Submitted:
06 February 2026
Posted:
09 February 2026
You are already at the latest version
Abstract
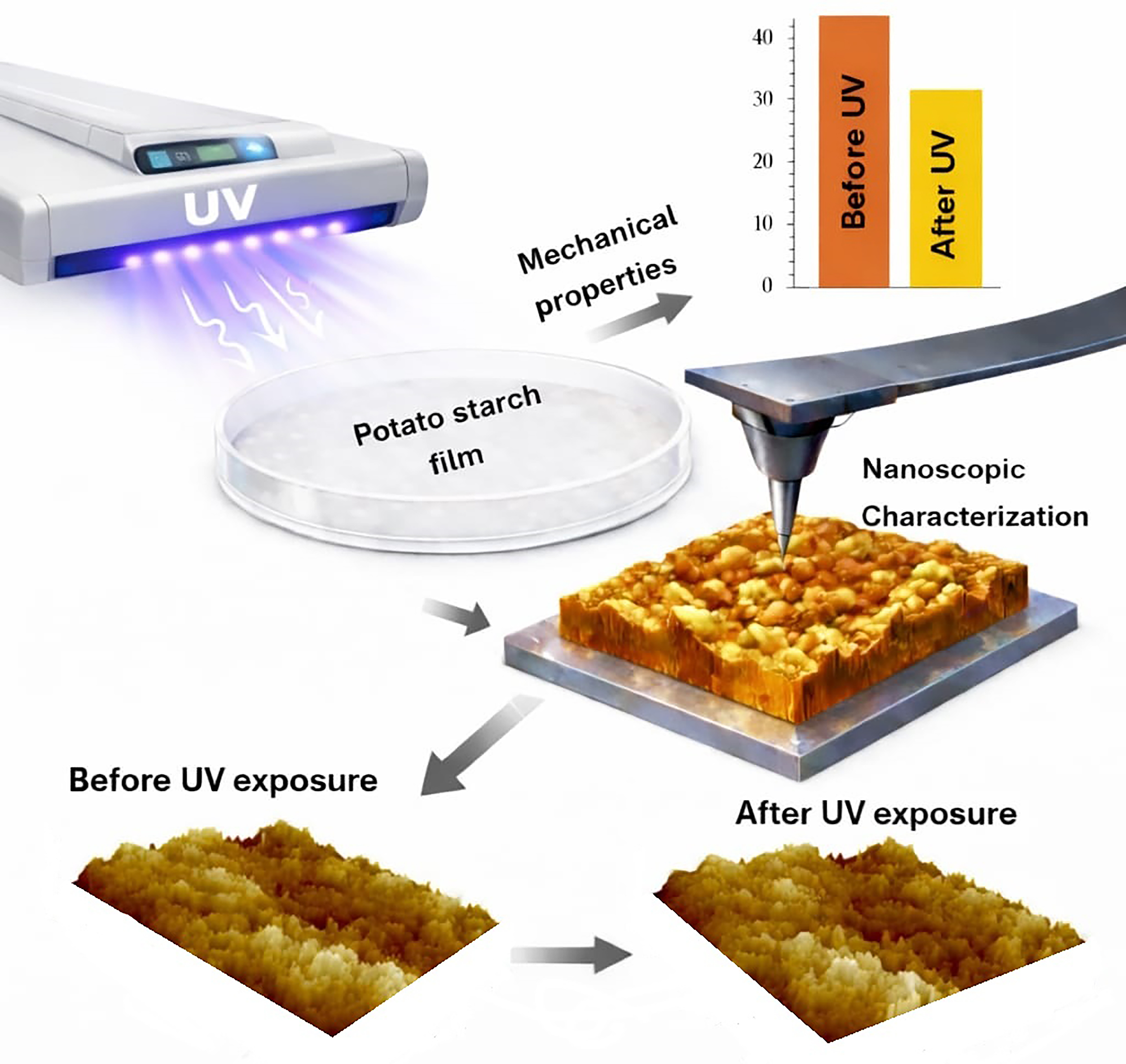
Keywords:
1. Introduction
2. Materials and Methods
2.1. Starch Extraction
2.2. Film Preparation
2.3. Optical Characterization
2.4. Mechanical Properties
2.5. Fourier Transform Infrared Spectroscopy (FTIR)
2.6. UV-Visible Optical Absorption and Opacity
2.8. Moisture Content (MC)
2.9. Water Total Soluble Matter (TSM)
2.10. Atomic Force Microscopy (AFM) Characterization
2.11. UV Exposure
2.12. Statistical Analysis
3. Results and Discussion
3.1. Optical Characterization of the Films
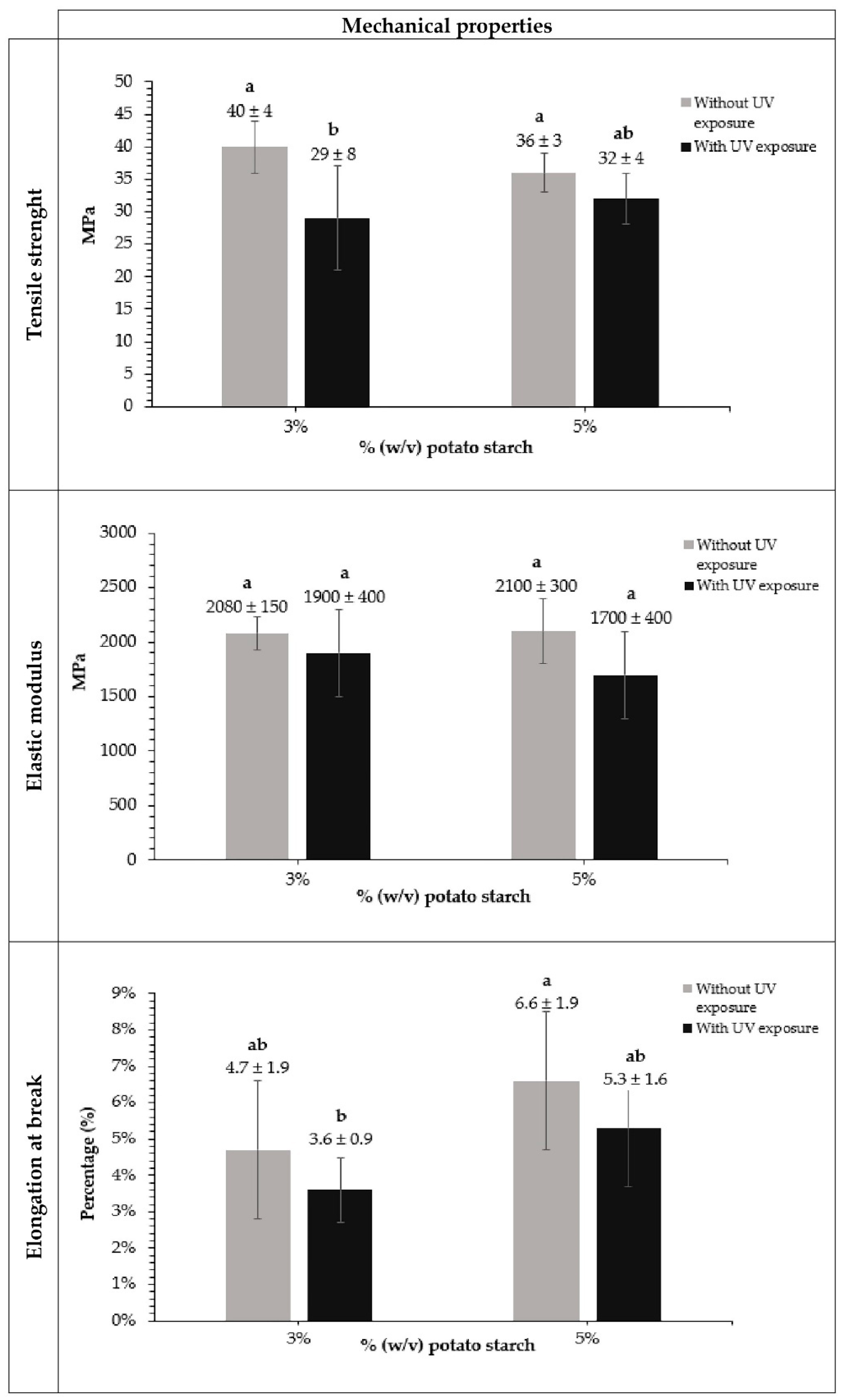
3.2. Mechanical Properties
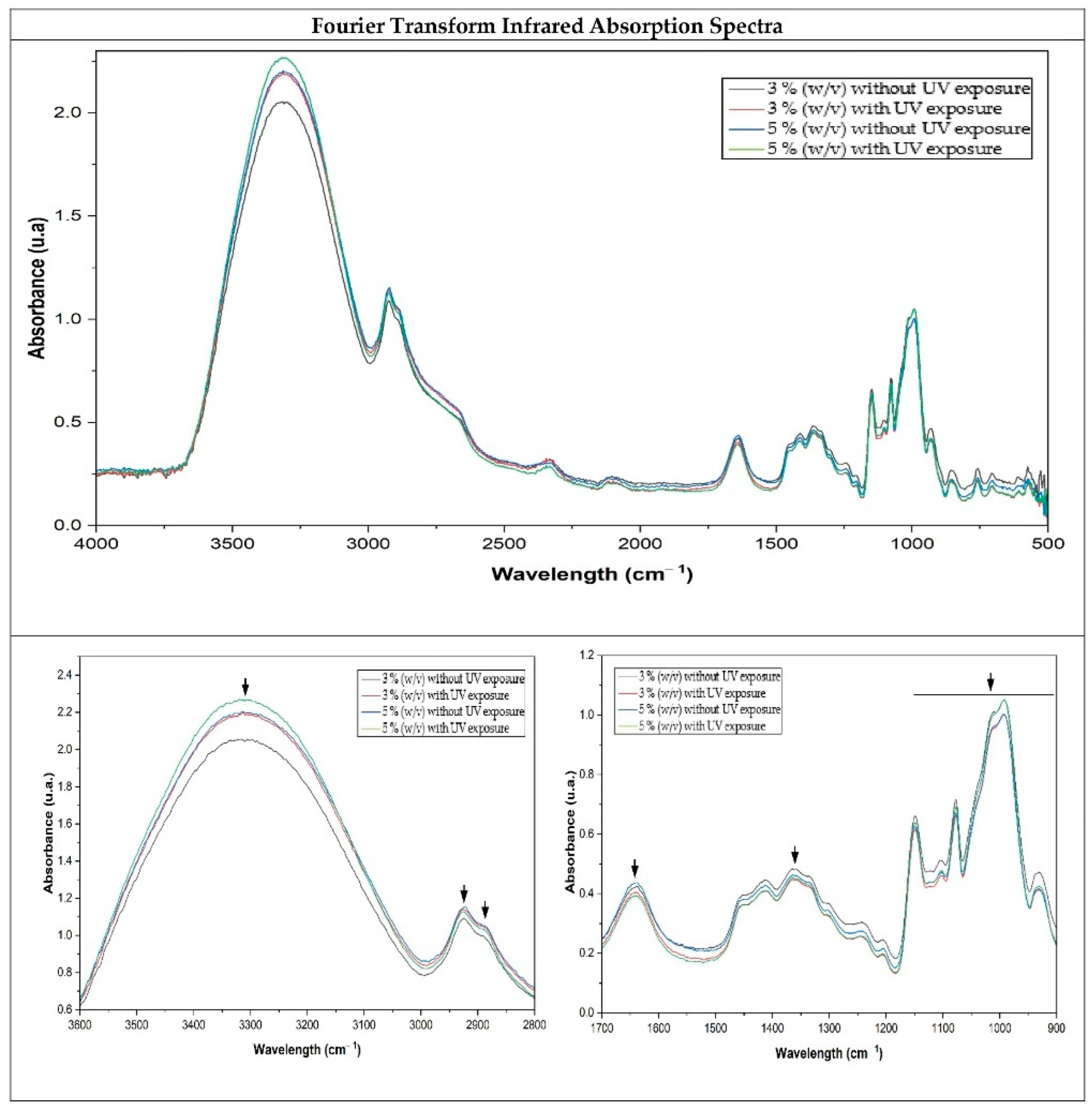
3.3. Fourier Transform Infrared Spectroscopy (FTIR) Absorption
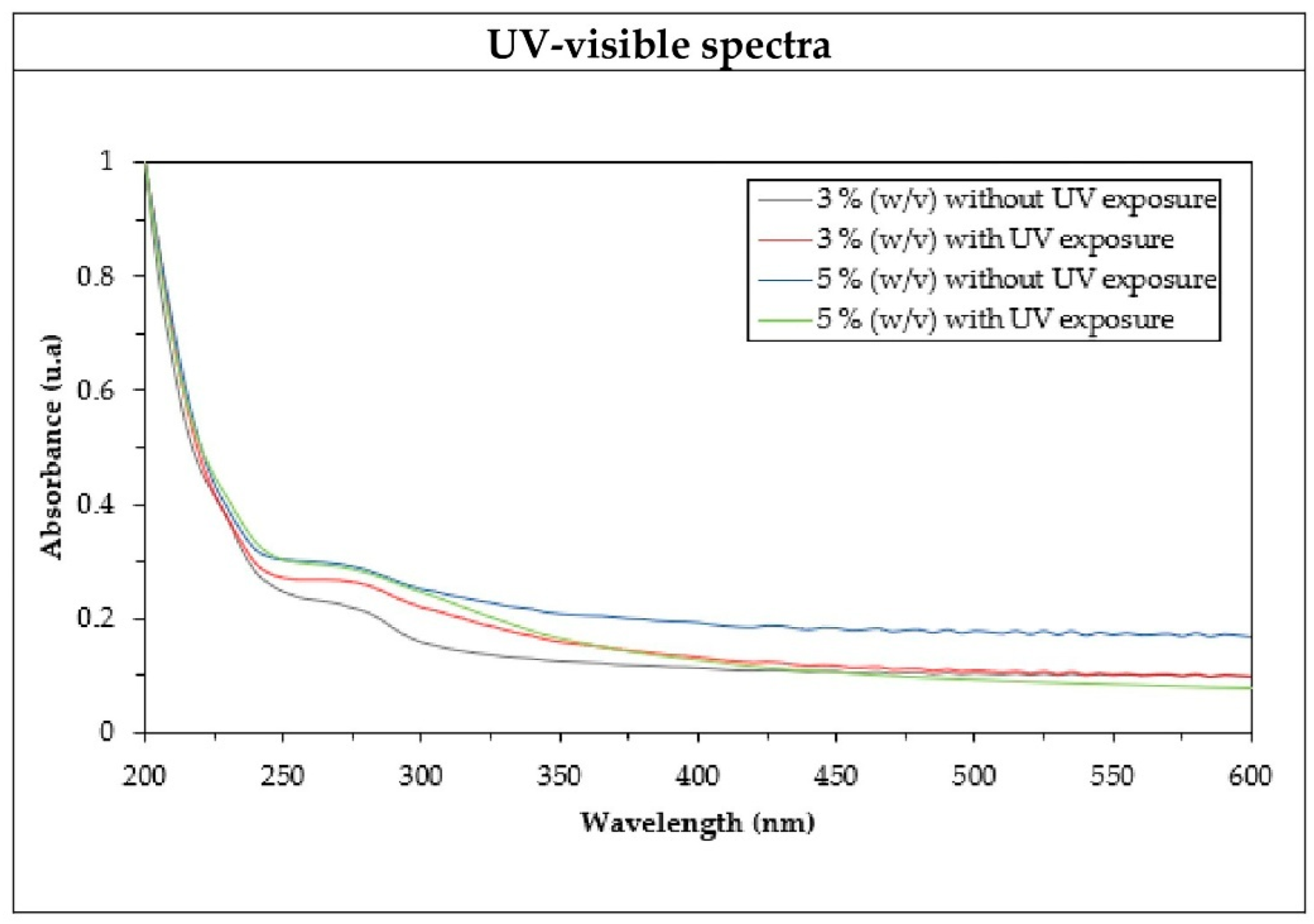
3.4. Optical Absorption
3.5. Moisture Content (MC)
3.6. Water Total Soluble Matter (TSM)
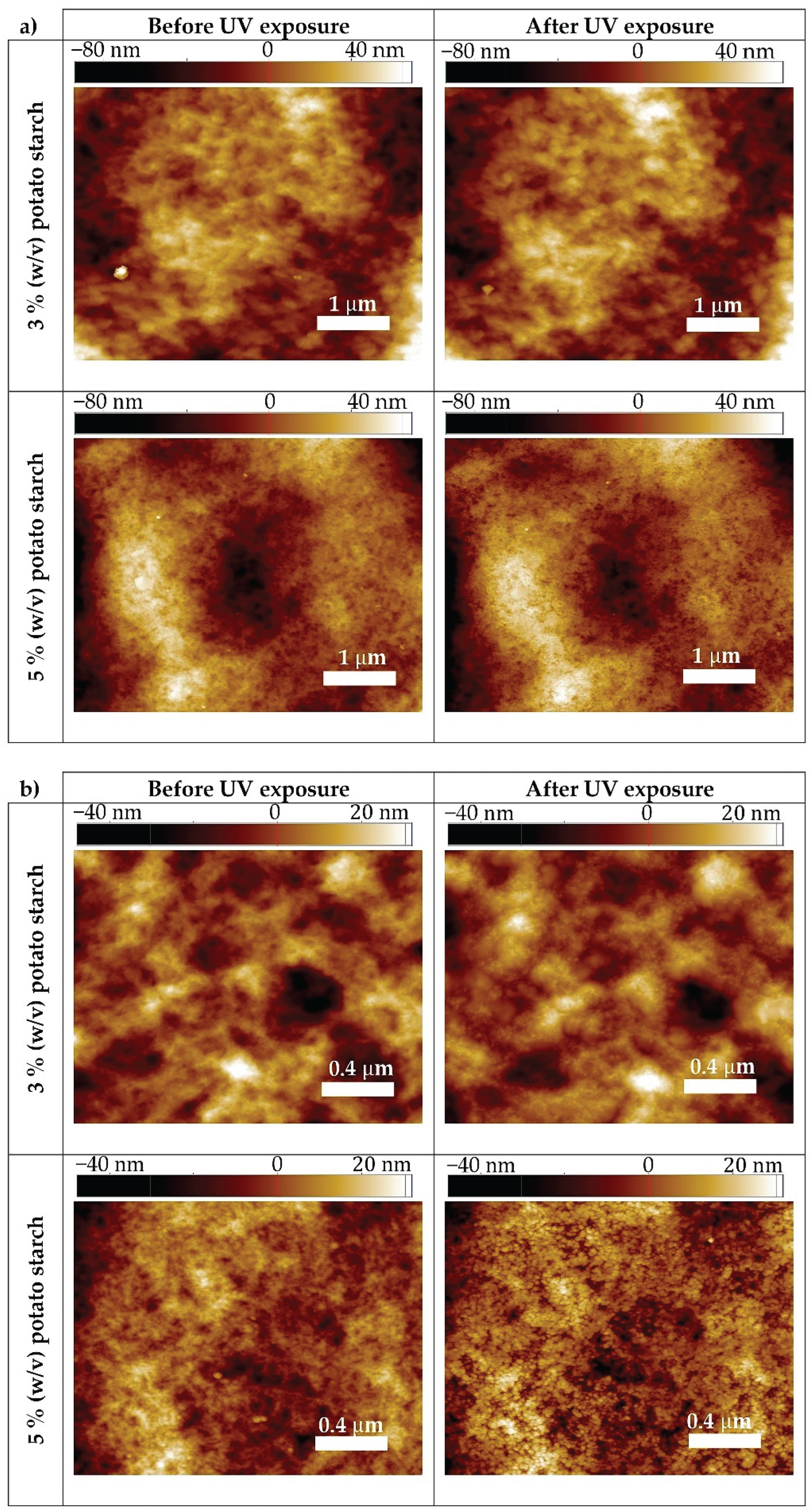
3.8. Nanoscopic Characterization
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Borrelle, S.B.; Ringma, J.; Law, K.L.; Monnahan, C.C.; Lebreton, L.; McGivern, A.; Murphy, E.; Jambeck, J.; Leonard, G.H.; Hilleary, M.A.; et al. Predicted Growth in Plastic Waste Exceeds Efforts to Mitigate Plastic Pollution. Science 2020, 369, 1515–1518. [CrossRef]
- Fayshal, M.A. Current Practices of Plastic Waste Management, Environmental Impacts, and Potential Alternatives for Reducing Pollution and Improving Management. Heliyon 2024, 10, e40838. [CrossRef]
- Rana, R.; Mishra, A.; Goswami, R.; Ahmad, A.; Ahmad, W. Plastics and the Environment: Challenges, Impacts, and Pathways to Sustainability. Integr. Environ. Assess. Manage. 2025, vjaf193. [CrossRef]
- Kibria, Md.G.; Masuk, N.I.; Safayet, R.; Nguyen, H.Q.; Mourshed, M. Plastic Waste: Challenges and Opportunities to Mitigate Pollution and Effective Management. Int. J. Environ. Res. Public Health 2023, 17, 20. [CrossRef]
- Shashoua, Y.; Peydaei, A.; Mortensen, M.N.; Kanstrup, A.B.; Gregory, D.J. Physio-Chemical Degradation of Single-Use Plastics in Natural Weather and Marine Environments. Environ. Pollut. 2024, 357, 124414. [CrossRef]
- Yousafzai, S.; Farid, M.; Zubair, M.; Naeem, N.; Zafar, W.; Zaman Asam, Z.U.; Farid, S.; Ali, S. Detection and Degradation of Microplastics in the Environment: A Review. Environ. Sci.: Adv. 2025, 4, 1142–1165. [CrossRef]
- Dziobak, M.K.; Fahlman, A.; Wells, R.S.; Takeshita, R.; Smith, C.; Gray, A.; Weinstein, J.; Leslie B.H. First evidence of microplastic inhalation among freeranging small cetaceans. PLoS ONE 2024, 19, 10, e0309377. [CrossRef]
- Pradel, A.; Hufenus, R.; Schneebeli, M.; Mitrano, D.M. Impact of contaminant size and density on their incorporation into sea ice. Nat. Commun. 2025, 16, 4375. [CrossRef]
- Isobe, N.; Ishii, S.; Nomaki, H. Progress and prospects in polymer science addressing plastic pollution in marine environments, including the deep-sea floor. Curr. Opin. Chem. Eng. 2025, 47, 101089. [CrossRef]
- Yin Feng, T.; Nur Hidayah, M.I.; Han Lyn, F.; Nur Hanani, Z.A. Physical and Functional Properties of Pulsed-Ultraviolet Treated Starch Based Films with Papaya Leaf Extract. Sustainable Food Technol. 2025, 3, 1986–1995. [CrossRef]
- Oliveira, M.; Almeida, M. The why and how of micro(nano)plastic research. TrAC Trends Anal. Chem. 2019, 114, 196–201. [CrossRef]
- Yousefi, M.; Mazaheri, Y.; Soltani, M.; Rezagholizade-shirvan, A.; Sadighara, P.; Askari, E.; Salehi, A.; Alikord, M.; Shokri, S. Microplastics as emerging contaminants in the food chain: Assessing exposure and threats to consumers, Food Biosci. 2025, 74, 108035. [CrossRef]
- Hu, C.J.; Garcia, M.A.; Nihart, A.; Liu, R.; Yin, L.; Adolphi, N.; Gallego, D.F.; Kang, H.; Campen, M.J.; Yu, X. Microplastic presence in dog and human testis and its potential association with sperm count and weights of testis and epididymis, Toxicol. Sci. 2024, 200, 2, 235–240. [CrossRef]
- Tavakolpournegari, A.; Villacorta, A.; Morataya-Reyes, M.; Arribas Arranz, J.; Banaei, G.; Pastor, S.; Velázquez, A.; Marcos, R.; Hernández, A.; Annangi, B. Harmful effects of true-to-life nanoplastics derived from PET water bottles in human alveolar macrophages. Environ. Pollut. 2024. 348, 123823. [CrossRef]
- Kossalbayev, B.D.; Belkozhayev, A.M.; Abaildayev, A.; Kadirshe, D.K.; Tastambek, K.T.; Kurmanbek, A.; Toleutay, G. Biodegradable Packaging from Agricultural Wastes: A Comprehensive Review of Processing Techniques, Material Properties, and Future Prospects. Polymers 2025, 17, 2224. [CrossRef]
- Balart, R.; Garcia-Garcia, D.; Fombuena, V.; Quiles-Carrillo, L.; Arrieta, M.P. Biopolymers from Natural Resources. Polymers 2021, 13, 2532. [CrossRef]
- Arruda, T.R.; Machado, G.d.O.; Marques, C.S.; Souza, A.L.d.; Pelissari, F.M.; Oliveira, T.V.d.; Silva, R.R.A. An Overview of Starch-Based Materials for Sustainable Food Packaging: Recent Advances, Limitations, and Perspectives. Macromol 2025, 5, 19. [CrossRef]
- Lescano, M.; Vásquez, N.; Tarrillo, S.; Yoplac, I.; Velásquez-Barreto, F.F. Development and Optimization of Biofilms Made from Potato or Arracacha. Starch-Stärke 2021, 73, 2100075. [CrossRef]
- Magnaghi, L.S.; Trigueros, E.; Biesuz, R. A Statistical Approach toward Biofilms Hydrophilicity Characterization: Integrating ANOVA, DOE, and PCA for Starch/Glycerol/CMC Films Case Study. ACS Appl. Bio Mater. 2025, 8, 5, 3691–3696. [CrossRef]
- Ilvis, P.; Acosta, J.; Arancibia, M.; Casado, S. Nanoscopic Characterization of Starch-Based Biofilms Extracted from Ecuadorian Potato (Solanum tuberosum) Varieties. Polymers 2024, 16, 1873. [CrossRef]
- Aurora-Vigo, E.F.; Paucar-Menacho, L.M.; Anaya-Esparza, L.M.; Schmiele, M. Oca (Oxalis tuberosa Mol.): An Andean Tuber With Promising Physicochemical, Technological and Nutritional Properties for Potential Industrial Applications. Plant Foods for Hum. Nutr. 2025, 80, 179. [CrossRef]
- Vera, W.; Quevedo-Olaya, J.L.; Minchán-Velayarce, H.; Samaniego-Rafaele, C.; Rodríguez-León, A.; Salvador-Reyes, R.; Quispe-Santivañez, G.W. From Ethnobotany to Food Innovation: Applications and Functional Potential of Mashua (Tropaeolum tuberosum). Foods 2025, 14, 4091. [CrossRef]
- Luera-Quiñones, S.; Correa, M.J.; Moreno-Rojo, C.; Salvador-Reyes, R.; Paucar-Menacho, L.M. Mashua (Tropaeolum tuberosum Ruiz & Pavón): Nutritional Composition, Bioactive Compounds, and Functional Potential as an Andean Natural Ingredient. Foods 2025, 14, 4198. [CrossRef]
- Pico, C.; De La Vega, J.; Tubón, I.; Arancibia, M.; Casado, S. Nanoscopic Characterization of Starch Biofilms Extracted from the Andean Tubers Ullucus Tuberosus, Tropaeolum Tuberosum, Oxalis Tuberosa, and Solanum Tuberosum. Polymers 2022, 14, 4116. [CrossRef]
- Karnwal, A.; Rauf, A.; Jassim, A.Y.; Selvaraj, M.; Al-Tawaha, A.R.M.S.; Kashyap, P.; Kumar, D.; Malik, T. Advanced Starch-Based Films for Food Packaging: Innovations in Sustainability and Functional Properties. Food Chem.: X 2025, 29, 102662. [CrossRef]
- Zhou, J.; Zhang, J.; Ma, Y.; Tong, J. Surface Photo-Crosslinking of Corn Starch Sheets. Carbohydr. Polym. 2008, 74, 405–410. [CrossRef]
- Fathi, N.; Almasi, H.; Pirouzifard, M.K. Effect of Ultraviolet Radiation on Morphological and Physicochemical Properties of Sesame Protein Isolate Based Edible Films. Food Hydrocoll. 2018, 85, 136–143. [CrossRef]
- Farhan, A.; Hani, N.M. Characterization of Edible Packaging Films Based on Semi-Refined Kappa-Carrageenan Plasticized with Glycerol and Sorbitol. Food Hydrocoll. 2017, 64, 48–58. [CrossRef]
- Orsuwan, A.; Sothornvit, R. Development and Characterization of Banana Flour Film Incorporated with Montmorillonite and Banana Starch Nanoparticles. Carbohydr. Polym. 2017, 174, 235–242. [CrossRef]
- Arancibia, MY; Alemán, A.; López-Caballero, ME; Gómez-Guillén, MC; Montero, P. Development of active films of chitosan isolated by mild extraction with added protein concentrate from shrimp waste. Food Hydrocoll. 2015, 43, 91–99. [CrossRef]
- Salazar, D.; Arancibia, M.; Casado, S.; Viteri, A.; López-Caballero, M.E.; Montero, M.P. Green Banana (Musa Acuminata AAA) Wastes to Develop an Edible Film for Food Applications. Polymers 2021, 13, 3183. [CrossRef]
- Casado, S. Studying friction while playing the violin: Exploring the stick–slip phenomenon. Beilstein J. Nanotechnol. 2017, 8, 159–166. [CrossRef]
- Casado, S.; Lobo, M.D.V.T.; Paino, C.L. Dynamics of plasma membrane surface related to the release of extracellular vesicles by mesenchymal stem cells in culture. Sci. Rep. 2017, 7, 6767. [CrossRef]
- Singh, N.; Singh, J.; Kaur, L.; Sodhi, N.S.; Gill, B.S. Morphological, thermal and rheological properties of starches from different botanical sources. Food Chem. 2003, 81(2), 219-231. [CrossRef]
- Quispe, M.; V. Lopez, O.; A. Villar, M. Oxidative Degradation of Thermoplastic Starch Induced by UV Radiation. J. Renewable Mater. 2019, 7, 383–391. [CrossRef]
- El-Hiti, G.A.; Ahmed, D.S.; Yousif, E.; Al-Khazrajy, O.S.A.; Abdallh, M.; Alanazi, S.A. Modifications of Polymers through the Addition of Ultraviolet Absorbers to Reduce the Aging Effect of Accelerated and Natural Irradiation. Polymers 2021, 14, 20. [CrossRef]
- Tarique, J.; Sapuan, S.M.; Khalina, A. Effect of Glycerol Plasticizer Loading on the Physical, Mechanical, Thermal, and Barrier Properties of Arrowroot (Maranta Arundinacea) Starch Biopolymers. Sci. Rep. 2021, 11, 13900. [CrossRef]
- Taweechat, C.; Wongsooka, T.; Rawdkuen, S. Properties of Banana (Cavendish Spp.) Starch Film Incorporated with Banana Peel Extract and Its Application. Molecules 2021, 26, 1406. [CrossRef]
- Dutta, D.; Sit, N. Comparison of Properties of Films Prepared from Potato Starch Modified by Annealing and Heat–Moisture Treatment. Starch-Stärke 2022, 74, 2200110. [CrossRef]
- Vamadevan, V.; Bertoft, E. Structure-function relationships of starch components. Starch – Stärke 2014, 67, 1–2, 55–68. [CrossRef]
- Domene-López, D.; García-Quesada, J.C.; Martin-Gullon, I.; Montalbán, M.G. Influence of Starch Composition and Molecular Weight on Physicochemical Properties of Biodegradable Films. Polymers 2019, 11, 1084. [CrossRef]
- Dai, L.; Zhang, J.; Cheng, F. Effects of Starches from Different Botanical Sources and Modification Methods on Physicochemical Properties of Starch-Based Edible Films. Int. J. Biol. Macromol. 2019, 132, 897–905. [CrossRef]
- Bergo, P.; Sobral, P.J.A.; Prison, J.M. Effect of glycerol on physical properties of cassava starch films. J. Food Processing Preserv. 2010, 34, 401–410. [CrossRef]
- Pereira, J.F.; Garcia Lonni, A.A.S.; Mali, S. Development of biopolymeric films with addition of vitamin C and catuaba extract as natural antioxidants. Prep. Biochem. Biotechnol. 2022, 52, 1–10. [CrossRef]
- Wu, D.; Samanta, A.; Srivastava, R.K.; Hakkarainen, M. Starch-Derived Nanographene Oxide Paves the Way for Electrospinnable and Bioactive Starch Scaffolds for Bone Tissue Engineering. Biomacromolecules 2017, 18, 1582–1591. [CrossRef]
- Pinto, J.; Dias, M.; Amaral, J.; Ivanov, M.; Paixão, J.A.; Coimbra, M.A.; Ferreira, P.; Pereira, E.; Gonçalves, I. Influence of UV degradation of bioplastics on the amplification of mercury bioavailability in aquatic environments. Marine Pollution Bulletin 2022, 180, 113806. [CrossRef]
- Yin, P.; Zhang, Y.; Liu, X.; Wang, J.; Chen, L. Surface cross-linked thermoplastic starch with different UV treatment effects studied by FTIR, mechanical and thermal analysis. RSC Advances 2020, 10, 35677–35686. [CrossRef]
- Bajer, D.; Kaczmarek, H.; Bajer, K. The Structure and Properties of Different Types of Starch Exposed to UV Radiation: A Comparative Study. Carbohydr. Polym. 2013, 98, 477–482. [CrossRef]
- Gutiérrez-Silva, K.; Capezza, A.J.; Gil-Castell, O.; Badia-Valiente, J.D. UV-C and UV-C/H₂O-Induced Abiotic Degradation of Films of Commercial PBAT/TPS Blends. Polymers 2025, 17, 1173. [CrossRef]
- Shahabi-Ghahfarrokhi, I.; Goudarzi, V.; Babaei-Ghazvini, A. Production of Starch Based Biopolymer by Green Photochemical Reaction at Different UV Region as a Food Packaging Material: Physicochemical Characterization. Int. J. Biol. Macromol. 2019, 122, 201–209. [CrossRef]
- Santacruz, S.; Rivadeneira, C.; Castro, M. Edible Films Based on Starch and Chitosan. Effect of Starch Source and Concentration, Plasticizer, Surfactant’s Hydrophobic Tail and Mechanical Treatment. Food Hydrocoll. 2015, 49, 89–94. [CrossRef]
- Pérez-Córdoba, L.J.; Galecio-Rojas, M.; Peña-Carrasco, F.; Ibarz, A.; Velezmoro-Sánchez, C.; Martínez-Tapia, P. Effect of ultraviolet irradiation on physicochemical and disintegrability properties of tunta starch–tara gum nanocomposite films reinforced with starch nanocrystals. Journal of Polymers and the Environment 2023, 31, 299–311. [CrossRef]
- Mahzan, S.; Fitri, M.; Zaleha, M. UV Radiation Effect towards Mechanical Properties of Natural Fibre Reinforced Composite Material: A Review. IOP Conf. Ser.: Mater. Sci. Eng. 2017, 165, 012021. [CrossRef]
- Uyarcan, M.; Güngör, S.C. Improving Functional Properties of Starch-Based Films by Ultraviolet (UV-C) Technology: Characterization and Application on Minced Meat Packaging. Int. J. Biol. Macromol. 2024, 282, 137085. [CrossRef]
- Velásquez-Castillo, L.E.; Leite, M.A.; Tisnado, V.J.A.; Ditchfield, C.; Sobral, P.J.D.A.; Moraes, I.C.F. Cassava Starch Films Containing Quinoa Starch Nanocrystals: Physical and Surface Properties. Foods 2023, 12, 576. [CrossRef]

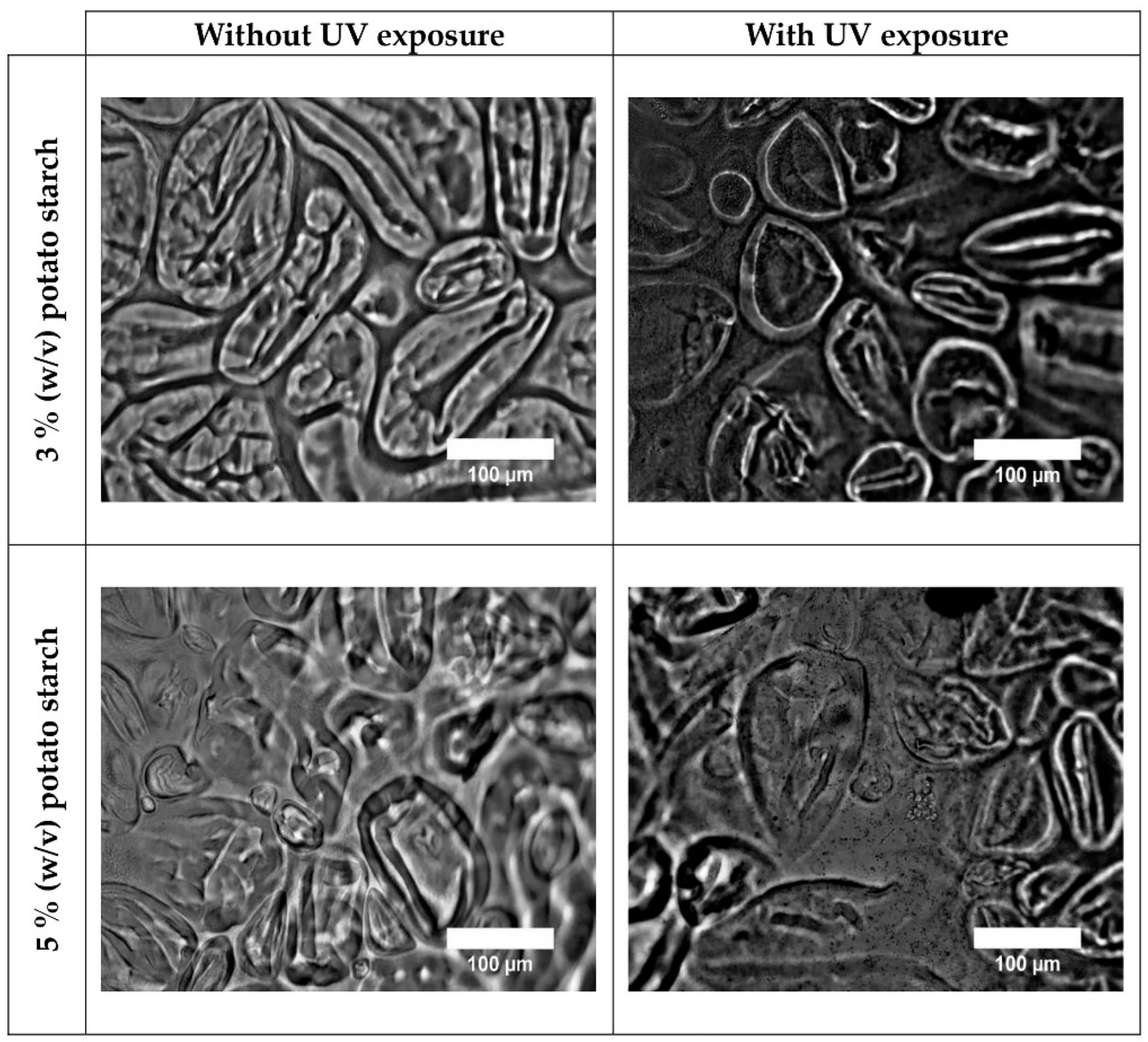
| % (w/v) potato starch | Treatment | Thickness (µm) |
|---|---|---|
| 3 | Without UV exposure | 82 ± 23ab |
| 3 | With UV exposure | 71 ± 15b |
| 5 | Without UV exposure | 114 ± 14a |
| 5 | With UV exposure | 113 ± 12a |
| % (w/v) potato starch | Treatment | Opacity (AU/mm) |
|---|---|---|
| 3 | Without UV exposure | 1.00 ± 0.02b |
| 3 | With UV exposure | 2.12 ± 0.02a |
| 5 | Without UV exposure | 0.83 ± 0.02b |
| 5 | With UV exposure | 2.41 ± 0.02a |
| % (w/v) potato starch | Treatment | Moisture content (%) |
|---|---|---|
| 3 | Without UV exposure | 11.5 ± 0.5a |
| 3 | With UV exposure | 7.1 ± 0.8b |
| 5 | Without UV exposure | 11.6 ± 0.6a |
| 5 | With UV exposure | 6.8 ± 0.6b |
| % (w/v) potato starch | Treatment | TSM (%) |
|---|---|---|
| 3 | Without UV exposure | 28.1 ± 0. 6a |
| 3 | With UV exposure | 17.7 ± 0.8b |
| 5 | Without UV exposure | 29.2 ± 0.6a |
| 5 | With UV exposure | 18.6 ± 1.0b |
| % (w/v) potato starch | 2 x 2 µm² | 5 x 5 µm² | ||
| Before UV exposure |
After UV exposure |
Before UV exposure |
After UV exposure |
|
| 3 | 13 ± 6a | 13 ± 7a | 29 ± 9a | 31 ± 11a |
| 5 | 9 ± 5a | 10 ± 6a | 23 ± 8a | 26 ± 10a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




