Submitted:
03 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
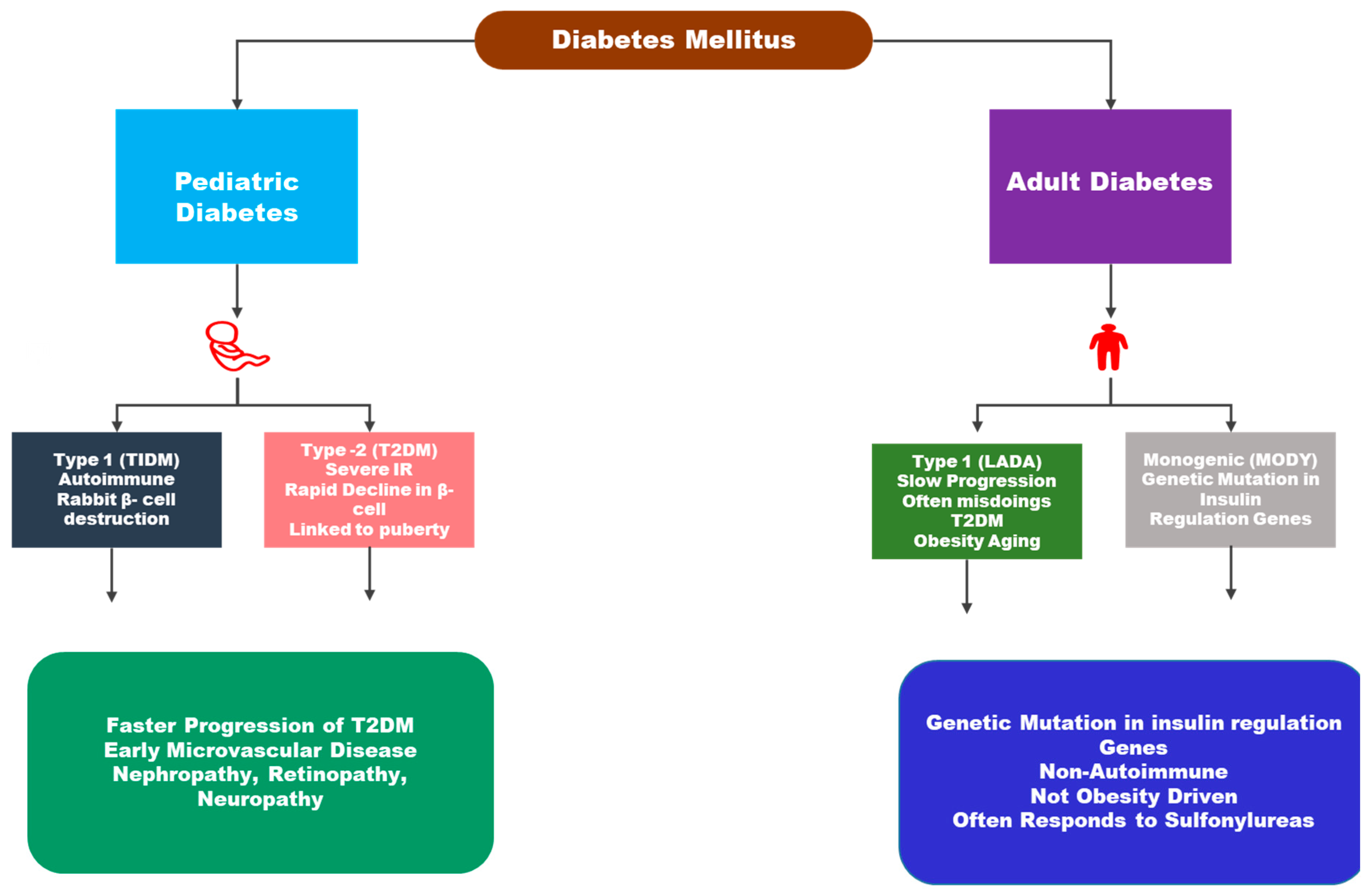
2. Pathophysiological Differences Between Pediatric and Adult Diabetes
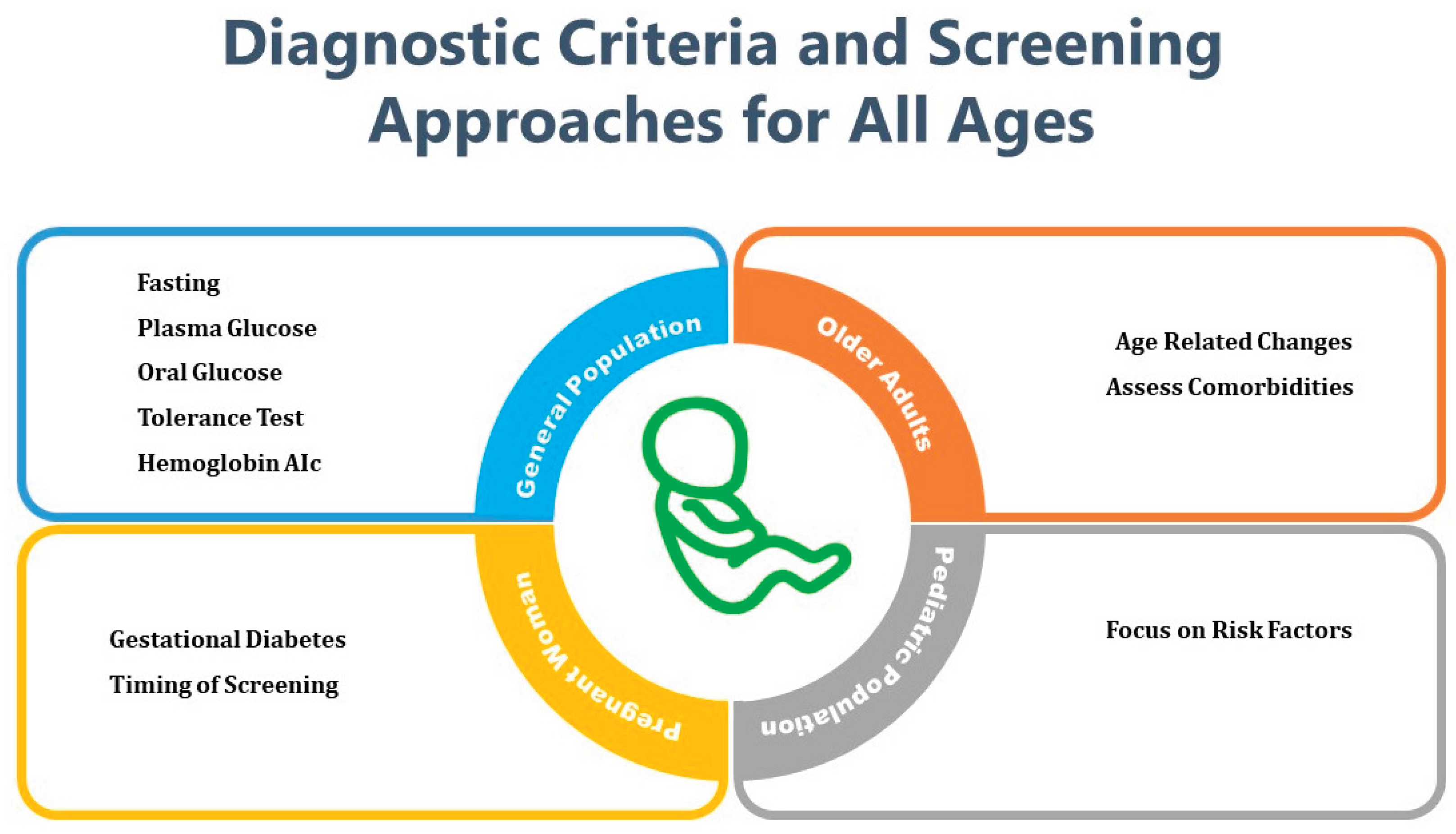
3. Diagnostic Criteria and Screening Approaches for All Ages
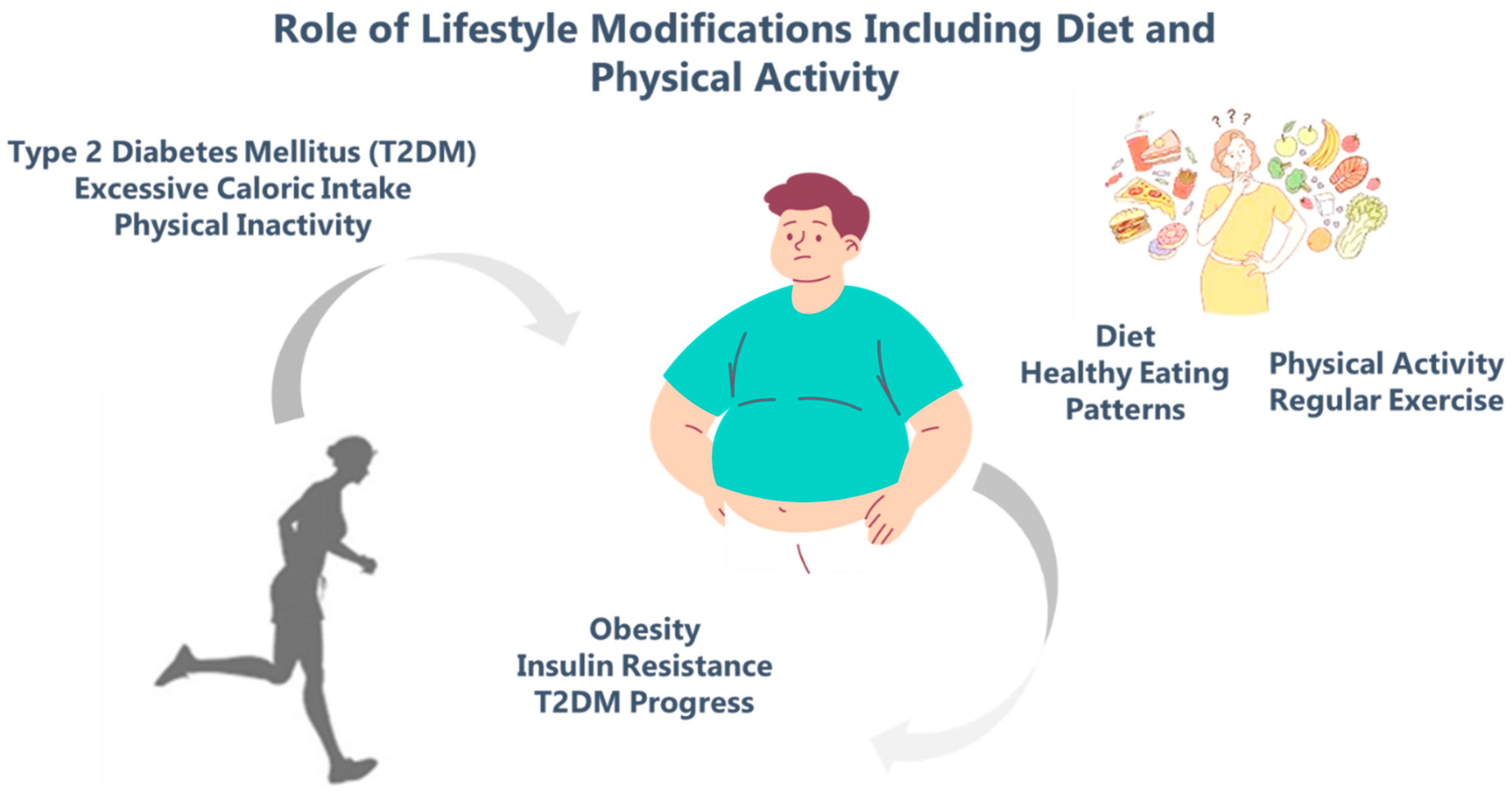
4. Role of Lifestyle Modifications Including Diet and Physical Activity
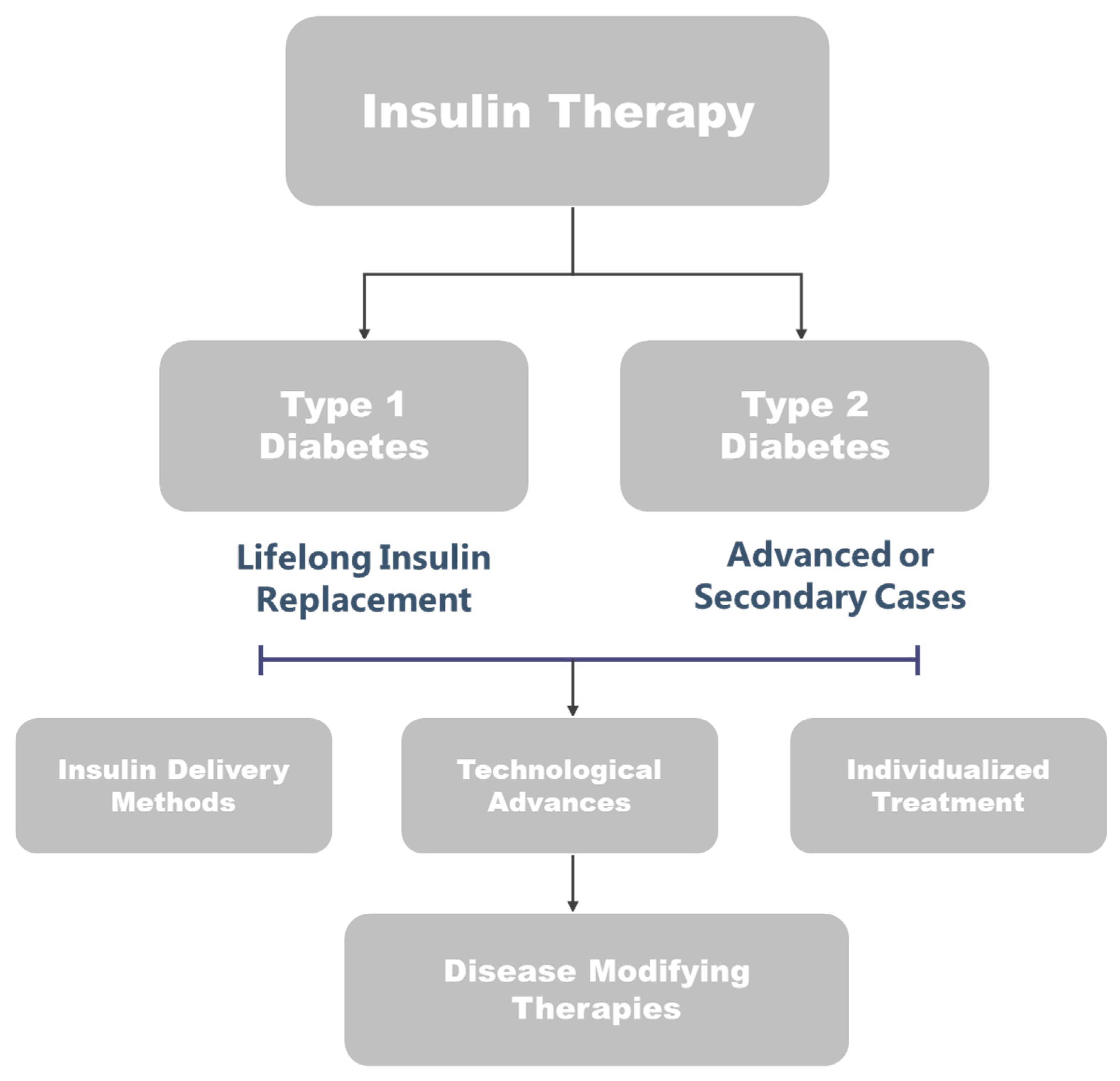
5. Insulin Therapy: Indications and Advances in Pediatric and Adult Diabetes
6. Oral and Injectable Non-Insulin Drugs for Diabetes Management
7. Technology in Diabetes Care: Pumps Continuous Glucose Monitoring and Automated Delivery
8. Transitioning from Pediatric to Adult Diabetes Care Services
9. Psychological and Behavioural Aspects of Diabetes Management
10. Future Directions and Emerging Therapies in Diabetes Treatment
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Diagnosis and Classification of Diabetes Mellitus. In Diabetes Care; 2009.
- Mayer-Davis, E.J.; Lawrence, J.M.; Dabelea, D.; Divers, J.; Isom, S.; Dolan, L.; Imperatore, G.; Linder, B.; Marcovina, S.; Pettitt, D.J.; et al. Incidence Trends of Type 1 and Type 2 Diabetes among Youths. N. Engl. J. Med. 2017. [Google Scholar] [CrossRef]
- Divers, J.; Mayer-Davis, E.J.; Lawrence, J.M.; Isom, S.; Dabelea, D.; Dolan, L.; Imperatore, G.; Marcovina, S.; Pettitt, D.J.; Pihoker, C.; et al. Trends in Incidence of Type 1 and Type 2 Diabetes Among Youths — Selected Counties and Indian Reservations, United States. MMWR. Morb. Mortal. Wkly. Rep. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ogle, G.D.; Wang, F.; Haynes, A.; Gregory, G.A.; King, T.W.; Deng, K.; Dabelea, D.; James, S.; Jenkins, A.J.; Li, X.; et al. Global Type 1 Diabetes Prevalence, Incidence, and Mortality Estimates 2025: Results from the International Diabetes Federation Atlas, 11th Edition, and the T1D Index Version 3.0. Diabetes Res. Clin. Pract. 2025, 225, 112277. [Google Scholar] [CrossRef]
- Roep, B.O.; Thomaidou, S.; van Tienhoven, R.; Zaldumbide, A. Type 1 Diabetes Mellitus as a Disease of the β-Cell (Do Not Blame the Immune System?). Nat. Rev. Endocrinol. 2021. [Google Scholar] [CrossRef]
- Rider, J.M. Treatment Considerations and Options for the Management of Diabetes Mellitus. J. Pharm. Pract. 2004. [Google Scholar] [CrossRef]
- Lascar, N.; Brown, J.; Pattison, H.; Barnett, A.H.; Bailey, C.J.; Bellary, S. Type 2 Diabetes in Adolescents and Young Adults. Lancet Diabetes Endocrinol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Gangwar, R.; Ahmad Zargar, A.; Kumar, R.; Sharma, A. Prevalence of Diabetes in India: A Review of IDF Diabetes Atlas 10th Edition. Curr. Diabetes Rev. 2023. [Google Scholar] [CrossRef]
- Hossain, M.J.; Al-Mamun, M.; Islam, M.R. Diabetes Mellitus, the Fastest Growing Global Public Health Concern: Early Detection Should Be Focused. Heal. Sci. Reports 2024. [Google Scholar] [CrossRef]
- Wilmot, E.; Idris, I. Early Onset Type 2 Diabetes: Risk Factors, Clinical Impact and Management. Ther. Adv. Chronic Dis. 2014. [Google Scholar] [CrossRef]
- Brauer, M.; Roth, G.A.; Aravkin, A.Y.; Zheng, P.; Abate, K.H.; Abate, Y.H.; Abbafati, C.; Abbasgholizadeh, R.; Abbasi, M.A.; Abbasian, M.; et al. Global Burden and Strength of Evidence for 88 Risk Factors in 204 Countries and 811 Subnational Locations, 1990–2021: A Systematic Analysis for the Global Burden of Disease Study 2021. Lancet 2024, 403, 2162–2203. [Google Scholar] [CrossRef] [PubMed]
- Chiang, J.L.; Maahs, D.M.; Garvey, K.C.; Hood, K.K.; Laffel, L.M.; Weinzimer, S.A.; Wolfsdorf, J.I.; Schatz, D. Type 1 Diabetes in Children and Adolescents: A Position Statement by the American Diabetes Association. In Diabetes Care; 2018. [Google Scholar]
- Atkinson, M.A.; Eisenbarth, G.S.; Michels, A.W. Type 1 Diabetes. Lancet 2014. [Google Scholar] [CrossRef] [PubMed]
- Bobadilla-Olaje, G.F.; Calleja-López, J.R.T.; Ruibal-Tavares, E.; Rivera-Rosas, C.N.; Aguilera-Duarte, L.J. Maturity-Onset Diabetes of the Young (MODY). Rev. Mex. Pediatr. 2022. [Google Scholar] [CrossRef]
- Soheilipour, F.; Kasbi, N.A.; Imankhan, M.; Eskandari, D. Complications and Treatment of Early-Onset Type 2 Diabetes. Int. J. Endocrinol. Metab. 2023, 21. [Google Scholar] [CrossRef]
- Kautzky-Willer, A.; Harreiter, J.; Pacini, G. Sex and Gender Differences in Risk, Pathophysiology and Complications of Type 2 Diabetes Mellitus. Endocr. Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Lotstein, D.S.; Seid, M.; Klingensmith, G.; Case, D.; Lawrence, J.M.; Pihoker, C.; Dabelea, D.; Mayer-Davis, E.J.; Gilliam, L.K.; Corathers, S.; et al. Transition from Pediatric to Adult Care for Youth Diagnosed with Type 1 Diabetes in Adolescence. Pediatrics 2013. [Google Scholar] [CrossRef]
- Pacifico, L.; Poggiogalle, E.; Cantisani, V.; Menichini, G.; Ricci, P.; Ferraro, F.; Chiesa, C. Pediatric Nonalcoholic Fatty Liver Disease: A Clinical and Laboratory Challenge. World J. Hepatol. 2010. [Google Scholar] [CrossRef]
- Elsayed, N.A.; Aleppo, G.; Aroda, V.R.; Bannuru, R.R.; Brown, F.M.; Bruemmer, D.; Collins, B.S.; Hilliard, M.E.; Isaacs, D.; Johnson, E.L.; et al. 2. Classification and Diagnosis of Diabetes: Standards of Care in Diabetes—2023. Diabetes Care 2023. [Google Scholar] [CrossRef]
- Sacks, D.B. A1C versus Glucose Testing: A Comparison. In Diabetes Care; 2011. [Google Scholar]
- Sacks, D.B.; Arnold, M.; Bakris, G.L.; Bruns, D.E.; Horvath, A.R.; Lernmark, Å.; Metzger, B.E.; Nathan, D.M.; Kirkman, M.S. Guidelines and Recommendations for Laboratory Analysis in the Diagnosis and Management of Diabetes Mellitus. Diabetes Care 2023. [Google Scholar] [CrossRef]
- Elliott, T.L.; Pfotenhauer, K.M. Classification and Diagnosis of Diabetes. Prim. Care - Clin. Off. Pract. 2022. [Google Scholar] [CrossRef]
- Rodbard, H.W. Diabetes Screening, Diagnosis, and Therapy in Pediatric Patients with Type 2 Diabetes CME/CE. In MedGenMed Medscape Gen. Med.; 2008. [Google Scholar]
- Seino, Y.; Nanjo, K.; Tajim, N.; Kadowaki, T.; Kashiwagi, A.; Araki, E.; Ito, C.; Inagaki, N.; Iwamoto, Y.; Kasuga, M.; et al. Report of the Committee on the Classification and Diagnostic Criteria of Diabetes Mellitus. J. Diabetes Investig. 2010. [Google Scholar] [CrossRef]
- Wang, H.; Li, N.; Chivese, T.; Werfalli, M.; Sun, H.; Yuen, L.; Hoegfeldt, C.A.; Elise Powe, C.; Immanuel, J.; Karuranga, S.; et al. IDF Diabetes Atlas: Estimation of Global and Regional Gestational Diabetes Mellitus Prevalence for 2021 by International Association of Diabetes in Pregnancy Study Group’s Criteria. Diabetes Res. Clin. Pract. 2022. [Google Scholar] [CrossRef]
- Davidson, M.B.; Schriger, D.L. Effect of Age and Race/Ethnicity on HbA1c Levels in People without Known Diabetes Mellitus: Implications for the Diagnosis of Diabetes. Diabetes Res. Clin. Pract. 2010. [Google Scholar] [CrossRef] [PubMed]
- Abebe, K.Z.; Scifres, C.; Simhan, H.N.; Day, N.; Catalano, P.; Bodnar, L.M.; Costacou, T.; Matthew, D.; Illes, A.; Orris, S.; et al. Comparison of Two Screening Strategies for Gestational Diabetes (GDM2) Trial: Design and Rationale. Contemp. Clin. Trials 2017. [Google Scholar] [CrossRef]
- Dallo, F.J.; Weller, S.C. Effectiveness of Diabetes Mellitus Screening Recommendations. Proc. Natl. Acad. Sci. U. S. A., 2003. [Google Scholar] [CrossRef]
- Wing, R.R.; Goldstein, M.G.; Acton, K.J.; Birch, L.L.; Jakicic, J.M.; Sallis, J.F.; Smith-West, D.; Jeffery, R.W.; Surwit, R.S. Behavioral Science Research in Diabetes: Lifestyle Changes Related to Obesity, Eating Behavior, and Physical Activity. In Diabetes Care; 2001. [Google Scholar]
- Comprehensive Medical Evaluation and Assessment of Comorbidities: Standards of Care in Diabetes—2024. Diabetes Care 2024. [CrossRef]
- Tuomilehto, J.; Lindström, J.; Eriksson, J.G.; Valle, T.T.; Hämäläinen, H.; Ilanne-Parikka, P.; Keinänen-Kiukaanniemi, S.; Laakso, M.; Louheranta, A.; Rastas, M.; et al. Prevention of Type 2 Diabetes Mellitus by Changes in Lifestyle among Subjects with Impaired Glucose Tolerance. N. Engl. J. Med. 2001. [Google Scholar] [CrossRef] [PubMed]
- Reduction of the Incidence of Type 2 Diabetes with Lifestyle Intervention or Metformin. In Int. Urol. Nephrol.; 2002.
- Esposito, K.; Maiorino, M.I.; Ceriello, A.; Giugliano, D. Prevention and Control of Type 2 Diabetes by Mediterranean Diet: A Systematic Review. Diabetes Res. Clin. Pract. 2010. [Google Scholar] [CrossRef]
- Ross, R.; Chaput, J.P.; Giangregorio, L.M.; Janssen, I.; Saunders, T.J.; Kho, M.E.; Poitras, V.J.; Tomasone, J.R.; El-Kotob, R.; McLaughlin, E.C.; et al. Canadian 24-Hour Movement Guidelines for Adults Aged 18-64 Years and Adults Aged 65 Years or Older: An Integration of Physical Activity, Sedentary Behaviour, and Sleep. In Proceedings of the Applied physiology, nutrition, and metabolism = Physiologie appliquee, nutrition et metabolisme, 2020. [Google Scholar]
- Cardiovascular Effects of Intensive Lifestyle Intervention in Type 2 Diabetes. Zeitschrift fur Gefassmedizin 2013.
- Qin, Y.; Aqeel, M.; Zhu, F.; Delp, E.J.; Eicher-Miller, H.A. Dietary Aspects to Incorporate in the Creation of a Mobile Image-Based Dietary Assessment Tool to Manage and Improve Diabetes. Nutrients 2021. [Google Scholar] [CrossRef]
- Sassi, F.; Cecchini, M.; Lauer, J.; Chisholm, D. IMPROVING LIFESTYLES, TACKLING OBESITY: THE HEALTH AND ECONOMIC IMPACT OF PREVENTION STRATEGIES. In OECD Heal. Work. Pap.; 2009. [Google Scholar]
- Malpass, A.; Andrews, R.; Turner, K.M. Patients with Type 2 Diabetes Experiences of Making Multiple Lifestyle Changes: A Qualitative Study. Patient Educ. Couns. 2009. [Google Scholar] [CrossRef]
- Goudy, K.S.; Tisch, R. Immunotherapy for the Prevention and Treatment of Type 1 Diabetes. Int. Rev. Immunol. 2005. [Google Scholar] [CrossRef]
- Candido, R.; Wyne, K.; Romoli, E. A Review of Basal-Bolus Therapy Using Insulin Glargine and Insulin Lispro in the Management of Diabetes Mellitus. Diabetes Ther. 2018. [Google Scholar] [CrossRef]
- Cengiz, E.; Sherr, J.L.; Weinzimer, S.A.; Tamborlane, W. V. New-Generation Diabetes Management: Glucose Sensor-Augmented Insulin Pump Therapy. Expert Rev. Med. Devices 2011. [Google Scholar] [CrossRef]
- Passerini, L.; Forlani, A.; Gregori, S. Advances in Regulatory Cell Therapy for Type 1 Diabetes: Emerging Strategies and Future Directions. Eur. J. Immunol. 2025, 55, 1–9. [Google Scholar] [CrossRef]
- Sherr, J.L.; Heinemann, L.; Fleming, G.A.; Bergenstal, R.M.; Bruttomesso, D.; Hanaire, H.; Holl, R.W.; Petrie, J.R.; Peters, A.L.; Evans, M. Automated Insulin Delivery: Benefits, Challenges, and Recommendations. A Consensus Report of the Joint Diabetes Technology Working Group of the European Association for the Study of Diabetes and the American Diabetes Association. Diabetologia 2023. [Google Scholar] [CrossRef]
- Padhi, S.; Nayak, A.K.; Behera, A. Type II Diabetes Mellitus: A Review on Recent Drug Based Therapeutics. In Biomed. Pharmacother.; 2020. [Google Scholar]
- Feingold, K.R. Oral and Injectable (Non-Insulin) Pharmacological Agents for Type 2 Diabetes; 2000. [Google Scholar]
- Padda, I.S.; Mahtani, A.U.; Parmar, M. Sodium-Glucose Transport Protein 2 (SGLT2) Inhibitors; 2022. [Google Scholar]
- Kalra, S.; Kesavadev, J.; Chadha, M.; Vijaya Kumar, G. Sodium-Glucose Cotransporter-2 Inhibitors in Combination with Other Glucose-Lowering Agents for the Treatment of Type 2 Diabetes Mellitus. Indian J. Endocrinol. Metab. 2018. [Google Scholar] [CrossRef] [PubMed]
- Blonde, L.; Dipp, S.; Cadena, D. Combination Glucose-Lowering Therapy Plans in T2DM: Case-Based Considerations. Adv. Ther. 2018. [Google Scholar]
- Bae, J.H. SGLT2 Inhibitors and GLP-1 Receptor Agonists in Diabetic Kidney Disease: Evolving Evidence and Clinical Application. Diabetes Metab. J. 2025, 49, 386–402. [Google Scholar] [CrossRef] [PubMed]
- Eckstein, M.L.; Williams, D.M.; O’Neil, L.K.; Hayes, J.; Stephens, J.W.; Bracken, R.M. Physical Exercise and Non-Insulin Glucose-Lowering Therapies in the Management of Type 2 Diabetes Mellitus: A Clinical Review. Diabet. Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Groop, L.; Widén, E.; Franssila-Kallunki, A.; Ekstrand, A.; Saloranta, C.; Schalin, C.; Eriksson, J. Different Effects of Insulin and Oral Antidiabetic Agents on Glucose and Energy Metabolism in Type 2 (Non-Insulin-Dependent) Diabetes Mellitus. Diabetologia 1989. [Google Scholar] [CrossRef]
- Easa, N.; Alany, R.G.; Carew, M.; Vangala, A. A Review of Non-Invasive Insulin Delivery Systems for Diabetes Therapy in Clinical Trials over the Past Decade. Drug Discov. Today 2019. [Google Scholar] [CrossRef]
- Zahid, M.; Dowlatshahi, S.; Kansara, A.H.; Sadhu, A.R. The Evolution of Diabetes Technology – Options Toward Personalized Care. Endocr. Pract. 2023. [Google Scholar] [CrossRef]
- Marks, B.E.; Williams, K.M.; Sherwood, J.S.; Putman, M.S. Practical Aspects of Diabetes Technology Use: Continuous Glucose Monitors, Insulin Pumps, and Automated Insulin Delivery Systems. J. Clin. Transl. Endocrinol. 2022. [Google Scholar] [CrossRef]
- Forlenza, G.P.; Buckingham, B.; Maahs, D.M. Progress in Diabetes Technology: Developments in Insulin Pumps, Continuous Glucose Monitors, and Progress towards the Artificial Pancreas. J. Pediatr. 2016. [Google Scholar] [CrossRef]
- Aleppo, G.; Webb, K.M. Integrated Insulin Pump and Continuous Glucose Monitoring Technology in Diabetes Care Today: A Perspective of Real-Life Experience with the MinimedTM 670G Hybrid Closed-Loop System. Endocr. Pract. 2018. [Google Scholar] [CrossRef] [PubMed]
- Pauley, M.E.; Tommerdahl, K.L.; Snell-Bergeon, J.K.; Forlenza, G.P. Continuous Glucose Monitor, Insulin Pump, and Automated Insulin Delivery Therapies for Type 1 Diabetes: An Update on Potential for Cardiovascular Benefits. Curr. Cardiol. Rep. 2022. [Google Scholar] [CrossRef]
- Klonoff, D.C.; Ahn, D.; Drincic, A. Continuous Glucose Monitoring: A Review of the Technology and Clinical Use. Diabetes Res. Clin. Pract. 2017. [Google Scholar] [CrossRef] [PubMed]
- Almurashi, A.M.; Rodriguez, E.; Garg, S.K. Emerging Diabetes Technologies: Continuous Glucose Monitors/Artificial Pancreases. J. Indian Inst. Sci. 2023. [Google Scholar] [CrossRef]
- Anandhakrishnan, A.; Hussain, S. Automating Insulin Delivery through Pump and Continuous Glucose Monitoring Connectivity: Maximizing Opportunities to Improve Outcomes. Diabetes, Obes. Metab. 2024, 26, 27–46. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, J.; Thomas, I.H.; Soleimanpour, S.A. Transition from Pediatric to Adult Care in Emerging Adults with Type 1 Diabetes: A Blueprint for Effective Receivership. Clin. Diabetes Endocrinol. 2019. [Google Scholar] [CrossRef]
- Peters, A.; Laffel, L. Diabetes Care for Emerging Adults: Recommendations for Transition from Pediatric to Adult Diabetes Care Systems. Diabetes Care 2012. [Google Scholar]
- Moses, J.; Kim, S.C. Transition from Pediatric to Adult Care. In Pediatric Inflammatory Bowel Disease; 2023; ISBN 9783031147449. [Google Scholar]
- Rajchert, J.; Rosa, M.; Pawłowska, J.; Parczewski, M.; Wawrzynowicz-Syczewska, M. Follow-up of Pediatric Liver Transplant Patients after Reaching Adulthood. Ann. Transplant. 2016. [Google Scholar] [CrossRef]
- O’Higgins, A.; Sebba, J.; Luke, N. What Is the Relationship between Being in Care and the Educational Outcomes of Children? Child Fam. Soc. Work 2015. [Google Scholar]
- White, P.H.; Cooley, W.C. Supporting the Health Care Transition from Adolescence to Adulthood in the Medical Home. Pediatrics 2018. [Google Scholar] [CrossRef]
- Mangoulia, P.; Milionis, C.; Vlachou, E.; Ilias, I. The Interrelationship between Diabetes Mellitus and Emotional Well-Being: Current Concepts and Future Prospects. Healthc. 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Kalra, S.; Jena, B.N.; Yeravdekar, R. Emotional and Psychological Needs of People with Diabetes. Indian J. Endocrinol. Metab. 2018. [Google Scholar]
- Chew, B.-H. Psychological Aspects of Diabetes Care: Effecting Behavioral Change in Patients. World J. Diabetes 2014. [Google Scholar] [CrossRef]
- Davies, M. Psychological Aspects of Diabetes Management. In Med. (United Kingdom); 2022. [Google Scholar]
- Tareen, R.S.; Tareen, K. Psychosocial Aspects of Diabetes Management: Dilemma of Diabetes Distress; Pediatr., 2017. [Google Scholar]
- Adili, F.; Larijani, B.; Haghighatpanah, M. Diabetic Patients: Psychological Aspects. In Proceedings of the Annals of the New York Academy of Sciences, 2006. [Google Scholar]
- Winterdijk, P.; Aanstoot, H.J.; Nefs, G. The Impact of Real-Time Sensor Technology on Quality of Life for Adults with Type 1 Diabetes: A Dutch National Survey. Diabetes Res. Clin. Pract. 2023. [Google Scholar] [CrossRef]
- de Wit, M.; Gajewska, K.A.; Goethals, E.R.; McDarby, V.; Zhao, X.; Hapunda, G.; Delamater, A.M.; DiMeglio, L.A. ISPAD Clinical Practice Consensus Guidelines 2022: Psychological Care of Children, Adolescents and Young Adults with Diabetes. Pediatr. Diabetes 2022. [Google Scholar] [CrossRef]
- Young-Hyman, D.; De Groot, M.; Hill-Briggs, F.; Gonzalez, J.S.; Hood, K.; Peyrot, M. Psychosocial Care for People with Diabetes: A Position Statement of the American Diabetes Association. Diabetes Care 2016. [Google Scholar] [CrossRef]
- Ansari, M.A.; Chauhan, W.; Shoaib, S.; Alyahya, S.A.; Ali, M.; Ashraf, H.; Alomary, M.N.; Al-Suhaimi, E.A. Emerging Therapeutic Options in the Management of Diabetes: Recent Trends, Challenges and Future Directions. Int. J. Obes. 2023. [Google Scholar] [CrossRef] [PubMed]
- Wagman, A.S.; Nuss, J.M. Current Therapies and Emerging Targets for the Treatment of Diabetes. Front. Med. Chem. - Online 2005. [Google Scholar] [CrossRef]
- Kąpa, M.; Koryciarz, I.; Kustosik, N.; Jurowski, P.; Pniakowska, Z. Future Directions in Diabetic Retinopathy Treatment: Stem Cell Therapy, Nanotechnology, and PPARα Modulation. J. Clin. Med. 2025, 14. [Google Scholar] [CrossRef]
- Sharma, R.; Borah, S.J.; Bhawna, N.; Kumar, S.; Gupta, A.; Kumari, V.; Kumar, R.; Dubey, K.K.; Kumar, V. Emerging Trends in Nano-Based Antidiabetic Therapeutics: A Path to Effective Diabetes Management. Mater. Adv. 2023. [Google Scholar] [CrossRef]
- Kopan, C.; Tucker, T.; Alexander, M.; Mohammadi, M.R.; Pone, E.; Lakey, J.R.T. Approaches in Immunotherapy, Regenerative Medicine, and Bioengineering for Type 1 Diabetes. Front. Immunol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Angiolillo, D. Antiplatelet Therapy in Diabetes: Efficacy and Limitations of Current Treatment Strategies and Future Directions. In Diabetes Care; 2009. [Google Scholar]
- Pimenta, C.; Bettiol, V.; Alencar-Silva, T.; Franco, O.L.; Pogue, R.; Carvalho, J.L.; Felipe, M.S.S. Advanced Therapies and Regulatory Framework in Different Areas of the Globe: Past, Present, and Future. Clin. Ther. 2021. [Google Scholar] [CrossRef] [PubMed]
- Guan, Z.; Li, H.; Liu, R.; Cai, C.; Liu, Y.; Li, J.; Wang, X.; Huang, S.; Wu, L.; Liu, D.; et al. Artificial Intelligence in Diabetes Management: Advancements, Opportunities, and Challenges. Cell Reports Med. 2023. [Google Scholar] [CrossRef]
| Component | Pediatric (Children) Population | Adult Population | Recent Advances |
| Diagnostic Approaches |
- HbA1c, Fasting Blood Glucose, OGTT- Autoantibody Testing (T1DM)- Genetic Testing (MODY) | - HbA1c, OGTT, Fasting Glucose- Risk Scoring Tools (e.g., ADA, FINDRISC)- Retinal and Renal Screening | - Continuous Glucose Monitoring (CGM)- AI-based Predictive Tools- Non-invasive Glucose Sensors |
| Insulin Therapy | - Multiple Daily Injections (MDI)- Insulin Pumps- Hybrid Closed-Loop Systems | - MDI or Premixed Insulin- Long-acting and Ultra-long-acting Analogues | - Smart Insulin Delivery Systems- Artificial Pancreas Systems- Personalized Insulin Algorithms |
| Pharmacologic Interventions | - Metformin (in pediatric T2DM)- GLP-1 Receptor Agonists (limited use) | - Metformin, SGLT2 Inhibitors, DPP-4 Inhibitors, GLP-1 RAs, Thiazolidinediones | - Dual GIP/GLP-1 Agonists (e.g., Tirzepatide)- Expanded use of SGLT2 inhibitors in T1DM (investigational) |
| Dietary Management | - Carbohydrate Counting- Low-Glycemic Index Diets- Parental Education Programs | - Mediterranean, DASH, or Low-Carb Diets- Structured Meal Planning | - Personalized Nutrition via CGM Data- Gut Microbiota-Based Dietary Modulation |
| Lifestyle Modifications | - Age-appropriate Exercise Programs- School and Family Engagement | - Structured Physical Activity- Smoking and Alcohol Cessation | - Mobile Health (mHealth) Tools- Digital Coaching Platforms- Behavioral Modification Apps |
| Monitoring and Follow-Up | - Pediatric Endocrinologist Follow-up- Monitoring of Growth and Puberty | - Routine Monitoring for Complications (CVD, Nephropathy, Retinopathy) | - Remote Patient Monitoring- Integrated EHR Systems with Predictive Analytics |
| Complication Prevention | - Early Screening for Microvascular Complications- Vaccination Compliance | - Cardiometabolic Risk Control- Nephropathy and Neuropathy Screening | - Use of Novel Biomarkers- Early Intervention with Renoprotective and Cardioprotective Drugs |
| Intervention | Target Group | Advantages | Disadvantages |
| Metformin | Adults | - First-line for Type 2 diabetes- Weight neutral or slight loss- Cardiovascular benefits | - GI side effects- Lactic acidosis (rare)- Contraindicated in renal impairment |
| Children (≥10 yrs) | - Approved for pediatric Type 2 diabetes- Delays insulin initiation | - GI intolerance- Limited monotherapy efficacy in some children | |
| Sulfonylureas | Adults | - Cost-effective- Rapid glucose reduction | - High risk of hypoglycemia- Weight gain |
| Children | - Used in neonatal and MODY forms of diabetes | - Not typically first-line- Risk of hypoglycemia | |
| DPP-4 Inhibitors | Adults | - Well-tolerated- No weight gain- Low hypoglycemia risk | - Moderate efficacy- Expensive |
| Children | - Limited pediatric data | - Off-label use in most settings- Cost-prohibitive in some regions | |
| SGLT2 Inhibitors | Adults | - Promotes weight loss- Cardiovascular and renal benefits | - Genital infections- Risk of ketoacidosis |
| Children (>10 yrs) | - Emerging evidence in adolescents- FDA approval for dapagliflozin (2023) | - Long-term safety unknown- Risk of dehydration and DKA | |
| GLP-1 Receptor Agonists |
Adults | - Weight loss- Improved cardiovascular outcomes- Delays insulin requirement | - Injectable- GI side effects- Expensive |
| Children (≥10 yrs) | - FDA-approved for pediatric Type 2 diabetes (liraglutide, 2020) | - GI intolerance- Fear of injection- High cost | |
| Basal-Bolus Insulin | Adults | - Flexible dosing- Better postprandial control | - Frequent injections- Hypoglycemia risk- Weight gain |
| Children | - Essential for Type 1 diabetes- Mimics physiologic insulin profile | - Requires strong parental involvement- Psychosocial burden | |
| Insulin Pumps (CSII) | Adults | - Improved glycemic control- Reduced variability- Quality of life | - High cost- Technical learning curve- Risk of DKA if malfunction |
| Children | - Enhanced quality of life- Better HbA1c control in some studies | - Device burden- Requires family support and monitoring | |
| Closed-Loop Systems | Adults | - Automated insulin delivery- Reduces hypo- and hyperglycemia | - Very expensive- Requires CGM calibration |
| Children | - Safe in children ≥6 yrs (e.g., MiniMed 780G)- Reduces parental stress | - High initial cost- Sensor wear and data interpretation challenges | |
| Low-Glycemic Index Diet | Adults | - Improves postprandial glucose- Sustainable eating habits | - Requires dietary planning- May conflict with cultural preferences |
| Children | - Prevents glucose spikes- Can be incorporated into school lunches | - Adherence challenges- Peer influence | |
| Carbohydrate Counting | Adults | - Supports flexible insulin dosing- Better control with bolus insulin | - Requires nutritional literacy- Time-consuming |
| Children | - Educational tool for families- Encourages healthy food choices | - Requires constant parental involvement- Difficult in social settings (e.g., school, parties) | |
| Mediterranean Diet | Adults | - Cardioprotective- Rich in fiber and healthy fats | - Can be expensive- May require lifestyle shift |
| Children | - Family-friendly and balanced | - Adherence in picky eaters or selective diets can be difficult |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




