1. Introduction
Renal cell carcinoma (RCC) accounts for approximately 2–3 % of all adult malignancies and remains one of the most aggressive urologic cancers. The disease is most commonly diagnosed between 60 and 70 years of age, although it may also occur in younger patients. Men are disproportionately affected, with a male-to-female ratio of approximately 1.5–2:1 [
1]. The most common histological subtype is clear cell renal cell carcinoma (ccRCC), accounting for approximately 75–80 % of all RCC cases. This predominance underscores its clinical significance, with important implications for diagnosis, therapeutic decision-making, and prognostic assessment [
2,
3,
4,
5,
6]. ccRCC is characterized by a more aggressive biological behavior and poorer prognosis compared to other subtypes, such as papillary or chromophobe RCC [
7,
8]. These observations highlight that timely identification of ccRCC is critical for accurate risk stratification, informed decisions between surgical intervention and active surveillance, and the optimal selection of systemic, targeted, or immunotherapeutic strategies [
4,
9,
10].
The genetic hallmark of ccRCC, most commonly involving alterations in the VHL gene, represents a key driver of tumor development, leading to pronounced vascularization and an elevated metastatic potential [
11]. Despite advances in imaging and modern early detection strategies, approximately 20–30 % of patients are diagnosed with metastatic disease at initial presentation [
12]. This is largely attributable to the frequently asymptomatic nature of early-stage RCC, with clinical manifestations becoming apparent only after regional or distant dissemination has occurred [
13]. Metastases in ccRCC most commonly involve the lungs (∼45 % of cases), bones (∼30 %), lymph nodes (∼22 %), and liver (∼20 %), reflecting the aggressive behavior of the disease [
14,
15,
16]. In contrast, metastasis to the urinary bladder is exceedingly rare, reported in approximately 2 % of cases, with some studies documenting only a few dozen instances worldwide [
17,
18,
19]. The rarity of bladder involvement is largely explained by the anatomical organization of the venous circulation and the relatively “isolated” pathway of the urinary tract. Several mechanisms have been proposed, including retrograde venous flow through the renal and gonadal venous plexuses, lymphatic reflux, and hematogenous microembolization [
20]. In select cases, tumor cell implantation via urinary “shedding” has also been suggested, particularly when synchronous involvement of the ureter or bladder is present [
21,
22]. Clinically, such metastases often present with gross hematuria, which can mimic primary urothelial carcinoma and potentially delay diagnosis [
17].
The present study aims to document an exceptionally rare case of ccRCC with synchronous bladder metastasis and to provide a comprehensive analysis of the diagnostic, surgical, and pathological aspects of such a presentation. The report integrates detailed imaging findings, histopathological and immunohistochemical assessments, as well as the operative strategy involving robot-assisted nephro-adrenalectomy with lymphadenectomy, illustrating a contemporary multidisciplinary approach to the management of advanced ccRCC. The novelty of this manuscript lies in its systematic presentation of the rare phenomenon of bladder metastasis, supported by clinical, pathological, and therapeutic observations, thereby expanding current knowledge on ccRCC biology and offering practical guidance for the recognition, differentiation, and individualized management of these patients. This case underscores the importance of long-term follow-up and careful identification of atypical metastatic sites to optimize prognosis and therapeutic outcomes in high-risk RCC patients.
2. Case Presentation
2.1. Patient Information
The patient is a 69-year-old male who was admitted to the Department of Nephrology and Urology at St. Marina University Hospital – Varna in October 2025, presenting with recurrent, painless gross hematuria and mild dysuria of approximately two weeks’ duration. His past medical history is notable for type 2 diabetes mellitus, arterial hypertension, and stage 2 chronic kidney disease according to KDIGO criteria. The patient reported no occupational exposures, is a non-smoker, and has no family history of malignancy.
2.2. Clinical Findings
Upon admission, the patient was hemodynamically stable and afebrile. Physical examination revealed normal cardiovascular and respiratory findings. The abdomen was soft and non-tender, without palpable renal masses or organomegaly. Laboratory tests showed mild normocytic anemia, slightly elevated serum creatinine, and borderline high urea. Urinalysis revealed significant hematuria, mild proteinuria, and glucosuria. The key laboratory results are summarized in
Table 1.
2.3. Diagnostic Assessment and Treatment Procedures
2.3.1. Imaging
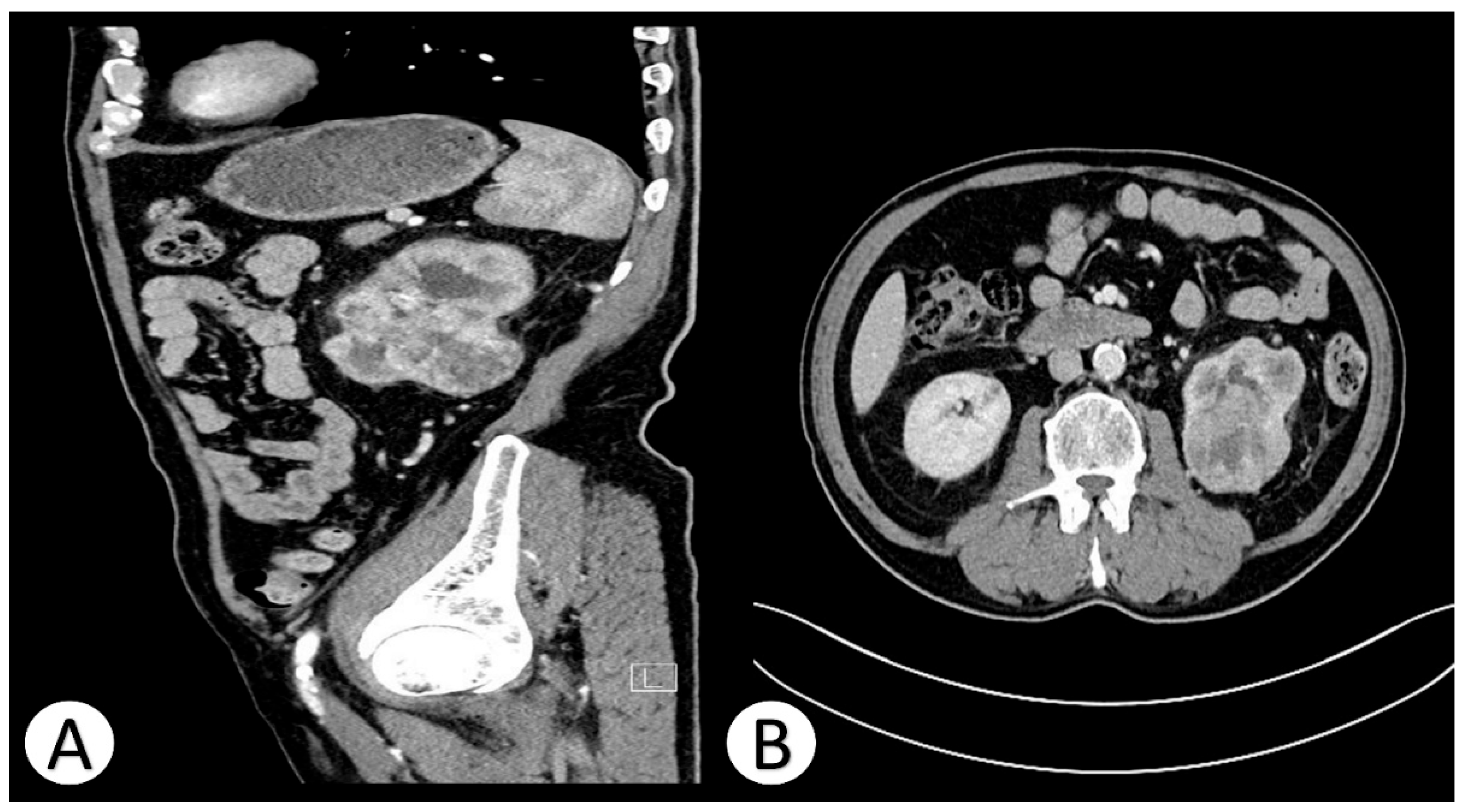
Contrast-enhanced computed tomography (CT) of the abdomen revealed a large (51 × 61 × 52 mm), heterogeneous, well-circumscribed mass originating from the mid to lower pole of the left kidney. The lesion exhibited mixed density with solid and necrotic components and heterogeneous contrast enhancement, consistent with a hypervascular renal tumor. Multiple bilateral pulmonary nodules (up to 24 mm) were also identified, highly suspicious for metastatic deposits. Additionally, para-aortic and paracaval lymphadenopathy (up to 18 mm) was visualized (
Figure 1).
2.3.2. Transurethral Resection (TUR)
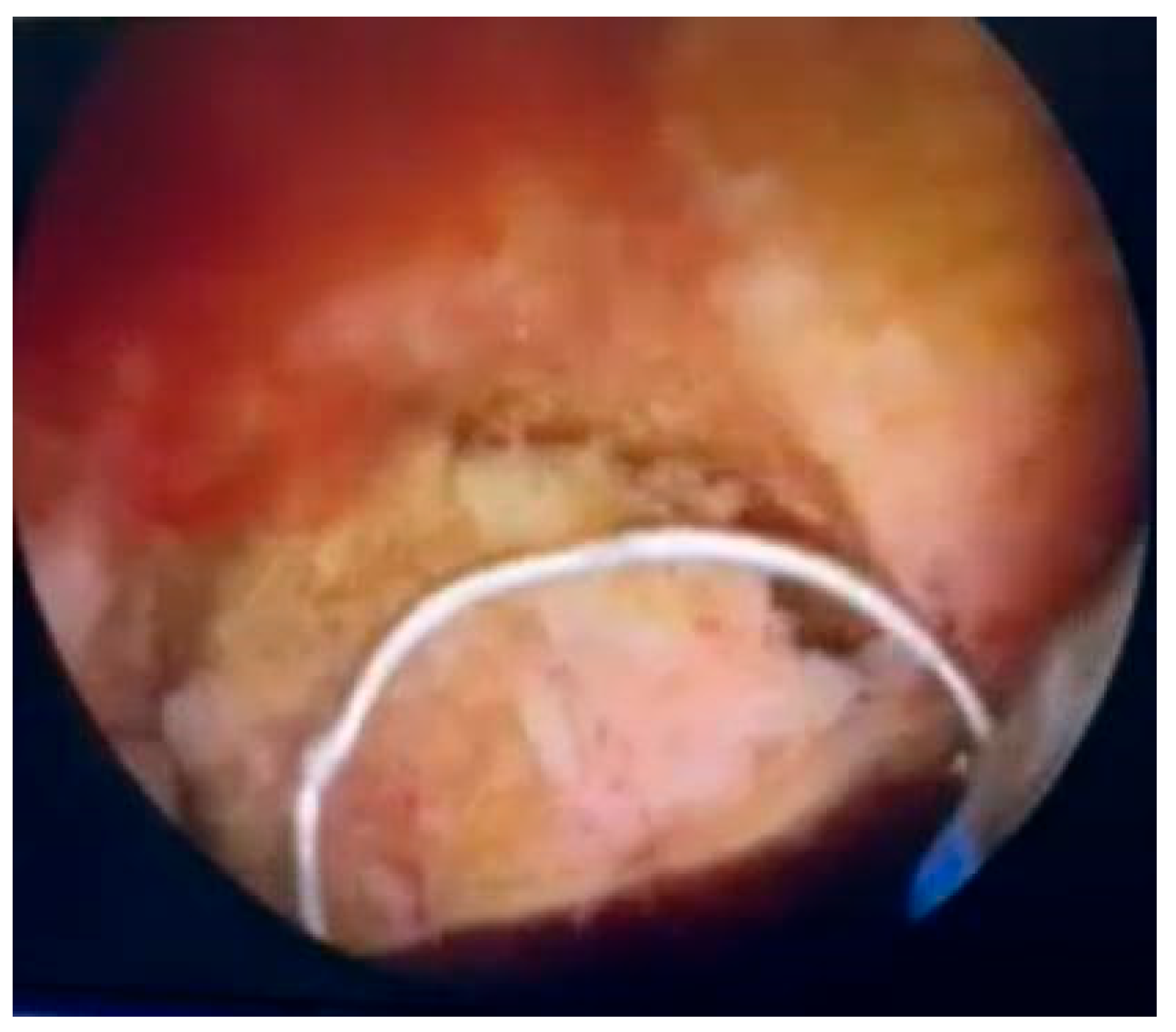
Due to marked bladder wall thickening and the presence of hematuria, the patient underwent transurethral resection (TUR) of a 4-cm papillary lesion located at the bladder dome on 3 October 2025. Intraoperatively, the lesion appeared highly vascularized with a yellowish hue (
Figure 2). The resection was complete, and hemostasis was adequately achieved. The excised tissue was submitted for histopathological and immunohistochemical analysis.
2.3.3. Histopathology and Immunohistochemistry (Post-TUR)
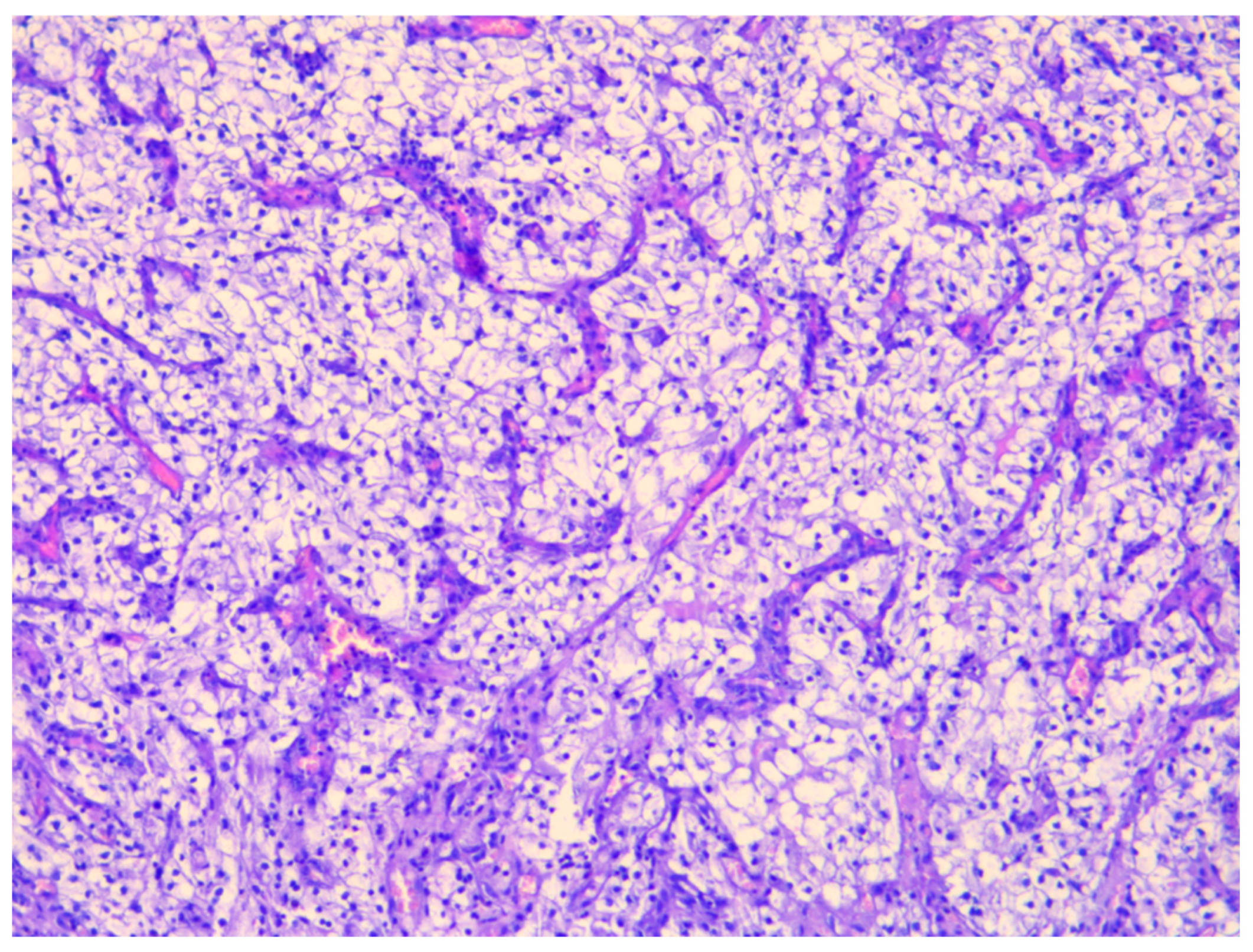
Histopathological examination revealed nests and alveolar arrangements of atypical clear cells with optically empty cytoplasm, well-defined cell borders, and hyperchromatic nuclei, organized around thin-walled, branching blood vessels. Foci of necrosis and hemorrhage were also observed. These morphological features were consistent with metastatic ccRCC, confirming secondary involvement of the urinary bladder from a primary renal tumor (
Figure 3).
Immunohistochemical profiling further corroborated the diagnosis of metastatic ccRCC with bladder involvement. Tumor cells exhibited positive staining for PAX8, CD10, and CAIX, and were negative for CK7 and GATA3. This immunophenotype is characteristic of ccRCC and allows clear distinction from primary urothelial carcinoma [
23,
24]. These findings underscore the critical importance of integrating detailed morphological assessment with immunohistochemical verification for the accurate identification of extrarenal metastatic sites of ccRCC and for guiding informed decisions regarding optimal therapeutic strategies.
2.3.4. Robot-Assisted Left Nephro-Adrenalectomy with Para-Aortic Lymphadenectomy
The patient underwent a planned radical robot-assisted left nephro-adrenalectomy with para-aortic lymphadenectomy on 28 October 2025, using the Da Vinci Xi surgical platform. This approach enabled a minimally invasive, precise, and oncologically safe excision of a large renal mass, including the adjacent adrenal gland and regional lymph nodes.
Intraoperatively, the left kidney exhibited marked hypertrophy (21 × 13 × 10 cm) and contained a nodular, yellow-ochre tumor mass measuring 7.5 cm, infiltrating the renal hilum and surrounding perirenal fat. The adrenal gland and several enlarged para-aortic lymph nodes were resected en bloc, allowing comprehensive histopathological assessment of tumor spread. No peritoneal deposits or other intra-abdominal abnormalities were identified.
The procedure was completed in 80 minutes without intraoperative complications. Hemodynamic parameters remained stable throughout, and the estimated blood loss was 200 mL. The robot-assisted approach provided exceptional precision during dissection near critical vascular structures and facilitated optimal oncologic resection, which is crucial for patients with locally advanced or metastatic ccRCC.
2.3.5. Histopathology Following Nephro-Adrenalectomy
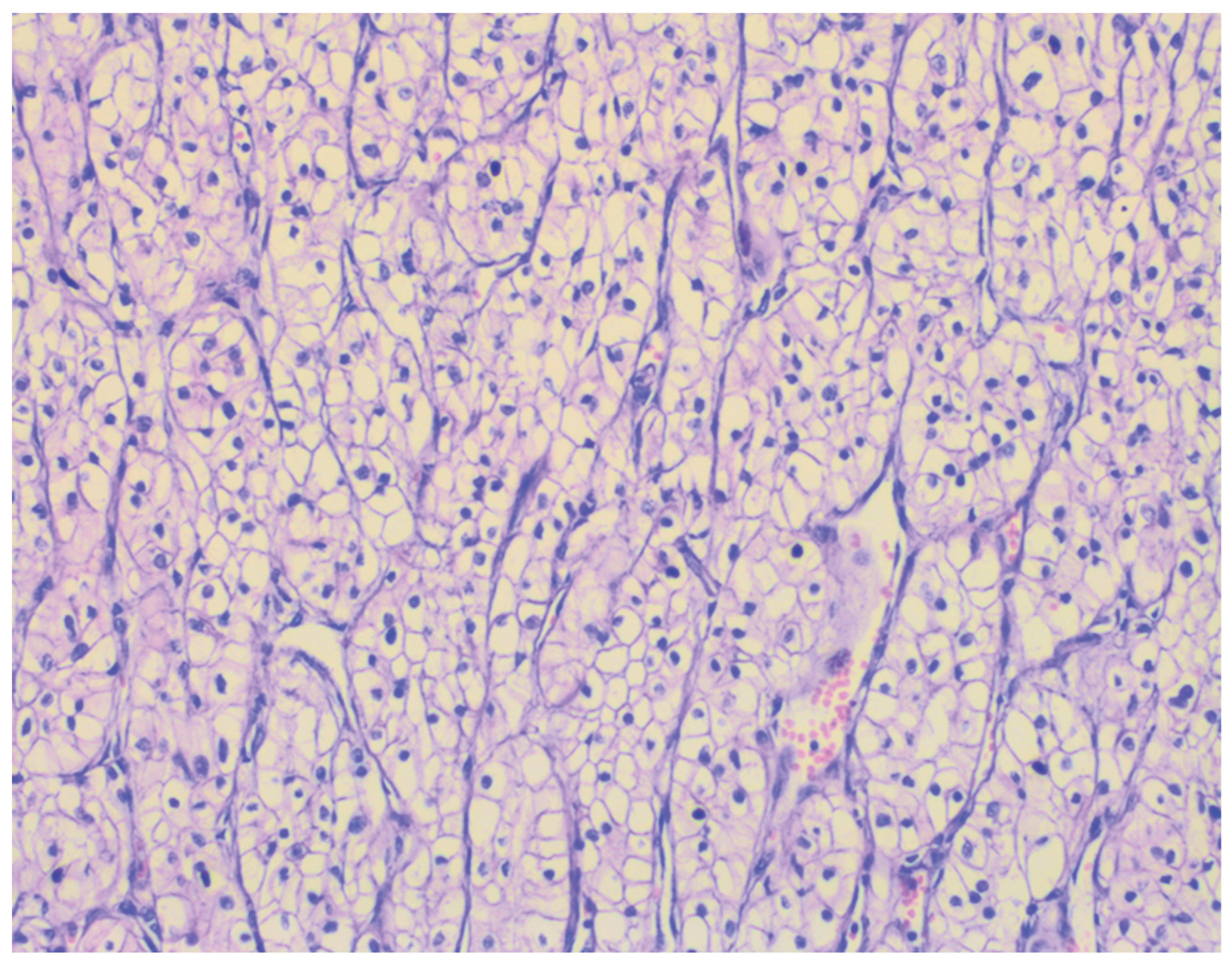
Histopathological analysis confirmed the diagnosis of ccRCC, staged as pT3aN1M1, ISUP grade 2, with evidence of lymphovascular invasion (LVI+). The tumor cells exhibited classic clear cytoplasm and delicate vascular architecture characteristic of ccRCC, correlating with a high potential for both local and distant metastasis.
Metastatic involvement of the adrenal gland and para-aortic lymph nodes was confirmed, establishing stage IV disease and indicating the need for ongoing systemic therapy [
25,
26]. Notably, the surgical resection margins, ureter, and hilar vessels remained free of tumor infiltration, demonstrating complete oncologic excision of the primary lesion and regional structures (
Figure 4). These findings underscore the critical importance of combining radical surgical resection with precise histopathological assessment for accurate oncologic staging and prognosis, as well as for the strategic planning of subsequent systemic therapy in patients with metastatic ccRCC.
2.4. Follow-Up and Outcomes
The postoperative course was uneventful. The patient remained afebrile, with satisfactory urine output and stable renal function, maintaining a serum creatinine level of 111 µmol/L. Overall clinical status was good, with no evidence of hemodynamic instability or perioperative complications. Five days following the robot-assisted procedure, the patient was discharged with clear instructions for regular follow-up and initiation of systemic oncologic therapy.
The standard therapeutic regimen comprised a combination of an immune checkpoint inhibitor (ICI) and a tyrosine kinase inhibitor (TKI), a strategy that has demonstrated efficacy in patients with stage IV metastatic ccRCC. This approach aims to control residual micrometastatic disease and improve long-term survival. The management exemplifies an integrated, multidisciplinary model for advanced RCC, combining radical surgical resection with targeted systemic therapy to optimize oncologic outcomes.
3. Discussion
This clinical case illustrates an exceptionally rare presentation of metastatic ccRCC with secondary involvement of the urinary bladder. Such occurrences are reported in only a small proportion of patients and are largely limited to isolated case reports and literature reviews, underscoring the importance of documenting these cases [
27]. Management of these patients requires careful consideration of the diagnostic challenges in differentiating metastatic lesions from primary urothelial carcinoma, as clinical symptoms and imaging findings frequently overlap. This is particularly relevant in the context of gross hematuria, with or without irritative lower urinary tract symptoms, which was also observed in our patient. Imaging findings may reveal solitary or multifocal bladder wall thickenings that are indistinguishable from primary bladder carcinoma. In this context, accurate identification of the metastatic origin of the bladder lesion through combined morphologic assessment and immunohistochemical profiling (PAX8+, CD10+, CAIX+, CK7–, GATA3–) was critical to confirm metastatic ccRCC and exclude primary urothelial carcinoma. These immunohistochemical markers have been previously described as highly specific and clinically valuable for differentiating ccRCC from urothelial tumors [
28].
In our patient, synchronous involvement of the kidney and urinary bladder supports a hematogenous route of dissemination [
17,
29,
30]. Transurethral resection (TUR) of the bladder lesion provided prompt symptomatic relief and enabled definitive diagnostic confirmation. Histopathology revealed classic clear cytoplasm, delicate vascular networks, and lymphovascular invasion, features associated with high metastatic potential and justifying comprehensive surgical management.
In this case, the decision to perform radical robot-assisted nephro-adrenalectomy with para-aortic lymphadenectomy using the Da Vinci Xi platform reflects a contemporary multidisciplinary approach to locally advanced urologic malignancies. Robotic surgery has become one of the most widely employed minimally invasive techniques in urology for the management of renal malignancies, including RCC, owing to enhanced three-dimensional visualization, articulated instruments with extensive freedom of movement, and tremor filtration, which collectively facilitate precise oncologic resection [
31,
32,
33,
34]. Key advantages of robotic surgery include reduced intraoperative blood loss and shorter hospital stays, as demonstrated in systematic reviews. Coco et al. (2025), in a systematic review of 30 studies including 5,432 patients undergoing robotic nephrectomy, reported significantly lower mean blood loss (−85 mL) and shorter hospitalization (−1.3 days) compared with open surgery, without compromising resection radicality [
33]. These findings are corroborated by other systematic reviews and retrospective analyses [31, 35−38]. Reduced intraoperative blood loss and expedited recovery are particularly important for patients with large or locally advanced tumors, as in the present case, minimizing surgical stress and early postoperative complications [
36]. Furthermore, the use of robotic systems markedly enhances intraoperative visualization and dissection precision, enabling surgeons to operate confidently within confined anatomical spaces while achieving negative resection margins without increasing the incidence of positive surgical edges—a critical prognostic determinant in oncologic procedures. These attributes render robotic technology particularly well-suited for complex urologic interventions, including nephron-sparing approaches such as partial nephrectomy, where preservation of renal parenchyma is essential [
31]. It is perhaps for this reason that, in a forward-looking perspective, some authors argue that the advent of minimally invasive surgery in the robotic era necessitates that comparative analyses be conducted not merely between open and laparoscopic approaches, but among the various robotic platforms themselves [
39]. Conversely, other sources highlight certain limitations of robotic surgery, including the absence of direct tactile feedback, which necessitates reliance on visual cues during delicate dissections, as well as longer operative times and higher costs associated with equipment and consumables [
35,
40,
41]. Systematic reviews indicate that, despite superior short-term outcomes—such as reduced intraoperative blood loss and shorter hospitalization—robust evidence demonstrating improved long-term survival compared with open or laparoscopic surgery remains limited, underscoring the necessity for careful patient selection and specialized surgical team training [
33,
42,
43,
44]. We fully concur with this assessment; however, we posit that generating more compelling evidence requires broader documentation and dissemination of similar clinical cases, a need that becomes particularly critical in the context of rare presentations, such as that observed in our patient.
Despite complete surgical resection, the presence of pulmonary, lymphatic, and adrenal metastases corresponded to stage IV disease, necessitating the initiation of systemic therapy. Over the past decade, the management of metastatic RCC has undergone a paradigm shift with the advent of ICI-based therapies, administered either as monotherapy or in combination with TKIs. These strategies stem from the recognition that modulation of the host immune response against tumor cells can potentiate antineoplastic efficacy and improve clinical outcomes beyond those achieved with conventional targeted therapies [
45]. The introduction of ICI + TKI combinations as first-line therapy for metastatic RCC is supported by multiple phase III clinical trials and meta-analyses, which have demonstrated substantially improved survival outcomes compared with monotherapy. Combinations of Programmed Death-1 (PD-1) or Programmed Death-Ligand 1 (PD-L1) inhibitors (e.g., nivolumab, pembrolizumab) with Vascular Endothelial Growth Factor (VEGF) inhibitors (e.g., axitinib, cabozantinib, lenvatinib) have shown superior progression-free survival and overall survival relative to standard sunitinib therapy in patients with metastatic RCC, thereby establishing these regimens as the new first-line standard of care. Meta-analyses further indicate that specific combinations, such as nivolumab + cabozantinib and pembrolizumab + lenvatinib, substantially reduce the risk of mortality compared with standard therapies (e.g., sunitinib), with this survival benefit maintained across diverse patient populations. Moreover, other regimens, including pembrolizumab + axitinib, avelumab + axitinib, and nivolumab + ipilimumab, have also demonstrated improved overall survival relative to traditional TKI monotherapy [
46]. Clinical registry data support the favorable outcomes of ICI + TKI therapy. In a retrospective study of 51 metastatic RCC patients, ICI + TKI achieved >80 % overall survival at 12 months, median progression-free survival of 19 months, and an objective response rate of ~69 % [
47]. While highly effective as first-line therapy, efficacy in second- or later-line settings decreases with prior treatments, though regimens remain safe and feasible in selected patients [
48]. Predictive biomarkers, including PD-L1 status, are under investigation but not yet fully reliable. Toxicity is variable, encompassing immune- and TKI-related adverse events, yet the overall safety profile allows sustained therapy and improved outcomes [
49]. Evidence from trials and real-world cohorts confirms ICI + TKI as an effective, safe first-line option for metastatic RCC, improving survival and response rates and highlighting the importance of multidisciplinary management [
50,
51,
52,
53,
54,
55,
56].
Against the backdrop of the proven benefits of contemporary immuno-oncologic strategies, clinical outcomes in patients with advanced and metastatic disease remain highly heterogeneous, largely dependent on tumor burden, the intrinsic biological characteristics of the neoplasm, and individual responses to systemic therapy. Literature data indicate that the median survival following the diagnosis of bladder metastases ranges from 6 to 30 months, underscoring the aggressive nature of the disease and the critical need to optimize therapeutic strategies [
57]. In this context, the present clinical case, together with an analysis of current evidence, highlights that early detection of metastatic spread, cytoreductive surgery when oncologically justified, and the implementation of modern systemic therapies constitute fundamental and mutually complementary components of comprehensive patient management. Such a multimodal and individualized approach not only provides the potential to prolong overall survival but also contributes to improved disease control, symptom alleviation, and enhanced quality of life. These objectives remain the paramount goals in the management of patients with advanced RCC.
4. Conclusion
The presented clinical case illustrates an exceptionally rare and clinically challenging presentation of ccRCC with synchronous bladder metastasis, highlighting the complexity of both diagnostic and therapeutic management in advanced disease. The atypical metastatic site, clinically manifesting as gross hematuria, necessitated an integrated, multimodal diagnostic approach, encompassing advanced imaging, detailed morphological assessment, and extensive immunohistochemical profiling to reliably distinguish metastatic ccRCC from primary urothelial carcinoma.
This case underscores the pivotal role of cytoreductive surgery, performed via robot-assisted nephro-adrenalectomy with lymphadenectomy, as part of a contemporary multidisciplinary framework for managing locally advanced and metastatic ccRCC. The Da Vinci Xi robotic platform enabled precise and oncologically adequate resection of the primary tumor and regional metastatic structures with minimal operative morbidity, thereby optimizing conditions for subsequent systemic therapy. Despite complete surgical excision, the presence of pulmonary, lymphatic, and adrenal metastases defined stage IV disease and warranted initiation of systemic therapy. Current evidence strongly supports the use of immune checkpoint inhibitors, alone or in combination with tyrosine kinase inhibitors, as first-line therapy in metastatic RCC, demonstrating significant improvements in both overall and progression-free survival. Nevertheless, clinical prognosis remains heterogeneous, particularly in patients with rare metastatic sites such as the bladder, emphasizing the importance of individualized therapeutic strategies.
In conclusion, this case highlights the necessity for heightened clinical vigilance, long-term follow-up, and interdisciplinary collaboration in ccRCC management. Furthermore, the documentation and publication of rare clinical presentations are critical to expanding understanding of tumor biology and behavior. A multimodal, personalized approach represents a cornerstone for optimizing oncological control, prolonging survival, and enhancing quality of life in patients with advanced RCC.
Author Contributions
For research articles with several authors, a short paragraph specifying their individual contributions must be provided. The following statements should be used “Conceptualization, M.B.-P., A.P., P.A., P.K., S. M., D. M., and S. S.-G.; methodology, M.B.-P., A.P., P.A., P.K., S. M., D. M., and S. S.-G.; software, M.B.-P., A.P., P.A., P.K., S. M; validation, M.B.-P., A.P., P.A., P.K., S. M., D. M., and S. S.-G.; formal analysis, M.B.-P., A.P., P.A., P.K., S. M., D. M., and S. S.-G.; investigation, M.B.-P., A.P., P.A., P.K., S. M., D. M., and S. S.-G.; resources, S. M., D. M., and S. S.-G. ; data curation, M.B.-P., A.P., P.A., P.K., S. M., D. M., and S. S.-G.; writing—original draft preparation, M.B.-P., D. M., and S. S.-G. ; writing—review and editing, S. S.-G. ; visualization, M.B.-P., A.P., P.A., P.K., S. M., D. M., and S. S.-G.; supervision, M.B.-P., S. M., D. M., and S. S.-G.; project administration, M.B.-P., A.P., P.A., P.K., S. M., D. M., and S. S.-G.; funding acquisition, M.B.-P., A.P., P.A., P.K., S. M., D. M., and S. S.-G. All authors have read and agreed to the published version of the manuscript.”.
Funding
Funding for this study was provided by the European Union – NextGenerationEU, within Scientific Group 3.2.3. RenEVA, under contract No. BG-RRP-2.004-0009-C02.
Institutional Review Board Statement
This study is retrospective and relies solely on medical data that had already been collected for other clinical purposes. Therefore, no approval was required from an ethics committee or an institutional review board, as no new interventions were performed on patients and all data were obtained from existing medical records.
Informed Consent Statement
Written informed consent was obtained from the patient for publication of this case report.
Data Availability Statement
No new datasets were generated or analyzed during the preparation of this case report.
Acknowledgments
The authors express their gratitude to the multidisciplinary teams from the Departments of Nephrology, Urology, and Pathology at University Hospital “St. Marina” – Varna, for their contribution to patient management. Special thanks to the patient and her family for their cooperation and consent to publication.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| ccRCC |
Clear cell renal cell carcinoma |
| CT |
Computed tomography |
| ICI |
Immune checkpoint inhibitors |
| PD-1 |
Programmed Death-1 |
| PD-L1 |
Programmed Death-Ligand 1 |
| RCC |
Renal cell carcinoma |
| TKI |
Tyrosine kinase inhibitor |
| TUR |
Transurethral resection |
References
- Escudier, B.; Eisen, T.; Porta, C.; Patard, J.J.; Khoo, V.; Algaba, F.; Mulders, P.; Kataja, V. on behalf of the ESMO Guidelines Working Group. Renal cell carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2012, 23 (Suppl. 7), vii65–vii71. [Google Scholar] [CrossRef] [PubMed]
- Capitanio, U.; Montorsi, F. Renal cancer. Lancet 2016, 387, 894–906. [Google Scholar] [CrossRef] [PubMed]
- Chevrier, S.; Levine, J.H.; Zanotelli, V.R.T.; Silina, K.; Schulz, D.; Bacac, M.; Ries, C.H.; Ailles, L.; Jewett, M.A.S.; Moch, H.; van den Broek, M.; Beisel, C.; Stadler, M.B.; Gedye, C.; Reis, B.; Pe’er, D.; Bodenmiller, B. An immune atlas of clear cell renal cell carcinoma. Cell 2017, 169, 736–749.e18. [Google Scholar] [CrossRef] [PubMed]
- Padala, S.A.; Barsouk, A.; Thandra, K.C.; Saginala, K.; Mohammed, A.; Vakiti, A.; Rawla, P.; Barsouk, A. Epidemiology of renal cell carcinoma. World J. Oncol. 2020, 11, 79–87. [Google Scholar] [CrossRef]
- Rose, T.L.; Kim, W.Y. Renal cell carcinoma: A review. JAMA 2024, 332, 1001–1010. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2024. CA Cancer J. Clin. 2024, 74, 7–33. [Google Scholar] [CrossRef]
- Cheville, J.C.; Lohse, C.M.; Zincke, H.; Weaver, A.L.; Blute, M.L. Comparisons of outcome and prognostic features among histologic subtypes of renal cell carcinoma. Am. J. Surg. Pathol. 2003, 27, 612–624. [Google Scholar] [CrossRef]
- Deng, J.; Li, L.; Xia, H.; Guo, J.; Wu, X.; Yang, X.; Hong, Y.; Chen, Q.; Hu, J. A comparison of the prognosis of papillary and clear cell renal cell carcinoma: Evidence from a meta-analysis. Med. 2019, 98, e16309. [Google Scholar] [CrossRef]
- Schiavoni, V.; Campagna, R.; Pozzi, V.; Cecati, M.; Milanese, G.; Sartini, D.; Salvolini, E.; Galosi, A.B.; Emanuelli, M. Recent Advances in the Management of Clear Cell Renal Cell Carcinoma: Novel Biomarkers and Targeted Therapies. Cancers 2023, 15, 3207. [Google Scholar] [CrossRef]
- Sepp, T.; Poyhonen, A.; Uusküla, A.; Kotsar, A.; Veitonmäki, T.; Tammela, T.L.J.; Baburin, A.; Murtola, T.J. Renal cancer survival in clear cell renal cancer compared to other types of tumor histology: A population-based cohort study. PLoS ONE 2025, 20, e0329000. [Google Scholar] [CrossRef]
- Pham, D.X.; Hsu, T. Tumor-initiating and metastasis-initiating cells of clear-cell renal cell carcinoma. J. Biomed. Sci. 2025, 32, 17. [Google Scholar] [CrossRef] [PubMed]
- Motzer, R.J.; Jonasch, E.; Agarwal, N.; Alva, A.; Bagshaw, H.; Baine, M.; Beckermann, K.; Carlo, M.I.; Choueiri, T.K.; Costello, B.A.; Derweesh, I.H.; Desai, A.; Ged, Y.; George, S.; Gore, J.L.; Gunn, A.; Haas, N.; Johnson, M.; Kapur, P.; King, J.; Kyriakopoulos, C.; Lam, E.T.; Lara, P.N.; Lau, C.; Lewis, B.; Madoff, D.C.; Manley, B.; Michaelson, M.D.; Mortazavi, A.; Ponsky, L.; Ramalingam, S.; Shuch, B.; Smith, Z.L.; Sosman, J.; Sweis, R.; Zibelman, M.; Schonfeld, R.; Stein, M.; Gurski, L.A. NCCN Guidelines® Insights: Kidney Cancer, Version 2.2024. J. Natl. Compr. Canc. Netw. 2024, 22, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Ouellet, S.; Binette, A.; Nguyen, A.; et al. Metastatic renal cell carcinoma initially presenting with hematochezia and subsequently with vaginal bleeding: A case report. BMC Urol. 2018, 18, 4. [Google Scholar] [CrossRef] [PubMed]
- Dudani, S.; de Velasco, G.; Wells, J.C.; et al. Evaluation of clear cell, papillary, and chromophobe renal cell carcinoma metastasis sites and association with survival. JAMA Netw. Open 2021, 4, e2021869. [Google Scholar] [CrossRef]
- Rony, M.; Ratone, J.P.; Walz, J.; Pignot, G.; Caillol, F.; Pesenti, C.; et al. Gastrointestinal metastases from primary renal cell cancer: A single center review. Front. Oncol. 2021, 11, 644301. [Google Scholar] [CrossRef]
- Lee, C.H.; Kang, M.; Kwak, C.; Ko, Y.H.; Kim, J.K.; Park, J.Y.; Bang, S.; Seo, S.I.; Suh, J.; Song, W.; Song, C.; Lee, H.H.; Chung, J.; Jeong, C.W.; Jo, J.K.; Choi, S.H.; Choi, J.; Choi, C.; Choo, S.H.; Han, J.H.; Hwang, E.C. Sites of metastasis and survival in metastatic renal cell carcinoma: Results from the Korean Renal Cancer Study Group database. J. Korean Med. Sci. 2024, 39, e293. [Google Scholar] [CrossRef]
- Doo, S.W.; Kim, W.B.; Kim, B.K.; Yang, W.J.; Yoon, J.H.; Jin, S.Y.; Song, Y.S. Metastasis of renal cell carcinoma to the bladder. Korean J. Urol. 2013, 54, 69–72. [Google Scholar] [CrossRef]
- Manav, A.N.; Şahin, G. Rare metastasis of renal cell carcinoma to the bladder with a new hypothesis and literature review: Case report. Preprint 2024, Version 1; Research Square. [CrossRef]
- Ali, R.N.; Anwaar, A.; Irfan, W.; Hameed, F.; Haq, I.U.; Asif Raza, M.; Karamat, R.I.; Akilimali, A. Metastatic clear renal cell carcinoma in urinary bladder at presentation—A rare incidental case report and literature review. Clin. Case Rep. 2025, 13, e70199. [Google Scholar] [CrossRef]
- Arpita, S.; Manoj, J.; Kaushik, S.; Anish, S. Metastasis of renal cell carcinoma to urinary bladder: A rare case report with review of literature. J. Lab. Physicians 2017, 9, 322–324. [Google Scholar] [CrossRef]
- Adhiguna, M.A.; Pramod, S.V.; Noegroho, B.S.; Safriadi, F.; Hernowo, B.S. Case of metachronous clear cell bladder metastasis on low risk localized renal cell carcinoma patient. Int. J. Surg. Case Rep. 2022, 96, 107158. [Google Scholar] [CrossRef]
- Agarwal, V.; Yuvaraja, T.B.; Waigankar, S.; Shah, A.; Asari, A.; Kulkarni, B.; R, A. Solitary Metachronous Drop Metastasis of a Rare Variant of Renal Cell Carcinoma to Ipsilateral Ureteric Stump: A Case Report. Bull. Urooncol 2023, 22, 46–49. Available online: https://uroonkolojibulteni.com/pdf/7970e68b-97ce-406f-9dd8-2946cf184dcf/articles/uob.galenos.2022.2022.7.1/UOB-22-46-En.pdf (accessed on 4 February 2026). [CrossRef]
- Carvalho, J.C.; Thomas, D.G.; McHugh, J.B.; Shah, R.B.; Kunju, L.P. p63, CK7, PAX8 and INI-1: An optimal immunohistochemical panel to distinguish poorly differentiated urothelial cell carcinoma from high-grade tumours of the renal collecting system. Histopathology 2012, 60, 597–608. [Google Scholar] [CrossRef] [PubMed]
- Alshenawy, H.A. Immunohistochemical panel for differentiating renal cell carcinoma with clear and papillary features. J. Microsc. Ultrastruct. 2015, 3, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Roskoski, R., Jr. Combination immune checkpoint and targeted protein kinase inhibitors for the treatment of renal cell carcinomas. Pharmacol. Res. 2024, 203, 107181. [Google Scholar] [CrossRef]
- Medscape. Renal Cell Carcinoma Treatment Protocols. Medscape eMedicine. 2025. Available online: https://emedicine.medscape.com/article/2007277-overview (accessed on 5 February 2026).
- Romero Uribe, J.L.; Cornejo Davila, V.; Cruz Ruiz, J. Synchronous bladder metastasis from chromophobe renal cell carcinoma: A report of a rare case. Cureus 2025, 17, e86234. [Google Scholar] [CrossRef]
- Zachos, I.; Mitrakas, L.; Karasavvidou, F.; Karatzas, A.; Tzortzis, V. Clear-cell renal cell carcinoma single urinary bladder metastasis: A case report and review of the literature. J. Surg. Case Rep. 2022, 2022, rjac475. [Google Scholar] [CrossRef]
- Stamatakos, P.V.; Glykas, L.; Fragkoulis, C.; Theodoropoulou, G.; Pappa, S. Bladder metastasis of renal cell carcinoma: A case report and review of the literature. Hellenic Urol. 2021, 33, 59–60. [Google Scholar] [CrossRef]
- Urushibara, M.; Nagata, M.; Okumura, T.; et al. Bladder metastasis with additional metastases in multiple other organs 4 years after radical nephrectomy for clear cell renal cell carcinoma: A case report and review of the literature. J. Med. Case Rep. 2022, 16, 131. [Google Scholar] [CrossRef]
- Kallingal, G.J.; Swain, S.; Darwiche, F.; Punnen, S.; Manoharan, M.; Gonzalgo, M.L.; Parekh, D.J. Robotic partial nephrectomy with the Da Vinci Xi. Adv. Urol. 2016, 2016, 9675095. [Google Scholar] [CrossRef]
- Teo, X.L.; Lim, S.K. Robot-assisted nephroureterectomy: Current perspectives. Robotic Surg. 2016, 3, 37–48. [Google Scholar] [CrossRef]
- Coco, D.; Leanza, S.; Viola, M.G.; Coco, D. Systematic review of robotic nephrectomy for kidney cancer. J. Kidney Cancer VHL 2025, 12, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Nightingale, Grup Florence. Robotic Surgery Da Vinci Xi. Florence Hospital. Available online: https://www.florence.com.tr/bg-bg/medical-technologies/robotic-surgery-da-vinci-xi (accessed on 4 February 2026).
- Wu, Z.; Li, M.; Liu, B.; Cai, C.; Ye, H.; Lv, C.; Yang, Q.; Sheng, J.; Song, S.; Qu, L.; Xiao, L.; Sun, Y.; Wang, L. Robotic versus open partial nephrectomy: A systematic review and meta-analysis. PLoS ONE 2014, 9, e94878. [Google Scholar] [CrossRef] [PubMed]
- Crocerossa, F.; Carbonara, U.; Cantiello, F.; Marchioni, M.; Ditonno, P.; Mir, M.C.; Porpiglia, F.; Derweesh, I.; Hampton, L.J.; Damiano, R.; Autorino, R. Robot-assisted radical nephrectomy: A systematic review and meta-analysis of comparative studies. Eur. Urol. 2021, 80, 428–439. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Kobari, Y.; Iizuka, J.; Kondo, T.; Ishida, H.; Tanabe, K.; Takagi, T. Robot-assisted laparoscopic versus open partial nephrectomy for renal cell carcinoma in patients with severe chronic kidney disease. Int. J. Urol. 2022, 29, 1349–1355. [Google Scholar] [CrossRef]
- Calpin, G.G.; Ryan, F.R.; McHugh, F.T.; McGuire, B.B. Comparing the outcomes of open, laparoscopic and robot-assisted partial nephrectomy: A network meta-analysis. BJU Int. 2023, 132, 353–364. [Google Scholar] [CrossRef]
- Gupta, R.; Wang, H.; Xu, T.; Wang, Z.; Qiao, Z.; Li, J.; Ding, D.; Rahemi, M.H.; Bao, Y.; Dong, Z.; Liu, F.; Feng, Y.; Xu, Y.; Xu, W. Comparable perioperative performance between three robotic system for robot-assisted partial nephrectomy to treat early renal cancer. Sci. Rep. 2025, 15, 31402. [Google Scholar] [CrossRef]
- Murphy, D.; Challacombe, B.; Khan, M.S.; Dasgupta, P. Robotic technology in urology. Postgrad. Med. J. 2006, 82, 743–747. [Google Scholar] [CrossRef]
- Ahmed, K.; Khan, M.S.; Vats, A.; Nagpal, K.; Priest, O.; Patel, V.; Vecht, J.A.; Ashrafian, H.; Yang, G.Z.; Athanasiou, T.; Darzi, A. Current status of robotic assisted pelvic surgery and future developments. Int. J. Surg. 2009, 7, 431–440. [Google Scholar] [CrossRef]
- Jeong, I.G.; Khandwala, Y.S.; Kim, J.H.; et al. Association of robotic-assisted vs laparoscopic radical nephrectomy with perioperative outcomes and health care costs, 2003 to 2015. JAMA 2017, 318, 1561–1568. [Google Scholar] [CrossRef]
- Kızılay, F.; Turna, B.; Apaydın, E.; Semerci, B. Comparison of long-term outcomes of laparoscopic and robot-assisted laparoscopic partial nephrectomy. Kaohsiung J. Med. Sci. 2019, 35, 238–243. [Google Scholar] [CrossRef]
- Tang, A.B.; Lamaina, M.; Childers, C.P.; Mak, S.S.; Ruan, Q.; Begashaw, M.M.; Bergman, J.; Booth, M.S.; Shekelle, P.G.; Wilson, M.; Gunnar, W.; Maggard-Gibbons, M.; Girgis, M.D. Perioperative and long-term outcomes of robot-assisted partial nephrectomy: A systematic review. Am. Surg. 2021, 87, 21–29. [Google Scholar] [CrossRef]
- Stühler, V.; Rausch, S.; Maas, J.M.; Stenzl, A.; Bedke, J. Combination of immune checkpoint inhibitors and tyrosine kinase inhibitors for the treatment of renal cell carcinoma. Expert Opin. Biol. Ther. 2021, 21, 1215–1226. [Google Scholar] [CrossRef]
- Wang, J.; Li, X.; Li, M.; Liu, Q.; Xie, Z.; Si, X.; Yang, L.; Wang, Z.; Ding, D. Unveiling the best immune checkpoint inhibitor-based therapy for metastatic renal cell carcinoma in the first-line setting: an updated systematic review and Bayesian network analysis. Ther. Adv. Med. Oncol. 2025, 17, 17588359251353259. [Google Scholar] [CrossRef]
- Iinuma, K.; Yamada, T.; Kameyama, K.; Taniguchi, T.; Kawada, K.; Ishida, T.; Nagai, S.; Enomoto, T.; Ueda, S.; Takagi, K.; Kawase, M.; Takeuchi, S.; Kawase, K.; Kato, D.; Takai, M.; Nakane, K.; Koie, T. The efficacy and safety of immune checkpoint inhibitor and tyrosine kinase inhibitor combination therapy for advanced or metastatic renal cell carcinoma: A multicenter retrospective real-world cohort study. Cancers 2023, 15, 947. [Google Scholar] [CrossRef]
- Yang, Y.; Psutka, S.P.; Parikh, A.B.; Li, M.; Collier, K.; Miah, A.; Mori, S.V.; Hinkley, M.; Tykodi, S.S.; Hall, E.; Thompson, J.A.; Yin, M. Combining immune checkpoint inhibition plus tyrosine kinase inhibition as first and subsequent treatments for metastatic renal cell carcinoma. Cancers 2022, 11, 3106–3114. [Google Scholar] [CrossRef] [PubMed]
- Quhal, F.; Mori, K.; Bruchbacher, A.; Resch, I.; Mostafaei, H.; Pradere, B.; Schuettfort, V.M.; Laukhtina, E.; Egawa, S.; Fajkovic, H.; Remzi, M.; Shariat, S.F.; Schmidinger, M. First-line immunotherapy-based combinations for metastatic renal cell carcinoma: A systematic review and network meta-analysis. Eur. Urol. Oncol. 2021, 4, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Rini, B.I.; Plimack, E.R.; Stus, V.; Gafanov, R.; Hawkins, R.; Nosov, D.; Pouliot, F.; Alekseev, B.; Soulières, D.; Melichar, B.; Vynnychenko, I.; Kryzhanivska, A.; Bondarenko, I.; Azevedo, S.J.; Borchiellini, D.; Szczylik, C.; Markus, M.; McDermott, R.S.; Bedke, J.; Tartas, S.; Chang, Y.-H.; Tamada, S.; Shou, Q.; Perini, R.F.; Chen, M.; Atkins, M.B.; Powles, T.; KEYNOTE-426 Investigators. Pembrolizumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N. Engl. J. Med. 2019, 380, 1116–1127. [Google Scholar] [CrossRef] [PubMed]
- Tung, I.; Sahu, A. Immune checkpoint inhibitor in first-line treatment of metastatic renal cell carcinoma: A review of current evidence and future directions. Front. Oncol. 2021, 11, 707214. [Google Scholar] [CrossRef]
- Bosma, N.A.; Warkentin, M.T.; Gan, C.L.; Karim, S.; Heng, D.Y.C.; Brenner, D.R.; Lee-Ying, R.M. Efficacy and Safety of First-Line Systemic Therapy for Metastatic Renal Cell Carcinoma: A Systematic Review and Network Meta-Analysis. Eur. Urol. Open Sci. 2022, 37, 14–26. [Google Scholar] [CrossRef]
- Lalani, A.A.; Heng, D.Y.C.; Basappa, N.S.; Wood, L.; Iqbal, N.; McLeod, D.; Soulières, D.; Kollmannsberger, C. Evolving landscape of first-line combination therapy in advanced renal cancer: A systematic review. Ther. Adv. Med. Oncol. 2022, 14, 17588359221108685. [Google Scholar] [CrossRef]
- Yanagisawa, T.; Mori, K.; Kawada, T.; Katayama, S.; Uchimoto, T.; Tsujino, T.; Nishimura, K.; Adachi, T.; Toyoda, S.; Nukaya, T.; Fukuokaya, W.; Urabe, F.; Murakami, M.; Yamanoi, T.; Bekku, K.; Komura, K.; Takahara, K.; Hashimoto, T.; Fujita, K.; Azuma, H.; Ohno, Y.; Shiroki, R.; Uemura, H.; Araki, M.; Kimura, T. On behalf of the JKFOOT Study Group. First-Line Therapy for Metastatic Renal Cell Carcinoma: A Propensity Score-Matched Comparison of Efficacy and Safety. Urol. Oncol. 2024, 42, 374.e21–374.e29. [Google Scholar] [CrossRef]
- Motzer, R.J.; Escudier, B.; Burotto, M.; Powles, T.; Apolo, A.B.; Bourlon, M.T.; Shah, A.Y.; Porta, C.; Suárez, C.; Barrios, C.H.; Richardet, M.; Gurney, H.; Kessler, E.R.; Tomita, Y.; Bedke, J.; Scheffold, C.; Askelson, M.; Panzica, J.; Zhang, J.; van Kooten Losio, M.; Choueiri, T.K. Final Analysis of Nivolumab Plus Cabozantinib for Advanced Renal Cell Carcinoma from the Randomized Phase III CheckMate 9ER Trial. Ann. Oncol. 2026, 37, 33–43. [Google Scholar] [CrossRef]
- Zhong, X.; Huang, T.; Peng, Y.; Zhang, Y.; Li, J.; Chen, H.; Wang, L.; Liu, Q.; Zhou, S.; Gao, P.; Li, X.; Yang, H. First-line PD-1 Blockades Combined with TKIs for Metastatic Non-Clear Cell Renal Cell Carcinoma: A Real-World Multicenter Retrospective Study. Cancer Immunol. Immunother. 2026, 75, 48. [Google Scholar] [CrossRef]
- Talbi, O.; Hinaje, K.; Maadin, K.; Amaadour, L.; Oualla, K.; Benbrahim, Z.; Arifi, S.; Mellas, N. Prognostic Factors of Metastatic Renal Cell Carcinoma: Analysis of 95 Patients. Am. J. Biomed. Sci. Res. 2024, 22, 344–347. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |