Submitted:
01 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
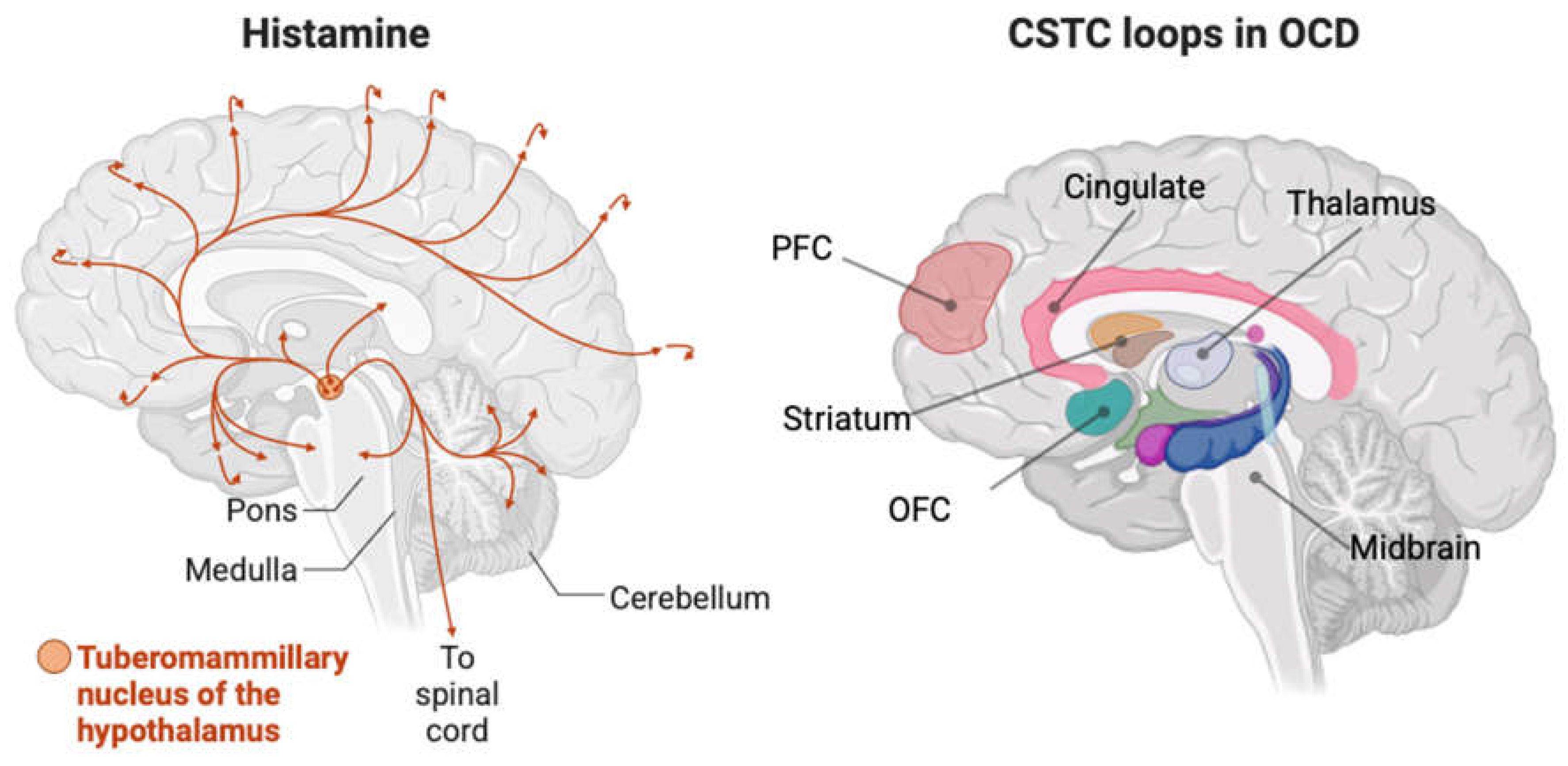
Neuroanatomy of the Brain Histamine System and Its Relevance to OCD Circuits
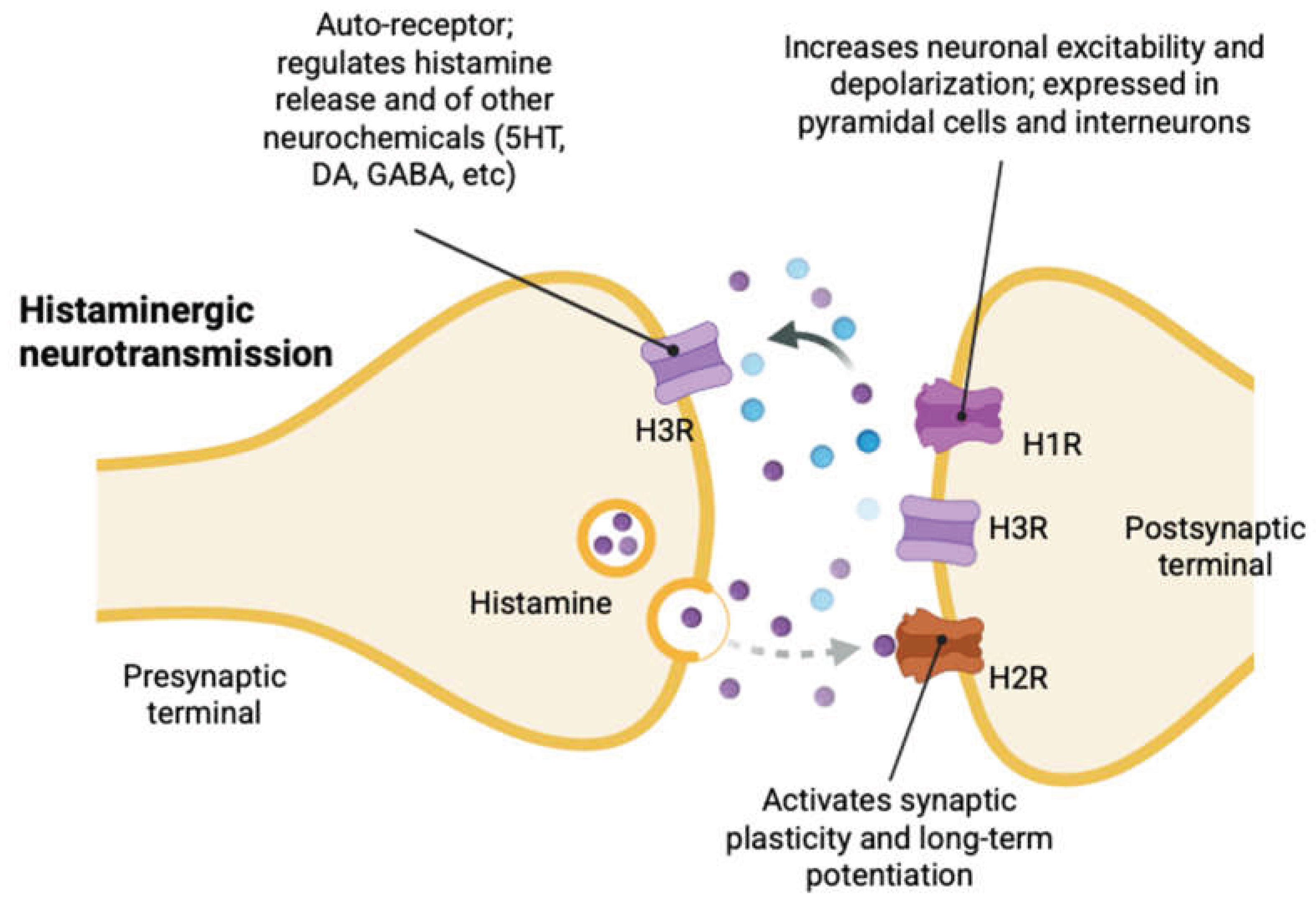
Histamine Receptor Pharmacology and Microcircuit Mechanisms in OCD
Histamine, Uncertainty, and Learning: A Computational Framework for OCD
Developmental and Genetic Considerations: Histamine as a Vulnerability Factor for Compulsivity
Alternative Neuromodulatory Accounts and Why Histamine Might Have Been Overlooked
Translational and Clinical Implications: Histamine as a Candidate Target for Circuit-Informed Investigation
Outstanding Questions for Future Research
Conclusions
References
- Stein, D.J. Obsessive-compulsive disorder. Nat Rev Dis Primers 2019, 5(1), p. 52. [Google Scholar] [CrossRef]
- Calza, J. Altered Cortico-Striatal Functional Connectivity During Resting State in Obsessive-Compulsive Disorder. Front Psychiatry 2019, 10, 319. [Google Scholar] [CrossRef]
- Cotovio, G. Obsessive-compulsive disorder secondary to focal brain lesions: from lesions to networks. medRxiv 2025. [Google Scholar] [CrossRef]
- Baumgarten, H.G.; Grozdanovic, Z. Role of serotonin in obsessive-compulsive disorder. Br J Psychiatry Suppl 1998, 35, 13–20. [Google Scholar] [CrossRef]
- Mota, P.; Pico-Perez, M.; Morgado, P. Mapping dopamine’s role in obsessive-compulsive disorder: A scoping review of neural circuits, brain regions, and behavioral implications. J Psychiatr Res 2025, 194, 11–20. [Google Scholar] [CrossRef]
- Haas, H.; Panula, P. The role of histamine and the tuberomamillary nucleus in the nervous system. Nat Rev Neurosci 2003, 4(2), 121–30. [Google Scholar] [CrossRef]
- Green, M. Histamine in the central nervous system. Proc West Pharmacol Soc 1978, 21, 337–9. [Google Scholar] [PubMed]
- Baronio, D. Histaminergic system in brain disorders: lessons from the translational approach and future perspectives. Ann Gen Psychiatry 2014, 13(1), p. 34. [Google Scholar] [CrossRef]
- Brown, R.E.; Stevens, D.R.; Haas, H.L. The physiology of brain histamine. Prog Neurobiol 2001, 63(6), 637–72. [Google Scholar] [CrossRef] [PubMed]
- Bruckner, R.; Heekeren, H.R.; Nassar, M.R. Understanding learning through uncertainty and bias. Commun Psychol 2025, 3(1), p. 24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Y. Targeting presynaptic H3 heteroreceptor in nucleus accumbens to improve anxiety and obsessive-compulsive-like behaviors. Proc Natl Acad Sci U S A 2020, 117(50), 32155–32164. [Google Scholar] [CrossRef]
- Panula, P.; Nuutinen, S. The histaminergic network in the brain: basic organization and role in disease. Nat Rev Neurosci 2013, 14(7), 472–87. [Google Scholar] [CrossRef]
- Yin, H.H.; Knowlton, B.J. The role of the basal ganglia in habit formation. Nat Rev Neurosci 2006, 7(6), 464–76. [Google Scholar] [CrossRef]
- Gittis, A.H.; Kreitzer, A.C. Striatal microcircuitry and movement disorders. Trends Neurosci 2012, 35(9), 557–64. [Google Scholar] [CrossRef]
- Airaksinen, M.S.; Panula, P. The histaminergic system in the guinea pig central nervous system: an immunocytochemical mapping study using an antiserum against histamine. J Comp Neurol 1988, 273(2), 163–86. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M. The representation of decision variables in orbitofrontal cortex is longitudinally stable. Cell Rep 2024, 43(10), p. 114772. [Google Scholar] [CrossRef]
- Carter, C.S. Anterior cingulate cortex, error detection, and the online monitoring of performance. Science 1998, 280(5364), 747–9. [Google Scholar] [CrossRef] [PubMed]
- Bolam, J.P.; Ellender, T.J. Histamine and the striatum. Neuropharmacology 2016, 106, 74–84. [Google Scholar] [CrossRef]
- Wall, N.R. Differential innervation of direct- and indirect-pathway striatal projection neurons. Neuron 2013, 79(2), 347–60. [Google Scholar] [CrossRef]
- Peng, J.Y. Receptor and Ionic Mechanism of Histamine on Mouse Dorsolateral Striatal Neurons. Mol Neurobiol 2023, 60(1), 183–202. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.X. Histamine Modulation of the Basal Ganglia Circuitry in the Motor Symptoms of Parkinson’s Disease. CNS Neurosci Ther 2025, 31(2), p. e70308. [Google Scholar] [CrossRef]
- Han, S. Histaminergic Control of Corticostriatal Synaptic Plasticity during Early Postnatal Development. J Neurosci 2020, 40(34), 6557–6571. [Google Scholar] [CrossRef] [PubMed]
- Ellenbroek, B.A. Histamine H(3) receptors, the complex interaction with dopamine and its implications for addiction. Br J Pharmacol 2013, 170(1), 46–57. [Google Scholar] [CrossRef]
- Lee, K.H. Histamine modulates thalamocortical activity by activating a chloride conductance in ferret perigeniculate neurons. Proc Natl Acad Sci U S A 2004, 101(17), 6716–21. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.Y.; Kalimo, H.; Panula, P. The histaminergic system in human thalamus: correlation of innervation to receptor expression. Eur J Neurosci 2002, 15(7), 1125–38. [Google Scholar] [CrossRef]
- Weeland, C.J. The thalamus and its subnuclei-a gateway to obsessive-compulsive disorder. Transl Psychiatry 2022, 12(1), p. 70. [Google Scholar] [CrossRef] [PubMed]
- Tasaka, K. Excitatory effect of histamine on the arousal system and its inhibition by H1 blockers. Brain Res Bull 1989, 22(2), 271–5. [Google Scholar] [CrossRef]
- Thakkar, M.M. Histamine in the regulation of wakefulness. Sleep Med Rev 2011, 15(1), 65–74. [Google Scholar] [CrossRef]
- Thangam, E.B. The Role of Histamine and Histamine Receptors in Mast Cell-Mediated Allergy and Inflammation: The Hunt for New Therapeutic Targets. Front Immunol 2018, 9, 1873. [Google Scholar] [CrossRef]
- Tashiro, M. Roles of histamine in regulation of arousal and cognition: functional neuroimaging of histamine H1 receptors in human brain. Life Sci 2002, 72(4-5), 409–14. [Google Scholar] [CrossRef]
- Luo, T.; Leung, L.S. Endogenous histamine facilitates long-term potentiation in the hippocampus during walking. J Neurosci 2010, 30(23), 7845–52. [Google Scholar] [CrossRef]
- Schlicker, E. Modulation of neurotransmitter release via histamine H3 heteroreceptors. Fundam Clin Pharmacol 1994, 8(2), 128–37. [Google Scholar] [CrossRef] [PubMed]
- Pauls, D.L. Obsessive-compulsive disorder: an integrative genetic and neurobiological perspective. Nat Rev Neurosci 2014, 15(6), 410–24. [Google Scholar] [CrossRef] [PubMed]
- Gillan, C.M. Functional neuroimaging of avoidance habits in obsessive-compulsive disorder. Am J Psychiatry 2015, 172(3), 284–93. [Google Scholar] [CrossRef]
- Voon, V. Disorders of compulsivity: a common bias towards learning habits. Mol Psychiatry 2015, 20(3), 345–52. [Google Scholar] [CrossRef]
- Vaghi, M.M. Action-Outcome Knowledge Dissociates From Behavior in Obsessive-Compulsive Disorder Following Contingency Degradation. Biol Psychiatry Cogn Neurosci Neuroimaging 2019, 4(2), 200–209. [Google Scholar] [CrossRef] [PubMed]
- Behrens, T.E. Learning the value of information in an uncertain world. Nat Neurosci 2007, 10(9), 1214–21. [Google Scholar] [CrossRef]
- Mathys, C.D. Uncertainty in perception and the Hierarchical Gaussian Filter. Front Hum Neurosci 2014, 8, 825. [Google Scholar] [CrossRef]
- Fradkin, I. Doubting what you already know: Uncertainty regarding state transitions is associated with obsessive compulsive symptoms. PLoS Comput Biol 2020, 16(2), e1007634. [Google Scholar] [CrossRef]
- Scammell, T.E. Histamine: neural circuits and new medications. Sleep 2019, 42(1). [Google Scholar] [CrossRef]
- Szechtman, H.; Woody, E. Obsessive-compulsive disorder as a disturbance of security motivation. Psychol Rev 2004, 111(1), 111–27. [Google Scholar] [CrossRef]
- Porkka-Heiskanen, T. The effect of REM sleep deprivation on histamine concentrations in different brain areas. Life Sci 1994, 54(22), 1719–26. [Google Scholar] [CrossRef]
- Segalas, C. Sleep disturbances in obsessive-compulsive disorder: influence of depression symptoms and trait anxiety. BMC Psychiatry 2021, 21(1), 42. [Google Scholar] [CrossRef] [PubMed]
- Panula, P.; Sundvik, M.; Karlstedt, K. Developmental roles of brain histamine. Trends Neurosci 2014, 37(3), 159–68. [Google Scholar] [CrossRef] [PubMed]
- Carthy, E.; Ellender, T. Histamine, Neuroinflammation and Neurodevelopment: A Review. Front Neurosci 2021, 15, 680214. [Google Scholar] [CrossRef]
- Ercan-Sencicek, A.G. L-histidine decarboxylase and Tourette’s syndrome. N Engl J Med 2010, 362(20), 1901–8. [Google Scholar] [CrossRef]
- Baldan, L.C. Histidine decarboxylase deficiency causes tourette syndrome: parallel findings in humans and mice. Neuron 2014, 81(1), 77–90. [Google Scholar] [CrossRef]
- Jindachomthong, K. White matter abnormalities in the Hdc knockout mouse, a model of tic and OCD pathophysiology. Front Mol Neurosci 2022, 15, 1037481. [Google Scholar] [CrossRef]
- Yanai, K. Histamine H1 receptors in human brain visualized in vivo by [11C]doxepin and positron emission tomography. Neurosci Lett 1992, 137(2), 145–8. [Google Scholar] [CrossRef]
- Sara, S.J.; Bouret, S. Orienting and reorienting: the locus coeruleus mediates cognition through arousal. Neuron 2012, 76(1), 130–41. [Google Scholar] [CrossRef]
- Howells, F.M.; Stein, D.J.; Russell, V.A. Synergistic tonic and phasic activity of the locus coeruleus norepinephrine (LC-NE) arousal system is required for optimal attentional performance. Metab Brain Dis 2012, 27(3), 267–74. [Google Scholar] [CrossRef]
- Sarter, M. Unraveling the attentional functions of cortical cholinergic inputs: interactions between signal-driven and cognitive modulation of signal detection. Brain Res Brain Res Rev 2005, 48(1), 98–111. [Google Scholar] [CrossRef]
- Huang, Z. Dynamic responses of striatal cholinergic interneurons control behavioral flexibility. Sci Adv 2024, 10(51), p. eadn2446. [Google Scholar] [CrossRef]
- Marder, E. Neuromodulation of neuronal circuits: back to the future. Neuron 2012, 76(1), 1–11. [Google Scholar] [CrossRef]
- Esbenshade, T.A. The histamine H3 receptor: an attractive target for the treatment of cognitive disorders. Br J Pharmacol 2008, 154(6), 1166–81. [Google Scholar] [CrossRef]
- Zou, Y. Pitolisant alleviates brain network dysfunction and cognitive deficits in a mouse model of Alzheimer’s disease. Transl Psychiatry 2025, 15(1), p. 126. [Google Scholar] [CrossRef]
- Li, S.; Yang, J. Pitolisant for treating patients with narcolepsy. Expert Rev Clin Pharmacol 2020, 13(2), 79–84. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



