Submitted:
05 February 2026
Posted:
06 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction – the Metaverse and the Virtual Worlds of Science
2. Digital Twins as the Basis for Building A Virtual World
2.1. Changing the Concept of Modeling
2.2. Computational Platform
3. Fundamentals of A Virtual Free-Radical Polymerization Experiment
3.1. A General Concept of the Experiment
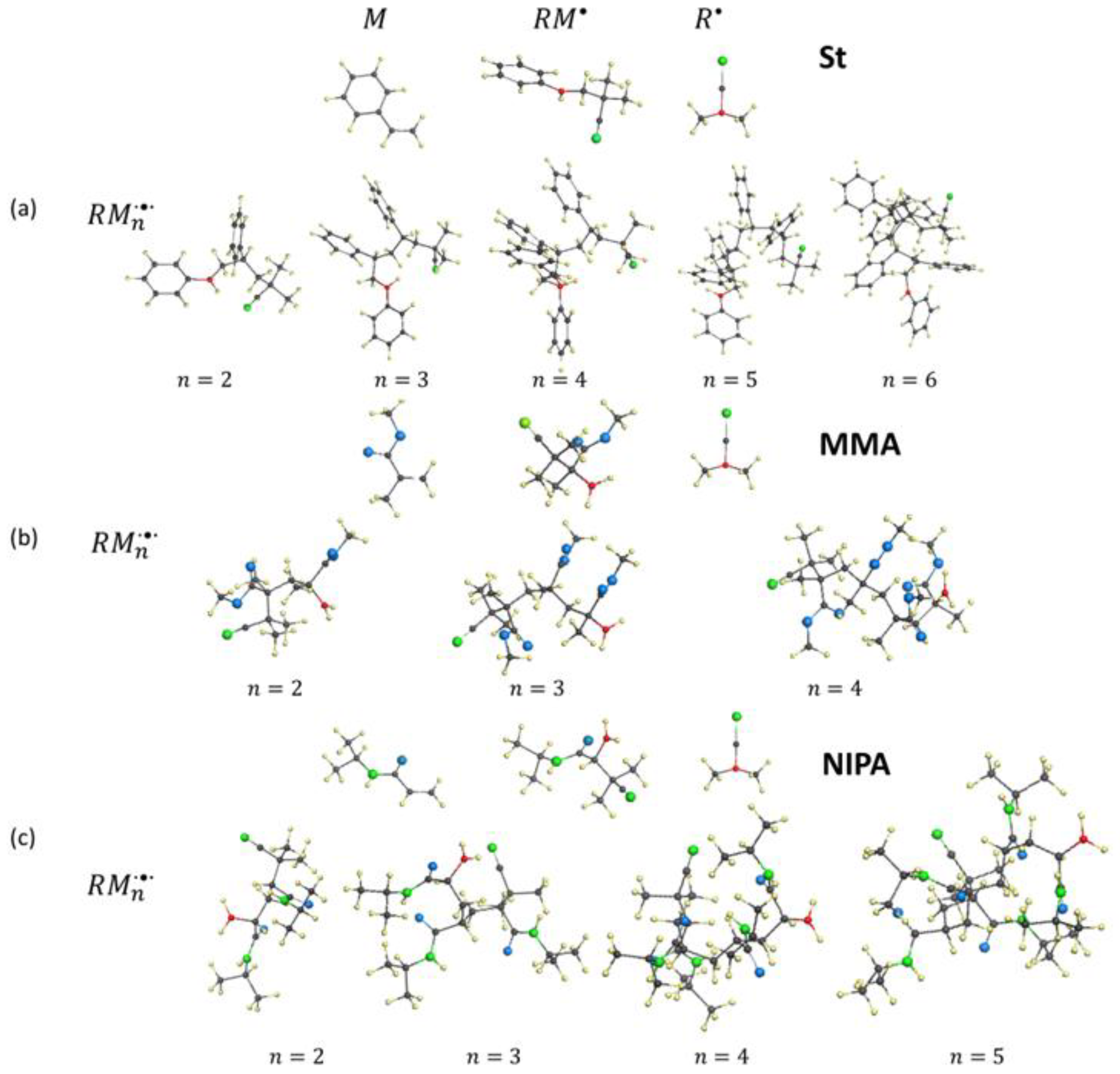
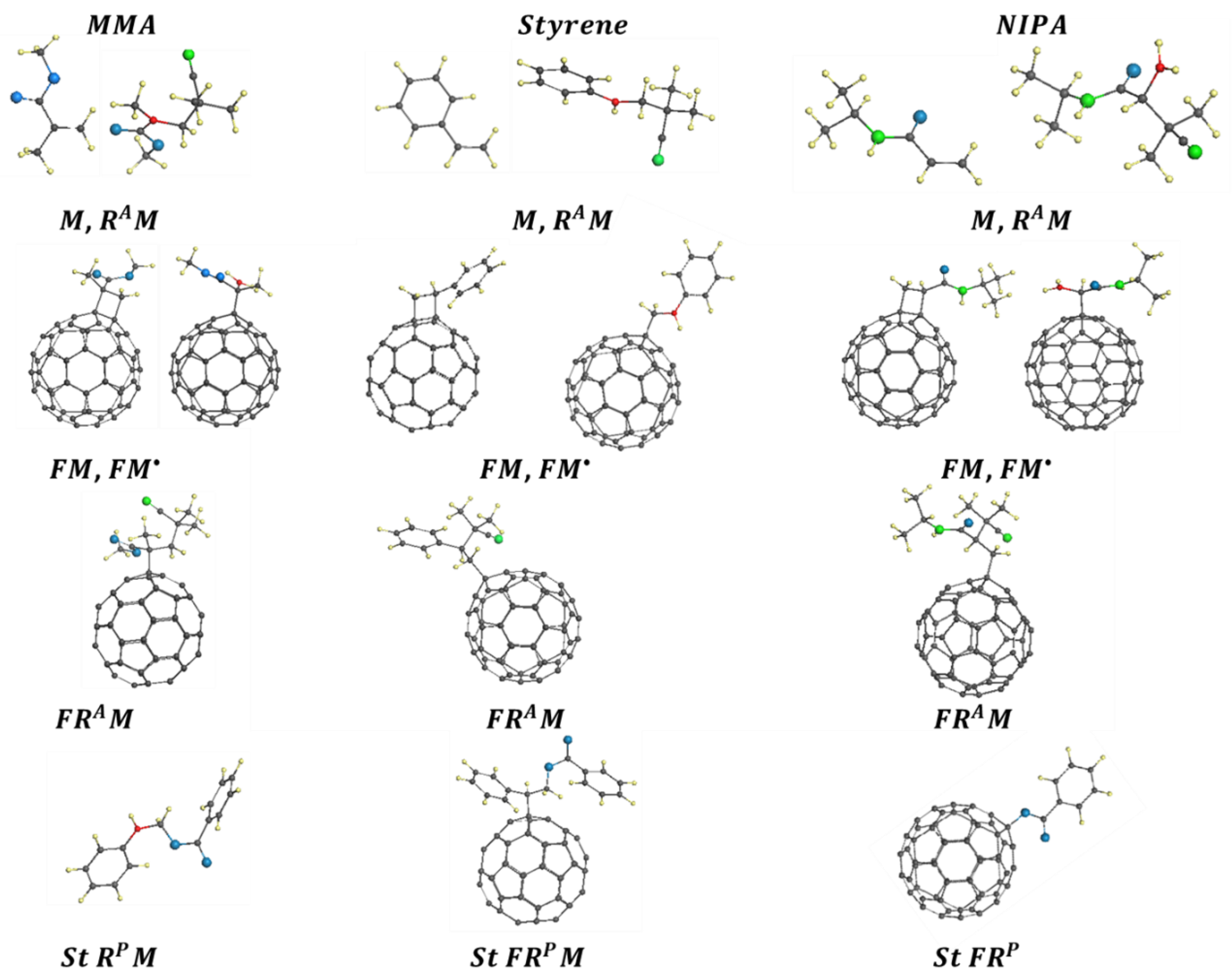
3.2. Dramatis Personae
3.3. Elementary Reactions
- Table 1. Nomination of elementary reactions and/or digital twins related to the initial stage of free radical copolymerization of vinyl monomers with stable radicals.
3.4. Virtual Device
3.5. Descriptors of Virtual Polymerization
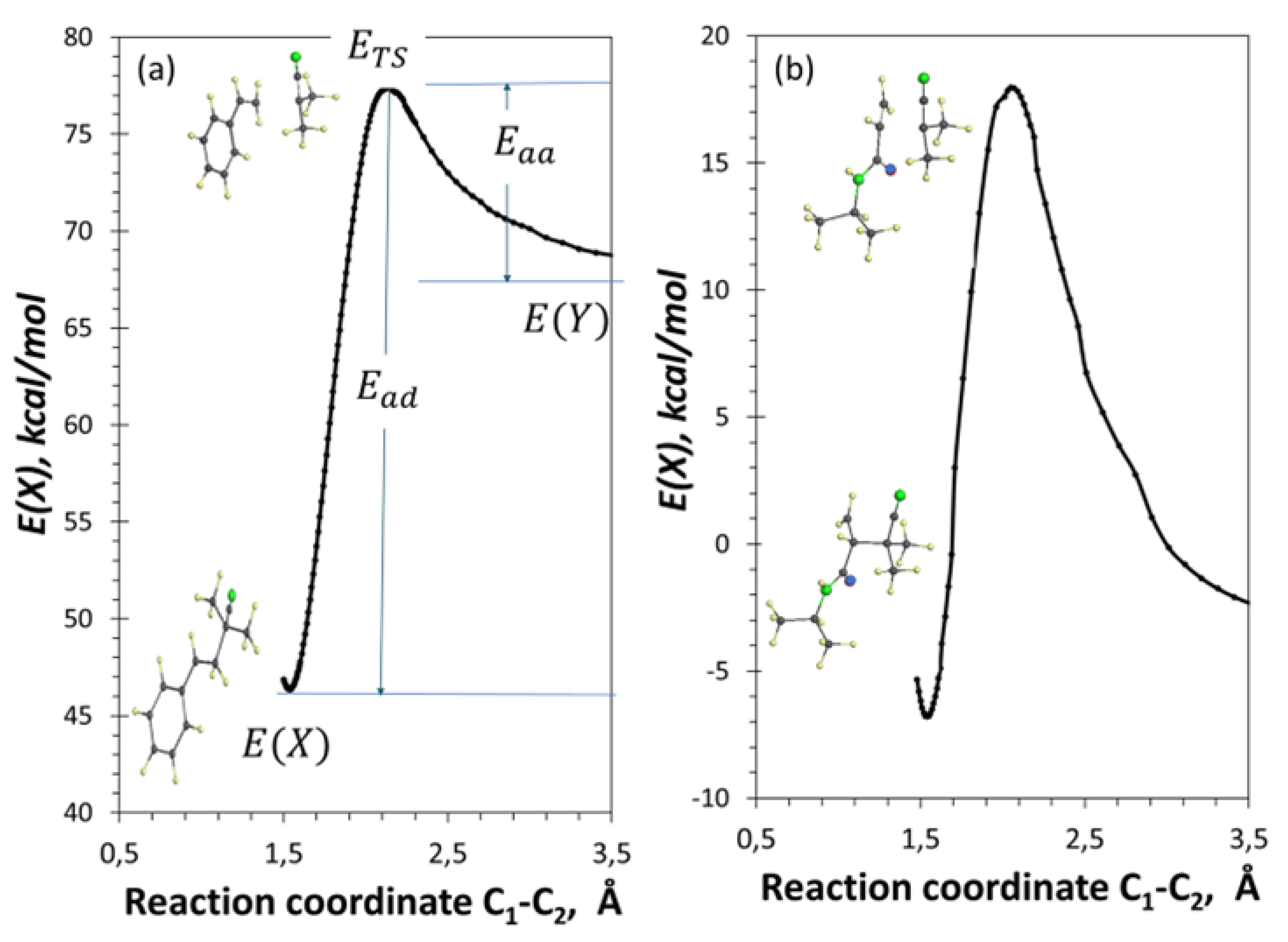
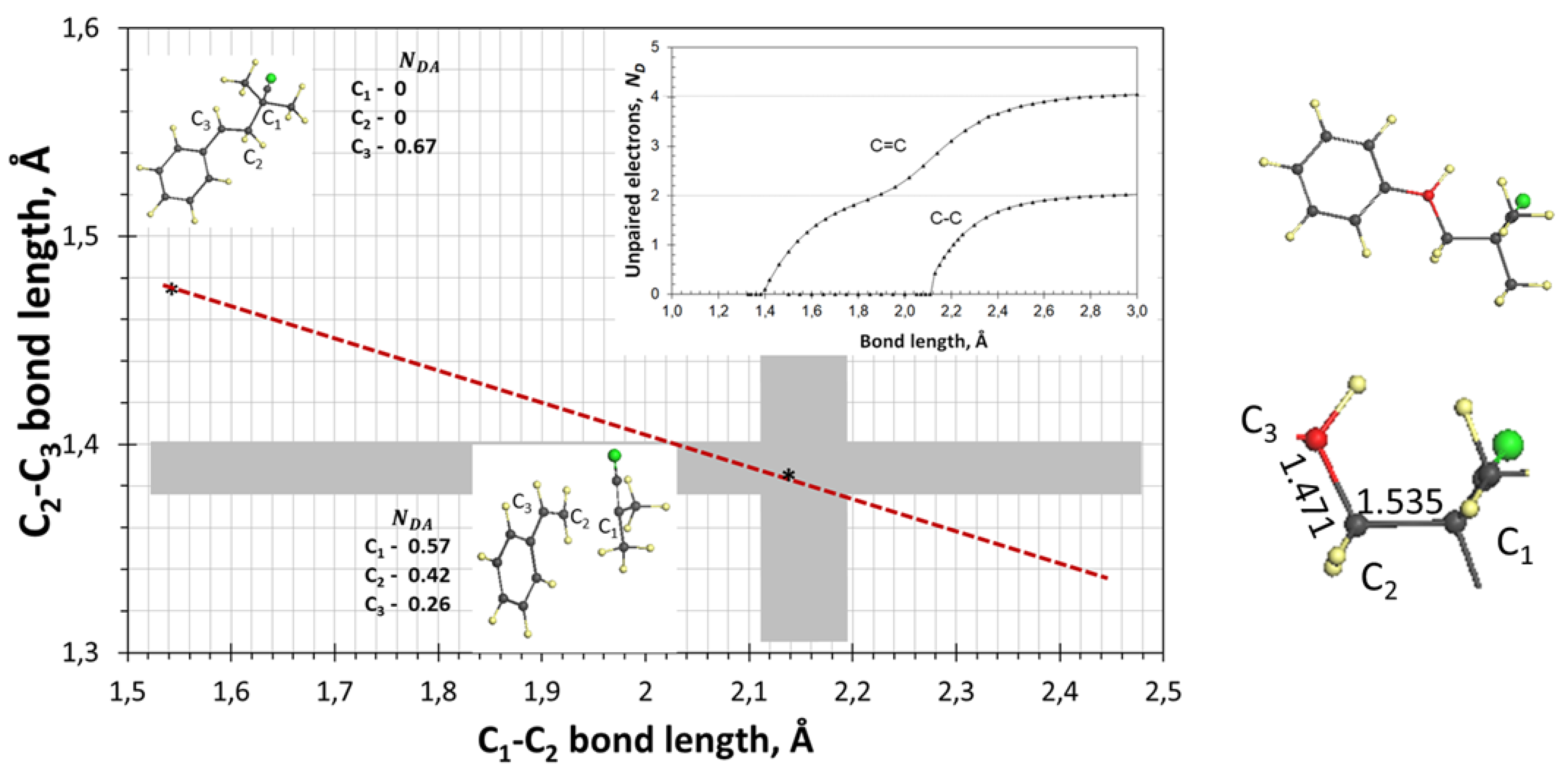
3.6. Transition State
3.7. Digital Polymerization Passports
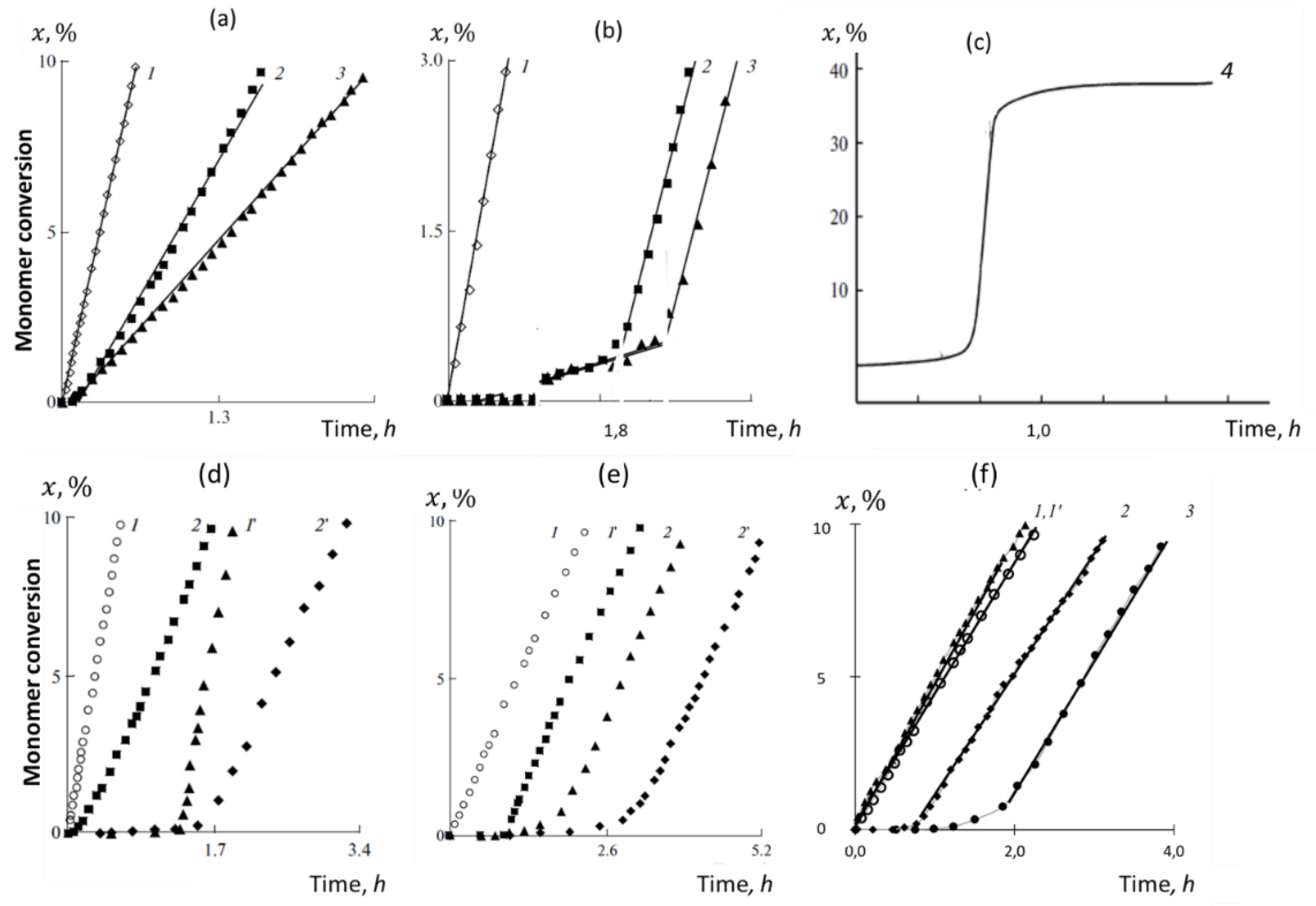
4. Virtual Free-Radical Polymerization of Vinyl Monomers
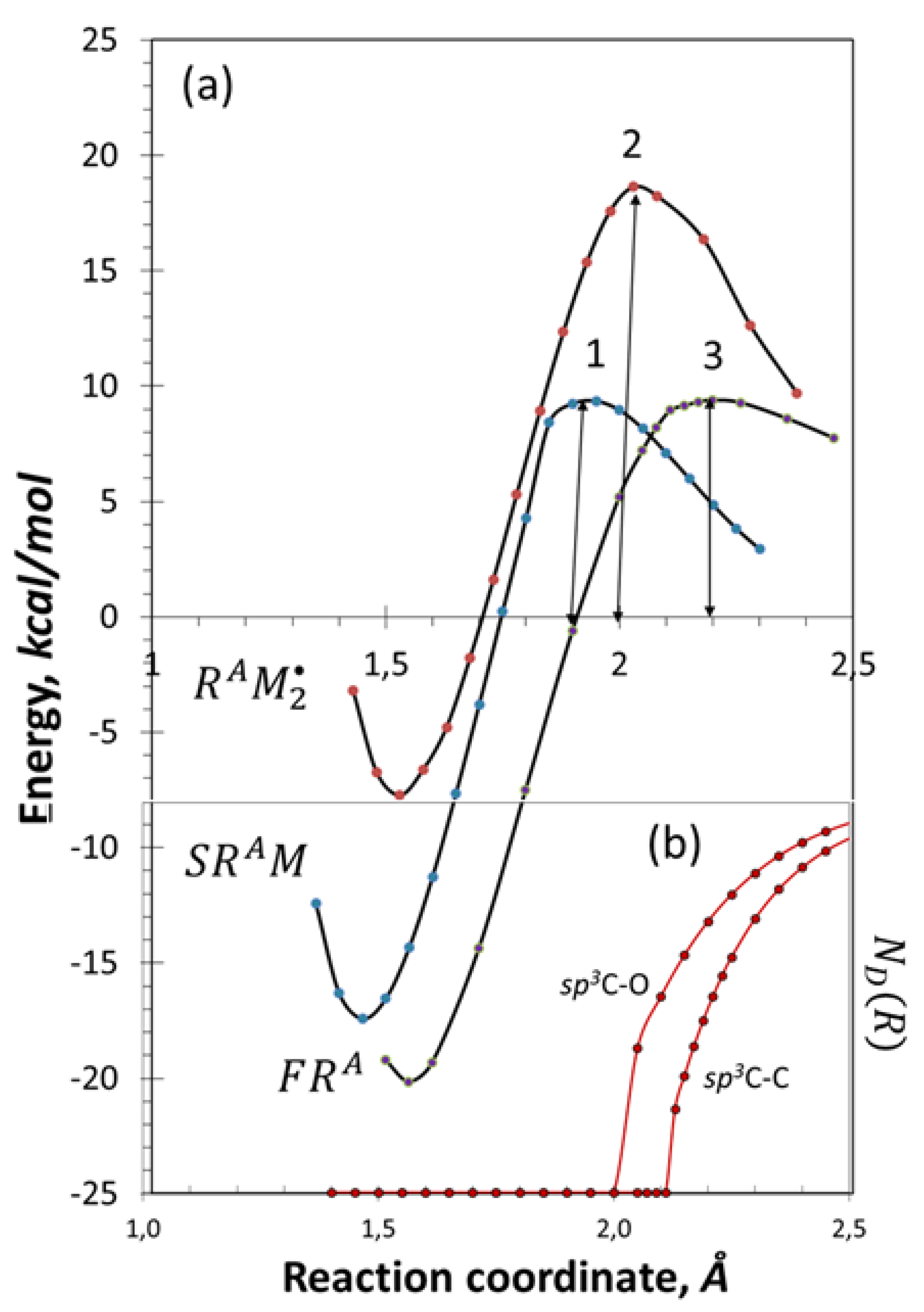
4.1. Initiation of Free-Radical Polymerization and Chain Propagation
4.2. Chain Break and Polymerization Termination
5. Virtual Free-Radical Copolymerization of Vinyl Monomers with Stable Radicals
5.1. Brief Review of Empirical Observations
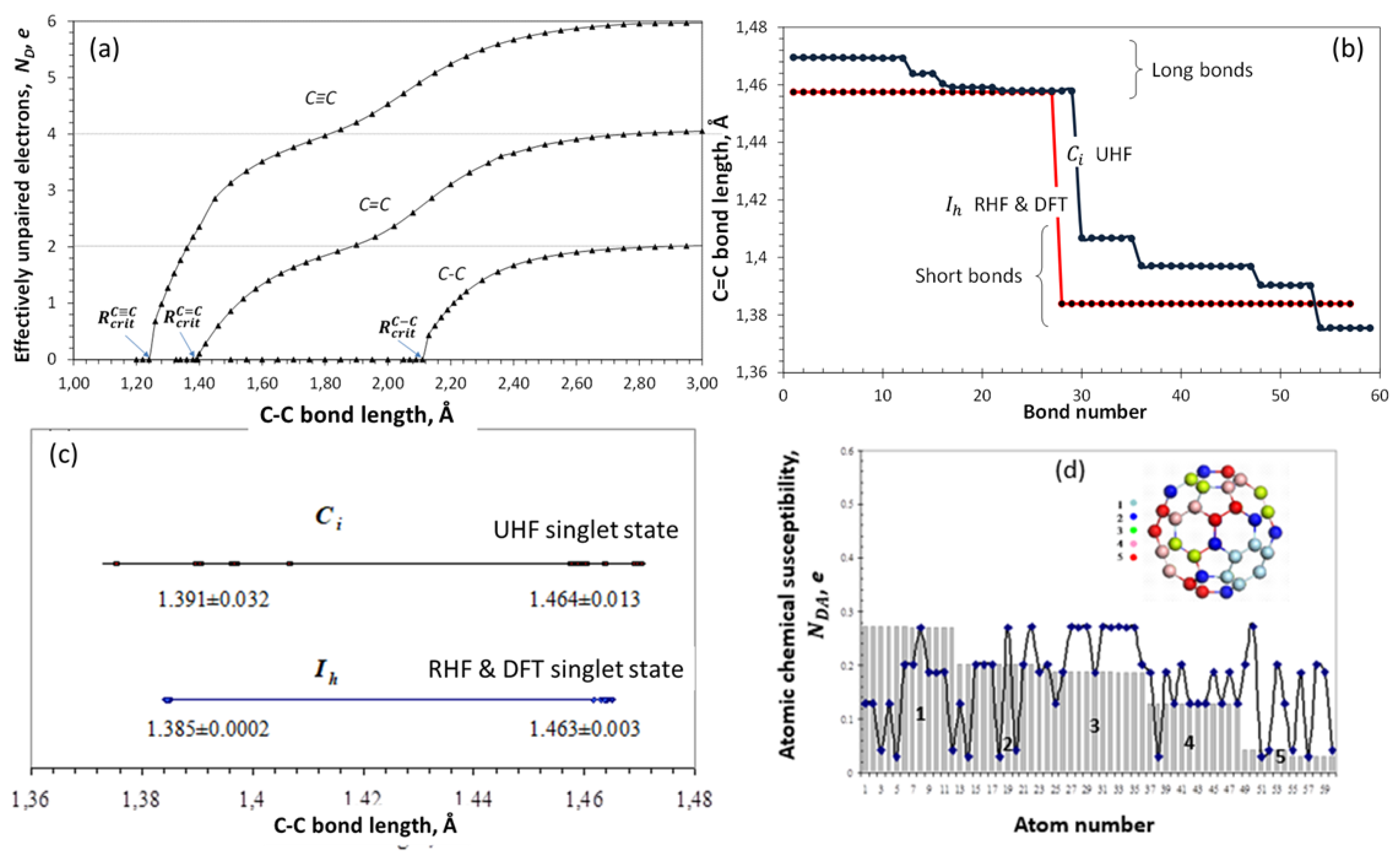
5.2. A Brief Excursion Into the Spin Theory of Fullerenes
5.2.1. the Origins of the Radicality of Fullerene C60
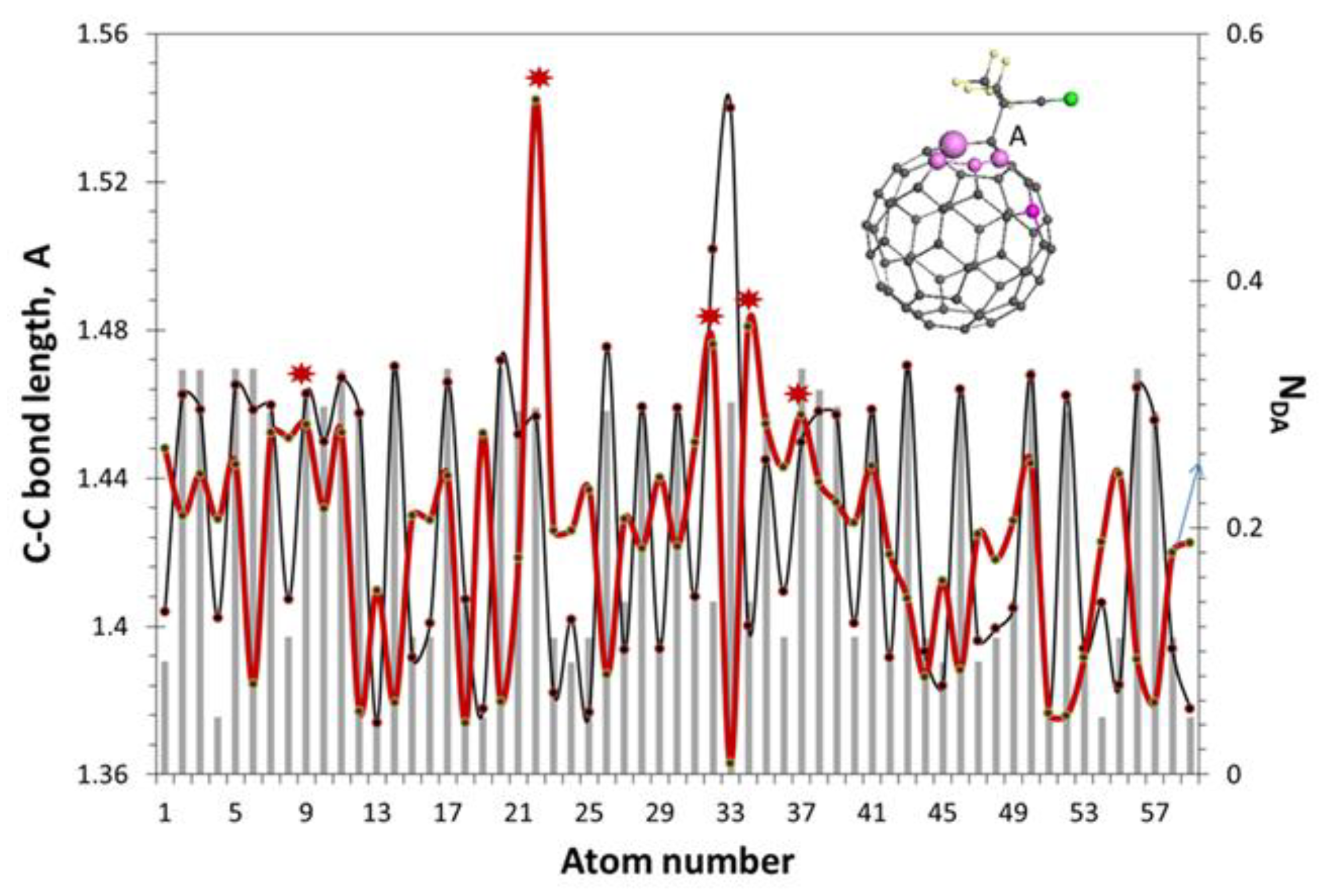
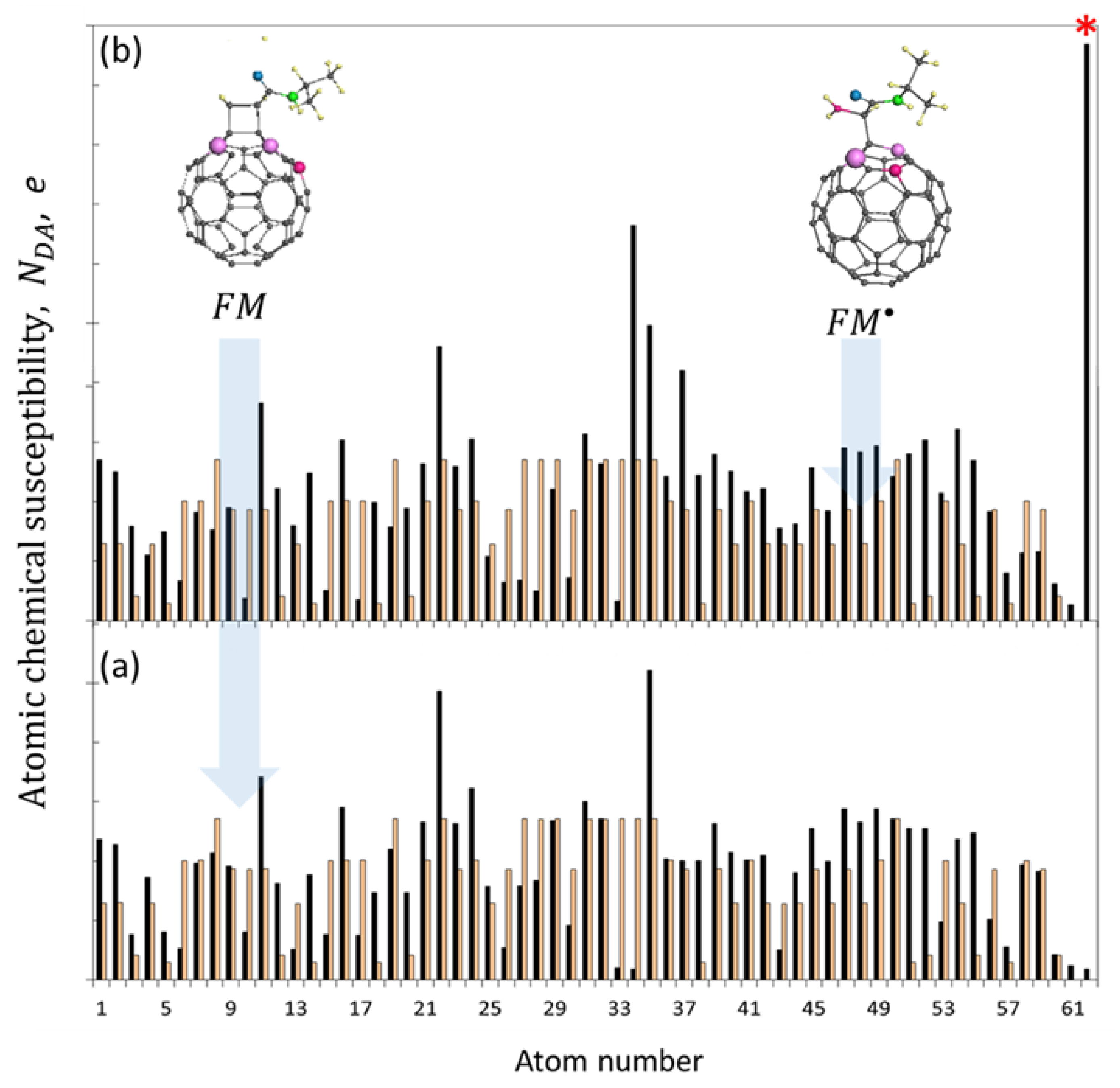
5.2.2. Spin Density Algorithm for A Virtual Addition Reaction
5.3. Chain Reaction of Polymerization in View of Spin Theory
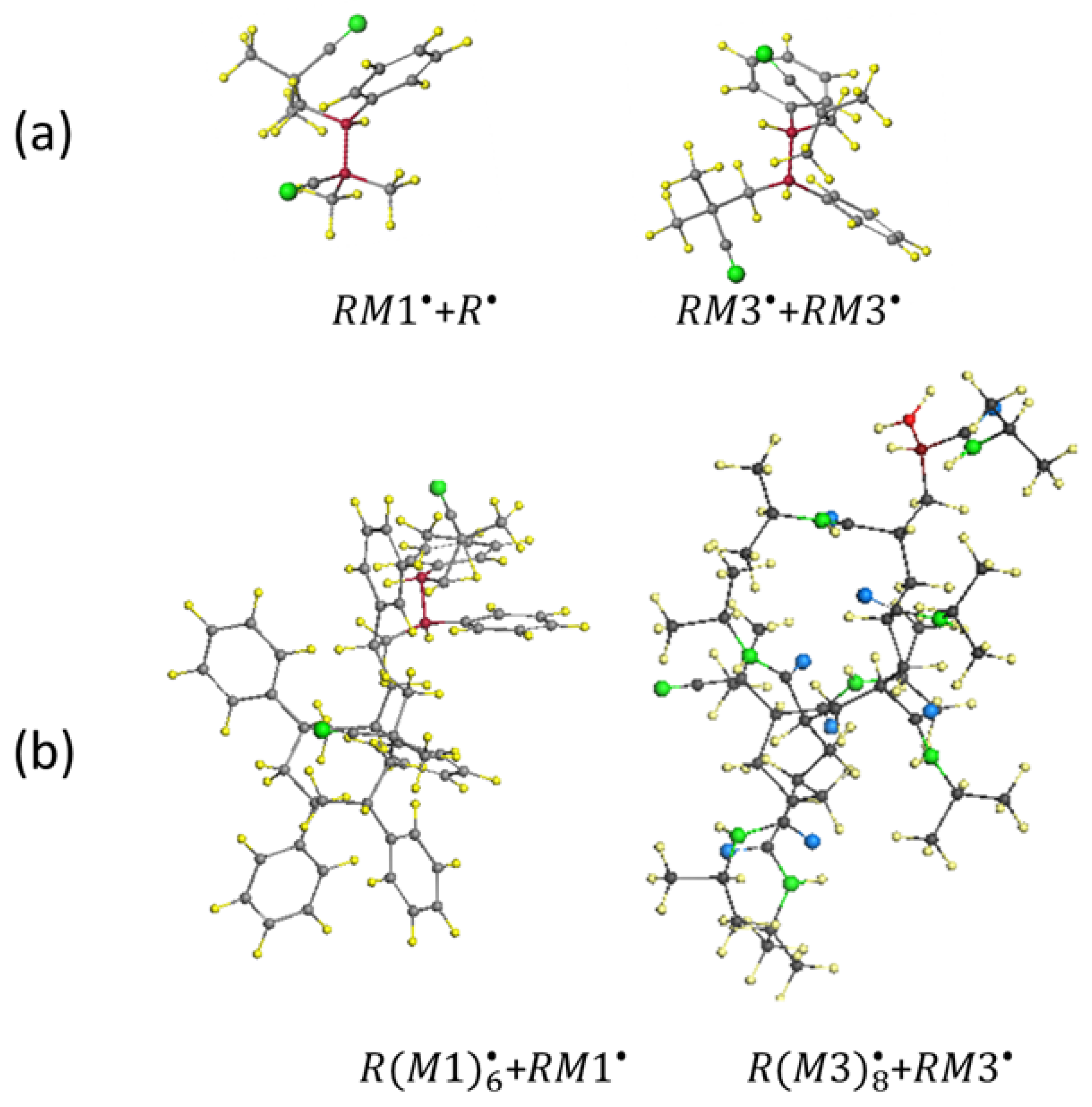
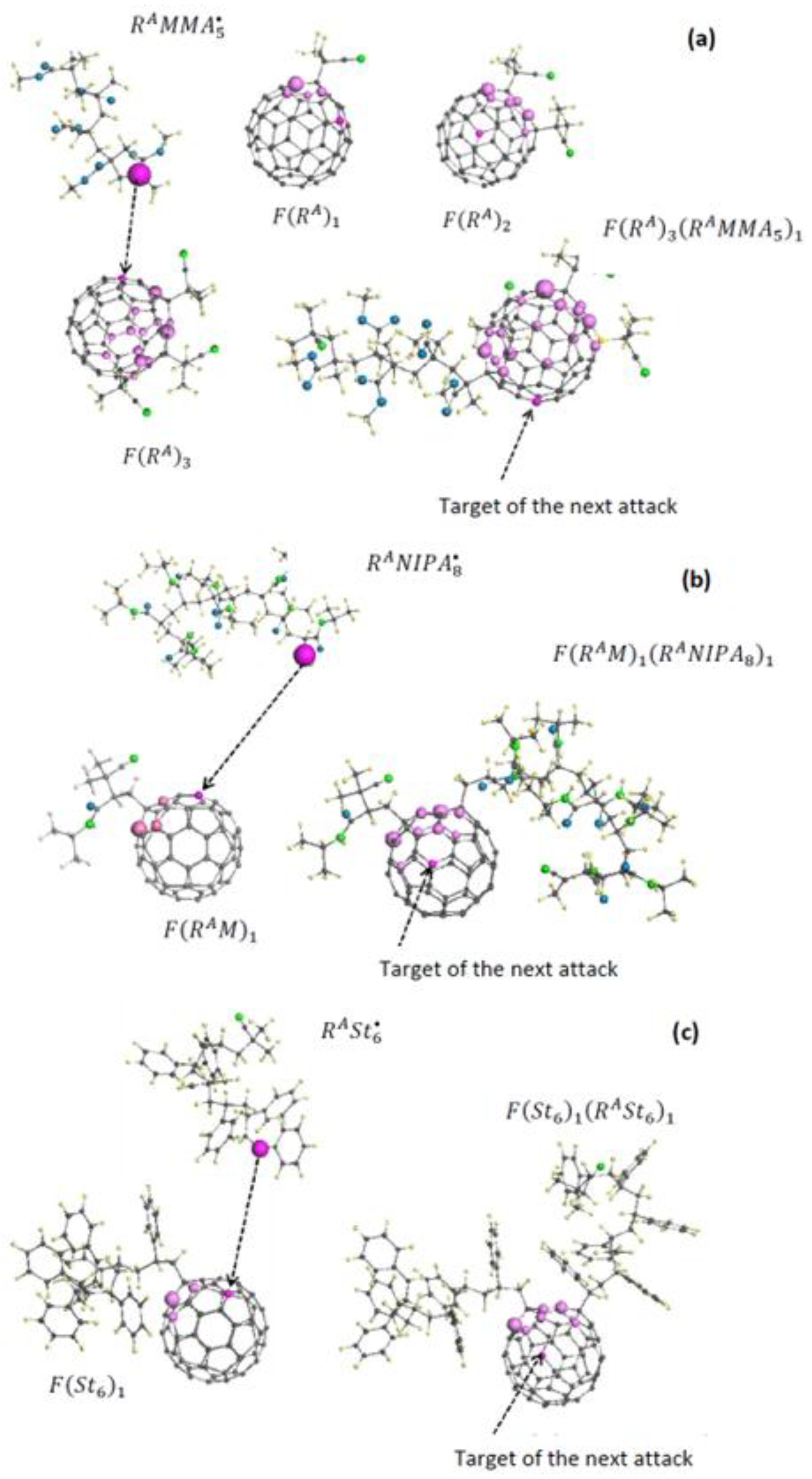
5.4. Virtual Free-Radical Copolymerization of Vinyl Monomers with C60 Fullerene
- 4. the Fullerene-Associated Reaction Sequence in Vrsa Forms A Series
5.4. Virtual Star-Branched Polymers of C60 Fullerene
6. Conclusive Remarks
Acknowledgments
References
- Shabason VE, Malaykin SN. Razumnaya metavselennaya. Ot tsifrovykh prilozheniy k novoy srede obitaniya. [Reasonable metaverse. From digital applications to a new environment]. Moscow, LitRes Publ., Juli 19 2022.
- Ball M. The Metaverse: And How It Will Revolutionize Everything. Liveright: New York, July 25, 2022.
- Li, H., Li, B. The state of metaverse research: a bibliometric visual analysis based on CiteSpace. J Big Data 11, 14 (2024). [CrossRef]
- Sheka EF. Metaverse and virtual worlds of science. Trends Comput Sci Inf Technol. 2024. 9, 103-105. [CrossRef]
- M. Liu, S. Fang, H. Dong, C. Xu. Review of digital twin about concepts, technologies, and industrial applications. J. Manufact. Syst. 58 (2021) 346–361. [CrossRef]
- Handbook of Digital Twins. Ed. Lv, Zh, CRC Press, Taylor and Francis, Boca Raton, 2024. [CrossRef]
- S. Waffenschmidt. Digital twin in the Industry 4.0: interview with a pioneer. t-systems.com›de/en/newsroom/best-practice/ issue 03, 2018.
- D. H. Gelernter (1991). Mirror Worlds: or the Day Software Puts the Universe in a Shoebox—How It Will Happen and What It Will Mean. Oxford; New York: Oxford University Press. ISBN 978-0195079067. OCLC 23868481.
- R. Piascik, et al., Technology Area 12: Materials, Structures, Mechanical Systems, and Manufacturing Road Map. 2010, NASA Office of Chief Technologist.
- Tao, F.; Cheng, J.; Qi, Q.; Zhang, M.; Zhang, H.; Sui, F. Digital Twin-Driven Product Design, Manufacturing and Service with Big Data. The International Journal of Advanced Manufacturing Technology 2018, 94 (9–12), 3563–3576. [CrossRef]
- Rasheed, A.; San, O.; Kvamsdal, T. Digital twins: Values, challenges and enablers from a modeling perspective. IEEE Access 2020. [CrossRef]
- Lazzari, S.; Lischewski, A.; Orlov, Y.; Deglmann, P.; Daiss, A.; Schreiner, E.; Vale, H. Toward a digital polymer reaction engineering. Adv. Chem. Eng. 2020, 56, 187-230.
- Zobel-Roos, S.; Schmidt, A.; Uhlenbrock, L.; Ditz, R.; Köster, D.; Strube, J. Digital Twins in Biomanufacturing; 2020; pp 181–262. [CrossRef]
- Gargalo, C. L.; de las Heras, S. C.; Jones, M. N.; Udugama, I.; Mansouri, S. S.; Krühne, U.; Gernaey, K. V. Towards the Development of Digital Twins for the Bio-Manufacturing Industry; 2020; pp 1–34. [CrossRef]
- Sinner, P.; Daume, S.; Herwig, C.; Kager, J. Usage of Digital Twins Along a Typical Process Development Cycle; 2020; pp 71–96. [CrossRef]
- Stanko, M.; Stommel, M. Digital Twin of the Polyurethane Rotational Moulding Process. In Advances in Polymer Processing 2020; Springer Berlin Heidelberg: Berlin, Heidelberg, 2020; pp 324–335. [CrossRef]
- Scheper, T.; Beutel, S.; McGuinness, N.; Heiden, S.; Oldiges, M.; Lammers, F.; Reardon, K. F. Digitalization and Bioprocessing: Promises and Challenges; 2020; pp 57–69. [CrossRef]
- Hürkamp, A.; Lorenz, R.; Ossowski, T.; Behrens, B.-A.; Dröder, K. Simulation-Based Digital Twin for the Manufacturing of Thermoplastic Composites. Procedia CIRP 2021, 100, 1–6. [CrossRef]
- Andronas, D.; Kokotinis, G.; Makris, S. On Modelling and Handling of Flexible Materials: A Review on Digital Twins and Planning Systems. Procedia CIRP 2021, 97, 447–452. [CrossRef]
- Qi, Q.; Tao, F.; Hu, T.; Anwer, N.; Liu, A.; Wei, Y.; Wang, L.; Nee, A. Y. C. Enabling Technologies and Tools for Digital Twin. J Manuf Syst 2021, 58, 3–21. [CrossRef]
- Lo, C. K.; Chen, C. H.; Zhong, R. Y. A Review of Digital Twin in Product Design and Development. Advanced Engineering Informatics 2021, 48, 101297. [CrossRef]
- Lebedev, I. V.; Ivanov, S. I.; Safarov, R. R.; Men’shutina, N. V. Digital Twins for the Porous Structures of Aerogels with the Use of the Cellular Automation Approach and Bezier Curves. Theoretical Foundations of Chemical Engineering 2023, 57 (4), 482– 488. [CrossRef]
- Wang, Y.; Tao, F.; Zuo, Y.; Zhang, M.; Qi, Q. Digital-Twin-Enhanced Quality Prediction for the Composite Materials. Engineering 2023, 22, 23–33. [CrossRef]
- Fernández-León, J.; Keramati, K.; Baumela, L.; González, C. A Digital Twin for Smart Manufacturing of Structural Composites by Liquid Moulding. The International Journal of Advanced Manufacturing Technology 2024, 130 (9–10), 4679–4697. [CrossRef]
- Isoko, K.; Cordiner, J. L.; Kis, Z.; Moghadam, P. Z. Bioprocessing 4.0: A Pragmatic Review and Future Perspectives. Digital Discovery 2024, 3 (9), 1662–1681. [CrossRef]
- Alajmi, A. Twinning the Future: Implementing Digital Twin Technology in the Optimisation of Fibre-Reinforced Polymers. MATEC Web of Conferences 2024, 401, 11005. [CrossRef]
- Jaboviste, K.; Sadoulet-Reboul, E.; Teloli, R. O.; Chevallier, G. Characterizing the Nonlinear Behavior of Viscoelastic Materials: A Bayesian Approach Combining Oberst Beam Experiments and Digital-Twin Simulations. Mech Syst Signal Process 2024, 208, 110978. [CrossRef]
- CEN and CENELEC. CEN and CENELEC Workshop Agreement: Methodology for the Data-Driven Management of Production; 50751:2024; 2024. https://www.cencenelec.eu/media/CEN-CENELEC/CWAs/RI/2024/cwa50751_2024.pdf (accessed 2024-12-19).
- Mitchell, A.; Wei, X,; Sun, R.; Yamamura, K.; Ye, L.; Corney, J.; Yu, N. Digital manufacturing for advanced manufacturing technologies: A review of successful implementation for plasma processing digital twin framework development. Available on 26 September 2024 at SSRN: https://ssrn.com/abstract=4846321 or. [CrossRef]
- E.F. Sheka, V.A. Popova. Virtual vibrational spectrometer for sp2 carbon clusters. 2. Fullerene C60 and its isomers. FNCN, 2021, 29, No. 12, 974-981. [CrossRef]
- E.F. Sheka and N. A. Popova. Virtual vibrational spectrometer for sp2 carbon clusters and dimers of fullerene C60. FNCN, 2022, 30, 777-786. [CrossRef]
- E.F. Sheka. Virtual vibrational spectrometry of stable radicals—necklaced graphene molecules. Nanomaterials 2022, 12(4), 597. [CrossRef]
- E.F.Sheka, N.A. Popova. Virtual vibrational analytics of reduced graphene oxide. Int. Journ. Mol. Sci. 2022, 23, 6978. [CrossRef]
- E.F.Sheka. Digital Twins solve the mystery of Raman spectra of parental and reduced graphene oxides. Nanomaterials, 2022, 12, 4209. [CrossRef]
- Е.Ф. Шека. Цифрoвые двoйники в технoлoгии графена. РЭНСИТ: Радиoэлектрoника. Нанoсистемы. Инфoрмациoнные технoлoгии, 2023, 15(1):59-80. [CrossRef]
- E.F. Sheka. Digital Twins in graphene technology. Chapter 47 of Handbook of Digital Twins, Ed. Lv, Zh, CRC Press, Taylor and Francis, Boca Raton, 2024, 19 pages. [CrossRef]
- E.F. Sheka. Virtual free-radical polymerization of vinyl monomers in view of digital twins. Polymers, 2023, 15, 2999. Published. [CrossRef]
- E.F. Sheka. Digitalization of free-radical polymerization. arXiv:2312.14163 [physics.chem-ph], 2023. [CrossRef]
- E.F. Sheka. The triumph of the spin chemistry of fullerene C60 in the light of its free radical copolymerization with vinyl monomers. Int. Journ. Mol. Sci. 2024, 25, 1317. [CrossRef]
- E.F. Sheka. Digital twins’ kinetics of virtual free-radical copolymerization of vinyl monomers with stable radicals. 1. Methyl methacrylate. FNCN, 2024. [CrossRef]
- E.F. Sheka. Digital twins’ kinetics of virtual free-radical copolymerization of vinyl monomers with stable radicals. 2. Styrol. arXiv:2311.02752 [cond-mat.mtrl-sci]. [CrossRef]
- E.F. Sheka. Digital twins’ kinetics of virtual free-radical copolymerization of vinyl monomers with stable radicals. 3. NIPA. arXiv:2311.06866 [cond-mat.mtrl-sci]. [CrossRef]
- Ladik, J., André, JM., Seel, M. (eds) Quantum Chemistry of Polymers — Solid State Aspects. NATO ASI Series, vol 123. Springer, Dordrecht, 1984.
- J. Ladik, Quantum Theory of Polymers as Solids. Plenum Press, New York, 1988.
- C. Tosy (ed). Proceedings of the First Donegani Scientific Workshop on Strategies for Computer Chemistry. October 12–13, 1987. Kluwer Academic Publishing: New York, Boston, Dordrecht, London, Moscow, 1989.
- Quantum Chemistry Aided Design of Organic Polymers: An Introduction to the Quantum Chemistry of Polymers and its Applications. Eds. J.-M. Andre, J. Delhalle, J.-L. Bredas, World Sci. Publ.: Singapoure, 1991.
- Computational Modeling of Polymers. Ed. J. Bicerano. CRC Press: Boca Raton, 1992.
- H. Fischer, L. Radom. Factors controlling the addition of carbon-centered radicals to alkenes. An experimental and theoretical perspective. Angew. Chem. Int. Ed. 2001, 40, 1340 – 1371.
- Heuts, J.P.A. Theory of radical reactions. In Handbook of Radical Polymerization, Eds. K. Matyjaszewski, T.P. Davis. John Wiley and Sons: Hoboken, 2002, pp. 1-76.
- Theory and Applications of Computational Chemistry: The First Forty Years. Eds. C. Dykstra, G. Frenking, K. Kim, G. Scuseria. Elsevier B.V.: Amsterdam, 2005.
- J.-M. Andre. Computational quantum chemistry on polymer chains: aspects of the last half century. In Theory and Applications of Computational Chemistry: The First Forty Years. Eds. C. Dykstra, G. Frenking, K. Kim, G. Scuseria. Elsevier B.V.: Amsterdam, 2005, pp. 1011-1045.
- M. L. Coote. Quantum-chemical modeling of free-radical polymerization. Macromol. Theory Simul. 2009, 18, 388–400.
- D. Gigmes, D. Bertin, C. Lefay, Y. Guillaneuf. Kinetic modeling of nitroxide-mediated polymerization: Conditions for living and controlled polymerization. Macromol. Theory Simul. 2009, 18, 402–419. [CrossRef]
- P. Deglmann, I. Mueller, F. Becker, A. Schaefer, K.-D. Hungenberg, H. Weiß. Prediction of propagation rate coefficients in free radical solution polymerization based on accurate quantum chemical methods: Vinylic and related monomers, including acrylates and acrylic acid. Macromol. React. Eng. 2009, 3, 496–515. [CrossRef]
- M. Dossi, G. Storti, D. Moscatelli. Initiation kinetics in free-radical polymerization: Prediction of thermodynamic and kinetic parameters based on ab initio calculations. Macromol. Theory Simul. 2010, 19, 170–178. [CrossRef]
- E. Mavroudakis, D. Cuccato, D. Moscatelli. On the use of quantum chemistry for the determination of propagation, copolymerization, and secondary reaction kinetics in free radical polymerization. Polymers 2015, 7, 1789-1819. [CrossRef]
- F. Ruipérez. Application of quantum chemical methods in polymer chemistry. Int. Rev. Phys. Chem. 2019, 38, 343-403. [CrossRef]
- Y. Hayashi, S. Kawauchi. Development of a quantum chemical descriptor expressing aromatic/quinoidal character for designing narrow-bandgap π-conjugated polymers. Polym. Chem., 2019, 10, 5584–5593. [CrossRef]
- J. Gay. Molecular Dynamics Studies of Polymer Systems. Department of Chemical Engineering at Worcester Polytechnic Institute: Worcester, 2012.
- T. E. Gartne, A. Jayaraman, Modeling and simulations of polymers: A roadmap. Macromol. 2019, 52, 755−786.
- C. Micheletti, P. Hauke, P. Faccioli. Polymer physics by quantum computing, Phys.Rev. Letts 2021, 127, 080501. [CrossRef]
- H.W. Starkweather, H.W.; Taylor, G.B. The kinetics of the polymerization of vinyl acetate. JACS 1930, 52, 4708-4714.
- Semenov, N.N. Tsepnyie Reakcii (Chain Reactions) Goschimizdat: Moskva, 1934 (in Russian).
- А.Bagdasar’yan, Kh.S., Teoriya radikal’noi polimerizatsii (Free_Radical Polymerization Theory), Moscow: Nauka, 1966. (in Russian).
- Gol’dfein, M.D.; Kozhevnikov, N.V.; Trubnikov, A.V. Kinetika i mekhanizm regulirovaniya protsessov obrazovaniya polimerov (Kinetics and Control of Polymerization Processes), Saratov: Saratov. Gos. Univ., 1989.
- Pross, A. Theoretical and Physical Principles of Organic Reactivity, Wiley, New York, 1995.
- Handbook of Radical Polymerization, Eds. K. Matyjaszewski, T. P. Davis. John Wiley & Sons, Inc. 2002.
- Handbook of Vinyl Polymers 2: Radical Polymerization, Process, and Technology, New Edition. Eds. M. Mishra, Y. Yagci. CRC Press: Boca Raton, 2019.
- Loy, D.A.; Assink, R.A. Synthesis of a C60-p-xylylene copolymer. J. Am. Chem. Soc. 1992, 114, 3977 – 3978.
- Bunker, C.E.; Lawson, G.E.; Sun, Y.P. Fullerene-styrene random copolymers. novel optical properties. Macromolecules 1995, 28, 3744 – 3746. [CrossRef]
- Cao, T.; Webber, S.E. Free-radical copolymerization of fullerenes with styrene. Macromolecules 1995, 28, 3741 – 3743. [CrossRef]
- Cao, T.; Webber, S.E. Free radical copolymerization of styrene and C60. Macromolecules 1996, 29, 3826 – 3830. [CrossRef]
- Camp, A. G.; Lary, A.; Ford, W. T. Free-radical copolymerization of methyl methacrylate and styrene with C60. Macromolecules 1995. 28, 7959–7961. [CrossRef]
- Sun, Y.P.; Lawson, G.E.; Bunker, C.E.; Johnson, R.A.; Ma, B.; Farmer, C.; Riggs, J.E.; Kitaygorodskiy, A. Preparation and characterization of fullerene−styrene copolymers. Macromolecules 1996, 29, 8441 – 8448. [CrossRef]
- Cao, T.; Webber, S.E. Radical copolymerization of styrene and C60. Macromolecules 1995, 28, 3741–3743.
- Geckeler, KE; Arsalani, N. Synthesis and properties of hydrophilic polymers. 4. Preparation and characterization of poly (oxyethylene) telechelics with different aromatic termini. J. Macromol. Sci., Part A: Pure and Appl. Chem. 1996, 33, 1165-1179. [CrossRef]
- Steward, D.; Imrie, C. T. Role of C60 in the free radical polymerisation of styrene. Chem. Commun. 1996, 13, 1383-1384.
- Chen, Y.; Lin, K.C. Radical polymerization of styrene in the presence of C60. J. Polym. Sci. A, Polym. Chem. 1999, 37, 2969 – 2975.
- Ford, W. T.; Graham, T. D.; Mourey, T. H. Incorporation of C60 into poly(methyl methacrylate) and polystyrene by radical chain polymerization produces branched structures. Macromolecules 1997, 30, 6422–6429. [CrossRef]
- Ford, W. T.; Nishioka, T.; McCleskey, S. C.; Mourey, T. H.; Kahol, P. Structure and radical mechanism of formation of copolymers of C60 with styrene and with methyl methacrylate. Macromolecules, 2000, 33, 2413–2423. [CrossRef]
- Nayak, P.L.; Yang, K.; Dhal, P.K.; Alva, S.; Kumar, J.; Tripathy, S.K. Polyelec-trolyte-containing fullerene I: synthesis and characterization of the copolymers of 4-vinylbenzoic acid with C60. Chem. Mater. 1998, 10, 2058 – 2066. [CrossRef]
- Jiang, G.; Zheng, Q. Synthesis and application of new fullerene derivative. J. Appl. Polym. Sci. 2005, 97, 2182 – 2185. [CrossRef]
- Kurmaz, S.V.; Pyryaev, A.N.; Obraztsova, N.A. Effect of fullerene on the radical homo_ and copolymerization of n_vinylpyrrolidone and (di)methacrylates. Polym. Sci., Ser. B, 2011, 53, 497–504. [CrossRef]
- Atovmyan, E.G.; Grishchuk, A.A.; Estrina, G.A.; Estrin, Ya.I. Formation of star-like water-soluble polymeric structures in the process of radical polymerization of N-isopropyl acrylamide in the presence of C60. Russ. Chem. Bull., Int. Ed. 2016, 65, 2082—2088. [CrossRef]
- Atovmyan, E. G. On the relationship between the fullerene reactivity and degree of substitution. Russ. Chem. Bull., Int. Ed. 2017, 66, 567-570. [CrossRef]
- Yumagulova, R. Kh.; Kuznetsov, S. I.; Diniakhmetova, D. R.; Frizen, A. K.; Kraikin, V. A.; Kolesov. S. V. On the initial stage of the free-radical polymerizations of styrene and methyl methacryate in the presense of fullerene C60. Kinetics and Catalysis 2016, 57, 380–387.
- Yumagulova, R. Kh.; Kolesov, S.V. Specific features of reactions between fullerene C60 and radicals stabilized by conjugation in the process of radical polymerization. Bullet, Bashkir University 2020, 25. 47-51. [CrossRef]
- Martin, N.; Giacalone, F. (Eds.) Fullerene Polymers. Synthesis, Properties and Applications. Wiley-VCH: Weinheim, 2009.
- Denisov, E. T. Constanty skorosni gomoliticheskikh zhidkofaznykh reakciy (Rate constants of homolytic liquid-phase reactions) Moskwa: Nauka. 1971.
- Denisov, E. T.; Sarkisov, O. M.; Likhtenshtein, G. I. Chemical Kinetics: Fundamentals and Recent Developments, Elsevier, Amsterdam, 2003.
- E.T. Denisov, I.B. Afanas’ev. Oxidation and Antioxidants in Organic Chemistry and Biology. Boca Raton, Florida: CRC Press. Taylor and Francis Group, 2005.
- H. Zipse. Radical stability—a theoretical perspective. Top Curr. Chem. 2006, 263: 163–189. [CrossRef]
- A.I. Krylov. The quantum chemistry of open-shell species. In Reviews in Computational Chemistry. Eds, A. L. Parrill, K. B. Lipkowitz. Vol. 30, First Edition. John Wiley & Sons, Inc.: Hoboken, 2017, pp. 151-224.
- Y. Huang, E. Egap. Open-shell organic semiconductors: an emerging class of materials with novel properties. Polym. Journ., 2018, 50, 603–614. doi.org/10.1038/s41428-018-0070-6.
- Z.X. Chen, Y. Li, F. Huang. Persistent and stable organic radicals: Design, synthesis, and applications. Chem 2021, 7, 288–332. [CrossRef]
- G. Kaplan. Symmetry properties of the electron density and following from it limits on the KS-DFT applications. Mol. Phys. 2018, 116, 658-665. [CrossRef]
- Sheka E F, Popova N A, Popova V A. Physics and chemistry of graphene. Emergentness, magnetism, mechanophysics and mechanochemistry Phys. Usp. 2018, 61, 645-691.
- E.F. Sheka, Chemical susceptibility of fullerenes in view of Hartree–Fock approach. Int. J. Quant. Chem. 2007, 107, 2803–2816. [CrossRef]
- Zayets, V. A. CLUSTER-Z1: Quantum-Chemical Software for Calculations in the s,p-Basis. Inst. Surf. Chem. Nat. Ac. Sci. of Ukraine: Kiev, 1990; (in Russian).
- Berzigiyarov, P.K.; Zayets, V.A.; Ginzburg, I.Ya.; et al. NANOPACK: Parallel codes for semiempirical quantum chemical calculations of large systems in the sp- and spd-basis. Int. J. Quantum Chem. 2002, 88, 449-462.
- Dewar M.J.S.; Zoebisch, E.G.; Healey, E.F.; Stewart, J.J.P. AM1: A new general-purpose quantum mechanical molecular model. J.Amer.Chem.Soc. 1985, 107, 3902-3909.
- Sheka, E.F. Fullerenes. Nanochemistry, Nanomagnetism, Nanomedicine, Nanophotonics. CRC Press, Taylor and Francis Group, Boca Raton, 2011.
- Sheka, E.F. Spin Chemical Physics of Graphene; Pan Stanford: Singapore, 2018.
- Sheka, E. F. Virtual vibrational spectrometry of stable radicals—necklaced graphene molecules. Nanomat. 2022, 12, 597.
- Mauri, A.; Consonni, V.; Todeschini, R. (2017). Molecular Descriptors. In: Leszczynski, J., Kaczmarek-Kedziera, A., Puzyn, T., G. Papadopoulos, M., Reis, H., K. Shukla, M. (eds) Handbook of Computational Chemistry. Springer, Cham.
- E. T. Denisov, O. M. Sarkisov, G. I. Likhtenshtein, Chemical Kinetics: Fundamentals and Recent Developments, Elsevier, Amsterdam 2003. 2015, 70, 111-161.
- Е.Ф. Шека. Спинoвая химия фуллерена. Издательствo РУДН, Мoсква, 2025.
- E.F.Sheka Stretching and breaking of chemical bonds, correlation of electrons, and radical properties of covalent species, Adv. Quant. Chem.
- Sheka, E.F.; Razbirin, B. S.; Nelson, D. K. Continuous symmetry of C60 fullerene and its derivatives. J. Phys. Chem. A 2011, 115, 3480-3490. [CrossRef]
- Löwdin, P-O. Quantum theory of many-particle systems. III. Extension of the Hartree-Fock scheme to include degenerate systems and correlation effects. Phys. Rev.1955, 97, 1509-1520. [CrossRef]
- Löwdin, P-O Correlation problem in many-electron quantum mechanics. 1. Review of different approaches and discussion of some current ideas. Adv. Chem. Phys. 1958, 2, 209-322.
- Fucutome, H. Unrestricted Hartree–Fock theory and its applications to molecules and chemical reactions. Int. J. Quant. Chem.1981, 20, 955-964.
- Takatsuka, K.; Fueno, T.; Yamaguchi, K. Distribution of odd electrons in ground-state molecules. Theor. Chim. Acta 1978, 48 175-183. [CrossRef]
- Staroverov, V. N.; Davidson, E. R. Distribution of effectively unpaired electrons. Chem. Phys. Lett. 2000, 330, 161-168. [CrossRef]
- Sheka, E.F. Spin effects of sp2 nanocarbons in light of unrestricted Hartree-Fock approach and spin-orbit coupling theory, in Quantum Systems in Physics, Chemistry, and Biology: Advances in Concepts and Applications (Tadjer, A., Pavlov, R., Maruani, J., Brändas, E.J. and Delgado-Barrio, G., eds.) Progress in Theoretical Chemistry and Physics 30, Springer, Switzerland, pp. 39-63. 2017.
- Sheka, E.F. 2011. Computational synthesis of hydrogenated fullerenes from C60 to C60H60. J Mol Mod: 17, 1973-1984. [CrossRef]
- Sheka, E.F. 2010. Step-wise computational synthesis of fullerene C60 derivatives. 1.Fluorinated fullerenes C60F2k. JETP 111, 395-412. [CrossRef]
- D. Zouraris (и еще 48 автoрoв). CompSafeNano project: NanoInformatics approaches for safe-by-design nanomaterials. Compt. Struct. Biotech. J. 2025, 29, 13-26. [CrossRef]
| Set of digital twins | ||||
| Virtual reaction solution for styrene () | ||||
| 8.50 (1) 2) | ||||
| 12.06 (2) 2) 6.12 - 16.49 (2-6) 2,3) |
||||
| Virtual reaction solution for methyl methacrylate () | ||||
| 12.28 (1) 2) | ||||
| 10.46 (2) 2) | ||||
| Virtual reaction solution for () | ||||
| 19.09 (1) 2) | ||||
| 8.39 (2) 2) 7.74 (3) 2,3) 8.49 (4) 2,3) |
||||
| () |
() |
|||||||
| Set of digital twins | ||||||||
| 2-bond | 1-bond | |||||||
| - | - | - | ||||||
| - | - | - | ||||||
| - | - | |||||||
| Virtual reaction solution for methyl methacrylate | ||||||||
| 10.46 (2) 2) | 12.28 (1) 2) | > 20 (2) 3) | ||||||
| - | - | 11.58 | ||||||
| - | - | 9.40 | ||||||
| Virtual reaction solution for styrene | ||||||||
| 12.06 (2) 2) 6.12 - 16.49 (2-6) 2,4) |
8.50 (1) 2) | 24.52 (2) 3) | 8.38 (1) | |||||
| - | - | - | 9.73 | |||||
| 7.02 5) | ||||||||
| - | - | - | 9.41 | |||||
| - | 8.78 (2)2,5 | 2.79 (1) 2,5) | 28.82 5) | |||||
| 11.25 (2) 2) | - | - | ||||||
| Virtual reaction solution for | ||||||||
| 8.39 (2) 2) 7.74 (3) 2,4) 8.49 (4) 2,4) |
19.09 (1) 2) | 17.29 (1) 3) 27.79 (2) |
20.01 | |||||
| - | - | 0.023 | ||||||
| - | - | 9.398 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




