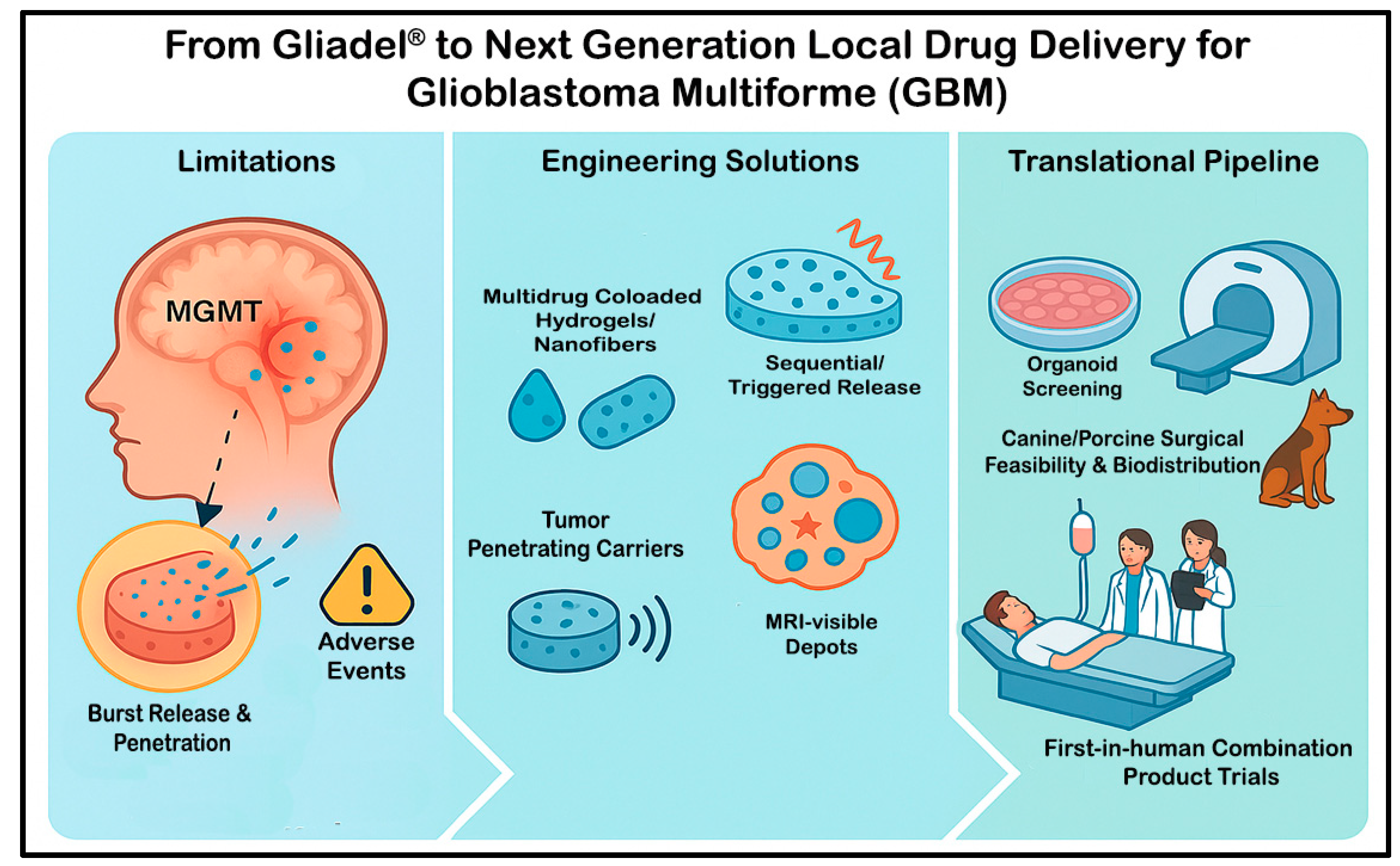
1. Introduction
Glioblastoma multiforme (GBM), now defined in the WHO CNS5 under adult-type diffuse gliomas as “Glioblastoma, IDH-wildtype” (CNS WHO grade 4) meeting histologic or molecular criteria (TERT promoter mutation, EGFR amplification, or +7/−10 signature), [
1] remains uniformly lethal despite maximal multimodal therapy. Standard care since 2005 consists of maximal safe resection followed by chemoradiation. [
2,
3] The median overall survival with contemporary therapy rarely exceeds 15–21 months, underscoring the need for better local control at the resection margin where most recurrences arise. [
4,
5] Local drug delivery has gained popularity because it bypasses the blood–brain barrier (BBB), potentially increasing drug concentrations in the surgical cavity and in the surrounding infiltrating tumor while minimizing systemic exposure. [
6]
Despite ongoing improvements in surgery, radiotherapy, and systemic treatments, studies on patterns of failure consistently indicate that most recurrences occur within 2 cm of the original tumor site. [
7] This suggests that residual cells at the resection margin are not being effectively treated, even when dose constraints are met. [
7,
8] In this setting, the resection cavity serves as a convenient site for treatment, allowing neurosurgeons to insert implants, catheters, or injectable depots during surgery, with the goal of maintaining high drug levels around the cavity rim for a prolonged time. [
6,
9] Local approaches, including biodegradable wafers, convection-enhanced delivery (CED), brachytherapy, and in situ–forming depots, are therefore increasingly viewed as complementary to systemic therapy rather than competitors, aiming to intensify treatment where microscopic disease is densest while limiting systemic toxicity. [
9,
10]
The BCNU–polyanhydride wafer (Gliadel wafer) is the first proof-of-concept and the only currently approved method for local intracranial delivery of chemotherapy, according to the U.S. Food and Drug Administration (FDA). [
11,
12] Randomized trials demonstrated modestly improved outcomes compared to placebo, both in recurrent malignant glioma (median survival 7.1 versus 5.3 months) and in newly diagnosed high-grade glioma (median 13.9 versus 11.6 months; 29% relative risk reduction in death). [
12,
13] These trials predated or only partially overlapped the Temozolomide (TMZ) era, and subsequent observational series in the TMZ era have shown heterogeneity of the benefit of Gliadel wafer. [
14,
15]
This review critically examines the limitations of the Gliadel wafer and proposes new design and translational strategies for next-generation local therapies for GBM. Addressing these limitations can enhance the efficacy and safety of these local drug-delivery systems for patients with GBM (
Figure 1).
2. Current Limitations of Gliadel Wafer
Despite establishing the feasibility of local chemotherapy, Gliadel wafers provide only incremental benefit in selected patients because their performance is constrained by tumor biology, pharmacokinetics, and device limitations. On the biology side, single-agent carmustine is blunted by MGMT-mediated repair and related DNA-damage responses, so durable control of a heterogeneous, infiltrative GBM is not achieved. Pharmacokinetically, the polifeprosan-20 matrix delivers a short, front-loaded burst of BCNU over only a few days, and concentrations fall steeply from the cavity wall, typically reaching just millimeters into the surrounding brain. Practical coverage is further limited by irregular cavity geometry and, when present, cerebrospinal fluid (CSF) communications. Safety profile and adverse events such as edema, seizures, wound-healing problems, CSF leak or infection, and cyst formation can occur. The wafer is brittle and moisture-sensitive (frozen storage and careful placement are required), and, therefore, full dosing is not always achievable. Together, these factors define the ceiling of first-generation wafers and motivate the engineering and translational strategies outlined in
Section 3 and
Section 4.
2.1. Limited Efficacy of Carmustine as a Single Agent
BCNU (carmustine) is a chloroethylnitrosourea that alkylates DNA at the O^6 position of guanine; the O^6-chloroethyl adduct can evolve into interstrand cross-links that kill tumor cells. [
16,
17] The DNA repair protein MGMT removes O^6 adducts in a single step and is a major determinant of nitrosourea and other O^6-alkylator resistance. High MGMT activity blunts drug effect, whereas MGMT promoter methylation correlates with greater alkylator responsiveness. [
18,
19] Mismatch repair and broader DNA damage responses further shape sensitivity to nitrosoureas and TMZ, enabling rapid selection of resistant clones under treatment pressure. [
17]
Across pivotal randomized trials, the survival gains with Gliadel wafer were modest, typically weeks to a few months, underscoring that single-agent BCNU cannot control a heterogeneous, often MGMT-proficient infiltrative tumor. [
13,
20] In addition, carmustine has not been shown to have a significant therapeutic effect on GBM stem cells, which are believed to drive tumor recurrence. [
21]
In the TMZ era, benefit appears most plausible in well-selected patients – for example, those with near-gross-total resection. Yet contemporary series report variable results, and some guidelines note limited routine use in Europe. [
22,
23]
At the cellular level, GBM stem-like cells (GSCs) and other treatment-tolerant subpopulations exhibit high DNA-repair capacity, quiescence, and metabolic plasticity, which collectively reduce their sensitivity to BCNU and TMZ. [
24,
25] These cells often reside at the infiltrative margin, around perivascular niches, and along white-matter tracts – regions that extend beyond the short diffusion radius of carmustine from the cavity wall. [
26,
27] Heterogeneous transcriptional programs (e.g., proneural, classical, mesenchymal) and dynamic phenotypic switching under treatment pressure further argue against relying on a single alkylator, and instead support rational local combinations that simultaneously target DNA damage response, invasion, and stem-cell survival pathways. [
28,
29]
2.2. Pharmacokinetic Constraints
The polyanhydride matrix (polifeprosan 20) hydrolyzes after implantation, producing an early burst of carmustine followed by a rapid decline in release; experimental work shows that most of the drug elutes over several days, while the polymer remnants persist for weeks. [
30]
Human pharmacokinetics mirror this behavior: systemic BCNU is detectable for about 24h, with a maximal blood concentration occurring about 3h after placement, supporting the evidence of limited, short-lived output from the implant. [
31,
32]
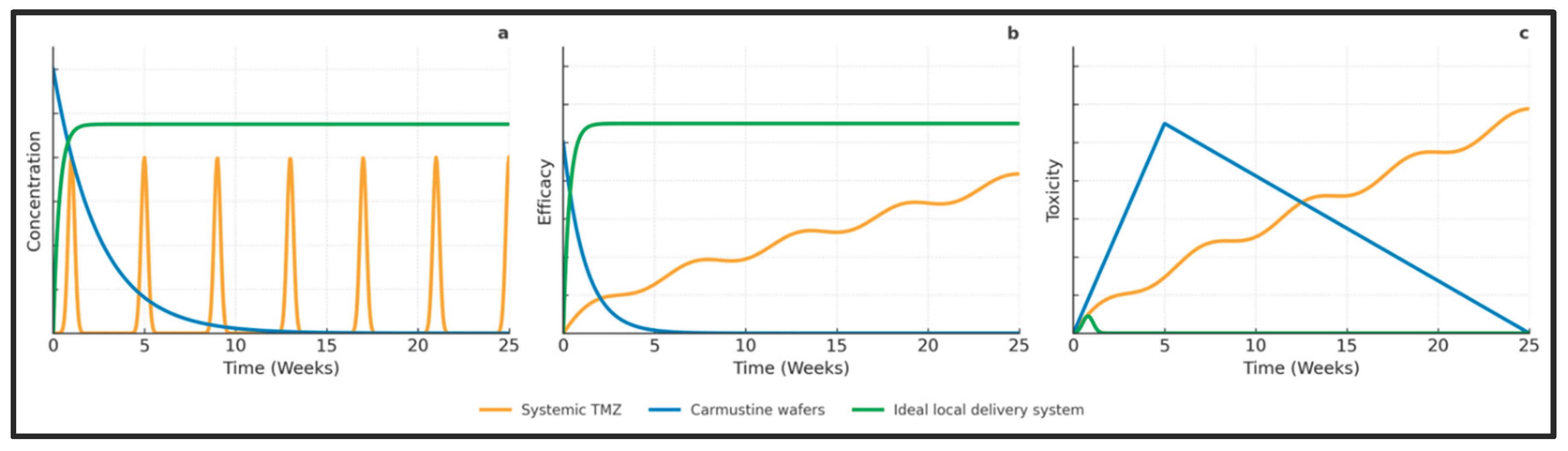
Drug levels decrease rapidly as they move away from the cavity wall, so significant drug concentrations are confined to a few millimeters into the surrounding tissue. This leaves any deeper invasive disease untreated. Both computer simulations and animal studies support the existence of this sharp concentration gradient. [
33,
34]
Reports on diffusion distances differ by model; however, even the more optimistic studies suggest typical penetration is in the millimeter range. [
20]
In real-world practice, these pharmacokinetic challenges are further complicated by the shape of the resection cavity. [
9,
20] Surgeons often need to cut wafers into smaller pieces, leave some surfaces exposed, or avoid placing wafers near sulci, ventricles, or cisterns to minimize migration and cerebrospinal fluid obstruction. [
20,
35] Consequently, only a portion of the peri-cavity surface contacts the drug-eluting polymer directly, creating a steep concentration gradient. [
20,
36] This means that cells just a few millimeters away often receive subtherapeutic doses, which explains why tumors tend to recur just beyond the expected diffusion zone. [
7,
36] Developing more uniform and conformal depots remains an important goal for future treatments, and these conceptual relationships between drug exposure, efficacy, and toxicity for systemic TMZ, Gliadel wafers, and an idealized local delivery platform are summarized in
Figure 2.
2.3. Safety, Adverse Effects, and Handling Constraints
Post-implant issues include peri-cavitary edema, seizures, wound-healing problems, CSF leak/meningitis, cyst formation, and intracranial hypertension. The FDA label reports seizures in up to 37% of patients with recurrent disease and cautions about edema and wound-healing risks that warrant close monitoring. [
32]
Case series and reviews report instances of peri-cavitary edema that are severe enough to exert pressure on surrounding structures, sometimes leading to hydrocephalus or displacement of wafers, especially when the cavities are connected to CSF pathways. [
37,
38]
Although systemic exposure is minimal and temporary, blood levels can be detected for approximately 24 hours following placement. As a result, the mutagenic and teratogenic risks associated with carmustine necessitate precautions related to contraception and breastfeeding. [
32]
Each wafer contains 7.7 mg of carmustine embedded in polifeprosan-20, which is a polymer composed of bis(p-carboxyphenoxy) propane and sebacic acid in a 20:80 ratio. It is recommended to use up to eight wafers to cover the affected area. [
32]
Labels and technical sheets specify frozen storage (≤ –20 °C) and careful intraoperative handling due to moisture sensitivity and fragility; in practice, irregular cavity geometry often prevents achieving the full eight-wafer dose. [
32]
Meta-analyses and large retrospective series confirm that these device-related toxicities are not rare, particularly when wafers are combined with standard postoperative chemoradiation. [
39,
40] Higher rates of cerebral edema, wound dehiscence, and intracranial infection have been reported compared with historical controls, and some series describe cystic degeneration of the cavity that can necessitate re-operation or long-term CSF diversion. [
41,
42] In patients with limited performance status or multiple comorbidities, the clinical impact of such complications may outweigh the modest survival benefit. [
13,
40] Consequently, many centers limit Gliadel use to carefully selected patients – typically those with good functional status (e.g., Karnofsky ≥70), supratentorial tumors amenable to near-gross-total resection, and cavities without direct communication to the ventricular system or basal cisterns. [
13,
35] Intraoperatively, best practices include gentle placement under direct visualization, avoiding multilayer stacking in narrow recesses, and striving for watertight dural closure to minimize CSF leak and wafer migration. [
20,
35] Even with meticulous technique, however, the wafer’s brittleness, fixed dimensions, and moisture sensitivity make it challenging to achieve uniform coverage of complex cavities, underscoring the need for more conformable, robust local delivery systems. [
6,
43]
3. Emerging Strategies to Overcome Current Limitations
Local therapy for GBM is evolving from single-agent, fast-eluting wafers toward combination depots and smarter materials that relatively match the biology and geometry of the disease. The most promising directions pair rational drug combinations with delivery matrices that release for months, conform to complex cavities, and spread the drug deeper into the peri-resection cavity parenchyma.
3.1. Multidrug Local Delivery Systems
Given GBM’s heterogeneity, stem cells, and rapid adaptive resistance, local drug combinations are more likely to succeed than replicating the first-generation single-agent approach. Numerous drugs have been discovered or repurposed to target GBM cells and have shown significant therapeutic efficacy in in vitro and in vivo studies. However, the ideal combination to eradicate the heterogeneous cell populations in Glioblastoma remains to be identified. [
44]
In orthotopic models, co-loaded polymer implants have outperformed single-drug wafers – for example, TMZ and BCNU released from a single biodegradable wafer produced stronger tumor control than either agent alone, establishing feasibility for intracavitary combination chemotherapy. [
45]
Similar results have been shown with TMZ and paclitaxel packaged in a photopolymerizable hydrogel or pellets, which provided sustained release, better conformity to complex margins, and improved survival compared with one-drug depots. [
46,
47,
48,
49] Together, these data suggest that, when used locally, combinations can complement each other mechanistically without increasing surgical morbidity or systemic toxicity. [
20]
A key challenge is carefully choosing which drugs to combine locally rather than empirically co-loading multiple cytotoxics. [
50,
51] Modern genomic and transcriptomic profiling reveals substantial inter- and intra-tumoral variation in pathways such as RTK/RAS/PI3K, cell-cycle regulation, and DNA repair, suggesting that ideal combinations will pair a DNA-damaging backbone (e.g., alkylators or topoisomerase inhibitors) with agents targeting compensatory survival pathways, invasion, or stem-cell maintenance. [
52,
53] Local depots also offer an opportunity to co-deliver agents that are poorly tolerated systemically – such as high-dose alkylators or radiosensitizers – while keeping systemic exposure low, potentially widening the therapeutic window when combined with radiotherapy and systemic TMZ. [
20,
54]
3.2. Engineering Improved Wafer Systems
Modern materials engineering offers several levers to correct the pharmacokinetic and spatial limits of current wafers. [
55] Hydrogels and nanofiber meshes can be tuned for long-lived, low-burst release and sequential dosing. For example, layered or core–sheath meshes can provide an initial radiosensitizer treatment followed by extended chemotherapy. Additionally, conformal patches can increase surface area without increasing cavity pressure. [
56,
57,
58]
In situ–forming, tissue-adhesive hydrogels improve retention on irregular walls and have delivered therapeutic proteins and antibodies in primary human GBM models, supporting their use as conformal depots after resection. To push the drug beyond the millimeter-scale rim, carriers can be equipped with tumor-penetrating ligands (e.g., RGD/iRGD) or built using layer-by-layer (LbL) surface chemistry; both approaches increase brain-tissue distribution and glioma-cell uptake in vivo, with LbL nanoparticles also enabling MRI visualization of local spread. [
59,
60]
Imaging-visible depots are feasible, MRI-monitored hydrogels have been tracked longitudinally in the brain, opening the door to noninvasive placement checks and dose-response readouts in early trials. [
61] In addition, triggerable systems (e.g., ultrasound/sonodynamic-responsive formulations) are being developed to boost release or add orthogonal cytotoxic mechanisms while preserving intracranial biocompatibility. [
62]
Beyond simply prolonging drug release, advanced biomaterials can be designed to actively influence the surrounding tissue environment. Hydrogels with customizable stiffness, porosity, and degradation rates can be tailored to facilitate deeper tissue penetration or to align with nearby blood vessels. [
43,
63] Electrospun meshes can be layered or patterned to establish spatial drug gradients that better target infiltrative tumor margins. [
57] Thermoresponsive nanocomposite hydrogels, injected as liquids and solidifying at body temperature, can create soft depots lining surgical cavities, as shown in orthotopic glioblastoma models, where these hydrogels provide extended intracortical drug release with minimal initial burst. [
64,
65] Emerging stimuli-responsive depots can also be activated by external triggers, such as magnetic fields or focused ultrasound, to control drug release on demand. [
66,
67] For instance, hydrogels containing magnetic nanoparticles can generate mild heat under alternating magnetic fields, speeding up drug diffusion and improving tissue penetration. [
66] Ultrasound-responsive systems may allow noninvasive enhancements of local therapy at specific times. [
67,
68] Additionally, nano-engineered hydrogels that co-deliver chemotherapeutic drugs with immunomodulators or metabolic agents have been shown to remodel the immune and stromal microenvironment, increasing T-cell infiltration and reducing tumor recurrence in preclinical glioma models. [
63,
69] These advances suggest that future drug-delivery wafers could become programmable, visible reservoirs capable of controlled drug release, rather than passive, degrading discs. [
6,
69]
3.3. Targeted Therapeutics
In addition to cytotoxic drugs, local delivery systems can provide targeted therapies like oncolytic viruses and small-molecule or antibody treatments.
Oncolytic viruses preferentially replicate in cancer cells, sparing normal cells, and induce direct oncolysis and promote immunogenic cell death. A phase I study of DNX-2401 (Delta-24-RGD), an oncolytic adenovirus, reported that among 37 patients with malignant glioma (including GBM and IDH-wildtype), 20% survived longer than 3 years and 12% achieved greater than a 95% reduction in tumor enhancement. [
70] Another phase II trial using intratumoral oncolytic herpes virus G47Δ in 19 adult patients with residual or recurrent supratentorial GBM demonstrated a 1-year survival rate of 84.2% with a median overall survival of 20.2 months from therapy initiation. [
71] Finally, in a multicenter phase 1/2 study combining oncolytic DNX-2401 virotherapy with pembrolizumab, no dose-limiting toxicities were observed, and the 12-month overall survival was 52.7%, compared with only 20% in the control group. [
72]
Local or intracavitary administration is particularly appealing for biological agents whose systemic use is limited by toxicity, immunogenicity, or poor BBB penetration. [
73,
74] Catheter-based CED has already enabled focal infusion of recombinant toxins, radiolabeled antibodies, and immune agonists deep within the brain, and similar pharmacokinetic principles can be harnessed by biodegradable depots positioned along the cavity wall. [
75,
76] By co-localizing oncolytic viruses, cytokines, or checkpoint modulators with regions of greatest residual tumor burden, future platforms may amplify intratumoral immune activation while keeping systemic exposure low and reducing the risk of off-target organ toxicity. [
69,
77]
Small-molecule/antibody conjugates for local delivery can be engineered as locoregional depots to release molecularly selective payloads – including small-molecule conjugates, antibody fragments, or peptide-decorated nanoparticles – so that uptake is driven by tumor-specific ligands while exposure remains concentrated around the cavity. Integrin-targeted designs (e.g., RGD/iRGD) improve cellular entry and tissue spread in GBM models, and can be embedded within hydrogels to sustain release and widen coverage at the infiltrative edge. [
78,
79]
CD44-directed systems, often built on hyaluronic-acid (HA) backbones, show preferential uptake by GBM cells versus normal glia and have enhanced antitumor activity in orthotopic studies – making HA-NP/hydrogel composites attractive for postoperative intracavitary delivery. [
80,
81] Hydrogel–nanoparticle matrices have already demonstrated feasibility in in-cavity placement and survival gains in rodents, supporting the platform concept for conjugated or ligand-guided agents after resection. [
10,
64]
That said, translation to patients remains challenging: for example, the EGFR-targeted ADC depatuxizumab mafodotin (ABT-414) failed to improve overall survival in a phase 3 population despite biological rationale, underscoring the need to pair smarter targeting with appropriate local delivery and patient selection. [
82] Recent engineering advances (e.g., micro-mesh and conformal hydrogel depots that carry nanoparticles or antibody fragments) aim to provide sustained, spatially uniform release compatible with neurosurgical workflow, but clinical validation in GBM is still pending. [
83]
Another emerging direction is cell-based local immunotherapy. Early clinical experience with dual-targeting CAR-T cells directed against antigens such as EGFR and IL13Rα2 has shown that intracavitary or intraventricular dosing is feasible, and that organoid-based avatars can help interpret response patterns and toxicity. [
84,
85] Embedding CAR-T cells, engineered macrophages, or dendritic-cell vaccines within supportive hydrogels or porous scaffolds is being explored preclinically to prolong cell persistence, guide their migration into the infiltrative rim, and shield them from hostile microenvironmental cues. [
86] Although still at a conceptual stage, these strategies highlight how intracavitary depots could eventually function as multifunctional “immune niches” that release drugs, living effector cells, and even diagnostic reporters in a coordinated fashion.
4. Translational Path: Beyond Traditional Preclinical Models
The Gliadel wafer was approved many years ago for local chemotherapy treatment of GBM and served as an early proof-of-concept for delivering drugs directly into the brain. However, its preclinical testing used 9L gliosarcoma cells in rats, a model now known to poorly predict how human GBM behaves. These limitations include a lack of tumor diversity, unnatural growth patterns, and failure to mimic interactions between the tumor and the immune system, all of which contribute to the high failure rate when translating findings to clinical settings. [
87,
88] Therefore, next-generation local drug-delivery platforms require preclinical models that better reflect the molecular, genetic, and immunological context of human GBM.
4.1. Efficacy Preclinical Studies
Patient-derived xenografts (PDXs) and GBM organoids (GBOs) have emerged as robust tools for bridging this gap. [
89] Derived from patient tumor tissue or short-term cultured cells, these models preserve tumor heterogeneity and molecular characteristics, allowing realistic assessment of therapeutic efficacy. [
90] Implantation in immunocompetent mice under short-term immunosuppression enables evaluation of therapies in the context of an intact immune system, capturing pro- and anti-tumor immune responses. [
91]
Beyond serving as generic efficacy platforms, PDX and organoid systems are increasingly being used in “functional precision medicine” workflows in which each patient’s tumor generates a small biobank of models. [
92,
93] These patient-derived models can be screened ex vivo against panels of systemic and locally deliverable agents, including potential intracavitary drug combinations, with results feeding back into clinical decision-making within clinically relevant time frames. [
92,
94] For local drug-delivery research, such pipelines enable evaluation of which drug cocktails (e.g., alkylators plus targeted agents or immunomodulators) are most active against an individual tumor before embedding them into wafers or hydrogels, thereby de-risking early-phase trials and aligning intracavitary therapy with patient-specific vulnerabilities. [
6,
93]
To enhance clinical relevance, standardized rodent tumor resection systems have been developed. [
95] These minimally invasive devices combine suction and a precision cutting blade to remove tumors through the original burr hole, reducing operative time, blood loss, and postoperative morbidity, while preserving tissue viability for downstream analyses. This method aids in researching perioperative biology and intra-cavity drug delivery, providing a more accurate representation of how surgery affects treatment responses. [
95]
Translationally, more predictive biological models must be matched with clinically meaningful endpoints. Incorporating standardized patient-reported outcomes (PROs) and neurocognitive testing into trials of local therapies will be essential to ensure that modest survival gains are not offset by deterioration in function or quality of life (QOL). [
96,
97] Recent work in glioblastoma shows that even when overall survival is poor, carefully delivered chemoradiotherapy can still improve specific dimensions of health-related QOL, such as emotional and social functioning, emphasizing the importance of capturing patient-centred benefits. [
98,
99] Contemporary neuro-oncology frameworks now recognize perceived cognition, communication difficulty, seizures, physical functioning, and symptomatic adverse events as core PRO domains that should accompany radiographic and survival endpoints in brain-tumor trials. [
96,
100] Local delivery platforms that prolong steroid dependence, increase seizure burden, or impair wound healing could negatively affect these domains even if progression-free survival improves, whereas technologies that reduce hospitalizations or allow earlier initiation of systemic therapy may offer net QOL advantages that deserve equal weight in clinical decision-making. [
100,
101]
4.2. Safety and Biodistribution Preclinical Studies
For safety and pharmacokinetic considerations, small animals are insufficient because their brain size, tissue structure, and drug distribution differ significantly from those of humans. Large-animal models such as dogs allow researchers to optimize implant size, drug payload, and release rates in conditions that more closely mimic human physiology.
By combining efficacy studies using PDX or GBO models, including those with resected tumors, with large-animal safety and pharmacokinetic testing, we can create a comprehensive, translational framework. This approach enhances the evaluation of advanced local drug-delivery systems, bringing us closer to effective human treatments.
Spontaneous gliomas in pet dogs have become particularly valuable in this context because they more closely recapitulate the size, microenvironment, and clinical course of human GBM than induced rodent tumors. [
102,
103] In a landmark study, CED of CPT-11-loaded liposomes into canine gliomas achieved robust intratumoral distribution on real-time MRI and produced volumetric responses that correlated with the volume of distribution, validating both the model and the delivery technique. [
102] Later work using biodegradable TMZ-loaded microcylinders implanted into partially resected canine tumors demonstrated the feasibility of postsurgical intracavitary chemotherapy and yielded detailed pharmacokinetic and safety data that would be difficult to obtain in human subjects. [
104] These naturally occurring canine models also capture clinically relevant variables such as tumor-related seizures, chronic steroid use, and heterogeneous supportive care – factors that influence edema, wound healing, and device performance. [
103] As such, they are well-suited to identify device-specific complications (e.g., cyst formation, edema around depots, catheter malfunction) before first-in-human trials. [
102,
104] Integrating large-animal studies with rodent models and patient-derived organoids can create a multi-tiered translational pipeline in which candidate local therapies are first screened for efficacy, then stress-tested for distribution and safety in realistic brains, and finally advanced into adaptive early-phase clinical trials. [
92,
102]
Early-phase human studies also offer an opportunity to refine regulatory science around intracavitary treatments. [
105,
106] Unlike purely systemic drugs, local depots and CED systems are combination products whose performance depends as much on surgical workflow, catheter positioning, and imaging guidance as on the pharmacology of the payload itself. [
9,
106] Harmonized reporting standards for implant placement, intra-operative complications, and longitudinal imaging changes – analogous to existing RANO and RANO-PRO initiatives for outcome assessment – would facilitate cross-platform comparison and accelerate the identification of best practices. [
96,
100] At the same time, phase I–II trials should incorporate detailed pharmacokinetic and pharmacodynamic sampling from re-resected tissue whenever ethically feasible, as exemplified by chronic topotecan CED studies that correlated intratumoral exposure with changes in proliferation and metabolism. [
105,
106] Such data help clarify whether disappointing clinical results reflect inadequate drug delivery, intrinsic resistance, or off-target toxicity, thereby guiding rational iteration of both drug combinations and device architecture. [
105,
106]
5. Conclusions
The use of Gliadel wafers after tumor resection has shown that intracavitary chemotherapy is feasible and can offer modest survival benefits, but overall outcomes in GBM remain poor due to the limited efficacy of single-agent carmustine, short and localized drug exposure, and device-related complications. Next-generation locoregional systems must therefore go beyond “more drug for longer” by combining multiple or targeted agents to overcome resistance, enabling controlled, sustained, and homogeneous drug delivery, and minimizing toxicity. Practically, ideal intracavitary platforms should handle multidrug payloads with independently tunable release, conform to irregular cavity geometries without causing mass effect, degrade predictably with good biocompatibility in blood and CSF, and remain compatible with repeat surgery, re-irradiation, and advanced imaging, while features like MRI visibility and externally triggered release could turn them into true theranostic devices. Progress will depend on integrating robust preclinical testing (patient-derived xenografts, organoids, large-animal models) with adaptive, biomarker-driven clinical trials in well-selected surgical candidates, using correlative measures such as cavity-wall biopsies, advanced MRI, and circulating tumor DNA to link local pharmacokinetics to biological and clinical effects. Ultimately, improving on Gliadel will require the coordinated advance of materials science, rational drug combinations, rigorous translational modeling, and carefully designed clinical implementation.
Author Contributions
Conceptualization: A.K., S.A. Methodology: A.K. Data curation: A.K. Formal analysis and investigation: A.K., M.J.K., H.C., Y.T., S.A. Writing – original draft: A.K. Writing – review & editing: A.K., M.J.K., H.C., Y.T., S.A. Supervision: S.A.
Funding
This research received no funding.
Ethics Statements
This narrative review relies solely on previously published data and does not involve any new human or animal research; IRB/ethics approval was not necessary.
Conflicts of interest
The author reports no conflicts related to this manuscript.
References
- Louis DN, Perry A, Wesseling P, et al. The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro Oncol. Aug 2 2021;23(8):1231-1251. [CrossRef]
- Stupp R, Mason WP, van den Bent MJ, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. Mar 10 2005;352(10):987-96. [CrossRef]
- Stupp R, Taillibert S, Kanner A, et al. Effect of Tumor-Treating Fields Plus Maintenance Temozolomide vs. Maintenance Temozolomide Alone on Survival in Patients With Glioblastoma: A Randomized Clinical Trial. Jama. Dec 19 2017;318(23):2306-2316. [CrossRef]
- Kotecha R, Odia Y, Khosla AA, Ahluwalia MS. Key Clinical Principles in the Management of Glioblastoma. JCO Oncol Pract. Apr 2023;19(4):180-189. [CrossRef]
- Langhans M, Popp I, Grosu AL, et al. Recurrence analysis of glioblastoma cases based on distance and dose information. Radiother Oncol. Jun 2023;183:109600. [CrossRef]
- Bastiancich C, Malfanti A, Préat V, Rahman R. Rationally designed drug delivery systems for the local treatment of resected glioblastoma. Adv Drug Deliv Rev. Oct 2021;177:113951. [CrossRef]
- Sherriff J, Tamangani J, Senthil L, et al. Patterns of relapse in glioblastoma multiforme following concomitant chemoradiotherapy with temozolomide. Br J Radiol. Feb 2013;86(1022):20120414. [CrossRef]
- Zheng L, Zhou ZR, Yu Q, et al. The Definition and Delineation of the Target Area of Radiotherapy Based on the Recurrence Pattern of Glioblastoma After Temozolomide Chemoradiotherapy. Front Oncol. 2020;10:615368. [CrossRef]
- Cha GD, Jung S, Choi SH, Kim DH. Local Drug Delivery Strategies for Glioblastoma Treatment. Brain Tumor Res Treat. Jul 2022;10(3):151-157. [CrossRef]
- Gazaille C, Sicot M, Saulnier P, Eyer J, Bastiat G. Local Delivery and Glioblastoma: Why Not Combining Sustained Release and Targeting? Front Med Technol. 2021;3:791596. [CrossRef]
- Bota DA, Desjardins A, Quinn JA, Affronti ML, Friedman HS. Interstitial chemotherapy with biodegradable BCNU (Gliadel) wafers in the treatment of malignant gliomas. Ther Clin Risk Manag. Oct 2007;3(5):707-15.
- Brem H, Piantadosi S, Burger PC, et al. Placebo-controlled trial of safety and efficacy of intraoperative controlled delivery by biodegradable polymers of chemotherapy for recurrent gliomas. The Polymer-brain Tumor Treatment Group. Lancet. Apr 22 1995;345(8956):1008-12. [CrossRef]
- Westphal M, Hilt DC, Bortey E, et al. A phase 3 trial of local chemotherapy with biodegradable carmustine (BCNU) wafers (Gliadel wafers) in patients with primary malignant glioma. Neuro Oncol. Apr 2003;5(2):79-88. [CrossRef]
- Ashby LS, Smith KA, Stea B. Gliadel wafer implantation combined with standard radiotherapy and concurrent followed by adjuvant temozolomide for treatment of newly diagnosed high-grade glioma: a systematic literature review. World J Surg Oncol. Aug 24 2016;14(1):225. [CrossRef]
- Chowdhary SA, Ryken T, Newton HB. Survival outcomes and safety of carmustine wafers in the treatment of high-grade gliomas: a meta-analysis. J Neurooncol. Apr 2015;122(2):367-82. [CrossRef]
- Bodell WJ, Aida T, Berger MS, Rosenblum ML. Repair of O6-(2-chloroethyl)guanine mediates the biological effects of chloroethylnitrosoureas. Environ Health Perspect. Oct 1985;62:119-26. [CrossRef]
- Nikolova T, Roos WP, Krämer OH, Strik HM, Kaina B. Chloroethylating nitrosoureas in cancer therapy: DNA damage, repair and cell death signaling. Biochim Biophys Acta Rev Cancer. Aug 2017;1868(1):29-39. [CrossRef]
- Gerson SL, Willson JK. O6-alkylguanine-DNA alkyltransferase. A target for the modulation of drug resistance. Hematol Oncol Clin North Am. Apr 1995;9(2):431-50.
- Margison GP, Povey AC, Kaina B, Santibáñez Koref MF. Variability and regulation of O6-alkylguanine-DNA alkyltransferase. Carcinogenesis. Apr 2003;24(4):625-35. [CrossRef]
- Wait SD, Prabhu RS, Burri SH, Atkins TG, Asher AL. Polymeric drug delivery for the treatment of glioblastoma. Neuro Oncol. Mar 2015;17 Suppl 2(Suppl 2):ii9-ii23. [CrossRef]
- Hersh AM, Gaitsch H, Alomari S, Lubelski D, Tyler BM. Molecular Pathways and Genomic Landscape of Glioblastoma Stem Cells: Opportunities for Targeted Therapy. Cancers (Basel). Jul 31 2022;14(15). [CrossRef]
- d'Avella D, DellaPuppa A. Safety and efficacy of Gliadel wafers for newly diagnosed and recurrent glioblastomas. Acta Neurochir (Wien). Aug 2012;154(8):1379-81. [CrossRef]
- Mangraviti A, Tyler B, Brem H. Interstitial chemotherapy for malignant glioma: Future prospects in the era of multimodal therapy. Surg Neurol Int. 2015;6(Suppl 1):S78-84. [CrossRef]
- Auffinger B, Spencer D, Pytel P, Ahmed AU, Lesniak MS. The role of glioma stem cells in chemotherapy resistance and glioblastoma multiforme recurrence. Expert Rev Neurother. 2015;15(7):741-52. [CrossRef]
- Singh N, Miner A, Hennis L, Mittal S. Mechanisms of temozolomide resistance in glioblastoma - a comprehensive review. Cancer Drug Resist. 2021;4(1):17-43. [CrossRef]
- Brooks LJ, Clements MP, Burden JJ, et al. The white matter is a pro-differentiative niche for glioblastoma. Nat Commun. Apr 12 2021;12(1):2184. [CrossRef]
- Brooks MD, Sengupta R, Snyder SC, Rubin JB. Hitting Them Where They Live: Targeting the Glioblastoma Perivascular Stem Cell Niche. Curr Pathobiol Rep. Jun 1 2013;1(2):101-110. [CrossRef]
- Fedele M, Cerchia L, Pegoraro S, Sgarra R, Manfioletti G. Proneural-Mesenchymal Transition: Phenotypic Plasticity to Acquire Multitherapy Resistance in Glioblastoma. Int J Mol Sci. Jun 4 2019;20(11). [CrossRef]
- Wang Z, Zhang H, Xu S, Liu Z, Cheng Q. The adaptive transition of glioblastoma stem cells and its implications on treatments. Signal Transduct Target Ther. Mar 23 2021;6(1):124. [CrossRef]
- Dang W, Daviau T, Brem H. Morphological characterization of polyanhydride biodegradable implant gliadel during in vitro and in vivo erosion using scanning electron microscopy. Pharm Res. May 1996;13(5):683-91. [CrossRef]
- Arbor Pharmaceuticals, LLC. GLIADEL Wafer (carmustine implant) [prescribing information]. Atlanta, GA: Arbor Pharmaceuticals, LLC; February 2025. Accessed November 6, 2025. https://gliadel.com/wp-content/uploads/2025/02/Gliadel-Prescribing-Information.pdf.
- US Food and Drug Administration. GLIADEL Wafer (carmustine implant) [prescribing information]. Silver Spring, MD: US Food and Drug Administration; 2018. Accessed November 6, 2025. https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/020637s029lbl.pdf.
- Healy AT, Vogelbaum MA. Convection-enhanced drug delivery for gliomas. Surg Neurol Int. 2015;6(Suppl 1):S59-67. [CrossRef]
- Wang CC, Li J, Teo CS, Lee T. The delivery of BCNU to brain tumors. J Control Release. Aug 27 1999;61(1-2):21-41. [CrossRef]
- Price SJ, Whittle IR, Ashkan K, Grundy P, Cruickshank G. NICE guidance on the use of carmustine wafers in high grade gliomas: a national study on variation in practice. Br J Neurosurg. Jun 2012;26(3):331-5. [CrossRef]
- Bodell WJ, Bodell AP, Giannini DD. Levels and distribution of BCNU in GBM tumors following intratumoral injection of DTI-015 (BCNU-ethanol). Neuro Oncol. Jan 2007;9(1):12-9. [CrossRef]
- Weber EL, Goebel EA. Cerebral edema associated with Gliadel wafers: two case studies. Neuro Oncol. Jan 2005;7(1):84-9. [CrossRef]
- Health Products Regulatory Authority. Gliadel 7.7 mg Implant: Summary of Product Characteristics. Dublin, Ireland: Health Products Regulatory Authority; February 28, 2025. Accessed November 6, 2025. https://assets.hpra.ie/products/Human/23877/Licence_PA22701-003-001_28022025163520.pdf.
- Ricciardi L, Manini I, Cesselli D, et al. Carmustine Wafers Implantation in Patients With Newly Diagnosed High Grade Glioma: Is It Still an Option? Front Neurol. 2022;13:884158. [CrossRef]
- Xiao ZZ, Wang ZF, Lan T, et al. Carmustine as a Supplementary Therapeutic Option for Glioblastoma: A Systematic Review and Meta-Analysis. Front Neurol. 2020;11:1036. [CrossRef]
- Dörner L, Ulmer S, Rohr A, Mehdorn HM, Nabavi A. Space-occupying cyst development in the resection cavity of malignant gliomas following Gliadel® implantation--incidence, therapeutic strategies, and outcome. J Clin Neurosci. Mar 2011;18(3):347-51. [CrossRef]
- McGirt MJ, Villavicencio AT, Bulsara KR, Friedman HS, Friedman AH. Management of tumor bed cysts after chemotherapeutic wafer implantation. Report of four cases. J Neurosurg. May 2002;96(5):941-5. [CrossRef]
- Zhong Z, Gan L, Feng Z, et al. Hydrogel local drug delivery systems for postsurgical management of tumors: Status Quo and perspectives. Mater Today Bio. Dec 2024;29:101308. [CrossRef]
- Alomari S, Zhang I, Hernandez A, et al. Drug Repurposing for Glioblastoma and Current Advances in Drug Delivery-A Comprehensive Review of the Literature. Biomolecules. Dec 13 2021;11(12). [CrossRef]
- Shapira-Furman T, Serra R, Gorelick N, et al. Biodegradable wafers releasing Temozolomide and Carmustine for the treatment of brain cancer. J Control Release. Feb 10 2019;295:93-101. [CrossRef]
- Quinn JA, Jiang SX, Carter J, et al. Phase II trial of Gliadel plus O6-benzylguanine in adults with recurrent glioblastoma multiforme. Clin Cancer Res. Feb 1 2009;15(3):1064-8. [CrossRef]
- Slika H, Shahani A, Gattu K, et al. Intracranial Nanogel Pellets Carrying Temozolomide and Paclitaxel for Adjuvant Brain Cancer Therapy. Mol Pharm. Jan 6 2025;22(1):131-141. [CrossRef]
- Weingart J, Grossman SA, Carson KA, et al. Phase I trial of polifeprosan 20 with carmustine implant plus continuous infusion of intravenous O6-benzylguanine in adults with recurrent malignant glioma: new approaches to brain tumor therapy CNS consortium trial. J Clin Oncol. Feb 1 2007;25(4):399-404. [CrossRef]
- Zhao M, Bozzato E, Joudiou N, et al. Codelivery of paclitaxel and temozolomide through a photopolymerizable hydrogel prevents glioblastoma recurrence after surgical resection. J Control Release. Sep 10 2019;309:72-81. [CrossRef]
- Alghamdi M, Gumbleton M, Newland B. Local delivery to malignant brain tumors: potential biomaterial-based therapeutic/adjuvant strategies. Biomater Sci. Sep 14 2021;9(18):6037-6051. [CrossRef]
- Bastiancich C, Danhier P, Préat V, Danhier F. Anticancer drug-loaded hydrogels as drug delivery systems for the local treatment of glioblastoma. J Control Release. Dec 10 2016;243:29-42. [CrossRef]
- Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature. Oct 23 2008;455(7216):1061-8. [CrossRef]
- Verhaak RG, Hoadley KA, Purdom E, et al. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell. Jan 19 2010;17(1):98-110. [CrossRef]
- Bow H, Hwang LS, Schildhaus N, et al. Local delivery of angiogenesis-inhibitor minocycline combined with radiotherapy and oral temozolomide chemotherapy in 9L glioma. J Neurosurg. Mar 2014;120(3):662-9. [CrossRef]
- Gaitsch H, Hersh AM, Alomari S, Tyler BM. Dendrimer Technology in Glioma: Functional Design and Potential Applications. Cancers (Basel). Feb 8 2023;15(4). [CrossRef]
- Han D, Serra R, Gorelick N, et al. Multi-layered core-sheath fiber membranes for controlled drug release in the local treatment of brain tumor. Sci Rep. Nov 29 2019;9(1):17936. [CrossRef]
- Li J, Xu W, Li D, et al. Locally Deployable Nanofiber Patch for Sequential Drug Delivery in Treatment of Primary and Advanced Orthotopic Hepatomas. ACS Nano. Jul 24 2018;12(7):6685-6699. [CrossRef]
- Hersh AM, Alomari S, Tyler BM. Crossing the Blood-Brain Barrier: Advances in Nanoparticle Technology for Drug Delivery in Neuro-Oncology. Int J Mol Sci. Apr 9 2022;23(8). [CrossRef]
- Pickering AJ, Lamson NG, Marand MH, Hwang W, Straehla JP, Hammond PT. Layer-by-Layer Polymer Functionalization Improves Nanoparticle Penetration and Glioblastoma Targeting in the Brain. ACS Nano. Dec 12 2023;17(23):24154-24169. [CrossRef]
- Shadab A, Farokhi S, Fakouri A, et al. Hydrogel-based nanoparticles: revolutionizing brain tumor treatment and paving the way for future innovations. Eur J Med Res. Feb 4 2025;30(1):71. [CrossRef]
- Kim JI, Kim B, Chun C, Lee SH, Song SC. MRI-monitored long-term therapeutic hydrogel system for brain tumors without surgical resection. Biomaterials. Jun 2012;33(19):4836-42. [CrossRef]
- Mess G, Anderson T, Kapoor S, et al. Sonodynamic Therapy for the Treatment of Glioblastoma Multiforme in a Mouse Model Using a Portable Benchtop Focused Ultrasound System. J Vis Exp. Feb 10 2023;(192). [CrossRef]
- Yang J, Wang Z, Ma C, et al. Advances in Hydrogels of Drug Delivery Systems for the Local Treatment of Brain Tumors. Gels. Jun 17 2024;10(6). [CrossRef]
- Bastiancich C, Bianco J, Vanvarenberg K, et al. Injectable nanomedicine hydrogel for local chemotherapy of glioblastoma after surgical resection. J Control Release. Oct 28 2017;264:45-54. [CrossRef]
- Ding L, Wang Q, Shen M, et al. Thermoresponsive nanocomposite gel for local drug delivery to suppress the growth of glioma by inducing autophagy. Autophagy. Jul 3 2017;13(7):1176-1190. [CrossRef]
- Beola L, Iturrioz-Rodríguez N, Pucci C, Bertorelli R, Ciofani G. Drug-Loaded Lipid Magnetic Nanoparticles for Combined Local Hyperthermia and Chemotherapy against Glioblastoma Multiforme. ACS Nano. Sep 26 2023;17(18):18441-18455. [CrossRef]
- Sun Y, Chen LG, Fan XM, Pang JL. Ultrasound Responsive Smart Implantable Hydrogels for Targeted Delivery of Drugs: Reviewing Current Practices. Int J Nanomedicine. 2022;17:5001-5026. [CrossRef]
- Arrieta VA, Gould A, Kim KS, et al. Ultrasound-mediated delivery of doxorubicin to the brain results in immune modulation and improved responses to PD-1 blockade in gliomas. Nat Commun. Jun 6 2024;15(1):4698. [CrossRef]
- Mohammadzadeh V, Atapour-Mashhad H, Shahvali S, et al. Hydrogels as advanced drug delivery platforms for cancer immunotherapy: promising innovations and future outlook. J Nanobiotechnology. Jul 28 2025;23(1):545. [CrossRef]
- Lang FF, Conrad C, Gomez-Manzano C, et al. Phase I Study of DNX-2401 (Delta-24-RGD) Oncolytic Adenovirus: Replication and Immunotherapeutic Effects in Recurrent Malignant Glioma. J Clin Oncol. May 10 2018;36(14):1419-1427. [CrossRef]
- Todo T, Ito H, Ino Y, et al. Intratumoral oncolytic herpes virus G47∆ for residual or recurrent glioblastoma: a phase 2 trial. Nat Med. Aug 2022;28(8):1630-1639. [CrossRef]
- Nassiri F, Patil V, Yefet LS, et al. Oncolytic DNX-2401 virotherapy plus pembrolizumab in recurrent glioblastoma: a phase 1/2 trial. Nat Med. Jun 2023;29(6):1370-1378. [CrossRef]
- Rui Y, Green JJ. Overcoming delivery barriers in immunotherapy for glioblastoma. Drug Deliv Transl Res. Dec 2021;11(6):2302-2316. [CrossRef]
- van Solinge TS, Nieland L, Chiocca EA, Broekman MLD. Advances in local therapy for glioblastoma - taking the fight to the tumour. Nat Rev Neurol. Apr 2022;18(4):221-236. [CrossRef]
- Bailly C, Vidal A, Bonnemaire C, et al. Potential for Nuclear Medicine Therapy for Glioblastoma Treatment. Front Pharmacol. 2019;10:772. [CrossRef]
- Rolfe NW, Dadario NB, Canoll P, Bruce JN. A Review of Therapeutic Agents Given by Convection-Enhanced Delivery for Adult Glioblastoma. Pharmaceuticals (Basel). Jul 23 2024;17(8). [CrossRef]
- Haddad AF, Young JS, Aghi MK. Using viral vectors to deliver local immunotherapy to glioblastoma. Neurosurg Focus. Feb 2021;50(2):E4. [CrossRef]
- Branco F, Cunha J, Mendes M, Vitorino C, Sousa JJ. Peptide-Hitchhiking for the Development of Nanosystems in Glioblastoma. ACS Nano. Jul 2 2024;18(26):16359-16394. [CrossRef]
- Wang B, Tang D, Cui J, Jiang H, Yu J, Guo Z. RGD-based self-assembling nanodrugs for improved tumor therapy. Front Pharmacol. 2024;15:1477409. [CrossRef]
- Chen C, Fan R, Wang Y, et al. Hyaluronic Acid-Conjugated Nanoparticles for the Targeted Delivery of Cabazitaxel to CD44-Overexpressing Glioblastoma Cells. J Biomed Nanotechnol. Apr 1 2021;17(4):595-605. [CrossRef]
- Hayward SL, Wilson CL, Kidambi S. Hyaluronic acid-conjugated liposome nanoparticles for targeted delivery to CD44 overexpressing glioblastoma cells. Oncotarget. Jun 7 2016;7(23):34158-71. [CrossRef]
- Lassman AB, Pugh SL, Wang TJC, et al. Depatuxizumab mafodotin in EGFR-amplified newly diagnosed glioblastoma: A phase III randomized clinical trial. Neuro Oncol. Feb 14 2023;25(2):339-350. [CrossRef]
- Di Mascolo D, Guerriero I, Pesce C, Spanò R, Palange AL, Decuzzi P. μMESH-Enabled Sustained Delivery of Molecular and Nanoformulated Drugs for Glioblastoma Treatment. ACS Nano. Aug 8 2023;17(15):14572-14585. [CrossRef]
- Bagley SJ, Logun M, Fraietta JA, et al. Intrathecal bivalent CAR T cells targeting EGFR and IL13Rα2 in recurrent glioblastoma: phase 1 trial interim results. Nat Med. May 2024;30(5):1320-1329. [CrossRef]
- Logun M, Wang X, Sun Y, et al. Patient-derived glioblastoma organoids as real-time avatars for assessing responses to clinical CAR-T cell therapy. Cell Stem Cell. Feb 6 2025;32(2):181-190.e4. [CrossRef]
- Kass L, Thang M, Zhang Y, et al. Development of a biocompatible 3D hydrogel scaffold using continuous liquid interface production for the delivery of cell therapies to treat recurrent glioblastoma. Bioeng Transl Med. Nov 2024;9(6):e10676. [CrossRef]
- Buahin KG, Brem H. Interstitial chemotherapy of experimental brain tumors: comparison of intratumoral injection versus polymeric controlled release. J Neurooncol. Nov 1995;26(2):103-10. [CrossRef]
- Tamargo RJ, Myseros JS, Epstein JI, Yang MB, Chasin M, Brem H. Interstitial chemotherapy of the 9L gliosarcoma: controlled release polymers for drug delivery in the brain. Cancer Res. Jan 15 1993;53(2):329-33.
- Percie du Sert N, Hurst V, Ahluwalia A, et al. The ARRIVE guidelines 2.0: Updated guidelines for reporting animal research. PLoS Biol. Jul 2020;18(7):e3000410. [CrossRef]
- Jacob F, Ming GL, Song H. Generation and biobanking of patient-derived glioblastoma organoids and their application in CAR T cell testing. Nat Protoc. Dec 2020;15(12):4000-4033. [CrossRef]
- Slika H, Karimov Z, Alimonti P, et al. Preclinical Models and Technologies in Glioblastoma Research: Evolution, Current State, and Future Avenues. Int J Mol Sci. Nov 14 2023;24(22). [CrossRef]
- Mann B, Artz N, Darawsheh R, Kram DE, Hingtgen S, Satterlee AB. Opportunities and challenges for patient-derived models of brain tumors in functional precision medicine. NPJ Precis Oncol. Feb 14 2025;9(1):47. [CrossRef]
- Ratliff M, Kim H, Qi H, et al. Patient-Derived Tumor Organoids for Guidance of Personalized Drug Therapies in Recurrent Glioblastoma. Int J Mol Sci. Jun 12 2022;23(12). [CrossRef]
- Alcaniz J, Winkler L, Dahlmann M, et al. Clinically relevant glioblastoma patient-derived xenograft models to guide drug development and identify molecular signatures. Front Oncol. 2023;13:1129627. [CrossRef]
- Alomari S, Kedda J, Malla AP, et al. Implementation of Minimally Invasive Brain Tumor Resection in Rodents for High Viability Tissue Collection. J Vis Exp. May 9 2022;(183). [CrossRef]
- Dirven L, Armstrong TS, Blakeley JO, et al. Working plan for the use of patient-reported outcome measures in adults with brain tumours: a Response Assessment in Neuro-Oncology (RANO) initiative. Lancet Oncol. Mar 2018;19(3):e173-e180. [CrossRef]
- Scheepens JCC, Taphoorn MJB, Koekkoek JAF. Patient-reported outcomes in neuro-oncology. Curr Opin Oncol. Nov 1 2024;36(6):560-568. [CrossRef]
- Pelak DMJ, Hummer A, Hug PE, et al. Patient-reported outcomes, neurocognitive functioning and oncologic results of pencil-beam-scanning proton beam therapy for CNS WHO G2 and G3 IDH1-mutant diffuse adult glioma: A single institution experience. Int J Radiat Oncol Biol Phys. Nov 10 2025; [CrossRef]
- Taphoorn MJ, Stupp R, Coens C, et al. Health-related quality of life in patients with glioblastoma: a randomised controlled trial. Lancet Oncol. Dec 2005;6(12):937-44. [CrossRef]
- Vera E, Christ A, Grajkowska E, et al. Relationship between RANO-PRO Working Group standardised priority constructs and disease progression among malignant glioma patients: A retrospective cohort study. EClinicalMedicine. Jan 2023;55:101718. [CrossRef]
- Noll K, King AL, Dirven L, Armstrong TS, Taphoorn MJB, Wefel JS. Neurocognition and Health-Related Quality of Life Among Patients with Brain Tumors. Hematol Oncol Clin North Am. Feb 2022;36(1):269-282. [CrossRef]
- Dickinson PJ, LeCouteur RA, Higgins RJ, et al. Canine spontaneous glioma: a translational model system for convection-enhanced delivery. Neuro Oncol. Sep 2010;12(9):928-40. [CrossRef]
- José-López R. Chemotherapy for the treatment of intracranial glioma in dogs. Front Vet Sci. 2023;10:1273122. [CrossRef]
- Hicks J, Platt S, Stewart G, et al. Intratumoral temozolomide in spontaneous canine gliomas: feasibility of a novel therapy using implanted microcylinders. Vet Med Sci. Feb 2019;5(1):5-18. [CrossRef]
- Spinazzi EF, Argenziano MG, Upadhyayula PS, et al. Chronic convection-enhanced delivery of topotecan for patients with recurrent glioblastoma: a first-in-patient, single-centre, single-arm, phase 1b trial. Lancet Oncol. Nov 2022;23(11):1409-1418. [CrossRef]
- Upadhyayula PS, Spinazzi EF, Argenziano MG, Canoll P, Bruce JN. Convection Enhanced Delivery of Topotecan for Gliomas: A Single-Center Experience. Pharmaceutics. Dec 30 2020;13(1). [CrossRef]
- Tabet A, Jensen MP, Parkins CC, Patil PG, Watts C, Scherman OA. Designing Next-Generation Local Drug Delivery Vehicles for Glioblastoma Adjuvant Chemotherapy: Lessons from the Clinic. Adv Healthc Mater. Feb 2019;8(3):e1801391. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).