Submitted:
04 February 2026
Posted:
04 February 2026
You are already at the latest version
Abstract
Keywords:
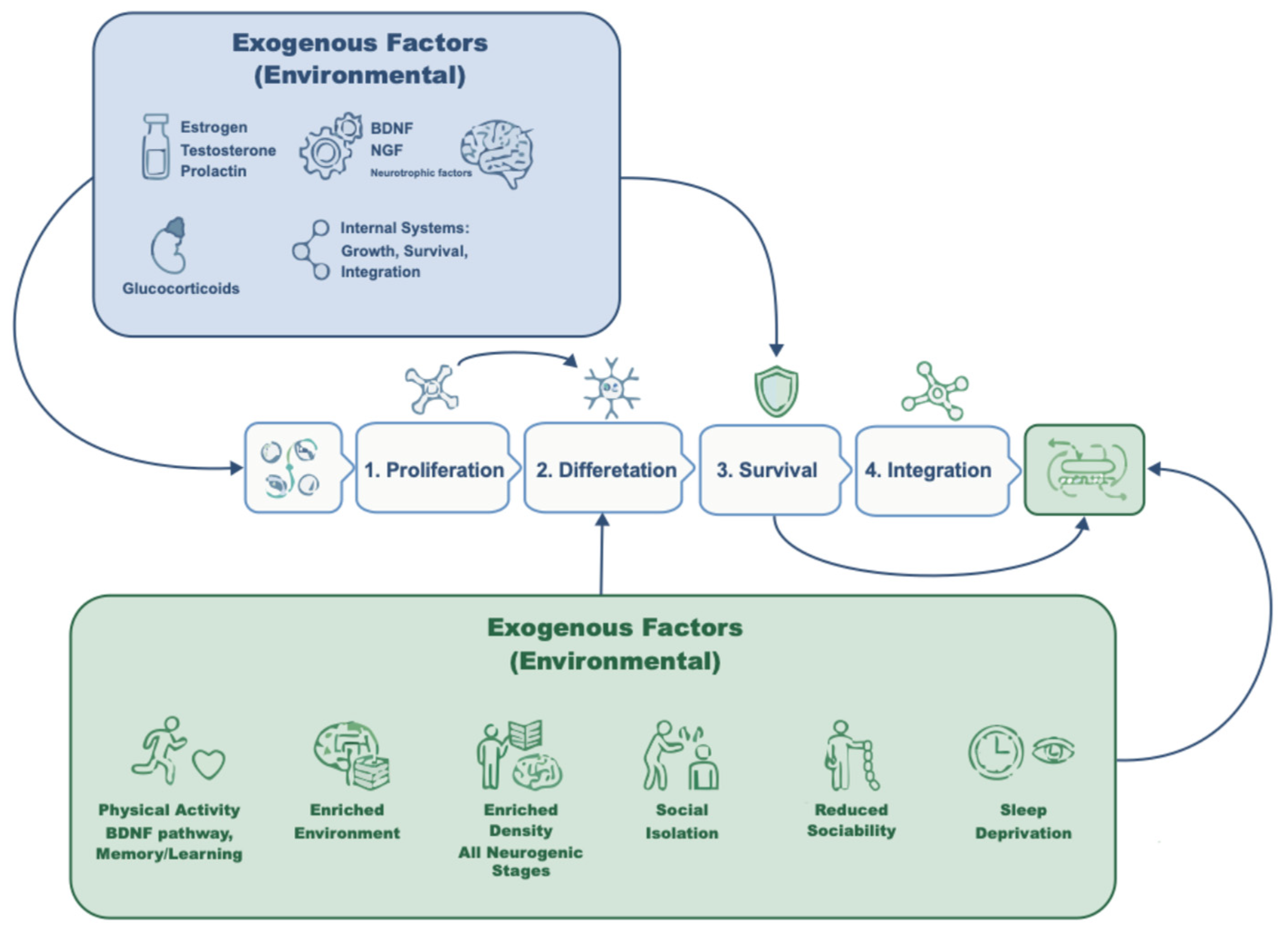
1. Introduction
2. Socialization
3. Isolation
4. Social Hierarchy
5. Social Buffering
6. Social Learning & Social Memory
7. Sex Differences and Sexual Dimorphism
8. Parenting
9. Sexual Interaction
- recruitment of mesolimbic dopamine circuits as a socially rewarding stimulus;
- estradiol-dependent increases in progenitor proliferation across the estrous cycle;
- testosterone-dependent enhancement of new-neuron survival; and
- experience-driven modulation of reward and stress pathways that jointly support adaptive hippocampal plasticity.
11. Comparative Models
12. Conclusions and Future Directions
Funding
Conflicts of Interest
Abbreviations
| AHN | Adult Hippocampal Neurogenesis |
| AVP | Arginine Vasopressin |
| BDNF | Brain-Derived Neurotrophic Factor |
| BrdU | Bromodeoxyuridine |
| CA1 | Cornu Ammonis 1 |
| CA2 | Cornu Ammonis 2 |
| CA3 | Cornu Ammonis 3 |
| DCX | Doublecortin |
| DG | Dentate Gyrus |
| dDG | Dorsal Dentate Gyrus |
| DHT | Dihydrotestosterone |
| EEG | Electroencephalography |
| ERα | Estrogen Receptor Alpha |
| ERβ | Estrogen Receptor Beta |
| HPA axis | Hypothalamic–Pituitary–Adrenal Axis |
| HPG axis | Hypothalamic–Pituitary–Gonadal Axis |
| HVC | High Vocal Center |
| LH | Luteinizing Hormone |
| mPFC | Medial Prefrontal Cortex |
| NAc | Nucleus Accumbens |
| NGF | Nerve Growth Factor |
| OXT | Oxytocin |
| PFC | Prefrontal Cortex |
| RA | Robust Nucleus of the Arcopallium |
| RNA-seq | RNA Sequencing |
| SVZ | Subventricular Zone |
| TrkB | Tropomyosin Receptor Kinase B |
| vDG | Ventral Dentate Gyrus |
| VTA | Ventral Tegmental Area |
| ΔFosB | Truncated FosB Transcription Factor |
References
- Taverna, E.; Götz, M.; Huttner, W.B. The Cell Biology of Neurogenesis: Toward an Understanding of the Development and Evolution of the Neocortex. Annu. Rev. Cell Dev. Biol. 2014, 30, 465–502. [Google Scholar] [CrossRef] [PubMed]
- Gross, C.G. Neurogenesis in the Adult Brain: Death of a Dogma. Nat Rev Neurosci 2000, 1, 67–73. [Google Scholar] [CrossRef]
- Jorgensen, C.; Wang, Z. Hormonal Regulation of Mammalian Adult Neurogenesis: A Multifaceted Mechanism. Biomolecules 2020, 10, 1151. [Google Scholar] [CrossRef] [PubMed]
- Kempermann, G. Environmental Enrichment, New Neurons and the Neurobiology of Individuality. Nat Rev Neurosci 2019, 20, 235–245. [Google Scholar] [CrossRef]
- Jorgensen, C. Adult Mammalian Neurogenesis and Motivated Behaviors. Integrative Zoology 2018, 13, 655–672. [Google Scholar] [CrossRef]
- Liu, P.Z.; Nusslock, R. Exercise-Mediated Neurogenesis in the Hippocampus via BDNF. Front. Neurosci. 2018, 12, 52. [Google Scholar] [CrossRef]
- Choi, D.-H.; Lee, K.-H.; Lee, J. Effect of Exercise-Induced Neurogenesis on Cognitive Function Deficit in a Rat Model of Vascular Dementia. Molecular Medicine Reports 2016, 13, 2981–2990. [Google Scholar] [CrossRef]
- Ma, C.-L.; Ma, X.-T.; Wang, J.-J.; Liu, H.; Chen, Y.-F.; Yang, Y. Physical Exercise Induces Hippocampal Neurogenesis and Prevents Cognitive Decline. Behavioural Brain Research 2017, 317, 332–339. [Google Scholar] [CrossRef]
- Duzel, E.; Van Praag, H.; Sendtner, M. Can Physical Exercise in Old Age Improve Memory and Hippocampal Function? Brain 2016, 139, 662–673. [Google Scholar] [CrossRef]
- Bekinschtein, P.; Oomen, C.A.; Saksida, L.M.; Bussey, T.J. Effects of Environmental Enrichment and Voluntary Exercise on Neurogenesis, Learning and Memory, and Pattern Separation: BDNF as a Critical Variable? Seminars in Cell & Developmental Biology 2011, 22, 536–542. [Google Scholar] [CrossRef] [PubMed]
- García-Gómez, L.; Castillo-Fernández, I.; Perez-Villalba, A. In the Pursuit of New Social Neurons. Neurogenesis and Social Behavior in Mice: A Systematic Review. Front. Cell Dev. Biol. 2022, 10, 1011657. [Google Scholar] [CrossRef]
- Opendak, M.; Briones, B.A.; Gould, E. Social Behavior, Hormones and Adult Neurogenesis. Frontiers in Neuroendocrinology 2016, 41, 71–86. [Google Scholar] [CrossRef]
- Leal-Galicia, P.; Chávez-Hernández, M.E.; Mata, F.; Mata-Luévanos, J.; Rodríguez-Serrano, L.M.; Tapia-de-Jesús, A.; Buenrostro-Jáuregui, M.H. Adult Neurogenesis: A Story Ranging from Controversial New Neurogenic Areas and Human Adult Neurogenesis to Molecular Regulation. IJMS 2021, 22, 11489. [Google Scholar] [CrossRef]
- Leblanc, H.; Ramirez, S. Linking Social Cognition to Learning and Memory. J. Neurosci. 2020, 40, 8782–8798. [Google Scholar] [CrossRef]
- Lemos, C.; Salti, A.; Amaral, I.M.; Fontebasso, V.; Singewald, N.; Dechant, G.; Hofer, A.; El Rawas, R. Social Interaction Reward in Rats Has Anti-stress Effects. Addiction Biology 2021, 26, e12878. [Google Scholar] [CrossRef] [PubMed]
- Seebacher, F.; Krause, J. Physiological Mechanisms Underlying Animal Social Behaviour. Phil. Trans. R. Soc. B 2017, 372, 20160231. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.M. Social Regulation of Adult Neurogenesis: A Comparative Approach. Frontiers in Neuroendocrinology 2016, 41, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Bell, M.R.; De Lorme, K.C.; Figueira, R.J.; Kashy, D.A.; Sisk, C.L. Adolescent Gain in Positive Valence of a Socially Relevant Stimulus: Engagement of the Mesocorticolimbic Reward Circuitry. Eur J of Neuroscience 2013, 37, 457–468. [Google Scholar] [CrossRef]
- Ibi, D.; Takuma, K.; Koike, H.; Mizoguchi, H.; Tsuritani, K.; Kuwahara, Y.; Kamei, H.; Nagai, T.; Yoneda, Y.; Nabeshima, T.; et al. Social Isolation Rearing-induced Impairment of the Hippocampal Neurogenesis Is Associated with Deficits in Spatial Memory and Emotion-related Behaviors in Juvenile Mice. Journal of Neurochemistry 2008, 105, 921–932. [Google Scholar] [CrossRef]
- Rivera, D.S.; Lindsay, C.B.; Oliva, C.A.; Codocedo, J.F.; Bozinovic, F.; Inestrosa, N.C. Effects of Long-Lasting Social Isolation and Re-Socialization on Cognitive Performance and Brain Activity: A Longitudinal Study in Octodon Degus. Sci Rep 2020, 10, 18315. [Google Scholar] [CrossRef]
- Arzate-Mejía, R.G.; Lottenbach, Z.; Schindler, V.; Jawaid, A.; Mansuy, I.M. Long-Term Impact of Social Isolation and Molecular Underpinnings. Front. Genet. 2020, 11, 589621. [Google Scholar] [CrossRef]
- He, J.; Crews, F.T. Neurogenesis Decreases during Brain Maturation from Adolescence to Adulthood. Pharmacology Biochemistry and Behavior 2007, 86, 327–333. [Google Scholar] [CrossRef]
- Duarte-Guterman, P.; Yagi, S.; Chow, C.; Galea, L.A.M. Hippocampal Learning, Memory, and Neurogenesis: Effects of Sex and Estrogens across the Lifespan in Adults. Hormones and Behavior 2015, 74, 37–52. [Google Scholar] [CrossRef]
- Galea, L.A.M.; Wainwright, S.R.; Roes, M.M.; Duarte-Guterman, P.; Chow, C.; Hamson, D.K. Sex, Hormones and Neurogenesis in the Hippocampus: Hormonal Modulation of Neurogenesis and Potential Functional Implications. J Neuroendocrinology 2013, 25, 1039–1061. [Google Scholar] [CrossRef]
- McCormick, C.M.; Nixon, F.; Thomas, C.; Lowie, B.; Dyck, J. Hippocampal Cell Proliferation and Spatial Memory Performance after Social Instability Stress in Adolescence in Female Rats. Behavioural Brain Research 2010, 208, 23–29. [Google Scholar] [CrossRef]
- Lu, L.; Bao, G.; Chen, H.; Xia, P.; Fan, X.; Zhang, J.; Pei, G.; Ma, L. Modification of Hippocampal Neurogenesis and Neuroplasticity by Social Environments. Experimental Neurology 2003, 183, 600–609. [Google Scholar] [CrossRef]
- Rizzi, S.; Bianchi, P.; Guidi, S.; Ciani, E.; Bartesaghi, R. Neonatal Isolation Impairs Neurogenesis in Thedentate Gyrus of the Guinea Pig. Hippocampus 2007, 17, 78–91. [Google Scholar] [CrossRef] [PubMed]
- Bloch, J.; Kaeser, M.; Sadeghi, Y.; Rouiller, E.M.; Redmond, D.E.; Brunet, J. Doublecortin-positive Cells in the Adult Primate Cerebral Cortex and Possible Role in Brain Plasticity and Development. J of Comparative Neurology 2011, 519, 775–789. [Google Scholar] [CrossRef] [PubMed]
- Dranovsky, A.; Picchini, A.M.; Moadel, T.; Sisti, A.C.; Yamada, A.; Kimura, S.; Leonardo, E.D.; Hen, R. Experience Dictates Stem Cell Fate in the Adult Hippocampus. Neuron 2011, 70, 908–923. [Google Scholar] [CrossRef] [PubMed]
- Lieberwirth, C.; Liu, Y.; Jia, X.; Wang, Z. Social Isolation Impairs Adult Neurogenesis in the Limbic System and Alters Behaviors in Female Prairie Voles. Hormones and Behavior 2012, 62, 357–366. [Google Scholar] [CrossRef]
- Westenbroek, C.; Den Boer, J.A.; Veenhuis, M.; Ter Horst, G.J. Chronic Stress and Social Housing Differentially Affect Neurogenesis in Male and Female Rats. Brain Research Bulletin 2004, 64, 303–308. [Google Scholar] [CrossRef]
- Biggio, F.; Mostallino, M.C.; Talani, G.; Locci, V.; Mostallino, R.; Calandra, G.; Sanna, E.; Biggio, G. Social Enrichment Reverses the Isolation-Induced Deficits of Neuronal Plasticity in the Hippocampus of Male Rats. Neuropharmacology 2019, 151, 45–54. [Google Scholar] [CrossRef]
- Scaccianoce, S.; Del Bianco, P.; Paolone, G.; Caprioli, D.; Modafferi, A.M.E.; Nencini, P.; Badiani, A. Social Isolation Selectively Reduces Hippocampal Brain-Derived Neurotrophic Factor without Altering Plasma Corticosterone. Behavioural Brain Research 2006, 168, 323–325. [Google Scholar] [CrossRef]
- Kowiański, P.; Lietzau, G.; Czuba, E.; Waśkow, M.; Steliga, A.; Moryś, J. BDNF: A Key Factor with Multipotent Impact on Brain Signaling and Synaptic Plasticity. Cell Mol Neurobiol 2018, 38, 579–593. [Google Scholar] [CrossRef]
- Castrén, E.; Antila, H. Neuronal Plasticity and Neurotrophic Factors in Drug Responses. Mol Psychiatry 2017, 22, 1085–1095. [Google Scholar] [CrossRef]
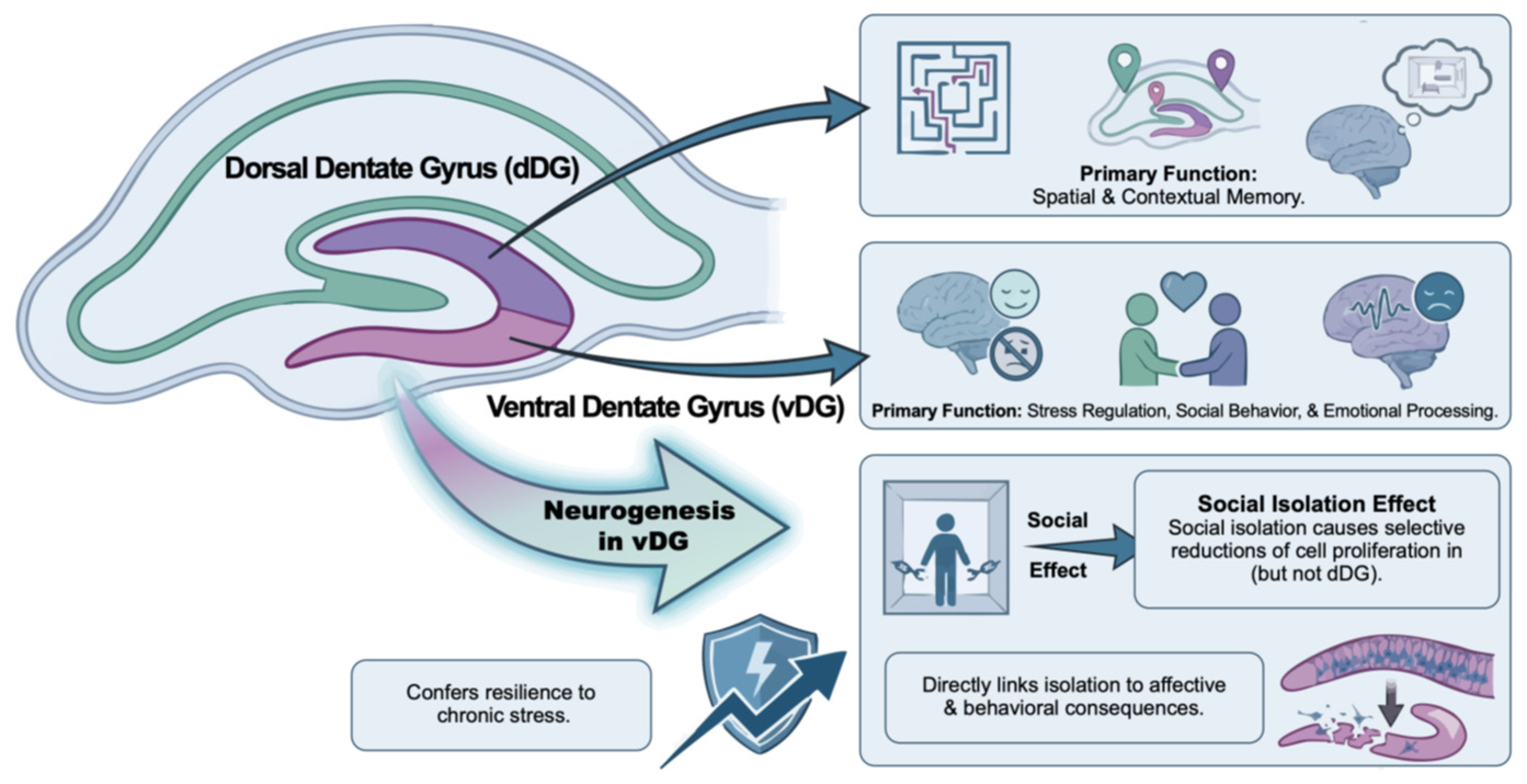
- Anacker, C.; Luna, V.M.; Stevens, G.S.; Millette, A.; Shores, R.; Jimenez, J.C.; Chen, B.; Hen, R. Hippocampal Neurogenesis Confers Stress Resilience by Inhibiting the Ventral Dentate Gyrus. Nature 2018, 559, 98–102. [Google Scholar] [CrossRef] [PubMed]
- Hanif, S.; Sclar, M.; Lee, J.; Nichols, C.; Likhtik, E.; Burghardt, N.S. Social Isolation during Adolescence Differentially Affects Spatial Learning in Adult Male and Female Mice. Learn. Mem. 2025, 32, a054059. [Google Scholar] [CrossRef]
- Karimian, M.; Famitafreshi, H.; Fanaei, H.; Attari, F.; Fatima, S. Social Isolation Is Associated with Reduced Neurogenesis, Impaired Spatial Working Memory Performance, and Altered Anxiety Levels in Male Rats. OAAP 2015, 87. [Google Scholar] [CrossRef]
- Dimonte, S.; Sikora, V.; Bove, M.; Morgese, M.G.; Tucci, P.; Schiavone, S.; Trabace, L. Social Isolation from Early Life Induces Anxiety-like Behaviors in Adult Rats: Relation to Neuroendocrine and Neurochemical Dysfunctions. Biomedicine & Pharmacotherapy 2023, 158, 114181. [Google Scholar] [CrossRef]
- Skelly, M.J.; Chappell, A.E.; Carter, E.; Weiner, J.L. Adolescent Social Isolation Increases Anxiety-like Behavior and Ethanol Intake and Impairs Fear Extinction in Adulthood: Possible Role of Disrupted Noradrenergic Signaling. Neuropharmacology 2015, 97, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Flashner, B.; Chiu, M.; Ver Hoeve, E.; Luz, S.; Bhatnagar, S. Social Isolation in Adolescence Alters Behaviors in the Forced Swim and Sucrose Preference Tests in Female but Not in Male Rats. Physiology & Behavior 2012, 105, 269–275. [Google Scholar] [CrossRef]
- Hysing, M.; Petrie, K.J.; Harvey, A.G.; Sivertsen, B. From Loneliness to Depression: A Longitudinal Diagnostic Study among Norwegian University Students. Soc Psychiatry Psychiatr Epidemiol 2025. [Google Scholar] [CrossRef]
- Csikós, V.; Dóra, F.; Láng, T.; Darai, L.; Szendi, V.; Tóth, A.; Cservenák, M.; Dobolyi, A. Social Isolation Induces Changes in the Monoaminergic Signalling in the Rat Medial Prefrontal Cortex. Cells 2024, 13, 1043. [Google Scholar] [CrossRef]
- Benfato, I.D.; Quintanilha, A.C.S.; Henrique, J.S.; Souza, M.A.; Rosário, B.D.A.; Beserra Filho, J.I.A.; Santos, R.L.O.; Ribeiro, A.M.; Le Sueur Maluf, L.; De Oliveira, C.A.M. Effects of Long-Term Social Isolation on Central, Behavioural and Metabolic Parameters in Middle-Aged Mice. Behavioural Brain Research 2022, 417, 113630. [Google Scholar] [CrossRef]
- Paz, C.; Mascialino, G.; Adana-Díaz, L.; Rodríguez-Lorenzana, A.; Simbaña-Rivera, K.; Gómez-Barreno, L.; Troya, M.; Paez, M.I.; Cárdenas, J.; Gerstner, R.M.; et al. Behavioral and Sociodemographic Predictors of Anxiety and Depression in Patients under Epidemiological Surveillance for COVID-19 in Ecuador. PLoS ONE 2020, 15, e0240008. [Google Scholar] [CrossRef]
- Blanchard, D.C.; Spencer, R.L.; Weiss, S.M.; Blanchard, R.J.; McEwen, B.; Sakai, R.R. Visible Burrow System as a Model of Chronic Social Stress: Behavioral and Neuroendocrine Correlates. Psychoneuroendocrinology 1995, 20, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Kozorovitskiy, Y.; Gould, E. Dominance Hierarchy Influences Adult Neurogenesis in the Dentate Gyrus: Figure 1. J. Neurosci. 2004, 24, 6755–6759. [Google Scholar] [CrossRef] [PubMed]
- Pallé, A.; Zorzo, C.; Luskey, V.E.; Mcgreevy, K.R.; Fernández, S.; Trejo, J.L. Social Dominance Differentially Alters Gene Expression in the Medial Prefrontal Cortex without Affecting Adult Hippocampal Neurogenesis or Stress and Anxiety-like Behavior. The FASEB Journal 2019, 33, 6995–7008. [Google Scholar] [CrossRef] [PubMed]
- Falconer, E.M.; Galea, L.A.M. Sex Differences in Cell Proliferation, Cell Death and Defensive Behavior Following Acute Predator Odor Stress in Adult Rats. Brain Research 2003, 975, 22–36. [Google Scholar] [CrossRef]
- Holmes, M.M.; Galea, L.A.M. Defensive Behavior and Hippocampal Cell Proliferation: Differential Modulation by Naltrexone during Stress. Behavioral Neuroscience 2002, 116, 160–168. [Google Scholar] [CrossRef]
- Hardy, M.P.; Sottas, C.M.; Ge, R.; McKittrick, C.R.; Tamashiro, K.L.; McEwen, B.S.; Haider, S.G.; Markham, C.M.; Blanchard, R.J.; Blanchard, D.C.; et al. Trends of Reproductive Hormones in Male Rats During Psychosocial Stress: Role of Glucocorticoid Metabolism in Behavioral Dominance1. Biology of Reproduction 2002, 67, 1750–1755. [Google Scholar] [CrossRef]
- Blanchard, R.J.; Yudko, E.; Dulloog, L.; Blanchard, D.C. Defense Changes in Stress Nonresponsive Subordinate Males in a Visible Burrow System. Physiology & Behavior 2001, 72, 635–642. [Google Scholar] [CrossRef]
- Wu, M.V.; Shamy, J.L.; Bedi, G.; Choi, C.-W.J.; Wall, M.M.; Arango, V.; Boldrini, M.; Foltin, R.W.; Hen, R. Impact of Social Status and Antidepressant Treatment on Neurogenesis in the Baboon Hippocampus. Neuropsychopharmacol 2014, 39, 1861–1871. [Google Scholar] [CrossRef]
- Peragine, D.E.; Simpson, J.A.; Mooney, S.J.; Lovern, M.B.; Holmes, M.M. Social Regulation of Adult Neurogenesis in a Eusocial Mammal. Neuroscience 2014, 268, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Golden, S.A.; Covington, H.E.; Berton, O.; Russo, S.J. A Standardized Protocol for Repeated Social Defeat Stress in Mice. Nat Protoc 2011, 6, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, V.; Han, M.-H.; Graham, D.L.; Berton, O.; Renthal, W.; Russo, S.J.; LaPlant, Q.; Graham, A.; Lutter, M.; Lagace, D.C.; et al. Molecular Adaptations Underlying Susceptibility and Resistance to Social Defeat in Brain Reward Regions. Cell 2007, 131, 391–404. [Google Scholar] [CrossRef]
- Morrison, K.E.; Bader, L.R.; McLaughlin, C.N.; Cooper, M.A. Defeat-Induced Activation of the Ventral Medial Prefrontal Cortex Is Necessary for Resistance to Conditioned Defeat. Behavioural Brain Research 2013, 243, 158–164. [Google Scholar] [CrossRef]
- Jasnow, A.M.; Cooper, M.A.; Huhman, K.L. N-Methyl-d-Aspartate Receptors in the Amygdala Are Necessary for the Acquisition and Expression of Conditioned Defeat. Neuroscience 2004, 123, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Hodges, T.E.; Louth, E.L.; Bailey, C.D.C.; McCormick, C.M. Adolescent Social Instability Stress Alters Markers of Synaptic Plasticity and Dendritic Structure in the Medial Amygdala and Lateral Septum in Male Rats. Brain Struct Funct 2019, 224, 643–659. [Google Scholar] [CrossRef]
- McCormick, C.M.; Smith, K.; Baumbach, J.L.; De Lima, A.P.N.; Shaver, M.; Hodges, T.E.; Marcolin, M.L.; Ismail, N. Adolescent Social Instability Stress Leads to Immediate and Lasting Sex-Specific Changes in the Neuroendocrine-Immune-Gut Axis in Rats. Hormones and Behavior 2020, 126, 104845. [Google Scholar] [CrossRef]
- Adhikari, A.; Topiwala, M.A.; Gordon, J.A. Single Units in the Medial Prefrontal Cortex with Anxiety-Related Firing Patterns Are Preferentially Influenced by Ventral Hippocampal Activity. Neuron 2011, 71, 898–910. [Google Scholar] [CrossRef]
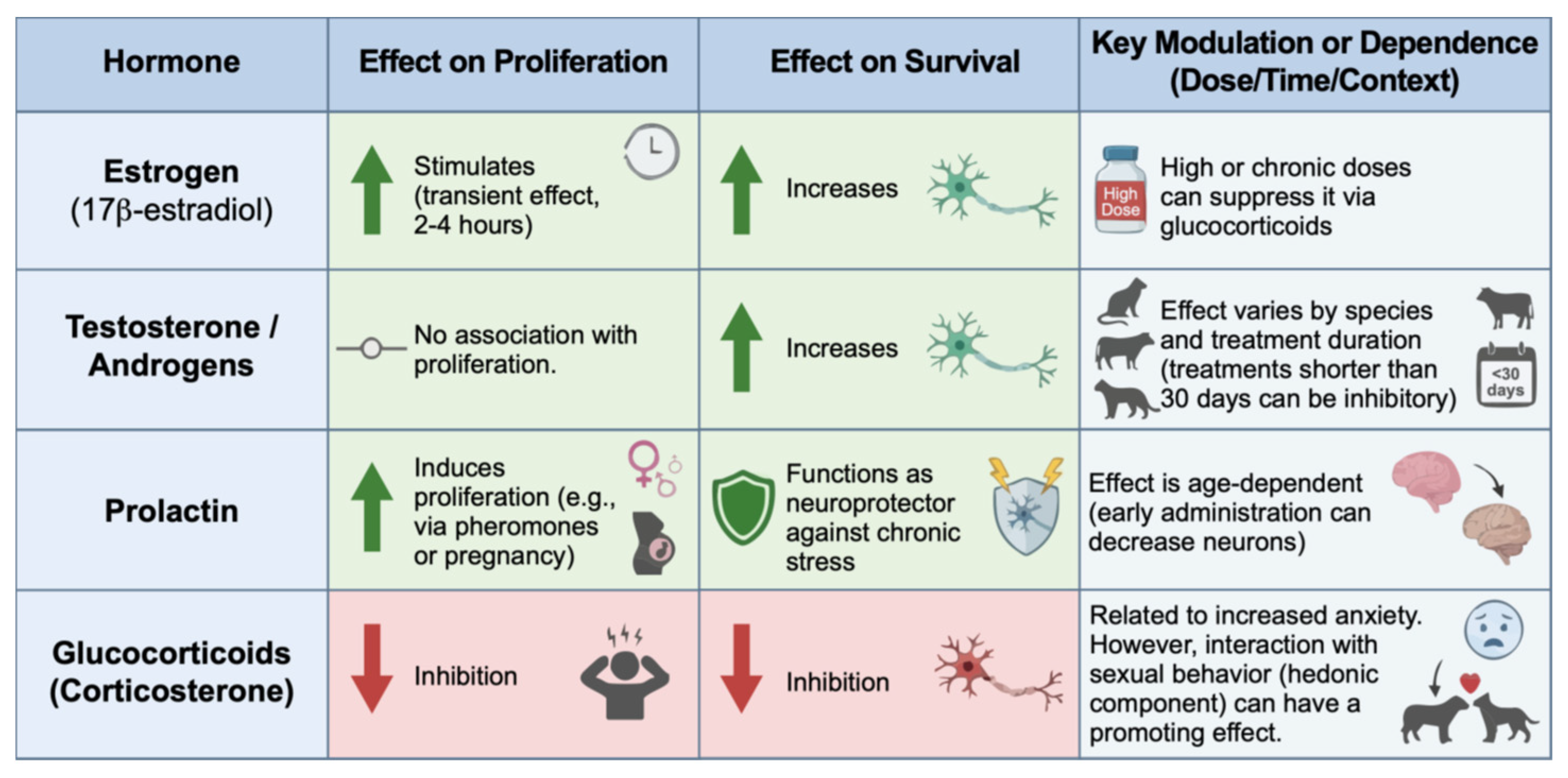
- Hamson, D.K.; Wainwright, S.R.; Taylor, J.R.; Jones, B.A.; Watson, N.V.; Galea, L.A.M. Androgens Increase Survival of Adult-Born Neurons in the Dentate Gyrus by an Androgen Receptor-Dependent Mechanism in Male Rats. Endocrinology 2013, 154, 3294–3304. [Google Scholar] [CrossRef] [PubMed]
- Spritzer, M.D.; Ibler, E.; Inglis, W.; Curtis, M.G. Testosterone and Social Isolation Influence Adult Neurogenesis in the Dentate Gyrus of Male Rats. Neuroscience 2011, 195, 180–190. [Google Scholar] [CrossRef] [PubMed]
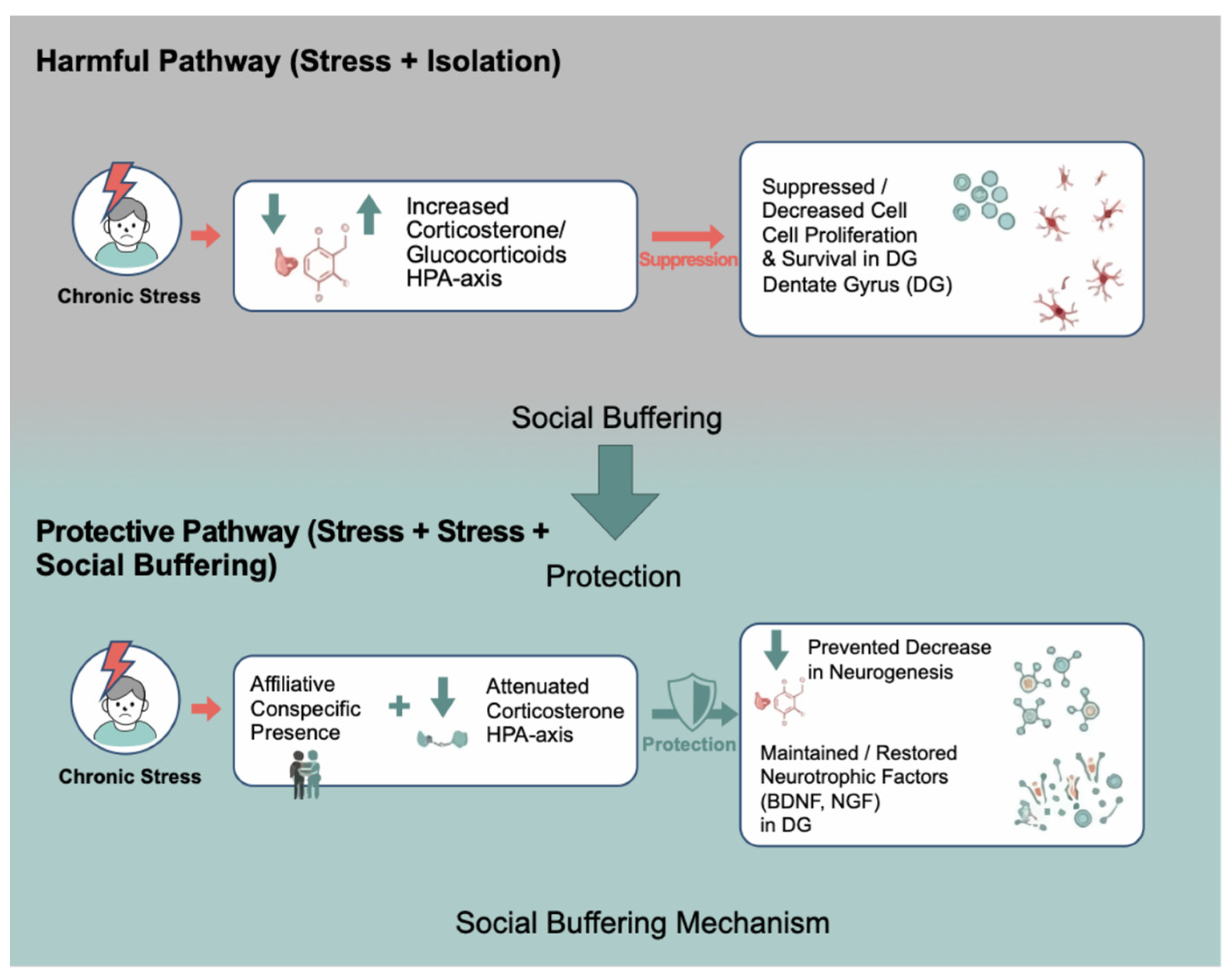
- Schoenfeld, T.J.; Gould, E. Stress, Stress Hormones, and Adult Neurogenesis. Experimental Neurology 2012, 233, 12–21. [Google Scholar] [CrossRef]
- Hostinar, C.E.; Sullivan, R.M.; Gunnar, M.R. Psychobiological Mechanisms Underlying the Social Buffering of the Hypothalamic–Pituitary–Adrenocortical Axis: A Review of Animal Models and Human Studies across Development. Psychological Bulletin 2014, 140, 256–282. [Google Scholar] [CrossRef]
- Gunnar, M.R.; Donzella, B. Social Regulation of the Cortisol Levels in Early Human Development. Psychoneuroendocrinology 2002, 27, 199–220. [Google Scholar] [CrossRef]
- Kiyokawa, Y.; Takeuchi, Y.; Mori, Y. Two Types of Social Buffering Differentially Mitigate Conditioned Fear Responses. Eur J of Neuroscience 2007, 26, 3606–3613. [Google Scholar] [CrossRef]
- Kiyokawa, Y.; Hiroshima, S.; Takeuchi, Y.; Mori, Y. Social Buffering Reduces Male Rats’ Behavioral and Corticosterone Responses to a Conditioned Stimulus. Hormones and Behavior 2014, 65, 114–118. [Google Scholar] [CrossRef]
- Caldwell, H.K. Oxytocin and Vasopressin: Powerful Regulators of Social Behavior. Neuroscientist 2017, 23, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Bielsky, I.F.; Young, L.J. Oxytocin, Vasopressin, and Social Recognition in Mammals. Peptides 2004, 25, 1565–1574. [Google Scholar] [CrossRef]
- Cilz, N.I.; Cymerblit-Sabba, A.; Young, W.S. Oxytocin and Vasopressin in the Rodent Hippocampus. Genes Brain and Behavior 2019, 18, e12535. [Google Scholar] [CrossRef]
- Wang, X.; Zhan, Y. Regulation of Social Recognition Memory in the Hippocampal Circuits. Front. Neural Circuits 2022, 16, 839931. [Google Scholar] [CrossRef]
- Lopez-Rojas, J.; De Solis, C.A.; Leroy, F.; Kandel, E.R.; Siegelbaum, S.A. A Direct Lateral Entorhinal Cortex to Hippocampal CA2 Circuit Conveys Social Information Required for Social Memory. Neuron 2022, 110, 1559–1572.e4. [Google Scholar] [CrossRef]
- Cope, E.C.; Waters, R.C.; Diethorn, E.J.; Pagliai, K.A.; Dias, C.G.; Tsuda, M.; Cameron, H.A.; Gould, E. Adult-Born Neurons in the Hippocampus Are Essential for Social Memory Maintenance. eNeuro 2020, 7, ENEURO.0182–20. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, B.M.M.; Moreira, F.A.; Massensini, A.R.; Moraes, M.F.D.; Pereira, G.S. Enriched Environment Increases Neurogenesis and Improves Social Memory Persistence in Socially Isolated Adult Mice: Neurogenesis Supports Social Memory Persistence. Hippocampus 2014, 24, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Bangasser, D.A.; Wicks, B. Sex-specific Mechanisms for Responding to Stress. J of Neuroscience Research 2017, 95, 75–82. [Google Scholar] [CrossRef]
- Gobinath, A.R.; Mahmoud, R.; Galea, L.A.M. Influence of Sex and Stress Exposure across the Lifespan on Endophenotypes of Depression: Focus on Behavior, Glucocorticoids, and Hippocampus. Front. Neurosci. 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Bonilla-Jaime, H.; Vázquez-Palacios, G.; Arteaga-Silva, M.; Retana-Márquez, S. Hormonal Responses to Different Sexually Related Conditions in Male Rats. Hormones and Behavior 2006, 49, 376–382. [Google Scholar] [CrossRef]
- Spritzer, M.D.; Galea, L.A.M. Testosterone and Dihydrotestosterone, but Not Estradiol, Enhance Survival of New Hippocampal Neurons in Adult Male Rats. Developmental Neurobiology 2007, 67, 1321–1333. [Google Scholar] [CrossRef]
- Shingo, T.; Gregg, C.; Enwere, E.; Fujikawa, H.; Hassam, R.; Geary, C.; Cross, J.C.; Weiss, S. Pregnancy-Stimulated Neurogenesis in the Adult Female Forebrain Mediated by Prolactin. Science 2003, 299, 117–120. [Google Scholar] [CrossRef]
- Leuner, B.; Glasper, E.R.; Gould, E. Parenting and Plasticity. Trends in Neurosciences 2010, 33, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Pawluski, J.L.; Galea, L.A.M. Reproductive Experience Alters Hippocampal Neurogenesis during the Postpartum Period in the Dam. Neuroscience 2007, 149, 53–67. [Google Scholar] [CrossRef]
- Bale, T.L.; Epperson, C.N. Sex Differences and Stress across the Lifespan. Nat Neurosci 2015, 18, 1413–1420. [Google Scholar] [CrossRef] [PubMed]
- Leuner, B.; Mirescu, C.; Noiman, L.; Gould, E. Maternal Experience Inhibits the Production of Immature Neurons in the Hippocampus during the Postpartum Period through Elevations in Adrenal Steroids. Hippocampus 2007, 17, 434–442. [Google Scholar] [CrossRef]
- Hillerer, K.M.; Neumann, I.D.; Couillard-Despres, S.; Aigner, L.; Slattery, D.A. Lactation-induced Reduction in Hippocampal Neurogenesis Is Reversed by Repeated Stress Exposure. Hippocampus 2014, 24, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Maeng, L.Y.; Shors, T.J. Once a Mother, Always a Mother: Maternal Experience Protects Females from the Negative Effects of Stress on Learning. Behavioral Neuroscience 2012, 126, 137–141. [Google Scholar] [CrossRef]
- Torner, L. Actions of Prolactin in the Brain: From Physiological Adaptations to Stress and Neurogenesis to Psychopathology. Front. Endocrinol. 2016, 7. [Google Scholar] [CrossRef]
- Saltzman, W.; Ziegler, T.E. Functional Significance of Hormonal Changes in Mammalian Fathers. J Neuroendocrinology 2014, 26, 685–696. [Google Scholar] [CrossRef]
- Glasper, E.R.; Kozorovitskiy, Y.; Pavlic, A.; Gould, E. Paternal Experience Suppresses Adult Neurogenesis without Altering Hippocampal Function in Peromyscus Californicus. J of Comparative Neurology 2011, 519, 2271–2281. [Google Scholar] [CrossRef]
- Glasper, E.R.; Hyer, M.M.; Katakam, J.; Harper, R.; Ameri, C.; Wolz, T. Fatherhood Contributes to Increased Hippocampal Spine Density and Anxiety Regulation in California Mice. Brain and Behavior 2016, 6, e00416. [Google Scholar] [CrossRef]
- Cárdenas, E.F.; Kujawa, A.; Humphreys, K.L. Neurobiological Changes during the Peripartum Period: Implications for Health and Behavior. Social Cognitive and Affective Neuroscience 2020, 15, 1097–1110. [Google Scholar] [CrossRef]
- Aguirre, R.M.H.; González, M.H.; Hernández, M.P.; Gutiérrez, C.D.C.A.; Guevara, M.Á. Observing Baby or Sexual Videos Changes the Functional Synchronization between the Prefrontal and Parietal Cortices in Mothers in Different Postpartum Periods. Social Neuroscience 2020, 15, 489–504. [Google Scholar] [CrossRef]
- Kim, P.; Leckman, J.F.; Mayes, L.C.; Feldman, R.; Wang, X.; Swain, J.E. The Plasticity of Human Maternal Brain: Longitudinal Changes in Brain Anatomy during the Early Postpartum Period. Behavioral Neuroscience 2010, 124, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Pfaus, J.G. Pathways of Sexual Desire. The Journal of Sexual Medicine 2009, 6, 1506–1533. [Google Scholar] [CrossRef]
- Paredes, R.G.; Ågmo, A. Has Dopamine a Physiological Role in the Control of Sexual Behavior? Progress in Neurobiology 2004, 73, 179–225. [Google Scholar] [CrossRef]
- Barha, C.K.; Lieblich, S.E.; Galea, L.A.M. Different Forms of Oestrogen Rapidly Upregulate Cell Proliferation in the Dentate Gyrus of Adult Female Rats. J Neuroendocrinology 2009, 21, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Tanapat, P.; Hastings, N.B.; Reeves, A.J.; Gould, E. Estrogen Stimulates a Transient Increase in the Number of New Neurons in the Dentate Gyrus of the Adult Female Rat. J. Neurosci. 1999, 19, 5792–5801. [Google Scholar] [CrossRef]
- Pawluski, J.L.; Brummelte, S.; Barha, C.K.; Crozier, T.M.; Galea, L.A.M. Effects of Steroid Hormones on Neurogenesis in the Hippocampus of the Adult Female Rodent during the Estrous Cycle, Pregnancy, Lactation and Aging. Frontiers in Neuroendocrinology 2009, 30, 343–357. [Google Scholar] [CrossRef]
- Fares, J.; Bou Diab, Z.; Nabha, S.; Fares, Y. Neurogenesis in the Adult Hippocampus: History, Regulation, and Prospective Roles. International Journal of Neuroscience 2019, 129, 598–611. [Google Scholar] [CrossRef]
- Nicolis Di Robilant, V.; Scardigli, R.; Strimpakos, G.; Tirone, F.; Middei, S.; Scopa, C.; De Bardi, M.; Battistini, L.; Saraulli, D.; Farioli Vecchioli, S. Running-Activated Neural Stem Cells Enhance Subventricular Neurogenesis and Improve Olfactory Behavior in P21 Knockout Mice. Mol Neurobiol 2019, 56, 7534–7556. [Google Scholar] [CrossRef] [PubMed]
- Overall, R.W.; Walker, T.L.; Leiter, O.; Lenke, S.; Ruhwald, S.; Kempermann, G. Delayed and Transient Increase of Adult Hippocampal Neurogenesis by Physical Exercise in DBA/2 Mice. PLoS ONE 2013, 8, e83797. [Google Scholar] [CrossRef]
- Barha, C.K.; Ishrat, T.; Epp, J.R.; Galea, L.A.M.; Stein, D.G. Progesterone Treatment Normalizes the Levels of Cell Proliferation and Cell Death in the Dentate Gyrus of the Hippocampus after Traumatic Brain Injury. Experimental Neurology 2011, 231, 72–81. [Google Scholar] [CrossRef]
- Ormerod, B.K.; Lee, T.T.; Galea, L.A.M. Estradiol Initially Enhances but Subsequently Suppresses (via Adrenal Steroids) Granule Cell Proliferation in the Dentate Gyrus of Adult Female Rats. J. Neurobiol. 2003, 55, 247–260. [Google Scholar] [CrossRef]
- Mahmoud, R.; Wainwright, S.R.; Galea, L.A.M. Sex Hormones and Adult Hippocampal Neurogenesis: Regulation, Implications, and Potential Mechanisms. Frontiers in Neuroendocrinology 2016, 41, 129–152. [Google Scholar] [CrossRef]
- Nugent, B.; Tobet, S.; Lara, H.; Lucion, A.; Wilson, M.; Recabarren, S.; Paredes, A. Hormonal Programming Across the Lifespan. Horm Metab Res 2012, 44, 577–586. [Google Scholar] [CrossRef]
- Nogami, H.; Hoshino, R.; Ogasawara, K.; Miyamoto, S.; Hisano, S. Region-Specific Expression and Hormonal Regulation of the First Exon Variants of Rat Prolactin Receptor mRNA in Rat Brain and Anterior Pituitary Gland. J Neuroendocrinology 2007, 19, 583–593. [Google Scholar] [CrossRef]
- Mak, G.K.; Enwere, E.K.; Gregg, C.; Pakarainen, T.; Poutanen, M.; Huhtaniemi, I.; Weiss, S. Male Pheromone–Stimulated Neurogenesis in the Adult Female Brain: Possible Role in Mating Behavior. Nat Neurosci 2007, 10, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Czéh, B.; Müller-Keuker, J.I.H.; Rygula, R.; Abumaria, N.; Hiemke, C.; Domenici, E.; Fuchs, E. Chronic Social Stress Inhibits Cell Proliferation in the Adult Medial Prefrontal Cortex: Hemispheric Asymmetry and Reversal by Fluoxetine Treatment. Neuropsychopharmacol 2007, 32, 1490–1503. [Google Scholar] [CrossRef] [PubMed]
- Mazzucco, C.A.; Lieblich, S.E.; Bingham, B.I.; Williamson, M.A.; Viau, V.; Galea, L.A.M. Both Estrogen Receptor α and Estrogen Receptor β Agonists Enhance Cell Proliferation in the Dentate Gyrus of Adult Female Rats. Neuroscience 2006, 141, 1793–1800. [Google Scholar] [CrossRef] [PubMed]
- McClure, R.E.S.; Barha, C.K.; Galea, L.A.M. 17β-Estradiol, but Not Estrone, Increases the Survival and Activation of New Neurons in the Hippocampus in Response to Spatial Memory in Adult Female Rats. Hormones and Behavior 2013, 63, 144–157. [Google Scholar] [CrossRef]
- Allen, K.M.; Fung, S.J.; Rothmond, D.A.; Noble, P.L.; Shannon Weickert, C. Gonadectomy Increases Neurogenesis in the Male Adolescent Rhesus Macaque Hippocampus: Gonadectomy And Adolescent Neurogenesis. Hippocampus 2014, 24, 225–238. [Google Scholar] [CrossRef]
- Carrier, N.; Kabbaj, M. Extracellular Signal-Regulated Kinase 2 Signaling in the Hippocampal Dentate Gyrus Mediates the Antidepressant Effects of Testosterone. Biological Psychiatry 2012, 71, 642–651. [Google Scholar] [CrossRef]
- Schoenfeld, T.J.; Rada, P.; Pieruzzini, P.R.; Hsueh, B.; Gould, E. Physical Exercise Prevents Stress-Induced Activation of Granule Neurons and Enhances Local Inhibitory Mechanisms in the Dentate Gyrus. J. Neurosci. 2013, 33, 7770–7777. [Google Scholar] [CrossRef] [PubMed]
- Potretzke, S.; Ryabinin, A.E. The Prairie Vole Model of Pair-Bonding and Its Sensitivity to Addictive Substances. Front. Psychol. 2019, 10, 2477. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.E.; Domínguez-Ordoñez, R.; Young, L.J.; Camacho, F.J.; Ávila-González, D.; Paredes, R.G.; Díaz, N.F.; Portillo, W. Pair-Bonding and Social Experience Modulate New Neurons Survival in Adult Male and Female Prairie Voles (Microtus Ochrogaster). Front. Neuroanat. 2022, 16, 987229. [Google Scholar] [CrossRef]
- Amrein, I.; Becker, A.S.; Engler, S.; Huang, S.; Müller, J.; Slomianka, L.; Oosthuizen, M.K. Adult Neurogenesis and Its Anatomical Context in the Hippocampus of Three Mole-Rat Species. Front. Neuroanat. 2014, 8. [Google Scholar] [CrossRef]
- Oosthuizen, M.K.; Amrein, I. Trading New Neurons for Status: Adult Hippocampal Neurogenesis in Eusocial Damaraland Mole-Rats. Neuroscience 2016, 324, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xu, N.-N.; Hao, Z.-Z.; Liu, S. Zoological Research 2023, 44, 315–322. [CrossRef]
- Kornack, D.R.; Rakic, P. Continuation of Neurogenesis in the Hippocampus of the Adult Macaque Monkey. Proc. Natl. Acad. Sci. U.S.A. 1999, 96, 5768–5773. [Google Scholar] [CrossRef]
- Larson, T.A.; Wang, T.-W.; Gale, S.D.; Miller, K.E.; Thatra, N.M.; Caras, M.L.; Perkel, D.J.; Brenowitz, E.A. Postsynaptic Neural Activity Regulates Neuronal Addition in the Adult Avian Song Control System. Proc. Natl. Acad. Sci. U.S.A. 2013, 110, 16640–16644. [Google Scholar] [CrossRef]
- Brenowitz, E.A.; Larson, T.A. Neurogenesis in the Adult Avian Song-Control System. Cold Spring Harb Perspect Biol 2015, 7, a019000. [Google Scholar] [CrossRef] [PubMed]
- Ganz, J.; Brand, M. Adult Neurogenesis in Fish. Cold Spring Harb Perspect Biol 2016, 8, a019018. [Google Scholar] [CrossRef] [PubMed]
- Teles, M.C.; Faustino, F.; Chanfana, C.; Cunha, A.; Esteves, M.; Oliveira, R.F. Social Enhancement of Adult Neurogenesis in Zebrafish Is Not Regulated by Cortisol. Neuroscience 2023, 509, 51–62. [Google Scholar] [CrossRef] [PubMed]
| Age / Stage | Species / Model | Duration / Timing of Isolation | Observed Effects | Reference |
| Early life | Rats | From early life (postnatal) into development; long-term. | Early-life social isolation leads to anxiety-like behavior in adulthood, plus marked neuroendocrine and neurochemical alterations (e.g., oxytocin, ghrelin, melatonin, monoamines). | [39] |
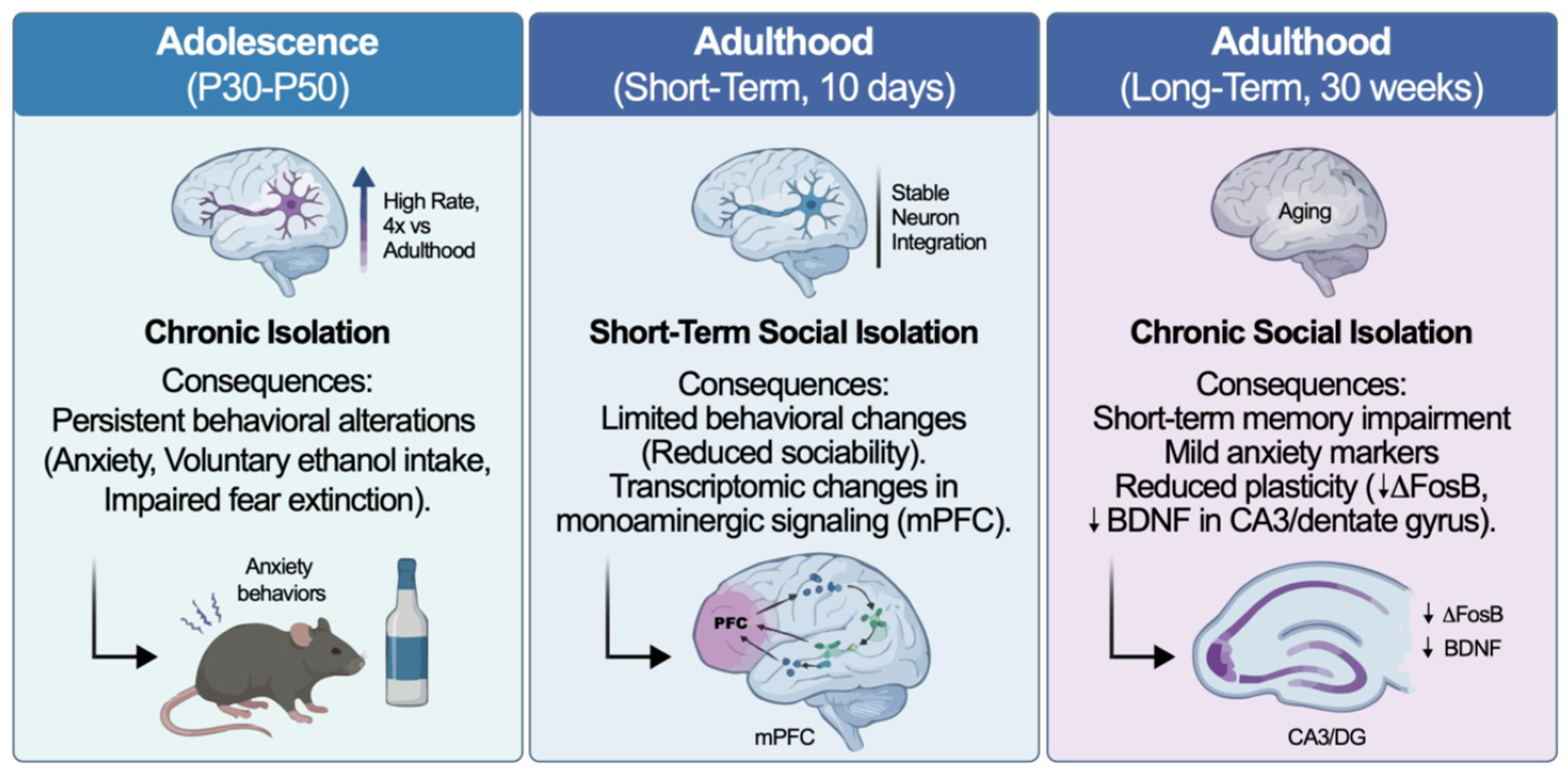
| Adolescence | Rats | Chronic isolation P30–P50 (adolescence). | Adolescent isolation produces stable changes in adult behavior, with altered forced swim and sucrose preference (active coping and reward-related behavior), particularly in females. | [41] |
| Rats | Chronic isolation during adolescence. | Adolescent social isolation increases anxiety-like behavior, voluntary ethanol intake, and difficults conditioned fear extinction on adulthood. | [40] | |
| Early Adulthood | Humans (young adults / students) | Longitudinal exposure to high loneliness / isolation. | Loneliness and social isolation predict major depressive episodes and poorer mental health over time, even after controlling for baseline mental health. | [42] |
| Adulthood | Rats | Short-term isolated housing in adulthood | Short-term social isolation (10 days) produced limited behavioral changes (mostly reduced sociability), while RNA-seq of mPFC revealed significant differential expression of genes related to dopaminergic and monoaminergic signalling. | [43] |
| Mice | Long term (30 weeks). | Long-term adult social isolation impaired short-term memory and produced mild anxiety-related markers, together with reduced ΔFosB activity in the dentate gyrus and lower BDNF expression in CA3, indicating diminished hippocampal plasticity. | [44] | |
| Humans | Short-term mandatory at-home quarantine. | Elevated anxiety and depressive symptoms; behavioral (physical inactivity, poor sleep, increased media use) and sociodemographic factors (female sex, lower education) significantly predicted symptom severity. | [45] |
| Phase | Dominant Hormones | Neural / Behavioral effects |
| Pregnancy | Rise in estrogens, progesterone, cortisol, and a strong prolactin surge. | Prolactin-driven SVZ proliferation and increased olfactory bulb neurogenesis prepare maternal circuits. |
| Early Postpartum (0–3 months) | Sharp drop in estrogens/progesterone; prolactin and oxytocin remain elevated; cortisol peaks around birth then declines. | Enhanced PFC–parietal and reward-circuit activity to infant cues; heightened maternal sensitivity. |
| Lactation | Sustained prolactin and oxytocin; altered HPA activity with elevated or modulated glucocorticoids. | Reduced hippocampal neurogenesis under baseline conditions; stress exposure can normalize neurogenesis and glucocorticoid levels. |
| Later Postpartum (≥4 months) | Gradual normalization of prolactin, oxytocin, and cortisol; increasing hormonal variability across women. | Refinement of prefrontal–parietal synchronization and continued tuning of maternal cortical networks. |
| Paternal Care | Increased prolactin and oxytocin; reduced testosterone; species-specific glucocorticoid/vasopressin changes. | Paternal experience suppresses hippocampal neurogenesis; fatherhood increases dendritic spine density and reduces anxiety-like behavior. |
| Hormone / system | Effect on DG/SVZ neurogenesis / plasticity | Relation to social behavior | References |
| Estradiol (ovarian estrogens) | Rapid but transient increase in progenitor proliferation in the dentate gyrus after acute administration; effect peaks within hours and disappears by 24–48 h. | Estradiol levels fluctuate across the estrous cycle and reproductive states, coordinating hippocampal plasticity with female reproductive physiology and sexual receptivity. | [96] |
| Androgens (testosterone, DHT) | Do not enhance proliferation, but increase survival of adult-born granule cells in the DG through androgen receptor–dependent mechanisms. | Androgen-sensitive pathways link hippocampal plasticity to male reproductive status and social/sexual contexts, where testosterone fluctuates with competition and mating. | [79] |
| Prolactin | Pregnancy-stimulated proliferation in the SVZ and increased neurogenesis in the olfactory bulb; effects are blocked by prolactin antagonism and mimicked by prolactin infusion. | These changes are interpreted as preparatory adaptations for maternal behavior, enhancing olfactory processing of offspring-related cues. | [80] |
| Oxytocin / vasopressin | Modulate hippocampal synaptic transmission and plasticity (e.g., CA2, DG) rather than directly altering proliferation; influence how existing circuits encode socially relevant information. | Oxytocin and vasopressin signaling in the hippocampus and related regions is critical for social recognition and social memory, supporting affiliative and pair-bonding behaviors. | [71] |
| BDNF and neurotrophins | Support survival, dendritic growth and functional integration of adult-born neurons in DG, and mediate activity-dependent reorganization of hippocampal circuits. | BDNF levels are strongly modulated by experience (exercise, enrichment, stress) and are tightly linked to learning, memory, and affective regulation in social contexts. | [34] |
| Monoaminergic and stress/inflammatory pathways (dopamine, serotonin, glucocorticoids, cytokines) | Chronic social isolation and social defeat reduce DG cell proliferation and neuronal activity and decrease BDNF expression in hippocampal subfields (DG ΔFosB↓, CA3 BDNF↓), while acute pharmacological modulation of monoamines can reverse or buffer some of these effects. | Social reward and stress are transduced by dopaminergic and monoaminergic circuits: dopamine in VTA–NAc supports sexual and social reward; long-term isolation or defeat alters monoaminergic gene expression in mPFC and produces anxiety/depression-like behavior, with associated reductions in hippocampal plasticity. | [43,44,95,108] |
| Model / species group | Type of sociality | Adult neurogenesis – key findings | Evolutionary / functional particularities | References |
| Prairie vole (Microtus ochrogaster) | Social monogamy; stable pair bonds; biparental care | Adult-born cells in the dentate gyrus and olfactory bulb are modulated by cohabitation with mating and opposite-sex social exposure. These social conditions enhance survival and differentiation of new neurons, with sex-dependent effects, aligning with mechanisms underlying pair-bond formation. | Specialized oxytocin and vasopressin systems in mesolimbic–social circuits support selective partner preference and long-term attachments, making prairie voles a canonical model of mammalian social bonding. | [114,115] |
| Naked mole-rat and eusocial mole-rats | Eusociality: reproductive castes; cooperative colonies | Eusocial mole-rats show markedly reduced adult hippocampal neurogenesis relative to surface-dwelling rodents, despite preserved progenitor markers. In naked mole-rats, new cells exhibit slow maturation, and neurogenesis is low but sustained across the lifespan, consistent with exceptional longevity. | Subterranean, hypoxic ecology and extreme lifespan correlate with a small, developmentally protracted hippocampus and atypical expression of calcium-binding proteins. These features are often interpreted as indicators of neoteny-like brain organization in mole-rats. | [116,117] |
| Nonhuman primates (macaques and other monkeys) | Complex dominance hierarchies; alliances; long-term social relationships | Adult hippocampal neurogenesis is clearly present but occurs at ~10-fold lower rates than in rodents. New granule cells mature over many months, and AHN declines with age, reaching very low levels in older primates. | Large brains, extended developmental trajectories, and sophisticated social cognition rely more on synaptic and structural plasticity than on high neuronal turnover, positioning primates as a key translational bridge for understanding human AHN. | [118,119] |
| Songbirds (oscine songbirds: canaries, zebra finches, etc.) | Learned vocal communication; seasonal territoriality; mate attraction via song | Song-control nuclei (HVC, RA, Area X) exhibit robust adult neurogenesis. Recruitment and survival of new neurons are regulated by photoperiod, gonadal hormones, singing activity, and social context, tightly linking neurogenesis to song learning and seasonal song plasticity. | Songbirds are the classic demonstration that adult-born projection neurons directly support a socially relevant learned behavior. Strong seasonal dynamics make them an ideal model to study endocrine, environmental, and social regulation of neurogenesis at a circuit level. | [120,121] |
| Teleost fish (e.g., zebrafish, trout) | Schooling / shoaling; group living; rich social signaling | Teleost fish display extensive adult neurogenesis across multiple proliferative zones along the rostro–caudal brain. Although the cited work on social plasticity does not measure neurogenesis directly, it demonstrates that social context modulates neural activity and gene expression in circuits involved in social behavior, highlighting the high adaptability of teleost neural systems. | Their lifelong brain growth and exceptional regenerative capacity make teleosts a valuable comparative model for studying neural plasticity, offering a clear contrast with the limited neurogenic potential observed in adult mammals. | [122,123] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




