Submitted:
14 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
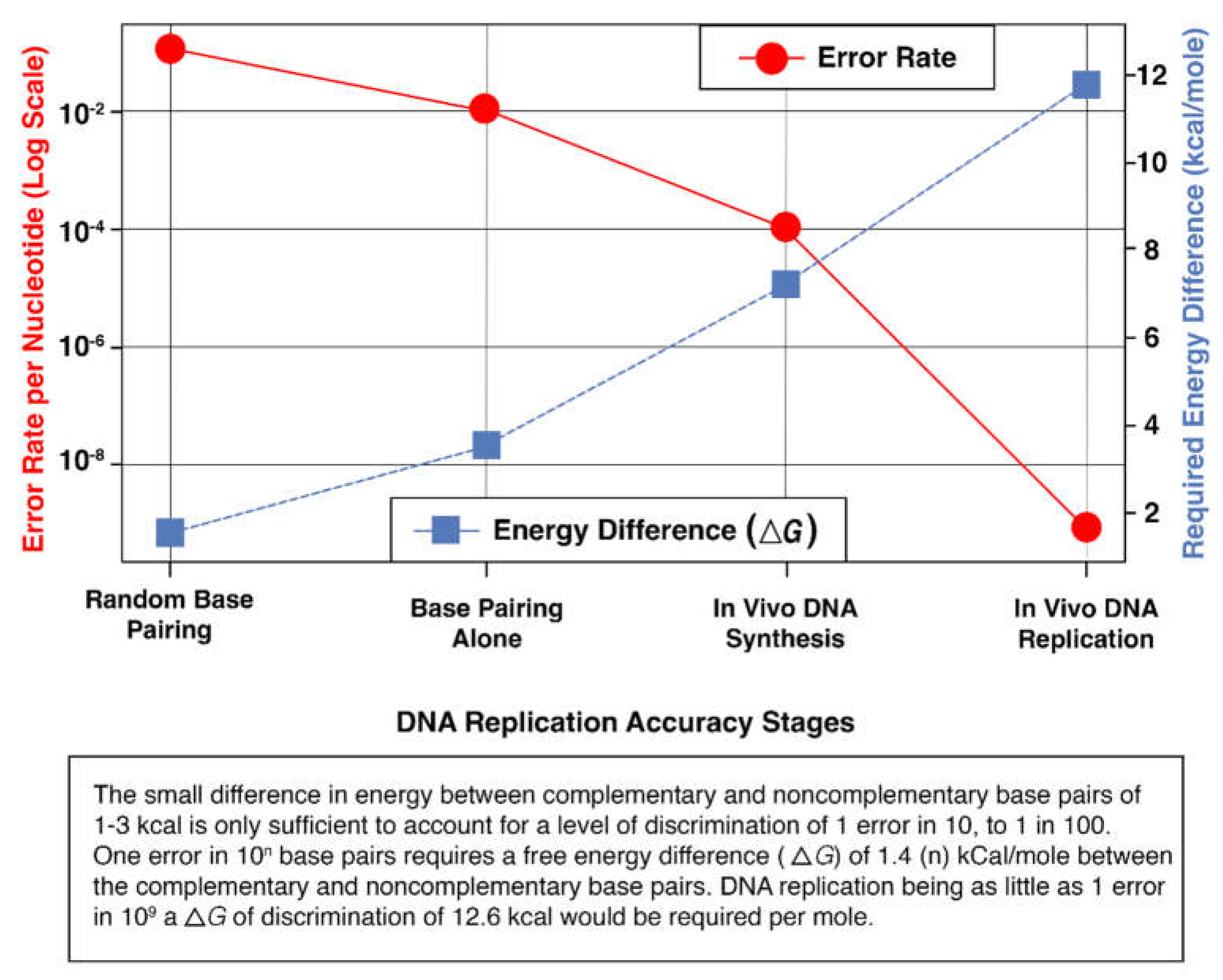
2.1. Genetic and Metabolic Constraints on Proofreading Fidelity
2.2. Approach and Rationale
2.3. Analysis and Synthesis
3. Results
3.1. The Hallmarks of Cancer
3.1.1. The Hallmarks of Cancer and Genomic Instability
3.1.2. DNA Replication Errors and Genomic Instability in Carcinogenesis
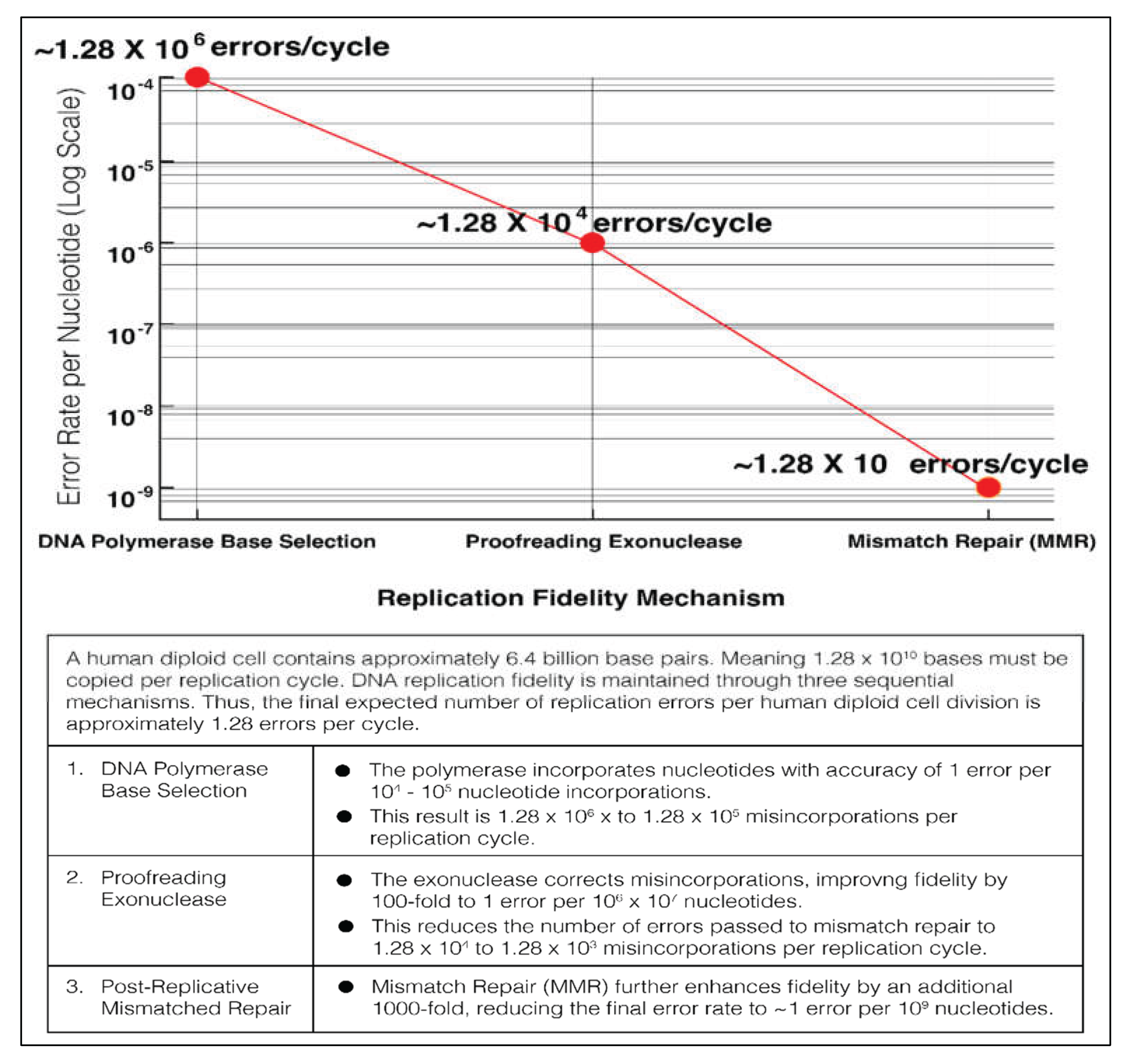
3.1.3. DNA Replication and Fidelity in Eukaryotes
3.1.4. Inhibition of Proofreading Activity is Mutagenic
3.1.5. Inhibition of Proofreading Activity is Carcinogenic
3.2. Energy Metabolism in Obesity and Associated Disorders
3.2.1. The Hallmarks of Metabolic Syndrome
3.2.2. Obesity and Associated Disorders Are Characterized by Impaired Energy Metabolism
3.2.3. Impaired Energy Metabolism in Obesity: The Central Role of AMPK
3.2.4. AMP Regulates Energy Production and Consumption
3.2.5. Impaired AMPK Activity is Linked with Carcinogenesis
3.2.6. AMPK Activation Prevents These Cancers
4. Discussion
- The mutation rate is metabolically regulated via energy-sensitive proofreading modulation.
- This regulation is integrated within the single 122 kDa core of the DNA polymerase complex, linking impaired energy metabolism directly to genomic instability.
- “Genomic Instability and mutation”. Impaired energy metabolism, increasing AMP-mediated inhibition of DNA polymerase proofreading accelerates stochastic mutation accumulation and genomic instability.
- “Dysregulated cellular energetics”, obesity-associated AMPK impairment represents a primary metabolic lesion that proceeds and facilitates genomic instability.
- “Sustaining proliferative signaling”: an increased mutational burden raises the probability of activating driver mutations.
- “Resisting Cell Death”: Energy stress-induced prioritization of survival over fidelity favors short-term viability at the cost of long-term genomic integrity.
- “Tumor evolution and heterogeneity”: A higher mutation rate increases clonal diversity and accelerates evolutionary selection.
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N Engl J Med 2003, 348, 1625–1638. [Google Scholar] [CrossRef]
- Bhaskaran, K.; Douglas, I.; Forbes, H.; dos-Santos-Silva, I.; Leon, D.A.; Smeeth, L. Body-mass index and risk of 22 specific cancers: a population-based cohort study of 5.24 million UK adults. Lancet 2014, 384, 755–765. [Google Scholar] [CrossRef]
- Lauby-Secretan, B.; Scoccianti, C.; Loomis, D.; Grosse, Y.; Bianchini, F.; Working, G.; Straif; K; International Agency for Research on Cancer Handbook. Body Fatness and Cancer--Viewpoint of the IARC Working Group. N Engl J Med 2016, 375, 794–798. [Google Scholar] [CrossRef] [PubMed]
- Win, A.K.; Dowty, J.G.; Antill, Y.C.; English, D.R.; Baron, J.A.; Young, J.P.; Giles, G.G.; Southey, M.C.; Winship, I.; Lipton, L.; et al. Body mass index in early adulthood and endometrial cancer risk for mismatch repair gene mutation carriers. Obstet Gynecol 2011, 117, 899–905. [Google Scholar] [CrossRef] [PubMed]
- Win, A.K.; Dowty, J.G.; English, D.R.; Campbell, P.T.; Young, J.P.; Winship, I.; Macrae, F.A.; Lipton, L.; Parry, S.; Young, G.P.; et al. Body mass index in early adulthood and colorectal cancer risk for carriers and non-carriers of germline mutations in DNA mismatch repair genes. Br J Cancer 2011, 105, 162–169. [Google Scholar] [CrossRef]
- Li, X.; Jansen, L.; Chang-Claude, J.; Hoffmeister, M.; Brenner, H. Risk of Colorectal Cancer Associated With Lifetime Excess Weight. JAMA Oncol 2022, 8, 730–737. [Google Scholar] [CrossRef]
- Sung, H.; Siegel, R.L.; Rosenberg, P.S.; Jemal, A. Emerging cancer trends among young adults in the USA: analysis of a population-based cancer registry. Lancet Public Health 2019, 4, e137–e147. [Google Scholar] [CrossRef]
- Simoneau, J.A.; Veerkamp, J.H.; Turcotte, L.P.; Kelley, D.E. Markers of capacity to utilize fatty acids in human skeletal muscle: relation to insulin resistance and obesity and effects of weight loss. FASEB J 1999, 13, 2051–2060. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E.; Harlan, D.M.; Archer, M.C.; Bergenstal, R.M.; Gapstur, S.M.; Habel, L.A.; Pollak, M.; Regensteiner, J.G.; Yee, D. Diabetes and cancer: a consensus report. Diabetes Care 2010, 33, 1674–1685. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: the next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov 2022, 12, 31–46. [Google Scholar] [CrossRef]
- Loeb, L.A.; Springgate, C.F.; Battula, N. Errors in DNA replication as a basis of malignant changes. Cancer Res 1974, 34, 2311–2321. [Google Scholar]
- Loeb, L.A. Human Cancers Express a Mutator Phenotype: Hypothesis, Origin, and Consequences. Cancer Res 2016, 76, 2057–2059. [Google Scholar] [CrossRef]
- Alexandrov, L.B.; Nik-Zainal, S.; Wedge, D.C.; Aparicio, S.A.; Behjati, S.; Biankin, A.V.; Bignell, G.R.; Bolli, N.; Borg, A.; Borresen-Dale, A.L.; et al. Signatures of mutational processes in human cancer. Nature 2013, 500, 415–421. [Google Scholar] [CrossRef]
- Vogelstein, B.; Fearon, E.R.; Hamilton, S.R.; Kern, S.E.; Preisinger, A.C.; Leppert, M.; Nakamura, Y.; White, R.; Smits, A.M.; Bos, J.L. Genetic alterations during colorectal-tumor development. N Engl J Med 1988, 319, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Tomasetti, C.; Vogelstein, B.; Parmigiani, G. Half or more of the somatic mutations in cancers of self-renewing tissues originate prior to tumor initiation. Proc Natl Acad Sci U S A 2013, 110, 1999–2004. [Google Scholar] [CrossRef]
- Tomasetti, C.; Vogelstein, B. Cancer etiology. Variation in cancer risk among tissues can be explained by the number of stem cell divisions. Science 2015, 347, 78–81. [Google Scholar] [CrossRef] [PubMed]
- Tomasetti, C.; Li, L.; Vogelstein, B. Stem cell divisions, somatic mutations, cancer etiology, and cancer prevention. Science 2017, 355, 1330–1334. [Google Scholar] [CrossRef]
- Vogelstein, B.; Papadopoulos, N.; Velculescu, V.E.; Zhou, S.; Diaz, L.A., Jr.; Kinzler, K.W. Cancer genome landscapes. Science 2013, 339, 1546–1558. [Google Scholar] [CrossRef]
- Byrnes, J.J.; Downey, K.M.; Black, V.L.; So, A.G. A new mammalian DNA polymerase with 3’ to 5’ exonuclease activity: DNA polymerase delta. Biochemistry 1976, 15, 2817–2823. [Google Scholar] [CrossRef]
- Goscin, L.P.; Byrnes, J.J. DNA polymerase delta: one polypeptide, two activities. Biochemistry 1982, 21, 2513–2518. [Google Scholar] [CrossRef]
- Lee, M.Y.; Tan, C.K.; So, A.G.; Downey, K.M. Purification of deoxyribonucleic acid polymerase delta from calf thymus: partial characterization of physical properties. Biochemistry 1980, 19, 2096–2101. [Google Scholar] [CrossRef] [PubMed]
- Bravo, R.; Frank, R.; Blundell, P.A.; Macdonald-Bravo, H. Cyclin/PCNA is the auxiliary protein of DNA polymerase-delta. Nature 1987, 326, 515–517. [Google Scholar] [CrossRef] [PubMed]
- Prelich, G.; Tan, C.K.; Kostura, M.; Mathews, M.B.; So, A.G.; Downey, K.M.; Stillman, B. Functional identity of proliferating cell nuclear antigen and a DNA polymerase-delta auxiliary protein. Nature 1987, 326, 517–520. [Google Scholar] [CrossRef] [PubMed]
- Syvaoja, J.; Linn, S. Characterization of a large form of DNA polymerase delta from HeLa cells that is insensitive to proliferating cell nuclear antigen. J Biol Chem 1989, 264, 2489–2497. [Google Scholar] [CrossRef]
- Syvaoja, J.; Suomensaari, S.; Nishida, C.; Goldsmith, J.S.; Chui, G.S.; Jain, S.; Linn, S. DNA polymerases alpha, delta, and epsilon: three distinct enzymes from HeLa cells. Proc Natl Acad Sci U S A 1990, 87, 6664–6668. [Google Scholar] [CrossRef]
- Burgers, P.M.; Bambara, R.A.; Campbell, J.L.; Chang, L.M.; Downey, K.M.; Hubscher, U.; Lee, M.Y.; Linn, S.M.; So, A.G.; Spadari, S. Revised nomenclature for eukaryotic DNA polymerases. Eur J Biochem 1990, 191, 617–618. [Google Scholar] [CrossRef]
- Lu, C.D.; Byrnes, J.J. PCNA-dependent DNA polymerase delta from rabbit bone marrow. Biochemistry 1992, 31, 12403–12409. [Google Scholar] [CrossRef]
- Chung, D.W.; Zhang, J.A.; Tan, C.K.; Davie, E.W.; So, A.G.; Downey, K.M. Primary structure of the catalytic subunit of human DNA polymerase delta and chromosomal location of the gene. Proc Natl Acad Sci U S A 1991, 88, 11197–11201. [Google Scholar] [CrossRef] [PubMed]
- Kesti, T.; Frantti, H.; Syvaoja, J.E. Molecular cloning of the cDNA for the catalytic subunit of human DNA polymerase epsilon. J Biol Chem 1993, 268, 10238–10245. [Google Scholar] [CrossRef] [PubMed]
- Bebenek, A.; Ziuzia-Graczyk, I. Fidelity of DNA replication-a matter of proofreading. Curr Genet 2018, 64, 985–996. [Google Scholar] [CrossRef] [PubMed]
- Byrnes, J.J.; Downey, K.M.; Que, B.G.; Lee, M.Y.; Black, V.L.; So, A.G. Selective inhibition of the 3’ to 5’ exonuclease activity associated with DNA polymerases: a mechanism of mutagenesis. Biochemistry 1977, 16, 3740–3746. [Google Scholar] [CrossRef]
- Que, B.G.; Downey, K.M.; So, A.G. Mechanisms of selective inhibition of 3’ to 5’ exonuclease activity of Escherichia coli DNA polymerase I by nucleoside 5’-monophosphates. Biochemistry 1978, 17, 1603–1606. [Google Scholar] [CrossRef]
- Kunkel, T.A.; Sabatino, R.D.; Bambara, R.A. Exonucleolytic proofreading by calf thymus DNA polymerase delta. Proc Natl Acad Sci U S A 1987, 84, 4865–4869. [Google Scholar] [CrossRef]
- Thomas, D.C.; Roberts, J.D.; Sabatino, R.D.; Myers, T.W.; Tan, C.K.; Downey, K.M.; So, A.G.; Bambara, R.A.; Kunkel, T.A. Fidelity of mammalian DNA replication and replicative DNA polymerases. Biochemistry 1991, 30, 11751–11759. [Google Scholar] [CrossRef]
- Goldsby, R.E.; Lawrence, N.A.; Hays, L.E.; Olmsted, E.A.; Chen, X.; Singh, M.; Preston, B.D. Defective DNA polymerase-delta proofreading causes cancer susceptibility in mice. Nat Med 2001, 7, 638–639. [Google Scholar] [CrossRef]
- Albertson, T.M.; Ogawa, M.; Bugni, J.M.; Hays, L.E.; Chen, Y.; Wang, Y.; Treuting, P.M.; Heddle, J.A.; Goldsby, R.E.; Preston, B.D. DNA polymerase epsilon and delta proofreading suppress discrete mutator and cancer phenotypes in mice. Proc Natl Acad Sci U S A 2009, 106, 17101–17104. [Google Scholar] [CrossRef]
- Cancer Genome Atlas, N. Comprehensive molecular characterization of human colon and rectal cancer. Nature 2012, 487, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Research, N.; Kandoth, C.; Schultz, N.; Cherniack, A.D.; Akbani, R.; Liu, Y.; Shen, H.; Robertson, A.G.; Pashtan, I.; Shen, R.; et al. Integrated genomic characterization of endometrial carcinoma. Nature 2013, 497, 67–73. [Google Scholar] [CrossRef]
- Church, D.N.; Briggs, S.E.; Palles, C.; Domingo, E.; Kearsey, S.J.; Grimes, J.M.; Gorman, M.; Martin, L.; Howarth, K.M.; Hodgson, S.V.; et al. DNA polymerase epsilon and delta exonuclease domain mutations in endometrial cancer. Hum Mol Genet 2013, 22, 2820–2828. [Google Scholar] [CrossRef]
- Palles, C.; Cazier, J.B.; Howarth, K.M.; Domingo, E.; Jones, A.M.; Broderick, P.; Kemp, Z.; Spain, S.L.; Guarino, E.; Salguero, I.; et al. Germline mutations affecting the proofreading domains of POLE and POLD1 predispose to colorectal adenomas and carcinomas. Nat Genet 2013, 45, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Rayner, E.; van Gool, I.C.; Palles, C.; Kearsey, S.E.; Bosse, T.; Tomlinson, I.; Church, D.N. A panoply of errors: polymerase proofreading domain mutations in cancer. Nat Rev Cancer 2016, 16, 71–81. [Google Scholar] [CrossRef]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2012, 2, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Park, V.S.; Pursell, Z.F. POLE proofreading defects: Contributions to mutagenesis and cancer. DNA Repair (Amst) 2019, 76, 50–59. [Google Scholar] [CrossRef]
- Reaven, G.M. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 1988, 37, 1595–1607. [Google Scholar] [CrossRef]
- Ruderman, N.; Prentki, M. AMP kinase and malonyl-CoA: targets for therapy of the metabolic syndrome. Nat Rev Drug Discov 2004, 3, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Mootha, V.K.; Lindgren, C.M.; Eriksson, K.F.; Subramanian, A.; Sihag, S.; Lehar, J.; Puigserver, P.; Carlsson, E.; Ridderstrale, M.; Laurila, E.; et al. PGC-1alpha-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat Genet 2003, 34, 267–273. [Google Scholar] [CrossRef]
- Patti, M.E.; Butte, A.J.; Crunkhorn, S.; Cusi, K.; Berria, R.; Kashyap, S.; Miyazaki, Y.; Kohane, I.; Costello, M.; Saccone, R.; et al. Coordinated reduction of genes of oxidative metabolism in humans with insulin resistance and diabetes: Potential role of PGC1 and NRF1. Proc Natl Acad Sci U S A 2003, 100, 8466–8471. [Google Scholar] [CrossRef]
- Petersen, K.F.; Dufour, S.; Befroy, D.; Garcia, R.; Shulman, G.I. Impaired mitochondrial activity in the insulin-resistant offspring of patients with type 2 diabetes. N Engl J Med 2004, 350, 664–671. [Google Scholar] [CrossRef]
- Ruderman, N.B.; Carling, D.; Prentki, M.; Cacicedo, J.M. AMPK, insulin resistance, and the metabolic syndrome. J Clin Invest 2013, 123, 2764–2772. [Google Scholar] [CrossRef]
- Esposito, K.; Chiodini, P.; Colao, A.; Lenzi, A.; Giugliano, D. Metabolic syndrome and risk of cancer: a systematic review and meta-analysis. Diabetes Care 2012, 35, 2402–2411. [Google Scholar] [CrossRef]
- Ramaiah, A.; Hathaway, J.A.; Atkinson, D.E. Adenylate as a Metabolic Regulator. Effect on Yeast Phosphofructokinase Kinetics. J Biol Chem 1964, 239, 3619–3622. [Google Scholar] [CrossRef]
- Krebs, H. The Croonian Lecture, 1963. Gluconeogenesis. Proc R Soc Lond B Biol Sci 1964, 159, 545–564. [Google Scholar] [CrossRef]
- Yeh, L.A.; Lee, K.H.; Kim, K.H. Regulation of rat liver acetyl-CoA carboxylase. Regulation of phosphorylation and inactivation of acetyl-CoA carboxylase by the adenylate energy charge. J Biol Chem 1980, 255, 2308–2314. [Google Scholar] [CrossRef]
- Ferrer, A.; Caelles, C.; Massot, N.; Hegardt, F.G. Activation of rat liver cytosolic 3-hydroxy-3-methylglutaryl coenzyme A reductase kinase by adenosine 5’-monophosphate. Biochem Biophys Res Commun 1985, 132, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Carling, D.; Zammit, V.A.; Hardie, D.G. A common bicyclic protein kinase cascade inactivates the regulatory enzymes of fatty acid and cholesterol biosynthesis. FEBS Lett 1987, 223, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Hardie, D.G.; Carling, D. The AMP-activated protein kinase: a multisubstrate regulator of lipid metabolism. Trends Biochem Sci 1989, 14, 20–23. [Google Scholar] [CrossRef]
- Hawley, S.A.; Selbert, M.A.; Goldstein, E.G.; Edelman, A.M.; Carling, D.; Hardie, D.G. 5’-AMP activates the AMP-activated protein kinase cascade, and Ca2+/calmodulin activates the calmodulin-dependent protein kinase I cascade, via three independent mechanisms. J Biol Chem 1995, 270, 27186–27191. [Google Scholar] [CrossRef] [PubMed]
- Gowans, G.J.; Hawley, S.A.; Ross, F.A.; Hardie, D.G. AMP is a true physiological regulator of AMP-activated protein kinase by both allosteric activation and enhancing net phosphorylation. Cell Metab 2013, 18, 556–566. [Google Scholar] [CrossRef]
- Hardie, D.G.; Hawley, S.A. AMP-activated protein kinase: the energy charge hypothesis revisited. Bioessays 2001, 23, 1112–1119. [Google Scholar] [CrossRef] [PubMed]
- Ross, F.A.; MacKintosh, C.; Hardie, D.G. AMP-activated protein kinase: a cellular energy sensor that comes in 12 flavours. FEBS J 2016, 283, 2987–3001. [Google Scholar] [CrossRef]
- Viollet, B.; Horman, S.; Leclerc, J.; Lantier, L.; Foretz, M.; Billaud, M.; Giri, S.; Andreelli, F. AMPK inhibition in health and disease. Crit Rev Biochem Mol Biol 2010, 45, 276–295. [Google Scholar] [CrossRef] [PubMed]
- Hardie, D.G. AMP-activated protein kinase - a journey from 1 to 100 downstream targets. Biochem J 2022, 479, 2327–2343. [Google Scholar] [CrossRef]
- Giardiello, F.M.; Brensinger, J.D.; Tersmette, A.C.; Goodman, S.N.; Petersen, G.M.; Booker, S.V.; Cruz-Correa, M.; Offerhaus, J.A. Very high risk of cancer in familial Peutz-Jeghers syndrome. Gastroenterology 2000, 119, 1447–1453. [Google Scholar] [CrossRef]
- Hemminki, A.; Markie, D.; Tomlinson, I.; Avizienyte, E.; Roth, S.; Loukola, A.; Bignell, G.; Warren, W.; Aminoff, M.; Hoglund, P.; et al. A serine/threonine kinase gene defective in Peutz-Jeghers syndrome. Nature 1998, 391, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Jenne, D.E.; Reimann, H.; Nezu, J.; Friedel, W.; Loff, S.; Jeschke, R.; Muller, O.; Back, W.; Zimmer, M. Peutz-Jeghers syndrome is caused by mutations in a novel serine threonine kinase. Nat Genet 1998, 18, 38–43. [Google Scholar] [CrossRef]
- Hawley, S.A.; Boudeau, J.; Reid, J.L.; Mustard, K.J.; Udd, L.; Makela, T.P.; Alessi, D.R.; Hardie, D.G. Complexes between the LKB1 tumor suppressor, STRAD alpha/beta and MO25 alpha/beta are upstream kinases in the AMP-activated protein kinase cascade. J Biol 2003, 2, 28. [Google Scholar] [CrossRef]
- Woods, A.; Johnstone, S.R.; Dickerson, K.; Leiper, F.C.; Fryer, L.G.; Neumann, D.; Schlattner, U.; Wallimann, T.; Carlson, M.; Carling, D. LKB1 is the upstream kinase in the AMP-activated protein kinase cascade. Curr Biol 2003, 13, 2004–2008. [Google Scholar] [CrossRef]
- Shaw, R.J.; Kosmatka, M.; Bardeesy, N.; Hurley, R.L.; Witters, L.A.; DePinho, R.A.; Cantley, L.C. The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc Natl Acad Sci U S A 2004, 101, 3329–3335. [Google Scholar] [CrossRef]
- Hardie, D.G.; Alessi, D.R. LKB1 and AMPK and the cancer-metabolism link - ten years after. BMC Biol 2013, 11, 36. [Google Scholar] [CrossRef]
- Gauthier, M.S.; O’Brien, E.L.; Bigornia, S.; Mott, M.; Cacicedo, J.M.; Xu, X.J.; Gokce, N.; Apovian, C.; Ruderman, N. Decreased AMP-activated protein kinase activity is associated with increased inflammation in visceral adipose tissue and with whole-body insulin resistance in morbidly obese humans. Biochem Biophys Res Commun 2011, 404, 382–387. [Google Scholar] [CrossRef]
- Xu, X.J.; Gauthier, M.S.; Hess, D.T.; Apovian, C.M.; Cacicedo, J.M.; Gokce, N.; Farb, M.; Valentine, R.J.; Ruderman, N.B. Insulin sensitive and resistant obesity in humans: AMPK activity, oxidative stress, and depot-specific changes in gene expression in adipose tissue. J Lipid Res 2012, 53, 792–801. [Google Scholar] [CrossRef]
- Sjostrom, L.; Gummesson, A.; Sjostrom, C.D.; Narbro, K.; Peltonen, M.; Wedel, H.; Bengtsson, C.; Bouchard, C.; Carlsson, B.; Dahlgren, S.; et al. Effects of bariatric surgery on cancer incidence in obese patients in Sweden (Swedish Obese Subjects Study): a prospective, controlled intervention trial. Lancet Oncol 2009, 10, 653–662. [Google Scholar] [CrossRef]
- Evans, J.M.; Donnelly, L.A.; Emslie-Smith, A.M.; Alessi, D.R.; Morris, A.D. Metformin and reduced risk of cancer in diabetic patients. BMJ 2005, 330, 1304–1305. [Google Scholar] [CrossRef] [PubMed]
- Soranna, D.; Scotti, L.; Zambon, A.; Bosetti, C.; Grassi, G.; Catapano, A.; La Vecchia, C.; Mancia, G.; Corrao, G. Cancer risk associated with use of metformin and sulfonylurea in type 2 diabetes: a meta-analysis. Oncologist 2012, 17, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Hawley, S.A.; Fullerton, M.D.; Ross, F.A.; Schertzer, J.D.; Chevtzoff, C.; Walker, K.J.; Peggie, M.W.; Zibrova, D.; Green, K.A.; Mustard, K.J.; et al. The ancient drug salicylate directly activates AMP-activated protein kinase. Science 2012, 336, 918–922. [Google Scholar] [CrossRef]
- Rothwell, P.M.; Fowkes, F.G.; Belch, J.F.; Ogawa, H.; Warlow, C.P.; Meade, T.W. Effect of daily aspirin on long-term risk of death due to cancer: analysis of individual patient data from randomised trials. Lancet 2011, 377, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Joharatnam-Hogan, N.; Cafferty, F.H.; Macnair, A.; Ring, A.; Langley, R.E. The role of aspirin in the prevention of ovarian, endometrial and cervical cancers. Womens Health (Lond) 2020, 16, 1745506520961710. [Google Scholar] [CrossRef]
- Neill, A.S.; Nagle, C.M.; Protani, M.M.; Obermair, A.; Spurdle, A.B.; Webb, P.M.; Australian National Endometrial Cancer Study; G. Aspirin, nonsteroidal anti-inflammatory drugs, paracetamol and risk of endometrial cancer: a case-control study, systematic review and meta-analysis. Int J Cancer 2013, 132, 1146–1155. [Google Scholar] [CrossRef]
- Verdoodt, F.; Friis, S.; Dehlendorff, C.; Albieri, V.; Kjaer, S.K. Non-steroidal anti-inflammatory drug use and risk of endometrial cancer: A systematic review and meta-analysis of observational studies. Gynecol Oncol 2016, 140, 352–358. [Google Scholar] [CrossRef]
- Zhang, D.; Bai, B.; Xi, Y.; Zhao, Y. Can Aspirin Reduce the Risk of Endometrial Cancer?: A Systematic Review and Meta-analysis of Observational Studies. Int J Gynecol Cancer 2016, 26, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Webb, P.M.; Na, R.; Weiderpass, E.; Adami, H.O.; Anderson, K.E.; Bertrand, K.A.; Botteri, E.; Brasky, T.M.; Brinton, L.A.; Chen, C.; et al. Use of aspirin, other nonsteroidal anti-inflammatory drugs and acetaminophen and risk of endometrial cancer: the Epidemiology of Endometrial Cancer Consortium. Ann Oncol 2019, 30, 310–316. [Google Scholar] [CrossRef]
- Movahedi, M.; Bishop, D.T.; Macrae, F.; Mecklin, J.P.; Moeslein, G.; Olschwang, S.; Eccles, D.; Evans, D.G.; Maher, E.R.; Bertario, L.; et al. Obesity, Aspirin, and Risk of Colorectal Cancer in Carriers of Hereditary Colorectal Cancer: A Prospective Investigation in the CAPP2 Study. J Clin Oncol 2015, 33, 3591–3597. [Google Scholar] [CrossRef]
- Bosetti, C.; Santucci, C.; Gallus, S.; Martinetti, M.; La Vecchia, C. Aspirin and the risk of colorectal and other digestive tract cancers: an updated meta-analysis through 2019. Ann Oncol 2020, 31, 558–568. [Google Scholar] [CrossRef]
- Sikavi, D.R.; Wang, K.; Ma, W.; Drew, D.A.; Ogino, S.; Giovannucci, E.L.; Cao, Y.; Song, M.; Nguyen, L.H.; Chan, A.T. Aspirin Use and Incidence of Colorectal Cancer According to Lifestyle Risk. JAMA Oncol 2024, 10, 1354–1361. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, G.R.; Carling, D. AMP-activated protein kinase: the current landscape for drug development. Nat Rev Drug Discov 2019, 18, 527–551. [Google Scholar] [CrossRef] [PubMed]
- Radman, M. Fidelity and infidelity. Nature 2001, 413, 115. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



