Submitted:
03 February 2026
Posted:
06 February 2026
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Results
2.1. Extraction of D. viscosa Roots
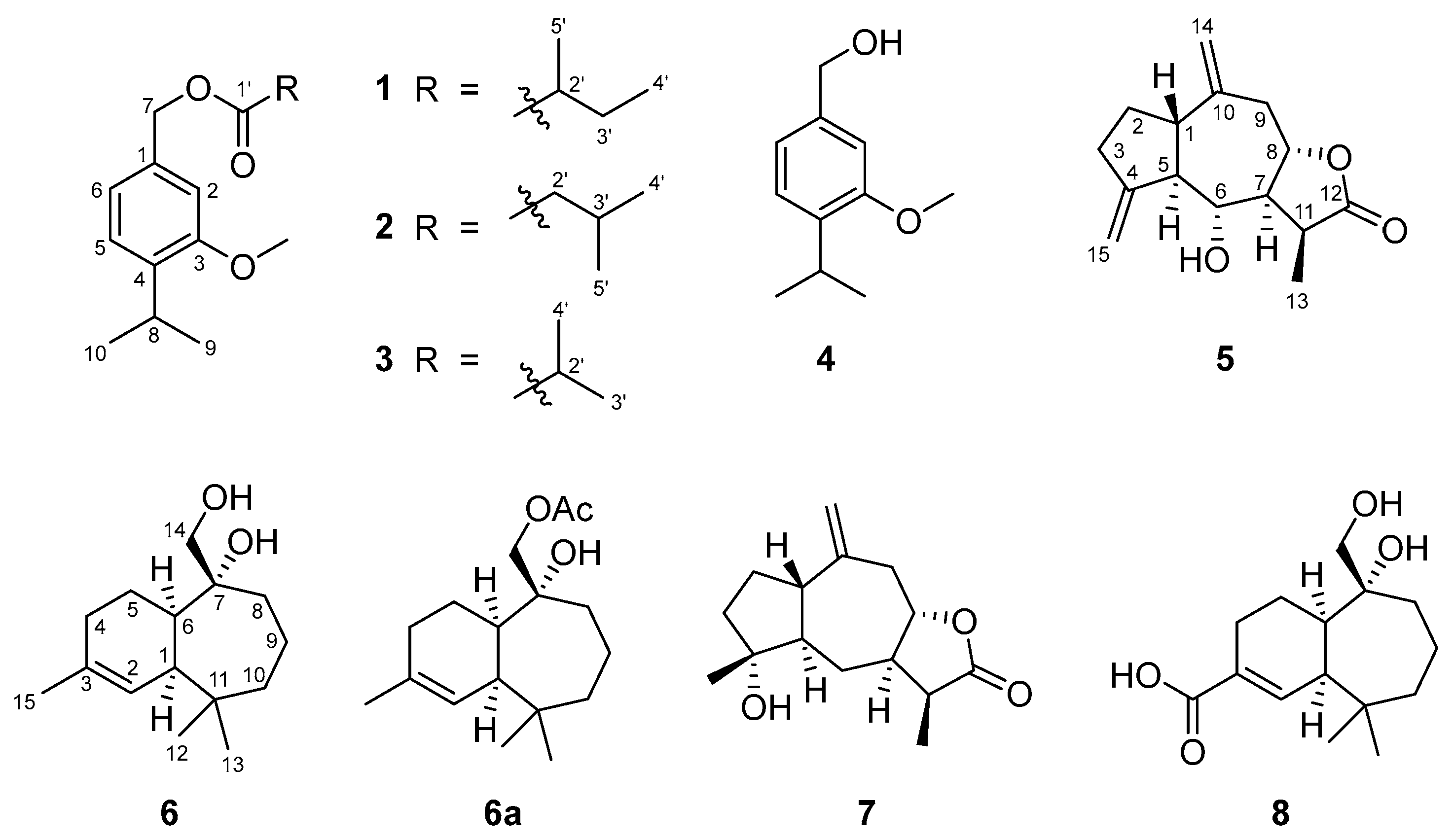
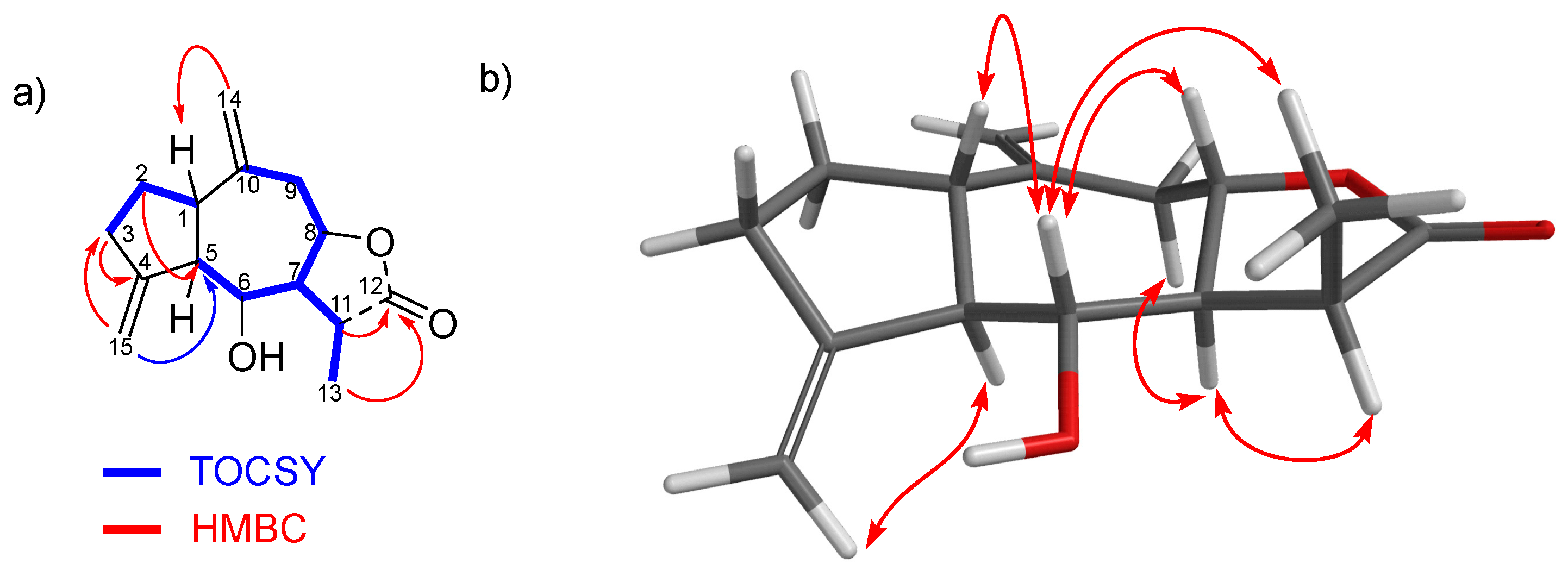
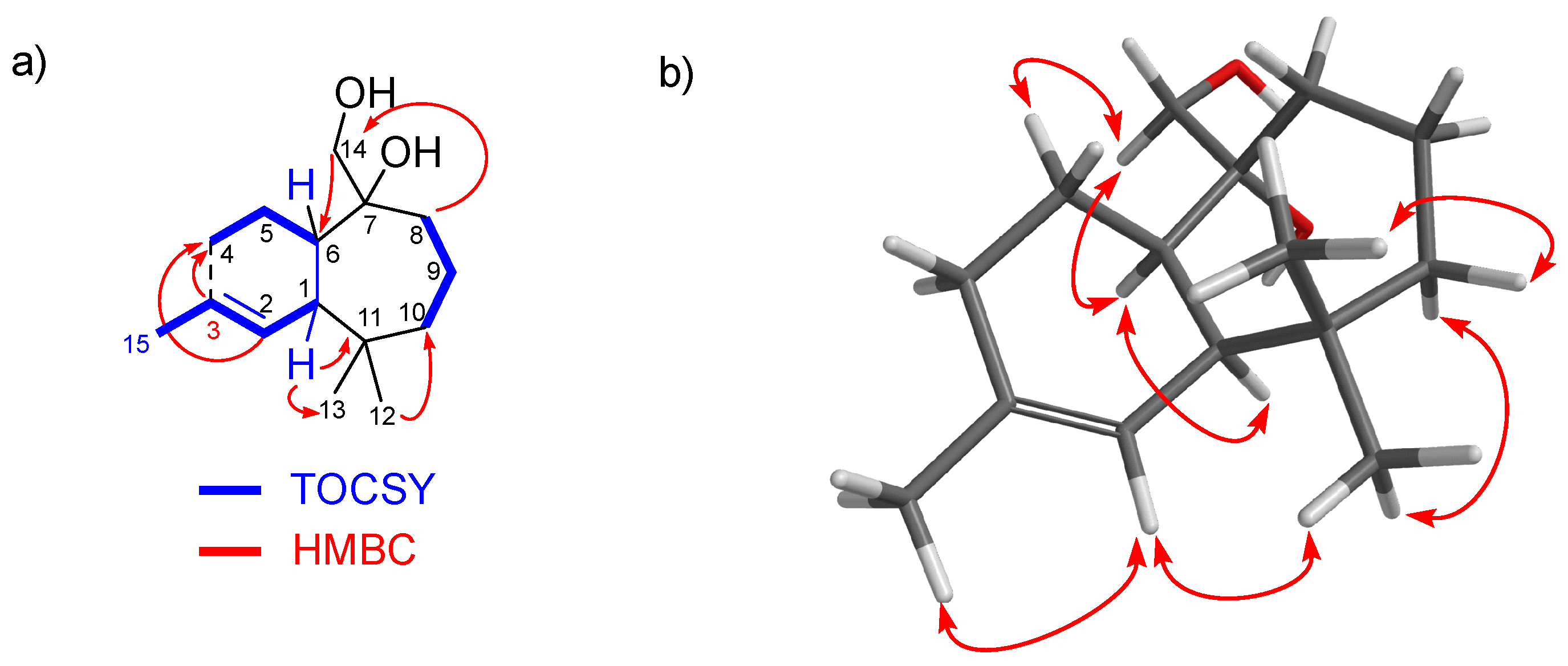
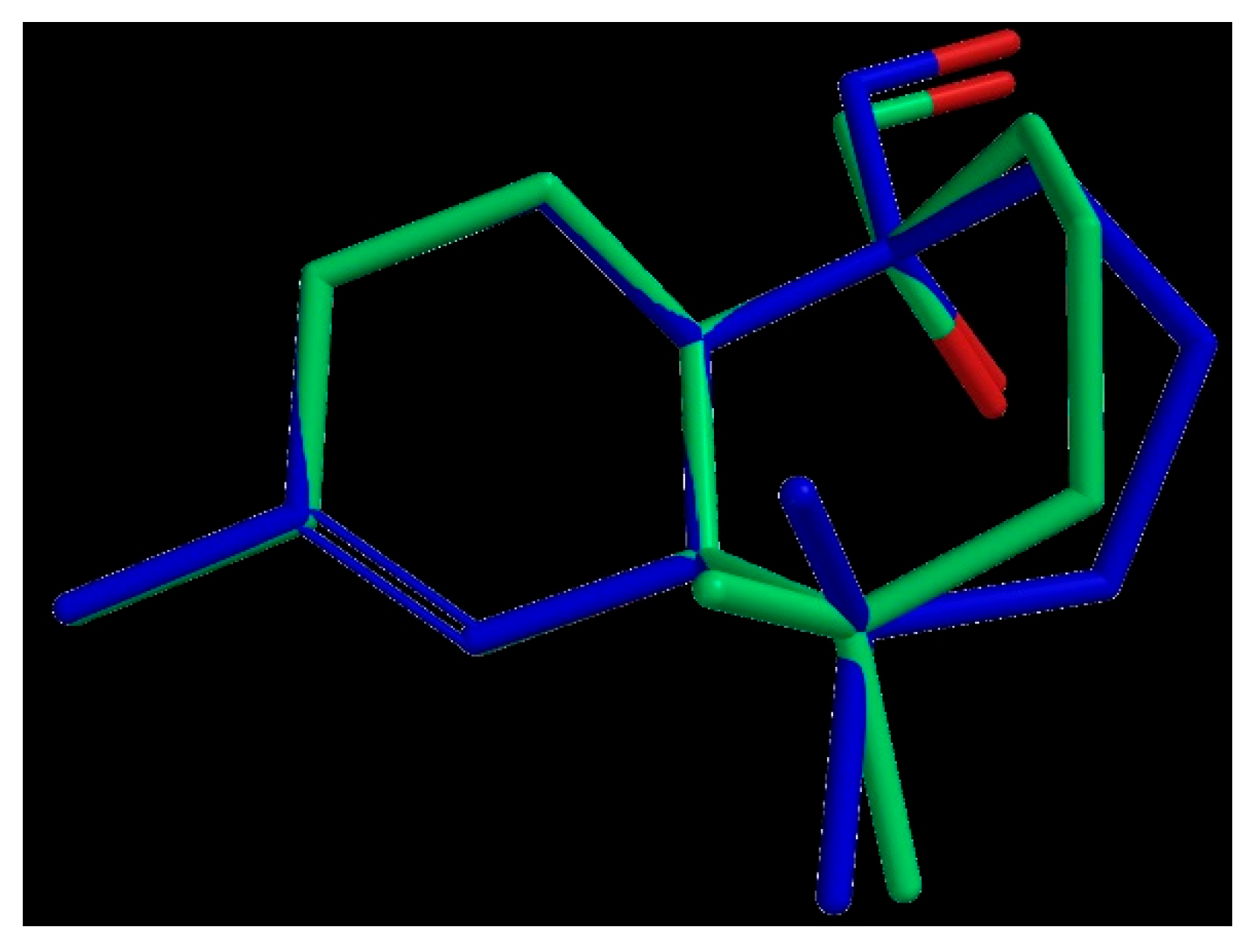
2.2. Structural Elucidation of New Natural Products
2.3. Plant Protection Effects
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Material, Extraction and Isolation
4.3. Derivatization of Natural Product 6
4.4. Spectroscopical Data (1H and 13C NMR) of Natural Products 1-8
4.5. Computational Calculations of 13C NMR Spectrum and Optical Rotation
4.6. Biopesticide Assays
4.6.1. Antifeedant Activity
4.6.2. Nematicidal Activity
4.6.3. Phytotoxic Effect
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Acheuk, F.; Basiouni, S.; Shehata, A. A.; Dick, K.; Hajri, H.; Lasram, S.; Yilmaz, M.; Emecki, M.; Tsiamis, G.; Spona-Friedl, M.; May-Simera, H.; Eisenreich, W.; Ntouglas, S. Status and prospects of botanical biopesticides in Europe and Mediterranean countries. Biomolecules 2022, 12, 311. [Google Scholar] [CrossRef]
- Lee, G. H.; Choi, K. C. Adverse effects of pesticides on the functions of immune system. Comp. Biochem. Physiol. C. 2020, 235, 108789. [Google Scholar] [CrossRef]
- Lo, C.C. Effect of pesticides on soil microbial community. J. Environ. Sci. Health B. 2010, 45(5), 348–359. [Google Scholar] [CrossRef]
- Villaverde, J. J.; Sandín-España, P.; Sevilla-Morán, B.; López-Goti, C.; Alonso-Prados, J. L. Biopesticides from natural products: Current development, legislative framework, and future trends. BioRes. 2016, 11(2), 5618–5640. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, A. Biopesticides: present status and the future prospects. J. Fertil Pestic. 2015, 6(2), 100–129. [Google Scholar] [CrossRef]
- Zaki, M. Natural products from Dittrichia Viscosa (Mini-Review). RHAZES: Green Appl. Chem. 2020, 9, 30–46. [Google Scholar] [CrossRef]
- Grauso, L.; Cesarano, G.; Zotti, M.; Ranesi, M.; Sun, W.; Bonanomi, G.; Lanzotti, V. Exploring Dittrichia viscosa (L.) Greuter phytochemical diversity to explain its antimicrobial, nematicidal and insecticidal activity. Phytochem. Rev. 2020, 19, 659–689. [Google Scholar] [CrossRef]
- Barrero, A. F.; Herrador, M. M.; Arteaga, P.; Catalán, J. V. Dittrichia viscosa L. Greuter: Phytochemistry and biological activity. Nat. Prod. Commun. 2008, 3(11), 1799–1804. [Google Scholar] [CrossRef]
- Rotundo, G.; Paventi, G.; Barberio, A.; De Cristofaro, A.; Notardonato, I.; Russo, M. V.; Germinara, G. S. Biological activity of Dittrichia viscosa (L.) Greuter extracts against adult Sitophilus granarius (L.) (Coleoptera, Curculionidae) and identification of active compounds. Sci. Rep. 2019, 9(1), 6429. [Google Scholar] [CrossRef]
- Araniti, F.; Lupini, A.; Sunseri, F.; Abenavoli, M. R. Allelopatic potential of Dittrichia viscosa (L.) W. Greuter mediated by VOCs: A physiological and metabolomic approach. PLoS One 2017, 12(1), 170161. [Google Scholar] [CrossRef]
- Murlanova, K.; Cohen, N.; Pinkus, A.; Vinnikova, L.; Pletnikov, M.; Kirby, M.; Gorelick, J.; Drori, E.; Pinhasov, A. Antidepressant-like effects of a chlorogenic acid-and cynarine-enriched fraction from Dittrichia viscosa root extract. Sci. Rep. 2022, 12(1), 3647. [Google Scholar] [CrossRef] [PubMed]
- Sladonja, B.; Poljuha, D.; Krapac, M.; Uzelac, M.; Mikulic-Petkovsek, M. Dittrichia viscosa: Native-Non Native Invader. Diversity 2021, 13(8), 380. [Google Scholar] [CrossRef]
- Perdikis, D.; Favas, C.; Lykouressis, D.; Fantinou, A. Ecological Relationships between Non-Cultivated Plants and Insect Predators in Agroecosystems: The Case of Dittrichia viscosa (Asteraceae) and Macrolophus melanotoma (Hemiptera: Miridae). Acta Oecol. 2007, 31(3), 299–306. [Google Scholar] [CrossRef]
- Rhimi, W.; Ben Salem, I.; Immediato, D.; Saidi, M.; Boulila, A.; Cafarchia, C. Chemical composition, antibacterial and antifungal activities of crude Dittrichia viscosa (L.) greuter leaf extracts. Molecules 2017, 22(7), 942. [Google Scholar] [CrossRef]
- Al-Qudah, M.; Al-Jaber, H.; Mayyas, A.; Abu-Orabi, S.; Abu Zarga, M. Chemical compositions of the essential oil from the jordanian medicinal plant Dittrichia viscosa. Jordan J. Chem. 2010, 5(4), 343–348. [Google Scholar]
- Gharred, N.; Dbeibia, A.; Falconieri, D.; Hammami, S.; Piras, A.; Dridi-Dhaouadi, S. Chemical composition, antibacterial and antioxidant activities of essential oils from flowers, leaves and aerial parts of Tunisian Dittrichia viscosa. J. Essent. Oil Res. 2019, 31(6), 582–589. [Google Scholar] [CrossRef]
- Gökbulut, A.; Özhana, O.; Satılmiş, B.; Batçioğlu, K.; Günal, S.; Şarer, E. Antioxidant and antimicrobial activities, and phenolic compounds of selected Inula species from Turkey. Nat. Prod. Commun. 2013, 8(4), 475–478. [Google Scholar] [CrossRef]
- Trimech, I.; Weiss, E. K.; Chedea, V. S.; Marin, D.; Detsi, A.; Ioannou, E.; Roussi, V.; Kefalas, P. Evaluation of anti-oxidant and acetylcholinesterase activity and identification of polyphenolics of the invasive weed Dittrichia viscosa. Phytochem. Anal. 2014, 25(5), 421–428. [Google Scholar] [CrossRef] [PubMed]
- Bohlmann, F.; Gupta, R. K. Ineupatorolide-like sesquiterpene lactones from Dittrichia viscosa. Phytochemistry. 1982, 21(6), 1443–1445. [Google Scholar] [CrossRef]
- Shtacher, G.; Kashman, Y. Chemical investigation of volatile constituents of Inula viscosa Ait. Tetrahedron 1971, 27(6), 1343–1349. [Google Scholar] [CrossRef]
- Muñoz-Rodríguez, P.; Carruthers, T.; Wood, J. R.; Williams, B. R.; Weitemier, K.; Kronmiller, B.; Ellis, D.; Anglin, N. L.; Longway, L.; Harris, S. A.; Rausher, M. D.; Kelly, S.; Liston, A.; Scotland, R. W. Reconciling Conflicting Phylogenies in the Origin of Sweet Potato and Dispersal to Polynesia. Curr. Biol. 2018, 28(8), 1246–1256. [Google Scholar] [CrossRef]
- Kim, J. H.; Jander, G. Myzus persicae (green peach aphid) feeding on Arabidopsis induces the formation of a deterrent indole glucosinolate. Plant J. 2007, 49(6), 1008–1019. [Google Scholar] [CrossRef]
- Greenslade, A. F. C.; Ward, J. L.; Martin, J. L.; Corol, D. I.; Clark, S. J.; Smart, L. E.; Aradottir, G. I. Triticum monococcum lines with distinct metabolic phenotypes and phloem-based partial resistance to the bird cherry-oat aphid Rhopalosiphum padi. Ann. Appl. Biol. 2016, 168(3), 435–449. [Google Scholar] [CrossRef]
- Alekcevetch, J. C.; de Lima Passianotto, A. L.; Ferreira, E. G. C.; Dos Santos, A. B.; da Silva, D. C. G.; Dias, W. P.; Belzile, F.; Abdelnoor, R. V.; Marcelino-Guimaraes, F.C. Genome-wide association study for resistance to the Meloidogyne javanica causing root-knot nematode in soybean. Theor. Appl. Genet. 2021, 134(3), 777–792. [Google Scholar] [CrossRef]
- Chaudhary, S.; Dutta, T. K.; Tyagi, N.; Shivakumara, T. N.; Papolu, P. K.; Chobhe, K. A.; Rao, U. Host-induced silencing of Mi-msp-1 confers resistance to root-knot nematode Meloidogyne incognita in eggplant. Transgenic Res. 28 2019, 327–340. [Google Scholar] [CrossRef]
- Kesraoui, S.; Andrés, M. F.; Berrocal-Lobo, M.; Soudani, S.; Gonzalez-Coloma, A. Direct and Indirect Effects of Essential Oils for Sustainable Crop Protection. Plants 2022, 11(16), 2144. [Google Scholar] [CrossRef] [PubMed]
- Dhekne, V. V.; Rao, A. S. Selective Oxidations with Lead Tetraacetate; A Convenient Preparation of 3-Methoxy-4-iso-Propylbehzylalcohol. Synt. Commun. 1978, 8(3), 135–141. [Google Scholar] [CrossRef]
- Jakupovic, J.; Schuster, A.; Bohlmann, F.; King, R. M.; Lander, N. S. Sesquiterpene lactones from Gnephosis species. Phytochemistry 1988, 27(10), 3181–3185. [Google Scholar] [CrossRef]
- Cheng, X.; Zeng, Q.; Ren, J.; Qin, J.; Zhang, S.; Shen, Y.; Zhu, J.; Zhang, F.; Chang, R.; Zhu, Y.; Zhang, W.; Jin, H. Sesquiterpene lactones from Inula falconeri, a plant endemic to the Himalayas, as potential anti-inflammatory agents. Eur. J. Med. 2011, 46(11), 5408–5415. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X. R.; Li, W. W.; Ren, J.; Zeng, Q.; Zhang, S. D.; Shen, Y. H.; Yan, S.-K.; Ye, J.; Jin, H.-Z.; Zhang, W.-D. Sesquiterpene lactones from Inula hookeri. Planta Med. 2012, 78(5), 465–471. [Google Scholar] [CrossRef]
- Da Silva, A. J.; Garcia, M.; Baker, P. M.; Rabi, J. A. 13C NMR spectra of natural products. 1—guaianolides. Org. Magn. Reson 1981, 16(3), 230–233. [Google Scholar] [CrossRef]
- Sülsen, V. P.; Martino, V. S. Sesquiterpene Lactones: Advances in their Chemistry and Biological Aspects, 1st ed.; Springer: Buenos Aires, 2018. [Google Scholar] [CrossRef]
- Ebihara, A.; Taguchi, R.; Jeelani, G.; Nozaki, T.; Suenaga, K.; Iwasaki, A. Kagimminols A and B, Cembrene-Type Diterpenes from an Okeania sp. Marine Cyanobacterium. J. Nat. Prod. 2024, 87(4), 16–23. [Google Scholar] [CrossRef]
- Nishiyama, M.; Tonouchi, A.; Maeda, H.; Hashimoto, M. DFT calculation–assisted stereo-structural assignment of arundifungin. Chirality 2020, 32(1), 17–31. [Google Scholar] [CrossRef]
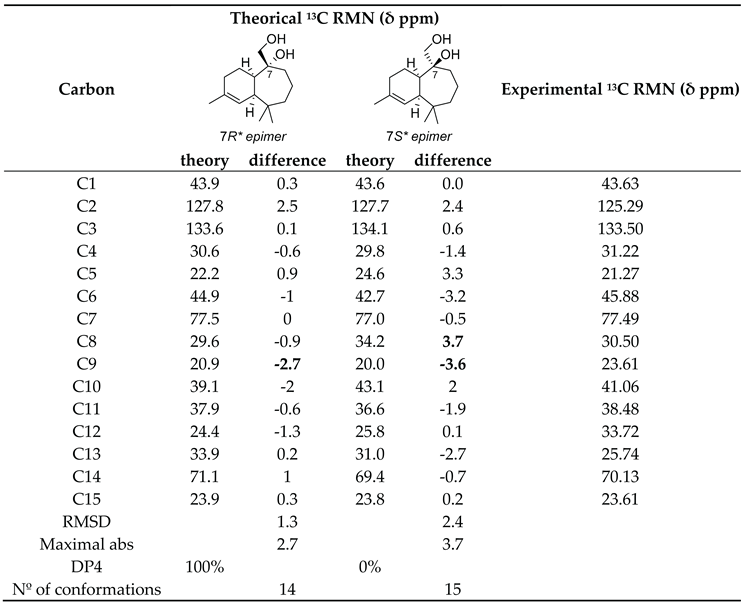
- Sánchez-Martínez, H. A.; Morán-Pinzón, J. A.; Del Olmo Fernández, E.; Eguiluz, D. L.; Adserias Vistue, J. F.; López-Pérez, J. L.; Guerrero de Leon, E. Synergistic Combination of NAPROC-13 and NMR (13)C DFT Calculations: A Powerful Approach for Revising the Structure of Natural Products. J. Nat. Prod. 2023, 86(10), 294–303. [Google Scholar] [CrossRef] [PubMed]
- Hehre, W.; Klunzinger, P.; Deppmeier, B.; Driessen, A.; Uchida, N.; Hashimoto, M.; Fukushi, E.; Takata, Y. Efficient Protocol for Accurately Calculating 13 C Chemical Shifts of Conformationally Flexible Natural Products: Scope, Assessment, and Limitations. J. Nat. Prod. 2019, 82(8), 299–306. [Google Scholar] [CrossRef] [PubMed]
- Smith, S. G.; Goodman, J. M. Assigning stereochemistry to single diastereoisomers by GIAO NMR calculation: The DP4 probability. J. Am. Chem. Soc. 2010, 132(37), 46–59. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, B. S. Issues and Challenges in Geomatics Education in Haryana State, India. Int. Arch. Photogramm. Remote Sens. Spatial Inf. Sci. 2012, 39, 37–40. [Google Scholar] [CrossRef]
- Singh, D.; Agarwal, S. K. Insecticidal principles of himalayan cedarwood oil. J. Chem. Ecol. 1988, 14, 1145–1151. [Google Scholar] [CrossRef]
- Valcárcel, F.; Olmeda, A. S.; González, M. G.; Andrés, M. F.; Navarro-Rocha, J.; González-Coloma, A. Acaricidal and insect antifeedant effects of essential oils from selected aromatic plants and their main components. Front. Agron. 2022, 3, 66–80. [Google Scholar] [CrossRef]
- Mamoci, E.; Cavoski, I.; Andrés, M. F.; Díaz, C. E.; González-Coloma, A. Chemical Characterization of the Aphid Antifeedant Extracts from Dittrichia viscosa and Ferula communis. Biochem. Syst. Ecol. 2012, 43, 101–107. [Google Scholar] [CrossRef]
- Reddy, S. E.; Kirti Dolma, S.; Koundal, R.; Singh, B. Chemical composition and insecticidal activities of essential oils against diamondback moth, Plutella xylostella (L.) (Lepidoptera: Yponomeutidae). Nat. Prod. Res. 2016, 30(16), 1834–1838. [Google Scholar] [CrossRef]
- González-Coloma, A.; Andrés, M. F.; Contreras, R.; Zúñiga, G. E.; Díaz, C. E. Sustainable production of insecticidal compounds from Persea indica. Plants 2022, 11(3), 418. [Google Scholar] [CrossRef]
- Oka, Y.; Ben-Daniel, B. H.; Cohen, Y. Control of Meloidogyne javanica by formulations of Inula viscosa leaf extracts. J. Nematol. 2006, 38(1), 46–51. [Google Scholar]
- Boari, A.; Vurro, M.; Calabrese, G. J.; Mahmoud, M. N. Z.; Cazzato, E.; Fracchiolla, M. Evaluation of Dittrichia viscosa (L.) Greuter dried Biomass for weed management. Plants 2021, 10(1), 1–11. [Google Scholar] [CrossRef]
- Dor, E.; Hershenhorn, J. Allelopathic effects of Inula viscosa leaf extracts on weeds. Allelopathy J. 2012, 30(2), 281–290. [Google Scholar]
- Andolfi, A.; Zermane, N.; Cimmino, A.; Avolio, F.; Boari, A.; Vurro, M.; Evidente, A. Inuloxins A–D, phytotoxic bi-and tri-cyclic sesquiterpene lactones produced by Inula viscosa: Potential for broomrapes and field dodder management. Phytochemistry 2013, 86, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Aparicio, M.; Masi, M.; Cimmino, A.; Vilariño, S.; Evidente, A. Allelopathic effect of quercetin, a flavonoid from Fagopyrum esculentum roots in the radicle growth of Phelipanche ramosa: quercetin natural and semisynthetic analogues were used for a structure-activity relationship investigation. Plants 2021, 10(3), 543. [Google Scholar] [CrossRef] [PubMed]
- Villani, A.; Zonno, M. C.; de Leonardis, S.; Vurro, M.; Paciolla, C. Inuloxin A Inhibits Seedling Growth and Affects Redox System of Lycopersicon Esculentum Mill. and Lepidium Sativum L. Biomolecules 2022, 12(2). [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Roberto, C.E.; Pinheiro, P.F.; de Assis Alves, T.; da Silva, J.A.; Praça-Fontes, M.M.; Soares, T.C.B. Phytogenotoxicity of thymol and semisynthetic thymoxyacetic acid in pre/post emergence of model plants and weeds. Environ. Sci. Pollut. 2023, 30, 38955–38969. [Google Scholar] [CrossRef]
- Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; et al. Gaussian 16 Revision C. 01, 2016. Gaussian Inc. Wallingford CT 1, 572.
- Lynch, B. J.; Zhao, Y.; Truhlar, D. G. Effectiveness of diffuse basis functions for calculating relative energies by density functional theory. J. Phys. Chem. A 2003, 107(9), 1384–1388. [Google Scholar] [CrossRef]
- Stephens, P. J.; Devlin, F. J.; Cheeseman, J. R.; Frisch, M. J. Calculation of Optical Rotation Using Density Functional Theory. J. Phys. Chem. A 2001, 105(22), 5356–5371. [Google Scholar] [CrossRef]
- Tomasi, J.; Mennucci, B.; Cammi, R. Quantum Mechanical Continuum Solvation Models. Chem. Rev. 2005, 105(8), 2999–3094. [Google Scholar] [CrossRef] [PubMed]
- Rueden, C. T.; Schindelin, J.; Hiner, M. C.; DeZonia, B. E.; Walter, A. E.; Arena, E. T.; Eliceiri, K. W. ImageJ2: ImageJ for the next generation of scientific image data. BMC Bioinform. 2017, 18, 529. [Google Scholar] [CrossRef]
- Ruiz-Vásquez, L.; Ruiz Mesia, L.; Caballero Ceferino, H. D.; Ruiz Mesia, W.; Andrés, M. F.; Díaz, C. E.; Gonzalez-Coloma, A. Antifungal and herbicidal potential of Piper essential oils from the Peruvian Amazonia. Plants 2022, 11(14), 1793. [Google Scholar] [CrossRef]
| Position | 5 | 6 | 8 |
|---|---|---|---|
| 1 | 2.23, m | 2.23, bs | 2.58, bs |
| 2a | 1.93, dt (11.1, 5.3) | 5.61, dd (5.6, 2.2) | 7.32, d (6.0) |
| 2b | 1.69, m | ||
| 3a | 2.40, m | ||
| 3b | 2.33, m | ||
| 4a | 2.01, bd (8.2) | 2.43, bd (18.6) | |
| 4b | 2.26, dt (18.6, 9.0) | ||
| 5a | 2.24, m | 1.70, m | 1.71, m |
| 5b | 1.44, m | 1.57, m | |
| 6 | 3.47, bt (9.5) | 1.96, m | 1.97, d (13.3) |
| 7 | 2.34, td (10.5, 7.6) | ||
| 8a | 4.54, td (11.1, 3.4) | 1.64, m | 1.66, m |
| 8b | 1.54, m | 1.54, m | |
| 9a | 3.17, dd (16.3, 3.1) | 1.69, m | 1.75, m |
| 9b | 2.63, dd (16.3, 11.6) | ||
| 10a | 1.90, m | 2.02, m | |
| 10b | 1.42, dq (14.4, 5.0) | 1.46, ddd (14.2, 6.6, 3.3) | |
| 11 | 2.92 p (7.7) | ||
| 12 | 1.01, s | 1.12, s | |
| 13a | 1.34, d (7.7) | 0.88, s | 0.89, s |
| 13b | |||
| 14a | 4.93, bs | 3.49, d (10.8) | 3.57, d (11.0) |
| 14b | 4.91, bs | 3.39, d (10.8) | 3.39, d (11.0) |
| 15a | 5.10, bs | 1.68, bs | |
| 15b | 4.99, bs |
| Position | 5 | 6 | 8 |
|---|---|---|---|
| 1 | 45.93 (CH) | 43.63 (CH) | 43.78 (CH) |
| 2 | 30.06 (CH2) | 125.31 (CH) | 146.49 (CH) |
| 3 | 34.20 (CH2) | 133.52 (C) | 128.93 (C) |
| 4 | 152.50 (C) | 31.22 (CH2) | 25.38 (CH2) |
| 5 | 54.94 (CH) | 21.27 (CH2) | 20.42 (CH2) |
| 6 | 71.19 (CH) | 45.86 (CH) | 45.92 (CH) |
| 7 | 54.83 (CH) | 77.49 (C) | 77.22 (C) |
| 8 | 76.66 (CH) | 30.49 (CH2) | 31.36 (CH2) |
| 9 | 39.64 (CH2) | 19.53 (CH2) | 19.27 (CH2) |
| 10 | 144.08 (C) | 41.05 (CH2) | 41.38 (CH2) |
| 11 | 39.06 (CH) | 38.47 (C) | 38.84 (C) |
| 12 | 179.14 (C) | 33.73 (CH3) | 33.50 (CH3) |
| 13 | 10.48 (CH3) | 25.74 (CH3) | 27.04 (CH3) |
| 14 | 110.67 (CH2) | 70.13 (CH2) | 69.65 (CH2) |
| 15 | 109.61 (CH2) | 26.60 (CH3) | 171.61 (C) |
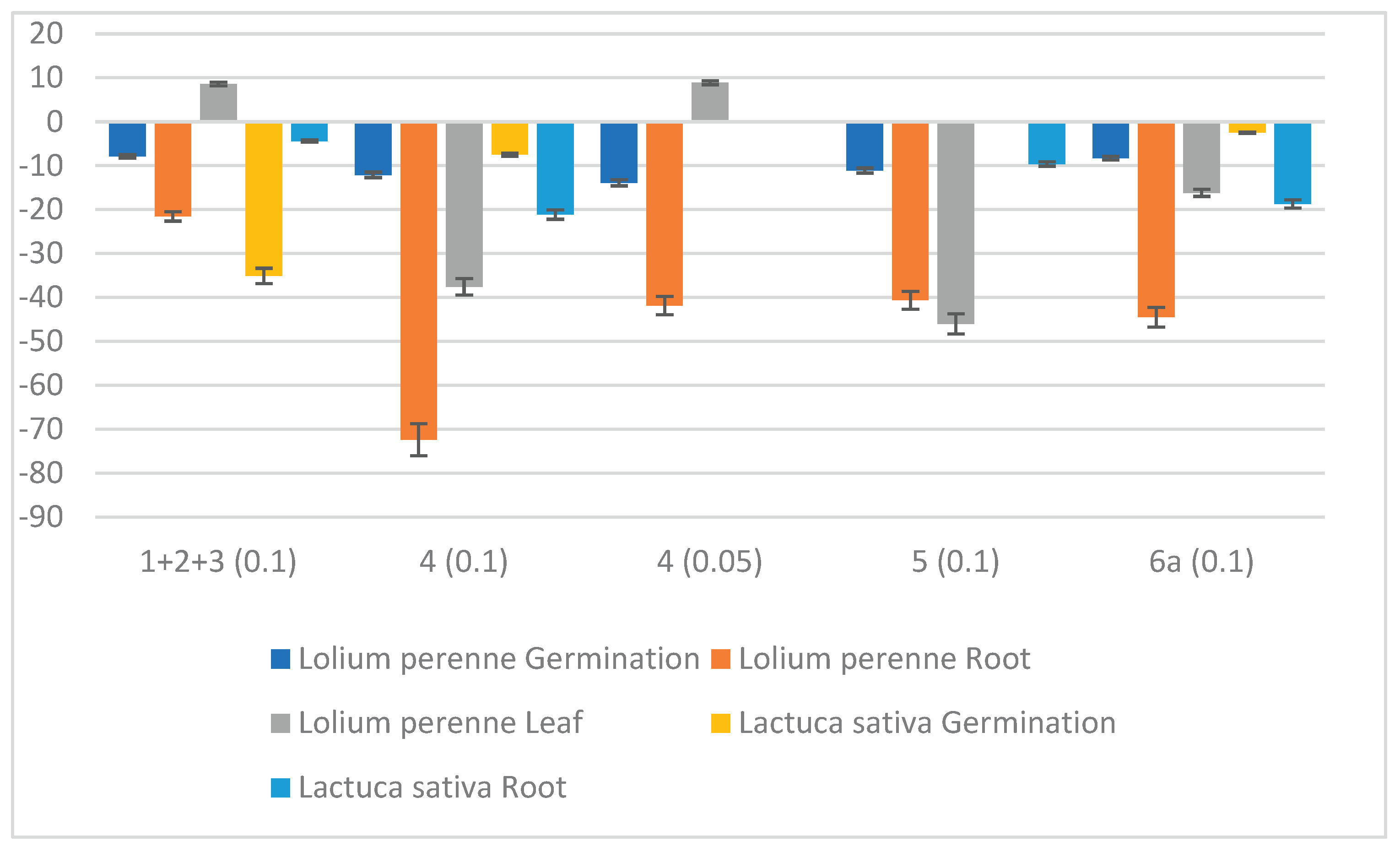
| Compound | μg/cm2 | S. littoralis | M. persicae | R. padi |
|---|---|---|---|---|
| %FI2 (N = 6-10) | %SI2 (N = 20) | |||
| 1+2+3 | 50 | 73.13 ± 10.01 | 70.33 ± 7.72 | 71.30 ± 6.12 |
| EC501 | 32.35 (23.82-43.93) | 16.54 (9.17-29.84) | 19.14 (11.41-32.08) | |
| 4 | 50 | 41.70 ± 8.91 | 57.65 ± 8.49 | 56.53 ± 8.03 |
| EC501 | >50 | ≈50 | ≈50 | |
| 5 | 50 | 3.60 ± 3.65 | 81.69 ± 5.21 | 65.41 ± 5.45 |
| EC501 | >50 | 18.71 (14.00-25.01) | ≈50 | |
| 6a | 50 | 86.69 ± 4.26 | 55.31 ± 7.43 | 52.45 ± 7.38 |
| EC501 | 22.10 (15.34-31.84) | >50 | ≈50 | |
| Compound | mg/mL | M. javanica | MLD |
|---|---|---|---|
| 1-3 | 0.5 | 25.83 ± 7.20 | >0.5 |
| 4 | 0.5 | 100.00 | 0.5 |
| 5 | 0.5 | 0.28 ± 0.12 | >0.5 |
| 6a | 0.5 | 0.00 | >0.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




