C.; Lou, Z.; Li, Q. Trigonelline reduced diabetic nephropathy and insulin resistance in type 2 diabetic rats through peroxisome proliferator-activated receptor-gamma. Exp Ther Med 2019, 18, 1331–1337. [Google Scholar] [CrossRef]
- Putra, I.; Fakhrudin, N.; Nurrochmad, A.; Wahyuono, S. A Review of Medicinal Plants with Renoprotective Activity in Diabetic Nephropathy Animal Models. Life (Basel) 2023, 13. [Google Scholar] [CrossRef]
- Rahman, M.A.; Akter, S.; Dorotea, D.; Mazumder, A.; Uddin, M.N.; Hannan, M.A.; Hossen, M.J.; Ahmed, M.S.; Kim, W.; Kim, B.; et al. Renoprotective potentials of small molecule natural products targeting mitochondrial dysfunction. 2022, 13–2022. [Google Scholar] [CrossRef]
- Uddin, M.J.; Kim, E.H.; Hannan, M.A.; Ha, H. Pharmacotherapy against Oxidative Stress in Chronic Kidney Disease: Promising Small Molecule Natural Products Targeting Nrf2-HO-1 Signaling. Antioxidants (Basel) 2021, 10. [Google Scholar] [CrossRef]
- Kaushal, G.P.; Chandrashekar, K.; Juncos, L.A. Molecular Interactions Between Reactive Oxygen Species and Autophagy in Kidney Disease. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef]
- Sridhar, S.; Botbol, Y.; Macian, F.; Cuervo, A.M. Autophagy and disease: always two sides to a problem. J Pathol 2012, 226, 255–273. [Google Scholar] [CrossRef]
- Wible, D.J.; Bratton, S.B. Reciprocity in ROS and autophagic signaling. Curr Opin Toxicol 2018, 7, 28–36. [Google Scholar] [CrossRef]
- Lin, T.A.; Wu, V.C.; Wang, C.Y. Autophagy in Chronic Kidney Diseases. Cells 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Ai, S.; Li, Y.; Zheng, H.; Wang, Z.; Liu, W.; Tao, J.; Li, Y.; Wang, Y. Global research trends and hot spots on autophagy and kidney diseases: a bibliometric analysis from 2000 to 2022. Front Pharmacol 2023, 14, 1275792. [Google Scholar] [CrossRef]
- Anders, H.J.; Schaefer, L. Beyond tissue injury-damage-associated molecular patterns, toll-like receptors, and inflammasomes also drive regeneration and fibrosis. J Am Soc Nephrol 2014, 25, 1387–1400. [Google Scholar] [CrossRef]
- Kinsey, G.R.; Li, L.; Okusa, M.D. Inflammation in acute kidney injury. Nephron Exp Nephrol 2008, 109, e102-107. [Google Scholar] [CrossRef] [PubMed]
- Akchurin, O.M.; Kaskel, F. Update on inflammation in chronic kidney disease. Blood Purif 2015, 39, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Pahwa, R.; Goyal, A.; Jialal, I. Chronic Inflammation. In StatPearls; StatPearls Publishing.
- Copyright © 2025. StatPearls Publishing LLC.: Treasure Island (FL), 2025.
- Banerjee, P.; Basu, A.; Wegiel, B.; Otterbein, L.E.; Mizumura, K.; Gasser, M.; Waaga-Gasser, A.M.; Choi, A.M.; Pal, S. Heme oxygenase-1 promotes survival of renal cancer cells through modulation of apoptosis- and autophagy-regulating molecules. J Biol Chem 2012, 287, 32113–32123. [Google Scholar] [CrossRef]
- Ding, Y.; Fu, X.; Wang, Q.; Liu, H.; Wang, H.; Wu, D. The Complex Interplay between Autophagy and NLRP3 Inflammasome in Renal Diseases. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Kim, J.W.; Nam, S.A.; Koh, E.S.; Kim, H.W.; Kim, S.; Woo, J.J.; Kim, Y.K. The Impairment of Endothelial Autophagy Accelerates Renal Senescence by Ferroptosis and NLRP3 Inflammasome Signaling Pathways with the Disruption of Endothelial Barrier. Antioxidants (Basel) 2024, 13. [Google Scholar] [CrossRef]
- Jianbing, H.; Xiaotian, L.; Jie, T.; Xueying, C.; Honge, J.; Bo, Z.; Lirong, H.; Lei, Z. The Effect of Allograft Inflammatory Factor-1 on Inflammation, Oxidative Stress, and Autophagy via miR-34a/ATG4B Pathway in Diabetic Kidney Disease. Oxid Med Cell Longev 2022, 2022, 1668000. [Google Scholar] [CrossRef]
- Kato, H.; Gruenwald, A.; Suh, J.H.; Miner, J.H.; Barisoni-Thomas, L.; Taketo, M.M.; Faul, C.; Millar, S.E.; Holzman, L.B.; Susztak, K. Wnt/β-catenin pathway in podocytes integrates cell adhesion, differentiation, and survival. Journal of Biological Chemistry 2011, 286, 26003–26015. [Google Scholar] [CrossRef]
- Fabian, S.L.; Penchev, R.R.; St-Jacques, B.; Rao, A.N.; Sipilä, P.; West, K.A.; McMahon, A.P.; Humphreys, B.D. Hedgehog-Gli pathway activation during kidney fibrosis. The American journal of pathology 2012, 180, 1441–1453. [Google Scholar] [CrossRef] [PubMed]
- White, E.S.; Mantovani, A.R. Inflammation, wound repair, and fibrosis: reassessing the spectrum of tissue injury and resolution. J Pathol 2013, 229, 141–144. [Google Scholar] [CrossRef]
- Kuppe, C.; Ibrahim, M.M.; Kranz, J.; Zhang, X.; Ziegler, S.; Perales-Patón, J.; Jansen, J.; Reimer, K.C.; Smith, J.R.; Dobie, R. Decoding myofibroblast origins in human kidney fibrosis. Nature 2021, 589, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Yanagita, M. Resident fibroblasts in the kidney: a major driver of fibrosis and inflammation. Inflammation and regeneration 2017, 37, 1–7. [Google Scholar] [CrossRef]
- Ferenbach, D.A.; Bonventre, J.V. Mechanisms of maladaptive repair after AKI leading to accelerated kidney ageing and CKD. Nature Reviews Nephrology 2015, 11, 264–276. [Google Scholar] [CrossRef] [PubMed]
- Venkatachalam, M.A.; Weinberg, J.M.; Kriz, W.; Bidani, A.K. Failed tubule recovery, AKI-CKD transition, and kidney disease progression. Journal of the American Society of Nephrology 2015, 26, 1765–1776. [Google Scholar] [CrossRef]
- Basile, D.P.; Bonventre, J.V.; Mehta, R.; Nangaku, M.; Unwin, R.; Rosner, M.H.; Kellum, J.A.; Ronco, C. Progression after AKI: understanding maladaptive repair processes to predict and identify therapeutic treatments. Journal of the American Society of Nephrology 2016, 27, 687–697. [Google Scholar] [CrossRef]
- Kumar, S. Cellular and molecular pathways of renal repair after acute kidney injury. Kidney international 2018, 93, 27–40. [Google Scholar] [CrossRef]
- Humphreys, B.D. Mechanisms of renal fibrosis. Annual review of physiology 2018, 80, 309–326. [Google Scholar] [CrossRef]
- Podestà, M.A.; Faravelli, I.; Ponticelli, C. Autophagy in lupus nephritis: a delicate balance between regulation and disease. Autoimmunity Reviews 2022, 21, 103132. [Google Scholar] [CrossRef]
- KIM, W.Y.; Nam, S.A.; Song, H.C.; Ko, J.S.; Park, S.H.; Kim, H.L.; Choi, E.J.; KIM, Y.S.; Kim, J.; Kim, Y.K. The role of autophagy in unilateral ureteral obstruction rat model. Nephrology 2012, 17, 148–159. [Google Scholar] [CrossRef]
- Ma, Z.; Li, L.; Livingston, M.J.; Zhang, D.; Mi, Q.; Zhang, M.; Ding, H.-F.; Huo, Y.; Mei, C.; Dong, Z. p53/microRNA-214/ULK1 axis impairs renal tubular autophagy in diabetic kidney disease. The Journal of clinical investigation 2020, 130, 5011–5026. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Song, Y.; Zhang, L.; Chen, Z.; Yang, C.; Liu, S.; Yuan, X.; Gao, H.; Ding, G.; Wang, H. Autophagy activation contributes to lipid accumulation in tubular epithelial cells during kidney fibrosis. Cell death discovery 2018, 4, 39. [Google Scholar] [CrossRef]
- Fu, Y.; Xiang, Y.; Wu, W.; Cai, J.; Tang, C.; Dong, Z. Persistent activation of autophagy after cisplatin nephrotoxicity promotes renal fibrosis and chronic kidney disease. Frontiers in Pharmacology 2022, 13, 918732. [Google Scholar] [CrossRef]
- Livingston, M.J.; Ding, H.-F.; Huang, S.; Hill, J.A.; Yin, X.-M.; Dong, Z. Persistent activation of autophagy in kidney tubular cells promotes renal interstitial fibrosis during unilateral ureteral obstruction. Autophagy 2016, 12, 976–998. [Google Scholar] [CrossRef] [PubMed]
- Gui, Z.; Suo, C.; Wang, Z.; Zheng, M.; Fei, S.; Chen, H.; Sun, L.; Han, Z.; Tao, J.; Ju, X. Impaired ATG16L-dependent autophagy promotes renal interstitial fibrosis in chronic renal graft dysfunction through inducing EndMT by NF-κB signal pathway. Frontiers in Immunology 2021, 12, 650424. [Google Scholar] [CrossRef] [PubMed]
- Hetz, C.; Zhang, K.; Kaufman, R.J. Mechanisms, regulation and functions of the unfolded protein response. Nature reviews Molecular cell biology 2020, 21, 421–438. [Google Scholar] [CrossRef]
- Wang, M.; Chen, Z.; Tang, Z.; Tang, S. Natural products derived from traditional Chinese medicines targeting ER stress for the treatment of kidney diseases. Renal Failure 2024, 46, 2396446. [Google Scholar] [CrossRef]
- Hasegawa, S.; Inagi, R. Organelle stress and crosstalk in kidney disease. Kidney360 2020, 1, 1157–1164. [Google Scholar] [CrossRef]
- Fan, Y.; Xiao, W.; Lee, K.; Salem, F.; Wen, J.; He, L.; Zhang, J.; Fei, Y.; Cheng, D.; Bao, H. Inhibition of reticulon-1a–mediated endoplasmic reticulum stress in early AKI attenuates renal fibrosis development. Journal of the American Society of Nephrology 2017, 28, 2007–2021. [Google Scholar] [CrossRef] [PubMed]
- Tavernier, Q.; Mami, I.; Rabant, M.; Karras, A.; Laurent-Puig, P.; Chevet, E.; Thervet, E.; Anglicheau, D.; Pallet, N. Urinary angiogenin reflects the magnitude of kidney injury at the infrahistologic level. Journal of the American Society of Nephrology 2017, 28, 678–690. [Google Scholar] [CrossRef]
- Yan, M.; Shu, S.; Guo, C.; Tang, C.; Dong, Z. Endoplasmic reticulum stress in ischemic and nephrotoxic acute kidney injury. Annals of medicine 2018, 50, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Matsui, T. Molecular mechanisms of macroautophagy, microautophagy, and chaperone-mediated autophagy. Journal of Nippon Medical School 2024, 91, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Aman, Y.; Schmauck-Medina, T.; Hansen, M.; Morimoto, R.I.; Simon, A.K.; Bjedov, I.; Palikaras, K.; Simonsen, A.; Johansen, T.; Tavernarakis, N. Autophagy in healthy aging and disease. Nature aging 2021, 1, 634–650. [Google Scholar] [CrossRef]
- Liu, F.; Zhao, L.; Wu, T.; Yu, W.; Li, J.; Wang, W.; Huang, C.; Diao, Z.; Xu, Y. Targeting autophagy with natural products as a potential therapeutic approach for diabetic microangiopathy. Frontiers in Pharmacology 2024, 15, 1364616. [Google Scholar] [CrossRef]
- Liu, J.; Livingston, M.J.; Dong, G.; Tang, C.; Su, Y.; Wu, G.; Yin, X.-M.; Dong, Z. Histone deacetylase inhibitors protect against cisplatin-induced acute kidney injury by activating autophagy in proximal tubular cells. Cell death & disease 2018, 9, 322. [Google Scholar]
- Tang, C.; Han, H.; Yan, M.; Zhu, S.; Liu, J.; Liu, Z.; He, L.; Tan, J.; Liu, Y.; Liu, H. PINK1-PRKN/PARK2 pathway of mitophagy is activated to protect against renal ischemia-reperfusion injury. Autophagy 2018, 14, 880–897. [Google Scholar] [CrossRef]
- Kaufman, D.R.; Papillon, J.; Larose, L.; Iwawaki, T.; Cybulsky, A.V. Deletion of inositol-requiring enzyme-1α in podocytes disrupts glomerular capillary integrity and autophagy. Molecular biology of the cell 2017, 28, 1636–1651. [Google Scholar] [CrossRef]
- Rashid, H.-O.; Yadav, R.K.; Kim, H.-R.; Chae, H.-J. ER stress: Autophagy induction, inhibition and selection. Autophagy 2015, 11, 1956–1977. [Google Scholar] [CrossRef] [PubMed]
- Hartleben, B.; Gödel, M.; Meyer-Schwesinger, C.; Liu, S.; Ulrich, T.; Köbler, S.; Wiech, T.; Grahammer, F.; Arnold, S.J.; Lindenmeyer, M.T. Autophagy influences glomerular disease susceptibility and maintains podocyte homeostasis in aging mice. The Journal of clinical investigation 2010, 120, 1084–1096. [Google Scholar] [CrossRef] [PubMed]
- Cybulsky, A.V. The intersecting roles of endoplasmic reticulum stress, ubiquitin–proteasome system, and autophagy in the pathogenesis of proteinuric kidney disease. Kidney international 2013, 84, 25–33. [Google Scholar] [CrossRef]
- Ruby, M.; Gifford, C.C.; Pandey, R.; Raj, V.S.; Sabbisetti, V.S.; Ajay, A.K. Autophagy as a Therapeutic Target for Chronic Kidney Disease and the Roles of TGF-β1 in Autophagy and Kidney Fibrosis. Cells 2023, 12. [Google Scholar] [CrossRef]
- Zhang, Z.; Sun, Y.; Xue, J.; Jin, D.; Li, X.; Zhao, D.; Lian, F.; Qi, W.; Tong, X. The critical role of dysregulated autophagy in the progression of diabetic kidney disease. Front Pharmacol 2022, 13, 977410. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Ma, Y.; Chen, X.; Liang, N.; Qu, S.; Chen, H. Hyperuricemia causes kidney damage by promoting autophagy and NLRP3-mediated inflammation in rats with urate oxidase deficiency. Dis Model Mech 2021, 14. [Google Scholar] [CrossRef]
- Wu, M.; Zhang, M.; Zhang, Y.; Li, Z.; Li, X.; Liu, Z.; Liu, H.; Li, X. Relationship between lysosomal dyshomeostasis and progression of diabetic kidney disease. Cell Death Dis 2021, 12, 958. [Google Scholar] [CrossRef]
- Yuan, Q.; Tang, B.; Zhang, C. Signaling pathways of chronic kidney diseases, implications for therapeutics. Signal Transduct Target Ther 2022, 7, 182. [Google Scholar] [CrossRef]
- Wang, H.; Gao, L.; Zhao, C.; Fang, F.; Liu, J.; Wang, Z.; Zhong, Y.; Wang, X. The role of PI3K/Akt signaling pathway in chronic kidney disease. Int Urol Nephrol 2024, 56, 2623–2633. [Google Scholar] [CrossRef]
- Miricescu, D.; Balan, D.G.; Tulin, A.; Stiru, O.; Vacaroiu, I.A.; Mihai, D.A.; Popa, C.C.; Papacocea, R.I.; Enyedi, M.; Sorin, N.A.; et al. PI3K/AKT/mTOR signalling pathway involvement in renal cell carcinoma pathogenesis (Review). Exp Ther Med 2021, 21, 540. [Google Scholar] [CrossRef]
- Muratsubaki, S.; Kuno, A.; Tanno, M.; Miki, T.; Yano, T.; Sugawara, H.; Shibata, S.; Abe, K.; Ishikawa, S.; Ohno, K.; et al. Suppressed autophagic response underlies augmentation of renal ischemia/reperfusion injury by type 2 diabetes. Scientific Reports 2017, 7, 5311. [Google Scholar] [CrossRef]
- R, H.; Y, X.; W, W.; X, S.; J, Q.; Z, Y.; B, L.; C, C.; T, Z.; J, L.-S.; et al. Deacetylation of nuclear LC3 drives autophagy initiation under starvation - PubMed. Molecular cell 02/05/2015, 57. [Google Scholar] [CrossRef]
- Sun, M.; Li, J.; Mao, L.; Wu, J.; Deng, Z.; He, M.; An, S.; Zeng, Z.; Huang, Q.; Chen, Z. p53 Deacetylation Alleviates Sepsis-Induced Acute Kidney Injury by Promoting Autophagy. Front Immunol 2021, 12, 685523. [Google Scholar] [CrossRef]
- Deng, J.K.; Zhang, X.; Wu, H.L.; Gan, Y.; Ye, L.; Zheng, H.; Zhu, Z.; Liu, W.J.; Liu, H.F. ROS-ERK Pathway as Dual Mediators of Cellular Injury and Autophagy-Associated Adaptive Response in Urinary Protein-Irritated Renal Tubular Epithelial Cells. J Diabetes Res 2021, 2021, 6614848. [Google Scholar] [CrossRef]
- Yang, J.; Yuan, L.; Liu, F.; Li, L.; Liu, J.; Chen, Y.; Lu, Y.; Yuan, Y. Molecular mechanisms and physiological functions of autophagy in kidney diseases. Front Pharmacol 2022, 13, 974829. [Google Scholar] [CrossRef]
- Wang, T.; Liu, C.; Jia, L. The roles of PKCs in regulating autophagy. J Cancer Res Clin Oncol 2018, 144, 2303–2311. [Google Scholar] [CrossRef]
- Sanz, A.B.; Sanchez-Niño, M.D.; Ramos, A.M.; Ortiz, A. Regulated cell death pathways in kidney disease. Nat Rev Nephrol 2023, 19, 281–299. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Mou, L.; Yi, Z.; Lin, Q.; Banu, K.; Wei, C.; Yu, X. Integrative informatics analysis identifies that ginsenoside Re improves renal fibrosis through regulation of autophagy. J Nat Med 2024, 78, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Jin, Q.; Yang, L.; Mao, H.; Ma, F.; Wang, Y.; Li, P.; Zhan, Y. Regulation of autophagy by natural polyphenols in the treatment of diabetic kidney disease: therapeutic potential and mechanism. Front Endocrinol (Lausanne) 2023, 14, 1142276. [Google Scholar] [CrossRef]
- Jia, Z.; Wang, K.; Zhang, Y.; Duan, Y.; Xiao, K.; Liu, S.; Ding, X. Icariin Ameliorates Diabetic Renal Tubulointerstitial Fibrosis by Restoring Autophagy via Regulation of the miR-192-5p/GLP-1R Pathway. Front Pharmacol 2021, 12, 720387. [Google Scholar] [CrossRef]
- Matboli, M.; Ibrahim, D.; Hasanin, A.H.; Hassan, M.K.; Habib, E.K.; Bekhet, M.M.; Afifi, A.M.; Eissa, S. Epigenetic modulation of autophagy genes linked to diabetic nephropathy by administration of isorhamnetin in Type 2 diabetes mellitus rats. Epigenomics 2021, 13, 187–202. [Google Scholar] [CrossRef]
- Ou, X.; Yu, Z.; Pan, C.; Zheng, X.; Li, D.; Qiao, Z.; Zheng, X. Paeoniflorin: a review of its pharmacology, pharmacokinetics and toxicity in diabetes. Frontiers in Pharmacology 2025, 16–2025. [Google Scholar] [CrossRef]
- Zhang, X.X.; Jiang, C.H.; Liu, Y.; Lou, D.X.; Huang, Y.P.; Gao, M.; Zhang, J.; Yin, Z.Q.; Pan, K. Cyclocarya paliurus triterpenic acids fraction attenuates kidney injury via AMPK-mTOR-regulated autophagy pathway in diabetic rats. Phytomedicine 2019, 64, 153060. [Google Scholar] [CrossRef]
- Teh, Y.M.; Mualif, S.A.; Mohd Noh, N.I.; Lim, S.K. The Potential of Naturally Derived Compounds for Treating Chronic Kidney Disease: A Review of Autophagy and Cellular Senescence. Int J Mol Sci 2024, 26. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhang, H.; Lv, S.; Zhu, C.; Gong, S.; Yu, X.; Wang, Y.; Huang, X.; Yuan, S.; Ding, X.; et al. Sulforaphane alleviates renal fibrosis through dual regulation on mTOR-mediated autophagy pathway. Int Urol Nephrol 2025, 57, 1277–1287. [Google Scholar] [CrossRef] [PubMed]
- Akaras, N.; Gur, C.; Kucukler, S.; Kandemir, F.M. Zingerone reduces sodium arsenite-induced nephrotoxicity by regulating oxidative stress, inflammation, apoptosis and histopathological changes. Chem Biol Interact 2023, 374, 110410. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Fan, Q.; Wang, X.; Li, L.; Lu, X.; Yue, Y.; Cao, X.; Liu, J.; Zhao, X.; Wang, L. Ursolic acid improves podocyte injury caused by high glucose. Nephrol Dial Transplant 2017, 32, 1285–1293. [Google Scholar] [CrossRef]
- Turgut, F.; Awad, A.S.; Abdel-Rahman, E.M. Acute Kidney Injury: Medical Causes and Pathogenesis. J Clin Med 2023, 12. [Google Scholar] [CrossRef]
- Zeng, Y.; Li, S.; Wu, J.; Chen, W.; Sun, H.; Peng, W.; Yu, X.; Yang, X. Autophagy inhibitors promoted aristolochic acid I induced renal tubular epithelial cell apoptosis via mitochondrial pathway but alleviated nonapoptotic cell death in mouse acute aritolochic acid nephropathy model. Apoptosis 2014, 19, 1215–1224. [Google Scholar] [CrossRef]
- Tan, X.; Zhu, H.; Tao, Q.; Guo, L.; Jiang, T.; Xu, L.; Yang, R.; Wei, X.; Wu, J.; Li, X.; et al. FGF10 Protects Against Renal Ischemia/Reperfusion Injury by Regulating Autophagy and Inflammatory Signaling. Front Genet 2018, 9, 556. [Google Scholar] [CrossRef]
- Kaushal, G.P.; Shah, S.V. Autophagy in acute kidney injury. Kidney international 2016, 89, 779–791. [Google Scholar] [CrossRef]
- Djavaheri-Mergny, M.; Amelotti, M.; Mathieu, J.; Besançon, F.; Bauvy, C.; Codogno, P. Regulation of autophagy by NFkappaB transcription factor and reactives oxygen species. Autophagy 2007, 3, 390–392. [Google Scholar] [CrossRef] [PubMed]
- Vijayan, A. Tackling AKI: prevention, timing of dialysis and follow-up. Nature Reviews Nephrology 2020, 17. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wang, Q.; Li, T.; Li, L.; Wang, C.; Xu, B.; Gong, X. Exploring therapeutic mechanisms of Chuan Huang Fang-II in the treatment of acute kidney injury on chronic kidney disease patients from the perspective of lipidomics. Ren Fail 2024, 46, 2356021. [Google Scholar] [CrossRef]
- Shi, Y.; Shi, X.; Zhao, M.; Chang, M.; Ma, S.; Zhang, Y. Ferroptosis: A new mechanism of traditional Chinese medicine compounds for treating acute kidney injury. Biomed Pharmacother 2023, 163, 114849. [Google Scholar] [CrossRef]
- Qi, J.; Xue, Q.; Kuang, L.; Xie, L.; Luo, R.; Nie, X. Berberine alleviates cisplatin-induced acute kidney injury by regulating mitophagy via PINK 1/Parkin pathway. Transl Androl Urol 2020, 9, 1712–1724. [Google Scholar] [CrossRef]
- Gong, X.; Duan, Y.; Zheng, J.; Ye, Z.; Hei, T.K. Tetramethylpyrazine Prevents Contrast-Induced Nephropathy via Modulating Tubular Cell Mitophagy and Suppressing Mitochondrial Fragmentation, CCL2/CCR2-Mediated Inflammation, and Intestinal Injury. Oxid Med Cell Longev 2019, 2019, 7096912. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Xu, B.; Sun, Y.; Lian, M.; Li, Y.; Lin, Y.; Chen, D.; Diao, Y.; Almoiliqy, M.; Wang, L. Paeoniflorin protects against intestinal ischemia/reperfusion by activating LKB1/AMPK and promoting autophagy. Pharmacol Res 2019, 146, 104308. [Google Scholar] [CrossRef]
- Li, H.; Tang, Y.; Wen, L.; Kong, X.; Chen, X.; Liu, P.; Zhou, Z.; Chen, W.; Xiao, C.; Xiao, P.; et al. Neferine reduces cisplatin-induced nephrotoxicity by enhancing autophagy via the AMPK/mTOR signaling pathway. Biochem Biophys Res Commun 2017, 484, 694–701. [Google Scholar] [CrossRef]
- Zuo, Z.; Li, Q.; Zhou, S.; Yu, R.; Wu, C.; Chen, J.; Xiao, Y.; Chen, H.; Song, J.; Pan, Y.; et al. Berberine ameliorates contrast-induced acute kidney injury by regulating HDAC4-FoxO3a axis-induced autophagy: In vivo and in vitro. Phytother Res 2024, 38, 1761–1780. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Jafar, T.H.; Nitsch, D.; Neuen, B.L.; Perkovic, V. Chronic kidney disease. The lancet 2021, 398, 786–802. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.; Okpechi, I.G.; Caskey, F.J.; Yang, C.-W.; Tonelli, M.; Jha, V. Perspectives on early detection of chronic kidney disease: the facts, the questions, and a proposed framework for 2023 and beyond. Kidney International 2023, 103, 1004–1008. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Choi, M.E. Autophagy in diabetic nephropathy. The Journal of endocrinology 2014, 224, R15. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.-A.; Wu, V.C.-C.; Wang, C.-Y. Autophagy in chronic kidney diseases. Cells 2019, 8, 61. [Google Scholar] [CrossRef]
- Periyasamy-Thandavan, S.; Jiang, M.; Schoenlein, P.; Dong, Z. Autophagy: molecular machinery, regulation, and implications for renal pathophysiology. American Journal of Physiology-Renal Physiology 2009, 297, F244–F256. [Google Scholar] [CrossRef]
- Di Tu, Q.; Jin, J.; Hu, X.; Ren, Y.; Zhao, L.; He, Q. Curcumin improves the renal autophagy in rat experimental membranous nephropathy via regulating the PI3K/AKT/mTOR and Nrf2/HO-1 signaling pathways. BioMed research international 2020, 2020, 7069052. [Google Scholar] [CrossRef]
- Liu, X.; Chen, J.; Sun, N.; Li, N.; Zhang, Z.; Zheng, T.; Li, Z. Ginsenoside Rb1 ameliorates autophagy via the AMPK/mTOR pathway in renal tubular epithelial cells in vitro and in vivo. International Journal of Biological Macromolecules 2020, 163, 996–1009. [Google Scholar] [CrossRef]
- Lu, R.; Chen, J.; Liu, B.; Lin, H.; Bai, L.; Zhang, P.; Chen, D.; Li, H.; Li, J.; Pang, Y. Protective role of Astragaloside IV in chronic glomerulonephritis by activating autophagy through PI3K/AKT/AS160 pathway. Phytotherapy Research 2020, 34, 3236–3248. [Google Scholar] [CrossRef]
- Tseng, C.-Y.; Yu, P.-R.; Hsu, C.-C.; Lin, H.-H.; Chen, J.-H. The effect of isovitexin on lipopolysaccharide-induced renal injury and inflammation by induction of protective autophagy. Food and Chemical Toxicology 2023, 172, 113581. [Google Scholar] [CrossRef]
- Yang, F.; Gao, Y.; Xie, S.; Yang, W.; Wang, Q.; Ye, W.; Sun, L.; Zhou, J.; Feng, X. Dietary phytosterol supplementation mitigates renal fibrosis via activating mitophagy and modulating the gut microbiota. Food & Function 2025, 16, 2316–2334. [Google Scholar] [CrossRef]
- Fan, L.; Chen, H.-h.; Liu, H.-j.; Chen, H.-j.; Zhu, L.-l.; Zhang, T. Experimental study on danggui shaoyao san improving renal fibrosis by promoting autophagy. Evidence-Based Complementary and Alternative Medicine 2022, 2022, 6761453. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.-J.; Lee, Y.-H.; Yeh, Y.-L.; Wu, W.-S.; Ho, C.-T.; Li, C.-Y.; Wang, B.-J.; Wang, Y.-J. Autophagy-inducing effect of pterostilbene: A prospective therapeutic/preventive option for skin diseases. journal of food and drug analysis 2017, 25, 125–133. [Google Scholar] [CrossRef]
- Zhang, D.; Zhang, H.; Lv, S.; Zhu, C.; Gong, S.; Yu, X.; Wang, Y.; Huang, X.; Yuan, S.; Ding, X. Sulforaphane alleviates renal fibrosis through dual regulation on mTOR-mediated autophagy pathway. International Urology and Nephrology 2025, 57, 1277–1287. [Google Scholar] [CrossRef]
- Tu, Y.; Gu, L.; Chen, D.; Wu, W.; Liu, H.; Hu, H.; Wan, Y.; Sun, W. Rhein inhibits autophagy in rat renal tubular cells by regulation of AMPK/mTOR signaling. Scientific reports 2017, 7, 43790. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Qin, X.; Xu, W.; Zhang, H.; Huang, S.; Yang, Y.; Qin, M.; Mi, Z.; Zhong, X. Herb pair of Rhubarb-Astragalus mitigates renal interstitial fibrosis through downregulation of autophagy via p38-MAPK/TGF-β1 and p38-MAPK/smad2/3 pathways. The International Journal of Biochemistry & Cell Biology 2024, 169, 106549. [Google Scholar]
- Kounatidis, D.; Vallianou, N.G.; Stratigou, T.; Voukali, M.; Karampela, I.; Dalamaga, M. The Kidney in Obesity: Current Evidence, Perspectives and Controversies. Curr Obes Rep 2024, 13, 680–702. [Google Scholar] [CrossRef]
- Martínez-Montoro, J.I.; Morales, E.; Cornejo-Pareja, I.; Tinahones, F.J.; Fernández-García, J.C. Obesity-related glomerulopathy: Current approaches and future perspectives. Obes Rev 2022, 23, e13450. [Google Scholar] [CrossRef]
- Hao, M.; Lv, Y.; Liu, S.; Guo, W. The New Challenge of Obesity - Obesity-Associated Nephropathy. Diabetes Metab Syndr Obes 2024, 17, 1957–1971. [Google Scholar] [CrossRef] [PubMed]
- Yau, K.; Kuah, R.; Cherney, D.Z.I.; Lam, T.K.T. Obesity and the kidney: mechanistic links and therapeutic advances. Nat Rev Endocrinol 2024, 20, 321–335. [Google Scholar] [CrossRef]
- Yao, E.; Yang, X.; Huang, X.; Mi, Y.; Wu, X.; Fang, M.; Huang, J.; Qiu, Y.; Hong, X.; Peng, L.; et al. Phytochemical wedelolactone reverses obesity by prompting adipose browning through SIRT1/AMPK/ PPARα pathway via targeting nicotinamide N-methyltransferase. Phytomedicine 2022, 94, 153843. [Google Scholar] [CrossRef]
- Axelsson, A.S.; Tubbs, E.; Mecham, B.; Chacko, S.; Nenonen, H.A.; Tang, Y.; Fahey, J.W.; Derry, J.M.J.; Wollheim, C.B.; Wierup, N.; et al. Sulforaphane reduces hepatic glucose production and improves glucose control in patients with type 2 diabetes. Sci Transl Med 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Bian, Z.; Zhang, M. Targeting the Nrf2 signaling pathway using phytochemical ingredients: A novel therapeutic road map to combat neurodegenerative diseases. Phytomedicine 2023, 109, 154582. [Google Scholar] [CrossRef]
- Mafra, D.; Borges, N.A.; Lindholm, B.; Shiels, P.G.; Evenepoel, P.; Stenvinkel, P. Food as medicine: targeting the uraemic phenotype in chronic kidney disease. Nat Rev Nephrol 2021, 17, 153–171. [Google Scholar] [CrossRef] [PubMed]
- Mykkänen, O.T.; Huotari, A.; Herzig, K.H.; Dunlop, T.W.; Mykkänen, H.; Kirjavainen, P.V. Wild blueberries (Vaccinium myrtillus) alleviate inflammation and hypertension associated with developing obesity in mice fed with a high-fat diet. PLoS One 2014, 9, e114790. [Google Scholar] [CrossRef] [PubMed]
- Roopchand, D.E.; Kuhn, P.; Rojo, L.E.; Lila, M.A.; Raskin, I. Blueberry polyphenol-enriched soybean flour reduces hyperglycemia, body weight gain and serum cholesterol in mice. Pharmacol Res 2013, 68, 59–67. [Google Scholar] [CrossRef]
- Du, J.; Shen, L.; Tan, Z.; Zhang, P.; Zhao, X.; Xu, Y.; Gan, M.; Yang, Q.; Ma, J.; Jiang, A.; et al. Betaine Supplementation Enhances Lipid Metabolism and Improves Insulin Resistance in Mice Fed a High-Fat Diet. Nutrients 2018, 10. [Google Scholar] [CrossRef]
- Mafra, D.; Esgalhado, M.; Borges, N.A.; Cardozo, L.; Stockler-Pinto, M.B.; Craven, H.; Buchanan, S.J.; Lindholm, B.; Stenvinkel, P.; Shiels, P.G. Methyl Donor Nutrients in Chronic Kidney Disease: Impact on the Epigenetic Landscape. J Nutr 2019, 149, 372–380. [Google Scholar] [CrossRef]
- Bahrampour, N.; Mirzababaei, A.; Hosseininasab, D.; Abaj, F.; Clark, C.C.T.; Mirzaei, K. High intake of dietary phytochemical index may be related to reducing risk of diabetic nephropathy: a case-control study. BMC Nutr 2023, 9, 14. [Google Scholar] [CrossRef]
- Urmila, A.; Armash, M.; Pavan, S.; Rachana, N.; Patil, R. Anti-Inflammatory phytochemicals for the treatment of diabetic nephropathy. Current Functional Foods 2023. [Google Scholar]
- Moreira, L.S.G.; Brum, I.; de Vargas Reis, D.C.M.; Trugilho, L.; Chermut, T.R.; Esgalhado, M.; Cardozo, L.; Stenvinkel, P.; Shiels, P.G.; Mafra, D. Cinnamon: an aromatic condiment applicable to chronic kidney disease. Kidney Res Clin Pract 2023, 42, 4–26. [Google Scholar] [CrossRef]
- Dinda, B.; Chakraborty, A. Pharmacokinetics and Metabolism of Phytochemicals Having Anti-obesity and Antidiabetic Activity; 2022; pp. 469–505. [Google Scholar]
- Wang, W.; Sun, W.; Cheng, Y.; Xu, Z.; Cai, L. Role of sirtuin-1 in diabetic nephropathy. J Mol Med (Berl) 2019, 97, 291–309. [Google Scholar] [CrossRef]
- Kim, H.; Dusabimana, T.; Kim, S.R.; Je, J.; Jeong, K.; Kang, M.C.; Cho, K.M.; Kim, H.J.; Park, S.W. Supplementation of Abelmoschus manihot Ameliorates Diabetic Nephropathy and Hepatic Steatosis by Activating Autophagy in Mice. Nutrients 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Sagoo, M.K.; Gnudi, L. Diabetic Nephropathy: An Overview. Methods Mol Biol 2020, 2067, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Jefferson, J.A.; Shankland, S.J.; Pichler, R.H. Proteinuria in diabetic kidney disease: a mechanistic viewpoint. Kidney Int 2008, 74, 22–36. [Google Scholar] [CrossRef]
- Kume, S.; Koya, D. Autophagy: A Novel Therapeutic Target for Diabetic Nephropathy. Diabetes Metab J 2015, 39, 451–460. [Google Scholar] [CrossRef]
- Kume, S.; Koya, D.; Uzu, T.; Maegawa, H. Role of nutrient-sensing signals in the pathogenesis of diabetic nephropathy. Biomed Res Int 2014, 2014, 315494. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Kim, S.; Lee, S.Y.; Koo, J.K.; Wang, Z.; Choi, M.E. Autophagy regulates TGF-β expression and suppresses kidney fibrosis induced by unilateral ureteral obstruction. J Am Soc Nephrol 2014, 25, 2835–2846. [Google Scholar] [CrossRef]
- Kume, S.; Yamahara, K.; Yasuda, M.; Maegawa, H.; Koya, D. Autophagy: emerging therapeutic target for diabetic nephropathy. Semin Nephrol 2014, 34, 9–16. [Google Scholar] [CrossRef]
- Jin, Y.; Liu, S.; Ma, Q.; Xiao, D.; Chen, L. Berberine enhances the AMPK activation and autophagy and mitigates high glucose-induced apoptosis of mouse podocytes. Eur J Pharmacol 2017, 794, 106–114. [Google Scholar] [CrossRef]
- Wei, Y.; Gao, J.; Qin, L.; Xu, Y.; Shi, H.; Qu, L.; Liu, Y.; Xu, T.; Liu, T. Curcumin suppresses AGEs induced apoptosis in tubular epithelial cells via protective autophagy. Exp Ther Med 2017, 14, 6052–6058. [Google Scholar] [CrossRef]
- Ezzat, S.M.; Abdallah, H.M.I.; Yassen, N.N.; Radwan, R.A.; Mostafa, E.S.; Salama, M.M.; Salem, M.A. Phenolics from Physalis peruviana fruits ameliorate streptozotocin-induced diabetes and diabetic nephropathy in rats via induction of autophagy and apoptosis regression. Biomed Pharmacother 2021, 142, 111948. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Han, X.; Wen, Q.; Qiu, X.; Deng, H.; Chen, Q. Protective Effect of Keluoxin against Diabetic Nephropathy in Type 2 Diabetic Mellitus Models. Evid Based Complement Alternat Med 2021, 2021, 8455709. [Google Scholar] [CrossRef]
- Chen, Y.; Zheng, Y.F.; Lin, X.H.; Zhang, J.P.; Lin, F.; Shi, H. Dendrobium mixture attenuates renal damage in rats with diabetic nephropathy by inhibiting the PI3K/Akt/mTOR pathway. Mol Med Rep 2021, 24. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, W.; Zhang, Z.; Hu, Y.; Zhang, X.; Sun, Y.; Lei, Q.; Sun, D.; Liu, T.; Fan, Y.; et al. Yishen capsule promotes podocyte autophagy through regulating SIRT1/NF-κB signaling pathway to improve diabetic nephropathy. Ren Fail 2021, 43, 128–140. [Google Scholar] [CrossRef]
- Amanat, M.; Lal, K.; Singh, T.G.; Singh, R. Molecular insights of diabetic nephropathy and chemical constituents-based treatment approach. In Phytochemistry Reviews; 2025. [Google Scholar] [CrossRef]
- Zhan, X.; Yan, C.; Chen, Y.; Wei, X.; Xiao, J.; Deng, L.; Yang, Y.; Qiu, P.; Chen, Q. Celastrol antagonizes high glucose-evoked podocyte injury, inflammation and insulin resistance by restoring the HO-1-mediated autophagy pathway. Mol Immunol 2018, 104, 61–68. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, H.; Wang, Q.; Zhou, X.; Lu, X.; Liu, T.; Zhan, Y.; Li, P. Chinese Herbal Medicine in Ameliorating Diabetic Kidney Disease via Activating Autophagy. J Diabetes Res 2019, 2019, 9030893. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gao, Y.; Tian, N.; Zhu, Z.; Wang, T.; Xu, J.; Wu, B.; Zhang, N. Astragaloside IV represses high glucose-induced mesangial cells activation by enhancing autophagy via SIRT1 deacetylation of NF-κB p65 subunit. Drug Des Devel Ther 2018, 12, 2971–2980. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.H.; Ding, D.F.; Yong, H.J.; Dong, C.L.; You, N.; Ye, X.L.; Pan, M.L.; Ma, J.H.; You, Q.; Lu, Y.B. Resveratrol transcriptionally regulates miRNA-18a-5p expression ameliorating diabetic nephropathy via increasing autophagy. Eur Rev Med Pharmacol Sci 2017, 21, 4952–4965. [Google Scholar]
- Huang, S.S.; Ding, D.F.; Chen, S.; Dong, C.L.; Ye, X.L.; Yuan, Y.G.; Feng, Y.M.; You, N.; Xu, J.R.; Miao, H.; et al. Resveratrol protects podocytes against apoptosis via stimulation of autophagy in a mouse model of diabetic nephropathy. Sci Rep 2017, 7, 45692. [Google Scholar] [CrossRef]
- Al Shahrani, M.; Chandramoorthy, H.C.; Alshahrani, M.; Abohassan, M.; Eid, R.A.; Ravichandran, K.; Rajagopalan, P. Cassia auriculata leaf extract ameliorates diabetic nephropathy by attenuating autophagic necroptosis via RIP-1/RIP-3-p-p38MAPK signaling. J Food Biochem 2021, 45, e13810. [Google Scholar] [CrossRef]
- Yang, F.; Qu, Q.; Zhao, C.; Liu, X.; Yang, P.; Li, Z.; Han, L.; Shi, X. Paecilomyces cicadae-fermented Radix astragali activates podocyte autophagy by attenuating PI3K/AKT/mTOR pathways to protect against diabetic nephropathy in mice. Biomed Pharmacother 2020, 129, 110479. [Google Scholar] [CrossRef]
- Han, J.; Zhang, Y.; Shi, X.; Peng, Z.; Xing, Y.; Pang, X. Tongluo Digui decoction treats renal injury in diabetic rats by promoting autophagy of podocytes. J Tradit Chin Med 2021, 41, 125–132. [Google Scholar] [CrossRef]
- Zhang, Y.; Arzaghi, H.; Ma, Z.; Roye, Y.; Musah, S. Epigenetics of Hypertensive Nephropathy. Biomedicines 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Pitzer, A.; Elijovich, F.; Laffer, C.L.; Ertuglu, L.A.; Sahinoz, M.; Saleem, M.; Krishnan, J.; Dola, T.; Aden, L.A.; Sheng, Q.; et al. DC ENaC-Dependent Inflammasome Activation Contributes to Salt-Sensitive Hypertension. Circ Res 2022, 131, 328–344. [Google Scholar] [CrossRef]
- Bidani, A.K.; Griffin, K.A. Pathophysiology of hypertensive renal damage: implications for therapy. Hypertension 2004, 44, 595–601. [Google Scholar] [CrossRef]
- Laffer, C.L.; Elijovich, F.; Sahinoz, M.; Pitzer, A.; Kirabo, A. New Insights Into the Renin-Angiotensin System in Chronic Kidney Disease. Circ Res 2020, 127, 607–609. [Google Scholar] [CrossRef]
- Ngowi, E.E.; Sarfraz, M.; Afzal, A.; Khan, N.H.; Khattak, S.; Zhang, X.; Li, T.; Duan, S.F.; Ji, X.Y.; Wu, D.D. Roles of Hydrogen Sulfide Donors in Common Kidney Diseases. Front Pharmacol 2020, 11, 564281. [Google Scholar] [CrossRef]
- Hackfort, B.T.; Mishra, P.K. Emerging role of hydrogen sulfide-microRNA crosstalk in cardiovascular diseases. Am J Physiol Heart Circ Physiol 2016, 310, H802–812. [Google Scholar] [CrossRef] [PubMed]
- Weber, G.J.; Pushpakumar, S.B.; Sen, U. Hydrogen sulfide alleviates hypertensive kidney dysfunction through an epigenetic mechanism. Am J Physiol Heart Circ Physiol 2017, 312, H874–h885. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Ren, M.; Fan, D.; Mao, Y.; Zhu, Y.Z. Identification and Validation of the miR/RAS/RUNX2 Autophagy Regulatory Network in AngII-Induced Hypertensive Nephropathy in MPC5 Cells Treated with Hydrogen Sulfide Donors. Antioxidants (Basel) 2024, 13. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Hu, T.; Gao, B.; Xia, R.; Dai, X.; Wang, J. Network pharmacology and in vitro experiments reveal the autophagy mechanism of Yanggan-Yishui granules in improving hypertensive renal injury. Archives of Medical Science 2024. [Google Scholar] [CrossRef]
- Klahr, S. Obstructive nephropathy. Intern Med 2000, 39, 355–361. [Google Scholar] [CrossRef]
- Chevalier, R.L.; Cachat, F. Role of Angiotensin II in Chronic Ureteral Obstruction. Contributions to nephrology 2001, 135, 250–260. [Google Scholar] [CrossRef]
- Ichikawa, I.; Kuwayama, F.; Pope, J.C.t.; Stephens, F.D.; Miyazaki, Y. Paradigm shift from classic anatomic theories to contemporary cell biological views of CAKUT. Kidney Int 2002, 61, 889–898. [Google Scholar] [CrossRef]
- Bander, S.J.; Buerkert, J.E.; Martin, D.; Klahr, S. Long-term effects of 24-hr unilateral ureteral obstruction on renal function in the rat. Kidney Int 1985, 28, 614–620. [Google Scholar] [CrossRef]
- Chevalier, R.L.; Forbes, M.S.; Thornhill, B.A. Ureteral obstruction as a model of renal interstitial fibrosis and obstructive nephropathy. Kidney Int 2009, 75, 1145–1152. [Google Scholar] [CrossRef]
- Li, L.; Zepeda-Orozco, D.; Black, R.; Lin, F. Autophagy is a component of epithelial cell fate in obstructive uropathy. Am J Pathol 2010, 176, 1767–1778. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Ru, F.; Chen, X.; Chen, Z. Autophagy attenuates renal fibrosis in obstructive nephropathy through inhibiting epithelial-to-mesenchymal transition. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2021, 46, 601–608. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, Z.; Li, B.; Zhang, B.; Du, Y.; Liu, Y.; He, Y.; Chen, X. Curcumin attenuates renal interstitial fibrosis of obstructive nephropathy by suppressing epithelial-mesenchymal transition through inhibition of the TLR4/NF-кB and PI3K/AKT signalling pathways. Pharm Biol 2020, 58, 828–837. [Google Scholar] [CrossRef]
- Liu, Y.-F.; Wang, H.-H.; Geng, Y.-H.; Han, L.; Tu, S.-H.; Wang, H. Advances of berberine against metabolic syndrome-associated kidney disease: Regarding effect and mechanism. Frontiers in Pharmacology 2023, 14–2023. [Google Scholar] [CrossRef]
- Malhotra, B.; Kulkarni, G.T.; Dhiman, N.; Joshi, D.D.; Chander, S.; Kharkwal, A.; Sharma, A.K.; Kharkwal, H. Recent advances on Berberis aristata emphasizing berberine alkaloid including phytochemistry, pharmacology and drug delivery system. Journal of Herbal Medicine 2021, 27, 100433. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, Y.-s.; Chang, Z.-m.; Li, L.; Zhang, Y.; Lu, M.-m.; Zheng, X.; Li, M.; Shao, D.; Li, J.; et al. Berberine-loaded Janus nanocarriers for magnetic field-enhanced therapy against hepatocellular carcinoma. Chem Biol Drug Des 2017, 89, 464–469. [Google Scholar] [CrossRef]
- Lu, W.-B.; Chen, W.-W.; Li, R.-L. Determination of berberine chloride in mouse plasma after intragastric administration of Lianxiang microemulsion. Zhong yao cai = Zhongyaocai = Journal of Chinese medicinal materials 2010, 33, 957–960. [Google Scholar]
- Mei, D.; Mao, S.; Sun, W.; Wang, Y.; Kissel, T. Effect of chitosan structure properties and molecular weight on the intranasal absorption of tetramethylpyrazine phosphate in rats. European Journal of Pharmaceutics and Biopharmaceutics 2008, 70, 874–881. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, G.; Cui, W.; Zhang, Y.; Liang, X. Regulatory mechanisms of tetramethylpyrazine on central nervous system diseases: A review. Frontiers in Pharmacology 2022, 13–2022. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Cheng, Z.; Cheng, Y.; Xu, Y. Investigating the passage of tetramethylpyrazine-loaded liposomes across blood-brain barrier models in vitro and ex vivo. Materials Science and Engineering: C 2016, 69, 1010–1017. [Google Scholar] [CrossRef]
- Li, J.; Wei, J.; Wan, Y.; Du, X.; Bai, X.; Li, C.; Lin, Y.; Liu, Z.; Zhou, M.; Zhong, Z. TAT-modified tetramethylpyrazine-loaded nanoparticles for targeted treatment of spinal cord injury. Journal of Controlled Release 2021, 335, 103–116. [Google Scholar] [CrossRef] [PubMed]
- TAN Zuo, H.W. Effects of neferine nanoliposomes on cerebral anoxia and cerebral embolism in mice. Journal of Guangdong College of Pharmacy 2010, 377–379. [Google Scholar]
- Chen, Y.-Q.; Chen, H.-Y.; Tang, Q.-Q.; Li, Y.-F.; Liu, X.-S.; Lu, F.-H.; Gu, Y.-Y. Protective effect of quercetin on kidney diseases: From chemistry to herbal medicines. Frontiers in Pharmacology 2022, 13–2022. [Google Scholar] [CrossRef]
- Cai, T.; Wang, X.; Li, B.; Xiong, F.; Wu, H.; Yang, X. Deciphering the synergistic network regulation of active components from SiNiSan against irritable bowel syndrome via a comprehensive strategy: Combined effects of synephrine, paeoniflorin and naringin. Phytomedicine 2021, 86, 153527. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Fang, C.; Jiang, M.J. The therapeutic effect of Berberine on rodent models of kidney injury: a systematic review and meta - analysis. BMC Nephrology 2025, 26, 315. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.; Yan, Y. Therapeutic potential of sulforaphane in liver diseases: a review. Front. Pharmacol. 2023, 14–2023. [Google Scholar] [CrossRef] [PubMed]
| Animal Models with Disease | Phytochemicals | Doses and times | Alterations in Autophagy and renal outcome | Alterations in mechanism/pathway involved | References |
|---|---|---|---|---|---|
| Sepsis-induced AKI in rats | Resveratrol | 30 mg/kg for 5 days | Autophagy activation reduced renal tubular damage | Induction of p53 deacetylation, activation of deacetylase Sirt1 | [75] |
| Renal fibrosis, AKI models | Ginsenoside Re | - | Improves renal function, reduces fibrosis, and reduces autophagy | [80] | |
| DKD in experimental rats | Ferulic acid |
50 mg/kg, orally, daily for 8 weeks |
Ameliorates kidney injury, reduces apoptosis, inflammation, and defective autophagy | Modulates AGEs, MAPKs, NF-κB, induces autophagy | [15,81] |
| DKD | Polyphenols (general) |
NA |
Improve autophagy and lysosomal function, reduce fibrosis, apoptosis, and inflammation | SIRT1, mTOR, AMPK, TFEB nuclear transfer | [81] |
| STZ-induced rats | Icariin | 20, 40, and 80 mg/kg/d, orally for 12 weeks | Restore autophagy, inhibit tubulointerstitial fibrosis | GLP-1R activate, inhibit mTOR phosphorylation. | [82] |
| STZ/HFD-induced T2DM rats | Isorhamnetin | 50 mg/kg/d) for 4 or 8 weeks | Enhances renal function, improves glucose/lipid metabolism and autophagy | ↑ FYCO1, ULK-1, TECPR1, WIPI | [83] |
| SD rats + UNx + STZ | Emodin | 20–40 mg/kg, 12 weeks | Reduces apoptosis, podocyte injury, and fibrosis | Activates autophagy via AMPK/mTOR | [84] |
| Male Sprague Dawley rats with STZ | Cyclocarya paliurus triterpenic acids | 40–160 mg/kg (CPTL) and 160 mg/kg (CPTH) for 10 weeks | ↑ Autophagy; ↓ kidney injury and apoptosis | ↑ p-AMPK; ↓ p-mTOR | [85] |
| DKD mice serum | Tripterygium glycoside | In vitro: 1.25 μg/mL, 72 h | ↑ Autophagy; ↓ EMT and podocyte apoptosis | mTOR/Twist1 pathway | [84] |
| STZ-induced diabetic mice | Isorhapontigenin (polyphenol) |
- |
Activates autophagy, reduces oxidative stress, improves podocyte/endothelial cell damage | ↑AMPK/Nrf2 pathway, ↑Beclin-1, ↑Atg5, ↓P62 | [81] |
| DKD, nephrotic syndrome | Plantago asiatica/major (Hispidulin) |
- |
↑Autophagy; Prevents podocyte apoptosis via autophagy, improves proteinuria and kidney function | MAPK pathway (animal models) | [86] |
| Renal fibrosis (CKD model) | Sulforaphane |
- |
Alleviates renal fibrosis via dual regulation of autophagy in fibroblasts and tubular epithelial cells | mTOR-mediated autophagy pathway | [87] |
| Post-renal transplant, nephrotoxicity | Schisandra sphenanthera (deoxyschizandrin) | - | Enhances tacrolimus effect, nephroprotection via autophagy (limited human data) | Nrf2 pathway | [86] |
| Arsenic-induced nephrotoxicity in rats | Zingerone | 25 and 50 mg/kg | Decreases oxidative stress, inflammation, apoptosis, and kidney damage; Reduces excessive autophagy | Modulate AKT2 and FOXO1, Reduce NF-κB and IL-1β, TNF, IL-6, iNOS, COX-2, MAPK14, MAPK15, JNK | [88] |
| High glucose-induced podocyte injury | Ursolic acid |
In vitro: 5 μmol/L for 24 h (in vitro) |
Increases autophagy, improves podocyte injury | ↓ miR-21 → ↑ PTEN → ↓ PI3K/Akt/mTOR pathway | [89] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.





