Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
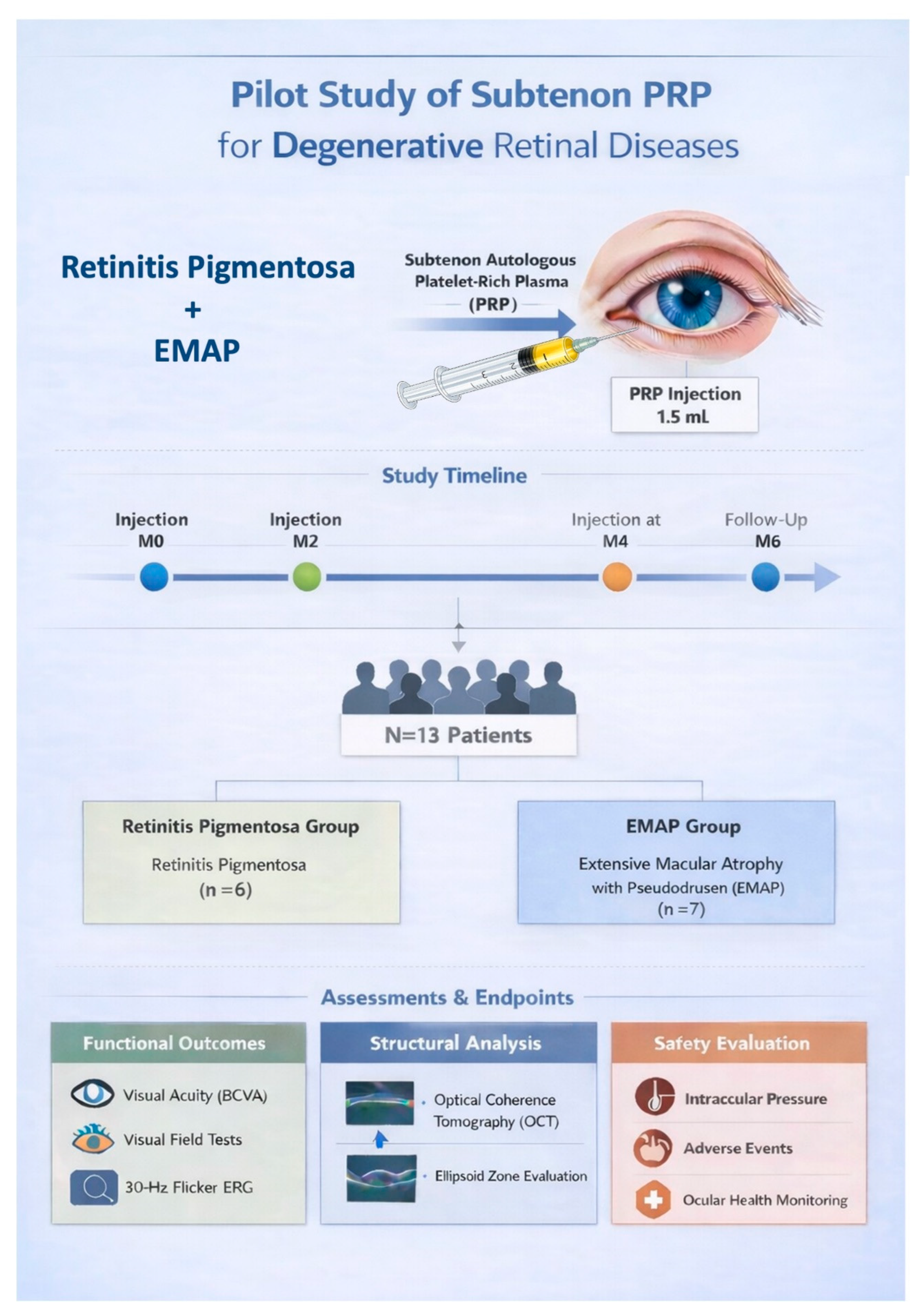
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethics
2.2. Study Population and Eligibility Criteria
- Age ≥ 18 years;
- Clinical diagnosis of retinitis pigmentosa (RP) or EMAP, confirmed by multimodal retinal imaging;
- Best-corrected visual acuity (BCVA) of counting fingers at 1 meter or better (≤ 1.9 logMAR) in the study eye;
- Measurable visual field using iCare COMPASS automated perimetry (10-2 or 24-2 strategy) with acceptable reliability indices;
- Clear ocular media allowing safe periocular injection and high-quality optical coherence tomography (OCT)imaging;
- Ability and willingness to comply with scheduled study visits.
- Exclusion Criteria
- Active ocular inflammation or infectious ocular disease;
- Active choroidal neovascularization or other macular diseases unrelated to RP or EMAP;
- Uncontrolled glaucoma (IOP > 21 mmHg despite treatment) or non-related optic neuropathies;
- Significant media opacity impairing imaging quality or injection safety;
- Recent ocular interventions that could confound outcomes (intravitreal therapy, periocular corticosteroid injection, or major intraocular surgery within 3 months);
- Known hypersensitivity to materials used in PRP preparation or injection;
- Coagulopathy or contraindications to periocular injections (platelet count < 100,000/µL or INR > 1.5);
- Pregnancy or breastfeeding;
- Uncontrolled systemic disease or participation in another interventional clinical trial within 3 months prior to enrollment.
2.3. Platelet-Rich Plasma Preparation
2.4. Intervention Protocol
- Baseline (Month 0),
- Month 2 (M2),
- Month 4 (M4).
2.5. Clinical Assessments and Follow-Up
2.6. Outcome Measures
- Best-corrected visual acuity (BCVA, logMAR);
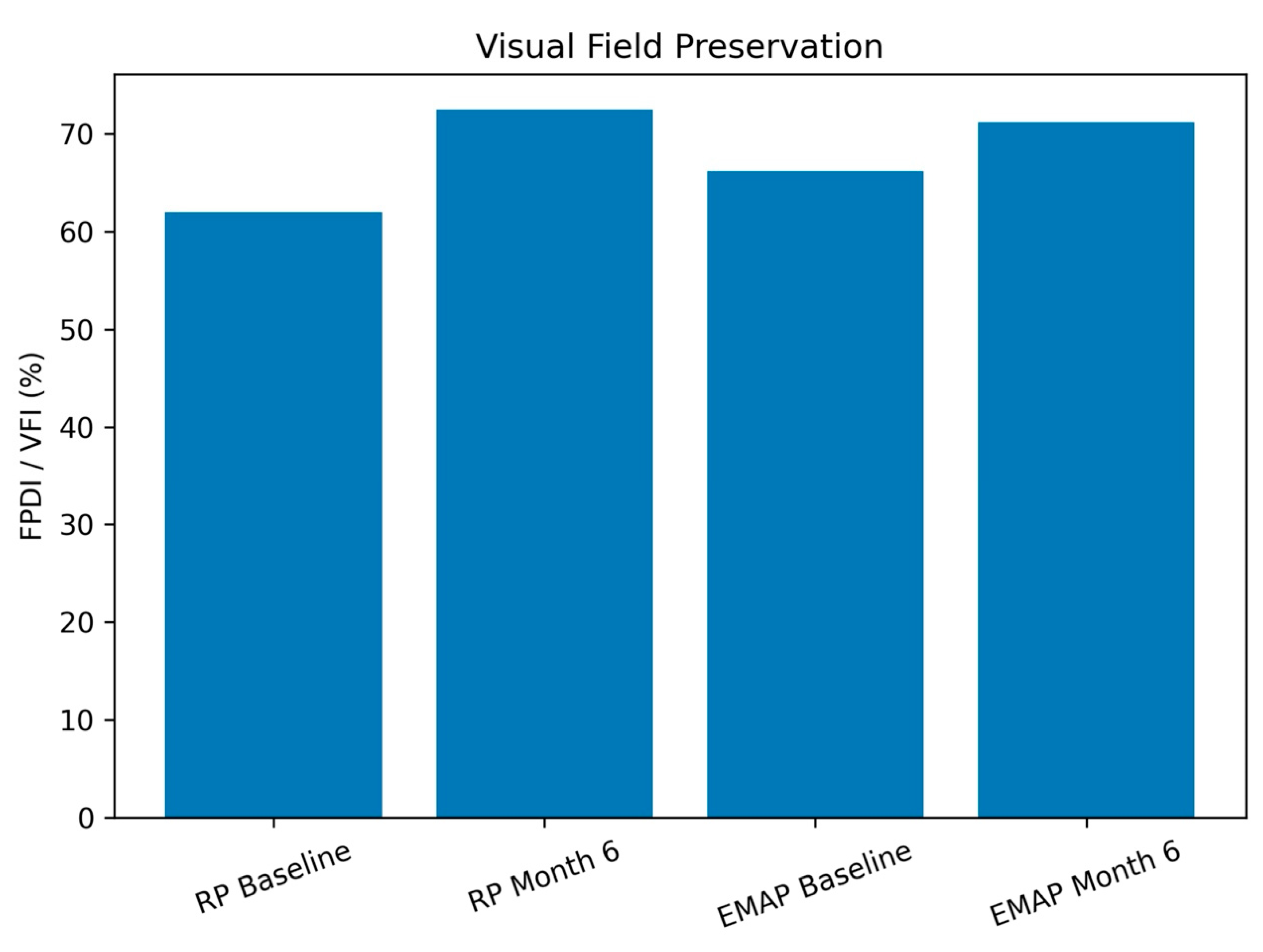
- Visual field preservation, assessed by Field Preservation Deviation Index (FPDI) and Mean Deviation (MD).
- When FPDI was unavailable, the Visual Field Index (VFI) was used as a surrogate.Secondary outcomes included:
- 30-Hz flicker ERG amplitude (phase 1, µV);
- Structural OCT parameters (central macular thickness and ellipsoid zone length);
- Safety outcomes, including IOP changes and ocular adverse events.
- Spectral-domain optical coherence tomography (SD-OCT) was performed at baseline and Month 6 to evaluate macular structure and to exclude confounding or safety-related retinal changes. OCT assessment included screening for cystoid macular edema (CME), choroidal neovascularization (CNV), central macular thickness, and qualitative evaluation of ellipsoid zone (EZ) integrity. Patients with active CME or CNV were excluded from enrollment. Throughout the study period, no new-onset CME or CNV was detected. Ellipsoid zone abnormalities were common at baseline and were considered reflective of underlying disease severity rather than treatment-related effects.
2.7. Statistical Analysis
3. Results
3.1. Study Population
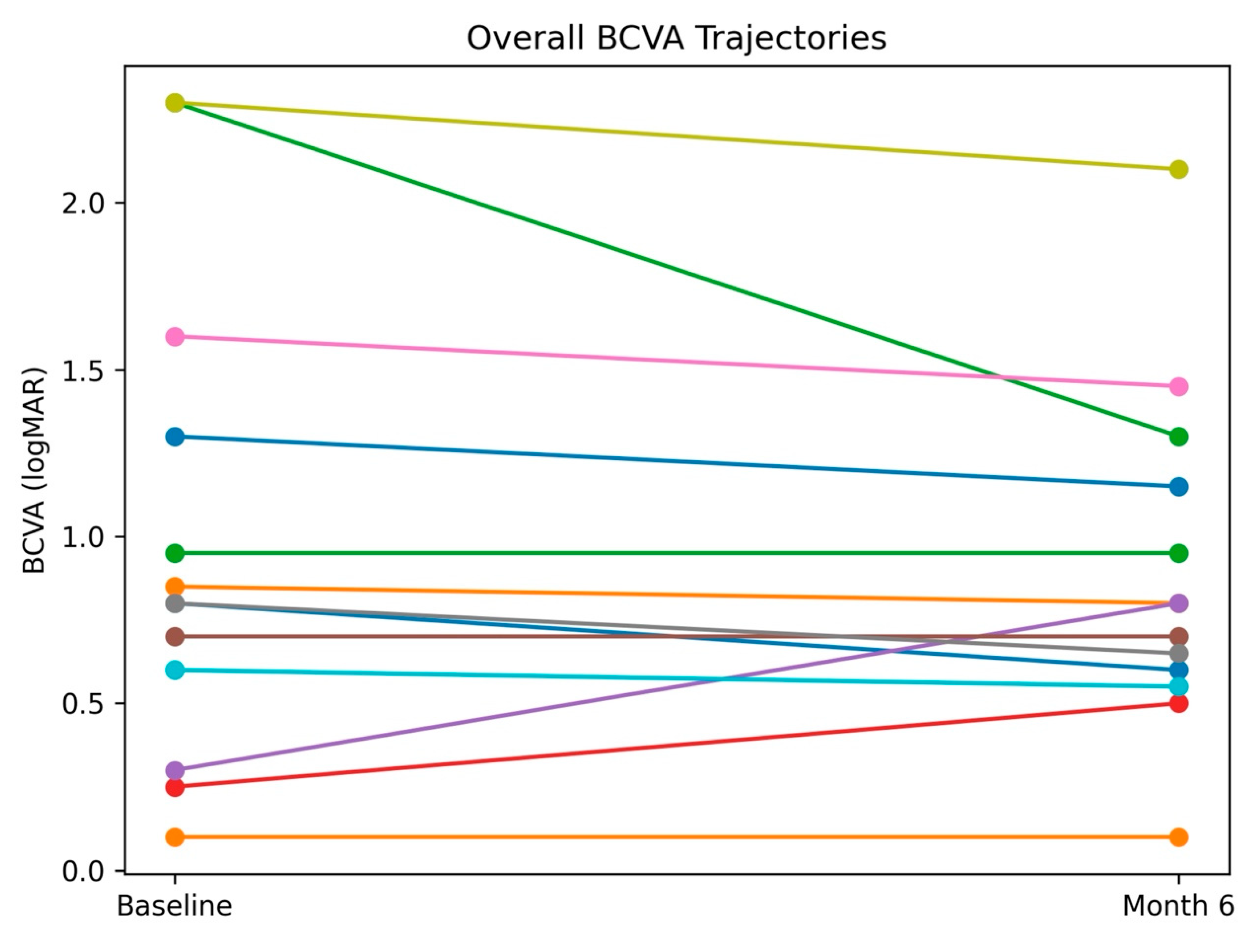
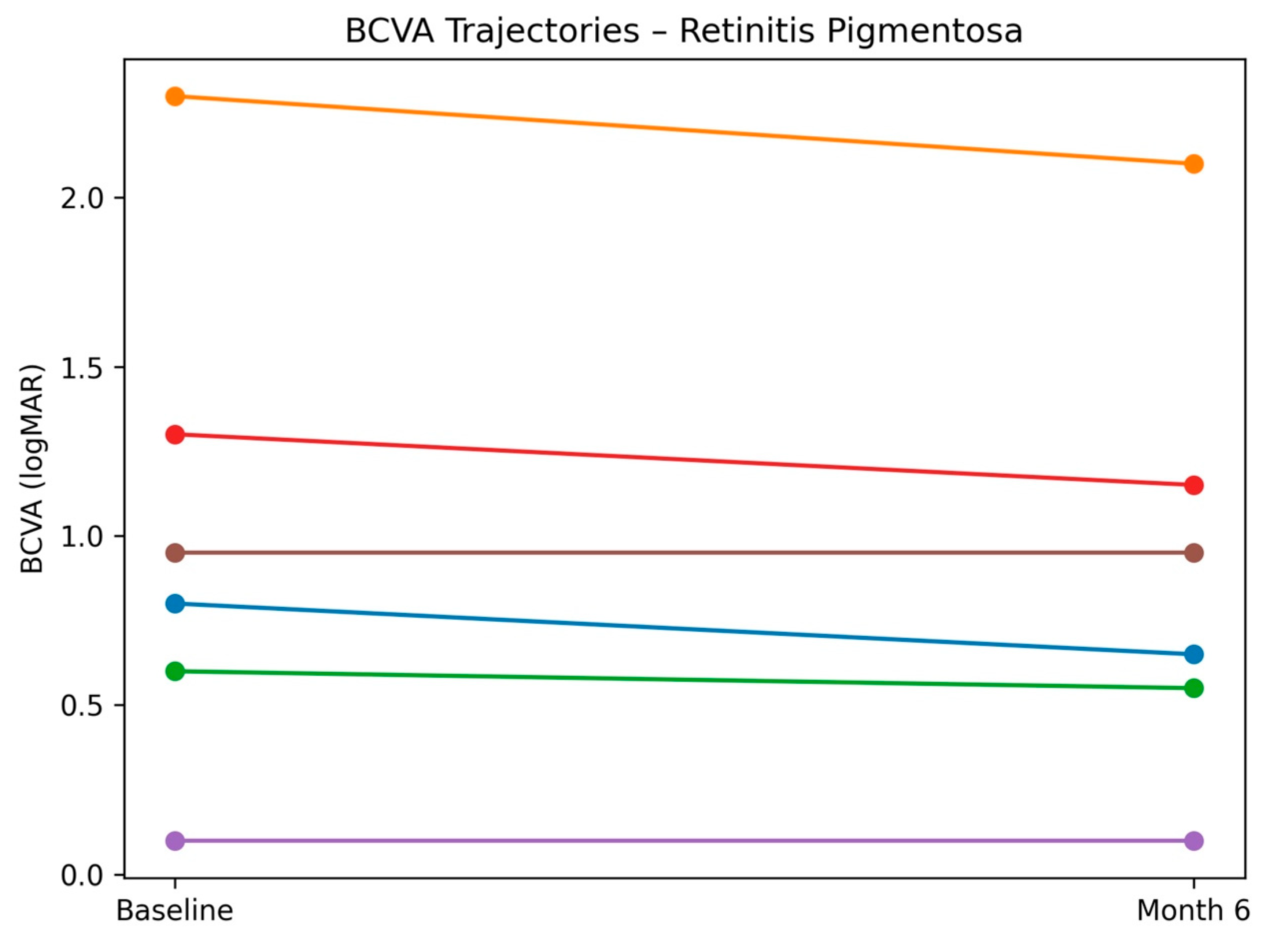
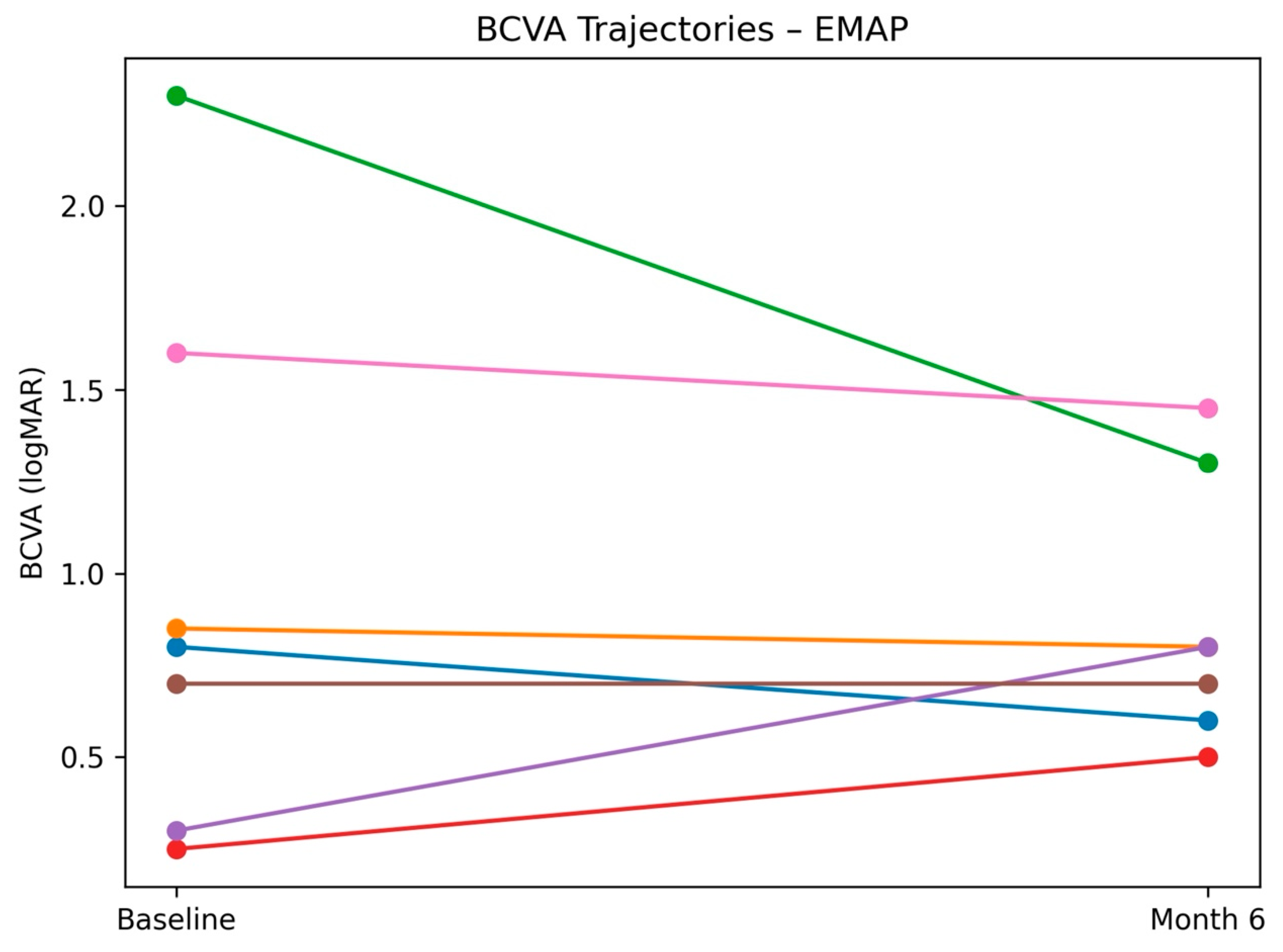
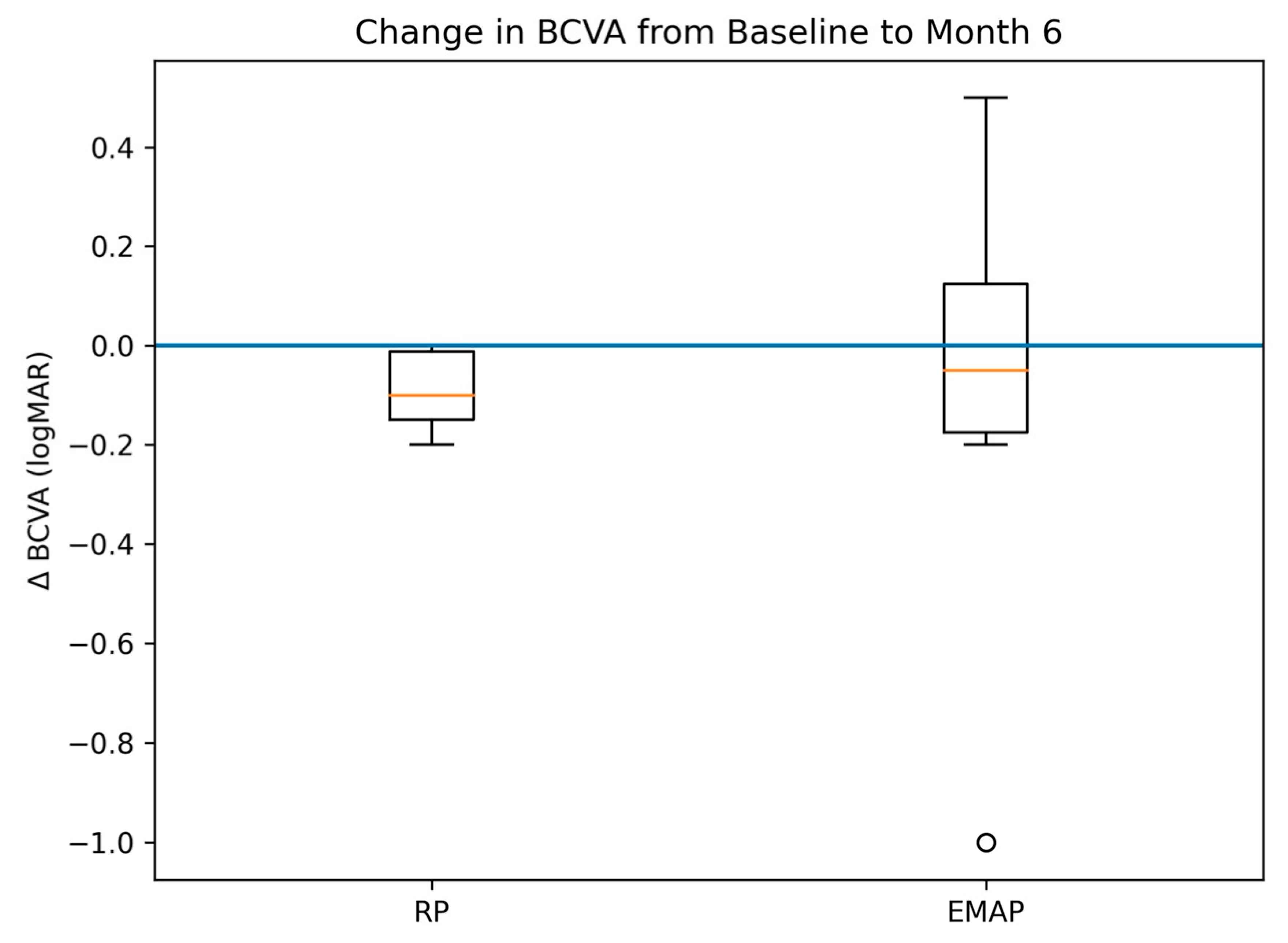
3.2. Best-Corrected Visual Acuity
3.3. Visual Field Outcomes
3.4. Electrophysiological Outcomes
3.5. Safety and Adverse Events
4. Discussion
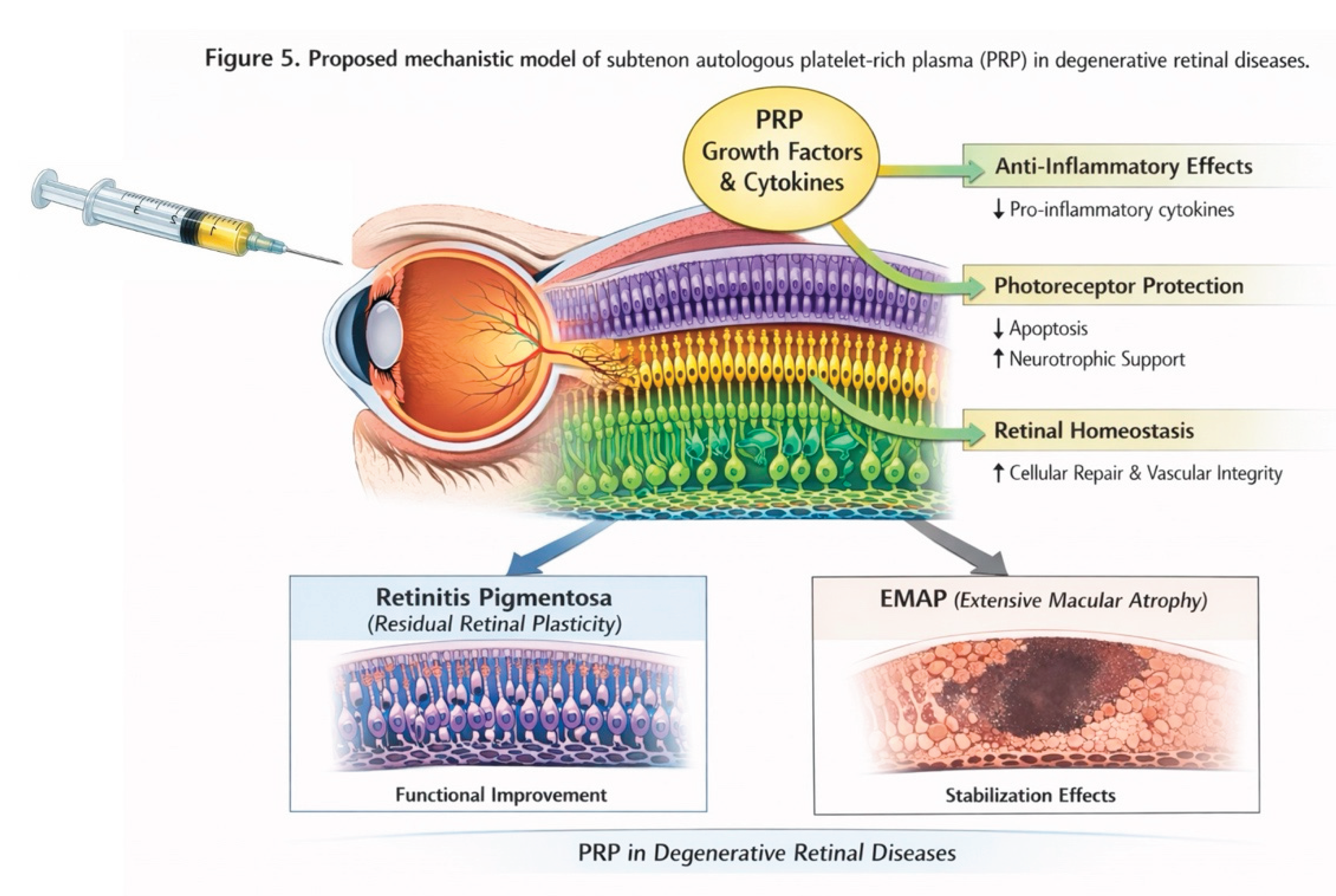
4.1. Differential Biological Substrate in RP and EMAP
4.2. Mechanistic Rationale for PRP Effects in RP
4.3. Limited Functional Plasticity in EMAP
4.4. Functional Outcomes Versus Structural and Electrophysiological Measures
4.5. Safety Considerations
4.6. Limitations and Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Clinical Trial Registration
References
- Hartong, DT; Berson, EL; Dryja, TP. Retinitis pigmentosa. Lancet 2006, 368(9549), 1795–1809. [Google Scholar] [CrossRef]
- Dias, MF; Joo, K; Kemp, JA; et al. Molecular genetics and emerging therapies for retinitis pigmentosa. Prog Retin Eye Res. 2018, 63, 107–131. [Google Scholar] [CrossRef]
- Hamel, CP; Defoort-Dhellemmes, S; Bron, A; et al. Extensive macular atrophy with pseudodrusen-like appearance: a new clinical and angiographic entity. Ophthalmology 2009, 116(4), 770–778. [Google Scholar] [CrossRef]
- Gliem, M; Hendig, D; Finger, RP; et al. Reticular pseudodrusen in eyes with extensive macular atrophy with pseudodrusen-like appearance. Retina 2013, 33(6), 1151–1158. [Google Scholar]
- Spaide, RF. Subretinal drusenoid deposits (reticular pseudodrusen): pathology and clinical significance. Retina 2010, 30(10), 1441–1451. [Google Scholar] [CrossRef] [PubMed]
- Balaratnasingam, C; et al. Progression of reticular pseudodrusen: a spectral domain OCT analysis. Invest Ophthalmol Vis Sci. 2014, 55(8), 5199–5205. [Google Scholar]
- Zarranz-Ventura, J; et al. Adalimumab for sight-threatening uveitis in patients with juvenile idiopathic arthritis. Br J Ophthalmol. 2014, 98(6), 736–741. [Google Scholar]
- Whitcup, SM; et al. Inflammation in retinal disease. Int J Inflam. 2013, 2013, 724648. [Google Scholar] [CrossRef] [PubMed]
- Parmeggiani, F; et al. Inflammation in retinal degenerations: epidemiology, pathogenetic role, and therapeutic implications. Prog Retin Eye Res. 2012, 31(5), 482–518. [Google Scholar]
- Rutar, M; Natoli, R; Provis, JM. Small interfering RNA targeting tumor necrosis factor-α in retinal microglia suppresses experimental photoreceptor degeneration. J Neuroinflammation 2012, 9, 221. [Google Scholar] [CrossRef]
- Siqueira, RC; Brandão, CC. The role of cytokines in degenerative retinal diseases: a comprehensive review. Biomedicines 2025, 13(7), 1724. [Google Scholar] [CrossRef] [PubMed]
- Forooghian, F; et al. TNF inhibition in the treatment of retinal disease: current status and future prospects. Eye 2010, 24(5), 747–753. [Google Scholar]
- Robson, AG; et al. Pattern and flicker ERGs in retinal and optic nerve disorders: diagnostic insights and applications. Prog Retin Eye Res. 2020, 79, 100859. [Google Scholar]
- Binns, AM; et al. Changes in implicit time of the cone flicker ERG in retinal degeneration. Doc Ophthalmol. 2012, 124(1), 1–12. [Google Scholar]
- Anitua, E; et al. Autologous platelet lysates for the treatment of ocular surface disorders. Curr Eye Res. 2016, 41(7), 873–882. [Google Scholar]
- López-Plandolit, S; et al. Autologous platelet-rich plasma eye drops in neurotrophic keratitis. Cornea 2011, 30(12), 1428–1430. [Google Scholar]
- Alio, JL; Arnalich-Montiel, F; Rodriguez, AE. The role of eye platelet-rich plasma in ophthalmology. Curr Pharm Biotechnol. 2012, 13(7), 1257–1265. [Google Scholar] [CrossRef]
- Arnalich, F; et al. Solid platelet-rich plasma in corneal surgery. Ophthalmol Ther. 2016, 5(1), 31–45. [Google Scholar] [CrossRef]
- Arslan, U; Özmert, E. Management of retinitis pigmentosa via platelet-rich plasma. Adv Ther. 2020, 37(5), 2390–2412. [Google Scholar] [CrossRef]
- Kahraman, N; Oner, A. Subtenon injection of autologous platelet-rich plasma in retinitis pigmentosa. Open J Ophthalmol. 2020, 10, 77–88. [Google Scholar] [CrossRef]
- Sahli, E; Arslan, U; Özmert, E; İdil, A. Subtenon autologous platelet-rich plasma injections in retinitis pigmentosa. Regen Med. 2021, 16(2), 131–143. [Google Scholar] [CrossRef] [PubMed]
- Sahli, E; Özmert, E; Günel, MD; Atilla, H. Factors affecting response to PRP therapy in retinitis pigmentosa. Int Ophthalmol. 2024, 44(1), 388. [Google Scholar] [CrossRef] [PubMed]
- Scholl, HPN; Strauss, RW; Singh, MS; et al. Emerging therapies for inherited retinal degeneration. Sci Transl Med. 2016, 8(368), 368rv6. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



