1. Introduction
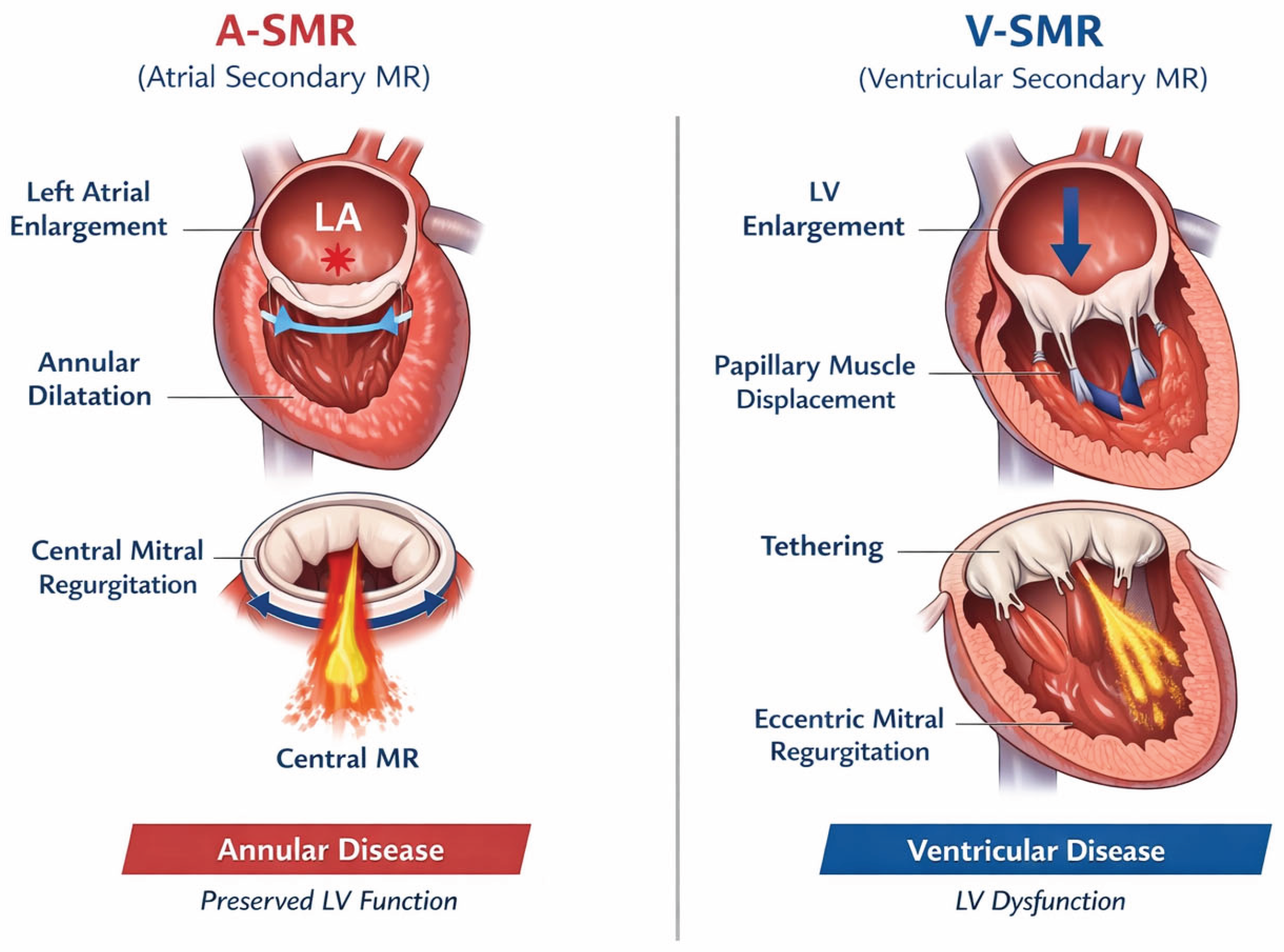
Functional mitral regurgitation (MR) has traditionally been regarded as a consequence of left ventricular remodeling and systolic dysfunction. In this classical framework, mitral valve incompetence develops secondary to ventricular dilatation, papillary muscle displacement, and leaflet tethering, while the valve leaflets themselves remain structurally normal [9]. This ventricular-centered paradigm has long guided both diagnostic classification and therapeutic strategies in patients with secondary MR.
In recent years, however, growing clinical and echocardiographic evidence has challenged this concept by identifying a distinct form of functional MR occurring in the presence of preserved left ventricular size and systolic function. In this setting, mitral regurgitation is primarily driven by left atrial enlargement and mitral annular dilatation rather than by ventricular distortion [10]. This entity, commonly referred to as atrial secondary mitral regurgitation (A-SMR) or atrial functional mitral regurgitation, represents a paradigm shift in the understanding of functional mitral valve disease and differs from the ventricular functional mitral regurgitation (V-SMR).
A-SMR is most frequently observed in patients with long-standing atrial fibrillation and in those with heart failure with preserved ejection fraction. These conditions are characterized by chronically elevated left atrial pressure and volume, which promote progressive atrial remodeling and annular enlargement over time [11]. As atrial size increases, leaflet coaptation becomes insufficient despite the absence of primary leaflet pathology, leading to clinically significant mitral regurgitation.
From an epidemiological standpoint, large observational studies and population-based datasets have shown that A-SMR is not an uncommon condition, particularly in elderly patients and in those with atrial fibrillation [12]. In a large unselected cohort, Chen et al. showed that although atrial functional MR accounted for approximately 39% of cases and purely ventricular functional MR for about 14%, nearly 60% of patients exhibited a significant ventricular contribution to mitral regurgitation when combined atrial–ventricular (dual) phenotypes were considered [3]. The dual functional mitral regurgitation phenotype refers to a mixed form of functional MR in which atrial-driven mechanisms (left atrial enlargement and annular dilatation) coexist with ventricular-driven mechanisms (left ventricular remodeling and leaflet tethering) [3]. This phenotype likely represents an advanced or progressive stage of disease, in which initial atrial dysfunction is accompanied over time by ventricular structural changes [3]. Importantly, these studies have also demonstrated that A-SMR is not a benign finding, as moderate-to-severe atrial-driven mitral regurgitation is associated with worsening symptoms and adverse clinical outcomes [12].
A-SMR differs from ventricular functional MR not only in terms of pathophysiology but also with respect to patterns of cardiac remodeling and disease progression. Comparative analyses have shown that atrial and ventricular functional MR are associated with distinct structural and clinical phenotypes, with atrial-driven disease exhibiting a stronger relationship with left atrial size, rhythm status, and annular geometry [13]. These differences suggest that therapeutic strategies effective in V-SMR may not be directly applicable to patients with A-SMR.
Despite its growing recognition, A-SMR remains insufficiently addressed in current clinical guidelines, without providing specific recommendations tailored to atrial-driven disease [5,14]. This lack of dedicated guidance contributes to uncertainty in clinical practice, particularly when choosing among medical therapy, rhythm control strategies, transcatheter interventions, and surgical repair.
Given these considerations, a comprehensive and mechanism-oriented evaluation of A-SMR is essential. Improved understanding of its pathophysiology, imaging characteristics, natural history, and response to different therapeutic approaches may help refine patient selection and optimize outcomes. In this context, cardiac surgery plays a pivotal role, as surgical annuloplasty directly targets the dominant anatomical substrate of A-SMR and offers the opportunity for concomitant treatment of atrial fibrillation [15].
2. Methodology and Study Selection
This narrative review summarizes current evidence on atrial secondary (atrial functional) mitral regurgitation with a focus on diagnostic definitions, imaging assessment, clinical outcomes, and implications for cardiac surgery. A targeted literature search was performed in PubMed/MEDLINE and Scopus from inception to December 2025 using combinations of the terms “atrial functional mitral regurgitation”, “atrial secondary mitral regurgitation”, “mitral annulus”, “atrial fibrillation”, “annuloplasty”, “mitral valve repair”, and “transcatheter edge-to-edge repair”. Additional records were identified by manual screening of reference lists of relevant articles. We prioritized English-language studies in adult populations, including observational cohorts, registry studies, interventional reports, meta-analyses, and contemporary reviews. Case reports were considered only when providing unique mechanistic or surgical insights. Given the narrative design, no formal risk-of-bias assessment was performed; however, evidence was critically appraised with attention to phenotype definition, imaging criteria, and outcome ascertainment.
3. Pathophysiology of Atrial Secondary Mitral Regurgitation
Atrial secondary mitral regurgitation is fundamentally an atrial-driven disease. In contrast to ventricular functional mitral regurgitation, in which mitral valve incompetence arises from left ventricular remodeling and papillary muscle displacement, A-SMR develops in the setting of preserved ventricular geometry and systolic function, with regurgitation primarily resulting from atrial and annular remodeling [16] (
Figure 1).
3.1. Left Atrial Remodeling and Annular Dilatation
Chronic elevation of left atrial pressure and volume represents the central trigger for structural remodeling in A-SMR. Long-standing atrial fibrillation (AF) and diastolic dysfunction lead to progressive atrial enlargement, wall stretch, and fibrosis, which in turn directly affect mitral annular size and geometry [17]. Because the mitral annulus is anatomically coupled to the left atrium, atrial dilatation translates into annular enlargement even in the absence of ventricular dilatation.
Annular dilatation in A-SMR predominantly involves the posterior and lateral segments of the annulus, resulting in a more circular and flattened configuration. This geometric alteration reduces the coaptation surface between the mitral leaflets despite preserved leaflet structure and mobility, thereby generating functional regurgitation [18].
3.2. Leaflet–Annulus Mismatch
Mitral leaflets may undergo adaptive enlargement in response to chronic loading conditions; however, in many patients with A-SMR this compensatory mechanism is insufficient to match the degree of annular dilatation. The resulting leaflet–annulus mismatch represents a key mechanism underlying atrial-driven mitral regurgitation [19]. Unlike V-SMR, leaflet tethering and subvalvular distortion are typically minimal, and coaptation failure occurs predominantly at the annular level.
This mechanistic distinction has important therapeutic implications, as interventions aimed at restoring annular size and geometry directly address the primary cause of regurgitation in A-SMR.
4. Imaging Assessment and Diagnostic Criteria
Imaging plays a central role in the diagnosis and management of atrial secondary mitral regurgitation. Because A-SMR is defined by preserved left ventricular geometry and the absence of primary mitral valve disease, a comprehensive imaging approach is essential to correctly identify the atrial-driven mechanism of regurgitation and to distinguish it from ventricular functional mitral regurgitation [20].
4.1. Standard Echocardiographic Evaluation
Transthoracic echocardiography (TTE) represents the first-line imaging modality for patients with suspected A-SMR. A systematic evaluation should document preserved left ventricular size and systolic function, exclude primary leaflet abnormalities, and assess the severity of mitral regurgitation. Marked left atrial enlargement is a hallmark finding and should prompt careful evaluation of annular dimensions and valve coaptation [21].
Mitral regurgitation jets in A-SMR are typically central or mildly posteriorly directed, reflecting symmetric annular dilatation rather than asymmetric leaflet tethering. Quantitative assessment of regurgitation severity should be interpreted with caution, as loading conditions and rhythm status may significantly influence Doppler measurements.
Transesophageal echocardiography (TEE) may be useful to exclude subtle leaflet pathology and to better define annular anatomy, particularly in patients being considered for interventional or surgical treatment.
4.2. Mechanistic and Geometric Parameters
Several echocardiographic parameters are particularly informative for recognizing atrial-driven mitral regurgitation. Measurements of mitral annular dimensions, including anteroposterior and commissural diameters, allow quantification of annular dilatation [20]. In contrast to V-SMR, indices of leaflet tethering such as tenting height and tenting area are typically minimal in A-SMR [22].
Assessment of leaflet length and coaptation depth provides additional insight into leaflet–annulus mismatch. In many patients with A-SMR, leaflet tissue is relatively preserved, whereas coaptation failure results from annular enlargement rather than from subvalvular distortion [20].
4.3. Advanced Imaging and Atrial Function
Three-dimensional echocardiography has improved the assessment of mitral annular geometry and leaflet coaptation in A-SMR. By enabling accurate reconstruction of annular shape and dynamic changes throughout the cardiac cycle, three-dimensional imaging facilitates refined phenotypic characterization and improves patient selection for intervention [23].
In addition, evaluation of left atrial function using strain imaging provides complementary information on the severity of atrial myopathy. Reduced atrial strain has been associated with advanced atrial remodeling and may help identify patients at increased risk of persistent or recurrent mitral regurgitation after treatment [20].
For several years, the use of cardiac magnetic resonance has become increasingly widespread in the field of valvular heart disease, particularly in mitral regurgitation. It allows an in-depth assessment of the mitral valve apparatus, leaflet morphology, and papillary muscles, and can also precisely characterize the impact of mitral regurgitation on left atrial and ventricular remodeling [24].
The DECAAF study demonstrated that the extent of left atrial fibrosis, quantified by delayed-enhancement MRI, is a strong and independent marker of atrial disease severity and clinical progression in patients with atrial fibrillation. Increasing degrees of atrial fibrosis were associated with worse clinical outcomes and reduced response to rhythm-control strategies, reflecting advanced atrial remodeling and loss of atrial compliance [25].
Cawley et al. performed a prospective head-to-head comparison between transthoracic echocardiography (TTE) and cardiac magnetic resonance (CMR) for quantification of chronic mitral and aortic regurgitation in 57 patients studied on the same day. The study demonstrated that CMR provides significantly lower intra- and interobserver variability than TTE for regurgitant volume and ventricular volume measurements, with particularly strong reproducibility for phase-contrast flow assessment of regurgitation. Although both modalities showed substantial variability in mitral regurgitation quantification, CMR tended to be more consistent than Doppler-based echocardiographic methods, especially when annular geometry and ventricular volumes were difficult to define. These findings highlight the limitations of echocardiography in complex or geometrically distorted valves and support the role of CMR as a complementary tool for accurate and reproducible quantification of regurgitation severity, particularly when precise serial assessment is required for clinical decision-making or timing of intervention [26].
5. Natural History and Clinical Impact
Atrial secondary mitral regurgitation is increasingly recognized as a progressive condition rather than a benign echocardiographic finding. Although mild forms of A-SMR may initially be well tolerated, longitudinal observations indicate that mitral regurgitation severity often increases over time as a consequence of ongoing atrial remodeling and progressive annular dilatation [27].
Progression of A-SMR is closely linked to the natural history of atrial fibrillation and atrial cardiomyopathy. Persistent elevation of left atrial pressure and volume overload promote further atrial enlargement, establishing a vicious cycle in which worsening mitral regurgitation contributes to additional atrial stretch and remodeling. This self-perpetuating mechanism may ultimately lead to stabilization of significant regurgitation that becomes less responsive to rhythm or medical therapies [28].
From a clinical perspective, increasing severity of A-SMR is associated with worsening symptoms, including exertional dyspnea, reduced exercise tolerance, and recurrent heart failure hospitalizations [28]. Importantly, observational studies indicate that moderate-to-severe atrial secondary mitral regurgitation is associated with worse clinical outcomes, independently of comorbid conditions and baseline cardiac function. [20].
Beyond symptom burden, A-SMR has important hemodynamic consequences. Chronic left atrial volume overload may lead to sustained elevation of pulmonary venous pressure and the development of pulmonary hypertension [2]. In advanced stages, pulmonary hypertension may result in right ventricular dysfunction and secondary tricuspid regurgitation, further complicating the clinical course and adversely affecting prognosis [23].
Timing appears to be a critical determinant of outcomes in patients with A-SMR. Earlier stages of disease, characterized by moderate annular dilatation and less advanced atrial remodeling, may retain partial reversibility, particularly when sinus rhythm can be restored [6]. In contrast, delayed intervention in the presence of severe atrial enlargement, persistent atrial fibrillation, and established pulmonary hypertension is associated with less favorable outcomes and a higher likelihood of residual or recurrent mitral regurgitation after treatment [2].
6. Non-Surgical Treatment Strategies
Non-surgical management of atrial secondary mitral regurgitation is primarily directed toward symptom control and treatment of associated conditions rather than correction of the underlying anatomical substrate. Because A-SMR is driven by atrial remodeling and annular dilatation, medical and rhythm-based therapies may influence disease severity in selected patients but generally do not provide definitive resolution of mitral regurgitation [5].
6.1. Medical Therapy
Medical therapy in patients with atrial secondary mitral regurgitation (A-SMR) should be considered within the framework of contemporary heart failure with preserved ejection fraction (HFpEF) management, a condition that frequently coexists with atrial-driven mitral regurgitation. Diuretic therapy remains essential for congestion relief and symptom control by reducing left atrial pressure; however, it does not directly address mitral annular dilatation or restore leaflet coaptation and therefore has limited impact on the structural progression of A-SMR [1]. Sodium–glucose cotransporter-2 (SGLT2) inhibitors have emerged as a foundational therapy in HFpEF, with large randomized trials demonstrating significant reductions in heart failure hospitalizations and improvements in patient-reported outcomes across the spectrum of preserved and mildly reduced ejection fraction [29,30]. In patients with A-SMR occurring in the HFpEF setting, SGLT2 inhibitors should therefore be regarded as part of optimized background therapy. While they do not directly modify annular geometry, their favorable effects on cardiac filling pressures and congestion may indirectly mitigate symptom burden and dynamic variability of mitral regurgitation severity [29,30]. Overall, pharmacological therapy in A-SMR should be viewed as supportive and prognostically beneficial, particularly for HFpEF and comorbidity management, but insufficient as a standalone strategy for correcting atrial-driven mitral regurgitation. Persistent moderate-to-severe A-SMR despite optimized medical therapy should prompt consideration of interventional or surgical treatment [1].
6.2. Rhythm and Rate Control Strategies
Given the central role of atrial fibrillation in the pathophysiology of A-SMR, rhythm control strategies are of particular interest. Restoration and maintenance of sinus rhythm may promote atrial reverse remodeling, improve atrial contractile function, and partially restore annular dynamics. In selected patients, particularly those with less advanced atrial remodeling and shorter duration of atrial fibrillation, rhythm control has been associated with a reduction in mitral regurgitation severity [6].
Catheter ablation represents an important rhythm control option in appropriately selected patients and may contribute to improvement in mitral regurgitation by reducing atrial size and pressure over time [31]. However, success rates decrease markedly in the presence of long-standing persistent atrial fibrillation and severe atrial enlargement, limiting the applicability of this approach in many patients with established A-SMR [31]. Rate control strategies, while important for symptom management, have a limited effect on atrial remodeling and are unlikely to substantially influence the course of atrial-driven mitral regurgitation [1,32].
6.3. Transcatheter Edge-To-Edge Repair
Transcatheter edge-to-edge repair has emerged as a therapeutic option for patients with severe A-SMR who remain symptomatic despite optimized medical and rhythm management and who are considered not eligible for surgery (Class IIb Level B Recommendation) [5]. By enhancing leaflet coaptation, this approach can reduce mitral regurgitation severity and improve symptoms in selected patients [7,33,34,35,36].
However, the predominantly annular nature of A-SMR raises concerns regarding long-term durability. Progressive annular dilatation and ongoing atrial remodeling may continue despite successful edge-to-edge repair, predisposing to residual or recurrent mitral regurgitation over time [4,37]. Advanced imaging studies have shown that patients with extreme annular enlargement and severe atrial myopathy derive less durable benefit from transcatheter repair [37].
Careful patient selection is therefore essential when considering transcatheter interventions in A-SMR. Patients with moderate annular dilatation, preserved leaflet tissue, and less advanced atrial remodeling appear more likely to benefit, whereas those with advanced atrial disease and severe tricuspid regurgitation may experience limited or transient improvement [4].
7. Surgical Implications in Atrial Secondary Mitral Regurgitation
Surgical treatment represents a cornerstone in the management of A-SMR, as it directly addresses the dominant anatomical mechanism underlying the disease. In contrast to V-SMR, where surgical outcomes are strongly influenced by left ventricular geometry and function, A-SMR is primarily an annular disorder occurring in the setting of preserved ventricular performance. This fundamental distinction has important implications for patient selection, timing of intervention, and choice of surgical technique [8,15].
7.1. Indications and Timing for Surgery
Surgical intervention should be considered in patients with severe A-SMR who remain symptomatic despite optimized medical therapy and appropriate rhythm or rate control strategies, in accordance with current guideline recommendations (Class IIa Level B Recommendation) [5]. Timing of intervention is particularly relevant, as surgery performed before the development of extreme atrial dilatation, advanced pulmonary hypertension, or right ventricular dysfunction is more likely to result in durable mitral valve repair and favorable clinical outcomes [38].
Delayed referral may attenuate the benefit of surgery, as advanced atrial remodeling and long-standing AF have been consistently associated with higher rates of recurrent mitral regurgitation following intervention [27]. These observations support consideration of earlier surgical referral in selected patients with progressive symptoms and echocardiographic evidence of worsening annular dilatation, before irreversible atrial and right-sided remodeling occurs [27].
7.2. Surgical Techniques
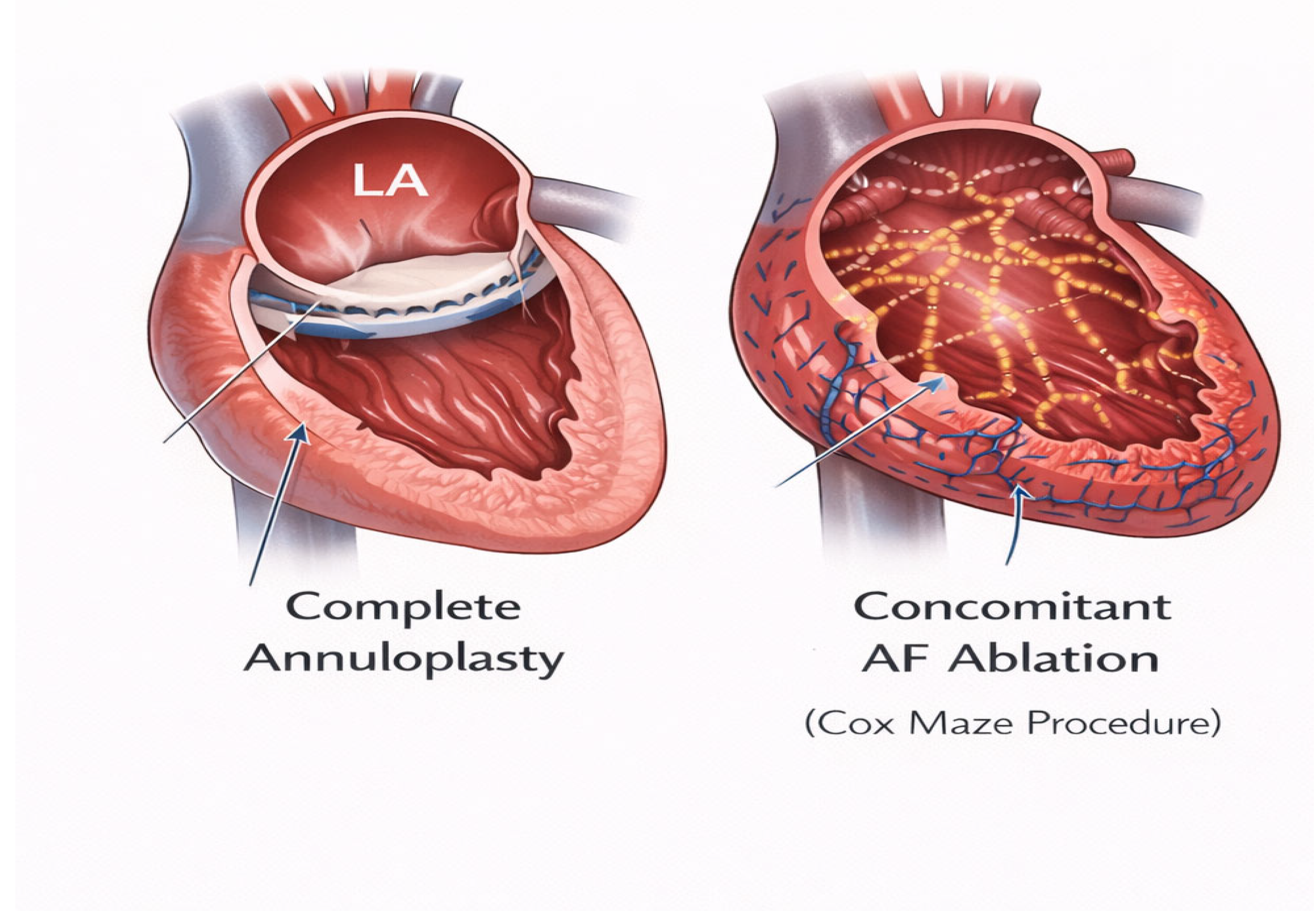
Mitral valve repair based on annuloplasty remains the preferred surgical strategy for A-SMR, as it directly targets the primary mechanism of annular dilatation [39] in the presence of structurally normal leaflets and an intact subvalvular apparatus (
Figure 2). Restoration of annular size and geometry improves leaflet coaptation and mitral competence without the need for ventricular or papillary muscle–directed interventions [8].
Early clinical evidence supporting this approach was provided by Vohra et al., who reported favorable mid-term outcomes following systematic mitral annuloplasty in patients with severe A-SMR due to isolated annular dilatation, frequently combined with concomitant atrial fibrillation surgery and tricuspid repair [40]. Subsequent studies have consistently confirmed annuloplasty as the foundation of surgical repair in this setting.
A comprehensive scoping review of the surgical literature further demonstrated that, despite substantial heterogeneity in diagnostic criteria and patient selection, mitral valve repair with ring annuloplasty represents the predominant surgical strategy across published A-SMR series. However, the use of adjunctive leaflet, atrial, and rhythm-control procedures remains highly variable, reflecting the absence of standardized treatment algorithms [41].
Disease progression plays a key role in determining the adequacy of isolated annuloplasty. As highlighted by Aranda-Domene and colleagues, A-SMR may evolve from an early stage dominated by annular dilatation and central regurgitation to more advanced stages characterized by atriogenic posterior leaflet tethering, anterior leaflet pseudoprolapse, and eccentric regurgitant jets. In such cases, annuloplasty alone may be insufficient, prompting consideration of more comprehensive surgical strategies [42].
Several authors have therefore advocated adjunctive techniques tailored to advanced atrial remodeling, including leaflet augmentation, atrial plication, and concomitant rhythm-control procedures [42]. Posterior leaflet augmentation with autologous pericardium has been explored as a means to improve coaptation; however, concerns remain regarding long-term durability due to leaflet shrinkage and stiffening [43].
The concept of A-SMR as a disease driven by atrial remodeling rather than ventricular distortion has been further reinforced by multiple surgical series demonstrating durable results with annuloplasty-based repair in patients with preserved ventricular geometry. However, additional procedures—including artificial chordal implantation, posterior leaflet augmentation with autologous pericardium, atrial plication, and routine tricuspid annuloplasty—were advocated for patients with giant left atria and markedly enlarged annuli [44]. Takahashi et al. reported sustained reduction of mitral regurgitation and symptomatic improvement following annuloplasty in patients with chronic atrial fibrillation, even in the absence of rhythm restoration, underscoring the primacy of annular pathology [45].
Nevertheless, outcomes appear less favorable in patients with excessive atriogenic or combined atriogenic–ventriculogenic leaflet tethering. Sakaguchi et al. demonstrated that recurrent mitral regurgitation following annuloplasty was associated with larger preoperative ventricular dimensions and more pronounced leaflet tethering, highlighting the importance of careful preoperative morphological assessment [46].
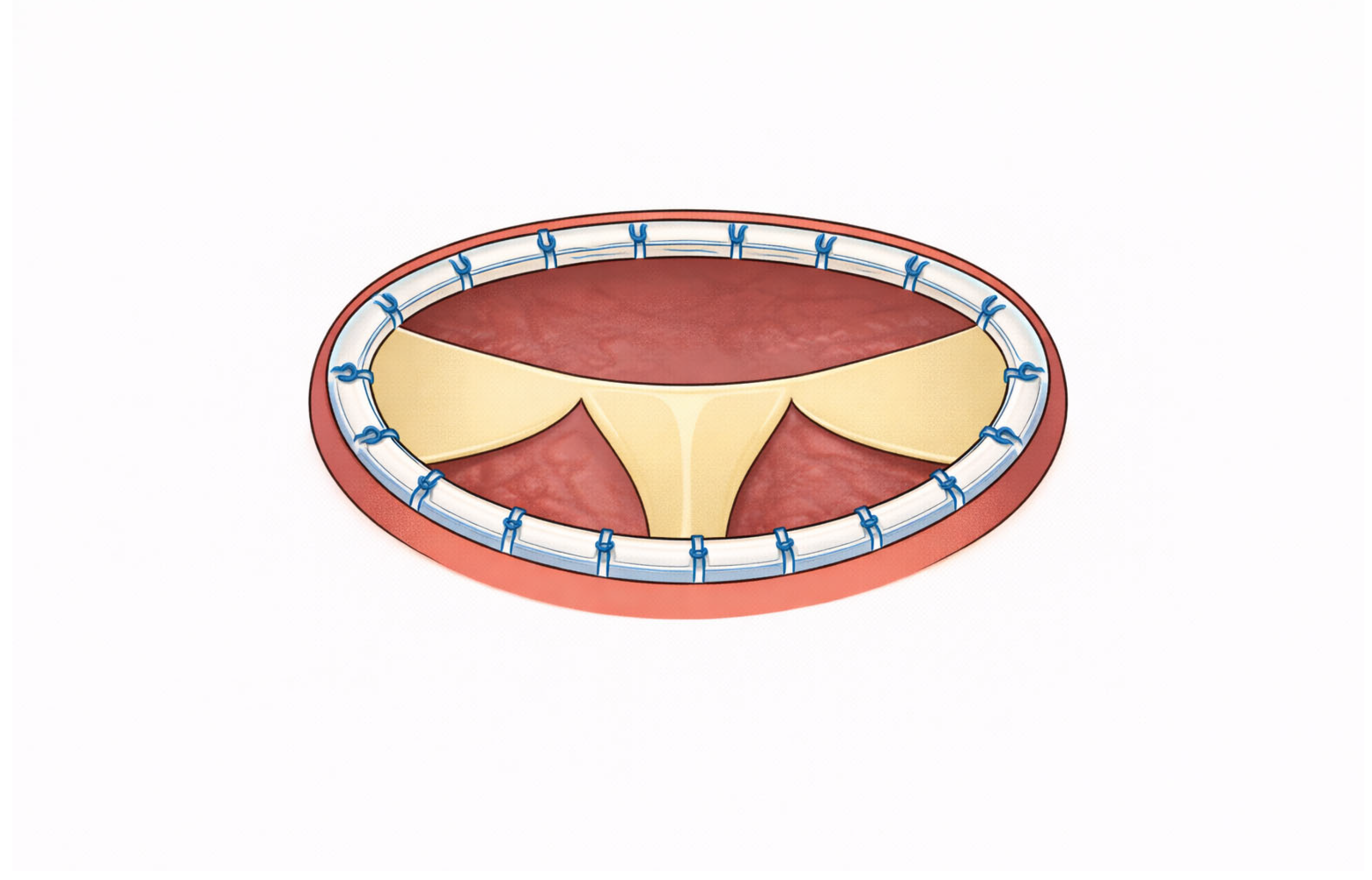
Given the frequent coexistence of atrial functional tricuspid regurgitation, several studies have emphasized the need for an integrated, dual-valve approach [40,44,45,47]. Combined mitral and tricuspid annuloplasty has been shown to result in sustained reduction of both regurgitations and improved functional status, while preoperative left atrial volume index has emerged as an important predictor of late cardiovascular events, reinforcing the prognostic role of advanced atrial remodeling [47] (
Figure 3). Notably, this concept has been acknowledged in the most recent 2025 ESC guidelines on valvular heart disease, which recommend concomitant tricuspid valve intervention in patients with severe tricuspid regurgitation undergoing left-sided valve surgery and suggest consideration of tricuspid repair even in moderate or mild regurgitation in the presence of annular dilatation, with the aim of preventing disease progression and right ventricular remodeling [5].
Left atrial plication has also been proposed as an adjunctive strategy in patients with giant left atria, with evidence suggesting improved correction of atriogenic posterior leaflet tethering and favorable mid-term durability in carefully selected patients [48] (
Table 1).
Parallel to these mechanistic advances, surgical strategies for A-SMR have evolved alongside the widespread adoption of minimally invasive mitral valve surgery [49]. Contemporary minimally invasive platforms allow the full spectrum of mitral repair and concomitant atrial procedures to be performed with outcomes comparable to conventional sternotomy, while offering a less invasive surgical framework [49]. In selected patients with A-SMR and HFpEF, minimally invasive annuloplasty has been associated with durable valve competence and improved long-term survival compared with medical therapy alone [50].
Comparative Outcomes and Predictors of Durability
Comparative surgical studies have further clarified the distinct prognostic profile of A-SMR. Compared with V-SMR, patients with A-SMR exhibit superior long-term survival and lower rates of reoperation following mitral valve surgery, despite similar early operative risk [15]. Importantly, etiological classification has emerged as a key determinant of outcome, supporting tailored surgical decision-making [51].
Within the A-SMR population, long-term follow-up studies extending up to 10 years have provided valuable insights into repair durability [52]. Mitral valve repair has consistently outperformed valve replacement in terms of survival, while recurrent regurgitation has been strongly associated with procedural factors rather than patient comorbidity alone. In particular, the use of partial band annuloplasty has been identified as an independent predictor of recurrent mitral regurgitation, supporting the preferential use of complete annuloplasty rings to ensure durable annular remodeling [52].
Consistent with evidence from predominantly ventricular functional mitral regurgitation populations, partial band annuloplasty has been associated with inferior durability compared with complete rings, reinforcing the importance of complete annular stabilization across etiological substrates [53].
Rigid or semi-rigid complete annuloplasty rings are therefore generally favored to prevent recurrent annular dilatation, with careful sizing required to balance effective annular reduction against the risk of functional mitral stenosis [15]. In contrast to ventricular functional mitral regurgitation, subvalvular repair techniques aimed at modifying papillary muscle position are typically unnecessary in A-SMR, reflecting the limited contribution of ventricular distortion to disease pathophysiology [15].
7.3. Concomitant Atrial Fibrillation Surgery and Left Atrial Appendage Closure
Given the central role of AF in the development and progression of A-SMR, concomitant surgical ablation should be strongly considered at the time of mitral valve repair. Surgical ablation strategies aim to restore sinus rhythm, reduce atrial fibrillation burden, and promote atrial reverse remodeling. This integrated approach addresses both the cause and the consequence of atrial-driven mitral regurgitation [54,55].
Chen et al. reported one of the largest surgical series evaluating mitral valve repair for A-SMR with a specific focus on the impact of concomitant surgical ablation for atrial fibrillation. In a cohort of 82 patients with persistent atrial fibrillation, preserved left ventricular systolic function, and moderate-to-severe A-SMR, all patients underwent mitral valve repair with annuloplasty, and 63% received concomitant Cox–Maze IV ablation (
Figure 2). Cox–Maze IV ablation is a standardized surgical ablation technique for atrial fibrillation that creates a predefined pattern of linear lesions in the atria using energy sources such as bipolar radiofrequency and/or cryoablation, reproducing the lesion set of the original cut-and-sew Cox–Maze III procedure while reducing operative complexity and morbidity. Surgical treatment was associated with significant improvement in functional status and reverse remodeling of both the left atrium and left ventricle, with no operative mortality. Importantly, concomitant surgical ablation was associated with a significantly lower rate of recurrent mitral regurgitation during mid-term follow-up, particularly in patients with smaller left atrial diameters (≤60 mm), whereas this benefit was not observed in those with advanced atrial enlargement [55].
Available evidence suggests that restoration of sinus rhythm may improve atrial function and annular dynamics, potentially enhancing the durability of mitral valve repair [6].
Ye et al. reported a large prospective cohort study evaluating outcomes of mitral valve repair combined with the Cox–Maze procedure in patients with A-SMR and heart failure with recovered ejection fraction. Among 312 consecutively enrolled patients, 247 underwent mitral valve repair plus surgical ablation after guideline-directed medical therapy, including 132 with full Left Ventricular Ejection Fraction (LVEF) recovery (≥50%) and 115 with partial recovery (40–50%). Operative mortality was low and comparable between groups, and overall 5-year survival reached 95% in the total cohort. Importantly, patients with LVEF ≥50% demonstrated significantly better long-term freedom from recurrent MR and AF compared with those with partial recovery, while survival and heart-failure rehospitalization rates were similar after inverse probability weighting adjustment. The authors concluded that mitral valve repair combined with Cox–Maze is safe and effective in patients with A-SMR and recovered systolic function, with more durable rhythm and valve outcomes observed in those achieving full ventricular recovery [56].
Song et al. compared surgical outcomes of mitral valve surgery in patients with A-SMR and degenerative mitral regurgitation (DMR) in a large multicenter cohort of 642 patients, including 82 propensity score–matched patients with A-SMR. Mitral valve repair was the predominant strategy in both groups and was associated with a similarly low rate of recurrent mitral regurgitation, with 5-year freedom from moderate or greater MR of 93.0% in A-SMR and 89.8% in DMR. Despite comparable durability of mitral repair, patients with A-SMR experienced higher rates of heart failure–related readmission and cardiac death at 5 years (88.6% vs 96.3% freedom from HF readmission), reflecting the impact of advanced atrial remodeling. Importantly, following concomitant Maze procedures, A-SMR patients showed a significantly higher incidence of junctional rhythm and permanent pacemaker implantation, highlighting a distinctive rhythm-related risk profile in this population. Overall, the study supports mitral valve repair as an effective surgical option for A-SMR while emphasizing the need for careful rhythm management and postoperative surveillance [57].
Patient selection remains critical, as the likelihood of maintaining sinus rhythm decreases with increasing atrial size and longer duration of atrial fibrillation [28].
Left atrial appendage (LAA) closure at the time of mitral valve intervention is often considered to reduce the long-term risk of stroke in patients with permanent AF, particularly in those with contraindications to long-term anticoagulation [32]. While dedicated randomized data in A-SMR populations are limited, expert consensus and recent evidence support the integration of LAA closure into the surgical strategy for selected patients with persistent or permanent AF, as part of a comprehensive approach to atrial remodeling and thromboembolic risk reduction [39,58,59].
7.4. Outcomes and Durability
Short- and mid-term outcomes following surgical repair of A-SMR are generally favorable in appropriately selected patients, with significant reduction in mitral regurgitation severity and improvement in functional status. However, recurrence of mitral regurgitation remains a concern, particularly in patients with advanced atrial remodeling or persistent atrial fibrillation [38].
Long-term outcome data specific to A-SMR remain limited, highlighting the need for prospective studies with extended follow-up. Nevertheless, from a pathophysiological perspective, surgical annuloplasty—particularly when combined with atrial fibrillation surgery—represents the most definitive treatment option for patients with atrial-driven mitral regurgitation who are suitable operative candidates [8,15]
8. Clinical–Surgical Decision-Making: A Proposed Algorithm
Clinical decision-making in atrial secondary mitral regurgitation should be guided by a mechanism-oriented approach that integrates clinical presentation, imaging findings, and procedural risk. Given the fundamental differences between atrial-driven and ventricular functional mitral regurgitation, accurate identification of A-SMR is the cornerstone of appropriate management and therapeutic selection [10,13].
Patients presenting with functional MR and HFpEF should undergo comprehensive echocardiographic evaluation to confirm the atrial mechanism. Key features supporting a diagnosis of A-SMR include significant left atrial enlargement, mitral annular dilatation, minimal leaflet tethering, and the absence of primary mitral valve disease [20,21,22]. Recognition of these characteristics is essential to avoid misclassification and inappropriate application of treatment strategies designed for V-SMR.
In asymptomatic or mildly symptomatic patients with early-stage A-SMR, initial management should focus on optimization of medical therapy and treatment of associated conditions, including HFpEF and AF [5,6]. Rhythm control strategies may be particularly beneficial in patients with limited atrial remodeling, as restoration of sinus rhythm can promote atrial reverse remodeling and reduce mitral regurgitation severity [6].
Patients with persistent symptoms despite optimized non-surgical management require further stratification based on atrial size, annular dimensions, pulmonary pressures, and right ventricular function. Progressive annular dilatation, persistent atrial fibrillation, and early pulmonary hypertension identify patients at higher risk of disease progression and adverse outcomes if intervention is delayed [27,28].
In patients who are suitable surgical candidates, mitral valve repair based on annuloplasty should be considered the preferred treatment strategy, particularly when intervention is performed before the development of advanced pulmonary hypertension or right ventricular dysfunction [8,15]. Concomitant surgical ablation of atrial fibrillation should be strongly encouraged, as it addresses the underlying atrial pathology and may enhance the durability of mitral repair [6,8].
TEER may be considered in selected patients who are deemed at prohibitive surgical risk or are not candidates for surgery. In these cases, careful patient selection is critical, as the annular-driven nature of A-SMR may limit long-term durability, particularly in the presence of extreme annular enlargement or advanced atrial myopathy [7,37].
Ultimately, management decisions in A-SMR should be individualized and guided by a multidisciplinary Heart Team approach. Integration of clinical status, imaging phenotype, procedural risk, and patient preferences is essential to align treatment strategy with disease mechanism and stage, thereby optimizing clinical outcomes.
9. Future Directions
Despite increasing recognition of A-SMR as a distinct clinical and mechanistic entity, several important knowledge gaps remain, particularly with regard to optimal timing of intervention, patient selection, and surgical strategy. Addressing these gaps will be essential to refine management algorithms and improve long-term outcomes. From a surgical standpoint, future research should prioritize prospective, phenotype-specific studies that clearly distinguish A-SMR from V-SMR and dual-mechanism functional mitral regurgitation [3,13,15]. The heterogeneity of current surgical series, often including mixed phenotypes, limits the ability to draw firm conclusions regarding durability and outcomes of mitral valve repair in pure A-SMR. Standardized diagnostic and imaging criteria integrating annular geometry, leaflet–annulus mismatch, atrial size, and atrial fibrosis burden are needed to improve surgical candidate selection and procedural planning [20,21,22,23,25]. The timing of surgical intervention represents a particularly relevant area for future investigation. Observational data suggest that surgical repair performed before the development of extreme atrial dilatation, advanced pulmonary hypertension, or right ventricular dysfunction is associated with superior durability and clinical outcomes [2,27,38]. However, prospective data defining optimal intervention thresholds are lacking. Future studies comparing early surgical referral versus delayed intervention in patients with progressive A-SMR may clarify whether timely annuloplasty-based repair can interrupt the self-perpetuating cycle of atrial remodeling and regurgitation progression. Further refinement of surgical techniques tailored to disease stage is also warranted. While complete rigid or semi-rigid annuloplasty is widely accepted as the cornerstone of repair in A-SMR [39,52], the role of adjunctive procedures—such as leaflet augmentation, atrial plication, or other atrial remodeling strategies—requires systematic evaluation. Identifying which patients benefit from isolated annuloplasty versus more complex repair strategies remains an unmet clinical need, particularly in advanced stages characterized by atriogenic leaflet tethering [42,43,44,46]. The integration of concomitant atrial fibrillation surgery represents another key area for future research. Available evidence suggests that surgical ablation may reduce atrial fibrillation burden, promote atrial reverse remodeling, and improve the durability of mitral valve repair in selected patients [6,55,56]. However, the benefit of ablation appears attenuated in patients with advanced atrial enlargement and extensive atrial fibrosis [25,28,55]. Future studies should aim to define atrial size, fibrosis extent, and AF duration thresholds beyond which the incremental benefit of surgical ablation becomes limited. Finally, comparative studies specifically focused on surgical repair versus transcatheter therapies in A-SMR populations are needed. While transcatheter edge-to-edge repair represents a valuable option for high-risk or inoperable patients [5,7,33,34,35,36], concerns remain regarding long-term durability in a predominantly annular disease characterized by progressive atrial remodeling [4,37]. Direct comparisons incorporating anatomical, functional, and patient-reported outcomes will be essential to better define the respective roles of surgical and transcatheter interventions within a mechanism-oriented treatment framework.
10. Conclusions
Atrial secondary mitral regurgitation is a distinct and increasingly recognized form of functional mitral regurgitation driven primarily by left atrial remodeling and mitral annular dilatation in the setting of preserved ventricular geometry and systolic function [1,3,10]. Its recognition challenges the traditional ventricular-centered paradigm of functional mitral valve disease and has major implications for diagnosis, risk stratification, and therapeutic decision-making. From a cardio-surgical perspective, A-SMR is fundamentally an annular disease, making mitral valve repair based on complete annuloplasty the most pathophysiologically sound and definitive therapeutic option in suitable candidates [8,15,39]. Surgical annuloplasty directly addresses the dominant mechanism of regurgitation and, when performed before advanced atrial and right-sided remodeling occurs, is associated with favorable durability and clinical outcomes [27,52]. Compared with ventricular functional mitral regurgitation, patients with A-SMR generally demonstrate superior long-term survival and lower rates of reoperation following surgical repair, underscoring the importance of etiological classification in surgical decision-making [15,51]. The frequent coexistence of atrial fibrillation and atrial functional tricuspid regurgitation further supports a comprehensive and integrated surgical strategy, combining mitral annuloplasty with concomitant atrial fibrillation ablation, tricuspid valve repair, and, when appropriate, left atrial appendage closure [40,44,45,47,55,56,57,58,59]. This approach targets both the cause and consequences of atrial remodeling and may enhance long-term valve competence and clinical stability. Nevertheless, surgical outcomes are strongly influenced by disease stage at the time of intervention. Advanced atrial enlargement, extensive atrial fibrosis, persistent atrial fibrillation, and established pulmonary hypertension are consistently associated with higher rates of recurrent mitral regurgitation and less favorable long-term outcomes [27]. These observations highlight the critical importance of timely referral and intervention before irreversible atrial and right-sided remodeling develops. In conclusion, atrial secondary mitral regurgitation should be managed through a mechanism-oriented, multidisciplinary Heart Team approach, with cardiac surgery playing a central role in eligible patients. Continued refinement of diagnostic criteria, surgical techniques, and timing strategies—supported by dedicated prospective studies—will be essential to optimize outcomes and fully define the role of surgical therapy in this increasingly prevalent and clinically relevant disease phenotype.
Author Contributions
Conceptualization, D.V. and G.C.; methodology, D.V.; software, E.M.N; validation, G.S., G.C. and E.M.N; formal analysis, D.V.; investigation, D.V.; resources, D.V.; data curation, G.C.; writing—original draft preparation, D.V.; writing—review and editing, D.V.; visualization, G.C.; supervision, G.S.; project administration, G.C.; funding acquisition, G.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
The authors have reviewed and edited the output and take full responsibility for the content of this publication.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| AF |
Atrial Fibrillation. |
| A-SMR |
Atrial Secondary Mitral Regurgitation |
| CMR |
Cardiac Magnetic Resonance |
| DMR |
Degenerative Mitral Regurgitation |
| HFpEF |
Heart Failure with preserved Ejection Fraction |
| Left Atrial Appendage |
LAA |
| LVEF |
Left Ventricular Ejection Fraction |
| MR |
Mitral Regurgitation |
| MRI |
Magnetic Resonance Imaging |
| SGLT2 |
Sodium–Glucose Cotransporter-2 |
| TEE |
Transesophageal echocardiography |
| TTE |
Transthoracic echocardiography |
| TEER |
Transcatheter Edge-To-Edge Repair |
| V-SMR |
Ventricular Secondary Mitral Regurgitation |
References
- Deferm, S; Bertrand, PB; Verbrugge, FH; Verhaert, D; Rega, F; Thomas, JD; Vandervoort, PM. Atrial Functional Mitral Regurgitation: JACC Review Topic of the Week. J Am Coll Cardiol. 2019, 73(19), 2465–2476. [Google Scholar] [CrossRef] [PubMed]
- Moonen, A; Ng, MKC; Playford, D; Strange, G; Scalia, GM; Celermajer, DS. Atrial functional mitral regurgitation: prevalence, characteristics and outcomes from the National Echo Database of Australia. Open Heart 2023, 10(1), e002180. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, QF; Zhou, X; Katsouras, CS; Ni, C; Zhu, H; Liu, C; Peng, Y; Ge, HB; Hong, C; Lin, WH; Zhou, XD. Atrial and ventricular functional mitral regurgitation: prevalence, characteristics, outcomes, and disease progression. Eur Heart J Cardiovasc Imaging 2025, 26(3), 545–556. [Google Scholar] [CrossRef] [PubMed]
- von Stein, P; von Stein, J; Hohmann, C; Wienemann, H; Guthoff, H; Körber, MI; Baldus, S; Pfister, R; Hahn, RT; Iliadis, C. Atrial Functional Mitral Regurgitation Subtypes Undergoing Transcatheter Edge-to-Edge Repair: Suboptimal Outcomes in Atriogenic Hamstringing. JACC Cardiovasc Imaging 2025, 18(1), 16–29. [Google Scholar] [CrossRef] [PubMed]
- Praz, F; ESC/EACTS Scientific Document Group. 2025 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J 2025, 46(44), 4635–4736. [Google Scholar] [CrossRef] [PubMed]
- Soulat-Dufour, L; Lang, S; Addetia, K; Ederhy, S; Adavane-Scheuble, S; Chauvet-Droit, M; Jean, ML; Nhan, P; Ben Said, R; Kamami, I; Issaurat, P; Capderou, E; Arnaud, C; Boccara, F; Lang, RM; Cohen, A. Restoring Sinus Rhythm Reverses Cardiac Remodeling and Reduces Valvular Regurgitation in Patients With Atrial Fibrillation. J Am Coll Cardiol. 2022, 79(10), 951–961. [Google Scholar] [CrossRef] [PubMed]
- Doldi, P; Stolz, L; Orban, M; Karam, N; Praz, F; Kalbacher, D; Lubos, E; Braun, D; Adamo, M; Giannini, C; Melica, B; Näbauer, M; Higuchi, S; Wild, M; Neuss, M; Butter, C; Kassar, M; Petrescu, A; Pfister, R; Iliadis, C; Unterhuber, M; Thiele, H; Baldus, S; von Bardeleben, RS; Schofer, N; Hagl, C; Petronio, AS; Massberg, S; Windecker, S; Lurz, P; Metra, M; Hausleiter, J. Transcatheter Mitral Valve Repair in Patients With Atrial Functional Mitral Regurgitation. JACC Cardiovasc Imaging 2022, 15(11), 1843–1851. [Google Scholar] [CrossRef] [PubMed]
- Wagner, CM; Brescia, AA; Watt, TMF; Bergquist, C; Rosenbloom, LM; Ceniza, NN; Markey, GE; Ailawadi, G; Romano, MA; Bolling, SF; Michigan Mitral Research Group. Surgical strategy and outcomes for atrial functional mitral regurgitation: All functional mitral regurgitation is not the same! J Thorac Cardiovasc Surg. Epub. 2024, 167(2), 647–655. [Google Scholar] [CrossRef] [PubMed]
- Huang, AL; Dal-Bianco, JP; Levine, RA; Hung, JW. Secondary Mitral Regurgitation: Cardiac Remodeling, Diagnosis, and Management. Struct Heart 2022, 7(3), 100129. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bartko, PE; Arfsten, H; Heitzinger, G; Pavo, N; Toma, A; Strunk, G; Hengstenberg, C; Hülsmann, M; Goliasch, G. A Unifying Concept for the Quantitative Assessment of Secondary Mitral Regurgitation. J Am Coll Cardiol. 2019, 73(20), 2506–2517. [Google Scholar] [CrossRef] [PubMed]
- Naser, JA; Michelena, HI; Lin, G; Scott, CG; Lee, E; Kennedy, AM; Noseworthy, PA; Pellikka, PA; Nkomo, VT; Pislaru, SV. Incidence, risk factors, and outcomes of atrial functional mitral regurgitation in patients with atrial fibrillation or sinus rhythm. Eur Heart J Cardiovasc Imaging 2023, 24(11), 1450–1457. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, C; Okada, A; Nishimura, K; Moriuchi, K; Amano, M; Takahama, H; Amaki, M; Hasegawa, T; Kanzaki, H; Fujita, T; Kobayashi, J; Yasuda, S; Izumi, C. Prognostic comparison of atrial and ventricular functional mitral regurgitation. Open Heart 2021, 8(1), e001574. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bartko, PE; Heitzinger, G; Arfsten, H; Pavo, N; Spinka, G; Andreas, M; Mascherbauer, J; Hengstenberg, C; Hülsmann, M; Goliasch, G. Disproportionate Functional Mitral Regurgitation: Advancing a Conceptual Framework to Clinical Practice. JACC Cardiovasc Imaging 2019, 12(10), 2088–2090. [Google Scholar] [CrossRef] [PubMed]
- Bao, C; Wei, K; Zhao, D; Zhang, J; Mei, J; Ma, N. Surgical Strategy for Atrial Functional Mitral Regurgitation With Atrial Fibrillation. J Am Heart Assoc. 2025, 14(19), e039019. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hirji, SA; Cote, CL; Javadikasgari, H; Malarczyk, A; McGurk, S; Kaneko, T. Atrial functional versus ventricular functional mitral regurgitation: Prognostic implications. J Thorac Cardiovasc Surg. 2022, 164(6), 1808–1815.e4. [Google Scholar] [CrossRef] [PubMed]
- Zoghbi, WA; Levine, RA; Flachskampf, F; Grayburn, P; Gillam, L; Leipsic, J; Thomas, JD; Kwong, RY; Vandervoort, P; Chandrashekhar, Y. Atrial Functional Mitral Regurgitation: A JACC: Cardiovascular Imaging Expert Panel Viewpoint. JACC Cardiovasc Imaging 2022, 15(11), 1870–1882. [Google Scholar] [CrossRef] [PubMed]
- Machino-Ohtsuka, T; Seo, Y; Ishizu, T; Sato, K; Sugano, A; Yamamoto, M; Hamada-Harimura, Y; Aonuma, K. Mechanistic Insights Into Atrial Functional Mitral Regurgitation - 3-Dimensional Echocardiographic Study. Circ J 2016, 80(10), 2240–8. [Google Scholar] [CrossRef] [PubMed]
- Uno, G; Omori, T; Shimada, S; Rader, F; Siegel, RJ; Shiota, T. Differences in mitral valve geometry between atrial and ventricular functional mitral regurgitation in patients with atrial fibrillation: a 3D transoesophageal echocardiography study. Eur Heart J Cardiovasc Imaging 2021, 22(10), 1106–1116. [Google Scholar] [CrossRef] [PubMed]
- Farhan, S; Silbiger, JJ; Halperin, JL; Zhang, L; Dukkipati, SR; Vogel, B; Kini, A; Sharma, S; Lerakis, S. Pathophysiology, Echocardiographic Diagnosis, and Treatment of Atrial Functional Mitral Regurgitation: JACC State-of-the-Art Review. J Am Coll Cardiol. Erratum in: J Am Coll Cardiol. 2023 Feb 21;81(7):711. doi: 10.1016/j.jacc.2023.01.003. PMID: 36480974. 2022, 80(24), 2314–2330. [Google Scholar] [CrossRef]
- Mesi, O; Gad, MM; Crane, AD; Ramchand, J; Puri, R; Layoun, H; Miyasaka, R; Gillinov, MA; Wierup, P; Griffin, BP; Kapadia, SR; Harb, SC. Severe Atrial Functional Mitral Regurgitation: Clinical and Echocardiographic Characteristics, Management and Outcomes. JACC Cardiovasc Imaging 2021, 14(4), 797–808. [Google Scholar] [CrossRef] [PubMed]
- Reid, A; Ben Zekry, S; Naoum, C; Takagi, H; Thompson, C; Godoy, M; Anastasius, M; Tarazi, S; Turaga, M; Boone, R; Webb, J; Leipsic, J; Blanke, P. Geometric differences of the mitral valve apparatus in atrial and ventricular functional mitral regurgitation. J Cardiovasc Comput Tomogr. Epub. 2022, 16(5), 431–441. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shoshan, J; Overtchook, P; Buithieu, J; Mousavi, N; Martucci, G; Spaziano, M; de Varennes, B; Lachapelle, K; Brophy, J; Modine, T; Baumbach, A; Maisano, F; Prendergast, B; Tamburino, C; Windecker, S; Piazza, N. Predictors of Outcomes Following Transcatheter Edge-to-Edge Mitral Valve Repair. JACC Cardiovasc Interv 2020, 13(15), 1733–1748. [Google Scholar] [CrossRef] [PubMed]
- Flachskampf, FA; Beyer, R; Fehske, W. Atrial functional mitral regurgitation: definition, features, and unknowns. Clin Res Cardiol 2025. [Google Scholar] [CrossRef] [PubMed]
- Garg, P; Pavon, AG; Penicka, M; Uretsky, S. Cardiovascular magnetic resonance imaging in mitral valve disease. Eur Heart J 2025, 46(7), 606–619. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Marrouche, NF; Wilber, D; Hindricks, G; Jais, P; Akoum, N; Marchlinski, F; Kholmovski, E; Burgon, N; Hu, N; Mont, L; Deneke, T; Duytschaever, M; Neumann, T; Mansour, M; Mahnkopf, C; Herweg, B; Daoud, E; Wissner, E; Bansmann, P; Brachmann, J. Association of atrial tissue fibrosis identified by delayed enhancement MRI and atrial fibrillation catheter ablation: the DECAAF study. JAMA Erratum in: JAMA. 2014 Nov 5;312(17):1805. PMID: 24496537. 2014, 311(5), 498–506. [Google Scholar] [CrossRef]
- Cawley, PJ; Hamilton-Craig, C; Owens, DS; Krieger, EV; Strugnell, WE; Mitsumori, L; D'Jang, CL; Schwaegler, RG; Nguyen, KQ; Nguyen, B; Maki, JH; Otto, CM. Prospective comparison of valve regurgitation quantitation by cardiac magnetic resonance imaging and transthoracic echocardiography. Circ Cardiovasc Imaging 2013, 6(1), 48–57. [Google Scholar] [CrossRef] [PubMed]
- Shin, JH; Lee, SH; Joo, HC; Youn, YN; Kim, JH; Lee, S. Long-term Outcomes of Mitral Valve Repair for Atrial Functional Mitral Regurgitation. Ann Thorac Surg 2025, 120(1), 71–78. [Google Scholar] [CrossRef] [PubMed]
- Naser, JA; Alexandrino, FB; Harada, T; Michelena, HI; Borlaug, BA; Eleid, MF; Lin, G; Scott, C; Kennedy, AM; Pellikka, PA; Nkomo, VT; Pislaru, SV. The Natural History of Atrial Functional Mitral Regurgitation. J Am Coll Cardiol. 2024, 83(16), 1495–1507. [Google Scholar] [CrossRef] [PubMed]
- Anker, SD. EMPEROR-Preserved Trial Investigators. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N Engl J Med. 2021, 385(16), 1451–1461. [Google Scholar] [CrossRef] [PubMed]
- Solomon, SD; DELIVER Trial Committees and Investigators. Dapagliflozin in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N Engl J Med. 2022, 387(12), 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Al-Hashedi, EM; Abdu, FA; Ahmad, A; Yu, Y; Li, R; Mohammed, AA; Wu, Y. Impact of Atrial Fibrillation Catheter Ablation on Functional Mitral Regurgitation. Am J Cardiol. 2025, 254, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Van Gelder, IC; ESC Scientific Document Group. 2024 ESC Guidelines for the management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J Erratum in: Eur Heart J. 2025 Nov 3;46(41):4349. doi: 10.1093/eurheartj/ehaf306. PMID: 39210723. 2024, 45(36), 3314–3414. [Google Scholar] [CrossRef]
- Masiero, G. Impact of Transcatheter Edge-to-Edge Mitral Valve Repair on Atrial Functional Mitral Regurgitation from the GIOTTO Registry. Am J Cardiol. 2024, 211, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Yoon, SH. Outcomes After Transcatheter Edge-to-Edge Mitral Valve Repair According to Mitral Regurgitation Etiology and Cardiac Remodeling. JACC Cardiovasc Interv. 2022, 15(17), 1711–1722. [Google Scholar] [CrossRef] [PubMed]
- Benito-González, T. Clinical and echocardiographic outcomes of transcatheter mitral valve repair in atrial functional mitral regurgitation. Int J Cardiol 2021, 345, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Popolo Rubbio, A. Transcatheter edge-to-edge mitral valve repair in atrial functional mitral regurgitation: insights from the multi-center MITRA-TUNE registry. Int J Cardiol 2022, 349, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T; Sugiura, A; Vogelhuber, J; Öztürk, C; Böhm, L; Wilde, N; Zimmer, S; Nickenig, G; Weber, M. Outcomes of transcatheter edge-to-edge repair for atrial functional mitral regurgitation. EuroIntervention 2024, 20(4), e250–e260. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rudolph, V; et al. Transcatheter repair versus surgery for atrial versus ventricular functional mitral regurgitation: a post hoc analysis of the MATTERHORN trial. Circulation 2025. [Google Scholar] [CrossRef]
- Kagiyama, N. Clinical Outcomes of Mitral Valve Surgery in Atrial Functional Mitral Regurgitation in the REVEAL-AFMR Registry. JAMA Netw Open 2024, 7(8), e2428032. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Vohra, HA; Whistance, RN; Magan, A; Sadeque, SA; Livesey, SA. Mitral valve repair for severe mitral regurgitation secondary to lone atrial fibrillation. Eur J Cardiothorac Surg 2012, 42(4), 634–7. [Google Scholar] [CrossRef] [PubMed]
- Amabile, A; Fereydooni, S; Mori, M; Hameed, I; Jung, J; Krane, M; Geirsson, A. Variable definitions and treatment approaches for atrial functional mitral regurgitation: A scoping review of the literature. J Card Surg. 2022, 37(5), 1182–1191. [Google Scholar] [CrossRef] [PubMed]
- Aranda-Domene, R; Canovas, SJ. Going further of mitral ring annuloplasty: the role of surgery in atrial functional mitral regurgitation. J Thorac Dis. 2023, 15(5), 2381–2384. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nappi, F. Atrial functional mitral regurgitation in cardiology and cardiac surgery. J Thorac Dis. 2024, 16(8), 5435–5456. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shibata, T; Takahashi, Y; Fujii, H; Morisaki, A; Abe, Y. Surgical considerations for atrial functional regurgitation of the mitral and tricuspid valves based on the etiological mechanism. Gen Thorac Cardiovasc Surg 2021, 69(7), 1041–1049. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Takahashi, Y; Abe, Y; Sasaki, Y; Bito, Y; Morisaki, A; Nishimura, S; Shibata, T. Mitral valve repair for atrial functional mitral regurgitation in patients with chronic atrial fibrillation. Interact Cardiovasc Thorac Surg 2015, 21(2), 163–8. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, T; Totsugawa, T; Orihashi, K; Kihara, K; Tamura, K; Hiraoka, A; Chikazawa, G; Yoshitaka, H. Mitral annuloplasty for atrial functional mitral regurgitation in patients with chronic atrial fibrillation. J Card Surg. 2019, 34(9), 767–773. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y; Abe, Y; Takashi, M; Fujii, H; Morisaki, A; Nishimura, S; Sakon, Y; Ito, K; Shintani, A; Shibata, T. Mid-term results of valve repairs for atrial functional mitral and tricuspid regurgitations. Gen Thorac Cardiovasc Surg 2020, 68(5), 467–476. [Google Scholar] [CrossRef] [PubMed]
- Matsumori, M; Kawashima, M; Aihara, T; Fujisue, J; Fujimoto, M; Fukase, K; Nomura, Y; Tanaka, H; Murakami, H; Mukohara, N. Efficacy of left atrial plication for atrial functional mitral regurgitation. Gen Thorac Cardiovasc Surg 2021, 69(3), 458–465. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- El-Andari, R; Watkins, AR; Fialka, NM; Kang, JJH; Bozso, SJ; Fatehi Hassanabad, A; Vasanthan, V; Adams, C; Cook, R; Moon, MC; Nagendran, J; Kent, W. Western Canadian Minimally Invasive Cardiac Surgery Working Group. Minimally Invasive Approaches to Mitral Valve Surgery: Where Are We Now? A Narrative Review. Can J Cardiol. 2024, 40(9), 1679–1689. [Google Scholar] [CrossRef] [PubMed]
- Balogh, Z; Mizukami, T; Bartunek, J; Collet, C; Beles, M; Albano, M; Katbeh, A; Casselman, F; Vanderheyden, M; Van Camp, G; Van Praet, F; Penicka, M. Mitral Valve Repair of Atrial Functional Mitral Regurgitation in Heart Failure with Preserved Ejection Fraction. J Clin Med. 2020, 9(11), 3432. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Carino, D; Lapenna, E; Ascione, G; Ruggeri, S; Del Forno, B; Castiglioni, A; Alfieri, O; De Bonis, M. Is mitral annuloplasty an effective treatment for severe atrial functional mitral regurgitation? J Card Surg. 2021, 36(2), 596–602. [Google Scholar] [CrossRef] [PubMed]
- Kawamoto, N; Fukushima, S; Kainuma, S; Ikuta, A; Tadokoro, N; Kakuta, T; Fujita, T. Mitral valve surgery for atrial functional mitral regurgitation: predicting recurrent mitral regurgitation and mid-term outcome. Gen Thorac Cardiovasc Surg 2022, 70(9), 761–769. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kwon, MH; Lee, LS; Cevasco, M; Couper, GS; Shekar, PS; Cohn, LH; Chen, FY. Recurrence of mitral regurgitation after partial versus complete mitral valve ring annuloplasty for functional mitral regurgitation. J Thorac Cardiovasc Surg. 2013, 146(3), 616–22. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q. Catheter Ablation or Surgical Therapy in Severe Atrial Functional Mitral Regurgitation Caused by Long-Standing Persistent Atrial Fibrillation-Propensity Score Analysis. J Am Heart Assoc. 2024, 13(23), e035695. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, J. Mitral valve repair and surgical ablation for atrial functional mitral regurgitation. Ann Transl Med. 2020, 8(21), 1420. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ye, Q; Zhao, Y; Bai, C; Liu, K; Zhao, C; Liu, Y; Li, Y; Wang, J. Outcome of mitral repair combined with Cox-maze procedure for atrial functional mitral regurgitation with heart failure with recovered ejection fraction. Eur J Cardiothorac Surg 2023, 64(2), ezad273. [Google Scholar] [CrossRef] [PubMed]
- Song, K; Lee, JH; Woo, HT; Kim, YS; Jang, WS; Chung, S; Cho, YH; Kim, WS; Sung, K. Surgical outcome of mitral valve surgery in atrial functional mitral regurgitation compared with degenerative etiology. J Thorac Cardiovasc Surg. 2025, 169(5), 1471–1482.e4. [Google Scholar] [CrossRef] [PubMed]
- Wagner, CM; Michigan Mitral Research Group. Surgical strategy and outcomes for atrial functional mitral regurgitation: All functional mitral regurgitation is not the same! J Thorac Cardiovasc Surg. 2024, 167(2), 647–655. [Google Scholar] [CrossRef] [PubMed]
- Pagnesi, M. Epidemiology, pathophysiology, diagnosis and management of atrial functional mitral regurgitation: An expert opinion paper. ESC Heart Fail 2025, 12(6), 3788–3805. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).