1. Introduction
Reactive Oxygen Species (ROS) are highly reactive, oxygen-containing molecules, including free radicals such as the superoxide anion (O2-) and non-radicals like hydrogen peroxide (H2O2), primarily produced as byproducts of aerobic metabolism within the mitochondrial electron transport chain [
1,
2]. While essential for processes such as redox signaling and innate immunity, their overproduction triggers oxidative stress. This stress leads to cellular damage by attacking and modifying vital biomolecules: initiating lipid peroxidation, which degrades cellular membranes; oxidatively modifying proteins to alter their function; and damaging DNA, potentially leading to mutations [
2]. Enzymatic antioxidants, including superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), alongside non-enzymatic antioxidants such as thiols and reduced glutathione (GSH), play pivotal roles in protecting cells against ROS-induced damage [
3,
4].
The chemotherapy drug cisplatin is a major cause of nephrotoxicity, often presenting as dose-limiting acute kidney injury (AKI). The primary histopathological manifestations are acute tubular necrosis (ATN) and apoptosis within the kidney’s proximal tubules, where the drug preferentially accumulates [
5,
6]. Cisplatin-induced toxicity involves complex mechanisms, including direct cytotoxicity, exacerbated oxidative stress, increased endoplasmic reticulum stress, DNA damage, mitochondrial dysfunction, inflammation, and renal vasoconstriction leading to ischemic injury [
5].
Malondialdehyde (MDA) is a key secondary end-product of lipid peroxidation and a widely used biomarker for assessing oxidative damage. An elevation in MDA concentration is interpreted as a direct indicator of pathological lipid peroxidation and oxidative stress in tissues [
7]. Superoxide Dismutase (SOD) is a crucial antioxidant enzyme that converts the superoxide radical into the less reactive hydrogen peroxide, serving as a primary enzymatic defense line [
8]. Total Antioxidant Status (TAS) and Total Oxidant Status (TOS) are comprehensive biochemical parameters used to evaluate the overall balance between oxidants and antioxidants. TOS provides an overall measure of all ROS, indicating the cumulative level of oxidation in tissue. Conversely, TAS reflects the combined action of both enzymatic and non-enzymatic antioxidant defense systems. Concomitant changes, such as a decrease in TAS and an increase in TOS, effectively indicate severe tissue damage [
9].
N-Acetylserotonin (NAS) (chemical name: N-Acetyl-5-hydroxytryptamine), or normelatonin, is a natural intermediate in the biosynthesis of melatonin from serotonin. Beyond its role as a precursor, NAS acts as an agonist for melatonin and tropomyosin receptor kinase B (TrkB) receptors. The activation of TrkB is linked to its potential antidepressant effects, promotion of neurogenesis, and neuroprotective properties in tissues such as the retina and hippocampus. Furthermore, NAS is a potent antioxidant, with some studies suggesting it may be more effective than melatonin in scavenging free radicals and protecting against oxidative damage, particularly lipid peroxidation [
10,
11,
12]. Although NAS is both the precursor and a metabolite of melatonin, it exerts several effects independent of melatonin against oxidative stress [
11].
As the precursor molecule of melatonin, NAS has been associated with various pleiotropic effects. NAS exhibits antioxidant [
11], anti-aging [
13,
14], neuroprotective [
11], and antidepressant [
14] properties, while also mitigating cognitive impairment [
14], and lipid peroxidation [
15,
16]. Furthermore, antihypertensive and antitumor activities have been reported [
17]. Protective effects have been documented in various tissues; specifically, NAS has been found to ameliorate damage in the brain [
18,
19], retina [
20], liver [
15,
18,
21], testes [
22], bone [
18], intervertebral disc [
4], and kidney [
13].
Melatonin and its metabolites exhibit antioxidative, anti-inflammatory, and anti-apoptotic effects. There are three identified melatonin receptors in mammals (MT1, MT2, and MT3). MT3 is found in the highest concentrations in the kidney and liver, and to a lesser extent in the heart, brain, and adipose tissue. MT3 is a cytosolic reductase enzyme possessing a melatonin-binding site; it has been identified as quinone reductase 2 (QR2) and inhibits the electron transfer reactions of quinones, thus playing a role in protecting the cell from oxidative stress. Notably, it has a higher binding affinity for NAS than for melatonin [
18].
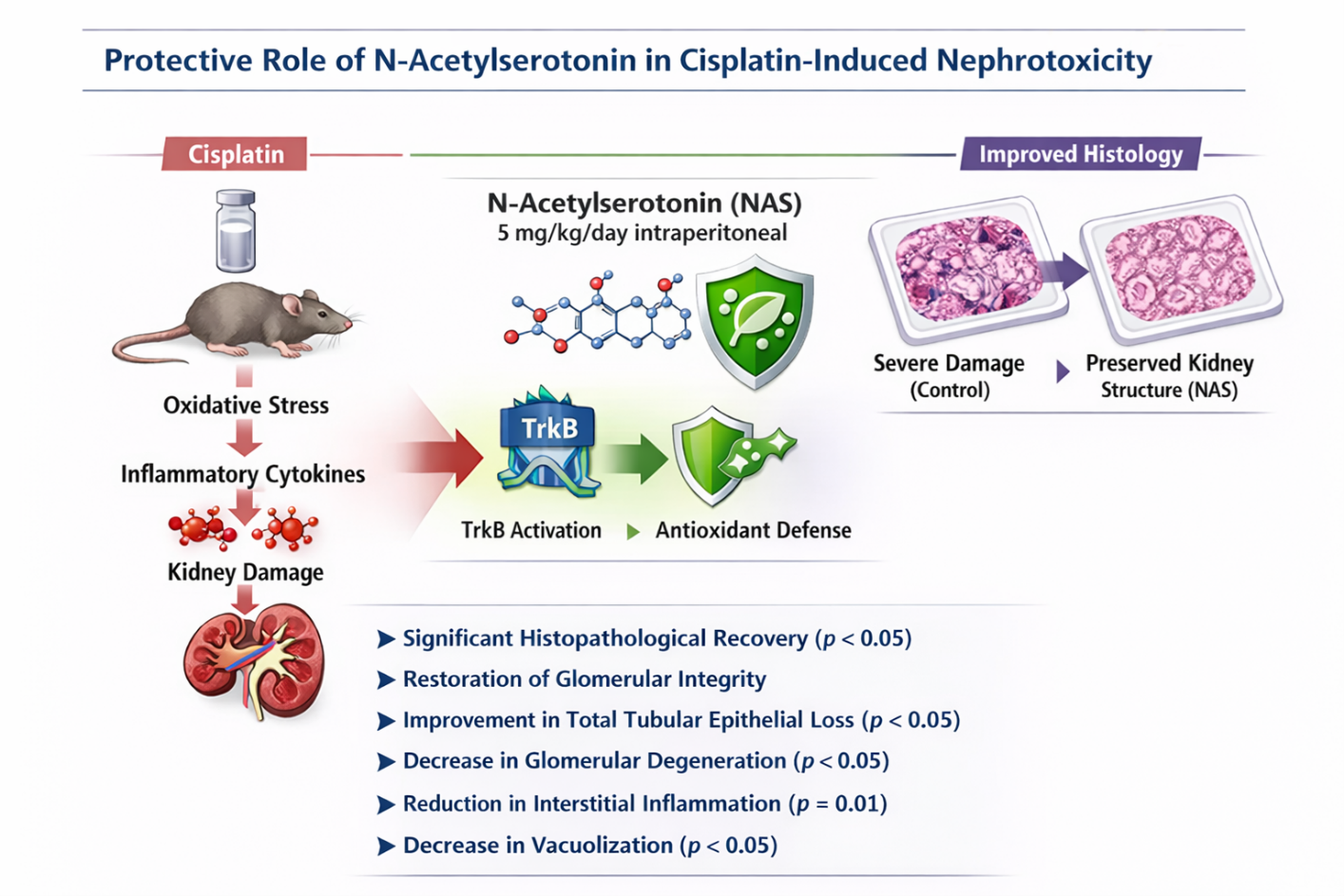
Considering that MT3 receptors are abundant in the kidney and exhibit a higher binding affinity for NAS than melatonin, this study aimed to investigate the effects of the potent antioxidant NAS on cisplatin-induced renal injury.
2. Materials and Methods
2.1. Experimental Procedure and Administered Drugs/Chemicals
This study was conducted at Balıkesir University’s Animal Experiments Laboratory, and the animals were obtained from the same facility. All experimental procedures were performed by researchers certified in animal experimentation.
A total of 35 two-month-old Wistar Albino rats (mean weight: 250 g, range: 190–280 g) were used. The rats were housed at room temperature (22–23 °C) under a 12-hour light/dark cycle for an acclimatization period of 7 days, with 7 rats per cage. They had free access to food and water.
Rats were randomly assigned to five experimental groups (n = 7 per group):
Control group (C): Received no treatment.
Sham group (S): Received an intraperitoneal (i.p.) injection of 0.5 mL of isotonic 1.2% ethanol solution daily (09:00 pm).
NAS group: Received daily i.p. injections of 5 mg/kg [
2,
4,
18] N-acetylserotonin (NAS) dissolved in 0.5 mL of isotonic 1.2% ethanol solution.
Cisplatin group (CP): Received daily i.p. injections of 0.5 mL isotonic 1.2% ethanol solution, and a single dose of 7.5 mg/kg cisplatin [
5] was administered i.p. on the fourth day.
CP+NAS group: Received daily i.p. injections of 5 mg/kg NAS dissolved in 0.5 mL of isotonic 1.2% ethanol solution, plus a single i.p. injection of 7.5 mg/kg cisplatin on the fourth day.
2.2. Termination of the Experiment and Collection of Blood and Tissue Samples
Six hours after the final procedures on the seventh day, the rats were anesthetized with ketamine. Following laparotomy and thoracotomy, 5 mL of blood was collected from each animal via intracardiac aspiration. Both kidneys were rapidly dissected; the right kidneys were fixed for pathological examination, while the left kidneys were stored for biochemical analysis.
2.3. Supply of Kits Used
Kidney tissue homogenates were used to measure levels of total oxidant status (TOS; μmol H2O2 Equiv./L), total antioxidant status (TAS; mmol Trolox Equiv./L), superoxide dismutase (SOD; U/mg protein), and malondialdehyde (MDA; μmol/g protein). Serum samples were analyzed for urea (mg/dL) and creatinine (mg/dL).
N-acetyl-5-hydroxytryptamine (NAS, CAS No. 1210-83-9; also known as normelatonin) was obtained from Sigma-Aldrich (USA). Cisplatin injectable solution (Cipintu®) was obtained from Onko Koçsel Pharmaceuticals (Kocaeli, Turkey). All other chemicals used for biochemical assays were of analytical grade and obtained from commercial sources.
2.4. Serum Sample Preparation
Serum samples were incubated at room temperature for 30–40 minutes and then centrifuged at 1500 × g for 20 minutes at 4 °C. Aliquoted serum samples were stored at −80 °C until analysis.
2.5. Renal Function Test Analysis
To assess renal function, serum urea and creatinine levels were analyzed via colorimetric methods using a Mindray BS-400 autoanalyzer (Shenzhen, China).
2.6. Preparation of Kidney Tissue Homogenates
Kidney tissues frozen at −80 °C were homogenized for MDA, SOD, TAS, and TOS analysis. First, tissues were washed with cold phosphate-buffered saline (PBS; 0.01 M, pH 7.4). Subsequently, tissues were weighed and cold PBS was added (0.9 mL per 0.1 g of tissue) to obtain a 10% (w/v) starting homogenate. Homogenization was performed using a Stuart SHM1 homogenizer (USA). The homogenates were centrifuged at 10,000 × g for 10 minutes at 4 °C (SIGMA 3-30K, Germany). The resulting supernatants were separated and stored at −80 °C. Total protein concentration in the supernatants was determined using the Bradford reagent (Clearband, Eco-Tech).
2.7. Biochemical Analyses
MDA levels were measured using a metabolic analysis kit (Elabscience, Cat. No. E-BC-K025-M, Houston, TX, USA) based on the thiobarbituric acid (TBA) method. The absorbance was measured at 532 nm using a Varioskan Flash Multimode Reader (Thermo Scientific, USA). Results are expressed as μmol/g protein.
Total SOD activity was measured with a hydroxylamine-based kit (Elabscience, Cat. No. E-BC-K019-M, USA) at a 1:150 dilution. Absorbance was recorded at 550 nm, and results are expressed as U/mg protein.
TAS and TOS levels were determined colorimetrically using specific kits (Rel Assay Diagnostics, Gaziantep, Turkey). TAS results are expressed as mmol Trolox equivalent/L, and TOS as μmol H2O2 equivalent/L.
2.8. Histopathological Evaluation
Kidney tissues were fixed in 10% neutral buffered formaldehyde for a minimum of 24 hours. Following standard dehydration and paraffin embedding, 4-µm-thick sections were cut using an automated microtome. Sections were stained with hematoxylin and eosin (H&E) and evaluated under a light microscope.
The histopathological examination assessed six morphological parameters: tubular epithelial loss, tubular dilatation, intraluminal cast formation, glomerular degeneration, interstitial inflammation, and vacuolization. Each parameter was scored on a semi-quantitative scale from 0 to 3 (0: absent, 1: mild, 2: moderate, 3: severe) [
23]. The evaluation was performed by a pathologist blinded to the experimental groups.
2.9. Ethical Approval
Approval for the study was received from the Balıkesir University Animal Experiments Ethics Committee (Decision No: 2025/7-1).
2.10. Statistical Evaluation
Statistical analyses were performed using SPSS version 23 (IBM Corp., Armonk, NY, USA). Data are expressed as mean ± standard deviation. The normality of data distribution was assessed using normality tests. Normally distributed numerical data were evaluated using one-way ANOVA for multiple group comparisons. When significant differences were detected, Post-Hoc tests (Tukey or Tamhane’s T2, depending on Levene’s test for equality of variances) were performed. For non-normally distributed data, the Kruskal-Wallis test was used for multiple comparisons, followed by the Mann-Whitney U test for pairwise comparisons. A p-value < 0.05 was considered statistically significant.
3. Results
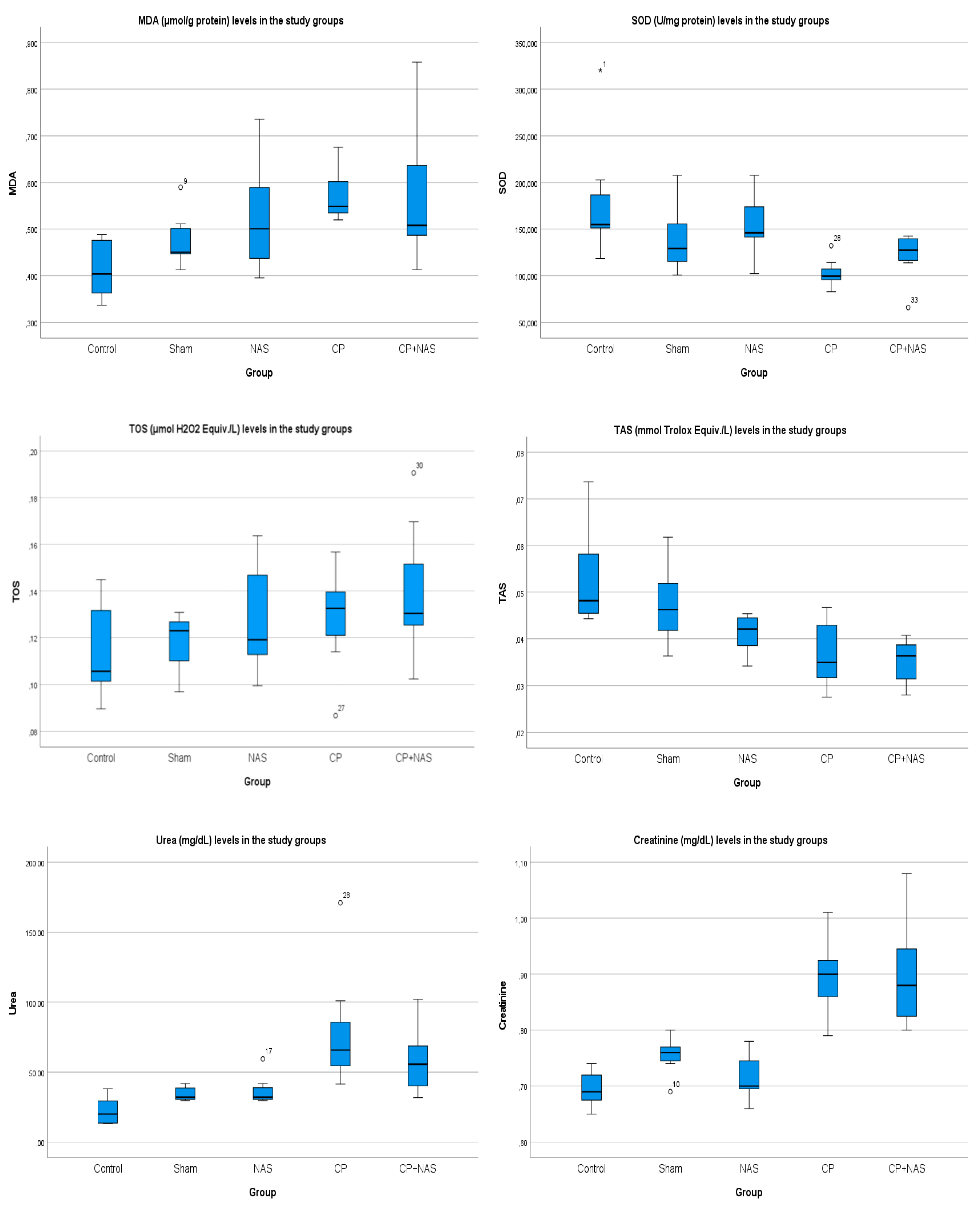
3.1. Malondialdehyde (MDA)
MDA levels in the kidney tissue were significantly higher in both the CP group (p <0.001) and the CP+NAS group (p <0.001) compared to the control group. No significant difference was observed between the sham, NAS, and control groups. Furthermore, MDA levels did not differ significantly between the CP and CP+NAS groups (
Table 1,
Figure 1).
3.2. Superoxide Dismutase (SOD)
SOD activities were not significantly different among the C, S, NAS, and CP+NAS groups. However, SOD levels were significantly lower in the CP group compared to the control group (p <0.01). Although SOD levels were slightly higher in the CP+NAS group compared to the CP group, this difference did not reach statistical significance (
Table 1,
Figure 1).
3.3. Total Oxidant Status (TOS)
No significant difference was detected in TOS levels among the five experimental groups studied.
3.4. Total Antioxidant Status (TAS)
TAS levels did not differ significantly between the C, S, and NAS groups. Conversely, TAS was significantly lower in the CP (p <0.01) and CP+NAS (p <0.01) groups compared to the control group. TAS levels were also significantly lower in the CP group compared to the sham group (p <0.05). Although TAS levels showed an upward trend in the CP+NAS group relative to the CP group, the difference was not statistically significant (
Table 1,
Figure 1).
3.5. Urea and Creatinine
Serum urea levels were highest in the CP group and lowest in the control group; however, these differences between the study groups were not statistically significant.
In contrast, serum creatinine levels did not differ between the C, S, and NAS groups. Creatinine concentrations in the CP group were significantly higher than those in the C (p = 0.001), S (p <0.05), and NAS (p <0.01) groups. Similarly, creatinine values in the CP+NAS group were significantly higher than in the C (p <0.05), S (p <0.05), and NAS (p <0.05) groups. No significant difference in creatinine levels was observed between the CP and CP+NAS groups.
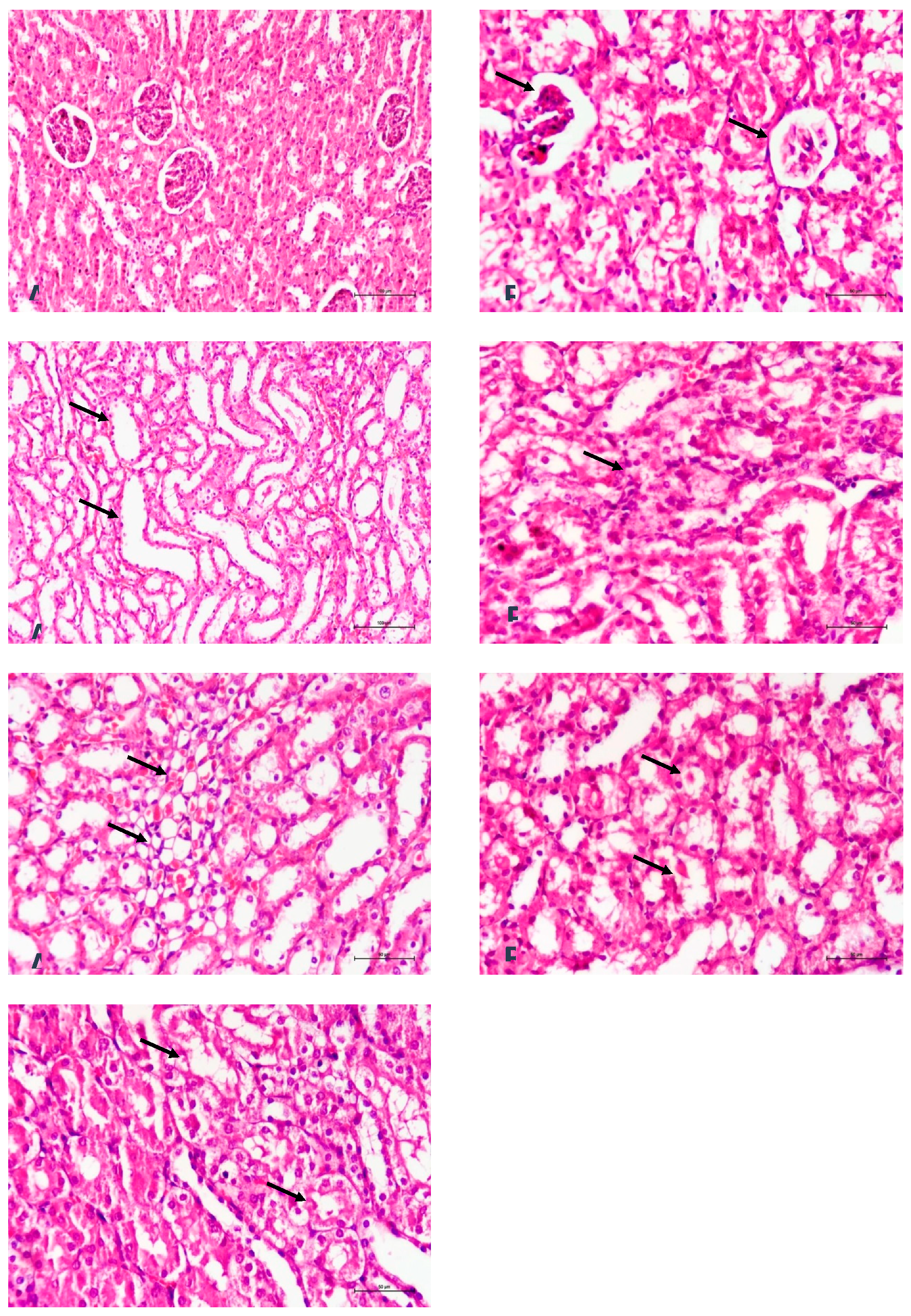
3.6. Pathological Examination Results
There was no significant difference between the control, sham, and NAS groups regarding any of the six histopathological parameters. The composite scores for all histopathological parameters in the CP group were significantly higher than those in the C, S, and NAS groups. Similarly, the total scores in the CP+NAS group were significantly higher than those in the C, S, and NAS groups.
Notably, scores for tubular epithelial loss (p <0.05), glomerular degeneration (p <0.05), interstitial inflammation (p = 0.01), and vacuolization (p <0.05) were significantly higher in the CP group compared to the CP+NAS group, indicating a protective effect of NAS treatment. However, intraluminal cast formation and glomerular degeneration scores did not differ significantly between the CP and CP+NAS groups. Total histopathological scores are summarized in
Table 2.
Representative images of histopathological findings from the renal tissue are presented in
Figure 2.
4. Discussion
In the present study, while biochemical alterations in serum and renal tissue remained relatively modest, the histopathological improvements provided by NAS were remarkably evident. At the administered dose and duration, cisplatin (CP) did not induce a significant elevation in blood urea levels. However, creatinine values rose substantially in both the CP and CP+NAS groups compared to the other experimental cohorts. NAS administration did not exert a significant corrective effect on these increased creatinine levels, suggesting that functional recovery may lag behind structural restoration.
Previous literature highlights the tissue-specific variability of NAS. Gesing et al. reported that NAS (20 mg/kg for 3 weeks) effectively attenuated MDA levels in the liver, brain, lungs, and testes in an aflatoxin B1-induced model; however, no effect was observed in the kidney as the toxin failed to induce lipid peroxidation in that specific tissue [
24]. Conversely, Oxenkrug et al. demonstrated that long-term, higher-dose oral NAS administration (20 mg/kg for 4 weeks) significantly reduced renal MDA levels, exhibiting potent antioxidant and anti-aging activity [
13] Similarly, Qingzhi Li et al. showed that NAS dose-dependently reduced hippocampal MDA elevation in a traumatic brain injury (TBI) model, where a 30 mg/kg dose successfully improved neurogenesis and mitigated cognitive impairments [
25]. While NAS has also been shown to reduce iron-induced lipid peroxidation in testicular tissue [
26], it did not significantly modulate MDA levels or demonstrate a lipid peroxidation-reducing effect at the specific dose and duration utilized in our study.
Interestingly, NAS slightly increased SOD levels in the CP+NAS group. The lack of difference in SOD levels between the CP+NAS and NAS-only groups is pivotal, as it suggests that NAS may directly enhance the antioxidant capacity within our renal injury model. Although not extensively studied in kidney tissues, NAS has shown corrective effects on MDA and SOD levels in liver cells under hydrogen peroxide-induced stress. This protection has been attributed to the upregulation of endogenous antioxidants and the suppression of mitochondria-dependent apoptotic pathways [
2]. Yu et al. similarly found that 5 mg/kg NAS significantly ameliorated hepatic ischemia-reperfusion injury by modulating MDA and SOD levels [
27]. While our study did not mirror these biochemical outcomes in terms of TOS or TAS differences, the robust structural preservation we observed suggests that NAS operates through pathways that prioritize cellular architecture over immediate systemic biochemical markers.
The most salient finding of our study was the profound corrective effect of NAS on tubular epithelial loss, glomerular degeneration, interstitial inflammation, and vacuolization in CP-induced renal injury. These findings align with previous reports indicating that the predominant toxic effects of CP occur within the tubules, specifically the proximal segments [
6,
28]. Findings suggest that the renal protective effect of NAS may occur through multiple pathways. NAS inhibit mitochondrial death pathways and autophagic activation in the brain, but these effects have not been studied in kidney tissue [
29]. While NAS studies have primarily focused on neuronal tissues, its nephroprotective potential is becoming increasingly apparent. The structural restoration observed here is particularly significant, as it indicates a localized preservation of the renal parenchyma.
It is well-established that the precursor (serotonin) and the subsequent metabolite (melatonin) of NAS possess protective effects in renal damage. Serotonin receptor activation stimulates mitochondrial biogenesis and homeostasis in renal injury [
30], while melatonin exerts anti-inflammatory effects by reducing IL1-alpha, IL1-beta, Mcp-1 and TGF-beta1mRNA levels, thereby mitigating ROS formation, fibrosis, and apoptosis [
31,
32,
33]. NAS appears to provide a unique bridge between these pathways.
Crucially, NAS is a potent and unique agonist of tropomyosin receptor kinase B (TrkB), the high-affinity receptor for Brain-Derived Neurotrophic Factor (BDNF) [
1,
34]. Unlike serotonin or melatonin, NAS rapidly activates TrkB, initiating downstream pro-survival cascades such as the PI3K/AKT and Ras-ERK pathways [
34,
35]. These pathways are essential for promoting cell survival and inhibiting apoptosis. In neuronal models, NAS has been shown to increase BCL-2 expression while decreasing BAX and cleaved caspases [
4,
12]. Furthermore, both melatonin and NAS have been found to inhibit mitochondrial death pathways, including the release of cytochrome c and Smac [
18,
29].
The significance of our findings is underscored by the widespread expression of TrkB receptors in the kidney, including proximal/distal tubule epithelial cells, the juxtaglomerular apparatus, and podocytes [
36,
37,
38]. TrkB signaling in the kidney plays dual roles in maintaining renal function and ensuring proper structural development [
38,
39,
40]. The pronounced histopathological improvement observed in our study—even in the absence of significant biochemical shifts—suggests that NAS targets these TrkB-dependent pro-survival properties to stabilize mitochondrial integrity and prevent cellular apoptosis in vulnerable renal tissues. The dose-dependent nature of NAS and its tissue-specific efficacy may explain the limited biochemical response in this study [
4,
15]. Factors such as tissue lipid content and the density of MT3/QR2 receptors likely influence the antioxidant potential of NAS in the renal environment [
41]. The fact that NAS effectively halted the progression of visible tissue damage (epithelial loss and inflammation) positions it as a promising candidate for renoprotective therapy. The main limitation of our study is that the observed protective effects were not investigated across a range of doses to fully delineate the molecular mechanisms. Future research should focus on the dose-response relationship to determine if higher concentrations might also trigger the expected biochemical shifts alongside the observed structural benefits.
5. Conclusions
In conclusion, our results demonstrate that NAS significantly ameliorates the histopathological manifestations of cisplatin-induced renal damage. While systemic biochemical changes were limited, the targeted protection of tubular and glomerular architecture is highly promising. Considering the unique TrkB-mediated pro-survival signaling and direct antioxidant properties of NAS, these findings warrant further investigation into its precise mechanisms as a therapeutic agent for acute kidney injury.
Author Contributions
Conceptualization, [S.Y., G.T., M.A.Y., B.A.A., C.Y., O.K.]; methodology, [S.Y., G.T., M.A.Y., B.A.A., C.Y., O.K.]; software, [S.Y.]; validation, [S.Y.]; formal analysis, [S.Y., G.T., M.A.Y., B.A.A., C.Y., O.K.]; investigation, [S.Y., G.T., M.A.Y., B.A.A., C.Y., O.K.]; resources, [S.Y.]; data curation, [S.Y.]; writing—original draft preparation, [S.Y., G.T., M.A.Y., B.A.A., C.Y., O.K.]; writing—review and editing, [S.Y.]; visualization, [S.Y.]; supervision, [S.Y.]; project administration, [S.Y.]; funding acquisition, [S.Y.]. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Balıkesir University Animal Experiments Ethics Committee (Decision No: 2025/7-1, Date of approval: 23/07/2025.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lezoualc’h, F.; Sparapani, M.; Behl, C. N-acetyl-serotonin (Normelatonin) and Melatonin Protect Neurons against Oxidative Challenges and Suppress the Activity of the Transcription Factor NF-κB. J. Pineal Res. 1998, 24, 168–178. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Yu, S.; Jiang, Z.; Liang, C.; Yu, W.; Li, J.; Du, X.; Wang, H.; Gao, X.; Wang, X. N-Acetyl-Serotonin Protects HepG2 Cells from Oxidative Stress Injury Induced by Hydrogen Peroxide. Oxid. Med. Cell. Longev. 2014, 2014, 1–15. [Google Scholar] [CrossRef]
- Söğüt, İ.; Oğlakçı, A.; Kartkaya, K.; Ol, K.K.; Söğüt, M.S.; Kanbak, G.; İnal, M.E. Effect of Boric Acid on Oxidative Stress in Rats with Fetal Alcohol Syndrome. Exp. Ther. Med. 2015, 9, 1023–1027. [Google Scholar] [CrossRef]
- Yidian, W.; Jihe, K.; Xudong, G.; Daxue, Z.; Mingqiang, L.; Xuewen, K. N-Acetylserotonin Protects Rat Nucleus Pulposus Cells Against Oxidative Stress Injury by Activating the PI3K/AKT Signaling Pathway. World Neurosurg. 2023, 176, e109–e124. [Google Scholar] [CrossRef] [PubMed]
- Akca, G.; Eren, H.; Tumkaya, L.; Mercantepe, T.; Horsanali, M.O.; Deveci, E.; Dil, E.; Yilmaz, A. The Protective Effect of Astaxanthin against Cisplatin-Induced Nephrotoxicity in Rats. Biomed. Pharmacother. 2018, 100, 575–582. [Google Scholar] [CrossRef]
- Verma, P.K.; Raina, R.; Sultana, M.; Singh, M.; Kumar, P. Total Antioxidant and Oxidant Status of Plasma and Renal Tissue of Cisplatin-Induced Nephrotoxic Rats: Protection by Floral Extracts of Calendula Officinalis Linn. Ren. Fail. 2016, 38, 142–150. [Google Scholar] [CrossRef]
- Ince, S.; Kucukkurt, I.; Cigerci, I.H.; Fatih Fidan, A.; Eryavuz, A. The Effects of Dietary Boric Acid and Borax Supplementation on Lipid Peroxidation, Antioxidant Activity, and DNA Damage in Rats. J. Trace Elem. Med. Biol. 2010, 24, 161–164. [Google Scholar] [CrossRef]
- Coban, F.K.; Ince, S.; Kucukkurt, I.; Demirel, H.H.; Hazman, O. Boron Attenuates Malathion-Induced Oxidative Stress and Acetylcholinesterase Inhibition in Rats. Drug Chem. Toxicol. 2015, 38, 391–399. [Google Scholar] [CrossRef]
- Demir, S.; Kazaz, I.O.; Kerimoglu, G.; Demir, E.A.; Colak, F.; Biyik, A.F.; Cansever, Y.; Mentese, A. Propolis Ameliorates Ischemia/Reperfusion-Induced Testicular Damage by Reducing Oxidative Stress. Rev. Int. Andrología 2023, 21, 100364. [Google Scholar] [CrossRef]
- Wölfler, A.; Abuja, P.M.; Schauenstein, K.; Liebmann, P.M. N-Acetylserotonin Is a Better Extra- and Intracellular Antioxidant than Melatonin. FEBS Lett. 1999, 449, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Diduk, R.; Galano, A.; Tan, D.X.; Reiter, R.J. N-Acetylserotonin and 6-Hydroxymelatonin against Oxidative Stress: Implications for the Overall Protection Exerted by Melatonin. J. Phys. Chem. B 2015, 119, 8535–8543. [Google Scholar] [CrossRef] [PubMed]
- Rui, T.; Wang, Z.; Li, Q.; Wang, H.; Wang, T.; Zhang, M.; Tao, L.; Luo, C. A TrkB Receptor Agonist N-Acetyl Serotonin Provides Cerebral Protection after Traumatic Brain Injury by Mitigating Apoptotic Activation and Autophagic Dysfunction. Neurochem. Int. 2020, 132, 104606. [Google Scholar] [CrossRef] [PubMed]
- Oxenkrug, G.; Requintina, P.; Bachurin, S. Antioxidant and Antiaging Activity of N -Acetylserotonin and Melatonin in the in Vivo Models. Ann. N. Y. Acad. Sci. 2001, 939, 190–199. [Google Scholar] [CrossRef]
- Oxenkrug, G.; Ratner, R. N-Acetylserotonin and Aging-Associated Cognitive Impairment and Depression. Aging Dis. 2012, 3, 330–338. [Google Scholar] [PubMed]
- Leaden, P.J.; Catalá, A. Melatonin and N-Acetyl Serotonin Inhibit Selectively Enzymatic and Non-Enzymatic Lipid Peroxidation of Rat Liver Microsomes. Prostaglandins Leukot. Essent. Fat. Acids 2007, 77, 29–35. [Google Scholar] [CrossRef]
- Gesing, A.; Karbownik-Lewinska, M. Protective Effects of Melatonin and N-Acetylserotonin on Aflatoxin B1-Induced Lipid Peroxidation in Rats. Cell Biochem. Funct. 2008, 26, 314–319. [Google Scholar] [CrossRef] [PubMed]
- OXENKRUG, G. Antioxidant Effects of N-Acetylserotonin: Possible Mechanisms and Clinical Implications. Ann. N. Y. Acad. Sci. 2005, 1053, 334–347. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Yang, Q.; Liu, Y.; Zhou, S.; Jiang, J.; Reiter, R.J.; Bhattacharya, P.; Cui, Y.; Yang, H.; Ma, H.; et al. The Multiple Protective Roles and Molecular Mechanisms of Melatonin and Its Precursor N-Acetylserotonin in Targeting Brain Injury and Liver Damage and in Maintaining Bone Health. Free Radic. Biol. Med. 2019, 130, 215–233. [Google Scholar] [CrossRef]
- Yoo, J.M.; Lee, B.D.; Sok, D.E.; Ma, J.Y.; Kim, M.R. Neuroprotective Action of N-Acetyl Serotonin in Oxidative Stress-Induced Apoptosis through the Activation of Both TrkB/CREB/BDNF Pathway and Akt/Nrf2/Antioxidant Enzyme in Neuronal Cells. Redox Biol. 2017, 11, 592–599. [Google Scholar] [CrossRef] [PubMed]
- Tosini, G.; Ye, K.; Iuvone, P.M. N-Acetylserotonin: Neuroprotection, Neurogenesis, and the Sleepy Brain. Neuroscientist 2012, 18, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Garcıa, J.J.; Reiter, R.J.; Karbownik, M.; Calvo, J.R.; Ortiz, G.G.; Tan, D.-X.; Martınez-Balların, E.; Acuna-Castroviejo, D. N-Acetylserotonin Suppresses Hepatic Microsomal Membrane Rigidity Associated with Lipid Peroxidation; 2001; Vol. 428. [Google Scholar]
- Karbownik, M.; Gitto, E.; Lewiñski, A.; Reiter, R.J. Relative Efficacies of Indole Antioxidants in Reducing Autoxidation and Iron-Induced Lipid Peroxidation in Hamster Testes. J. Cell. Biochem. 2001, 81, 693–699. [Google Scholar] [CrossRef]
- Yaman, T.; Akkoyun, H.T.; Bayramoğlu Akkoyun, M.; Karagözoğlu, F.; Melek, Ş.; Keleş, Ö.F.; Bengü, A.Ş. Assessment of the Effect of Sodium Tetraborate on Oxidative Stress, Inflammation, and Apoptosis in Lead-Induced Nephrotoxicity. Drug Chem. Toxicol. 2025, 48, 150–162. [Google Scholar] [CrossRef]
- Gesing, A.; Karbownik-Lewinska, M. Protective Effects of Melatonin and N -acetylserotonin on Aflatoxin B1-induced Lipid Peroxidation in Rats. Cell Biochem. Funct. 2008, 26, 314–319. [Google Scholar] [CrossRef]
- Li, Q.; Wang, P.; Huang, C.; Chen, B.; Liu, J.; Zhao, M.; Zhao, J. N-Acetyl Serotonin Protects Neural Progenitor Cells Against Oxidative Stress-Induced Apoptosis and Improves Neurogenesis in Adult Mouse Hippocampus Following Traumatic Brain Injury. J. Mol. Neurosci. 2019, 67, 574–588. [Google Scholar] [CrossRef] [PubMed]
- Karbownik, M.; Reiter, R.J.; Cabrera, J.; Garcia, J.J. Comparison of the Protective Effect of Melatonin with Other Antioxidants in the Hamster Kidney Model of Estradiol-Induced DNA Damage. Mutat. Res. - Fundam. Mol. Mech. Mutagen. 2001, 474, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Zheng, J.; Jiang, Z.; Shi, C.; Li, J.; Du, X.; Wang, H.; Jiang, J.; Wang, X. Protective Effect of N-Acetylserotonin against Acute Hepatic Ischemia-Reperfusion Injury in Mice. Int. J. Mol. Sci. 2013, 14, 17680–17693. [Google Scholar] [CrossRef]
- Pabla, N.; Dong, Z. Cisplatin Nephrotoxicity: Mechanisms and Renoprotective Strategies. Kidney Int. 2008, 73, 994–1007. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Wang, J.; Jiang, J.; Stavrovskaya, I.G.; Li, M.; Li, W.; Wu, Q.; Zhang, X.; Luo, C.; Zhou, S.; et al. N-Acetyl-Serotonin Offers Neuroprotection through Inhibiting Mitochondrial Death Pathways and Autophagic Activation in Experimental Models of Ischemic Injury. J. Neurosci. 2014, 34, 2967–2978. [Google Scholar] [CrossRef] [PubMed]
- Hurtado, K.; Scholpa, N.E.; Schnellmann, J.G.; Schnellmann, R.G. Serotonin Regulation of Mitochondria in Kidney Diseases. Pharmacol. Res. 2024, 203, 107154. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Han, Z.; Shi, Z.; Liu, C.; Lu, Q. Melatonin Alleviates Renal Injury in Mouse Model of Sepsis. Front. Pharmacol. 2021, 12, 1–8. [Google Scholar] [CrossRef]
- Huang, Y.S.; Ka, S.M.; Lu, K.C.; Chen, A.; Wu, C.C. Therapeutic Insights and Molecular Mechanism Linking Melatonin Signaling and Membranous Nephropathy (Review). Biomed. Reports 2025, 23. [Google Scholar] [CrossRef]
- Minich, D.M.; Henning, M.; Darley, C.; Fahoum, M.; Schuler, C.B.; Frame, J. Is Melatonin the “Next Vitamin D”?: A Review of Emerging Science, Clinical Uses, Safety, and Dietary Supplements. Nutrients 2022, 14, 3934. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.W.; Liu, X.; Pradoldej, S.; Tosini, G.; Chang, Q.; Iuvone, P.M.; Ye, K. N-Acetylserotonin Activates TrkB Receptor in a Circadian Rhythm. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 3876–3881. [Google Scholar] [CrossRef]
- Luo, X.; Zeng, H.; Fang, C.; Zhang, B.H. N-Acetylserotonin Derivative Exerts a Neuroprotective Effect by Inhibiting the NLRP3 Inflammasome and Activating the PI3K/Akt/Nrf2 Pathway in the Model of Hypoxic-Ischemic Brain Damage. Neurochem. Res. 2021, 46, 337–348. [Google Scholar] [CrossRef]
- Huber, L.J.; Hempstead, B.; Donovan, M.J. Neurotrophin and Neurotrophin Receptors in Human Fetal Kidney. Dev. Biol. 1996, 179, 369–381. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Armelloni, S.; Zennaro, C.; Wei, C.; Corbelli, A.; Ikehata, M.; Berra, S.; Giardino, L.; Mattinzoli, D.; Watanabe, S.; et al. BDNF Repairs Podocyte Damage by MicroRNA-Mediated Increase of Actin Polymerization. J. Pathol. 2015, 235, 731–744. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.-Y.; Sheu, W.H.-H.; Lee, I.-T. Brain-Derived Neurotrophic Factor Associated with Kidney Function. Diabetol. Metab. Syndr. 2023, 15, 16. [Google Scholar] [CrossRef]
- Kurajoh, M.; Kadoya, M.; Morimoto, A.; Miyoshi, A.; Kanzaki, A.; Kakutani-Hatayama, M.; Hamamoto, K.; Shoji, T.; Moriwaki, Y.; Yamamoto, T.; et al. Plasma Brain-Derived Neurotrophic Factor Concentration Is a Predictor of Chronic Kidney Disease in Patients with Cardiovascular Risk Factors - Hyogo Sleep Cardio-Autonomic Atherosclerosis Study. PLoS ONE 2017, 12, 0178686. [Google Scholar] [CrossRef] [PubMed]
- Cirrik, S.; Hacioglu, G.; Ayyildiz, S.N.; Tezcan, B.; Abidin, I.; Aydin-Abidin, S.; Noyan, T. Renal Response to Tunicamycin-Induced Endoplasmic Reticulum Stress in BDNF Heterozygous Mice. Adv. Clin. Exp. Med. 2019, 28, 1161–1170. [Google Scholar] [CrossRef] [PubMed]
- Fagali, N.; Catalá, A. The Antioxidant Behaviour of Melatonin and Structural Analogues during Lipid Peroxidation Depends Not Only on Their Functional Groups but Also on the Assay System. Biochem. Biophys. Res. Commun. 2012, 423, 873–877. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).