Submitted:
03 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Carbon in the Language of Covalent Bonds
2.1. Crystalline Carbon
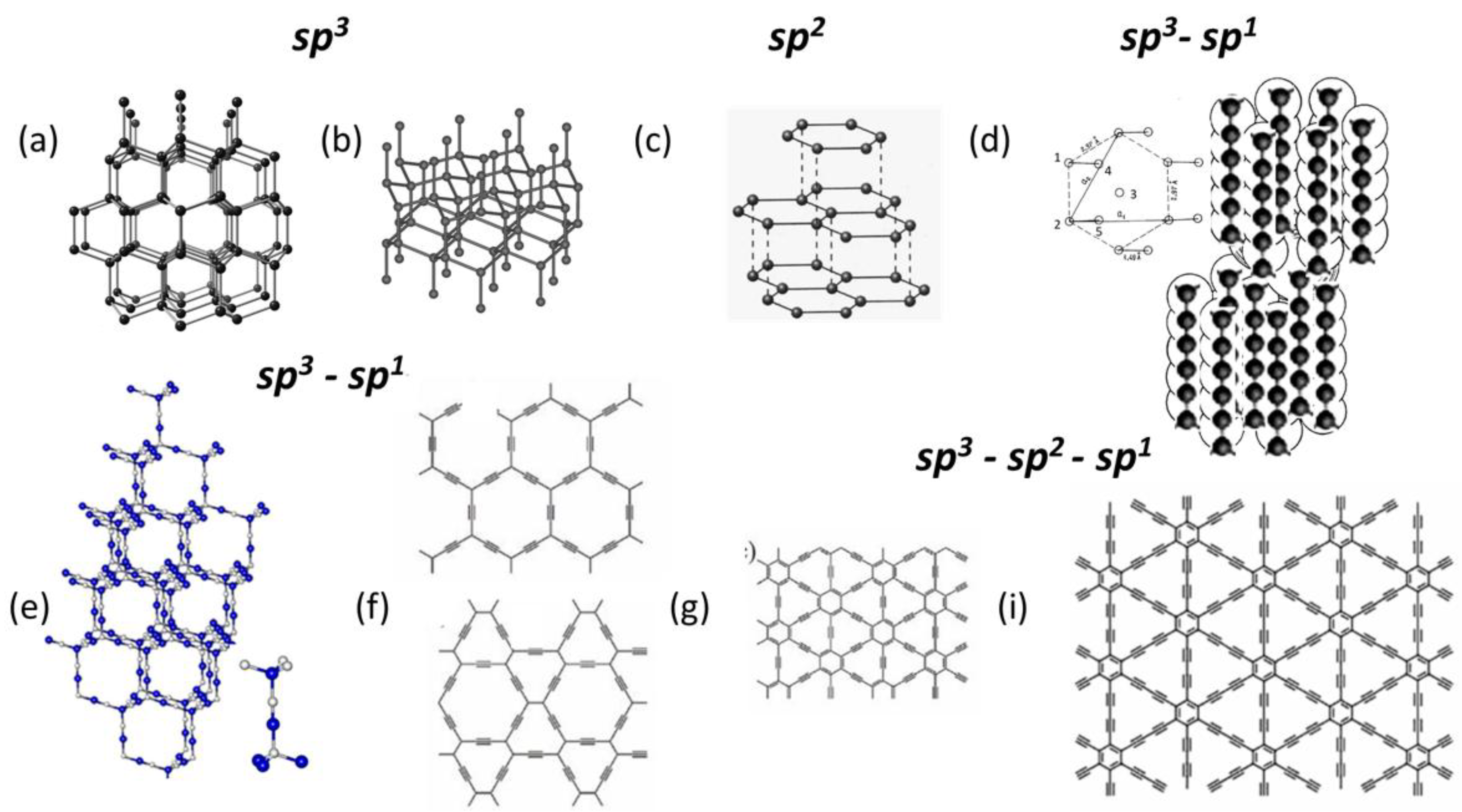
2.2. Amorphous Carbon
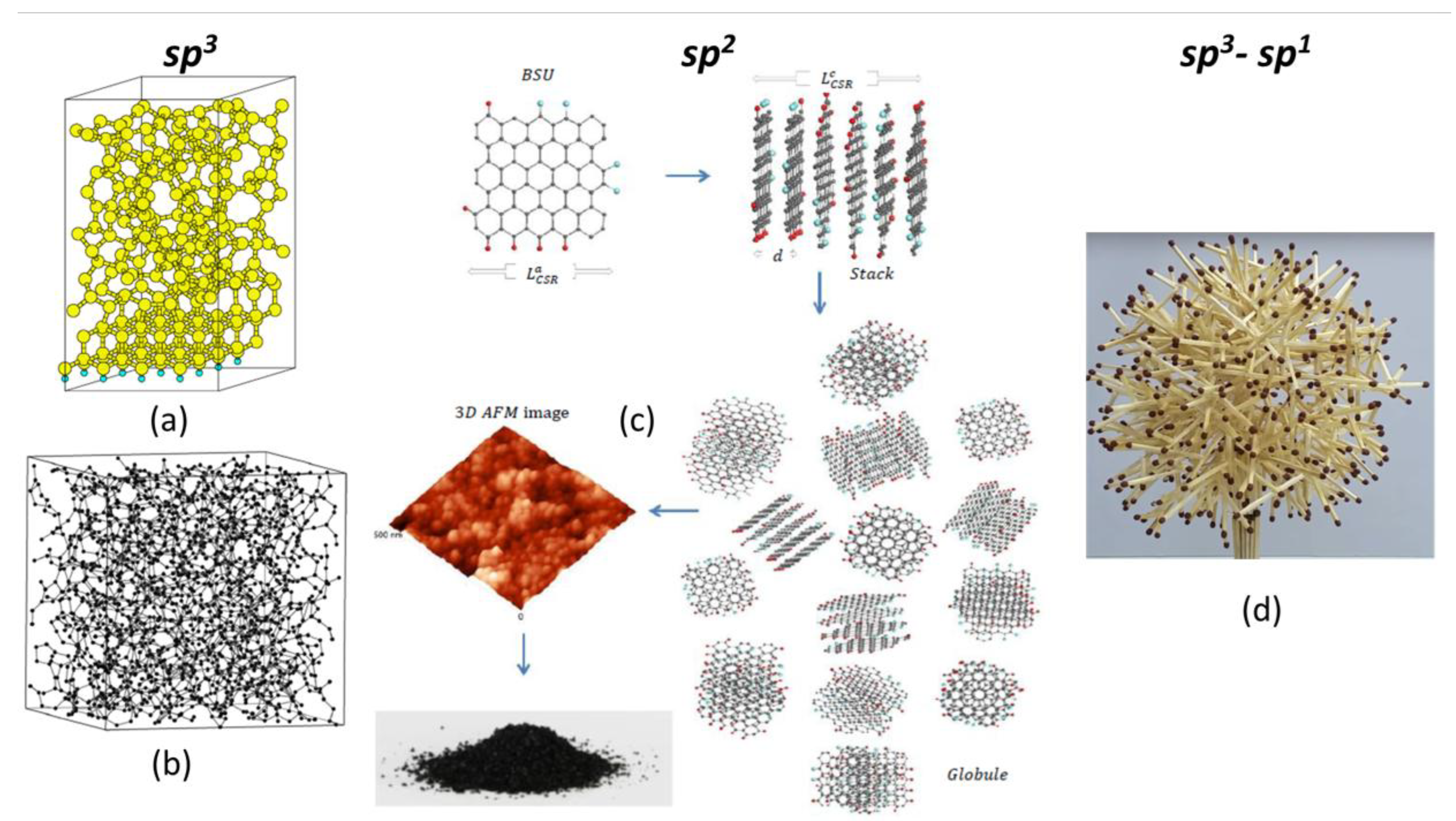
3. Lengths of Bonds as a Governing Factor of the Carbon Covalent Chemistry
3.1. Spin-Radical Concept of the Carbon Atoms’ Covalence
3.2. Spin Emergents of Carbon Covalent Bonds
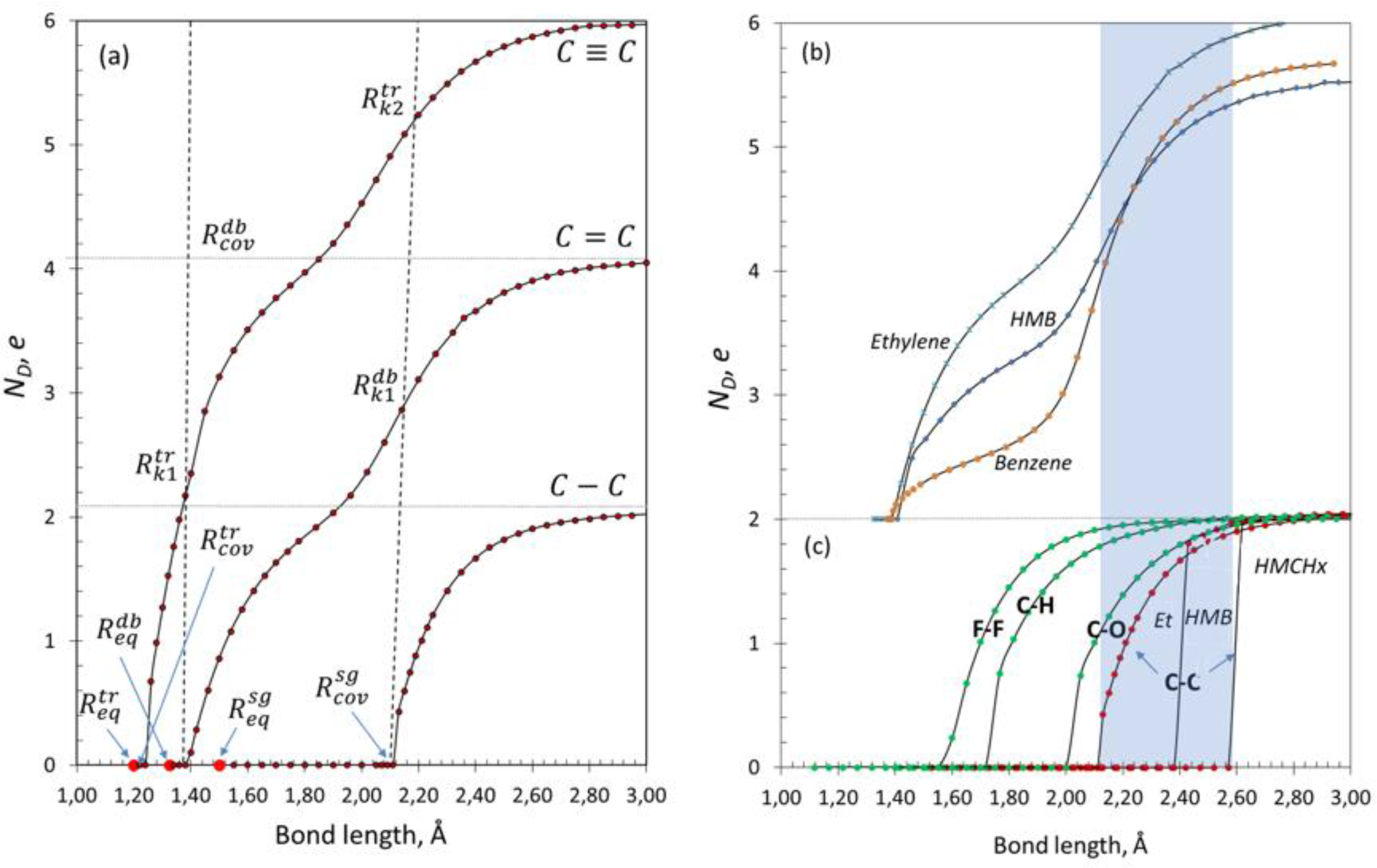
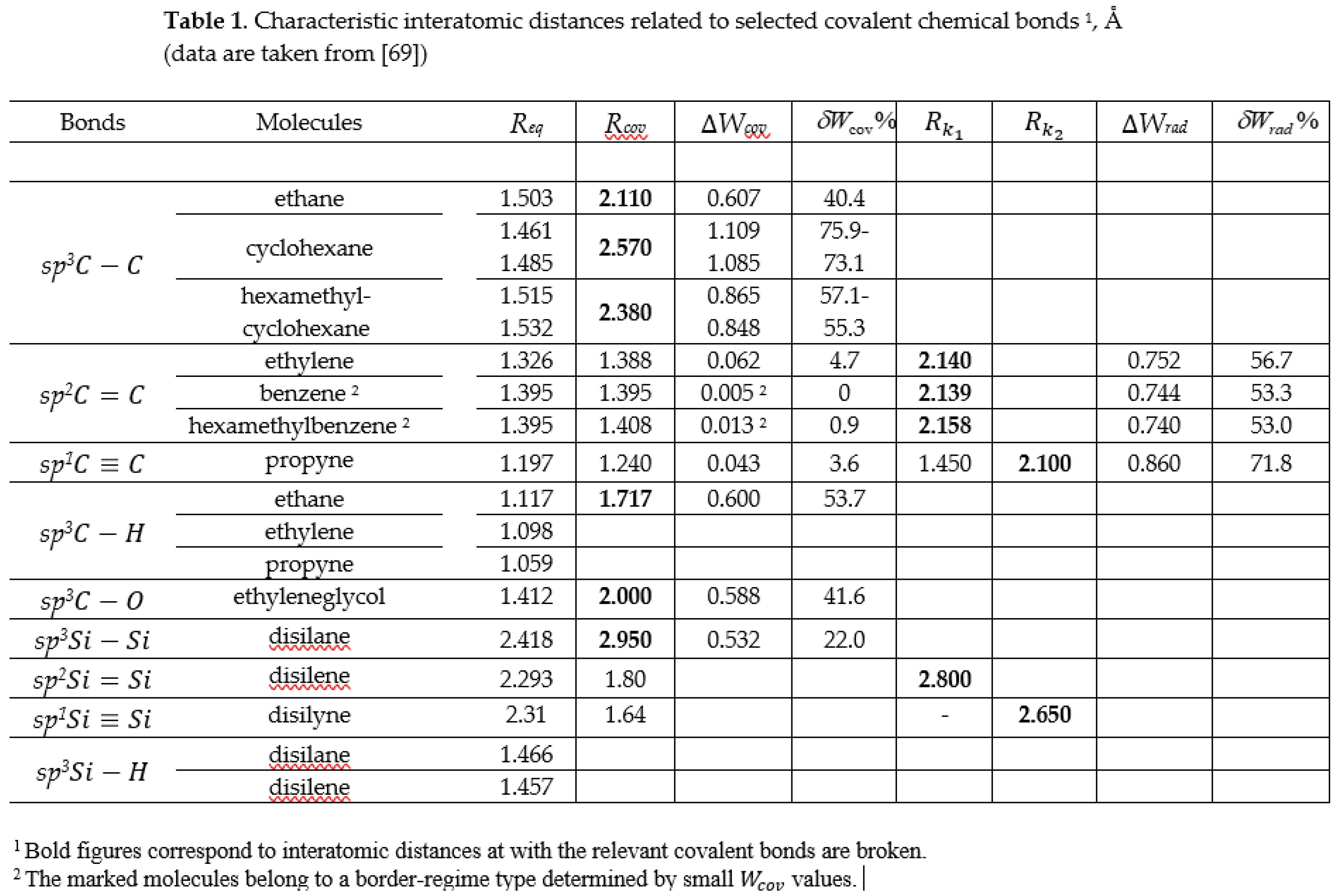
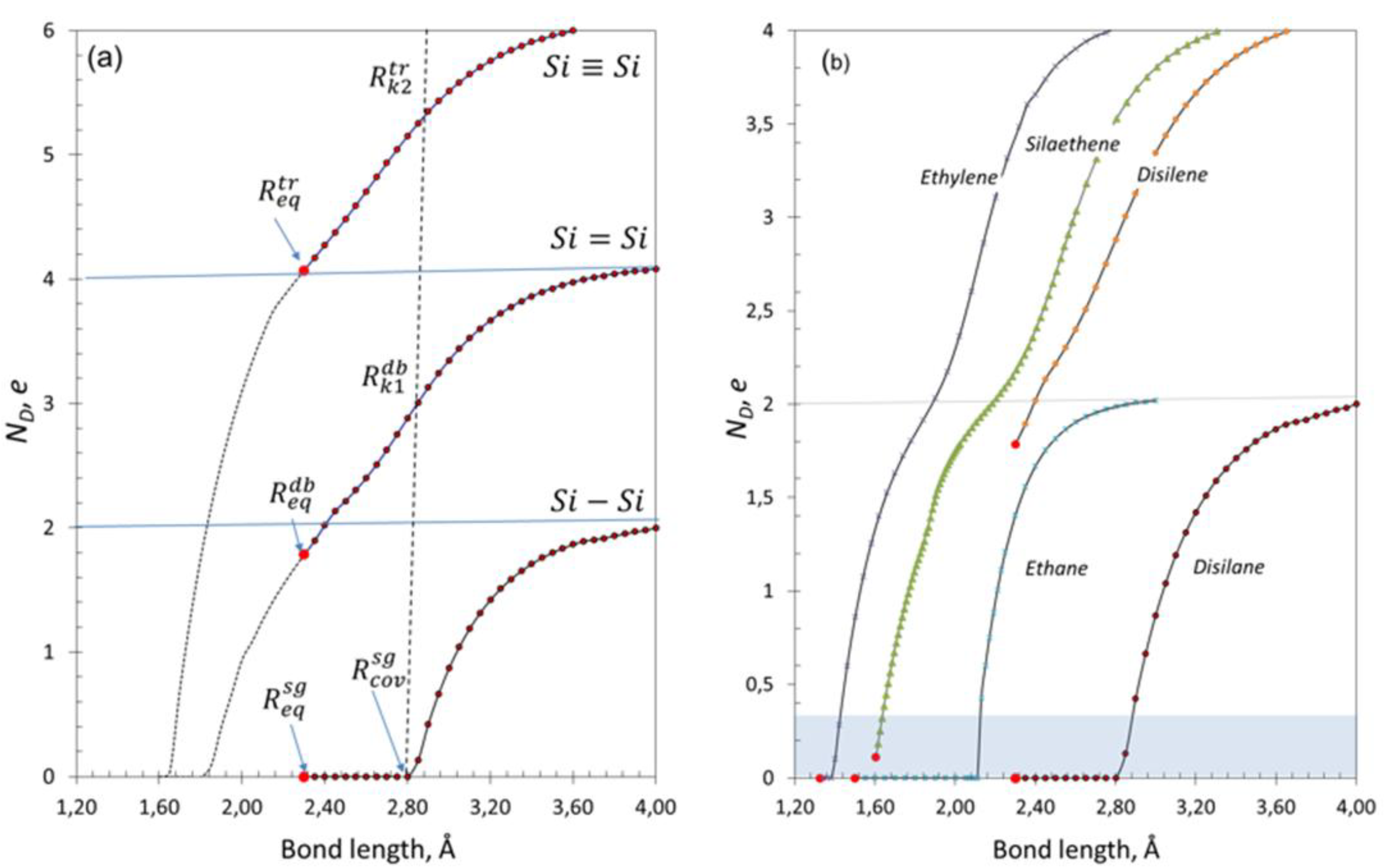
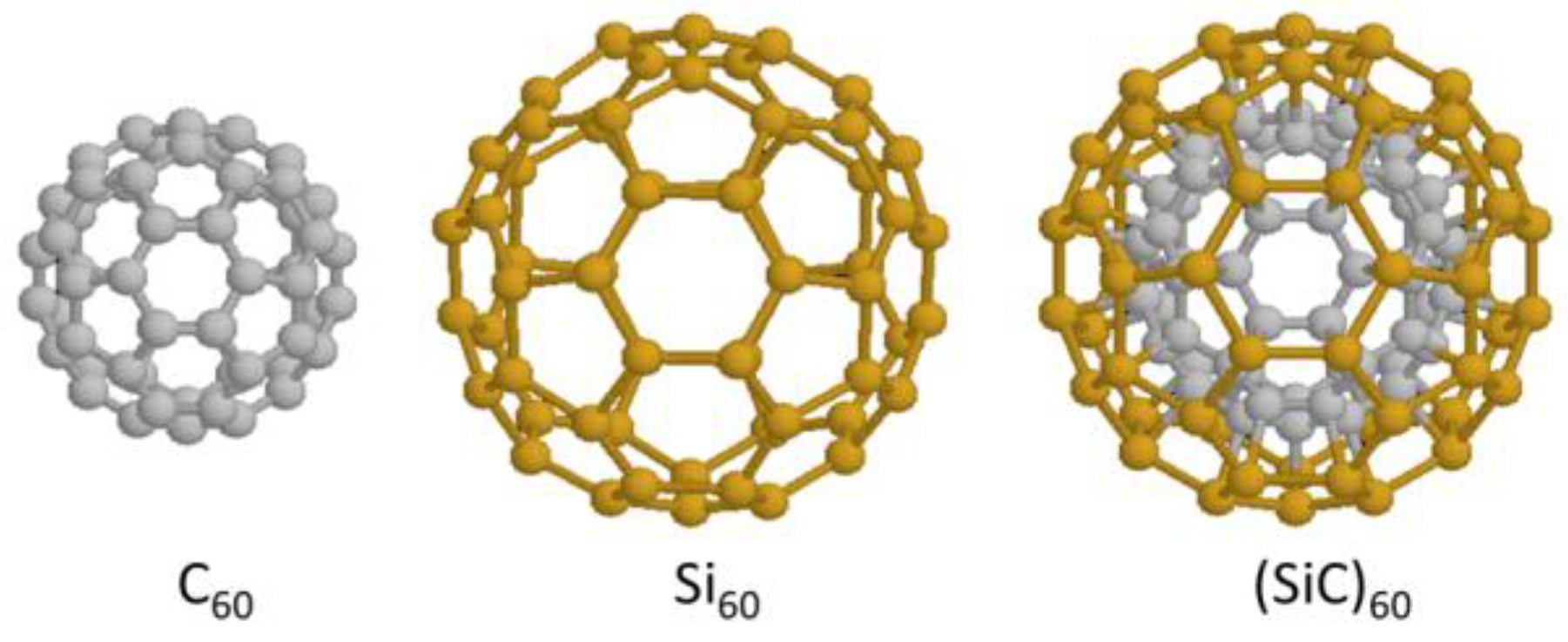
3.2. Spin Emergents of Silicon Covalent Bonds
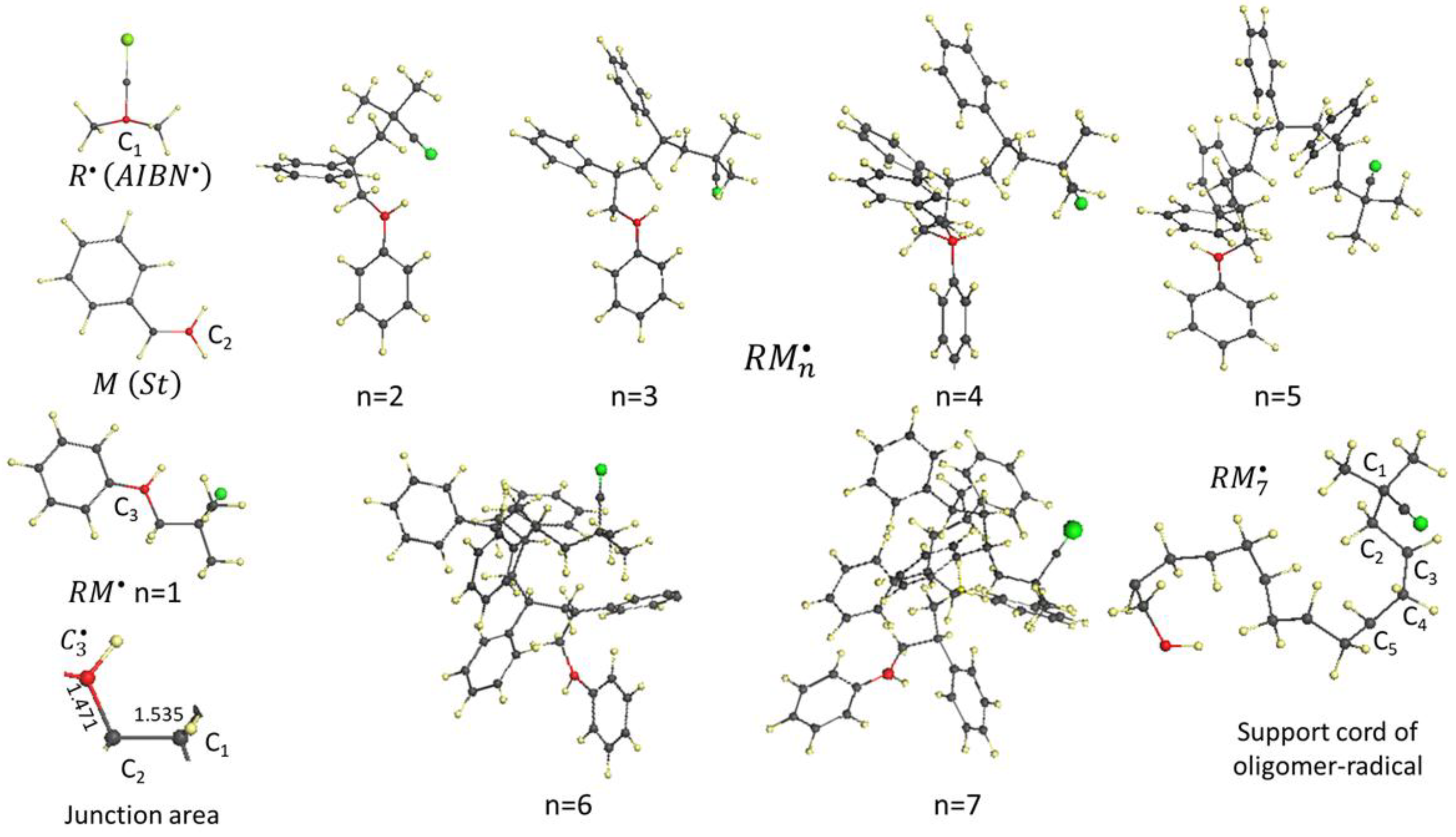
4. Spin Chemistry of Alkane Bonds
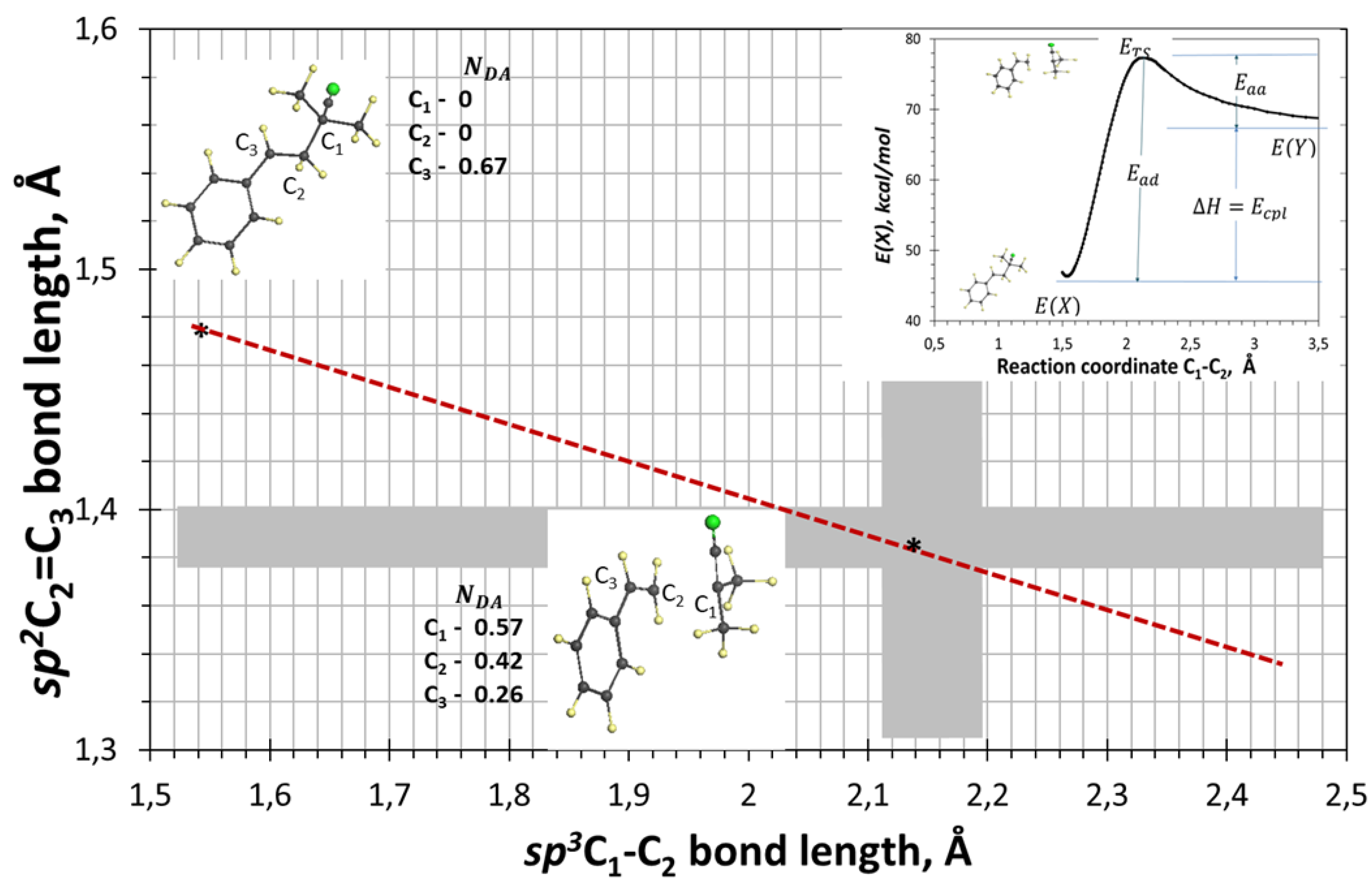
5. Spin Covalence of Alkene Bonds
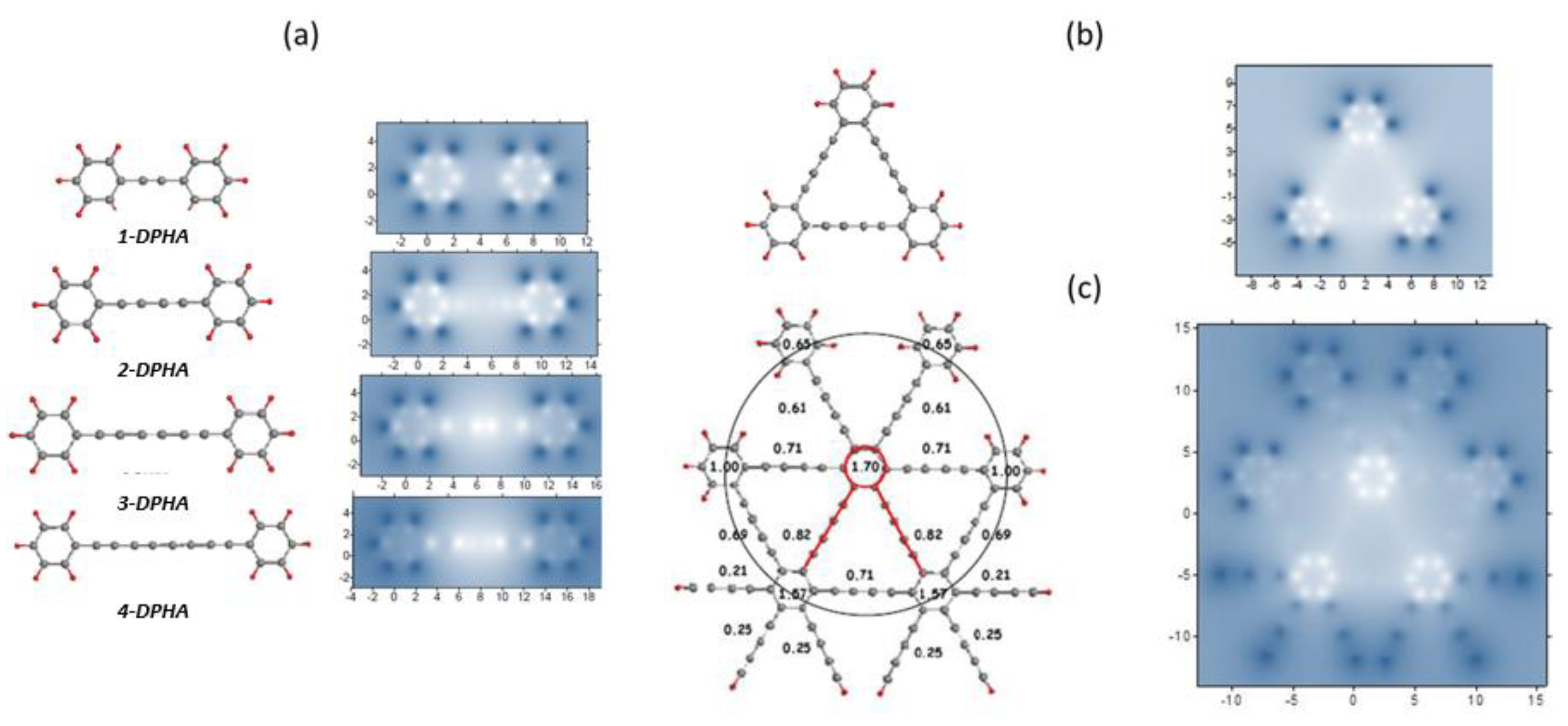
5.1. Aromatic Hydrocarbons
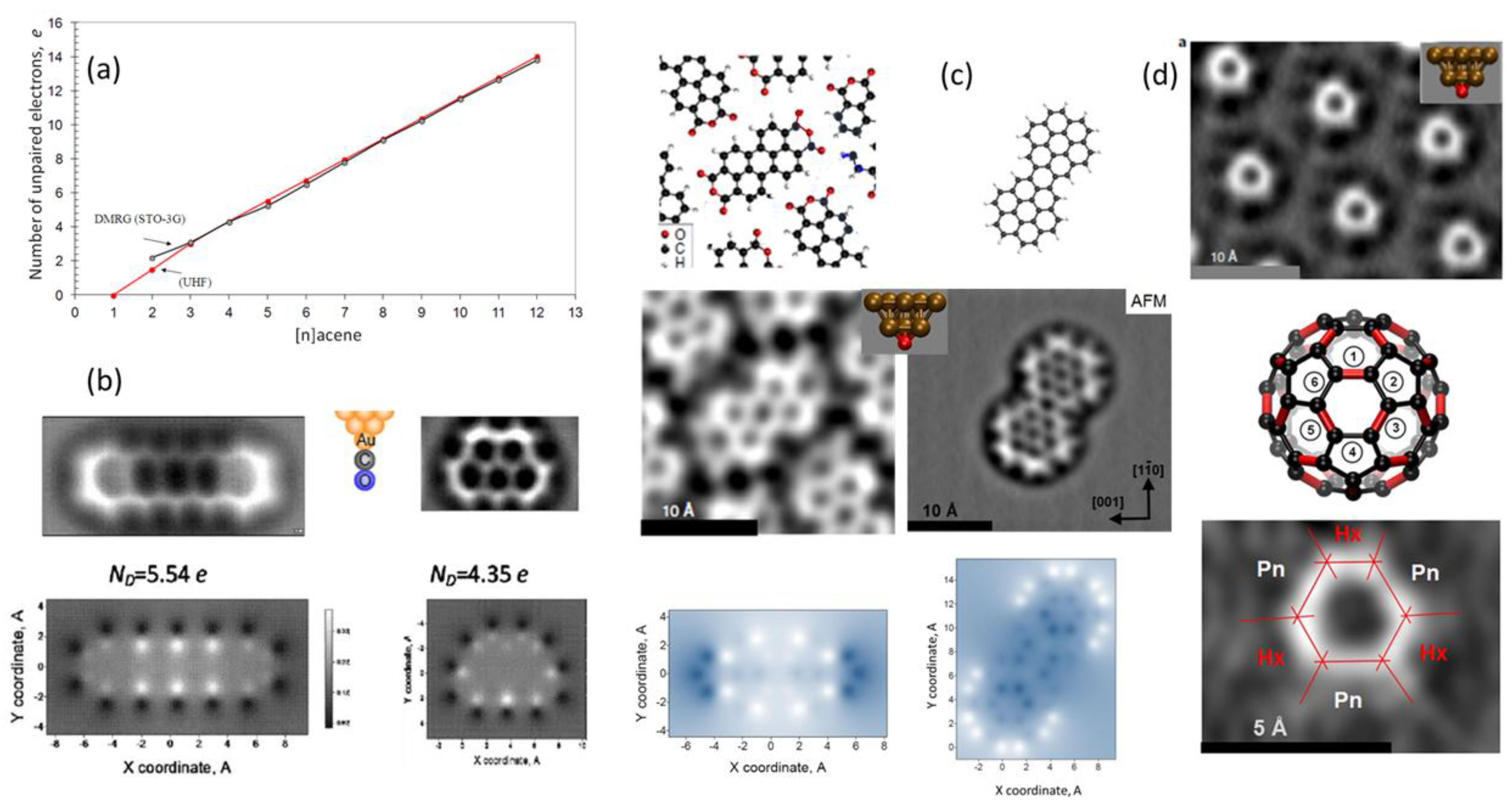
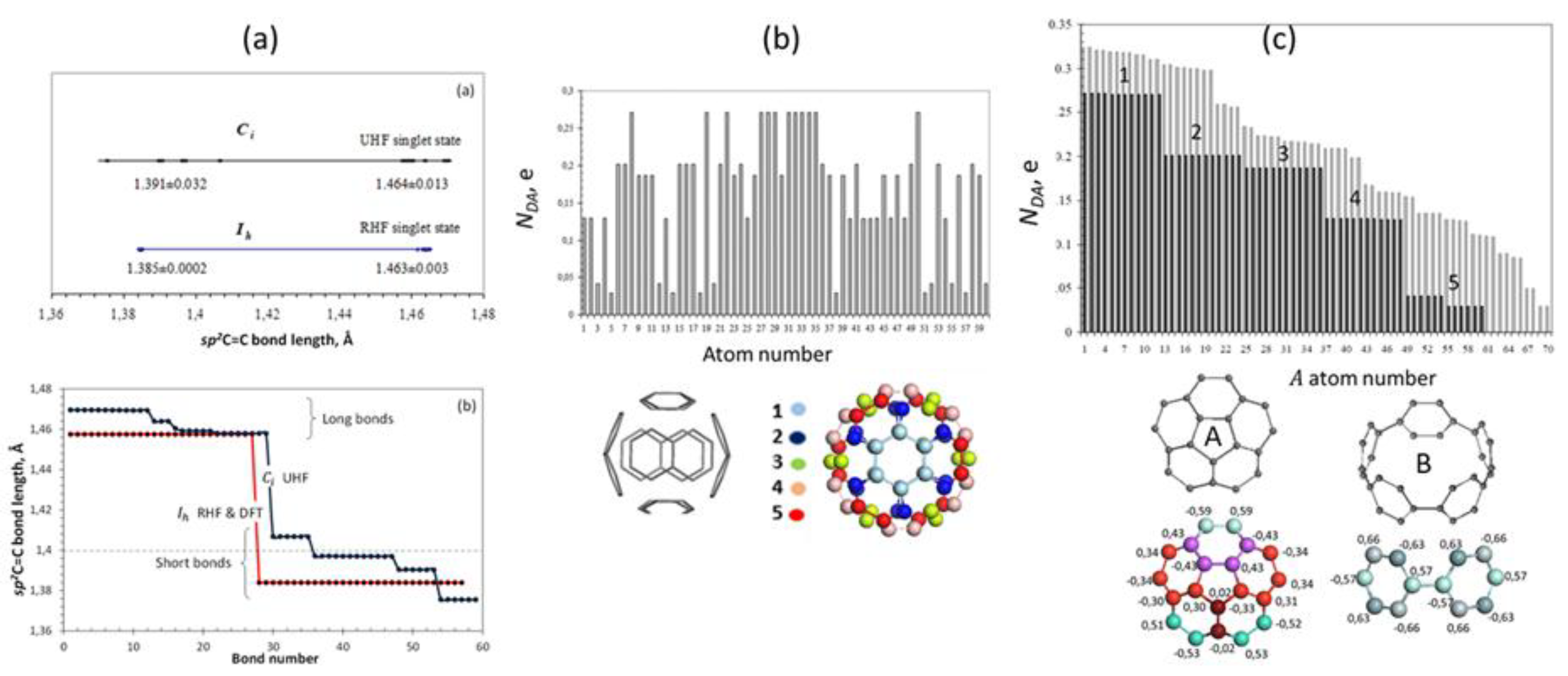
5.2. Fullerenes
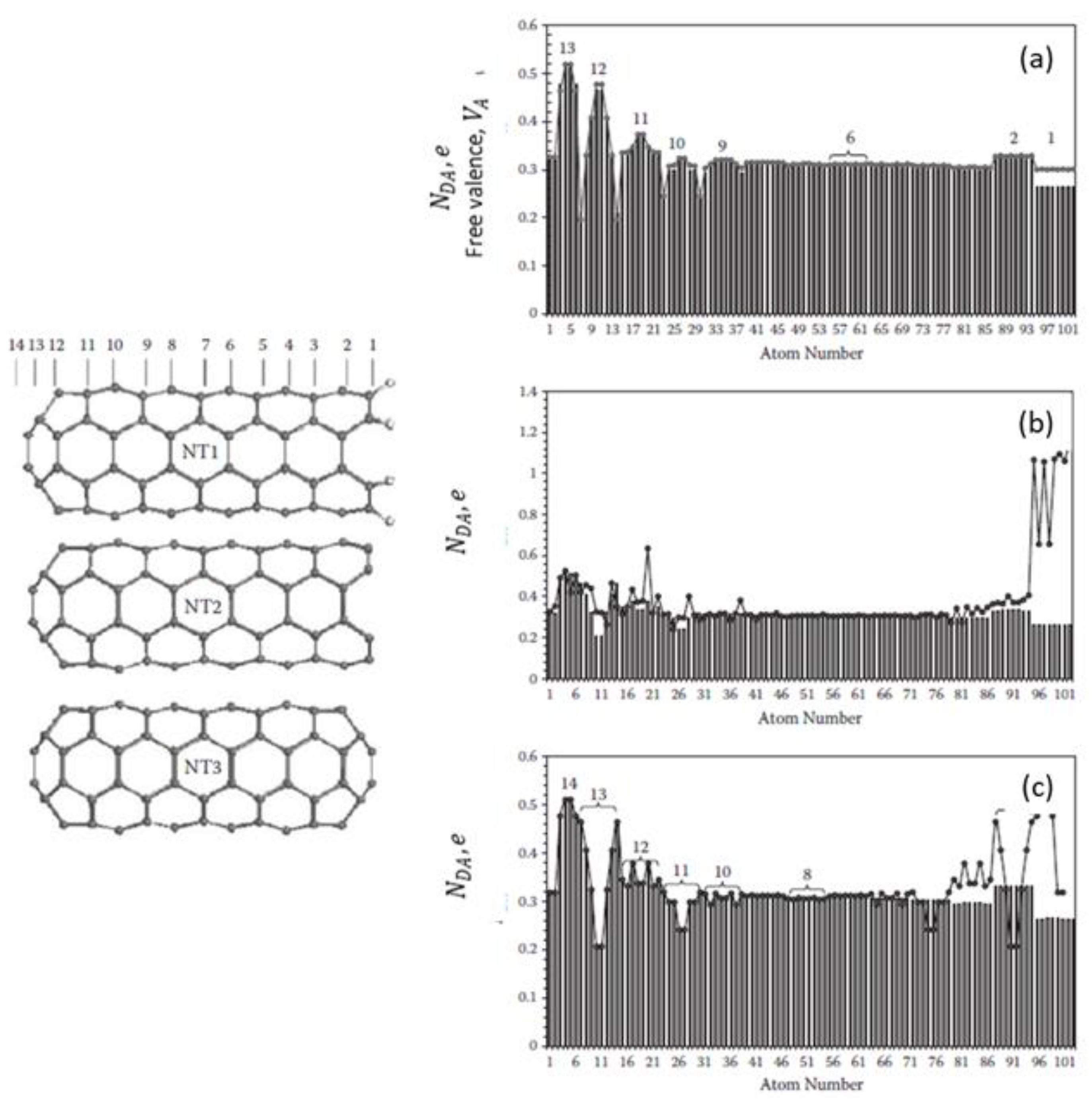
5.3. Carbon Nanotubes
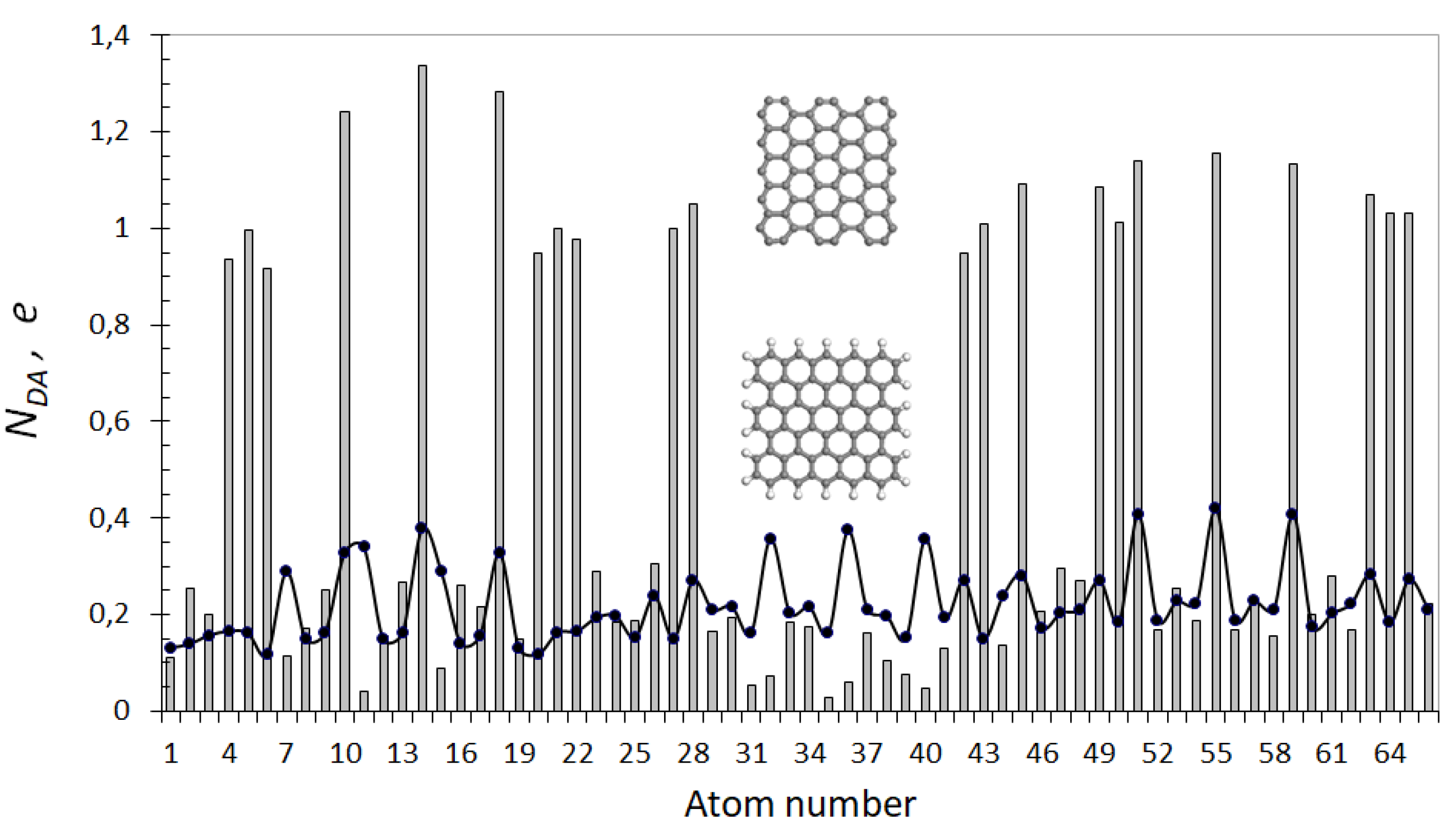
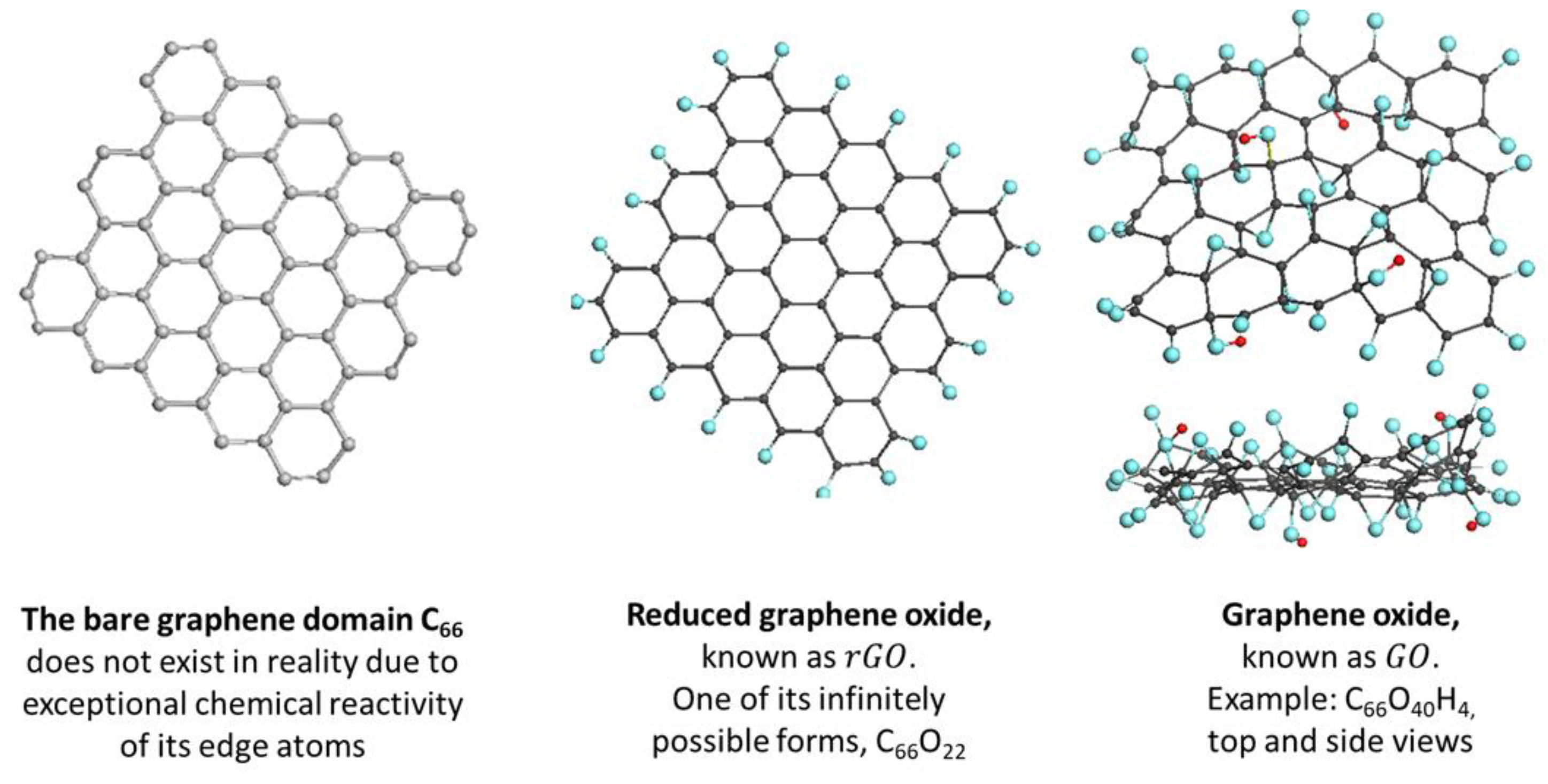
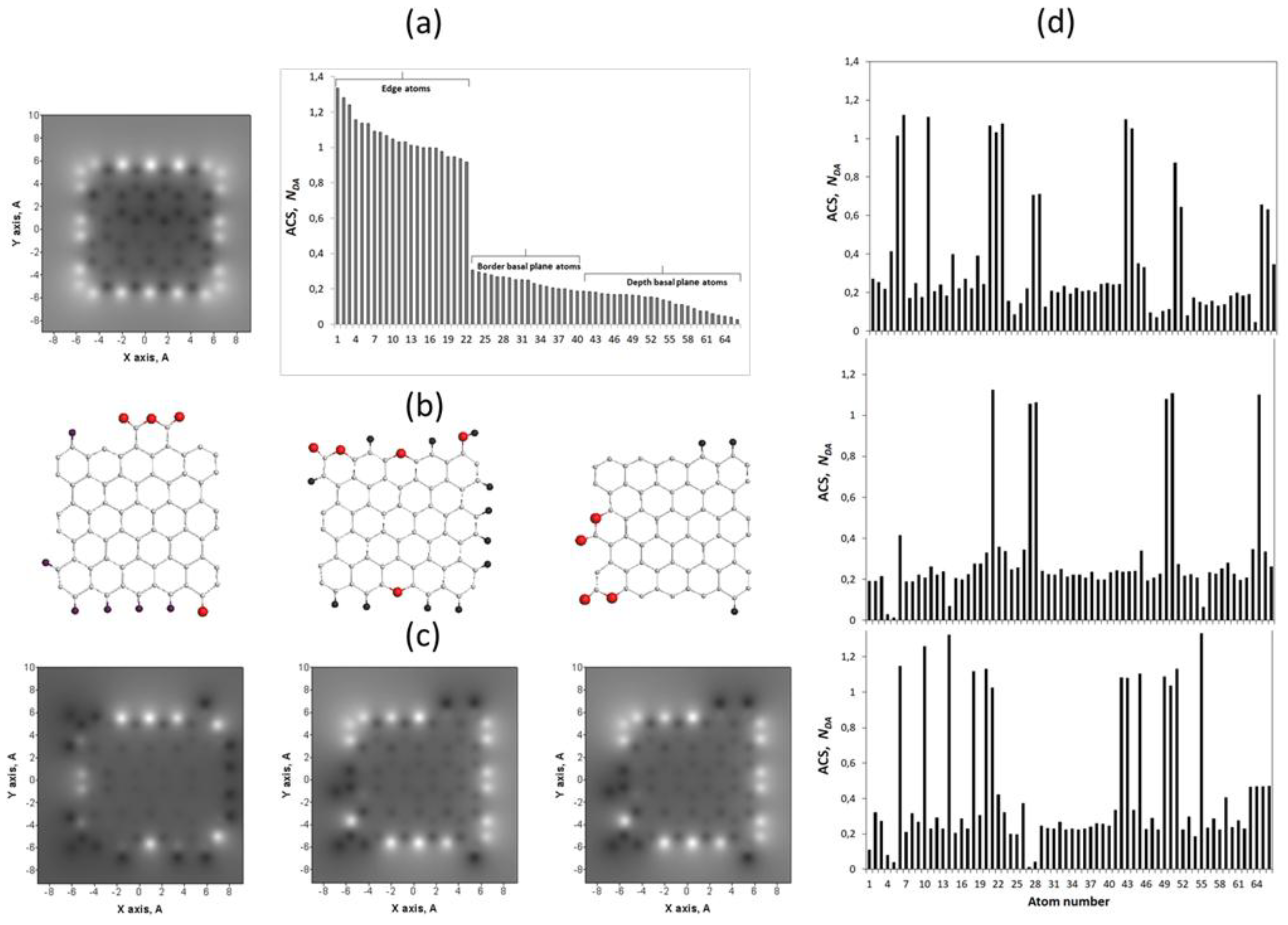
5.4. Graphene Domains
6. Spin Covalence of Alkyne Bonds
7. Carbon Catalysts in Light of Their Spin Covalence
7.1. Graphene Carbocatalysis
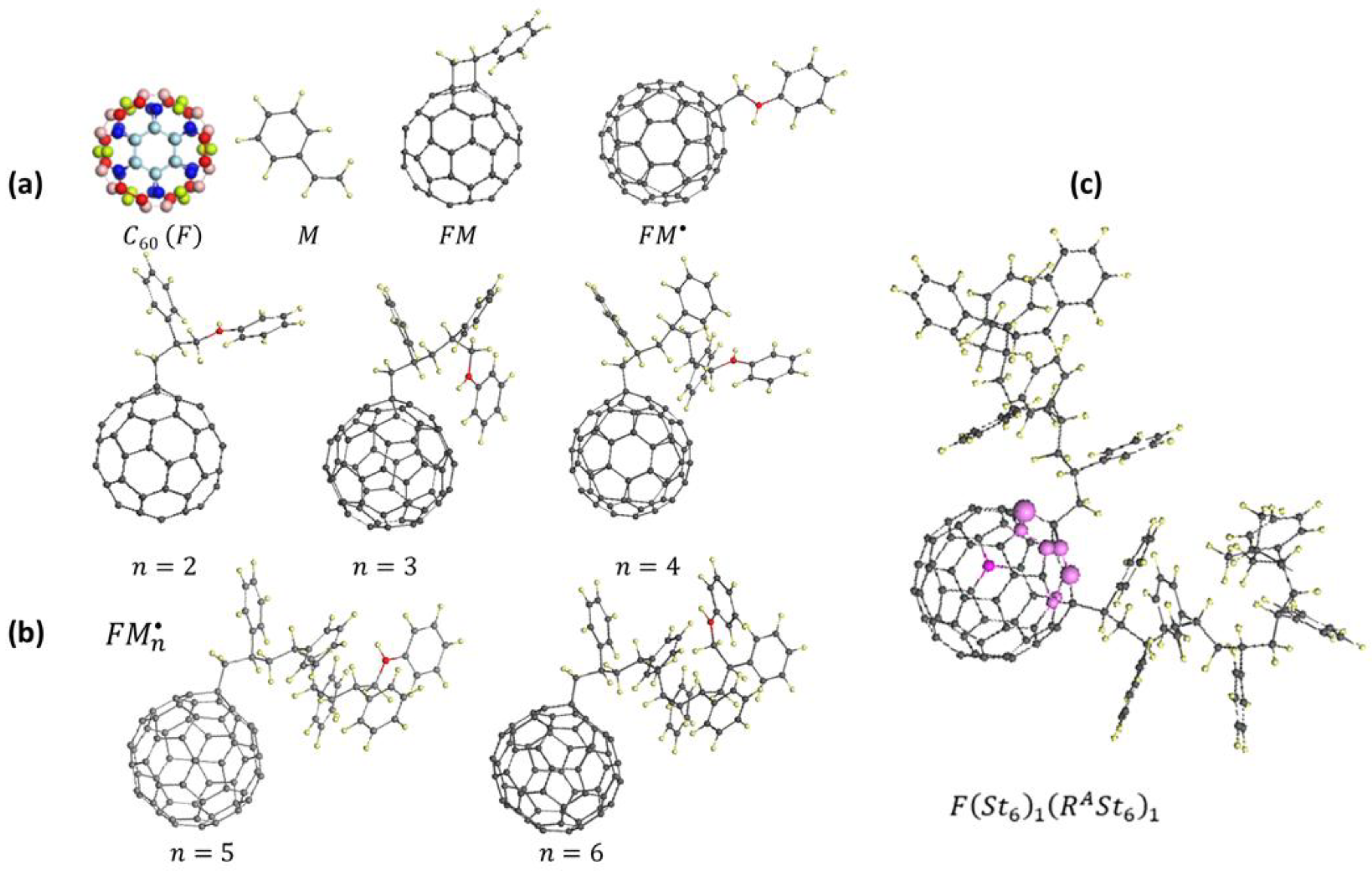
7.2. Fullerene Carbocatalysis
8. Aposteriori Reflections and Conclusive Remarks
Acknowledgments
References
- Allègre, CJ; Poirier, J-P; Humler, E; et al. The chemical composition of the Earth. Earth Planet Sci Lett. 1995, 134, 515–526. [Google Scholar] [CrossRef]
- Sladkov, A. M.; Kudryavtsev, Yu. P. Diamond, graphite, carbyne = allothropic forms of carbon. Priroda (Moscow) 1969, 5, 3. [Google Scholar]
- Bean, J. J.; Katiyar, N.K.; Forrest, R. M.; Zhou, X.; Goel, S. Resolving Lonsdaleite's decade-long controversy: Atomistic insights into a metastable diamond polymorph. Diam. Rel. Mat. 2025, 157, 112405. [Google Scholar] [CrossRef]
- Thapliyal, V.; Alabdulkarim, M. E.; Whelan, D. R.; Mainali, B.; Maxwell, J. L. A concise review of the Raman spectra of carbon allotropes. Diam. Rel. Mat. 2022, 127, 109180. [Google Scholar] [CrossRef]
- Ubbelohde, A.R. F. A. Lewis, Graphite and Its Crystal Compounds; Clarendon: Oxford, 1960. [Google Scholar]
- Kudryavtsev, Y.P. The discovery of carbyne. In Carbyne and Carbynoid Structures. Physics and Chemistry of Materials with Low-Dimensional Structures; Heimann, R.B., Evsyukov, S.E., Kavan, L., Eds.; Springer: Dordrecht, 1999; vol 21, pp. 1–6. [Google Scholar]
- Kudryavtsev, Y.P.; Evsyukov, S.E.; Guseva, M.B.; et al. Carbyne — the third allotropic form of carbon. Russ. Chem. Bull. 1993, 42, 399–413. [Google Scholar] [CrossRef]
- Kudryavtsev, Yu.P.; Evsyukov, S.E.; Babaev, V.G.; Gouseva, M.B. Oriented carbyne layers. Carbon 1992, 30, 213–221. [Google Scholar] [CrossRef]
- EL Goresy, A.; Donnay, G. A new allotropic form of carbon from the Ries Crater. Science 1968, 161, 363. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, A.G. Carbon: occurence of carbyne forms of carbon in natural graphite. Carbon 1979, 17, 21–24. [Google Scholar] [CrossRef]
- Bulychev, B. M.; Udod, I. A. Formation of ‘carbyne’ in the interaction of polyacetylene. Ross. Khim. Zh 1995, 39, 9–14. [Google Scholar]
- Heimann, R. B.; Evsyukov, S. E.; Kavan, L. (Eds.) Carbyne and Carbynoid Structures; Springer: Dordrecht, 1999. [Google Scholar]
- Heimann, R. B. The nature of carbyne – pros and cons. In Carbyne and Carbynoid Structures; Heinmann, R.B., Evsyukov, S.E., Kavan, L., Eds.; Springer: Dordrecht, 1999; pp. 1–15. [Google Scholar]
- A Hirsch, The era of carbon allotropes. Nat. Mater. 2010, 9, 868–871. [CrossRef]
- Jarowski, P.D.; Wodrich, M.D.; Wannere, C.S.; Schleyer, P.V.R.; Houk, K.N. How large is the conjugative stabilization of diynes? J. Am. Chem. Soc. 2004, 126, 15036–15037. [Google Scholar] [CrossRef] [PubMed]
- Itzhaki, L.; Altus, E.; Basch, H.; Hoz, S. Harder than diamond: determining the cross-sectional area and Young's modulus of molecular rods. Angew. Chem. Int. Ed. 2005, 44, 7432–7435. [Google Scholar] [CrossRef] [PubMed]
- Enyashin, A.N.; Ivanovskii, A.L. Structural, electronic, and elastic properties of Y-diamonds and their BN analogues. Diam. Rel. Mat. 2013, 38, 93–100. [Google Scholar] [CrossRef]
- Enyashin, A.N.; Ivanovskii, A.L. Structural and electronic properties of new 1D and 2D carbon allotropes with mixed sp1 - sp3 hybridization types. Chem. Phys. Lett. 2014, 609, 15–20. [Google Scholar] [CrossRef]
- Li, X.; Li, B-h.; He, Y-b.; Kang, F-y. A review of graphynes: Properties, applications and Synthesis. New Carbon Mat. 2020, 35, 619–629. [Google Scholar] [CrossRef]
- Baughman, A R. H.; Eckhardt, H. Structure-property predictions for new planar forms of carbon: Layered phases containing sp2 and sp atoms. J. Chern. Phys. 1997, 87, 6687. [Google Scholar] [CrossRef]
- Ivanovskii, L. Graphynes and graphdyynes. Progr. Solid State Chem. 2013, 41, 1–32. [Google Scholar] [CrossRef]
- Parvin, N.; Jin, Q.; Wei, Y.; Yu, R.; Zheng, B.; Huang, L. et.all. Few-layer graphdiyne nanosheets applied for multiplexed real-time DNA detection. Adv. Mater. 2017, 29, 1606755. [Google Scholar] [CrossRef]
- Li, G; Li, Y; Liu, H; et al. Architecture of graphdiyne nanoscale films. Chem. Communic 2010, 46, 3256–3258. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J; Gao, X; Liu, R; et al. Synthesis of graphdiyne nanowalls using acetylenic coupling reaction. JACS 2015, 137, 7596–7599. [Google Scholar] [CrossRef]
- Wang, S S; Liu, H B; Kan, X N; et al. Superlyophilicity-facilitated synthesis reaction at the microscale: ordered graphdiyne stripe arrays. Small 2017, 13, 1602265. [Google Scholar] [CrossRef] [PubMed]
- Matsuoka, R; Sakamoto, R; Hoshiko, K; et al. Crystalline graphdiyne nanosheets produced at a gas/liquid or liquid/liquid interface. JACS 2017, 139, 3145–3152. [Google Scholar] [CrossRef] [PubMed]
- Caro, M. A.; Zoubkoff, R.; Lopez-Acevedo, O.; Laurila, T. Atomic and electronic structure of tetrahedral amorphous carbon surfaces from density functional theory: Properties and simulation strategies. Carbon 2014, 77, 1168–1182. [Google Scholar] [CrossRef]
- Palomäki, T.; Wester, N.; Caro, M. A.; Sainio, S.; Protopopova, V.; Koskinen, J.; Laurila, T. Electron transport determines the electrochemical properties of tetrahedral amorphous carbon (ta-C) thin films. Electrochim. Acta 2017, 225, 1–10. [Google Scholar] [CrossRef]
- Gupta, S.; Bhaumik, A.; Sachan, R.; et al. Structural evolution of Q-carbon and nanodiamonds. JOM 2018, 70, 450–455. [Google Scholar] [CrossRef]
- Narayana, J.; Bhaumika, A.; Guptaa, S.; Haquea, A.; Sachan, R. Progress in Q-carbon and related materials with extraordinary properties. Mater. Res. Lett. 2018, 6, 353–364. [Google Scholar] [CrossRef]
- Sachan, R.; Gupta, S.; Narayan, J. Nonequilibrium structural evolution of Q-carbon and interfaces. ACS Appl. Mater. Interf. 2020, 12, 1330–1338. [Google Scholar] [CrossRef]
- Sheka, E.F.; Hołderna-Natkaniec, K.; Natkaniec, I.; Krawczyk, J. X.; Golubev, Ye.A.; Rozhkova, N.N.; Kim, V.V.; Popova, N.A.; Popova, V.A. Computationally supported neutron scattering study of natural and synthetic amorphous carbons. J. Phys. Chem. C 2019, 123, 15841–15850. [Google Scholar] [CrossRef]
- Golubev, Ye. A.; Rozhkova, N. N.; Kabachkov, E. N.; Shul'ga, Y. M.; Natkaniec-Hołderna, K.; Natkaniec, I.; Antonets, I. V.; Makeev, B. A.; Popova, N. A.; Popova, V. A.; Sheka, E. F. sp2 Amorphous carbons in view of multianalytical consideration: normal, expeсted and new. J. Non-Cryst. Solids 2019, 524, 119608. [Google Scholar] [CrossRef]
- Sheka, E. F.; Natkaniec, I.; Ipatova, E. U.; Golubev, Ye. A.; Kabachkov, E. N.; Popova, V. A. Heteroatom necklaces of sp2 amorphous carbons. XPS supported INS and DRIFT spectroscopy. FNCN 2020, 28, 1010–1029. [Google Scholar]
- Sheka, E.F.; Golubev, Y. A.; Popova, N. A. Graphene domain signature of Raman spectra of sp2 amorphous carbons. Nanomat 2020, 10. [Google Scholar] [CrossRef]
- Sheka, E.F.; Golubev, Ye.A.; Popova, N.A. Amorphous state of sp2 solid carbons. FNCN 2021, 29, 107–113. [Google Scholar] [CrossRef]
- Sheka, E.F. A neoteric view of sp2 amorphous carbon. Nanomat. 2023, 13, 1648. [Google Scholar] [CrossRef]
- Warren, B.E. X-ray diffraction in random layer lattices. Phys. Rev. B 1941, 59, 639–643. [Google Scholar] [CrossRef]
- Avdeev, M. V.; Tropin, T. V.; Aksenov, V. L.; Rosta, L.; Garamus, V. M.; Rozhkova, N. N. Pore structures in shungites as revealed by small-angle neutron scattering. Carbon 2006, 44, 954–961. [Google Scholar] [CrossRef]
- Golubev, Ye.A.; Ulyashev, V.V.; Veligzhanin, A.A. Porosity and structural parameters of Karelian shungites according to the data of small-angle synchrotron radiation scattering and microscopy. Crystallgr. Rep. 2016, 61, 66–77. [Google Scholar] [CrossRef]
- Duan, X.; Ao, Z.; Zhang, H.; Saunders, M.; Sun, H.; Shao, Z.; Wang, S. Nanodiamonds in sp2 /sp3 configuration for radical to nonradical oxidation: Core-shell layer dependence. Appl. Cat. B: Envirom 2018, 222, 176–181. [Google Scholar] [CrossRef]
- Sadovnichii, R.V.; Rozhkov, S.S.; Rozhkova, N.N. The use of shungite processing products in nanotechnology: Geological and mineralogical justification. Smart Nanocomp 2016, 7, 111–119. [Google Scholar]
- Garvie, L.A.J.; Buseck, P.R. Carbonaceous materials in the acid residue from the Orgueil carbonaceous chondrite meteorite. Meteor. Planet. Sci. 2006, 41, 633–642. [Google Scholar] [CrossRef]
- Taskaev, S.; Skokov, K.; Khovaylo, V.; Donner, W.; Faske, T.; Dudorov, A.; Gorkavyi, N.; Muratov, D. S.; Savosteenko, G.; Dyakonov, A.; Baek, W.; Kuklin, A.; Avramov, P.; Gutfleisc, O. Exotic carbon microcrystals in meteoritic dust of the Chelyabinsk superbolide: Experimental investigations and theoretical scenarios of their formation. Eur. Phys. J. Plus 2022, 137, 562. [Google Scholar] [CrossRef]
- Ivanovskii, AL. Graphynes and graphdyines. Prog. Solid State Chem. 2013, 41, 1–19. [Google Scholar] [CrossRef]
- Pauling, L. The Nature of the Chemical Bond; Cornell University Press, 1960. [Google Scholar]
- Locke, W. Introduction to Molecular Orbital Theory. 1997. retrieved May 18, 2005.
- March, N.H. Electron Density Theory of Atoms and Molecules; Academic Press, 1992. [Google Scholar]
- Frenking, G.; Shail, S. The Chemical Bond. Fundamental Aspects of Chemical Bonding; John Wiley and Sons, 2014. [Google Scholar]
- Frenking, G..; Shail, S. The Chemical Bond. Chemical Bonding Across the Periodic Table; John Wiley and Sons, 2014. [Google Scholar]
- Bader, R.F.W. Atoms in Molecules - A Quantum Theory; Oxford University Press: London, 1990. [Google Scholar]
- Gazquez, J.L.; del Campo, J.M.; Trickey, S.B.; Alvarez-Mendez, R.J.; Vela, A. Analysis of generalized gradient approximation for exchange energy. In Concepts and Methods in Modern Theoretical Chemistry. Vol. 1. Electronic Structure and Reactivity; Ghosh, S.K., Chattaraj, P.K., Eds.; CRC Press, Taylor and Francis Group: Boca Raton, 2013; pp. 295–307. [Google Scholar]
- Scherer, W.; Fisher, A.; Eickerling, G. The experimental density perspectives of chemical bonding. In The Chemical Bond: Fundamental Aspects of Chemical Bonding; Frenking, G., Shaik, S., Eds.; Wiley-VCH Verlag GmbH & Co. KGaA, 2014; pp. 309–343. [Google Scholar]
- Sanderson, R.T. Chemical Bonds and Bond Energy; Academic Press: New York, London, 1976. [Google Scholar]
- Darwent, B. de B. National Standard Reference Data Series; National Bureau of Standards 31: Washington, DC, 1970. [Google Scholar]
- Schreiner, P.R.; Chernish, L.V.; Gunchenko, P.A.; Tikhonchuk, E.Yu.; Hausmann, H.; Serafin, M.; Schlecht, S.; Dahl, J.E.P.; Carlson, R.M.K.; Fokin, A.A. Overcoming lability of extremely long alkane carbon–carbon bonds through dispersion forces. Nature 2011, 477, 308–311. [Google Scholar] [CrossRef]
- Mascal, M.; Hafezi, N.; Meher, N.K.; Fettinger, J.C. Oxatriquinane and oxatriquinacene: Extraordinary oxonium ions. JACS 2008, 130, 13532–13533. [Google Scholar] [CrossRef]
- Lane, J.R.; Contreras-García, J.; Piquemal, J.-P.; Miller, B.J. Are bond critical points really critical for hydrogen bonding? J. Chem. Theory Comput. 2013, 9, 3263–3266. [Google Scholar] [CrossRef]
- Boguslawski, K.; Tecmer, P.; Legeza, O.; Reiher, M. Entanglement measures for single- and multireference correlation effects. J. Phys. Chem. Lett. 2012, 3, 3129–3135. [Google Scholar] [CrossRef] [PubMed]
- Sheka, E F; Popova, N A; Popova, V A. Physics and chemistry of graphene. Emergentness, magnetism, mechanophysics and mechanochemistry. Phys. Usp. 2018, 61, 645–691. [Google Scholar] [CrossRef]
- Mayer, I. Covalent bonding: the role of exchange effects. J. Phys. Chem. A 2014, 118, 2543–2546. [Google Scholar] [CrossRef] [PubMed]
- Sheka, E.F. Stretching and breaking of chemical bonds, correlation of electrons, and radical properties of covalent species. Adv. Quant. Chem. 2015, 70, 111–161. [Google Scholar]
- Zayets, V. A. CLUSTER-Z1: Quantum-Chemical Software for Calculations in the s,p-Basis; Inst. Surf. Chem. Nat. Ac. Sci. of Ukraine; Kiev, 1990. [Google Scholar]
- Berzigiyarov, P.K.; Zayets, V.A.; Ginzburg, I.Ya.; et al. NANOPACK: Parallel codes for semiempirical quantum chemical calculations of large systems in the sp- and spd-basis. Int. J. Quantum Chem. 2002, 88, 449–462. [Google Scholar] [CrossRef]
- Dewar, M.J.S.; Zoebisch, E.G.; Healey, E.F.; Stewart, J.J.P. AM1: A new general- purpose quantum mechanical molecular model. JACS 1985, 107, 3902–3909. [Google Scholar] [CrossRef]
- Sheka, E.F. Chemical susceptibility of fullerenes in view of Hartree–Fock approach. Int. J. Quant. Chem. 2007, 107, 2803–2816. [Google Scholar] [CrossRef]
- Sheka, E.F. Fullerenes. Nanochemistry, Nanomagnetism, Nanomedicine, Nanophotonics, CRC Press, Taylor and Francis Group: Boca Raton, 2011.
- Sheka, E.F. Spin Teory of Fullerene; RUDN Press: Moscow, 2025. [Google Scholar]
- Sheka, E.F. Spin Chemical Physics of Graphene; Pan Stanford: Singapore, 2018. [Google Scholar]
- Sheka, E. F. Virtual vibrational spectrometry of stable radicals—necklaced graphene molecules. Nanomat. 2022, 12, 597. [Google Scholar] [CrossRef]
- Sheka, E. F. Spin effects of sp2 nanocarbons in light of unrestricted Hartree-Fock approach and spin-orbit coupling theory. In Quantum Systems in Physics, Chemistry, and Biology: Advances in Concepts and Applications; Tadjer, A., Pavlov, R., Maruani, J., Brändas, E.J., Delgado-Barrio, G., Eds.; Progress in Theoretical Chemistry and Physics; Springer: Switzerland, 2017; Volume 30, pp. 39–63. [Google Scholar]
- Nagase, S. Structure and reactions of compounds containing hevier main group elements. In The Transition State. A Theoretical Approach; Fueno, T., Ed.; Kodansha Ltd: Tokyo, Japan; Gordon and Breach Sci Pubs.: Amsterdam, The Netherlands, 1999; pp. 140–157. [Google Scholar]
- Cordero, B.; Gґomez, V.; Platero-Prats, A.E.; Revґes, M.; Echeverrґıa, J.; Cremades, E.; Barragґan, F.; Alvarez, S. Covalent radii revisited. Dalton Trans 2008, 2832–2838.
- Sheka, E.F. Why sp2-like nanosilicons should not form: Insight from quantum chemistry. Int. J. Quant. Chem. 2013, 113, 612–618. [Google Scholar] [CrossRef]
- Sheka, E.F. Silicene is a material phantom. Nanosystems: Phys. Chem. Math. 2016, 7, 983–1001. [Google Scholar] [CrossRef]
- Gunbas, G.; Hafezi, N.; Sheppard, W.L.; Olmstead, M.M.; Stoyanova, L.V.; Tham, F.S.; Meyer, M.P.; Mascal, M. Extreme oxatriquinanes and a record C–O bond length. Nat. Chem. 2012, 4, 1018–1023. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, R.; Iwamoto, T.; Kira, M. Fused tricyclic disilenes with highly strained Si-Si double bonds: Addition of a Si-Si single bond to a Si-Si double bond. Ang. Chem. Int. Ed. 2006, 45, 6371–6373. [Google Scholar] [CrossRef]
- Gusel’Nikov, L. E.; Flowers, M. C. The thermal decomposition of 1,1-dimethyl-1-silacyclobutane and some reactions of an unstable intermediate containing a silicon-carbon double bond. Chem. Commun. (London). 1967, 864–865. [Google Scholar]
- Brook, A.G.; Abdesaken, F.; Gutekunst, B.; Gutekunst, G.; Kallury, R.K. A solid silaethene: isolation and characterization. J. Chem. Soc. Chem. Comm. 1995, 24, 2473–2670. [Google Scholar] [CrossRef]
- Karni, M.; Apeloig, Y. The quest for a stable silyne, RSi≡CR_. The effect of bulky substituents. Sil. Chem. 2002, 1, 61–66. [Google Scholar]
- Sekiguchi, A.; Kinjo, R.; Ichinohe, M. A stable compound containing a silicon-silicon triple bond. Science 2004, 305, 1755–1757. [Google Scholar] [CrossRef]
- Sheka, E.F. Virtual Free-Radical Polymerization of Vinyl Monomers in View of Digital Twins. Polymers 2023, 15, 2999. [Google Scholar] [CrossRef]
- Sheka, E.F. Digital twins’ kinetics of virtual free-radical copolymerization of vinyl monomers with stable radicals. 2. Styrene. arXiv:2311.02752 [cond-mat.mtrl-sci], 2023.
- Sheka, E.F. Digitalization of free-radical polymerization. Adv. Quant. Chem. 2025, 92, 286–317. [Google Scholar]
- Blanquart, G. Effects of spin contamination on estimating bond dissociation energies of polycyclic aromatic hydrocarbons. J. Quant. Chem. 2015, 115, 796–801. [Google Scholar] [CrossRef]
- Kaplan, I. Problems in DFT with the total spin and degenerate states. Int. J. Quant. Chem. 2007, 107, 2595–2603. [Google Scholar] [CrossRef]
- Jacob, C.R.; Reiher, M. Spin in density-functional theory. Int. J.Quant. Chem. 2012, 112, 3661–3684. [Google Scholar] [CrossRef]
- Kaplan, I. Symmetry properties of the electron density and following from it limits on the KS-DFT applications. Mol. Phys. 2018, 116, 658–665. [Google Scholar] [CrossRef]
- Hachmann, J; Dorando, JJ; Avilés, M; Chan, GK-L. The radical character of the acenes: a density matrix renormalization group study. J. Chem. Phys. 2007, 127, 134309. [Google Scholar] [CrossRef]
- Spethmann, J.; Khanh, N. D.; Yoshimochi, H.; Takagi, R.; Hayami, S.; Motome, Y.; Wiesendanger, R.; Seki, S.; von Bergmann, K. SP-STM study of the multi-Q phases in GdRu2Si2. Phys. Rev. Mat. 2024, 8, 064404. [Google Scholar]
- Gross, L.; et al. Bond-order discrimination by atomic force microscopy. Science 2012, 337, 1326–1329. [Google Scholar] [CrossRef] [PubMed]
- Mistry, A.; et al. The synthesis and STM/AFM imaging of ‘olympicene’ benzo[cd]pyrenes. Chem. Eur. J. 2014, 21, 2011–2018. [Google Scholar] [CrossRef]
- Pavliček, N.; et al. Synthesis and characterization of triangulene. Nature Nanotech 2017, 12, 308–311. [Google Scholar] [CrossRef]
- Van der Lit, J.; et al. Suppression of electron-vibron coupling in graphene nanoribbons contacted via a single atom. Nat. Commn 2013, 4, 2023. [Google Scholar] [CrossRef]
- Mönig, H.; Amirjalayer, S.; Timmer, A.; et al. Quantitative assessment of intermolecular interactions by atomic force microscopy imaging using copper oxide tips. Nature Nanotech. 2018, 13, 371–375. [Google Scholar] [CrossRef]
- Hedberg, K.; Hedberg, L.; Bethune, D. S. lengths in free molecules of buckminsterfullerene, C60, from gas-phase electron diffraction. Science 1991, 234, 410–412. [Google Scholar] [CrossRef]
- Liu, S.; Lu, Y.-J.; Kappes, M. M.; Ibers, J. A. The structure of the C60 molecule: X-ray crystal structure determination of a twin at 110 K. Science 1991, 254, 408–410. [Google Scholar] [CrossRef] [PubMed]
- Leclercq, F.; Damay, P.; Foukani, M.; Chieux, P.; Bellisent-Funnel, M.C.; Rassat, A.; Fabre, C. Precise determination of the molecular geometry in fullerene C60 powder: A study of the structure factor by neutron scattering in a large momentum-transfer range. Phys. Rev. B 1993, 48, 2748–2758. [Google Scholar] [CrossRef]
- Yanonni, C.S.; Bernier, P.P.; Bethune, D.S.; Meijer, G.; Salem, J.K. NMR determination of the bond lengths in C60. J. Am. Chem. Soc. 1991, 113, 3190–3192. [Google Scholar] [CrossRef]
- Slovokhotov, Yu.L.; Moskaleva, I.V.; Shil’nikov, V.I. Molecular and crystal structures of C-60 derivatives: CSD statistics and theoretical modeling. Mol. Mater. 1996, 8, 117–124. [Google Scholar]
- Li, Y. Z.; Chander, M.; Patrin, J. C.; Weaver, J. H.; Chibante, L. P. F.; Smalley, R. E. Adsorption of individual С60 molecules on Si(111). Phys. Rew. B 1992, 1, 13837–13840. [Google Scholar] [CrossRef] [PubMed]
- Moriarty, P. J. Fullerene adsorption on semiconductor surfaces. Surf. Sci. Rep. 2010, 65, 175–227. [Google Scholar] [CrossRef]
- Sheka, E. F. ‘Chemical’ portrait of fullerenes. J. Struct. Chem. 2006, 47, 593–599. [Google Scholar] [CrossRef]
- Sheka, E.F.; Chernozatonskii, L.A. Bond length effect on odd electrons behavior in single-walled carbon nanotubes. J. Phys. Chem. C. 2007, 111, 10771–10779. [Google Scholar] [CrossRef]
- Sheka, E.F.; Chernozatonskii, L.A. Broken symmetry approach and chemical susceptibility of carbon nanotubes. Int. Journ. Quant. Chem. 2010, 110, 1466–1480. [Google Scholar] [CrossRef]
- Sheka, E.F.; Chernozatonskii, L. A. Broken spin symmetry approach to chemical reactivity and magnetism of graphenium species. J. Exp. Theor. Phys. 2010, 110, 121–132. [Google Scholar] [CrossRef]
- Gao, X.; Zhou, Z.; Zhao, Y.; Nagase, S.; Zhang, S. B.; Chen, Z. Comparative study of carbon and BN nanographenes: Ground electronic states and energy gap engineering. J. Phys. Chem. A 2008, 112, 12677–12682/. [Google Scholar] [CrossRef]
- Sheka, E.F. Molecular theory of graphene chemical modification. In Graphene Science Handbook: Mechanical and Chemical Properties, Vol.4; Aliofkhazraei, M., Ali, N., Miln, W.I., Ozkan, C. S., Mitura, S., Gervasoni, J., Eds.; CRC Press, Taylor and Francis Group: Boca Raton; pp. 312–338.
- Sheka, E F; Popova, N A; Popova, V A. Physics and chemistry of graphene. Emergentness, magnetism, mechanophysics and mechanochemistry. Phys. Usp. 2018, 61, 645–691. [Google Scholar] [CrossRef]
- Li, Y.; Xu, L.; Liu, H.; Li, Y. Graphdiyne and graphyne: From theoretical predictions to practical construction. Chem. Soc. Rev. 2014, 43, 2572–2586. [Google Scholar] [CrossRef]
- Liu, Z.; Yu, G.; Yao, H.; Liu, L.; Jiang, L.; Zheng, Y. A simple tight-binding model for typical graphyne structures. New J. Phys. 2014, 14, 113007. [Google Scholar] [CrossRef]
- Peng, Q.; Dearden, A.K.; Crean, J.; Han, L.; Liu, S.; Wen, X.; De, S. New materials graphyne, graphdiyne, graphone, and graphane: Review of properties, synthesis, and application in nanotechnology. Nanotech. Sci. Appl. 2014, 7, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Chalifoux, W.A.; Tykwinski, R.R. Synthesis of polyynes to model the sp-carbon allotrope carbine. Nature Chem. 2010, 2, 967–971. [Google Scholar] [CrossRef]
- Shi, L.; Rohringer, P.; Suenaga, K. Confined linear carbon chains as a route to bulk carbine. Nature Mat. 2016, 15, 634–639. [Google Scholar] [CrossRef] [PubMed]
- Xi, J.; Wang, D.; Shua, Z. Electronic properties and charge carrier mobilities of graphynes and graphdiynes from first principles. WIREs Comput. Mol. Sci. 2015, 5, 215–227. [Google Scholar] [CrossRef]
- Ahrendt, K. A.; Borths, C. J.; MacMillan, D. W. C. New strategies for organic catalysis: The first highly enantioselective organocatalytic Diels-Alder reaction. J. Am. Chem. Soc. 2000, 122, 4243–4244. [Google Scholar] [CrossRef]
- MacMillan, D.W.C. The advent and development of organocatalysis. Nature, 2008, 455|18 September 2008, 304-308. [CrossRef] [PubMed]
- Xiang, S-H.; Tan, B. Advances in asymmetric organocatalysis over the last 10 years. Nature Comm. 2020, 11, 3786. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.H.; Ahn, B.K.; Lee, J.Y.; Kim, H.S.; Ha, D.-C. Organocatalysis for the asymmetric Michael addition of cycloketones and α, β-unsaturated nitroalkenes. Catalysts 2021, 11, 1004. [Google Scholar] [CrossRef]
- Lee, J.; Kim, K.H.; Kwon, E.E. Biochar as a catalyst. Renew. Sustain. Energy Rev. 2017, 77, 70–79. [Google Scholar] [CrossRef]
- Donet, J.-B.; Bansal, R.C.; Wang, M.-J. Carbon Black. Science and Technology; Marcel Dekker: New York, NY, USA; Basel, Switzerland, 1993. [Google Scholar]
- Francisco, R.R. The role of carbon materials in heterogeneous catalysis. Carbon 1998, 36, 159–175. [Google Scholar] [CrossRef]
- Radovich, L.R. Carbon materials as adsorbents in aqueous solutions. In Chemistry and Physics of Carbon; Marcel Dekker: New York, NY, USA, 2001; Volume 27, pp. 227–406. [Google Scholar]
- Toyoda, M.; Tsumura, T.; Tryba, B.; Mozia, S.; Janus, M.; Morawski, A.W.; Inagaki, M. Carbon materials in photocatalysis. In Chemistry and Physics of Carbon; Radovich, L.R., Ed.; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2013; pp. 171–267. [Google Scholar]
- Machado, B.; Serp, P. Graphene-based materials for catalysis. Catal. Sci. Technol. 2012, 2, 54–75. [Google Scholar] [CrossRef]
- Serp, P.; Machado, B. (Eds.) Nanostructured Carbon Materials for Catalysis; Royal Society of Chemistry: Croydon, UK, 2015. [Google Scholar]
- Bandosz, T.J. Surface chemistry of carbon materials. Spectroscopic methods. In Carbon Materials for Catalysis; Serp, P., Figueiredo, J.L., Eds.; Wiley: Hoboken, NJ, USA, 2009; pp. 45–92. [Google Scholar]
- Bandosz, T.J. Nanoporous carbons: Looking beyond their perception as adsorbents, catalyst supports and supercapacitors. Chem. Rec. 2016, 16, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Matos, I.; Bernardo, M.; Fonseca, I. Porous Carbon: A versatile material for catalysis. Catal. Today 2017, 285, 194–203. [Google Scholar] [CrossRef]
- Yam, K.M.; Guo, N.; Jiang, Z.; Li, S.; Zhang, C. Graphene-based heterogeneous catalysis: Role of graphene. Catalysts 2020, 10, 53. [Google Scholar] [CrossRef]
- Salvio, R.; Molitermo, M.; Bella, M. Alkynes in organocatalysis. Asian J. Org. Chem. 2014, 3, 340–351. [Google Scholar] [CrossRef]
- Hopkinson, M.N.; Richter, C.; Schedler, M.; Glorius, F. An overview of N-heterocyclic carbenes. Nature 2014, 510, 485–406. [Google Scholar] [CrossRef]
- Fan, X.; Zhang, G.; Zhang, F. Multiple roles of graphene in heterogeneous catalysis. Chem. Soc. Rev. 2015, 44, 3023–3035. [Google Scholar] [CrossRef] [PubMed]
- Guo, N.; Yam, K.M.; Zhang, C. Substrate engineering of graphene reactivity: Towards high-performance graphene-based catalysts. npj 2D Mater. Appl. 2018, 2, 1. [Google Scholar] [CrossRef]
- Deng, D.; Novoselov, K.S.; Fu, Q.; Zheng, N.; Tian, Z.; Bao, X. Catalysis with two-dimensional materials and their heterostructures. Nat. Nanotech 2016, 11, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Fu, Q.; Zhang, Y.-Y.; Weng, X.; Li, H.; Chen, M.; Jin, L.; Dong, A.; Mu, R.; Jiang, P.; et al. Graphene cover-promoted metal-catalyzed reactions. Proc. Natl. Acad. Sci. USA 2014, 111, 17023–17028. [Google Scholar] [CrossRef] [PubMed]
- Sutter, P.; Sadowski, J.T.; Sutter, E.A. Chemistry under cover: Tuning metal–graphene interaction by Reactive Intercalation. J. Am. Chem. Soc. 2010, 132, 8175–8179. [Google Scholar] [CrossRef]
- Sheka, E.F. Virtual vibrational spectrometry of stable radicals—necklaced graphene molecules. Nanomat. 2022, 12, 597. [Google Scholar] [CrossRef] [PubMed]
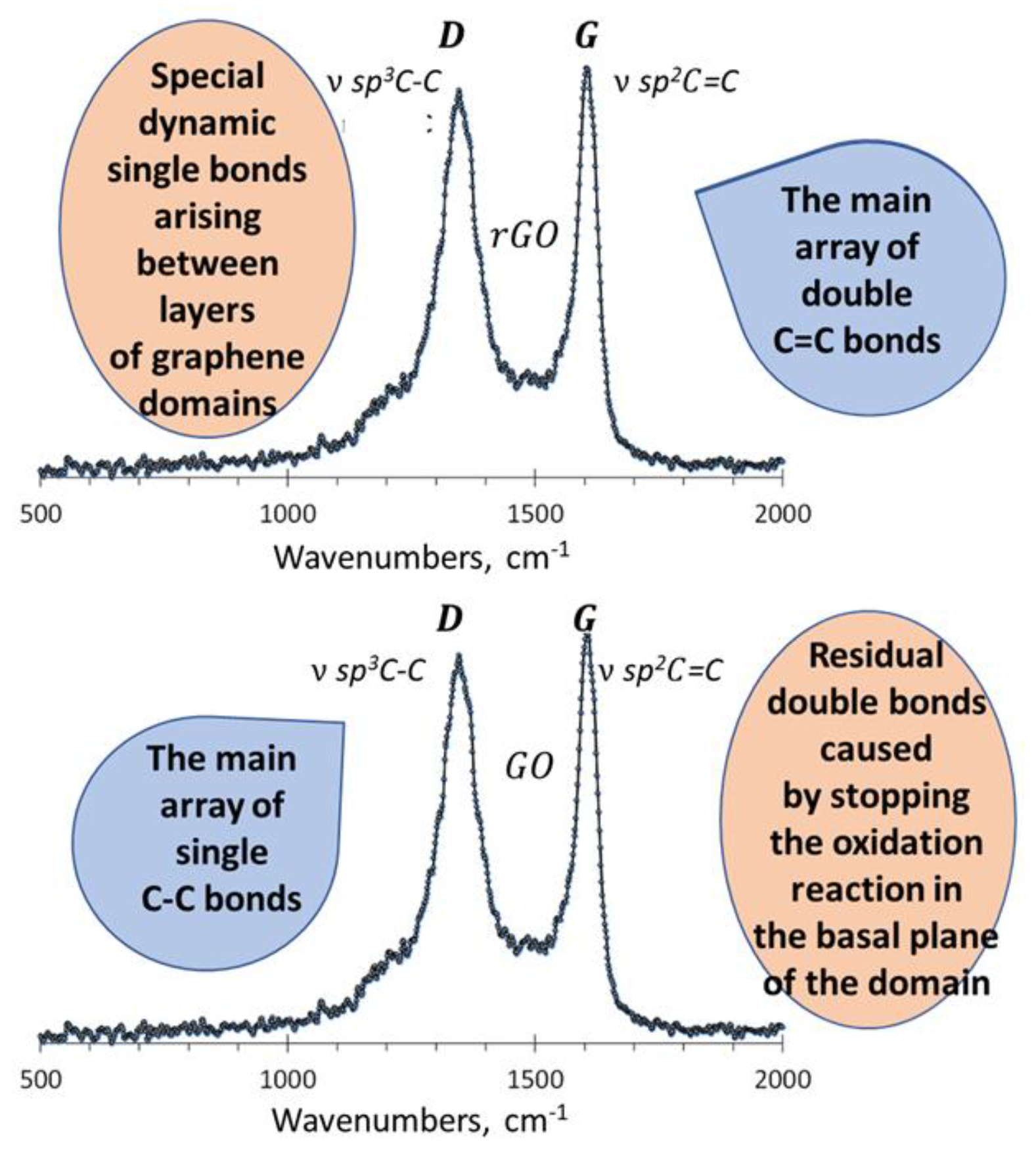
- Sheka, E.F. Digital Twins solve the mystery of Raman spectra of parental and reduced graphene oxides. Nanomat. 2022, 12, 4209. [Google Scholar] [CrossRef] [PubMed]
- Bansal, R.C.; Donnet, J.-B. Surface groups on carbon blacks. In Carbon Black. Science and Technology; Donet, J.-B., Bansal, R.C., Wang, M.-J., Eds.; Marcel Dekker: New York, NY, USA; Basel, Switzerland, 1993; pp. 152–181. [Google Scholar]
- Navalon, S.; Dhakshinamoorthy, A.; Alvaro, M.; Antonietti, M.; García, H. Active sites on graphene-based materials as metal-free catalysts. Chem. Soc. Rev. 2017, 46, 4501–4529. [Google Scholar] [CrossRef] [PubMed]
- Sheka, E.F. Graphene oxyhydride catalysts in view of spin radical chemistry. Materials 2020, 13, 565. [Google Scholar] [CrossRef] [PubMed]
- Radovich, L.R. Physicochemical properties of carbon materials: A brief overview. In Carbon Materials for Catalysis; Serp, P., Figueiredo, J. L., Eds.; Wiley, Hoboken, 2009; pp. 1–44. [Google Scholar]
- Rideal, K.; Wright, W.M. Low temperature oxidation at charcoal surfaces. Part I. The behaviour of charcoal in the absence of promoters. J. Chem. Soc. Trans. 1925, 127, 1347–1357. [Google Scholar] [CrossRef]
- Lonkar, S.P.; Abdala, A.A. Applications of graphene in catalysis. J. Thermodyn. Catal 2014, 5, 1–6. [Google Scholar]
- Chung, C; Kim, YK; Shin, D; Ryoo, SR; Hong, BH; et al. Biomedical applications of graphene and graphene oxide. Acc Chem Res 2013, 46, 2211–2224. [Google Scholar] [CrossRef]
- Rozhkov, S.; Goryunov, A.; Rozhkova, N. Molecular serum albumin unmask nanobio properties of molecular graphenes in shungite carbon nanoparticles. Int. J. Mol. Sci. 2024, 25, 2465. [Google Scholar] [CrossRef] [PubMed]
- Sadovnichii, R.V.; Rozhkova, N.N. The minerals associations of carbon-rich shungite rocks of the Maksovo deposit (the onega structure). Trans. KarRC RAS 2014, 1, 148–158. [Google Scholar]
- Sadovnichii, R.V.; Mikhaylina, A.A.; Rozhkova, N.N.; Inina, I.S. The Morphological and structural features of quartz of shungite rocks from Maksovo deposit. Trans. KarRC RAS 2016, 2, 73–88. [Google Scholar]
- Sadovnichii, R.V.; Rozhkov, S.S.; Rozhkova, N.N. The use of shungite processing products in nanotechnology: geological and mineralogical justification. Smart Nanocomp 2016, 7, 111–120. [Google Scholar]
- Chen, Y.; Guo, F.; Jachak, A.; Kim, S. P.; Datta, D.; Liu, J.; Kulaots, I.; Vaslet, C.; Jang, H. D.; Huang, J.; et al. Aerosol Synthesis of Cargo-Filled Graphene Nanosacks. Nano Lett. 2012, 12, 1996–2002. [Google Scholar] [CrossRef] [PubMed]
- Campra, P. Graphene oxide detection in aqueous suspension. Observational study in optical and electron microscopy. June 28, 2021. Available online: https://www.docdroid.net/rNgtxyh/microscopia-devial-.
- Yong, R.O. Scanning and transmission microscopy reveals graphene oxide in CoV-19 vaccines. Acta Sci. Med. Sci. 2022, 6, 98–111. [Google Scholar]
- Sheka, E.F. The triumph of the spin chemistry of fullerene C60 in the light of its free radical copolymerization with vinyl monomers. Int. J. Mol. Sci. 2024, 25, 1317. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.; Lawandy, N.M.; Suuberg, E.M. Fullerene-catalyzed oxidation of organic solvents. En. Fuels 1994, 8, 810–811. [Google Scholar] [CrossRef]
- Bagdasar’yan, K.S. Teoriya Radikal’noi Polimerizatsii (Free_Radical Polymerization Theory); Nauka: Moscow, Russia, 1966. [Google Scholar]
- Pross, A. Theoretical and Physical Principles of Organic Reactivity; Wiley: New York, NY, USA, 1995. [Google Scholar]
- Sheka, E.F. Intermolecular interaction in C60-based donor acceptor complexes. Int. J. Quant. Chem. 2004, 100, 388–406. [Google Scholar] [CrossRef]
- Sheka, E.F. Donor-acceptor origin of fullerene C60 dimerization. Int. J. Quant. Chem. 2007, 107, 2361–2371. [Google Scholar] [CrossRef]
- Neeman, E.M.; Ramón, J.; Moreno, A.; Huet, T.R. The gas phase structure of -pinene, a main biogenic volatile organic compound. J. Chem. Phys. 2017, 147, 214305. [Google Scholar] [CrossRef] [PubMed]
- Sheka, E.F. Step-wise computational synthesis of fullerene C60 derivatives. Fluorinated fullerenes C60F2k. J. Exp. Theor. Phys. 2010, 111, 397–414. [Google Scholar] [CrossRef]
- Sheka, E.F. Computational synthesis of hydrogenated fullerenes from C60 to C60H60. J. Mol. Mod. 2011, 17, 1973–1984. [Google Scholar] [CrossRef]
- Sheka, E.F.; Popova, N.A. Odd-electron molecular theory of the graphene hydrogenation. J. Mol. Model. 2012, 18, 3751–3768. [Google Scholar] [CrossRef] [PubMed]
- D. R. Dreyer, S. Park, C. W. Bielawski, R. S. The chemistry of graphene oxide. Chem. Soc. Rev. 2010, 39, 228–240. [CrossRef] [PubMed]
- Chen, D.; Feng, H.; Li, J. Graphene oxide: Preparation, functionalization, and electrochemical applications. Chem. Rev. 2012, 112, 6027−6053. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Bongiorno, A. Origin of the Chemical and kinetic stability of graphene oxide. Sci. Rep. 2013, 3, 2484. [Google Scholar] [CrossRef] [PubMed]
- Sheka, E.F.; Golubev, Y.A.; Popova, N.A. Graphene domain signature of Raman spectra of sp2 amorphous carbons. Nanomat. 2020, 10. [Google Scholar] [CrossRef]
- Sheka, E.F. Metaverse and virtual worlds of science. TCSIT 2024, 9, 103–105. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



