Submitted:
03 February 2026
Posted:
04 February 2026
You are already at the latest version
Abstract
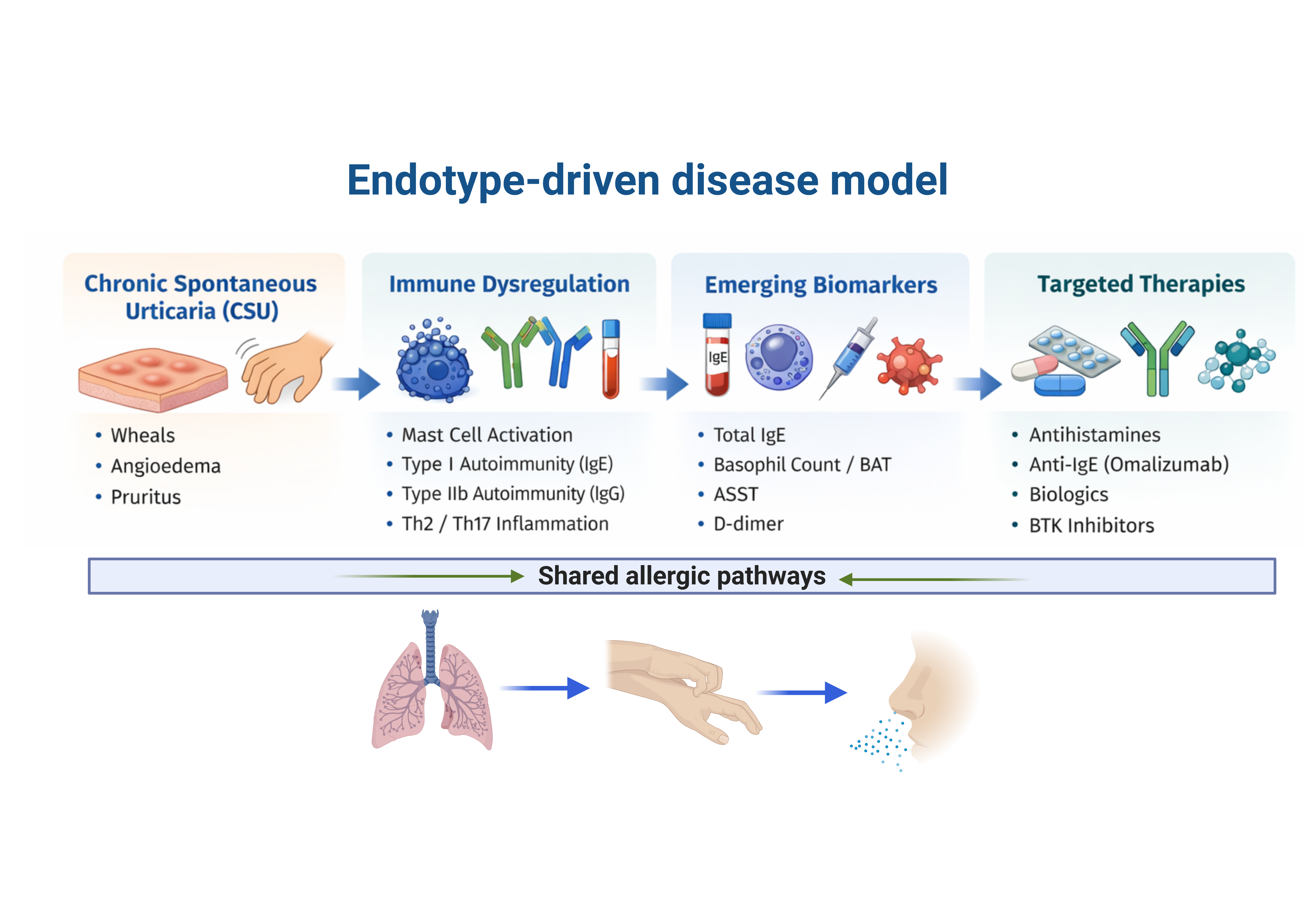
Keywords:
1. Introduction
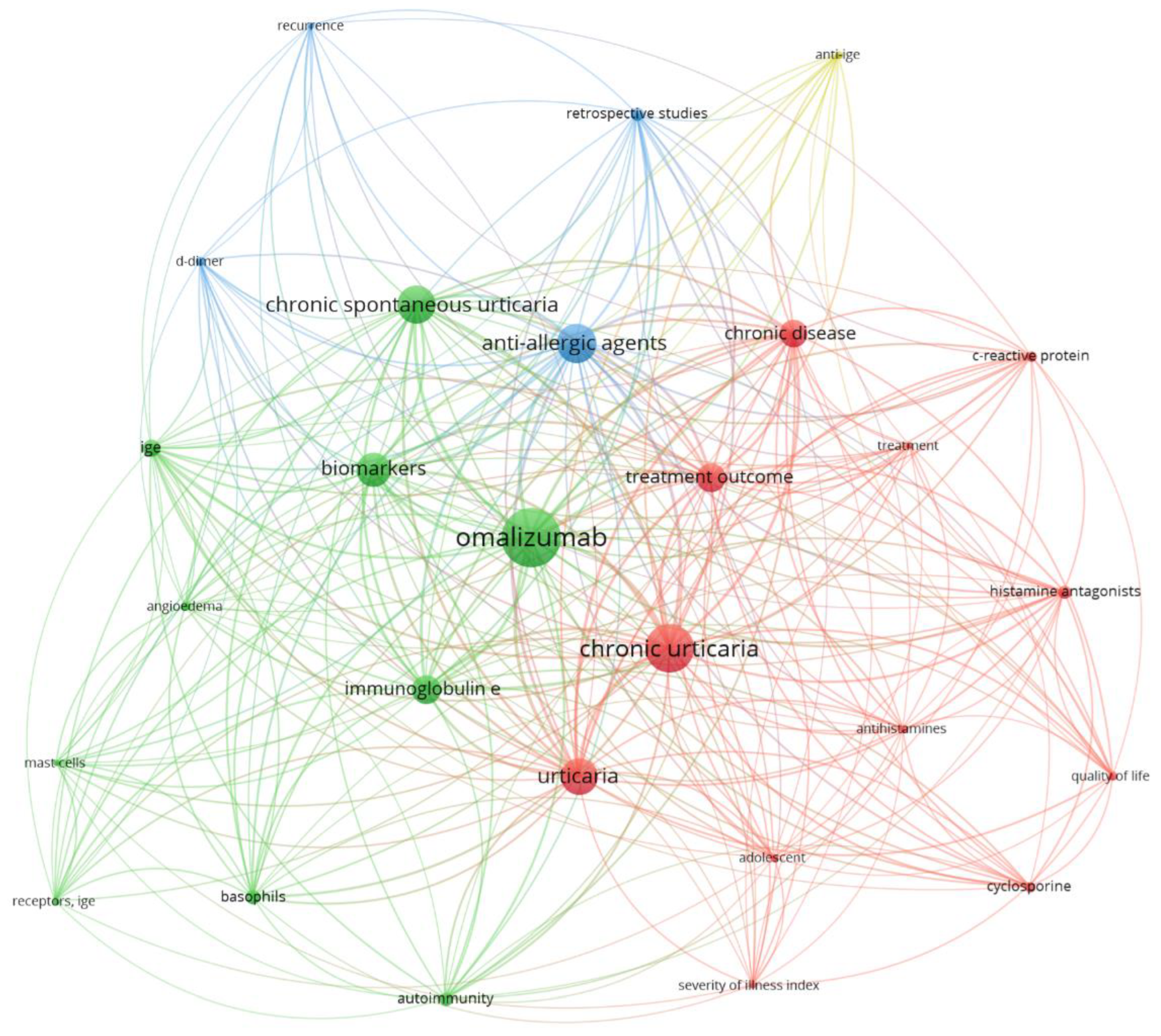
2. Classification and Epidemiological Aspects
3. Pathophysiological Mechanisms
3.1. Mast Cell and Basophil Activation
3.2. Autoimmune Endotypes
3.3. Inflammatory Networks
4. Emerging Biomarkers
5. Therapeutic Advances
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ASST | Autologous Serum Skin Test |
| BAT | Basophil Activation Test |
| BTK | Bruton’s Tyrosine Kinase |
| CU | Chronic Urticaria |
| CSU | Chronic Spontaneous Urticaria |
| EAACI | European Academy of Allergy and Clinical Immunology |
| FcεRI | High-Affinity IgE Receptor |
| GA²LEN | Global Allergy and Asthma European Network |
| H1 | Histamine Type 1 Receptor |
| IgE | Immunoglobulin E |
| IgG | Immunoglobulin G |
| MRGPRX2 | Mas-Related G Protein-Coupled Receptor X2 |
| RCT | Randomized Controlled Trial |
| Th2 | T Helper Type 2 Cells |
| Th17 | T Helper Type 17 Cells |
| WAO | World Allergy Organization |
References
- Zuberbier, T.; et al. Chronic urticaria: Unmet needs, emerging drugs, and new perspectives on personalised treatment. Lancet. 2024, 404, 1218–1231. [Google Scholar] [CrossRef]
- Kaplan, A.P.; Giménez-Arnau, A.M.; Saini, S.S. Chronic spontaneous urticaria: Focus on pathophysiology to unlock treatment advances. Allergy. 2022, 78, 389–401. [Google Scholar] [CrossRef]
- Bernstein, J.A.; Lang, D.M.; Khan, D.A. Chronic spontaneous urticaria. Immunol. Allergy Clin. North Am. 2024, 44, 381–398. [Google Scholar] [CrossRef]
- Kolkhir, P.; Church, M.K.; Weller, K.; Metz, M.; Schmetzer, O.; Maurer, M. Autoimmune chronic spontaneous urticaria. J. Allergy Clin. Immunol. 2022, 149, 1819–1831. [Google Scholar] [CrossRef]
- Greiner, A.N. Pathophysiology, diagnosis, and management of chronic spontaneous urticaria: A literature review. Clin. Rev. Allergy Immunol. 2022, 64, 194–211. [Google Scholar] [CrossRef]
- Kim, S.H.; et al. Advances in pathophysiology and therapeutic paradigm shifts in chronic spontaneous urticaria. Dermatol. Ther. 2025, 15, 1–18. [Google Scholar] [CrossRef]
- He, A.; et al. Chronic urticaria: Advances in understanding of the disease and clinical management. Clin. Rev. Allergy Immunol. 2021, 61, 424–448. [Google Scholar] [CrossRef]
- Yosipovitch, G.; et al. Current and emerging therapies for chronic spontaneous urticaria: A narrative review. Dermatol. Ther. 2023, 13, 1771–1794. [Google Scholar] [CrossRef] [PubMed]
- Asero, R.; et al. Biologics for chronic spontaneous urticaria: Toward a personalized treatment. Expert Rev. Clin. Immunol. 2022, 18, 1229–1240. [Google Scholar] [CrossRef] [PubMed]
- Giménez-Arnau, A.M.; et al. Recent updates in urticaria. Med. Clin. (Barc.). 2023, 161, 311–317. [Google Scholar] [CrossRef]
- Melchers, S.; et al. Chronic spontaneous urticaria—Status quo and future. Allergo J. Int. 2023, 32, 229–238. [Google Scholar] [CrossRef]
- Yolcu, S.; et al. Current view of chronic spontaneous urticaria. Astım Allerji İmmünoloji. 2023, 21, 67–76. [Google Scholar] [CrossRef]
- Puxeddu, I.; et al. Mast cell signaling and its role in urticaria. Ann. Allergy Asthma Immunol. 2024, 133, 256–265. [Google Scholar] [CrossRef]
- Grimalt, R. Chronic spontaneous urticaria: A changing paradigm. Allergies 2026, 6, 4. [Google Scholar] [CrossRef]
- Asero, R. Therapies for chronic spontaneous urticaria: Present and future. Pharmaceuticals. 2024, 17, 1499. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, J.A.; et al. Biomarkers for monitoring treatment response of omalizumab in patients with chronic urticaria. Int. J. Mol. Sci. 2023, 24, 11328. [Google Scholar] [CrossRef] [PubMed]
- Yanase, Y.; et al. Basophil characteristics as a marker of the pathogenesis of chronic spontaneous urticaria. Int. J. Mol. Sci. 2023, 24, 10320. [Google Scholar] [CrossRef]
- Sardina, D.S.; Valenti, G.; Papia, F.; Uasuf, C.G. Exploring machine learning techniques to predict the response to omalizumab in chronic spontaneous urticaria. Diagnostics 2021, 11, 2150. [Google Scholar] [CrossRef]
- Calzari, P.; et al. Omalizumab for chronic spontaneous urticaria in adults and adolescents: An eight-year real-life experience. J. Clin. Med. 2024, 13, 5610. [Google Scholar] [CrossRef]
- Manzoor, H.; et al. Meta-analysis of omalizumab dosing regimens in chronic spontaneous urticaria. Healthcare 2022, 10, 2579. [Google Scholar] [CrossRef]
- Bernstein, J.A.; et al. Why a complete response is the treatment aim in chronic spontaneous urticaria. J. Clin. Med. 2023, 12, 3561. [Google Scholar] [CrossRef] [PubMed]
- Manti, S.; et al. Monoclonal antibodies in the treatment of chronic spontaneous urticaria. J. Clin. Med. 2022, 11, 4453. [Google Scholar] [CrossRef]
- Robak, E.; et al. Bruton’s tyrosine kinase inhibitors for the treatment of immunological diseases. J. Clin. Med. 2022, 11, 2807. [Google Scholar] [CrossRef]
- Rizzi, A.; et al. Alarmins in Chronic Spontaneous Urticaria: Immunological Insights and Therapeutic Perspectives. Biomedicines. 2024, 12, 2765. [Google Scholar] [CrossRef]
- Bartosik, B.; et al. Chronic Spontaneous Urticaria—New Predictor on the Horizon? J. Clin. Med. 2024, 13, 6812. [Google Scholar] [CrossRef]
- Georgescu, M.; et al. Beyond the Skin: Exploring the Gut–Skin Axis in Chronic Spontaneous Urticaria and Other Inflammatory Skin Diseases. Biomedicines. 2025, 13, 2014. [Google Scholar] [CrossRef]
- Galletta, F.; et al. Efficacy and Safety of Omalizumab and Dupilumab in Pediatric Patients with Skin Diseases: An Observational Study J. Pers. Med. 2025, 15, 64. [Google Scholar] [CrossRef] [PubMed]
- Jutel, M.; et al. Nomenclature of allergic diseases and hypersensitivity reactions: Adapted to modern needs: An EAACI position paper. Allergy. 2023, 78, 2851–2874. [Google Scholar] [CrossRef]
- Chiang, V.; et al. Disproportionate rate of female referrals for suspected COVID-19 vaccine allergies. Allergy. 2022, 78, 308–310. [Google Scholar] [CrossRef]
- Pongdee, T. Omalizumab safety concerns. J. Allergy Clin. Immunol. Pract. 2024, 12, 01183-7. [Google Scholar] [CrossRef]
- Mulani, A.O.; et al. Transforming Drug Therapy with Deep Learning: The Future of Personalized Medicine. Drug Res. 2025, 75, 326–333. [Google Scholar] [CrossRef] [PubMed]
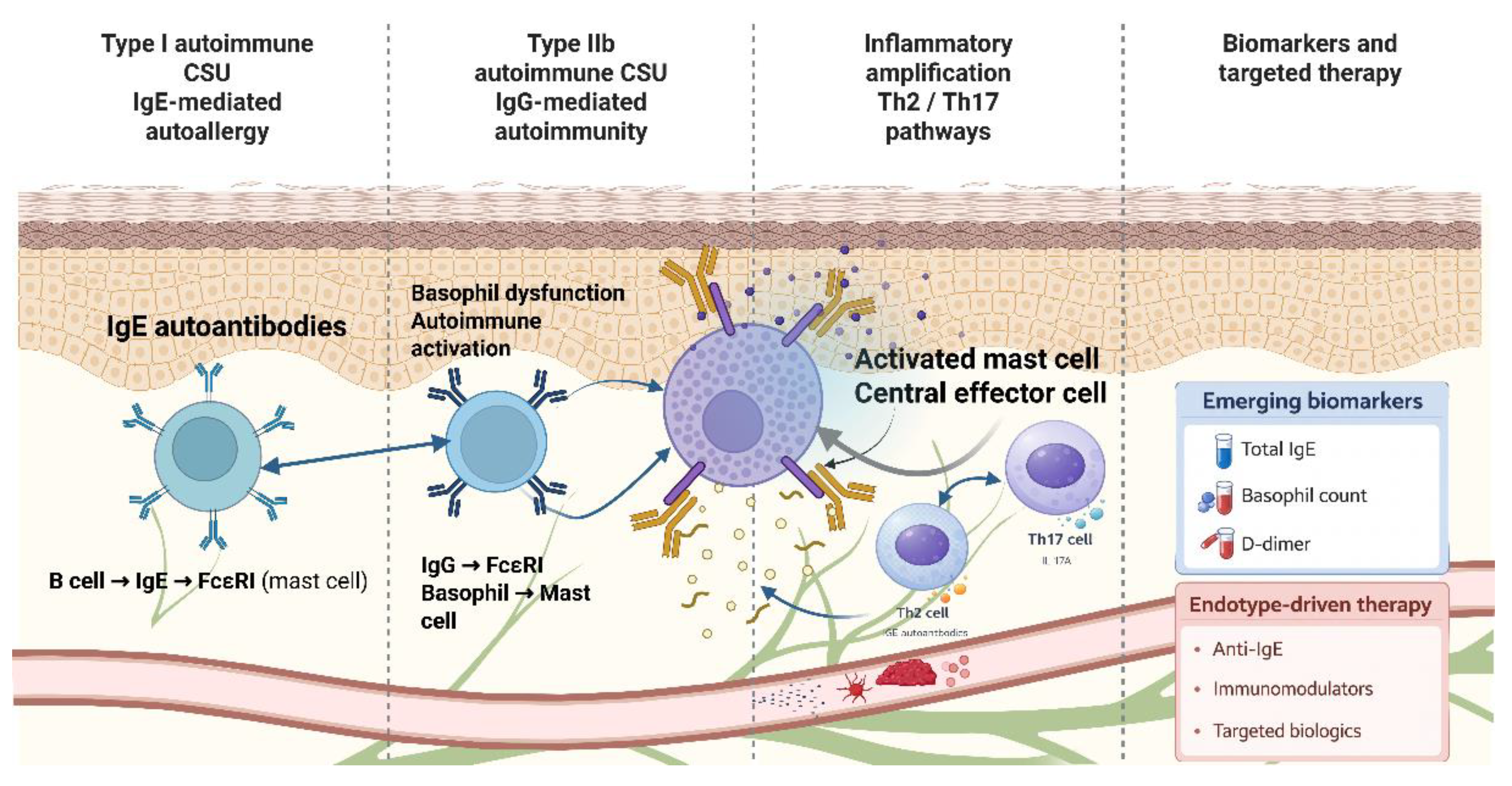
| Endotype | Dominant Immune Mechanism | Key Immunological Features | Representative Biomarkers | Therapeutic Implications |
|---|---|---|---|---|
| Type I autoimmune CSU | IgE-mediated autoallergy | Mast cell activation via FcεRI; Th2-skewed response | High total IgE | Good response to anti-IgE therapy |
| Type IIb autoimmune CSU | IgG-mediated autoimmunity | Autoantibodies against FcεRI or IgE; basophil dysfunction | Low IgE; basopenia; positive BAT/ASST | Reduced response to anti-IgE; benefit from immunomodulators |
| Inflammatory CSU | Innate and adaptive immune dysregulation | Th2/Th17 pathways; non-IgE mast cell activation | Normal IgE; elevated inflammatory markers | Potential benefit from targeted biologics |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




