Submitted:
05 February 2026
Posted:
06 February 2026
You are already at the latest version
Abstract
Keywords:
Aims and Premise
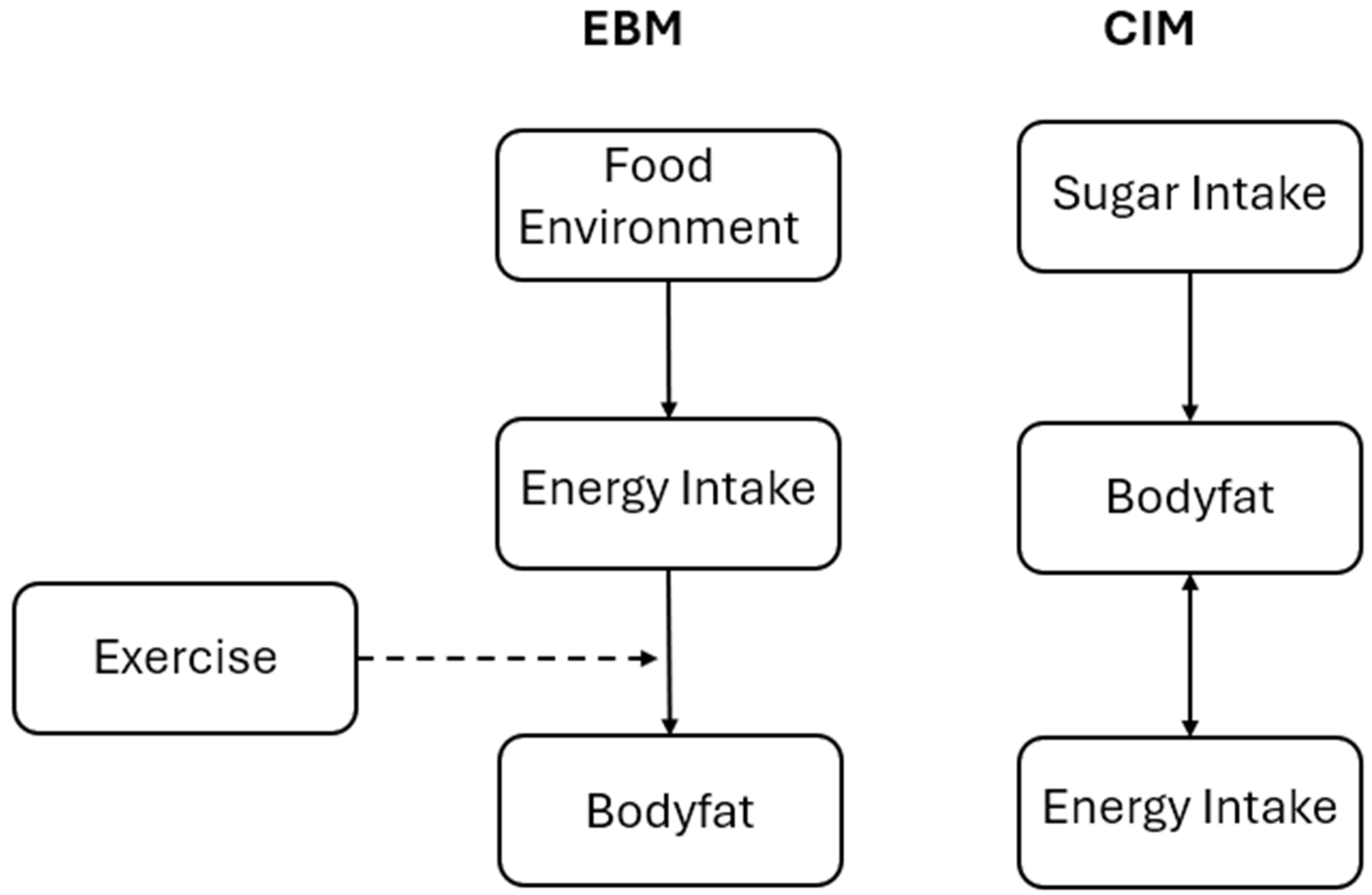
Background
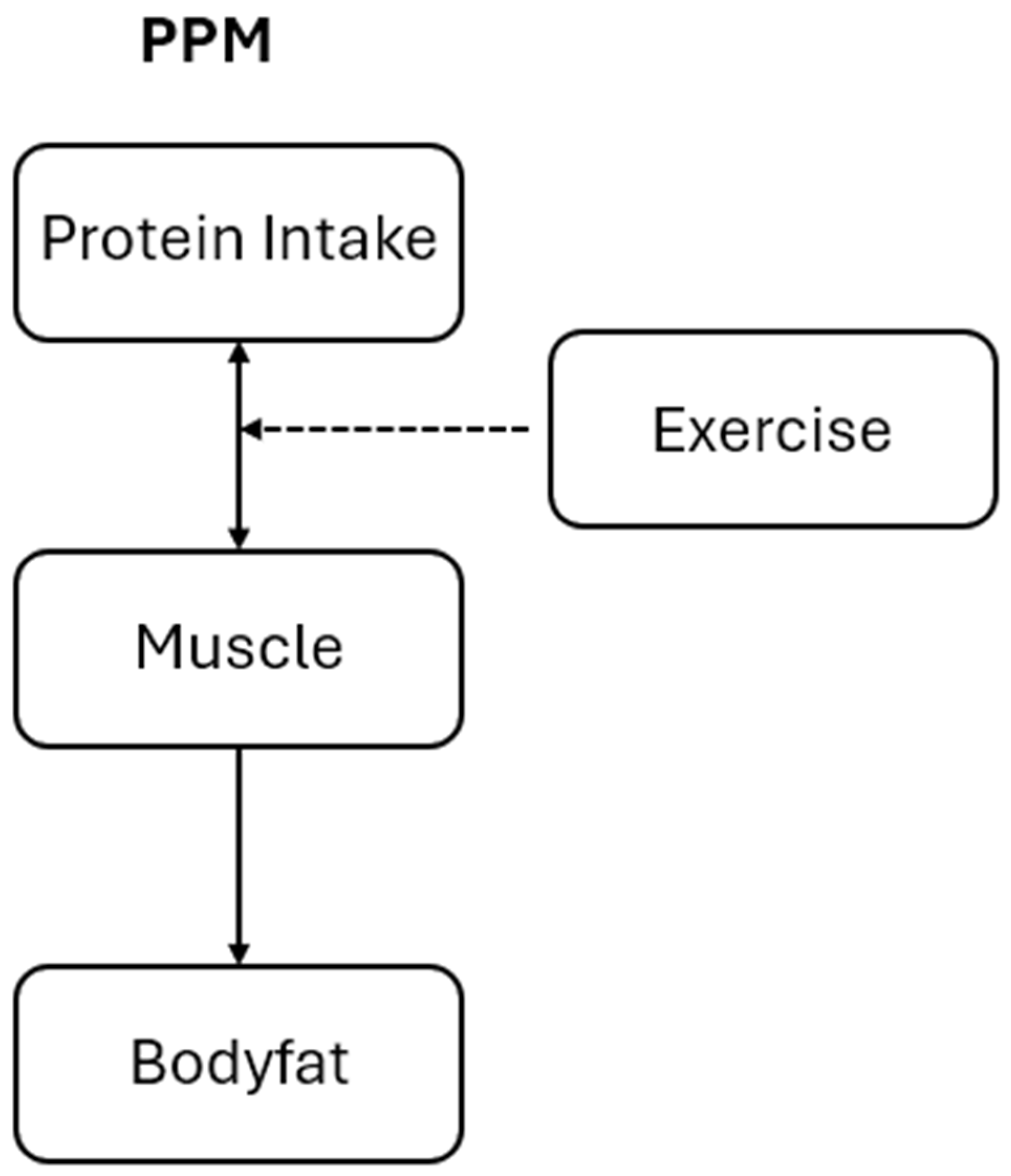
The Protein Partitioning Model
- lean mass increases with protein intake and energy availability, (recall that simply represents the fraction of calories not stored as fat and so remaining for maintaining lean body mass and energy needs), such that , assuming
- insulin sensitivity () improves with lean mass such as due to muscle such that (thus, protein intake affects fat accumulation indirectly by increasing lean mass, which improves insulin sensitivity and reduces the fraction of energy partitioned into fat), and
- fat storage fraction decreases as insulin sensitivity improves and so is inversely related to lean mass,
Discussion
Protein’s ROLE in lean Mass and Energy Partitioning
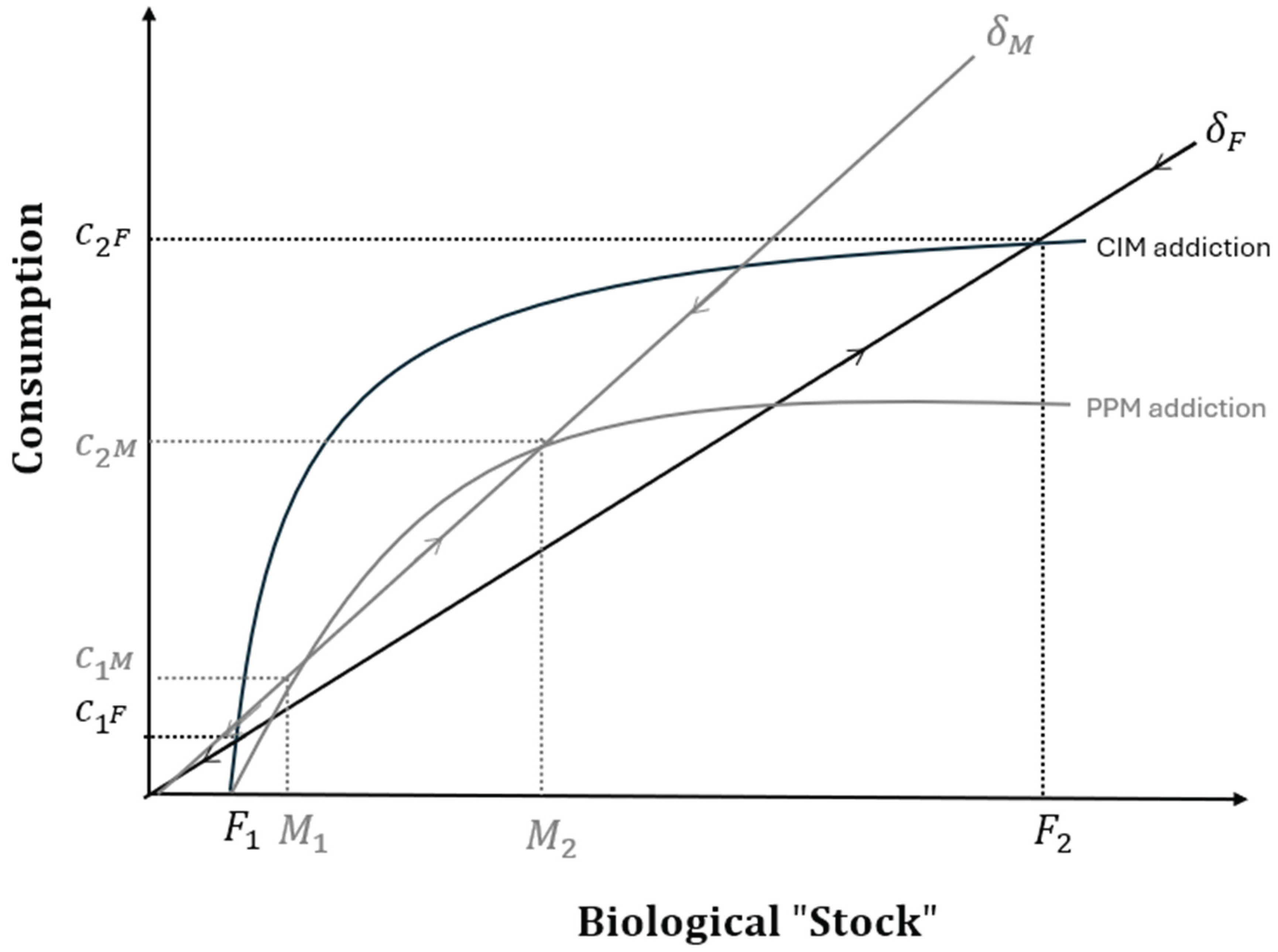
Food Addiction and Biological Stocks
Practical Implications
Summary and Conclusion
Statements and Declarations
Data Availability Statement
References
- Antonio, J.; Candow, D. G.; Forbes, S. C.; Ormsbee, M. J.; Saracino, P. G.; Roberts, J. Effects of dietary protein on body composition in exercising individuals. Nutrients 2020, 12(6), 1890. [Google Scholar] [CrossRef]
- Aragon, A. A.; Schoenfeld, B. J.; Wildman, R.; Kleiner, S.; VanDusseldorp, T.; Taylor, L.; Antonio, J. International society of sports nutrition position stand: diets and body composition. Journal of the International Society of Sports Nutrition 2017, 14(1), 16. [Google Scholar] [CrossRef]
- Balleine, B. W.; Dickinson, A. Goal-directed instrumental action: Contingency and incentive learning and their cortical substrates. Neuropharmacology 1998, 37(4-5), 407–419. [Google Scholar] [CrossRef]
- Beals, J. W.; Burd, N. A.; Moore, D. R.; Van Vliet, S. Obesity alters the muscle protein synthetic response to nutrition and exercise. Frontiers in nutrition 2019, 6, 87. [Google Scholar] [CrossRef]
- Becker, G. S.; Murphy, K. M. A Theory of Rational Addiction. Journal of Political Economy 1988, 96(4), 675–700. [Google Scholar] [CrossRef]
- Bosy-Westphal, A.; Kahlhöfer, J.; Lagerpusch, M.; Skurk, T.; Müller, M. J. Deep body composition phenotyping during weight cycling: relevance to metabolic efficiency and metabolic risk. Obesity Reviews 2015, 16, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Chiacchierini, G.; Naneix, F.; Peters, K. Z.; Apergis-Schoute, J.; Snoeren, E. M. S.; McCutcheon, J. E. Protein appetite drives macronutrient-related differences in ventral tegmental area neural activity. Journal of Neuroscience 2021, 41(23), 5080–5092. [Google Scholar] [CrossRef] [PubMed]
- Collins, K. A.; Ross, L. M.; Slentz, C. A.; Huffman, K. M.; Kraus, W. E. Differential effects of amount, intensity, and mode of exercise training on insulin sensitivity and glucose homeostasis: a narrative review. Sports Medicine-Open 2022, 8(1), 90. [Google Scholar] [CrossRef]
- Di Meo, S.; Iossa, S.; Venditti, P. Improvement of obesity-linked skeletal muscle insulin resistance by strength and endurance training. Journal of Endocrinology 2017, 234(3), R159–R181. [Google Scholar] [CrossRef]
- Dulloo, A. G.; Jacquet, J. The control of partitioning between protein and fat during human starvation: its internal determinants and biological significance. British Journal of Nutrition 1999, 82(5), 339–356. [Google Scholar] [CrossRef]
- English, K. L.; Paddon-Jones, D. Protecting muscle mass and function in older adults during bed rest. Current Opinion in Clinical Nutrition & Metabolic Care 2010, 13(1), 34–39. [Google Scholar]
- Griffioen-Roose, S.; Smeets, P. A.; van den Heuvel, E.; Boesveldt, S.; Finlayson, G.; de Graaf, C. Human protein status modulates brain reward responses to food cues. The American Journal of Clinical Nutrition 2014, 100(1), 113–122. [Google Scholar] [CrossRef] [PubMed]
- Gwin, J. A. The Effects of Dietary Protein at Breakfast and Across the Day on Appetite Control & Satiety, Food Intake, and Sleep Quality . Doctoral dissertation, Purdue University, 2018. [Google Scholar]
- Hall, K. D. A review of the carbohydrate–insulin model of obesity. European Journal of Clinical Nutrition 2017, 71(3), 323–326. [Google Scholar] [CrossRef]
- Hall, K. D.; Guo, J. Obesity energetics: Body weight regulation and the effects of diet composition. Gastroenterology 2017, 152(7), 1718–1727.e3. [Google Scholar] [CrossRef] [PubMed]
- Hall, K. D.; Chen, K. Y.; Guo, J.; Lam, Y. Y.; Leibel, R. L.; Mayer, L. E.; Reitman, M. L.; Rosenbaum, M.; Smith, S. R.; Waslh, B. T.; Ravussin, E. Energy expenditure and body composition changes after an isocaloric ketogenic diet in overweight and obese men. The American Journal of Clinical Nutrition 2016, 104(2), 324–333. [Google Scholar] [CrossRef]
- Hoertel, H. A.; Will, M. J.; Leidy, H. J. A randomized crossover, pilot study examining the effects of a normal protein vs. high protein breakfast on food cravings and reward signals in overweight/obese “breakfast skipping”, late-adolescent girls. Nutrition Journal 2014, 13, 1–8. [Google Scholar] [CrossRef]
- Horton, T. J.; Drougas, H.; Brachey, A.; Reed, G. W.; Peters, J. C.; Hill, J. O. Fat and carbohydrate overfeeding in humans: different effects on energy storage. The American Journal of Clinical Nutrition 1995, 62(1), 19–29. [Google Scholar] [CrossRef]
- Isacco, L.; Lazzer, S.; Pereira, B.; Fearnbach, N.; Montaurier, C.; Vermorel, M.; Rannou, F.; Boirie, Y.; Thivel, D. Association of protein-energy partitioning with body weight and body composition changes in adolescents with severe obesity. International Journal of Obesity 2022, 46(11), 2021–2028. [Google Scholar] [CrossRef] [PubMed]
- Kanaan, M. F.; Nait-Yahia, S.; Doucet, É. The effects of high protein intakes during energy restriction on body composition, energy metabolism and physical performance in recreational athletes. European Journal of Clinical Nutrition 2025, 1–9. [Google Scholar] [CrossRef]
- Khan, N.; Li, Z.; Ali, A.; Quan, B.; Kang, J.; Ullah, M.; Shafiq, M. Comprehensive transcriptomic analysis of myostatin-knockout pigs: insights into muscle growth and lipid metabolism. Transgenic Research 2025, 34(1), 12. [Google Scholar] [CrossRef]
- Layman, D. K. Dietary Guidelines should reflect new understandings about adult protein needs. Nutrition & Metabolism 2009, 6(1), 12. [Google Scholar] [CrossRef]
- Leidy, H. J.; Clifton, P. M.; Astrup, A.; Wycherley, T. P.; Westerterp-Plantenga, M. S.; Luscombe-Marsh, N. D.; Woods, S. C.; Mattes, R. D. The role of protein in weight loss and maintenance. The American Journal of Clinical Nutrition 2015, 101(6), 1320S–1329S. [Google Scholar] [CrossRef]
- Leidy, H. J.; Tang, M.; Armstrong, C. L. H.; Martin, C. B.; Campbell, W. W. The effects of consuming frequent, higher protein meals on appetite and satiety during weight loss in overweight/obese men. Obesity 2011, 19(4), 818–824. [Google Scholar] [CrossRef]
- Leong, D. P.; Teo, K. K.; Rangarajan, S.; Lopez-Jaramillo, P.; Avezum, A.; Orlandini, A.; Yusuf, S. Prognostic value of grip strength: findings from the Prospective Urban Rural Epidemiology (PURE) study. The Lancet 2015, 386(9990), 266–273. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, D. S.; Ebbeling, C. B. The carbohydrate-insulin model of obesity: beyond "calories in, calories out. JAMA Internal Medicine 2018, 178(8), 1098–1103. [Google Scholar] [CrossRef] [PubMed]
- Magkos, F.; Sørensen, T. I.; Raubenheimer, D.; Dhurandhar, N. V.; Loos, R. J.; Bosy-Westphal, A.; Clemmensen, C.; Hjorth, M. F.; Allison, D. B.; Taubes, G.; Astrup, A. On the pathogenesis of obesity: causal models and missing pieces of the puzzle. Nature Metabolism 2024, 6(10), 1856–1865. [Google Scholar] [CrossRef]
- McPherron, A. C.; Lee, S. J. Suppression of body fat accumulation in myostatin-deficient mice. The Journal of Clinical Investigation 2002, 109(5), 595–601. [Google Scholar] [CrossRef]
- Mettler, S.; Mitchell, N.; Tipton, K. D. Increased protein intake reduces lean body mass loss during weight loss in athletes. Medicine and Science in Sports and Exercise 2010, 42(2), 326–337. [Google Scholar] [CrossRef]
- Ottenheimer, D.; Richard, J. M.; Janak, P. H. Ventral pallidum encodes relative reward value earlier and more robustly than nucleus accumbens. Nature Communications 2018, 9(1), 4350. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S. Protein consumption and resistance exercise: maximizing anabolic potential. Sports Sci Exch 2013, 26(107), 1–5. [Google Scholar]
- Phillips, S. M. A brief review of critical processes in exercise-induced muscular hypertrophy. Sports Medicine 2014, 44 (Suppl 1), S71–S77. [Google Scholar] [CrossRef]
- Phillips, S. M.; van Loon, L. J. C. The impact of protein quality on the promotion of resistance exercise-induced changes in muscle mass. Nutrition & Metabolism 2016, 13, 64. [Google Scholar] [CrossRef]
- Ren, H.; Xiao, W.; Qin, X.; Cai, G.; Chen, H.; Hua, Z.; Bi, Y. Myostatin regulates fatty acid desaturation and fat deposition through MEF2C/miR222/SCD5 cascade in pigs. Communications Biology 2020, 3(1), 612. [Google Scholar] [CrossRef] [PubMed]
- Richter, E. A.; Sylow, L.; Hargreaves, M. Interactions between insulin and exercise. Biochemical Journal 2021, 478(21), 3827–3846. [Google Scholar] [CrossRef]
- Roh, E.; Choi, K. M. Health consequences of sarcopenic obesity: a narrative review. Frontiers in Endocrinology 2020, 11, 332. [Google Scholar] [CrossRef] [PubMed]
- Sardeli, A. V.; Komatsu, T. R.; Mori, M. A.; Gáspari, A. F.; Chacon-Mikahil, M. P. T. Resistance training prevents muscle loss induced by caloric restriction in obese elderly individuals: a systematic review and meta-analysis. Nutrients 2018, 10(4), 423. [Google Scholar] [CrossRef] [PubMed]
- Sartori, R.; Romanello, V.; Sandri, M. Mechanisms of muscle atrophy and hypertrophy: implications in health and disease. Nature Communications 2021, 12(1), 330. [Google Scholar] [CrossRef]
- Shulman, G. I. Cellular mechanisms of insulin resistance. Journal of Clinical Investigation 2000, 106(2), 171–176. [Google Scholar] [CrossRef] [PubMed]
- Simpson, S. J.; Raubenheimer, D. Obesity: the protein leverage hypothesis. Obesity Reviews 2005, 6(2), 133–142. [Google Scholar] [CrossRef]
- Simpson, S. J.; Raubenheimer, D. The nature of nutrition: a unifying framework from animal adaptation to human obesity; Princeton University Press, 2012. [Google Scholar]
- Srikanthan, P.; Karlamangla, A. S. Relative muscle mass is inversely associated with insulin resistance and prediabetes. The Journal of Clinical Endocrinology & Metabolism 2011, 96(9), 2898–2903. [Google Scholar] [CrossRef]
- Swinburn, B. A.; Sacks, G.; Hall, K. D.; McPherson, K.; Finegood, D. T.; Moodie, M. L.; Gortmaker, S. L. The global obesity pandemic: shaped by global drivers and local environments. The Lancet 2011, 378(9793), 804–814. [Google Scholar] [CrossRef] [PubMed]
- Tomé, D.; Chaumontet, C.; Even, P. C.; Darcel, N.; Azzout-Marniche, D. Protein status modulates the rewarding value of foods and meals to maintain an adequate protein intake. Physiology & Behavior 2019, 206, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.; Cooney, G. J.; Kraegen, E. W.; Bruce, C. R. Fatty acid metabolism, energy expenditure and insulin resistance in muscle. Journal of Endocrinology 2014, 220(2), T61–T79. [Google Scholar] [CrossRef] [PubMed]
- Vincent, H. K.; Raiser, S. N.; Vincent, K. R. The aging musculoskeletal system and obesity-related considerations with exercise. Ageing Research Reviews 2012, 11(3), 361–373. [Google Scholar] [CrossRef]
- Weaver, C. M.; Gordon, C. M.; Janz, K. F.; Kalkwarf, H. J.; Lappe, J. M.; Lewis, R.; Zemel, B. S. The National Osteoporosis Foundation’s position statement on peak bone mass development and lifestyle factors: a systematic review and implementation recommendations. Osteoporosis International 2016, 27(4), 1281–1386. [Google Scholar] [CrossRef]
- Westerterp, K. R. Exercise, energy balance and body composition. European Journal of Clinical Nutrition 2018, 72(9), 1246–1250. [Google Scholar] [CrossRef]
- Wolfe, R. R. The underappreciated role of muscle in health and disease. The American Journal of Clinical Nutrition 2006, 84(3), 475–482. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, W.; Liu, T.; Zhang, D. Association of grip strength with risk of all-cause mortality, cardiovascular diseases, and cancer in community-dwelling populations: a meta-analysis of prospective cohort studies. Journal of the American Medical Directors Association 2017, 18(6), 551–e17. [Google Scholar] [CrossRef]
- Wycherley, T. P.; Noakes, M.; Clifton, P. M.; Cleanthous, X.; Keogh, J. B.; Brinkworth, G. D. A high-protein diet with resistance exercise training improves weight loss and body composition in overweight and obese patients with type 2 diabetes. Diabetes Care 2010, 33(5), 969–976. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



