Submitted:
29 January 2026
Posted:
10 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
Construction of the Bioenergetic Index (BEI)
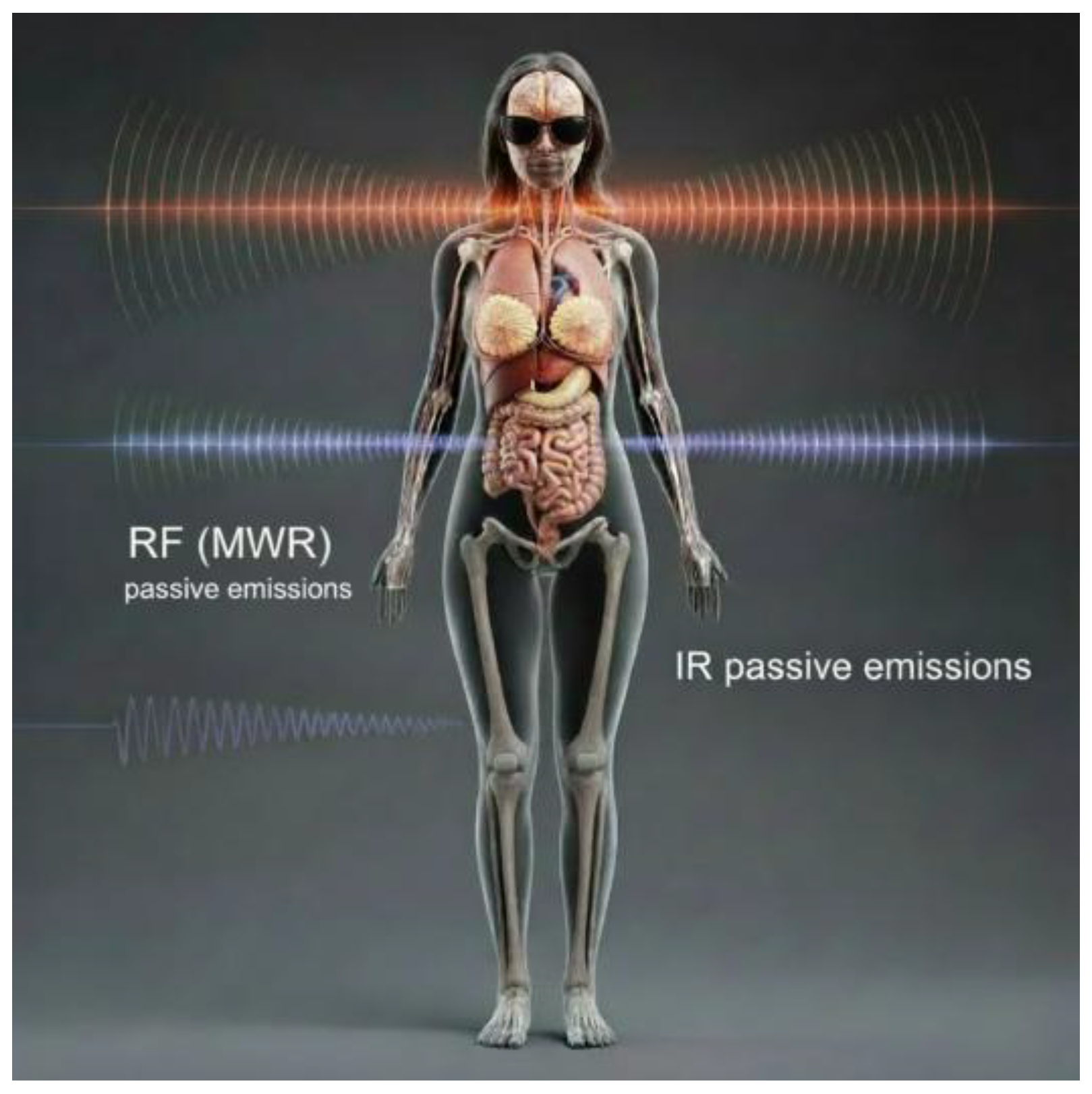
2. Materials and Methods
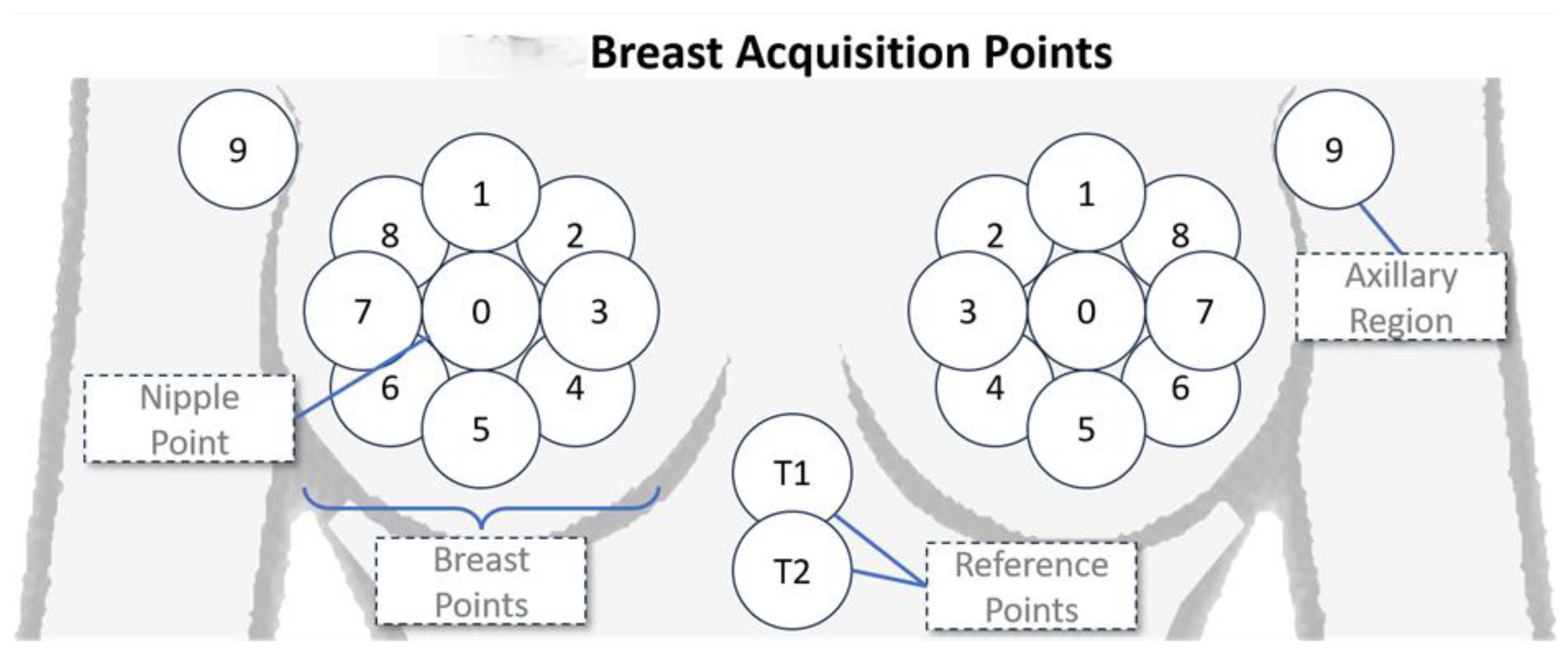
3. Results
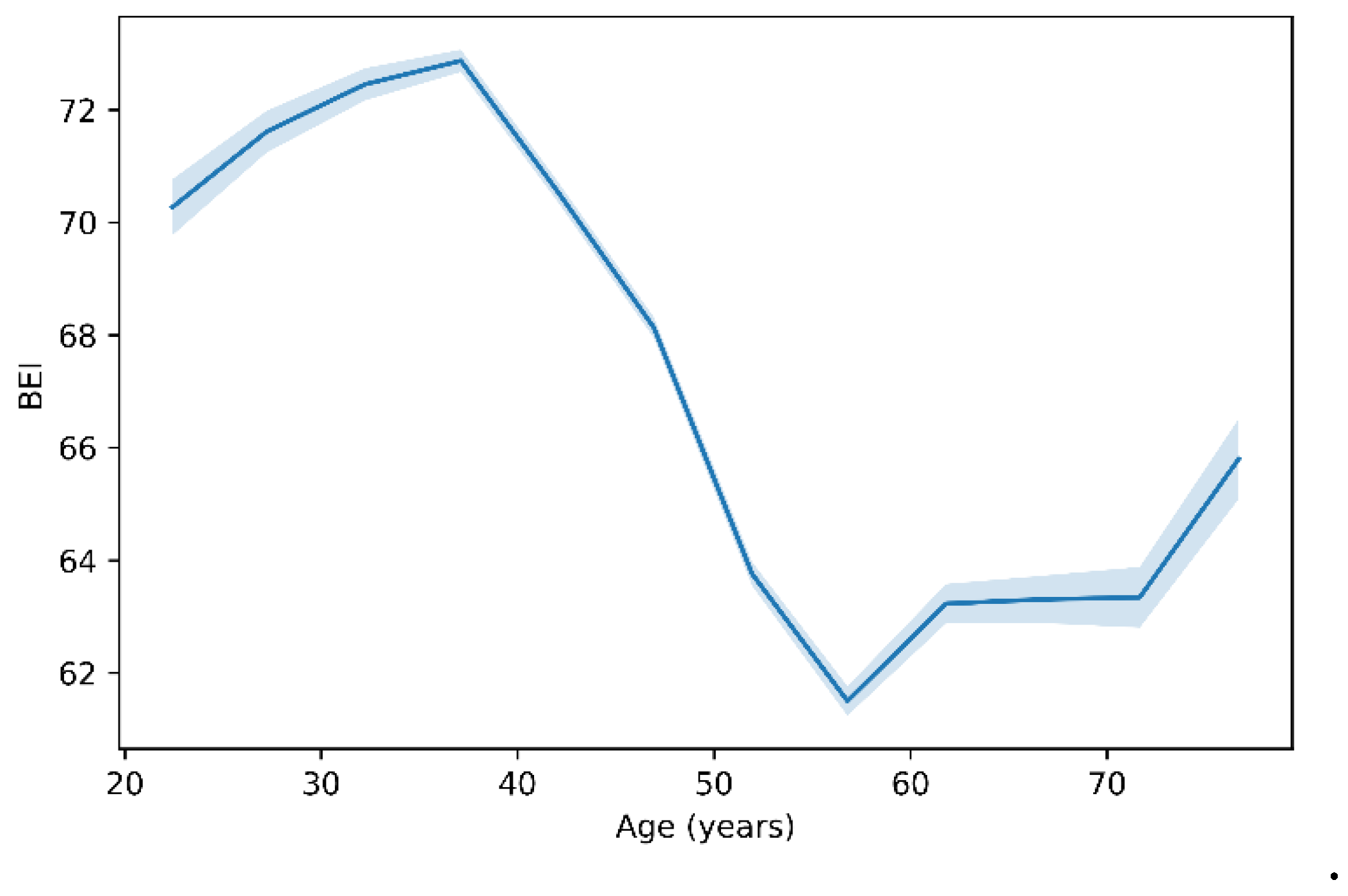
BEI Trends with Age
Biological Age Prediction
- Coefficient of Determination: To measure the proportion of variance in age explained by thermal features.
- Root Mean Square Error (RMSE): To quantify the standard deviation of the prediction errors (in years).
- Mean Absolute Error (MAE): To assess the average magnitude of errors (in years).
MWR-NetAge
The biological Meaning of BEI Ageing
Comparison to Existing Biological Aging Methods
| Clock type | Typical R2 | RMSE (years) | Key limitations |
| DNA methylation clocks (Horvath, Hannum, PhenoAge) | 0.70–0.92 | 3–7 | Costly lab tests; batch effects; delayed readout |
| Proteomic / metabolomic clocks | 0.65–0.90 | 4–8 | Highly invasive, expensive, assay-dependent |
| Wearable-derived clocks (HRV, sleep, activity) | 0.40–0.65 | 5–10 | High noise; long-term tracking required |
| Thermophysiological BEI (this work) | ≈0.8 | ≈4.8 | Local tissue signal; requires controlled setup |
- Completely non-invasive (no needles, no radiation, no contrast agents)
- Instant measurement (scan and result in <15 minutes)
- No consumables beyond device amortisation
- Low cost and easily scalable to population level
- Reflects real-time metabolic and microvascular physiology rather than static molecular marks
Clinical and Diagnostic Implications
Nonlinearity and Physiological Interpretation
- mitochondrial density
- estrogen-mediated vasodilation
- microvascular elasticity
- brown adipose activity
Nonlinearity and Physiological Interpretation
Strengths and Limitations
Key Strengths
- Very large clinical dataset (N > 36,000) spanning ages 20–80
- High-quality, standardised deep-tissue and surface thermal measurements
- Fully non-invasive, low-risk and operationally scalable modality
- Physiology-driven feature engineering (deep, surface, gradients, heterogeneity, BEI)
- Predictive models trained without chronological age as an input feature
- Robust reconstruction of both individual BioAge and the non-linear population-level ageing trajectory
Key Limitations
- Female-only, breast-imaging cohort; generalisation to mixed-sex populations and other anatomical regions remains to be demonstrated
- Retrospective, cross-sectional design rather than longitudinal follow-up
- Limited clinical annotation; lack of systematic metabolic, cardiovascular and inflammatory phenotypes to correlate with Age Delta
- Breast-specific anatomy and thermal field geometry; male torso and whole-body distributions are likely to differ and require separate calibration
Future Work
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cohen AA, Kennedy BK, et al. Biological aging: What is it and why is it important? Nat Rev Genet. 2023;24:357–372.
- van der Spoel E, Hughes DA, et al. Comparing biological age clocks based on DNA methylation, clinical biomarkers, and proteomics. Nat Aging. 2022;2:192–205.
- Goryanin I, Karbainov S, Shevelev O, et al. Passive microwave radiometry in biomedical studies. Drug Discov Today. 2020;25(4):757–763. [CrossRef]
- Galazis C, Wu H, Goryanin I. Breast cancer detection via multi-tiered self-contrastive learning in microwave radiometric imaging. Diagnostics (Basel). 2025;15(5):549. [CrossRef]
- Tarakanov AV, Tarakanov AA, Skorodumova EG, et al. Age-related changes in the temperature of the lumbar spine measured by passive microwave radiometry (MWR). Diagnostics (Basel). 2023;13(21):3294. [CrossRef]
- Goryanin I, Ivanov Y, Damms B, et al. Exploring the interface between quantum biology, microwave technology, and neuroscience. Drug Discov Today. 2025;30(7):104408. [CrossRef]
- Gavazzi G, Krause K-H. Ageing and heat production in humans: decline of mitochondrial thermogenesis. J Gerontol A Biol Sci Med Sci. 2020;75:1214–1222.
- McCulloch K, Din MU, et al. Brown adipose function declines nonlinearly with age. Cell Metab. 2021;33:998–1014.
- González-Alonso J. Microvascular function in aging and cardiometabolic disease. Nat Rev Cardiol. 2023;20:48–66.
- Ganio MS, et al. Autonomic thermoregulation and age: decline in heat dissipation and sweating function. Exp Gerontol. 2022;163:111810.
- Wang Z, et al. Nonlinear dynamics of human core temperature across the lifespan. Sci Rep. 2020;10:17074.
- Masuda Y, et al. Decline and plateau of body temperature in elderly individuals: nonlinear aging effects. Gerontology. 2023;69:35–46.
- Breiman L. Random forests. Mach Learn. 2001;45:5–32.
- Horvath S. DNA methylation age of human tissues and cell types. Genome Biol. 2013;14:R115.
- Hannum G, et al. Genome-wide methylation profiles reveal quantitative views of human aging rates. Mol Cell. 2013;49:359–367. [CrossRef]
- Levine ME, et al. An epigenetic biomarker of aging for lifespan and healthspan (DNAm PhenoAge). Aging (Albany NY). 2018;10:573–591.
- Lu AT, et al. DNA methylation GrimAge strongly predicts lifespan and healthspan. Aging (Albany NY). 2019;11:303–327. [CrossRef]
- Lehallier B, et al. Undulating changes in human plasma proteome profiles across the lifespan. Nat Med. 2019;25:1843–1850. [CrossRef]
- Tanaka T, et al. Plasma proteomic biomarker signature of age predicts health and life span. eLife. 2020;9:e61073.
- Hertel J, et al. Measuring biological age via metabonomics: the metabolic age score. J Proteome Res. 2016;15:400–410. [CrossRef]
- Mutz J, et al. Metabolomic age (MileAge) predicts health and life span. Sci Adv. 2024;10:eadp3743. [CrossRef]
- Pyrkov TV, et al. Deep longitudinal phenotyping of wearable sensor data reveals independent markers of longevity, stress and resilience. Aging (Albany NY). 2021;13:7900–7913. [CrossRef]
- Troncale JA. The aging process: physiologic changes and pharmacologic implications. Postgrad Med. 1996;99(5):111–114, 120–122.
- Lipsitz LA, Goldberger AL. Loss of “complexity” and aging: potential applications of fractals and chaos theory to senescence. JAMA. 1992;267(13):1806–1809.
- Goldberger AL, Peng C-K, Lipsitz LA. What is physiologic complexity and how does it change with aging and disease? Neurobiol Aging. 2002;23(1):23–26. [CrossRef]

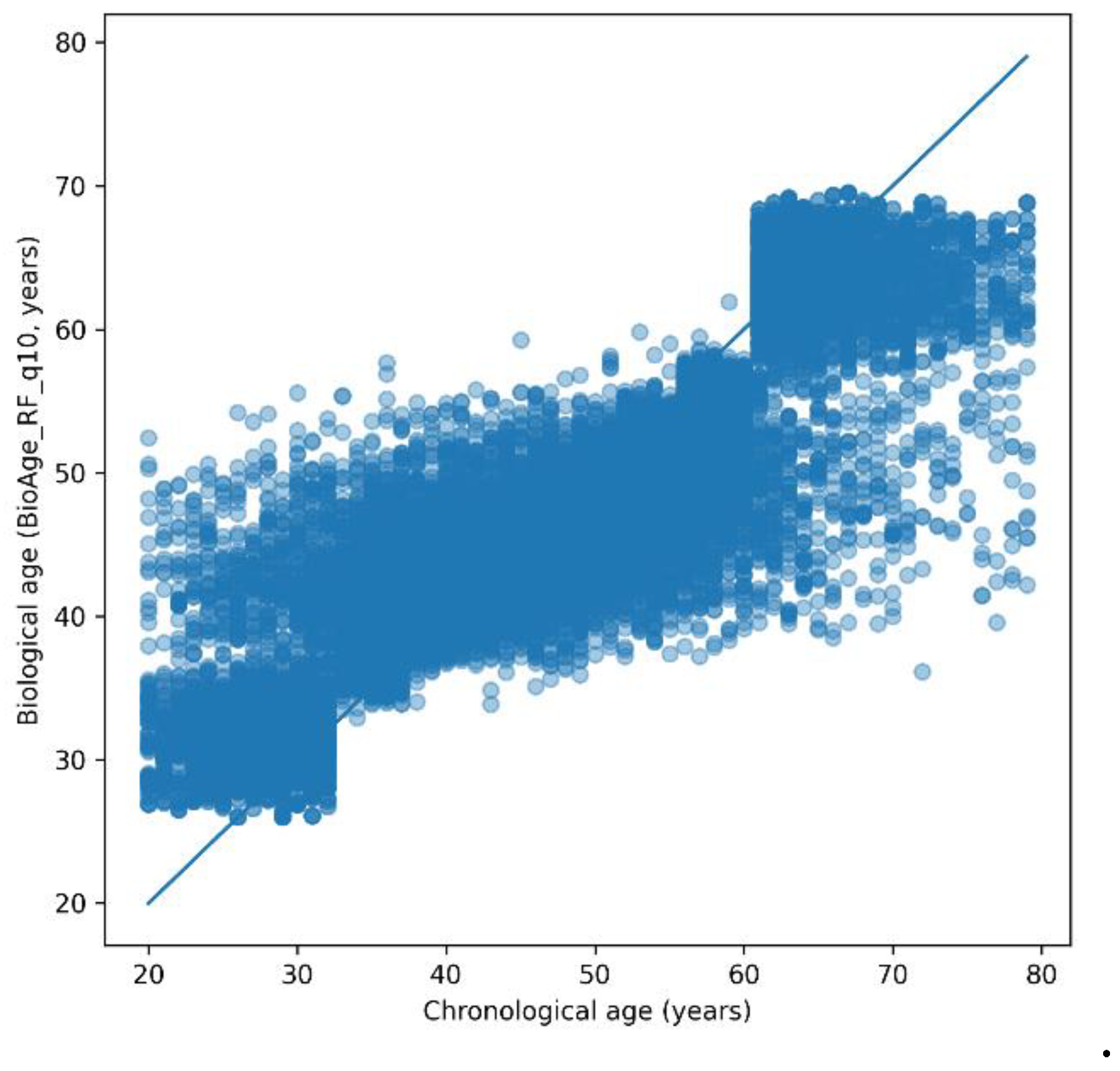
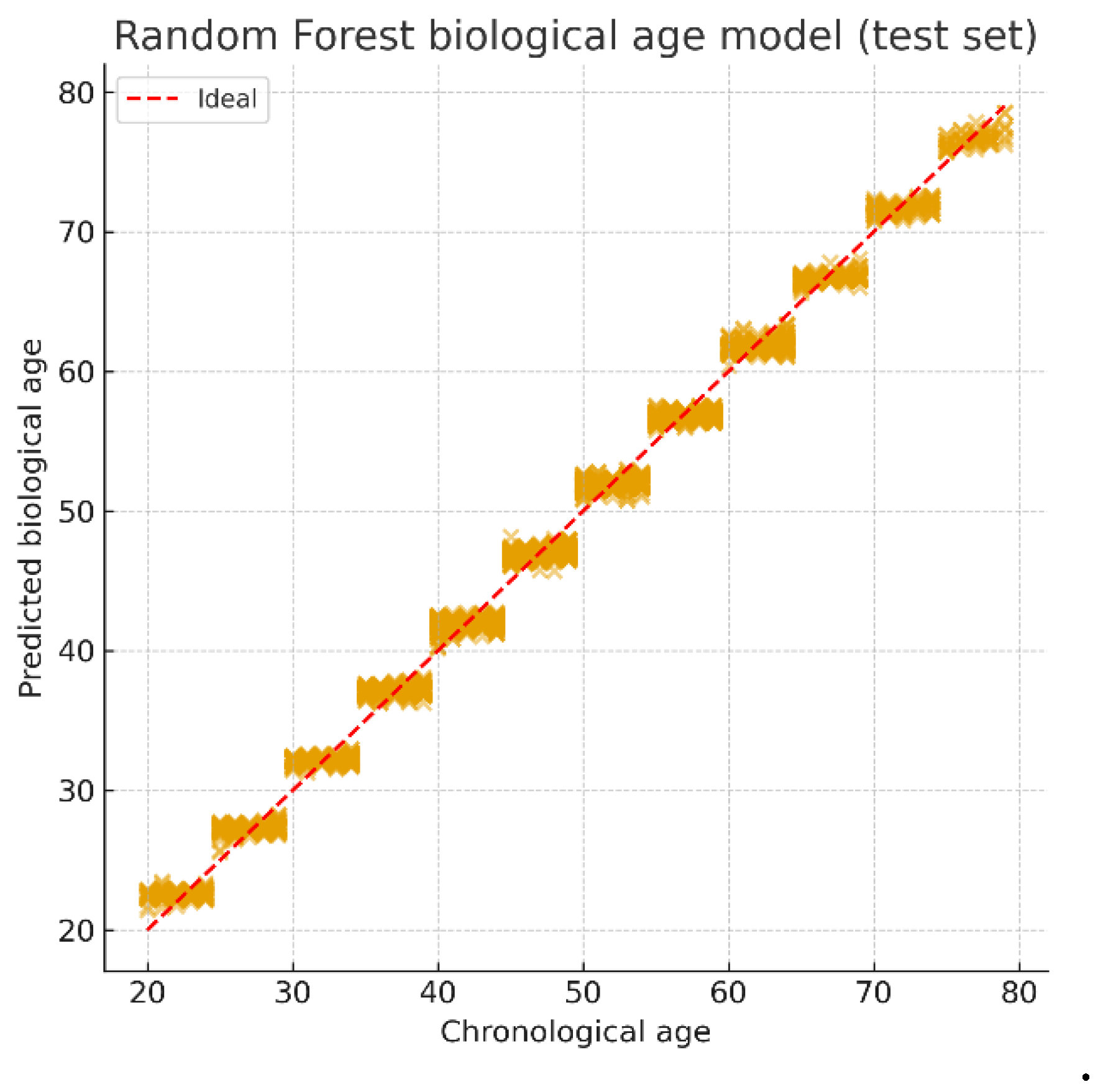
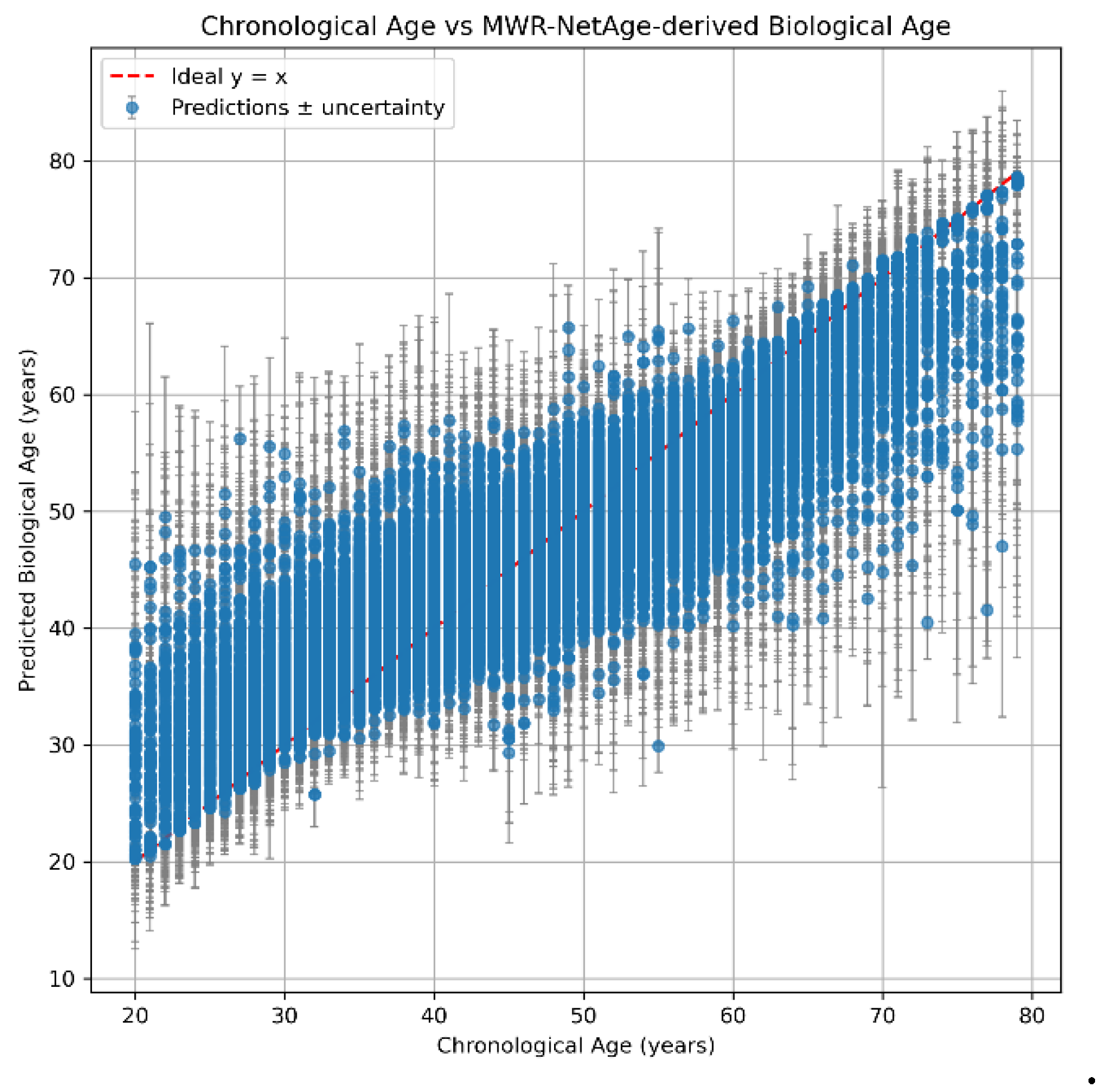
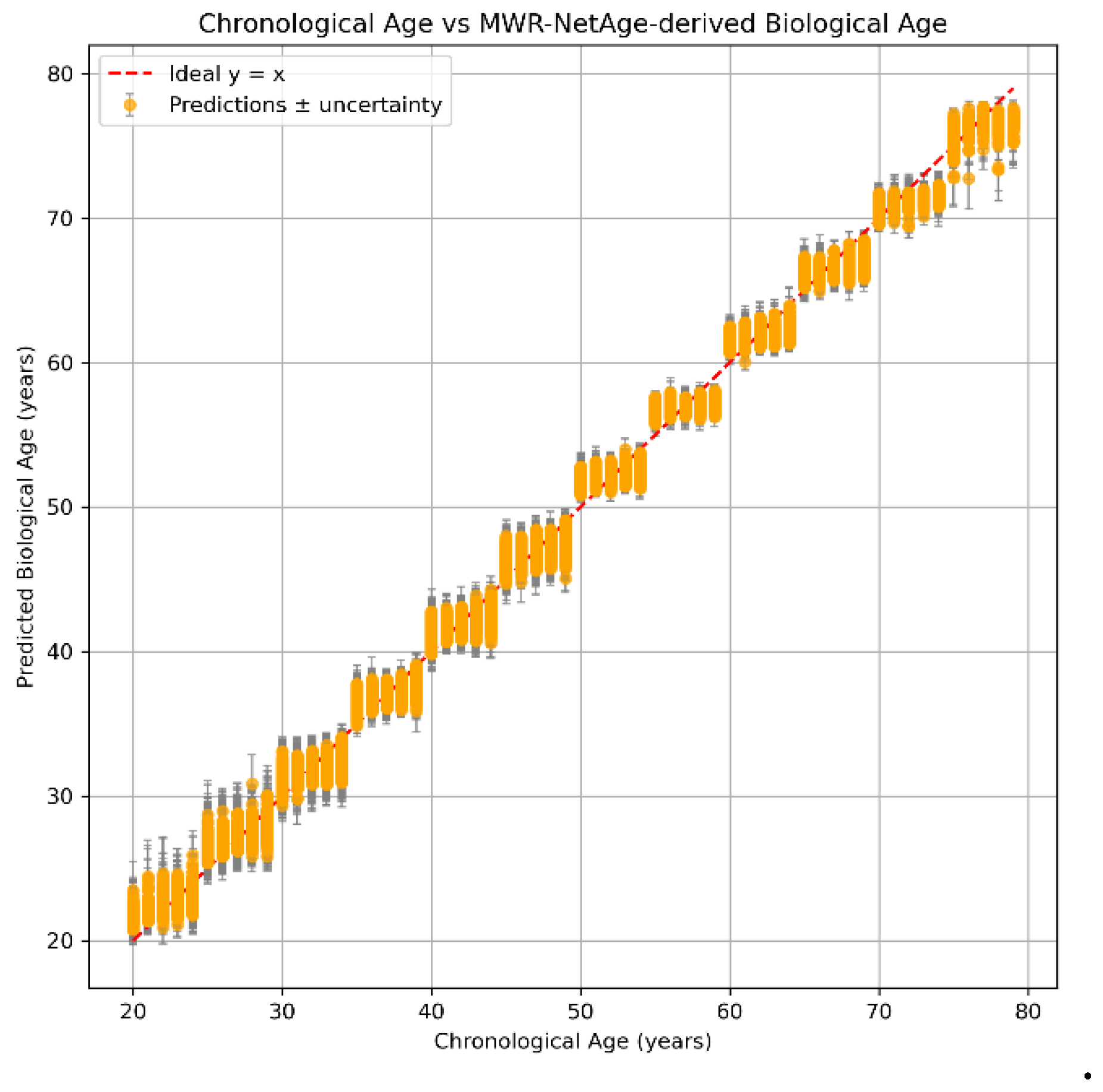
| Age group. | N (subjects) | RMSE (years) | R2 |
|---|---|---|---|
| < 30 years | 9,845 | 4.92 | 0.003 |
| 30–65 years | 24,464 | 6.18 | 0.081 |
| ≥ 65 years | 2,081 | 3.81 | 0.009 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




