Submitted:
27 January 2026
Posted:
03 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Definition
1.2. Manifestations, Origins, Causes, and Consequences
1.2.1. Hyperalgesia and Allodynia
- Allodynia
- Hyperalgesia
1.2.2. Origins
- Chronic Cutaneous Pain
- Chronic Pruritus
- Chronic Muscle Pain
- Chronic Visceral Pain
1.2.3. Causes
1.2.4. Chronic Inflammatory Pain (with Case Report)
- Case Report: Transition from Acute Inflammatory Pain to Chronic Pain
1.2.5. Chronic Neuropathic Pain (CNP) (with Case Report)
- Causes
- Symptoms
- Mechanisms
- Neuroplasticity
- Case Report: Chronic Peripheral Neuropathic Pain with Central Sensitization
- Reduced pinprick and temperature sensation in a bilateral stocking distribution
- Preserved vibration sense proximally
- Absent ankle reflexes
- Marked dynamic mechanical allodynia to light brushing
- Secondary hyperalgesia extending beyond the initially affected dermatomes
- Positive neuropathic symptoms (burning pain, electric shocks, allodynia)
- Negative symptoms (thermal hypoesthesia)
- Evidence of central sensitization, indicated by mirror-image pain and extraterritorial hyperalgesia
- Peripheral sensitization, driven by metabolic injury to nociceptive fibers, resulting in spontaneous activity and reduced activation thresholds.
- Neuroinflammation, initiated by nerve injury and sustained by immune–neuronal interactions involving Schwann cells and macrophages, leading to persistent peripheral input.
- Central sensitization, reflected by exaggerated pain responses, expanded receptive fields, and allodynia, likely mediated by NMDA-receptor–dependent synaptic plasticity in spinal DH neurons.
- Glial activation, with microglial and astrocytic release of cytokines and neurotrophic factors contributing to altered excitability and maintenance of pain states.
- Maladaptive neuroplasticity, extending to supraspinal structures including the THAL, ACC, AMY, and PFC, reinforcing the emotional–motivational dimension of pain
1.2.6. Peripheral and Central Sensitization
1.2.7. Neuroplasticity
- Human Brain Imaging
- Central Nervous System (CNS) Alterations
- Structural Alterations
- Changes in Connectivity
- Immune System Involvement
- Cognitive and Affective Changes
2. The Functional Structures of Chronic Pain
- Peripheral Sensitization
2.1. Changes in Nociceptors
- Cutaneous Nociceptors
- Muscle Nociceptors
- Visceral Nociceptors
- Peripheral Plasticity
- Neuromodulation
- Ion-channel Changes
2.1.1. Peripheral Sensitization to Inflammation
- Substance P (SP)
- Protons (H+)
- Glutamate
2.1.2. Peripheral Sensitization after Nerve Injury
- De-afferentation Pain
- Centralization of Primary Afferent Activity
- Mechanisms
- Oligodendrocytes
2.1.3. Changes in Sympathetic Modulation of Nociceptors
- Catecholamines
- Sprouting of Postganglionic Sympathetic Fibers within Dorsal-Root Ganglion (DRG)
2.2. Changes in the Spinal Cord
2.2.1. Loss of Inhibition
2.2.2. Changes in Presynaptic Inhibition (PSI)
- Inhibitory Parvalbumin (PV) INTs (INTs)
2.2.3. Increase in Dorsal-Root Reflexes (DRRs)
2.2.4. Shift in Anion Gradients
2.2.5. Changes in Intrinsic Neuron Properties
- Injury Discharge
- Wide-dynamic Range (WDR) Neurons
2.2.6. Changes in Receptive-Fields
2.2.7. Sensory Axonal Sprouting
- Peripheral Sprouting
- Central Sprouting
2.2.8. Changes in Network Structures
2.2.9. Synaptic Changes
- Spinal Short-term Plasticity (STP)
- Long-term Potentiation (LTP)
- Windup
- Nitric Oxide (NO)
- Changes in Synaptic Connections
2.2.10. Activation of the Immune System
- Myositis
- Chronic Stress
2.3. The PAG-Triad Connection
2.3.1. Peri-Aqueductal Gray (PAG)
2.3.2. RVM-CVLM-DReN Triad
2.3.3. Rostral Ventro-medial Medulla (RVM)
- Input/Output
- ON- and OFF-cells
- Imbalance between Inhibition and Facilitation
- μ-Opioid RVM Cells
- Wide-dynamic Range (WDR)Neurons
2.3.4. Caudal Ventro-Lateral Medulla (CVLM)
- Nociceptive Inputs
- Other Inputs
- Outputs
2.3.5. Dorsal Reticular Nucleus (DReN)
- Inputs
- Outputs
- Dorsal Horn (DH)-Dorsal Reticular Nucleus (DReN)-Cerebellum Connections
- Dorsal Reticular Nucleus (DReN)and Wide-dynamic Range (WDR) Neurons
- Dorsal Reticular Nucleus (DReN) and Wide-dynamic Range (WDR) Neurons in Diffuse Noxious Inhibitory Control (DNIC)
2.3.6. The RVM-VLM-DReN Triad in Chronic Pain
- Activity Changes
- Changes in Peri-aqueductal Gray (PAG) and Rostral Ventro-medial Medulla (RVM)
- Chronic Inflammatory Pain
- Locus Coeruleus→Dorsal Reticular Nucleus (DReN) Pathway
- Spinal Hypersensitivity
- Impact of Chronic Stress
2.4. Nucleus Tractus Solitarii (NTS)
- Inputs
- Outputs
- Functions
- Nucleus Tractus Solitarii (NTS) and Chronic Pain
2.5. Parabrachial Nucleus (PBN)
- Parabrachial Nucleus (PBN)-Peri-aqueductal Gray (PAG) Connection
- Parabrachial Nucleus (PBN)-Rostral Ventro-medial Medulla (RVM)Connection
- Parabrachial Nucleus (PBN)-Central Nucleus of Amygdala (CeA) Connection
2.6. Cerebellum
2.7. Midbrain Dopamine (DA) Cell Groups
- Neuronal Plasticity and Functional Connectivity
- Dorsal Raphé Nucleus (DRN)-Ventral Tegmental Area (VTA) Pathway
- Ventral Tegmental Area (VTA)-Nucleus Accumbens (NAc) Connection
- Ventral Tegmental Area (VTA)-Prefrontal Cortex (PFC) Connection
2.8. Locus Coeruleus (LC) and Other Noradrenergic (NA) Cell Groups
2.8.1. Locus Coeruleus (LC) and Subnucleus Reticularis Dorsalis (SRD)
- Human Brain Imaging
- Affective Dimensions
- Pain Chronification
- Locus Coeruleus (LC) and Descending Pain Modulation
- Diffuse Noxious Inhibitory Control (DNIC)
2.8.2. A1-A7 (NA) and Adrenergic Cell Groups
- A1, A2, C1, C2 Cell Groups
- A5 Cell Group
- A7 Cell Group
2.9. Raphé Nuclei (RN)
2.9.1. Dorsal Raphé Nucleus (DRN)
- Dorsal Raphé Nucleus (DRN) in Pain Regulation
- Anterior Cingulate Cortex (ACC)-Dorsal Raphé Nucleus (DRN)Connection
2.9.2. Nucleus Raphé Obscurus (NRO) and Nucleus Raphé Pallidus (NRP)
2.10. Hypothalamus (HYP)
2.10.1. Hypothalamic-Pituitary-Adrenal (HPA) Axis
2.10.2. Hypothalamic-Pituitary-Thyroid (HPT) Axis
2.10.3. Hypothalamic-Pituitary-Gonadal (HPG) Axis
2.11. Basal Ganglia (BG)
2.11.1. Striatum
2.11.2. Nucleus Accumbens (NAc)
- Inputs/Outputs
- Functions
- Alterations ofNucleus Accumbens (NAc)
- Cortico-Nucleus Accumbens (NAc)Connections
2.11.3. Subthalamic Nucleus (STN)
- Anterior Cingulate Cortex (ACC)-Subthalamic Nucleus (STN)Pathway
- Substantia Nigra Reticularis (SNr)-Subthalamic Nucleus (STN)-Parabrachial Nucleus (PBN) Pathway
2.11.4. Substantia Nigra Pars Reticularis (SNr)
2.12. Amygdala (AMY)
- Amygdalar Role in Pain Processing
- Unpleasant Pain Representation in Baso-lateral Amygdala (BLA)
- Amygdalar Hypertrophy and Changed Activity in Rats
- Amygdalar Activation or Hyper-activity in Humans
- Neuropathic Pain
- Disturbed Interactions between Amygdala (AMY) and Cortical Regions
- Baso-lateral Amygdala (BLA)-Prefrontal Cortex (PFC)-Peri-aqueductal Gray (PAG)-Spinal Cord Pathway
- Amygdala (AMY)-Nucleus Accumbens (NAc) Circuit
- Neuroplasticity in Central Nucleus of Amygdala (CeA)
2.13. Habenula (Hb)
2.14. Thalamus (THAL)
- Thalamic (THAL) Re-organization
- Thalamic (THAL) Discharge
- Posterior Paraventricular Nucleus (pPVT) Neurons
- Paraventricular Nucleus of THAL (PVT) to Baso-lateral Amygdala (BLA) Connection
- Thalamo-cortical Connections
- Nucleus Accumbens (NAc) to THAL Connection
2.15. Cerebral Cortex
- Re-organization
- Gray Matter Loss
2.15.1. Somatosensory Cortices
- Activity of S1
2.15.2. Primary Motor Cortex (M1)
- Primary Motor Cortex (M1) Stimulation
2.15.3. Prefrontal Cortex (PFC) and Associates
- Epigenetic Re-programming
- Prefrontal Cortex (PFC)-Nucleus Accumbens (NAc) Connection
- Prefrontal Cortex (PFC)-Amygdala (AMY) Connections
- Medial Prefrontal Cortex (mPFC)
- Cortical Re-organization
- Re-arrangement of Excitatory Inputs
- Changes inMedial Prefrontal Cortex (mPFC)Activity
- Medial Prefrontal Cortex (mPFC)-Thalamus (THAL) Connections
- Medial Prefrontal Cortex (mPFC)-Peri-aqueductal Gray (PAG) Connections
- Dorso-medial Prefrontal Cortex (dmPFC)
- Ventro-medial Prefrontal Cortex (vmPFC)
- Dorso-Lateral Prefrontal Cortex (dlPFC)
- Ventro-Lateral Prefrontal Cortex (vlPFC)
- Social Pain
- Ventro-Lateral Orbito-Frontal Cortex (vlOFC)
2.15.4. Anterior: Cingulate Cortex (ACC)
- Anterior Cingulate Cortex (ACC)Functions
- Nociceptive Inputs
- Anterior Cingulate Cortex (ACC)Connections
- Anterior Cingulate Cortex (ACC) during Neuropathic Pain
- Anterior Cingulate Cortex (ACC) Hyperexcitability
- Synaptic Transmission
- Anterior Cingulate Cortex (ACC) and Dorsal Horn (DH)
- Stress-induced Pain Facilitation
2.15.5. Insular Cortex (IC)
- Structure and Connections
- Inputs
- Outputs
- Insula´s Role in Pain
- Structural Changes
- Activity Changes
- Insular Cortex (IC) Stimulation
- Chronic Constriction Injury (CCI) of Infra-orbital Nerve
- Insular Cortex (IC)Connections to Thalamus (THAL) and Amygdala (AMY)
- Desending Pain Control
2.15.6. Hippocampus (HIPP)
- Structure
- Pain Processing
- Changes in Hippocampus (HIPP)Volume and Connectivity
- Changes in Firing Patterns
- Inflammation
- Neurogenesis
- Plasticity
2.15.7. Bed Nucleus of the Stria Terminalis (BNST)
3. Changes in the Motor Changes in the Motor and Associated Systems
3.1. Changes in Proprioceptive Functions
3.1.1. Changes in Muscle Spindle Afferents
- Sympathetic Innervation of Muscle Spindles
- Ion Channels
3.1.2. Changes in Golgi Tendon Organ (GTO) Afferents
- Ion Channels
3.1.3. Intermezzo
3.2. Changes in Spinal Somatic Reflexes
- Presynaptic (PSI) and Recurrent Inhibition
- Withdrawal Reaction
3.3. Pain and Posture
3.4. Pain and Locomotion
3.5. Musculo-Skeletal Pain Disorders
3.6. Cardio-Vascular Reactions
3.7. Respiratory Reactions
4. Clinical Syndromes
4.1. Phantom Pain
4.2. Post-Herpetic Neuropathy
4.3. Trigeminal Neuropathy (TN)
4.4. Pain After SCI
4.5. Chronic Widespread Non-Inflammatory Muscle Pain (CWP): Fibromyalgia, Myofascial Syndromes, Chronic Fatigue Syndrome (CFS)
4.6. Chronic or Recurrent Low Back Pain (cLBP)
4.7. Complex Regional Pain Syndrome (CRPS)
4.8. Summary
5. Concluding Remarks
Funding
Acknowledgments
Conflicts of Interest
Ethics Approval and Consent to Participate
Abbreviations:
References
- Abbári Rossi Manciopi, P; Rinaldi, NM; Moraes, R. Prehension combined with gait in individuals with chronic low back pain. Motor Control 2017, 21, 90–111. [Google Scholar] [CrossRef]
- Abdulla, FA; Moran, TD; Balasubramanyan, S; Smith, PA. Effects and consequences of nerve injury on the electrical properties of sensory neurons. Can J Physiol Pharmacol 2003, 81, 663–682. [Google Scholar] [CrossRef]
- Adamaszek, M; D´Agata, F; Ferrucci, R; Habas, C; Keulen, S; Kirkby, KC; Leggio, M; Mariёn, P; Molinari, M; Moulton, E; Orsi, L; Van Overwalle, F; Papadelis, C; Priori, A; Sacchetti, L; Schutter, DJ; Styliadis, C; Verhoeven, J. Consensus Paper: Cerebellum and emotion. Cerebellum 2017, 16, 552–576. [Google Scholar] [CrossRef] [PubMed]
- Allen, HN; Bobnar, HJ; Kolber, BJ. Left and right hemispheric lateralization of the amygdala in pain. Prog Neurobiol 2021, 196, 101891. [Google Scholar] [CrossRef]
- Alles, SRA; Smith, PA. Etiology and pharmacology of neuropathic pain. Pharmacol Rev 2018, 70, 315–347. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, P; Dieb, W; Hafidi, A; Voisin, DL; Dallel, R. Insular cortex representation of dynamic mechanical allodynia in trigeminal neuropathic rats. Neurobiol Dis 2009, 33, 89–95. [Google Scholar] [CrossRef]
- Anderson, EO; Schneider, ER; Bagriantsev, SN. Piezo2 in cutaneous and proprioceptive mechanotransduction in vertebrates. Curr Top Membr 2017, 79, 197–217. [Google Scholar] [PubMed]
- Antunes, GF; Pinheiro Campos, AC; Varin de Assis, D; Gouveia, FV; Dias de Jesus Seno, M; Pagano, RL; Ruiz Martinez, RC. Habenula activation patterns in a preclinical model of neuropathic pain accompanied by depressive-like behaviour. PloS One 2022, 17, e0271295. [Google Scholar] [CrossRef]
- Apkarian, AV; Baliki, MN; Geha, PY. Towards a theory of chronic pain. Prog Neurobiol 2009, 87, 81–97. [Google Scholar] [CrossRef]
- Arslan, D; Çevik, IÜ. Interactions between the painful disorders and the autonomic nervous system. Agri 2022, 34, 155–165. [Google Scholar] [CrossRef]
- Artola, A; Voisin, D; Dallel, R. PKCγ interneurons, a gateway to pathological pain in the dorsal horn. J Neural Transm (Vienna) 2020, 127, 527–540. [Google Scholar] [CrossRef]
- Ashyüce, YÖ; Demirel, A; Ülger, Ö. Investigation of joint position sense and balance in individuals with chronic idiopathic neck pain: A cross-sectional study. J Manipulative Physiol Ther 2022, 45, 188–195. [Google Scholar]
- Aternali, A; Katz, J. Recent advances in understanding and managing phantom limb pain. F1000Research 2019, 8, 1167. [Google Scholar] [CrossRef]
- Austin, J; Fiore, NT. Supraspinal neuroimmune crosstalk in chronic pain states. Curr Opin Physiol 2019, 11, 7–15. [Google Scholar] [CrossRef]
- Bai, Y; Chen, Y-B; Qiu, X-T; Chen, Y-B; Ma, L-T; Li, Y-Q; Sun, H-K; Zhank, M-M; Zhang, T; Chen, T; Fan, B-Y; Li, H; Li, Y-Q. Nucleus tractus solitarius mediates hyperalgesia induced by chronic pancreatitis in rats. World J Gastroenterol 2019, 25, 6077–6093. [Google Scholar] [CrossRef]
- Bak, MS; Park, H; Kim, SK. Neural plasticity in the brain during neuropathic pain. Biomedicines 2021, 9, 624. [Google Scholar] [CrossRef] [PubMed]
- Baker, PM; Mathis, V; Lecourtier, L; Simmons, SC; Nugent, FS; Hill, S; Mizumori, SJY. Lateral habenula beyond avoidance: Roles in stress, memory, and decision-making with implications for psychiatric disorders. Front Syst Neurosci 2022, 16, 826475. [Google Scholar] [CrossRef]
- Baliki, M; Al-Amin, HA; Atweh, SF; Jabei, M; Hawwa, N; Jabbur, SJ; Apkarian, AV; Saadé, NE. Attenuation of neuropathic manifestations by local block of the activities of the ventrolateral orbito-frontal area in the rat. Neuroscience 2003, 120, 1093–1104. [Google Scholar] [CrossRef] [PubMed]
- Bandler, R; Shipley, MT. Columnar organization in the midbrain periaqueductal gray: modules for emotional expression? Trends Neurosci 1994, 7, 379–389. [Google Scholar] [CrossRef]
- Bank, PJM; Peper, CE; Marinus, J; Beek, PJ; van Hilten, JJ. Motor consequences of experimentally induced limb pain: a systematic review. Eur J Pain 2013, 17, 145–157. [Google Scholar] [CrossRef]
- Bao, BB; Zhu, HY; Wei, HF; Li, J; Wang, ZB; Li, YH; Hua, XY; Zheng, MX; Zheng, XY. Altered intra- and inter-network brain functional connectivity in upper-limb amputees revealed through independent component analysis. Neural Regen Res 2022, 17, 2725–2729. [Google Scholar] [PubMed]
- Baron, R. Mechanisms of disease: neuropathic pain – a clinical perspective. Nature Clinical Practice Neurol 2006, 2, 95–106. [Google Scholar] [CrossRef]
- Basbaum, AI; Bautista, DM; Scherrer, G; Julius, D. Cellular and molecular mechanisms of pain. Cell 2009, 139, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Baumbauer, KM; Young, EE; Joynes, RL. Pain and learning in a spinal system: contradictory outcomes from common origins. Brain Res Rev 2009, 61, 124–143. [Google Scholar] [CrossRef]
- Benarroch, EE. Pain-autonomic interactions. Neurol Sci 2006, 27, S130–123. [Google Scholar] [CrossRef]
- Bennett, DL; Clark, AJ; Huang, J; Waxman, SG; Dib-Hajj, SD. The role of voltage-gated sodium channels in pain signaling. Physiol Rev 2019, 99, 1079–1151. [Google Scholar] [CrossRef]
- Benke, D. GABAB receptors and pain. Curr Top Behav Neurosci 2022, 52, 213–239. [Google Scholar]
- Berger, JV; Knaepen, L; Janssen, SPM; Jaken, RJP; Marcus, MAE; Joosten, EAJ; Deumens, R. Cellular and molecular insights into neuropathy-induced pain hypersensitivity for mechanism-based treatment approaches. Brain Res Rev 2011, 67, 282–310. [Google Scholar] [CrossRef]
- Bewick, GS. Synaptic-like vesicles and candidate transduction channels in mechanosensory terminals. J Anat 2015, 227, 194–213. [Google Scholar] [CrossRef]
- Bewick, GS; Banks, RW. Mechanotransduction in the muscle spindle. Pflügers Arch - Eur J Physiol 2015, 467, 175–190. [Google Scholar] [CrossRef] [PubMed]
- Bewick, GS; Banks, RW. Mechanotransduction channels in proprioceptive sensory nerve terminals: still an open question? Curr Opin Physiol 2021, 21, 90–104. [Google Scholar] [CrossRef]
- Binshtok, AM; Wang, H; Zimmermann, K; Amaya, F; Vardeh, D; Shi, L; Brenner, GJ; Ji, RR; Bean, BP; Woolf, CJ; Samad, TA. Nociceptors are interleukin-1beta sensors. J Neurosci 2008, 28, 14062–14073. [Google Scholar] [CrossRef]
- Birznieks, I; Burton, AR; Macefield, VG. The effects of experimental muscle and skin pain on the static stretch sensitivity of human muscle spindles in relaxed leg muscles. J Physiol (Lond) 2008, 586, 2713–2723. [Google Scholar] [CrossRef]
- Biurrun Manresa, JA; Brix Finnerup, NS; Johannesen, IL; Biering-Sørensen, F; Staehelin-Jensen, T; Arendt-Nielsen, L; Kæseler-Andersen, O. Central sensitization in spinal cord injured humans assessed by reflex receptive fields. Clin Neurophysiol 2014, 125, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Björnsdotter, M; Löken, L; Olausson, H; Vallbo, A; Wessberg, J. Somatotopic organization of gentle touch processing in the posterior insular cortex. J Neurosci 2009, 29, 9314–9320. [Google Scholar] [CrossRef] [PubMed]
- Bliss, TVP; Collingridge, GL; Kaang, B-K; Zhuo, M. Synaptic plasticity in the anterior cingulate cortex in acute and chronic pain. Nat Rev Neurosci 2016, 17, 485–946. [Google Scholar] [CrossRef]
- Boadas-Vaello, P; Homs, J; Reina, F; Carrera, A; Verdú, E. Neuroplasticity of supraspinal structures associated with pathological pain. Anat Rec 2017, 300, 1481–1501. [Google Scholar] [CrossRef]
- Boccella, S; Marabese, I; Guida, F; Luongo, L; Maione, S; Palazzo, E. The modulation of pain by metabotropic glutamate receptors 7 and 8 in the dorsal striatum. Curr Neuropharmacol 2020, 18, 34–50. [Google Scholar] [CrossRef] [PubMed]
- Bombardi, C; Grandis, A; Chiocchetti, R; Bertolami, R; Johansson, H; Lucchl. Immunohistochemical localization of alpha1a-adrenoreceptors in muscle spindles of rabbit masseter muscle. Tissue and Cell 2006, 38, 121–125. [Google Scholar] [CrossRef]
- Borchers, AT; Gershein, ME. The clinical relevance of complex regional pain syndrome type I: The emperorˈs new clothes. Autoimmun Rev 2017, 16, 22–33. [Google Scholar] [CrossRef]
- Borsook, D. Neurological diseases and pain. Brain 2012, 135, 320–344. [Google Scholar] [PubMed]
- Borsook, D; Youssef, AM; Simons, L; Elman, I; Ecclestone, C. When pain gets stuck: the evolution of pain chronification and treatment resistance. Pain 2018, 159, 2421–2436. [Google Scholar] [CrossRef]
- Boscan, P; Pickering, AE; Paton, JFR. The nucleus of the solitary tract: an integrating station for nociceptive and cardiorespiratory afferents. Exp Physiol 2002, 87, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Bostan, AC; Strick, PL. The basal ganglia and the cerebellum nodes in an integrated network. Nat Rev Neurosci 2018, 19, 338–350. [Google Scholar] [CrossRef] [PubMed]
- Bouchet, CA; Ingram, SL. Cannabinoids in the descending pain modulatory circuit: Role in inflammation. Pharmacol Ther 2020, 209, 107495. [Google Scholar] [CrossRef]
- Bouhassira, D; Bing, Z; Le Bars, D. Studies of the brain structures involved in diffuse noxious inhibitory controls: the mesencephalon. J Neurophysiol 1990, 64, 1712–1723. [Google Scholar] [CrossRef]
- Boyle, KA; Gradwell, MA.; Yasaka, T; Dickie, AC; Polgár, E; Ganley, RP; Orr, DPH; Watanabe, M; Abraira, VE; Kuehn, ED; Zimmerman, AL; Ginty, DD; Callister, RJ; Graham, BA, DI. Defining a spinal microcircuit that gates myelinated afferent Input: Implications for tactile allodynia. Cell Rep 2019, 28, 526–540.e6. [Google Scholar] [CrossRef] [PubMed]
- Bräscher, A-K; Becker, S; Hoeppli, M-E; Schweinhardt, P. Different brain circuitries mediating controllable and uncontrollable pain. J Neurosci 2016, 36, 5013–5025. [Google Scholar] [CrossRef]
- Braz, J; Solorzano, C; Wang, X; Basbaum, AI. Transmitting pain and itch messages: a contemporary view of the spinal cord circuits that generate gate control. Neuron 2014, 82, 522–536. [Google Scholar] [CrossRef]
- Brodal, A. Neurological anatomy. In relation to clinical medicine, 3rd edn; Oxford University Press: New York, 1981. [Google Scholar]
- Brodal, P. A neurobiologist´s attempt to understand persistent pain. Scand J Pain 2017, 15, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Bruehl, S; Bjørkholt Olsen, R; Tronstad, C; Sevre, K; Burns, JW; Schirmer, H; Nielsen, CS; Stubhaug, A; Rosseland, LA. Chronic pain-related changes in cardiovascular regulation and impact on comorbid hypertension in a general population: the Tromsø study. Pain 2018, 159, 119–127. [Google Scholar] [CrossRef]
- Bruehl, S; Chung, OY; Jirjis, JN; Biridepalli, S. Prevalence of clinical hypertension in patients with chronic pain compared to nonpain general medical patients. Clin J Pain 2005, 21, 147–153. [Google Scholar] [CrossRef]
- Bushnell, MC; Čeko, M; Low, LA. Cognitive and emotional control of pain and its disruption in chronic pain. Nat Rev Neurosci 2013, 14, 502–511. [Google Scholar] [CrossRef]
- Butler, SH. Pain “chronification“: what is the problem with this model? Scand J Pain 2022, 23, 419–423. [Google Scholar] [CrossRef]
- Camarena-Delgado, C; Llorca-Torralba, M; Suárez-Pereira, I; Bravo, L; López-Martín, C; Garcia-Partida, J; Mico, JA; Berrosco, E. Nerve injury induces transient locus coeruleus activation over time: role of the locus coeruleus-dorsal reticular nucleus pathway. Pain 2022, 163, 943–954. [Google Scholar] [CrossRef]
- Cao, L; DeLeo, JA. Immune system and pain. In Encyclopedia of neuroscience; Binder, MD, Hirokawa, N, Windhorst, U, Eds.; Springer-Verlag: Berlin Heidelberg, 2009; pp. pp 1919–1922. [Google Scholar]
- Capra, NF; Hisley, CK; Masri, RM. The influence of pain on masseter spindle afferent discharge. Arch Oral Biol 2007, 52, 387–390. [Google Scholar] [CrossRef] [PubMed]
- Carbone, E. Calcium channels – an overview. In Encyclopedia of neuroscience; Binder, MD, Hirokawa, N, Windhorst, U, Eds.; Springer-Verlag: Berlin Heidelberg, 2009; pp. pp 545–550. [Google Scholar]
- Cardoso-Cruz, H; Lima, D; Galhardo, V. Instability of spatial encoding by CA1 hippocampal place cells after peripheral nerve injury. Eur J Neurosci 2011, 33, 2255–2264. [Google Scholar] [CrossRef] [PubMed]
- Carlton, SM. Nociceptive primary afferents: they have a mind of their own. J Physiol (Lond) 2014, 592, 3403–3411. [Google Scholar] [CrossRef]
- Carrasco, C; Naziroğlu, M; Rodríguez, A; Patiente, JA. Neuropathic pain: Delving into the oxidative origin and the possible implication of transient receptor potential channels. Front Physiol 2018, 9, 95. [Google Scholar] [CrossRef]
- Cathenaut, L; Schlichter, R; Hugel, S. Short-term plasticity in the spinal nociceptive system. Pain 2023, 164, 2411–2424. [Google Scholar] [CrossRef] [PubMed]
- Cerveco, F. Hyperalgesia and allodynia. In Encyclopedia of neuroscience; Binder, MD, Hirokawa, N, Windhorst, U, Eds.; Springer-Verlag: Berlin Heidelberg, 2009; pp. pp 1876–1880. [Google Scholar]
- Cevikbas, F; Lerner, EA. Physiology and pathophysiology of itch. Physiol Rev 2020, 100, 945–982. [Google Scholar] [CrossRef]
- Chandler, DJ; Jensen, P; McCall, JG; Pickering, AE; Schwarz, LA; Totah, NK. Redefining NA neuromodulation of behavior: impacts of a modular locus coeruleus architecture. J Neurosci 2019, 39, 8239–8249. [Google Scholar] [CrossRef]
- Chapman, CR; Vierck, CJ. The transition of acute postoperative pain to chronic pain: An integrative overview of research on mechanisms. J Pain 2017, 18, 359.e1–359.e38. [Google Scholar] [CrossRef]
- Chen, J; Gao, Y; Bao, S-T; Wang, Y-D; Jia, T; Yin, C; Xiao, C; Zhou, C. Insula→amygdala and insula→thalamus pathways are involved in comorbid chronic pain and depression-like behavior in mice. J Neurosci 2024, 44, e2062232024. [Google Scholar] [CrossRef]
- Chen, Q; Zhao, M; Dong, J; Yang, K. Chronic restraint stress-induced hyperalgesia is modulated by the periaqueductal gray neurons projecting to the rostral ventromedial medulla in mice. Biochem Biophys Res Commun 2024, 710, 149875. [Google Scholar] [CrossRef]
- Chen, QL; Heinricher, MM. Plasticity in the link between pain-transmitting and pain-modulating systems in acute and persistent inflammation. J Neurosci 2019, 39, 2065–2079. [Google Scholar] [CrossRef] [PubMed]
- Chen, R; Cohen, LG; Halett, M. Nervous system reorganization following injury. Neurosci 2002, 111, 761–773. [Google Scholar] [CrossRef] [PubMed]
- Chen, T; Taniguchi, W; Chen, Q-Y; Tozaki-Saitoh, H; Song, Q; Liu, R-H; Koga, K; Matsuda, T; Kaito-Sugimura, Y; Wang, J; Li, Z-H; Lu, Y-C; Inoue, K; Tsuda, M; Li, Y-Q; Nakatsuka, T; Zhuo, M. Top-down descending facilitation of spinal sensory excitatory transmission from the anterior cingulate cortex. Nature Commun 2018, 9, 1886. [Google Scholar] [CrossRef]
- Chen, X; Tang, S-J. Neural circuitry polarization in the spinal dorsal horn (SDH): A novel form of dysregulated circuitry plasticity during pain pathogenesis. Cells 2024, 13, 398. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L; Duan, B; Huang, T; Zhang, Y; Chen, Y; Britz, O; Garcia-Campmany, L; Ren, X; Vong, L; Lowell, BB; Goulding, M; Wang, Y; Ma, Q. Identification of spinal circuits involved in touch-evoked dynamic mechanical pain. Nat Neurosci 2017, 20, 804–814. [Google Scholar] [CrossRef]
- Cheng, Y-R; Jiang, B-Y; Chen, C-C. Acid-sensing ion channels: dual function proteins for chemo-sensing and mechano-sensing. J Biomed Sci 2018, 25, 46. [Google Scholar] [CrossRef]
- Cheriyan, J; Sheets, PL. Altered excitability and local connectivity of mPFC-PAG neurons in a mouse model of neuropathic pain. J Neurosci 2018, 38, 4829–4839. [Google Scholar] [CrossRef]
- Chiang, C; Aston-Jones, G. Response of locus coeruleus neurons to foot shock stimulation is mediated by neurons in the rostral ventral medulla. Neuroscience 1993, 53, 705–715. [Google Scholar] [CrossRef]
- Chiang, MC; Bowen, A; Schier, LA; Tupone, D; Uddin, O; Heinricher, MM. Parabrachial complex: A hub for pain and aversion. J Neurosci 2019, 39, 8225–8230. [Google Scholar] [CrossRef] [PubMed]
- Chudler, EH; Dong, WK. The role of the basal ganglia in nociception and pain. Pain 1995, 60, 3–38. [Google Scholar] [CrossRef]
- Chung, K; Yoon, YW; Chung, JM. Sprouting sympathetic fibers form synaptic varicosities in the dorsal root ganglion of the rat with neuropathic injury. Brain Res 1997, 751, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Cichon, J; Blanck, TJJ; Gan, WB; Yang, G. Activation of cortical somatostatin interneurons prevents the development of neuropathic pain. Nat Neurosci 2017, 20, 1122–1132. [Google Scholar] [CrossRef]
- Cleland, CL; Hayward, L; Rymer, WZ. Neural mechanisms underlying the clasp-knife reflex in the cat. II. Stretch-sensitive muscular-free nerve endings. J Neurophysiol 1990, 64, 1319–1330. [Google Scholar] [CrossRef] [PubMed]
- Cleland, CL; Rymer, WZ. Functional properties of spinal interneurons activated by muscular free nerve endings and their potential contributions to the clasp-knife reflex. J Neurophysiol 1993, 69, 1181–1191. [Google Scholar] [CrossRef]
- Cobos, A; Lima, D; Almeida, A; Tavares, I. Brain afferents to the lateral caudal ventrolateral medulla: a retrograde and anterograde tracing study in the rat. Neuroscience 2003, 120, 485–498. [Google Scholar] [CrossRef]
- Coderre, TJ; Katz, J. Peripheral and central hyperexcitability: Differential signs and symptoms in persistent pain. Beh Brain Sci 1997, 20, 404–419. [Google Scholar] [CrossRef]
- Coizet, V; Dommett, EJ; Klop, EM; Redgrave, P; Overton, PG. The parabrachial nucleus is a critical link in the transmission of short latency nociceptive information to midbrain dopaminergic neurons. Neuroscience 2010, 168, 263–272. [Google Scholar] [CrossRef]
- Comitato, A; Bardoni, R. Presynaptic inhibition of pain and touch in the spinal cord: From receptors to circuits. Int J Mol Sci 2021, 22, 414. [Google Scholar] [CrossRef]
- Contreras, KM; Caillaud, M; Neddenriep, B; Bagdas, D; Roberts, JL; Ulker, E; White, AB; Aboulhosn, R; Toma, W; Khalefa, T; Adel, A; Mann, JA; Damaj, MI. Deficit in voluntary wheel running in chronic inflammatory and neuropathic pain models in mice: Impact of sex and genotype. Behav Brain Res 2021, 399, 113009. [Google Scholar] [CrossRef]
- Cook, AJ; Woolf, CJ; Wall, PD; MacMahon, SB. Dynamic receptive field plasticity in rat spinal cord dorsal horn following C-primary afferent input. Nature 1987, 325, 151–153. [Google Scholar] [CrossRef] [PubMed]
- Corder, G; Ahanonu, B; Grewe, BF; Wang, D; Schnitzer, MJ; Scherrer, G. An amygdalar neural ensemble that encodes the unpleasantness of pain. Science 2019, 363, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Costa, AR; Tavares, I; Martins, I. How do opioids control pain circuits in the brainstem during opioid-induced disorders and in chronic pain? Implications for the treatment of chronic pain. Pain 2024, 165, 324–336. [Google Scholar] [CrossRef]
- Coste, J; Voisin, DL; Miraucourt, L; Dallel, R; Luccarini, P. Dorsal horn NK1-expressing neurons control windup of downstream trigeminal nociceptive neurons. Pain 2008, 137, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Costigan, M; Scholz, J; Woolf, CJ. Neuropathic pain: a maladaptive response of the nervous system to damage. Annu Rev Neurosci 2009, 32, 1–32. [Google Scholar] [CrossRef]
- Craig, AD. Pain mechanisms: labeled lines versus convergence in central processing. Annu Rev Neurosci 2003, 26, 1–30. [Google Scholar] [CrossRef]
- Cui, C-X; Liu, H-Y; Yue, N; Du, Y-R; Che, L-M; Yu, J-S. Research progress on the mechanism of chronic neuropathic pain. IBRO Neurosci Rep 2023, 14, 80–85. [Google Scholar] [CrossRef]
- Dai, D; Li, W; Chen, A; Gao, X-F; Xiong, L. Lateral habenula and its potential roles in pain and related behaviors. ACS Chem Neurosci 2022, 13, 1108–1118. [Google Scholar] [CrossRef]
- Da Silva, JT; Seminowicz, DA. Neuroimaging of pain in animal models: a review of recent literature. Pain Rep 2019, 4, e732. [Google Scholar] [CrossRef]
- Davidson, S; Golden, JP; Copits, BA; Ray, PR; Vogt, SK; Valtcheva, MV; Schmidt, RE; Ghetti, A; Price, TJ; Gereau, 4th RW. Group II mGluRs suppress hyperexcitability in mouse and human nociceptors. Pain 2016, 157, 2081–2088. [Google Scholar] [CrossRef] [PubMed]
- Decherchi, P; Dousset, E; Grélot, L. Metabolic stability and physiological adaptation of muscle under conditions of exercise. Rev Neurol (Paris) 2004, 160, 297–305. [Google Scholar] [CrossRef]
- Article in French.
- De Felice, C; Herrero, JF; O´Brien, JA; Palmer, JA; Doyle, CA; Smith, AJ; Laird, JM; Belmonte, C; Cervero, F; Hunt, SP. Altered nociception, analgesia and aggression in mice lacking the receptor for substance P. Nature 1998, 392, 394–397. [Google Scholar] [CrossRef] [PubMed]
- De Felice, M; Ossipov, MH. Cortical and subcortical modulation of pain. Pain Manag 2016, 6, 111–120. [Google Scholar] [CrossRef]
- Delbono, O; Wang, Z-M; Messi, ML. Brainstem noradrenergic neurons: Identifying a hub at the intersection of cognition, motility, and skeletal muscle regulation. Acta Physiol (Oxf) 2022, 236, e13887. [Google Scholar] [CrossRef]
- De Ridder, D; Adhia, D; Vanneste, S. The anatomy of pain and suffering in the brain and its clinical implications. Neurosci Biobehav Rev 2021, 130, 125–1463. [Google Scholar] [CrossRef]
- De Ridder, D; De Mulder, G; Verstraeten, E; Sunaert, S; Moller, A. Somatosensory cortex stimulation for deafferentation pain. Acta Neurochir Suppl 2007, 97, 67–74. [Google Scholar]
- Derjean, D; Bertrand, S; Le Masson, G; Landry, M; Morisset, V; Nagy, F. Dynamic balance of metabotropic inputs causes dorsal horn neurons to switch functional states. Nat Neurosci 2003, 6, 274–281. [Google Scholar] [CrossRef] [PubMed]
- DeSantana, JM; Sluka, KA. Central mechanisms in the maintenance of chronic widespread noninflammatory muscle pain. Curr Pain Headache Rep 2008, 12, 338–343. [Google Scholar] [CrossRef]
- Devor, M. Rethinking the causes of pain in herpes zoster and postherpetic neuralgia: the ectopic pacemaker hyposthesis. Pain Rep 2018, 3, e702. [Google Scholar] [CrossRef] [PubMed]
- Dibaj, P; Brockmann, K; Gärtner, J. Dopamine-mediated yawning-fatigue syndrome with specific recurrent initiation and responsiveness to opioids. JAMA Neurol 2020, 77, 254. [Google Scholar] [CrossRef] [PubMed]
- Dibaj, P; Nadrigny, F; Steffens, H; Scheller, A; Hirrlinger, J; Schomburg, ED; Neusch, C; Kirchhoff, F. NO mediates microglial response to acute spinal cord injury under ATP control in vivo. Glia 2010, 58, 1133–44. [Google Scholar] [CrossRef]
- Dibaj, P; Safavi-Abbasi, S; Asadollahi, E. In vivo spectrally unmixed multi-photon imaging of longitudinal axon-glia changes in injured spinal white matter. Neurosci Lett 2024, 841, 137959. [Google Scholar] [CrossRef]
- Dibaj, P; Schomburg, ED. In vivo recording of nerve conduction velocity of spinal CNS fibers in the mouse. Physiol Res 2017, 66, 545–548. [Google Scholar] [CrossRef]
- Dibaj, P; Seeger, D; Gärtner, J; Petzke, F. Follow-up of a case of dopamine-mediated yawning-fatigue-syndrome responsive to opioids, successful desensitization via graded activity treatment. Neurol Int 2021, 13, 79–84. [Google Scholar] [CrossRef]
- Dibaj, P; Windhorst, U. Preprints 2024091515; Muscle fatigue in health and disease. 2024b.
- Dibaj, P; WindhorstU. Motor-control notions in health and disease. Preprints 2024b, 2024031799. [Google Scholar]
- Dib-Hajj, SD; Waxman, SG. Sodium channels in human pain disorders: Genetics and pharmacogenomics. Annu Rev Neurosci 2019, 42, 87–106. [Google Scholar] [CrossRef]
- Doan, HN; Chang, MC. Comprehensive mechanisms and non-invasive treatment for complex regional pain syndrome: a narrative review. J Pain Res 2025, 18, 4699–4726. [Google Scholar] [CrossRef]
- Doan, L; Manders, T; Wang, J. Neuroplasticity underlying the comorbidity of pain and depression. Neural Plast 2015, 2015, 504691. [Google Scholar] [CrossRef]
- Dogrul, BN; Machado Kopruszinski, C; Dolatyari Eslami, M; Watanabe, M; Luo, S; Moreira de Souza, LH; Vizin, RL; Yue, X; Palmiter, RD; Navratilova, E; Porreca, F. Descending facilitation from rostral ventromedial medulla mu opioid receptor-expressing neurons is necessary for maintenance of sensory and affective dimensions of chronic neuropathic pain. Pain 2025, 166, 153–159. [Google Scholar] [CrossRef]
- Dostrovsky, JO. Role of thalamus in pain. Prog Brain Res 2000, 129, 245–257. [Google Scholar]
- Duan-Mu, C-L; Zhang, X-N; Shi, H; Su, Y-S; Wan, H-Y; Wang, Y; Qu, Z-Y; He, W; Wang, X-Y; Jing, X-H. Electroacupuncture-induced muscular inflammatory pain relief was associated with activation of low-threshold mechanoreceptor neurons and inhibition of wide dynamic range neurons in spinal dorsal horn. Front Neurosci 2021, 8, 687173. [Google Scholar] [CrossRef] [PubMed]
- Echeverria-Villalobos, M; Tortorici, V; Brito, BE; Ryskamp, D; Uribe, A; Weaver, T. The role of neuroinflammation in the transition of acute to chronic pain and the opioid-induced hyperalgesia and tolerance. Front Pharmacol 2023, 14, 1297931. [Google Scholar] [CrossRef]
- Endo, T; Spenger, C; Westman, E; Tominaga, T; Olson, L. Reorganization of sensory processing below the level of spinal cord injury as revealed by fMRI. Exp Neurol 2008, 209, 155–160. [Google Scholar] [CrossRef]
- Evrard, HC. The organization of the primate insular cortex. Front Neuroanat 2019, 13, 43. [Google Scholar] [CrossRef] [PubMed]
- Fasick, V; Spengler, RN; Samankan, S; Nader, ND; Ignatowski, TA. The hippocampus and TNF: Common links between chronic pain and depression. Neurosci Biobehav Rev 2015, 53, 139–159. [Google Scholar] [CrossRef] [PubMed]
- Fekete, C; Lechan, RM. Negative feedback regulation of hypophysiotropic thyrotropin-releasing hormone (TRH) synthesizing neurons: role of neuronal afferents and type 2 deiodinase. Front Neuroendocrinol 2007, 28, 97–114. [Google Scholar] [CrossRef]
- Felix, R; Corzo-Lopez, A; Sandoval, A. Voltage-gated ion channels in neuropathic pain signaling. Life (Basel) 2025, 15, 888. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, EC; Pechincha, C; Luz, LL; Kokai, E; Szucs, P; Safronov, BV. Primary afferent-driven presynaptic inhibition of C-fiber inputs to spinal lamina I neurons. Prog Neurobiol 2020, 188, 101786. [Google Scholar] [CrossRef]
- Ferreira, BR; Misery, L. Psychopathology associated with chronic pruritus: A systematic review. Acta Derm Venereol 2023, 103, adv8488. [Google Scholar] [CrossRef]
- Finnerup, NB; Kuner, R; Jensen, TS. Neuropathic pain: from mechanisms to treatment. Physiol Rev 2021, 101, 259–301. [Google Scholar] [CrossRef]
- Flor, H. Cortical reorganisation and chronic pain: implications for rehabilitation. J Rehabil Med 2003, 41 Suppl, 66–72. [Google Scholar] [CrossRef]
- Fore, S; Palumbo, F; Pelgrims, R; Yaksi, E. Information processing in the vertebrate habenula. Semin Cell Dev Biol 2018, 8, 130–139. [Google Scholar] [CrossRef]
- Gackière, F; Vinay, L. Serotonergic modulation of post-synaptic inhibition and locomotor alternating pattern in the spinal cord. Front Neural Circuits 2014, 8, 102. [Google Scholar]
- Gamal-Eltrabily, M; Márquez-Morales, C; Martínez-Lorenzana, G; González-Hernández, A; Condés-Lara, M. Cortical modulation of nociception. Neuroscience 2021, 15(458), 256–270. [Google Scholar] [CrossRef] [PubMed]
- Gamboa-Esteves, FO; Tavares, I; Almeida, A; Batten, TF; McWilliam, PN; Lima, D. Projection sites of superficial and deep spinal dorsal horn cells in the nucleus tractus solitarii of the rat. Brain Res 2001, 921, 195–205. [Google Scholar] [CrossRef]
- Gangadharan, V; Zheng, H; Taberner, FJ; Landry, J; Nees, TA; Pistolec, J; Agarwal, N; Männich, D; Benes, V; Helmstaedter, M; Ommer, B; Lechner, SG; Kuner, T; Kuner, R. Neuropathic pain caused by miswiring and abnormal end organ targeting. Nature 2022, 606, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y; Mei, C; Chen, P; Chen, X. The contribution of neuro-immune crosstalk to pain in the peripheral nervous system and the spinal cord. Int Immunopharmacol 2022, 107, 108700. [Google Scholar] [CrossRef]
- Garcia, AD; Buffalo, EA. Anatomy and function of the primate entorhinal cortex. Annu Rev Vis Sci 2020, 6, 411–432. [Google Scholar] [CrossRef]
- Garcia-Larrea, L; Quesada, C. Cortical stimulation for chronic pain: from anecdote to evidence. Eur J Phys Rehabil Med 2022, 58, 290–305. [Google Scholar] [CrossRef] [PubMed]
- Gazerani, P; Cairns, BE. Activation of rat masticatory muscle afferent fibres by acidic pH. Somatosens Mot Res 2018, 35, 86–94. [Google Scholar] [CrossRef]
- Gebhart, GF. Pain. In Encyclopedia of neuroscience; Binder, MD, Hirokawa, N, Windhorst, U, Eds.; Springer-Verlag: Berlin Heidelberg, 2009; pp. pp 3063–3065. [Google Scholar]
- Giummarra, MJ; Gibson, SJ; Georgiou-Karistianis, N; Bradshaw, JL. Mechanisms underlying embodiment, disembodiment and loss of embodiment. Neurosci Biobehav Rev 2008, 32, 143–160. [Google Scholar] [CrossRef] [PubMed]
- Gong, N; Hagopian, G; Holmes, TC; Luo, D; Xu, X. Functional reorganization of local circuit connectivity in superficial spinal dorsal horn with neuropathic pain states. eNeuro 2019, 6, ENEURO.0272–19. [Google Scholar] [CrossRef] [PubMed]
- González-Hernández, A; Manzano-García, A; Martínez-Lorenzana, G; Tello-García, IA; Carranza, M; Arámburo, C; Condés-Lara, M. Peripheral oxytocin receptors inhibit the nociceptive input signal to spinal dorsal horn wide-dynamic-range neurons. Pain 2017, 158, 2117–2128. [Google Scholar] [CrossRef]
- Goudet, C; Magnaghi, V; Landry, M; Nagy, F; Gereau, RW, 4th; Pin, JP. Metabotropic receptors for glutamate and GABA in pain. Brain Res Rev 2009, 60, 43–56. [Google Scholar] [CrossRef]
- Gouveia, FV; Ibrahim, GM. Habenula as a neural substrate for aggressive Behavior. Front Psychiatry 2022, 13, 817302. [Google Scholar] [CrossRef]
- Grace, PM; Gaudet, AD; Staikopoulos, V; Maier, SF; Hutchinson, MR; Salvemini, D; Watkins, LR. Nitroxidative signaling mechanisms in pathological pain. Trends Neurosci 2016, 39, 862–879. [Google Scholar] [CrossRef]
- Gradwell, MA; Callister, RJ; Graham, BA. Reviewing the case for compromised spinal inhibition in neuropathic pain. J Neural Transm (Vienna) 2020, 127, 481–503. [Google Scholar] [CrossRef] [PubMed]
- Granata, G; Di lorio, R; Ilari, S; Angeloni, BM; Tomasello, F; Cimmino, AT; Carrarini, C; Marrone, A; lodice, F. Phantom limb syndrome: from pathogenesis to treatment. A narrative review. Neurol Sci 2024, 45, 4741–4755. [Google Scholar] [CrossRef]
- Grau, JW. Learning from the spinal cord: How the study of spinal cord plasticity informs our view of learning. Neurobiol Learning Memory 2014, 108, 155–171. [Google Scholar] [CrossRef]
- Graven-Nielsen, T; Arendt-Nielsen, L. Peripheral and central sensitization in musculoskeletal pain disorders: an experimental approach. Curr Rheumatol Rep 2002, 4, 313–321. [Google Scholar] [CrossRef]
- Graven-Nielsen, T; Arendt-Nielsen, L. Impact of clinical and experimental pain on muscle strength and activity. Curr Rheumatol Rep 2008, 10, 475–481. [Google Scholar] [CrossRef]
- Graven-Nielsen, T; Mense, S. The peripheral apparatus of muscle pain: evidence from animal and human studies. Clin J Pain 2001, 17, 2–10. [Google Scholar] [CrossRef]
- Greenwald, JD; Shafritz, KM. An integrative neuroscience framework for the treatment of chronic pain: from cellular alterations to behavior. Front Integr Neurosci 2018, 12, 18. [Google Scholar] [CrossRef]
- Grillner, S. How circuits for habits are formed within the basal ganglia. Proc Natl Acad Sci U S A 2025, 122, e2423068122. [Google Scholar] [CrossRef]
- Grillner, S; Robertson, S; Hellgren Kotaleski, J. Basal ganglia – A motion perspective. Compr Physiol 2020, 10, 1241–1275. [Google Scholar] [CrossRef]
- Groenewegen, HJ. The basal ganglia and motor control. Neural Plast 2003, 10, 107–120. [Google Scholar] [CrossRef]
- Guo, D; Hu, J. Spinal presynaptic inhibition in pain control. Neurosci 2014, 283, 95–106. [Google Scholar] [CrossRef]
- Ha, SO; Kim, JK; Hong, HS; Kim, DS; Cho, HJ. Expression of brain-derived neurotrophic factor in rat dorsal root ganglia, spinal cord and gracile nuclei in experimental models of neuropathic pain. Neuroscience 2001, 107, 301–309. [Google Scholar] [CrossRef]
- Hayoun-Vigouroux, M; Misery, L. Dermatological conditions inducing acute and chronic pain. Acta Derm Venereol 2022, 102, 284. [Google Scholar] [CrossRef] [PubMed]
- Heijmans, L; Mons, MR; Joosten, EA. A systematic review on descending serotonergic projections and modulation of spinal nociception in chronic neuropathic pain and after spinal cord stimulation. Mol Pain 2021, 17, 17448069211043965. [Google Scholar] [CrossRef] [PubMed]
- Heinricher, MM; Tavares, I; Leith, JL; Lumb, BM. Descending control of nociception: Specificity, recruitment and plasticity. Brain Res Rev 2008, 60, 214–225. [Google Scholar] [CrossRef] [PubMed]
- Hellström, F; Roatta, S; Thunberg, J; Passatore, M; Djupsjöbacka, M. Responses of muscle spindles in feline dorsal neck muscles to electrical stimulation of the cervical sympathetic nerve. Exp Brain Res 2005, 165, 328–342. [Google Scholar] [CrossRef]
- Hibi, D; Takamoto, K; Iwama, Y; Ebina, S; Nishimaru, H; Matsumoto, J; Takamura, Y; Yamazaki, M; Nishijo, H. Impaired hemodynamic activity in the right dorsolateral prefrontal cortex is associated with impairment of placebo analgesia and clinical symptoms in postherpetic neuralgia. IBRO Rep 2020, 8, 56–64. [Google Scholar] [CrossRef]
- Hiraga, S-I; Itokazu, T; Nishibe, M; Yamashita, T. Neuroplasticity related to chronic pain and its modulation by microglia. Inflamm Regen 2022, 42, 15. [Google Scholar] [CrossRef]
- Hochman, S; Shreckengost, J; Kimura, H; Quevedo, J. Presynaptic inhibition of primary afferents by depolarization: observations supporting nontraditional mechanisms. Ann NY Acad Sci 2010, 1198, 140–152. [Google Scholar] [CrossRef]
- Hoffmann, S; Beyer, C. A fatal alliance between microglia, inflammasomes, and central pain. Int J Mol Sci 2020, 21, 3764. [Google Scholar] [CrossRef]
- Hoheisel, U; Mense, S; Simons, DG; Yu, XM. Appearance of new receptive fields in rat (DH) neurons following noxious stimulation of skeletal muscle: a model for referral of muscle pain? Neurosci Lett 1993, 153, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Holden, JE; Naleway, E. Microinjection of carbachol in the lateral hypothalamus produces opposing actions on nociception mediated by alpha(1)- and alpha(2)-adrenoceptors. Brain Res 2001, 911, 27–36. [Google Scholar] [CrossRef]
- Holt, MK. The ins and outs of the caudal nucleus of the solitary tract: An overview of cellular populations and anatomical connections. J Neuroendocrinol 2022, 34, e13132. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W-L; Chen, CPc; Nikkhoo, M; Lin, C-F; Ching, CT-S; Niu, C-C; Cheng, C-H. Fatigue changes neck muscle control and deteriorates postural stability during arm movement perturbations in patients with chronic neck pain. Spine J 2020, 20, 530–537. [Google Scholar] [CrossRef]
- Hu, H; Cui, Y; Yang, Y. Circuits and functions of the lateral habenula in health and in disease. Nat Rev Neurosci 2020, 21, 277–295. [Google Scholar] [CrossRef]
- Huang, J; Gadotti, VM; Chen, L; Souza, IA; Huang, S; Wang, D; Ramakrishnan, C; Deisseroth, K; Zhang, Z; Zamponi, GW. A neuronal circuit for activating descending modulation of neuropathic pain. Nat Neurosci 2019, 22, 1659–1668. [Google Scholar] [CrossRef]
- Hucho, T; Levine, JD. Signaling pathways in sensitization: toward a nociceptor cell biology. Neuron 2007, 55, 365–376. [Google Scholar] [CrossRef]
- Hughes, DI; Todd, AJ. Central nervous system targets: Inhibitory interneurons in the spinal cord. Neurotherapeutics 2020, 17, 874–885. [Google Scholar] [CrossRef]
- Hulsebosch, CE; Hains, BC; Crown, ED; Carlton, SM. Mechanisms of chronic central neuropathic pain aftre spinal cord injury. Brain Res Rev 2009, 60, 202–213. [Google Scholar] [CrossRef]
- Huma, Z; Ireland, K; Maxwell, DL. The spino-bulbar-cerebellar pathway: Activation of neurons projecting to the lateral reticular nucleus in the rat in response to noxious mechanical stimuli. Neurosci Lett 2015, 591, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Humphries, MD; Prescott, TJ. The ventral basal ganglia, a selection mechanism at the crossroads of space, strategy, and reward. Prog Neurobiol 2010, 90, 385–417. [Google Scholar] [CrossRef] [PubMed]
- Hunt, SP; Mantyh, PW. The molecular dynamics of pain control. Nat Rev Neurosci 2001, 2, 83–91. [Google Scholar] [CrossRef]
- Inoue, K. Nociceptive signaling of P2X receptors in chronic pain states. Purinergic Signalling 2021, 17, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Inoue, K; Tsuda, M. Microglia in neuropathic pain: cellular and molecular mechanisms and therapeutic potential. Nat Rev Neurosci 2018, 19, 138–152. [Google Scholar] [CrossRef] [PubMed]
- Jänig, W. Neurobiology of visceral pain. Schmerz in German. 2014, 28, 233–251. [Google Scholar] [CrossRef]
- Jaggi, AS; Singh, N. Role of different brain areas in peripheral nerve injury-induced neuropathic pain. Brain Res 2011, 381, 187–201. [Google Scholar] [CrossRef]
- Jankowska, E. Interneuronal relay in spinal pathways from proprioceptors. Prog Neurobiol 1992, 38, 335–378. [Google Scholar] [CrossRef]
- Jarvis, MF. The neural-glial purinergic receptor ensemble in chronic pain states. Trends Neurosci 2010, 33, 48–57. [Google Scholar] [CrossRef]
- Jean, A. The nucleus tractus solitarius: neuroanatomic, neurochemical and functional aspects. Arch Int Physiol Biochim Biophys in French. 1991, 99, A3–52. [Google Scholar]
- Jefferson, T; Kelly, CJ; Martina, M. Differential rearrangement of excitatory inputs to the medial prefrontal cortex in chronic pain models. Front Neural Circuits 2021, 15, 791043. [Google Scholar] [CrossRef]
- Ji, R-R; Donnelly, CR; Nedergaard, M. Astrocytes in chronic pain and itch. 2019, 20, 667–685. [Google Scholar] [CrossRef] [PubMed]
- Jia, T; Wang, Y-D; Chen, J; Zhang, X; Cao, J-L; Xiao, C; Zhou, C. A nigro-subthalamo-parabrachial pathway modulates pain-like behaviors. Nat Commun 2022, 13, 7756. [Google Scholar] [CrossRef]
- Jiang, X; Wang, Y; Wan, R; Feng, B; Zhang, Z; Lin, Y; Wang, Y. The effect of high-definition transcranial direct current stimulation on pain processing in a healthy population: A single-blinded crossover controlled study. Neurosci Lett 2022, 767, 136304. [Google Scholar] [CrossRef]
- Joyce, MKP; Uchendu, S; Arnsten, AFT. Stress and inflammation target dorsolateral prefrontal cortex function: Neural mechanisms underlying weakened cognitive control. Biol Psychiatry 2025, 97, 359–371. [Google Scholar] [CrossRef]
- Julius, D; Basbaum, AI. Molecular mechanisms of nociception. Nature 2001, 413, 203–210. [Google Scholar] [CrossRef]
- Kahlaee, AH; Ghamkhar, L; Arab, AM. The association between neck pain and pulmonary function: A systematic review. Am J Phys Med Rehabil 2017, 96, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Kalezic, I; Bugaychenko, LA; Kostyukov, AI; PilyavskiiAI; Ljubisavljevid, M; Windhorst, U; Johansson, H. Fatigue-related depression of the feline monosynaptic gastrocnemius-soleus reflex. J Physiol 2004, 556, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Kamei, J; Ohsawa, M; Hayashi, S-S; Nakanishi, Y. Effect of chronic pain on morphine-induced respiratory depression in mice. Neuroscience 2011, 174, 224–233. [Google Scholar] [CrossRef]
- Kamibayashi, K; Nakazawa, K; Ogata, H; Obata, H; Akai, M; Shinohara, M. Invariable H-reflex and sustained facilitation of stretch reflex with heightened sympathetic outflow. J Electromyogr Kinesiol 2009, 19, 1053–1060. [Google Scholar] [CrossRef]
- Kanashiro, A; Hiroji Hiroki, C; Morais da Fonseca, D; Birbrair, A; Gomes Ferreira, R; Shimizu Bassi, G; Fonseca, MD; Kusuda, R; Martelossi Cebinelli, GC; Pinho da Silva, K; Wagner Wanderley, C; Menezes, GC; Alves-Fiho, JC; Oliveira, AG; Cunha, TM; Sampaio Pupo, A; Ulloa, L; Queiroz Cunha, F. The role of neutrophils in neuro-immune modulation. Pharmacol Res 2020, 151, 104580. [Google Scholar] [CrossRef]
- Kaushal, R; Taylor, BK; Jamal, AB; Zhang, L; Ma, F; Donahue, R; Westlund, KN. GABA-A receptor activity in the noradrenergic locus coeruleus drives trigeminal neuropathic pain in the rat; contribution of NAα1 receptors in the medial prefrontal cortex. Neuroscience 2016, 334, 148–159. [Google Scholar] [CrossRef]
- Kc, E; Moon, HC; Kim, S; Kim, HK; Won, SY; Hyun, S-H; Park, YS. Optical modulation on the nucleus accumbens core in the alleviation of neuropathica pain in chronic dorsal root ganglion compression rat model. Neuromodulation 2020, 23, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Kilburn-Watt, E; Banati, RB; Keay, KA. Altered thyroid hormones and behavioural change in a sub-population of rats following chronic constriction injury. J Neuroendocrinol 2010, 22, 960–970. [Google Scholar] [CrossRef]
- Kim, W; Angulo, MC. Unraveling the role of oligodendrocytes and myelin in pain. J Neurochem 2025, 169, e16206. [Google Scholar] [CrossRef]
- Kim, J-YV; Tillu, DV; Quinn, TL; Mejia, GL; Shy, A; Asiedu, MNK; Murad, E; Schumann, AP; Totsch, SK; Sorge, RE; Mantyn, PW; Dussor, G; Price, TJ. Spinal dopaminergic projections control the transition to pathological pain plasticity via a D1/D5-mediated mechanism. J Neurosci 2015, 35, 6307–6317. [Google Scholar] [CrossRef]
- Kiritoshi, T; Yakhnitsa, V; Singh, S; Wilson, TD; Chaudhry, S; Neugebauer, B; Torres-Rodriguez, JM; Lin, JL; Carrasquillo, Y; Neugebauer, V. Cells and circuits for amygdala neuroplasticity in the transition to chronic pain. Cell Rep 2024, 43, 114669. [Google Scholar] [CrossRef] [PubMed]
- Kissiwaa, SA; Bagley, EE. Central sensitization of the spino-parabrachial-amygdala pathway that outlasts a brief nociceptive stimulus. J Physiol 2018, 596, 4457–4473. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, L; Petersen, GL; Næsted NØrskov, K; Vase, L; Finnerup, N; Staehelin Jensen, T; Svensson, P. Review of neuroimaging studies related to pain modulation. Scand J Pain 2018, 2, 108–120. [Google Scholar] [CrossRef]
- Koerber, HR; Brown, PB. Quantitative analysis of (DH) cell receptive fields following limited deafferentation. J Neurophysiol 1995, 74, 2065–2076. [Google Scholar] [CrossRef]
- Khoutorsky, A; Price, TJ. Translational control mechanisms in persistemt pain. Trends Neurosci 2018, 41, 100–114. [Google Scholar] [CrossRef]
- Koutsikou, S; Apps, R; Lumb, BM. Top-down control of spinal sensorimotor circuits essential for survival. J Physiol (Lond) 2017, 595, 4151–4158. [Google Scholar] [CrossRef] [PubMed]
- Krause, T; Asseyer, S; Taskin, B; Flöel, A; Witte, AV; Mueller, K; Fiebach, JB; Villringer, K; Villringer, A; Jungehulsing, GJ. The cortical signature of central poststroke pain: Gray matter decreases in somatosensory, insular, and prefrontal Cortices. Cereb Cortex 2016, 26, 80–88. [Google Scholar] [CrossRef]
- Kummer, KK; Mitrić, M; Kalpachidou, T; Kress, M. The medial prefrontal cortex as a central hub for mental comorbidities associated with chronic pain. Int J Mol Sci 2020, 21, 3440. [Google Scholar] [CrossRef]
- Kuner, R; Flor, H. Structural plasticity and reorganisation in chronic pain. Nat Rev Neurosci 2016, 18, 20–30. [Google Scholar] [CrossRef]
- Kuner, R; Kuner, T. Cellular circuits in the brain and their modulation in acute and chronic pain. Physiol Rev 2021, 101, 213–258. [Google Scholar] [CrossRef]
- Kwiat, GC; Basbaum, AI. The origin of brainstem noradrenergic and serotonergic projections to the spinal cord dorsal horn in the rat. Somatosensory & motor research 1992, 9, 157–173. [Google Scholar]
- Labrakakis, C. The role of the insular cortex in pain. Int J Mol Sci 2023, 24, 5736. [Google Scholar] [CrossRef] [PubMed]
- Lançon, K; Séguéla, P. Dysregulated neuromodulation in the anterior cingulate cortex in chronic pain. Front Pharmacol 2023, 14, 128921. [Google Scholar] [CrossRef] [PubMed]
- Laurin, J; Pertici, V; Dousset, E; Marqueste, T; Decherchi, P. Group III and IV muscle afferents: role on central motor drive and clinical implications. Neuroscience 2015, 290, 543–551. [Google Scholar] [CrossRef]
- Lavenex, P; Amaral, DG. Hippocampal-neocortical interaction: a hierarchy of associativity. Hippocampus 2000, 10, 420–430. [Google Scholar] [CrossRef]
- Lee, C-H; Chen, C-C. Role of proprioceptors in chronic musculoskeletal pain. Exp Physiol 2023, 109, 45–54. [Google Scholar] [CrossRef]
- Lee, C-H; Lin, J-H; Lin, S-H; Chang, C-T; Wu, Y-W; Bewick, G; Banks, RW; Gründer, S; Hochgeschwender, U; Chen, C-C. A role for proprioceptors in sngception. Sci Adv 2025, 11, eabc5219. [Google Scholar] [CrossRef]
- Leite-Almeida, H; Valle-Fernandes, A; Almeida, A. Brain projections from the medullary dorsal reticular nucleus: an anterograde and retrograde tracing study in the rat. Neuroscience 2006, 140, 577–595. [Google Scholar] [CrossRef]
- Lesnak, J; Sluka, KA. Chronic non-inflammatory muscle pain: central and peripheral mediators. Curr Opin Physiol 2019, 11, 67–74. [Google Scholar] [CrossRef]
- Levinson, SR. Sodium channels. In Encyclopedia of neuroscience; Binder, MD, Hirokawa, N, Windhorst, U, Eds.; Springer-Verlag: Berlin Heidelberg, 2009; pp. pp 3759–3766. [Google Scholar]
- Lewin, GR; Lu, Y; Park, TJ. A plethora of painful molecules. Curr Opin Neurobiol 2004, 14, 443–449. [Google Scholar] [CrossRef]
- Li, J; Wei, Y; Zhou, J; Zou, H; Ma, L; Liu, C; Xiao, Z; Liu, X; Tan, X; Yu, T; Cao, S. Activation of locus coeruleus-spinal cord NA neurons alleviates neuropathic pain in mice via reducing neuroinflammation from astrocytes and microglia in spinal dorsal horn. J Neuroinflammation 2022, 19, 123. [Google Scholar] [CrossRef] [PubMed]
- Li, M; Yang, G. A mesocortical glutamatergic pathway modulates neuropathic pain independent of dopamine co-release. Nat Commun 2024, 15, 643. [Google Scholar] [CrossRef] [PubMed]
- Li, S; Cao, X; Li, Y; Tang, Y; Cheng, S; Zhang, D. Enhancing ventrolateral prefrontal cortex activation mitigates social pain and modifies subsequent social attitudes: Insights from TMS and fMRI. Neuroimage 2024, 292, 120620. [Google Scholar] [CrossRef]
- Li, T; Zhang, S; Kurata, J. Suppressed descending pain modulatory and enhanced sensorimotor networks in patients with chronic low back pain. J Anesth 2018, 32, 831–843. [Google Scholar] [CrossRef] [PubMed]
- Li, X-H; Miao, H-H; Zhuo, M. NMDA receptor dependent long-term potentiation in chronic pain. Neurochem Res 2019, 44, 531–538. [Google Scholar] [CrossRef]
- Li, Y; Xu, Z; Xie, H; Fu, R; Lo, WLA; Cheng, X; Yang, J; Ge, L; Yu, Q; Wang, C. Changes in cortical activation during upright stance in individuals with chronic low back pain: An fNIRS study. Front Hum Neurosci 2023, 17, 1085831. [Google Scholar] [CrossRef]
- Liang, S-H; Zhao, W-J; Yin, J-B; Chen, Y-B; Li, J-N; Feng, B; Lu, Y-C; Wang, J; Dong, Y-L; Li, Y-Q. A neural circuit from thalamic paraventricular nucleus to central amygdala for the facilitation of neuropathic pain. J Neurosci 2020, 40, 7837–7854. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y; Luo, M; Xu, Q; Zhang, S; Niu, S; Li, X; Sun, W; Song, M; Wang, L; Xing, X; Wang, J; Feng, M; Zhao, L; Chen, H; Sheng, R; Wang, Y. Microglia-neuronal communication mediated by P2X4R-BDNF-TrkB promotes synaptic plasticity and anterior cingulate cortex hyperactivity in muscle pain chronicity. Br J Anaesth 2025, 135, 710–722. [Google Scholar] [CrossRef]
- Lima, D; Almeida, A. The medullary dorsal reticular nucleus as a pronociceptive centre of the pain control system. Prog Neurobiol 2002, 66, 81–108. [Google Scholar] [CrossRef] [PubMed]
- Lin, C-CJ; Chen, W-N; Chen, C-J; Lin, Y-W; Zimmer, A; Chen, C-C. An antinociceptive role for substance P in acid-induced chronic muscle pain. Proc Natl Acad Sci U S A 2012, 109, E76-83. [Google Scholar] [CrossRef]
- Lin, S-H; Cheng, Y-R; Banks, RW; Min, M-Y; Bewick, GS; Chen, C-C. Evidence for the involvement of ASIC3 in sensory mechanotransduction in proprioceptors. Nat Commun 2016, 7, 11460. [Google Scholar] [CrossRef]
- Liu, D; Hu, S-W; Wang, D; Zhang, Q; Zhang, X; Ding, H-L; Cao, J-L. An ascending excitatory circuit from the dorsal raphe for sensory modulation of pain. J Neurosci 2024, 44, e0869232023. [Google Scholar] [CrossRef]
- Liu, M; Li, Y; Zhong, J; Xia, L; Dou, N. The effect of IL-6/Piezo2 on the trigeminal neuropathic pain. Aging (Albany NY) 2021, 13, 13615–13625. [Google Scholar] [CrossRef]
- Liu, JA; Yu, J; Cheung, CW. Immune actions on the peripheral nervous system in pain. Int J Mol Sci 2021, 22, 1448. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, J; Minoshima, S; Casey, KL. Keeping pain out of mind: the role of the dorsolateral prefrontal cortex in pain modulation. Brain 2003, 126, 1079–1091. [Google Scholar] [CrossRef]
- Lu, C; Yang, T; Zhao, H; Zhang, M; Meng, F; Fu, H; Xie, Y; Xu, H. Insular cortex is critical for the perception, modulation, and chronification of pain. Neurosci Bull 2016, 32, 191–201. [Google Scholar] [CrossRef]
- Lund, JP; Sadeghi, S; Athanassiadis, T; Salas, NC; Auclair, F; Thivierge, B; Arsenault, I; Rompré, P; Westberg, K-G; Kolta, A. Assessment of the potential role of muscle spindle mechanoreceptor afferents in chronic muscle pain in the rat masseter muscle. PLoS One 2010, 5, e11131. [Google Scholar] [CrossRef]
- Luo, Z; Liao, X; Luo, L; Fan, Q; Zhang, X; Guo, Y; Wang, F; Ye, Z; Luo, D. Extracellular ATP and cAMP signaling promote Piezo2-dependent mechanical allodynia after trigeminal nerve compression injury. J Neurochem 2022, 160, 376–391. [Google Scholar] [CrossRef] [PubMed]
- Lyubashina, OA; Sivachenko, IB; Mikhalkin, AA. Impaired visceral pain-related functions of the midbrain periaqueductal gray in rats with colitis. Brain Res Bull 2022, 182, 12–25. [Google Scholar] [CrossRef]
- Lyubashina, OA; Sivachenko, IB; Sokolov, AY. Differential responses of neurons in the rat caudal ventrolateral medulla to visceral and somatic noxious stimuli and their alterations in colitis. Brain Res Bull 2019, 152, 299–310. [Google Scholar] [CrossRef]
- Macefield, VG; Sverrisdottir, YB; Wallin, BG. Resting discharge of human muscle spindles is not modulated by increases in sympathetic drive. J Physiol 2003, 551, 1005–1011. [Google Scholar] [CrossRef] [PubMed]
- Madalena, KM; Lerch, JK. The effect of glucocorticoid and glucocorticoid receptor interactions on brain, spinal cord, and glial cell plasticity. Neural Plast 2017, 2017(2017), 8640970. [Google Scholar] [CrossRef]
- Maisky, VA; Pilyavskii, AI; Kalezic, I; Ljubisavljevic, M; Kostyukov, AI; Windhorst, U; Johansson, H. NADPH-diaphorase activity and c-fos expression in medullary neurons after fatiguing stimulation of hindlimb muscles in the rat. Auton Neurosci 2002, 101, 1–12. [Google Scholar] [CrossRef]
- Makovac, E; Venezia, A; Hohenschurz-Schmidt, D; Dipasquale, O; Jackson, JB; Medina, S; O´Daly, O; Williams, SCR; McMahon, SB; Howard, MA. The association between pain-induced autonomic reactivity and descending pain control is mediated by the periaqueductal grey. J Physiol 2021, 599, 5243–5260. [Google Scholar] [CrossRef]
- Maletic, V; Raison, CL. Neurobiology of depression, fibromyalgia and neuropathic pain. Front Biosci (Landmark Ed) 2009, 14, 5291–338. [Google Scholar] [CrossRef] [PubMed]
- Malfliet, A; Coppieters, I; Van Wilgen, P; Kregel, J; De Pauw, R; Dolphens, M; Ickmans, K. Brain changes associated with cognitive and emotional factors in chronic pain: A systematic review. Eur J Pain 2017, 21, 769–786. [Google Scholar] [CrossRef]
- Mansour, AR; Farmer, MA; Baliki, MN; Apkarian, AV. Chronic pain: The role of learning and brain plasticity. Restor Neurol Neurosci 2014, 32, 129–139. [Google Scholar] [CrossRef]
- Martin, WJ; Cao, Y; Basbaum, AI. Characterization of wide dynamic range neurons in the deep dorsal horn of the spinal cord in preprotachykinin-a null mice in vivo. J Neurophysiol 2004, 91, 1945–1954. [Google Scholar] [CrossRef]
- Martins, I; Carvalho, P; de Vries, MG; Teixeira-Pinto, A; Wilson, SP; Westerink, BHC; Tavares, I. Increased noradrenergic neurotransmission to a pain facilitatory area of the brain is implicated in facilitation of chronic pain. Anesthesiology 2015, 123, 642–653. [Google Scholar] [CrossRef]
- Martins, I; Tavares, I. Reticular formation and pain: The past and the future. Front Neuroanat 2017, 11, 51. [Google Scholar] [CrossRef] [PubMed]
- Mathie, A; Veale, EL. Neuronal potassium channels. In Encyclopedia of neuroscience; Binder, MD, Hirokawa, N, Windhorst, U, Eds.; Springer-Verlag: Berlin Heidelberg, 2009; pp. pp 2792–2797. [Google Scholar]
- Mazzitelli, M; Yakhnitsa, V; Neugebauer, B; Neugebauer, V. Optogenetic manipulations of CeA-CRF neurons modulate pain- and anxiety-like behaviors in neuropathic pain and control rats. Neuropharmacology 2022, 210, 109031. [Google Scholar] [CrossRef] [PubMed]
- McCarberg, B; Peppin, J. Pain pathways and nervous system plasticity: Learning and memory in pain. Pain Medv 2019, 20, 2421–2437. [Google Scholar] [CrossRef] [PubMed]
- McCloskey, DI; Prochazka, A. The role of sensory information in the guidance of voluntary movement. Somatosens Mot Res 1994, 11, 21–37. [Google Scholar] [CrossRef]
- McEwen, BS; Nasca, C; Gray, JD. Stress effects on neuronal structure: hippocampus, amygdala, and prefrontal cortex. Neuropsychopharmacology 2016, 41, 3–23. [Google Scholar] [CrossRef]
- McLachlan, EM; Jänig, W.; Devor, M; Michaelis, M. Peripheral nerve injury triggers noradrenergic sprouting within dorsal root ganglia. Nature 1993, 363, 543–546. [Google Scholar] [CrossRef]
- McMahon, SB; La Russa, F; Bennett, DLH. Crosstalk between the nociceptive and immune systems in host defence and disease. Nat Rev Neurosci 2015, 16, 389–402. [Google Scholar] [CrossRef]
- McMahon, SB; Wall, PD. Receptive fields of rat lamina 1 projection cells move to incorporate a nearby region of injury. Pain 1984, 19, 235–247. [Google Scholar] [CrossRef]
- Meeker, TJ; Schmid, A-C; Keaser, ML; Khan, SA; Gullapalli, RP; Dorsey, SG; Greenspan, JD; Seminowicz, DA. Tonic pain alters functional connectivity of the descending pain modulatory network involving amygdala, periaqueductal gray, parabrachial nucleus and anterior cingulate cortex. Neuroimage 2022, 256, 1192783. [Google Scholar] [CrossRef] [PubMed]
- Mense, S. What is different about muscle pain? Schmerz Article in German. 2003, 17, 459–463. [Google Scholar] [CrossRef]
- Mense, S. Mechanisms of transition from acute to chronic muscle pain. Orthopade Article in German. 2004, 33, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Mense, S. Muscle pain: mechanisms and clinical significance. Dtsch Ärztebl Int. 2008, 105, 214–219. [Google Scholar] [CrossRef]
- Merighi, A. Brain-derived neurotrophic factor, nociception, and pain. Biomolecules 2024, 14, 539. [Google Scholar] [CrossRef] [PubMed]
- Metz, M; Grundmann, S; Ständer, S. Pruritus: an overview of current concepts. Vet Dermatol 2011, 22, 121–131. [Google Scholar] [CrossRef]
- Metzger, M; Souza, R; Lima, LB; Bueno, D; Gonçalves, L; Sego, C; Donato, Jr J; Shammah-Lagnado, SL. Habenular connections with the dopaminergic and serotonergic system and their role in stress-related psychiatric disorders. Eur J Neurosci 2021, 53, 65–88. [Google Scholar] [CrossRef]
- Meylakh, N; Crawford, LS; Mills, EP; Macefield, VG; Vickers, ER; Macey, PM; Keay, KA; Henderson, LA. Altered corticobrainstem connectivity during spontaneous fluctuations in pain intensity in painful trigeminal neuropathy. eNeuro 2024, 11, ENEURO.0522–23. [Google Scholar] [CrossRef]
- Michaelis, M; Devor, M; Jänig, W. Sympathetic modulation of activity in rat dorsal root ganglion neurons changes over time following peripheral nerve injury. J Neurophysiol 1996, 76, 753–763. [Google Scholar] [CrossRef]
- Millan, MJ. The induction of pain: an integrative review. Prog Neurobiol 1999, 57, 1–164. [Google Scholar] [CrossRef] [PubMed]
- Mills, EP; Keay, KA; Henderson, LA. Brainstem pain-modulation circuitry and its plasticity in neuropathic pain: Insights from human brain imaging investigations. Front Pain Res (Lausanne) 2021, 2, 705345. [Google Scholar] [CrossRef] [PubMed]
- Mitsi, V; Zachariou, V. Modulation of pain, nociception, and analgesia by the brain reward center. Neuroscience 2016, 338, 81–92. [Google Scholar] [CrossRef] [PubMed]
- Moalem, G; Tracey, DJ. Immune and inflammatory mechanisms in neuropathic pain. Brain Res Rev 2006, 51, 240–264. [Google Scholar] [CrossRef]
- Moon, HC; Park, YS. Optogenetic stimulation of the anterior cingulate cortex modulates the pain processing in neuropathic pain: A review. J Mol Neurosci 2022, 72, 1–8. [Google Scholar] [CrossRef]
- Moore, KA; Baba, H; Woolf, CJ. Synaptic transmission and plasticity in the superficial dorsal horn. Prog Brain Res 2000, 129, 63–80. [Google Scholar]
- Mutso, AA; Radzicki, D; Baliki, MN; Huang, L; Banisadr, G; Centeno, MV; Radulovic, J; Martina, M; Miller, RJ; Apkarian, AV. Abnormalities in hippocampal functioning with persistent pain. J Neurosci 2012, 32, 5747–5756. [Google Scholar] [CrossRef]
- Nadrigny, F; Le Meur, K; Schomburg, ED; Safavi-Abbasi, S; Dibaj, P. Two-photon laser-scanning microscopy for single and repetitive imaging of dorsal and lateral spinal white matter in vivo. Physiol Res 2017, 66, 531–537. [Google Scholar] [CrossRef]
- Nagai, M; Kishi, K; Kato, S. Insular cortex and neuropsychiatric disorders: a review of recent literature. Eur Psychiatry 2007, 22, 387–394. [Google Scholar] [CrossRef]
- Nakamura, SI; Myers, RR. Injury to dorsal root ganglia alters innervation of spinal cord dorsal horn lamina involved in nociception. Spine (Phila Pa 1976) 2000, 25, 537–542. [Google Scholar] [CrossRef]
- Naser, PV; Kuner, R. Molecular, cellular and circuit basis of cholinergic modulation of pain. Neuroscience 2018, 387, 135–148. [Google Scholar] [CrossRef]
- Nelissen, S; Lemmens, E; Geurts, N; Kramer, P; Maurer, M; Hendriks, J; Hendrix, S. The role of mast cells in neuroinflammation. Acta Neuropathol 2013, 125, 637–650. [Google Scholar] [CrossRef]
- Neugebauer, V. Amygdala pain mechanisms. Handb Exp Pharmacol 2015, 227, 261–284. [Google Scholar] [PubMed]
- Neugebauer, V; Galhardo, V; Maione, S; Mackey, SC. Forebrain pain mechanisms. Brain Res Rev 2009, 60, 226–242. [Google Scholar] [CrossRef]
- Neugebauer, V; Li, W; Bird, GC; Han, JS. The amygdala and persistent pain. Neuroscientist 2004, 10, 221–234. [Google Scholar] [CrossRef] [PubMed]
- Neugebauer, V; Mazzitelli, M; Cragg, B; Ji, G; Navratilova, E; Porreca, F. Amygdala, neuropeptides, and chronic pain-related affective behaviors. Neuropharmacology 2020, 170, 108052. [Google Scholar] [CrossRef]
- Neugebauer, V; Presto, P; Yakhnitsa, V; Antenucci, N; Mendoza, B; Ji, G. Pain-related cortico-limbic plasticity and opioid signaling. Neuropharmacology 2023, 231, 109510. [Google Scholar] [CrossRef] [PubMed]
- Neziri, AY; Haesler, S; Petersen-Felix, S; Müller, M; Arendt-Nielsen, L; Biurrun Manresa, J; Andersen, OK; Curatolo, M. Generalized expansion of nociceptive reflex receptive fields in chronic pain patients. Pain 2010, 151, 798–805. [Google Scholar] [CrossRef]
- Nicol, GD; Vasko, MR. Unraveling the story of NGF-mediated sensitization of nociceptive sensory neurons: ON or OFF the Trks? Mol Interv 2007, 7, 26–41. [Google Scholar] [CrossRef]
- Nieuwenhuys, R; Voogd, J; van Huijzen, C. The human central nervous system. A synopsis and atlas; Springer: Berlin Heidelberg New York, 1978. [Google Scholar]
- Oane, I; Barborica, A; Mindruta, IR. Cingulate cortex: anatomy, structural and functional connectivity. J Clin Neurophysiol 2023, 40, 482–490. [Google Scholar] [CrossRef]
- Ogawa, SK; Watabe-Uchida, M. Organization of dopamine and serotonin system: Anatomical and functional mapping of monosynaptic inputs using rabies virus. Pharmacol Biochem Behav 2018, 174, 9–22. [Google Scholar] [CrossRef]
- O´Hearn, E; Molliver, ME. Organization of raphe-cortical projections in rat: a quantitative retrograde study. Brain Res Bull 1984, 3, 709–726. [Google Scholar] [CrossRef] [PubMed]
- Okada, S; Katagiri, A; Saito, H; Lee, J; Ohara, K; Iinuma, T; Iwata, K. Functional involvement of nucleus tractus solitarii neurons projecting to the parabrachial nucleus in trigeminal neuropathic pain. J Oral Sci 2019, 61, 370–378. [Google Scholar] [CrossRef]
- Oliver, KM; Florez-Paz, DM; Badea, TC; Mentis, GZ; Menon, V; de Nooij, JC. Molecular correlates of muscle spindle and Golgi tendon organ afferents. Nat Commun 2021, 12, 1451. [Google Scholar] [CrossRef]
- Ong, WY; Stohler, CS; Herr, DR. Role of the prefrontal cortex in pain processing. Mol Neurobiol 2019, 56, 1137–1166. [Google Scholar] [CrossRef]
- Ossipov, MH; Morimura, K; Porreca, F. Descending pain modulation and chronification of pain. Curr Opin Support Palliat Care 2014, 8, 143–151. [Google Scholar] [CrossRef] [PubMed]
- O´Sullivan, P. Diagnosis and classification of chronic low back pain disorders: maladaptive movement and motor control impairments as underlying mechanism. Man Ther 2005, 10, 242–255. [Google Scholar] [CrossRef] [PubMed]
- Pagliusi, M; Gomes, FV. The role of the rostral ventromedial medulla in stress responses. Brain Sci 2023, 13, 776. [Google Scholar] [CrossRef]
- Palmiter, RD. Parabrachial neurons promote nociplastic pain. Trends Neurosci S0166-2236 2024, 24)00127–9. [Google Scholar] [CrossRef]
- Panneton, WM; Gan, Q; Ariel, M. Injections of algesic solutions into muscle activate the lateral reticular formation: A nociceptive relay of the spinoreticulothalamic tract. PLoS One 2015, 10, e0130939. [Google Scholar] [CrossRef] [PubMed]
- Passatore, M; Roatta, S. Autonomic control of sensory receptors. In Encyclopedia of neuroscience; Binder, MD, Hirokawa, N, Windhorst, U, Eds.; Springer-Verlag: Berlin Heidelberg, 2009; pp. pp 245–250. [Google Scholar]
- Patel, JM; Dao, H. Chronic pruritus: A review of meurophysiology and associated immune neuromodulatory treatments. Skin Therapy Lett 2018, 23, 5–9. [Google Scholar]
- Patel, R; Dickenson, AH. A study of cortical and brainstem mechanisms of diffuse noxious inhibitory controls in anaesthetised normal and neuropathic rats. Eur J Neurosci 2020, 51, 952–962. [Google Scholar] [CrossRef]
- Pautrat, A; Rolland, M; Barthelemy, M; Baunez, C; Sinniger, V; Piallat, B; Savasta, M; Overton, PG; David, O; Coizet, V. Revealing a novel nociceptive network that links the subthalamic nucleus to pain processing. Elife 2018, 7, e36607. [Google Scholar] [CrossRef]
- Peirs, C; Williams, S-PG; Zhao, X; Walsh, CE; Gedeon, JY; Cagle, NE; Goldring, AC; Hioki, H; Liu, Z; Marell, PS; Seal, RP. Dorsal horn circuits for persistent mechanical pain. Neuron 2015, 87, 797–812. [Google Scholar] [CrossRef]
- Peng, B; Jiao, Y; Zhang, Y; Li, S; Chen, S; Xu, S; Gao, P; Fan, Y; Yu, W. Bulbospinal nociceptive ON and OFF cells related neural circuits and transmitters. Front Pharmacol 2023, 14, 1159753. [Google Scholar] [CrossRef]
- Pereira, AR; Alemi, M; Cerqueira-Nunes, M; Monteiro, C; Galhardo, V; Cardoso-Cruz, H. Dynamics of lateral habenula-ventral tegmental area microcircuit on pain-related cognitive dysfunctions. Neurol Int 2023, 15, 1303–1319. [Google Scholar] [CrossRef] [PubMed]
- Pertovaara, A. NA pain modulation. Prog Neurobiol 2006, 80, 53–83. [Google Scholar] [CrossRef]
- Pertovaara, A. The NA pain regulation system: a potential target for pain therapy. Eur J Pharmacol 2013, 716, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Peyron, R; Quesada, C; Fauchon, C. Cingulate-mediated approaches to treating chronic pain. Handb Clin Neurol 2019, 166, 317–326. [Google Scholar]
- Pezet, S; McMahon, SB. Neurotrophins: mediators and modulators of pain. Annu Rev Neurosci 2006, 29, 507–538. [Google Scholar] [CrossRef] [PubMed]
- Pilyavskii, AI; Maznychenko, AV; Maisky, VA; Kostyukov, AI; Hellström, F; Windhorst, U. Capsaicin-induced effects on c-fos expression and NADPH-diaphorase activity in the feline spinal cord. Eur J Pharmacol 2005, 521, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Pinho-Ribeiro, FA; Verri, WA, Jr.; Chiu, IM; Chiu, M. Nociceptor sensory neuron-immune interactions in pain and inflammation. Trends Neuroimmunol 2017, 38, 5–19. [Google Scholar] [CrossRef]
- Pinto, M; Lima, D; Tavares, I. Neuronal activation at the spinal cord and medullary pain control centers after joint stimulation: a c-fos study in acute and chronic articular inflammation. Neuroscience 2007, 147, 1076–1089. [Google Scholar] [CrossRef]
- Pinto-Ribeiro, F; Ansah, OB; Almeida, A; Pertovaara, A. Response properties of nociceptive neurons in the caudal ventrolateral medulla (CVLM) in monoarthritic and healthy control rats: modulation of responses by the paraventricular nucleus of the hypothalamus (PVN). Brain Res Bull 2011, 86, 82–90. [Google Scholar] [CrossRef]
- Poe, GR; Foote, S; Eschenko, O; Johansen, JP; Bouret, S; Aston-Jones, G; Harley, CW; Manahan-Vaughan, D; Weinshenker, D; Valentino, R; Berridge, C; Chandler, DJ; Waterhouse, B; Sara, SJ. Locus coeruleus: a new look at the blue spot. Nat Rev Neurosci 2020, 21, 644–659. [Google Scholar] [CrossRef]
- Porreca, F; Ossipov, MH; Gebhart, GF. Chronic pain and medullary descending facilitation. Trends Neurosci 2002, 25, 319–325. [Google Scholar] [CrossRef]
- Prescott, SA. Synaptic inhibition and disinhibition in the spinal dorsal horn. Prog Mol Biol Transl Sci 2015, 131, 359–383. [Google Scholar] [PubMed]
- Price, TJ; Cervero, F; Gold, MS; Hammond, DL; Prescott, SA. Chloride regulation in the pain pathway. Brain Res Rev 2009, 60, 149–170. [Google Scholar] [CrossRef]
- Price, TJ; Inyang, KE. Commonalities between pain and memory mechanisms and their meaning for understanding chronic pain. Prog Mol Biol Transl Sci 2015, 131, 409–434. [Google Scholar] [PubMed]
- Price, TJ; Ray, PR. Recent advances toward understanding the mysteries of the acute to chronic pain transition. Curr Opin Physiol 2019, 11, 42–50. [Google Scholar] [CrossRef]
- Proske, U; Gandevia, SC. Kinesthetic Senses. Compr Physiol 2018, 8, 1157–1183. [Google Scholar] [CrossRef]
- Quevedo, JN. Presynaptic inhibition. In Encyclopedia of neuroscience; Binder, MD, Hirokawa, N, Windhorst, U, Eds.; Springer-Verlag: Berlin Heidelberg, 2009; pp. pp 3266–3270. [Google Scholar]
- Radovanovic, D; Peikert, K; Lindström, M; Domelöff, FP. Sympathetic innervation of human muscle spindles. J Anat 2015, 226, 542–548. [Google Scholar] [CrossRef]
- Ramer, MS; Bisby, MA. Adrenergic innervation of rat sensory ganglia following proximal or distal painful sciatic neuropathy: distinct mechanisms revealed by anti-NGF treatment. Eur J Neurosci 1999, 11, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Ramer, MS; Thompson, SWN; McMahon, SB. Causes and consequences of sympathetic basket formation in dorsal root ganglia. Pain Suppl 1999, 82, S111–S120. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Jarquín, UN; Lazo-Gómez, R; Tovar-Y-Romo, LB; Tapia, R. Spinal inhibitory circuits and their role in motor neuron degeneration. Neuropharmacology 2014, 82, 101–107. [Google Scholar] [CrossRef]
- Raver, C; Uddin, O; Ji, Y; Li, Y; Cramer, N; Jenne, C; Morales, M; Masri, R; Keller, A. An amygdalo-parabrachial pathway regulates pain perception and chronic pain. J Neurosci 2020, 40, 3424–3442. [Google Scholar] [CrossRef]
- Ren, K; Dubner, R. Pain facilitation and activity-dependent plasticity in pain modulatory circuitry: role of BNDF-TrkB signaling and NMDA receptors. Mol Neurobiol 2007, 35, 224–235. [Google Scholar] [CrossRef] [PubMed]
- Ren, K; Dubner, R. Interactions between the immune and nervous systems in pain. Nat Med 2010, 16, 1267–1276. [Google Scholar] [CrossRef]
- Ren, K; Torres, R. Role of interleukin-1 during pain and inflammation. Brain Res Rev 2009, 60, 57–64. [Google Scholar] [CrossRef]
- Roatta, S; Windhorst, U; Ljubisavljevic, M; Passatore, M. Sympathetic modulation of muscle spindle afferent sensitivity to stretch in rabbit jaw closing muscles. J Physiol 2002, 540, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Roatta, S; Windhorst, U; Djupsjöbacka, M; Lytvynenko, S; Passatore, M. Effects of sympathetic stimulation on the rhythmical jaw movements produced by electrical stimulation of the cortical masticatory areas of rabbits. Exp Brain Res 2005, 162, 14–22. [Google Scholar] [CrossRef]
- Rocha, I; González-García, M; Carillo-Franco, L; Dawid-Milner, MS; López-González, MV. Influence of brainstem’s area A5 on sympathetic outflow and cardiorespiratory dynamics. Biology (Basel) 2024, 13, 161. [Google Scholar] [CrossRef]
- Roeckel, L-A; Le Coz, G-M; Gavériaux-Ruff, C; Simonin, Fréderic. Opioid-induced hyperalgesia: cellular and molecular mechanisms. Neuroscience 2016, 338, 160–182. [Google Scholar] [CrossRef]
- Rogers, M; Tang, L; Madge, DJ; Stevens, EB. The role of sodium channels in neuropathic pain. Sem Cell&Devel Biol 2006, 17, 571–581. [Google Scholar]
- Romero-Grimaldi, C; Berrocoso, E; Alba-Delgado, C; Madrigal, JL; Perez-Nievas, BG; Leza, JC; Mico, JA. Stress increases the negative effects of chronic pain on hippocampal neurogenesis. Anesth Analg 2015, 121, 1078–1088. [Google Scholar] [CrossRef] [PubMed]
- Roza, C; Bernal, L. Electrophysiological characterization of ectopic spontaneous discharge in axotomized and intact fibers upon nerve transection: a role in spontaneous pain? Pflugers Arch 2022, 474, 387–396. [Google Scholar] [CrossRef]
- Rudomin, P. In search of lost presynaptic inhibition. Exp Brain Res 2009, 196, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Rudomin, P; Schmidt, RF. Presynaptic inhibition in the vertebrate spinal cord revisited. Exp Brain Res 1999, 129, 1–37. [Google Scholar] [CrossRef]
- Rygh, LJ; Suzuki, R; Rahman, W; Wong, Y; Vonsy, JL; Sandhu, H; Webber, M; Hunt, S; Dickenson, AH. Local and descending circuits regulate long-term potentiation and zif268 expression in spinal neurons. Eur J Neurosci 2006, 24, 761–772. [Google Scholar] [CrossRef] [PubMed]
- Saab, CY. Pain-related changes in the brain: diagnostic and therapeutic potentials. Trends Neurosci 2012, 35, 629–637. [Google Scholar] [CrossRef]
- Saab, CY; Hains, BC. Remote neuroimmune signaling: a long-range mechanism of nociceptive network plasticity. Trends Neurosci 2009, 32, 110–117. [Google Scholar] [CrossRef]
- Saab, CY; Waxman, SG; Hains, BC. Alarm or curse? The pain of neuroinflammation. Brain Res Rev 2008, 58, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Saadé, NE; Jabbur, SJ. Nociceptive behavior in animal models for peripheral neuropathy: Spinal and supraspinal mechanisms. Prog Neurobiol 2008, 86, 22–47. [Google Scholar] [CrossRef] [PubMed]
- Safavi-Abbasi, S; Venezia, E; Sughrue, M; Dibaj, P. Effectiveness of mindfulness mediationon the quality of life, pain intensity, mobility and physical function in adults with chronic low back pain: a systemic review and meta-analysis. Discov Psychol 2025, 5, 183. [Google Scholar] [CrossRef]
- Salgado, S; Kaplitt, MG. The nucleus accumbens: a comprehensive review. Stereotact Funct Neurosurg 2015, 93, 75–93. [Google Scholar] [CrossRef]
- Salomons, TV; Johnstone, T; Backonja, M-M; Shackman, AJ; Davidson, RJ. Individual differences in the effects of perceived controllability on pain perception: critical role of the prefrontal cortex. J Cogn Neurosci 2007, 19, 993–1003. [Google Scholar] [CrossRef]
- Saman, Y; Arshad, Q; Dutia, M; Rea, P. Stress and the vestibular system. Int Rev Neurobiol 2020, 152, 221–236. [Google Scholar]
- Sanderson, A; Wang, SF; Elgueta-Cancino, E; Martinez-Valdes, E; Sanchis-Sanchez, E; Liew, B; Falla, D. The effect of experimental and clinical musculoskeletal pain on spinal and supraspinal projections to motoneurons and motor unit properties in humans: A systematic review. Eur J Pain 2021, 25, 1668–1701. [Google Scholar] [CrossRef] [PubMed]
- Sandkühler, J. Models and mechanisms of hyperalgesia and allodynia. Physiol Rev 2009, 89, 707–758. [Google Scholar] [CrossRef]
- Sarantopoulos, C; McCallum, B; Sapunar, D; Kwok, W-M; Hogan, Q. ATP-sensitive potassium channels in rat primary afferent neurons: the effect of neuropathic injury and gabapentin. Neurosci Lett 2003, 343, 185–189. [Google Scholar] [CrossRef]
- Sas, D; Gaudel, F; Verdier, D; Kolta, A. Hyperexcitability of muscle spindle afferents in jaw-closing muscles in experimental myalgia: Evidence for large primary afferents involvement in chronic pain. Exp Physiol 2024, 109, 100–111. [Google Scholar] [CrossRef]
- Schmidtko, A. Nitric oxide-mediated pain processing in the spinal cord. Handb Exp Pharmacol 2015, 227, 103–117. [Google Scholar]
- Schmidtko, A; Tegeder, I; Geisslinger, G. No NO, no pain? The role of nitric oxide and cGMP in spinal pain processing. Trends Neurosci 2009, 32, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Scholz, J; Woolf, CJ. Can we conquer pain? Nature Neurosci Suppl 2002, 5, 1062–1067. [Google Scholar] [CrossRef]
- Scholz, J; Woolf, CJ. The neuropathic pain triad: neurons, immune cells and glia. Nature Neurosci 2007, 10, 1361–1368. [Google Scholar] [CrossRef]
- Schomburg, ED; Dibaj, P; Steffens, H. Differentiation between Aδ and C fibre evoked nociceptive reflexes by TTX resistance and opioid sensitivity in the cat. Neurosci Res 2011, 69, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Schomburg, ED; Dibaj, P; Steffens, H. Role of L-DOPA in spinal nociceptive reflex activity: higher sensitivity of Aδ versus C fibre-evoked nociceptive reflexes to L-DOPA. Physiol Res 2011, 60, 701–703. [Google Scholar] [CrossRef] [PubMed]
- Schomburg, ED; Kalezic, I; Dibaj, P; Steffens, H. Reflex transmission to lumbar α-motoneurones in the mouse similar and different to those in the cat. Neurosci Res 2013, 76, 133–140. [Google Scholar] [CrossRef]
- Schomburg, ED; Steffens, H; Dibaj, P; Sears, TA. Major contribution of Aδ-fibres to increased reflex transmission in the feline spinal cord during acute muscle inflammation. Neurosci Res 2012, 72, 155–162. [Google Scholar] [CrossRef]
- Schomburg, ED; Steffens, H; Pilyavskii, AI; Maisky, VA; Brück, W; Dibaj, P; Sears, TA. Long lasting activity of nociceptive muscular afferents facilitates bilateral flexion pattern in the feline spinal cord. Neurosci Res 2015, 95, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Seminowicz, DA; Moayedi, M. The dorsolateral prefrontal cortex in acute and chronic pain. Pain 2017, 18, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Seno, MDJ; Assis, DV; Gouveia, F; Antunes, GF; Kuroki, M; Oliveira, CC; Santos, LCT; Pagano, RL; Martinez, RCR. The critical role of amygdala subnuclei in nociceptive and depressive-like behaviors in peripheral neuropathy. Sci Rep 2018, 8, 13608. [Google Scholar] [CrossRef]
- Serafini, RA; Pryce, KD; Zachariou, V. The mesolimbic dopamine system in chronic pain and associated affective comorbidities. Biol Psychiatry 2020, 87, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Shelton, L; Becerra, L; Borsook, D. Unmasking the mysteries of the habenula in pain and analgesia. Prog Neurobiol 2012, 96, 208–219. [Google Scholar] [CrossRef]
- Shoraka, O; Syed, M; Mandloi, S; Thalheimer, S; Naghizadeh Kashani, S; Heller, JE; Mohamed, FB; Sharan, AD; Talekan, KS; Matias, CM; HArrop, JS; Krisa, L; Alizadeh, M. Periaquaeductal gray connectivity in spinal cord injury-induced neuropathic pain. J Neuroimaging 2024, 34, 704–719. [Google Scholar] [CrossRef]
- Shortland, P; Kinman, E.; Molander, C. Sprouting of A-fibre primary afferents into lamina II in two rat models of neuropathic pain. Eur J Pain 1997, 1, 215–227. [Google Scholar] [CrossRef]
- Sluka, KA; Clauw, DJ. Neurobiology of fibromyalgia and chronic widespread pain. Neuroscience 2016, 338, 114–129. [Google Scholar] [CrossRef]
- Sluka, KA; Danielson, J; Rasmussen, L; DaSilva, LF. Exercise-induced pain requires NMDA receptor activation in the medullary raphe nuclei. Med Sci Sports Exerc 2012, 44, 420–427. [Google Scholar] [CrossRef]
- Sluka, KA; Gregory, NS. The dichotomized role for acid sensing ion channels in musculoskeletal pain and inflammation. Neuropharmacology 2015, 94, 58–63. [Google Scholar] [CrossRef]
- Sluka, KA; Winter, OC; Wemmie, JA. Acid-sensing ion channels: A new target for pain and CNS diseases. Curr Opin Drug Discov Devel 2009, 12, 693–704. [Google Scholar]
- Smith, LJ; Macefield, VG; Birznieks, I; Burton, AR. Effects of tonic muscle pain on fusimotor control of human muscle spindles during isometric ankle dorsiflexion. J Neurophysiol 2019, 121, 1143–1149. [Google Scholar] [CrossRef]
- Solodkin, A; Traub, RJ; Gebhart, GF. Unilateral hindpaw inflammation produces a bilateral increase in NADPH-diaphorase histochemical staining in the rat lumbar spinal cord. Neuroscience 1992, 51, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Sosa, MKS; Boorman, DC; Keay, KA. The impact of sciatic nerve injury and social interactions testing on glucocorticoid receptor expression in catecholaminergic medullary cell populations. Brain Res 2023, 1819, 148542. [Google Scholar] [CrossRef] [PubMed]
- Sotgiu, ML; Valente, M; Storchi, R; Caramenti, G; Biella, GEM. Contribution by DRt descending facilitatory pathways to maintenance of spinal neuron sensitization in rats. Brain Res 2008, 1188, 69–75. [Google Scholar] [CrossRef]
- Spooner, M-F; Robichaud, P; Carrier, JC; Marchand, S. Endogenous pain modulation during the formalin test in estrogen receptor beta knockout mice. Neuroscience 2007, 150, 675–680. [Google Scholar] [CrossRef]
- Staud, R. Abnormal endogenous pain modulation is a shared characteristic of many chronic pain conditions. Exp Rev Neurother 2012, 12, 577–585. [Google Scholar] [CrossRef]
- Stein, C; Clark, JD; Oh, U; Vasko, MR; Wilcox, GL; Overland, AC; Vanderah, TW; Spencer, RH. Peripheral mechanisms of pain and analgesia. Brain Res Rev 2009, 60, 90–113. [Google Scholar] [CrossRef] [PubMed]
- Stucky, CL; Mikesell, AR. Cutaneous pain in disorders affecting peripheral nerves. Neurosci Lett 2021, 765, 136233. [Google Scholar] [CrossRef]
- Suárez-Pereira, I; Llorca-Torralba, M; Bravo, L; Camarena-Delgado, C; Soriano-Mas, C; Berroscoso, E. The role of the locus coeruleus in pain and associated stress-related disorders. Biol Psychiatry 2022, 91, 786–797. [Google Scholar] [CrossRef]
- Sun, H; Li, Z; Qiu, Z; Shen, Y; Guo, Q; Hu, S-W; Ding, H-W; An, S; Cao, J-L. A common neuronal ensemble in nucleus accumbens regulates pain-like behaviour and sleep. Nat Commun 2023, 14, 4700. [Google Scholar] [CrossRef]
- Sun, L; Liu, R; Guo, F; Wen, M-Q; Ma, X-L; Li, K-Y; Sun, H; Xu, C-L; Li, Y-Y; Wu, M-Y; Zhu, Z-G; Li, X-J; Yu, Y-Q; Chen, Z; Li, X-Y; Duan, S. Parabrachial nucleus circuit governs neuropathic pain-like behavior. Nat Commun 2020, 11, 5974. [Google Scholar] [CrossRef]
- Sun, Z-C; Ma, S-B; Chu, W-G; Jia, D; Luo, C. Canonical transient receptor potential (TRPC) channels in nociception and pathological pain. Neural Plast 2020, 3764193. [Google Scholar] [CrossRef] [PubMed]
- Sunavsky, A; Hashmi, MA; Robertson, JW; Veinot, J; Hashmi, JA. The nucleus accumbens-prefrontal connectivity as a predictor of chronic low back pain. Pain 2025. [Google Scholar] [CrossRef]
- Svendsen, F; Tjølsen, A; Hole, K. LTP of spinal A and C-fibre evoked responses after electrical sciatic nerve stimulation. NeuroReport 1997, 8, 3427–3430. [Google Scholar] [CrossRef]
- Szok, D; Tajti, J; Nyári, A; Véscei, L. Therapeutic approaches for peripheral and central neuropathic pain. Behav Neurol 2019, 2019, 8685954. [Google Scholar] [CrossRef]
- Takayanagi, Y; Onaka, T. Roles of oxytocin in stress responses, allostasis and resilience. Int J Mol Sci 2021, 23, 150. [Google Scholar] [CrossRef]
- Talavera, K; Startek, JB; Alvarez-Collazo, J; Boonen, B; Alpizar, YA; Sanchez, A; Naert, R; Nilius, B. Mammalian transient receptor potential TRPA1 channels: From structure to disease. Physiol Rev 2020, 100, 725–803. [Google Scholar] [CrossRef] [PubMed]
- Tam, TH; Salter, MW. Purinergic signalling in spinal pain processing. Purinergic Signal 2021, 17, 49-54, 49-543. [Google Scholar] [CrossRef]
- Tanabe, M; Shimizu, S; Takabayashi, K; Honda, M; Ono, H. Functional alteration of inhibitory influences on spinal motor output in painful diabetic neuropathy in rats. Neurosci Lett 2005, 389, 152–156. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q-Q; Wu, Y; Tao, Q; Shen, Y; An, X; Liu, D; Xu, Z. Direct paraventricular thalamus-basolateral amygdala circuit modulates neuropathic pain and emotional anxiety. Neuropsychopharmacology 2024, 49, 455–466. [Google Scholar] [CrossRef]
- Tang, Y; Wang, M; Zheng, T; Xiao, Y; Wang, S; Han, F; Chen, G. Structural and functional brain abnormalities in postherpetic neuralgia: A systematic review of neuroimaging studies. Brain Res 2021, 1752, 147219. [Google Scholar] [CrossRef]
- Tashima, R; Koga, K; Yoshikawa, Y; Sekine, M; Watanabe, M; Tozaki-Saitoh, H; Furue, H; Yasaka, T; Tsuda, M. A subset of spinal dorsal horn interneurons crucial for gating touch-evoked pain-like behavior. Proc Natl Acad Sci U S A 2021, 118, e2021220118. [Google Scholar] [CrossRef]
- Tavares, I; Costa-Pereira, JT; Martins, I. Monoaminergic and opioidergic modulation of brainstem circuits: New insights into the clinical challenges of pain treatment? Front Pain Res (Lausanne) 2021, 2, 696515. [Google Scholar] [CrossRef] [PubMed]
- Tavares, I; Lima, D. From neuroanatomy to gene therapy: searching for new ways to manipulate the supraspinal endogenous pain modulatory system. J Anat 2007, 211, 261–268. [Google Scholar] [CrossRef]
- Taylor, BK; Westlund, KN. The NA locus coeruleus as a chronic pain generator. J Neurosci Res 2017, 95, 1336–1346. [Google Scholar] [CrossRef] [PubMed]
- Tewari, A; Jog, R; Jog, MS. The striatum and subthalamic nucleus as independent and collaborative structures in motor control. Front Syst Neurosci 2016, 10, 17. [Google Scholar] [CrossRef] [PubMed]
- Thompson, CS; Hiller, CE; Schabrun, SM. Altered spinal-level sensorimotor control related to pain and perceived instability in people with chronic ankle instability. J Sci Med Sport 2019, 22, 425–429. [Google Scholar] [CrossRef]
- Thompson, JM; Neugebauer, V. Cortico-limbic pain mechanisms. Neurosci Lett 2019, 702, 15–23. [Google Scholar] [CrossRef]
- Todd, AJ. Neuronal circuitry for pain processing in the dorsal horn. Nat Rev Neurosci 2010, 11, 823–836. [Google Scholar] [CrossRef]
- Todd, AJ. Plasticity of inhibition in the spinal cord. Handb Exp Pharmacol 2015, 227, 171–190. [Google Scholar]
- Todd, AJ. Identifying functional populations among the interneurons in laminae I-III of the spinal dorsal horn. Mol Pain 2017, 1744806917693003. [Google Scholar] [CrossRef]
- Toney, GM; Mifflin, SW. Sensory modalities conveyed in the hindlimb somatic afferent input to nucleus tractus solitarius. J Appl Physiol (1985) 2000, 88, 2062–2073. [Google Scholar] [CrossRef]
- Topham, L; Gregoire, S; Kang, H; Salmon-Divon, M; Lax, E; Millecamps, M; Szyf, M; Stone, LS. The transition from acute to chronic pain: dynamic epigenetic reprogramming of the mouse prefrontal cortex up to 1 year after nerve injury. Pain 2020, 161, 2394–2409. [Google Scholar] [CrossRef]
- Torres-Rodriguez, JM; Wilson, TD; Singh, S; Chaudhry, S; Adke, AP; Becker, JJ; Lin, JL; Martinez Gonzalez, S; Soler-Cedeño, O; Carrasquillo, Y. The parabrachial to central amygdala circuit is a key mediator of injury-induced pain sensitization. bioRxiv [Preprint] 2023. [Google Scholar]
- Treede, R-D. Gain control mechanisms in the nociceptive system. Pain 2016, 157, 1199–1204. [Google Scholar] [CrossRef] [PubMed]
- Tsantoulas, C; McMahon, SB. Opening paths to novel analgesics: the role of potassium channels in chronic pain. Trends Neurosci 2014, 37, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Tsantoulas, C; Mooney, ER; McNaughton, PA. HCN2 ion channels: basic science opens up possibilities for intervention in neuropathic pain. Biochem J 2016, 473, 2717–1236. [Google Scholar] [CrossRef]
- Tsay, A; Allen, TJ; Proske, U; Giummarra, MJ. Sensing the body in chronic pain: A review of psychophysical studies implicating altered body representation. Neurosci Biobehav Rev 2015, 52, 221–2323. [Google Scholar] [CrossRef]
- Tsuda, M; Koga, K; Chen, T; Zhuo, M. Neuronal and microglial mechanisms for neuropathic pain in the spinal dorsal horn and anterior cingulate cortex. J Neurochem 2017, 141, 486–498. [Google Scholar] [CrossRef]
- Van Oosterwijck, S; Billens, A; Cnockaert, E; Danneels, L; Mertens, T; Dhondt, E; Van Oosterwijck, J. Spinal hyperexcitability in patients with chronic musculoskeletal pain or headache as evidenced by alterations in the nociceptive withdrawal reflex: a systematic review and meta-analysis. Pain 2025, 166, 1002–1029. [Google Scholar] [CrossRef] [PubMed]
- Van Reij, RRI; Joosten, EAJ; van der Hoogen, NJ. Dopaminergic neurotransmission and genetic variation in chronification of post-surgical pain. Br J Anaesth 2019, 123, 853–864. [Google Scholar] [CrossRef]
- Vasic, V; Schmidt, MHH. Resilience and vulnerability to pain and inflammation in the hippocampus. Int J Mol Sci 2017, 18, 739. [Google Scholar] [CrossRef]
- Villanueva, L; Le Bars, D. The activation of bulbo-spinal controls by peripheral nociceptive inputs: diffuse noxious inhibitory controls. Biol Res 1995, 28, 113–125. [Google Scholar]
- Vogt, BA. Midcingulate cortex: Structure, connections, homologies, functions and diseases. J Chem Neuroanat 2016, 74, 28–46. [Google Scholar] [CrossRef]
- Vogt, BA. Cingulate cortex in the three limbic subsystems. Handb Clin Neurol 2019, 166, 39–51. [Google Scholar]
- Vos, BP; Benoist, JM; Gautron, M; Guilbaud, G. Changes in neuronal activities in the two ventral posterior medial thalamic nuclei in an experimental model of trigeminal pain in the rat by constriction of one infraorbital nerve. Somatosens Mot Res 2000, 17, 109–122. [Google Scholar] [PubMed]
- Wall, PD. Recruitment of ineffective synapses after injury. In Functional Recovery in Neurological Disease. Advances in Neurology; Waxman, S.G., Ed.; Raven Press: New York, 1988; Vol. 47, pp. 387–400. [Google Scholar]
- Wan, Y; Zhou, J; Li, H. The role of mechanosensitive piezo channels in chronic pain. J Pain Res 2024, 17, 4199–4212. [Google Scholar] [CrossRef] [PubMed]
- Wang, H; Ehnert, C; Brenner, GJ; Woolf, CJ. Bradykinin and peripheral sensitization. Biol Chem 2006, 387, 11–14. [Google Scholar] [CrossRef]
- Wang, L-L; Wang, H-B; Fu, F-H; Yu, L-C. Role of calcitonin gene-related peptide in pain regulation in the parabrachial nucleus of naive rats and rats with neuropathic pain. Toxicol Appl Pharmacol 2021, 414, 115428. [Google Scholar] [CrossRef]
- Wang, M; Tutt, JO; Dorricott, NO; Parker, KL; Russo, AF; Sowers, LP. Involvement of the cerebellum in migraine. Front Syst Neurosci 2022, 16, 984406. [Google Scholar] [CrossRef]
- Wang, QP; Nakai, Y. The dorsal raphe: an important nucleus in pain modulation. Brain Res Bull 1994, 34, 575–785. [Google Scholar]
- Wang, S-L; Chen, W-Y; Liu, Z-J; Huang, Y-G. Genetic evidence for causal association between hypertension and chronic pain: A Bidirectional Two-Sample Mendelian Randomization Study. Chin Med Sci J 2024, 39, 155–161. [Google Scholar]
- Wang, T; Ma, C. Peripheral nociceptors as immune sensors in the development of pain and itch. Adv Exp Med Biol 2016, 904, 77–85. [Google Scholar] [PubMed]
- Wang, X-Y; Jia, W-B; Xu, X; Chen, R; Wang, L-B; Su, X-J; Xu, P-F; Liu, X-Q; Wen, J; Song, X-Y; Liu, Y-Y; Zhang, Z; Liu, X-F; Zhang, Y. A glutamatergic DRN-VTA pathway modulates neuropathic pain and comorbid anhedonia-like behavior in mice. Nat Commun 2023, 14, 5124. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y-D; Bao, S-T; Gao, Y; Chen, J; Jia, T; Yin, C; Cao, J-L; Xiao, C; Zhou, C. The anterior cingulate cortex controls the hyper-activity in subthalamic neurons in male mice with comorbid chronic pain and depression. PLoS Biol 2024, 22, e3002518. [Google Scholar] [CrossRef] [PubMed]
- Wasner, G; Schattschneider, J; Binder, A; Baron, R. Complex regional pain syndrome – diagnostic, mechanisms, CNS involvement and therapy. Spinal Cord 2003, 41, 61–75. [Google Scholar] [CrossRef]
- Watanabe, M; Narita, M; Hamada, Y; Yamashita, A; Tamura, H; Ikegami, D; Kondo, T; Shinzato, T; Shimizu, T; Fukuchi, Y; Muto, A; Okano, H; Yamanaka, A; Tawfik, VL; Kuzumaki, N; Navratilova, E; Porreca, F; Narita, M. Activation of ventral tegmental area dopaminergic neurons reverses pathological allodynia resulting from nerve injury or bone cancer. Mol Pain 2018, 14, 1744806918756406. [Google Scholar] [CrossRef]
- Watkins, LR; Hutchinson, MR; Milligan, ED; Maier, SF. “Listening” and “talking” to neurons: implications of immune activation for pain control and increasing the efficacy of opioids. Brain Res Rev 2007, 56, 148–169. [Google Scholar] [CrossRef]
- Waxman, SG; Hains, BC. Fire and phantoms after spinal cord injury: Na+ channels and central pain. Trends Neurosci 2006, 29, 207–215. [Google Scholar] [CrossRef]
- Welman, FHSM; Smit, AE; Jongen, JLM; Tibboel, D; van der Geest, JN; Holstege, JC. Pain experience is somatotopically organized and overlaps with pain anticipation in the human cerebellum. Cerebellum 2018, 17, 447–460. [Google Scholar] [CrossRef]
- West, SJ; Bannister, K; Dickenson, AH; Bennett, DL. Circuitry and plasticity of the dorsal horn--toward a better understanding of neuropathic pain. Neuroscience 2015, 300, 254–275. [Google Scholar] [CrossRef]
- White, FA; Bhangoo, SK; Miller, RJ. Chemokines: integrators of pain and inflammation. Nat Rev Drug Discov 2005, 4, 834–844. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, T; DeLong, MR. Deep brain stimulation for movement disorders of basal ganglia origin: Restoring function or functionality? Neurotherapeutics 2016, 13, 264–283. [Google Scholar] [CrossRef]
- Wiech, K; Edwards, R; Moseley, LG; Berna, C; Ploner, M; Tracey, I. Dissociable neural mechanisms underlying the modulation of pain and anxiety? An FMRI pilot study. PLoS One 2014, 9, e110654. [Google Scholar] [CrossRef]
- Wiesenfeld-Hallin, Z; Aldskogius, H; Grant, G; Hao, J-X; Hoekfelt, T; Xu, X-J. Central inhibitory dysfunctions: mechanisms and clinical implications. Beh Brain Sci 1997, 20, 420–425. [Google Scholar] [CrossRef]
- Wilkinson, KA. Molecular determinants of mechanosensation in the muscle spindle. Curr Opin Neurobiol 2022, 74, 102542. [Google Scholar] [CrossRef] [PubMed]
- Willis, WD. Temperature perception and pain. In Comprehensive human physiology. From cellular mechanisms to integration; Greger, R, Windhorst, U, Eds.; Springer-Verlag: Berlin Heidelberg New York, 1996; vol 1, pp. pp 677–696. [Google Scholar]
- Willis, WD. Dorsal root potentials and dorsal root reflexes: a double-edged sword. Exp Brain Res 1999, 124, 395–421. [Google Scholar] [CrossRef] [PubMed]
- Willis, WD. Long-term potentiation in spinothalamic neurons. Brain Res Rev 2002, 40, 202–21. [Google Scholar] [CrossRef]
- Windhorst, U. Muscle proprioceptive feedback and spinal networks. Brain Res Bull 2007, 73, 155–202. [Google Scholar] [CrossRef]
- Windhorst, U. Spinal cord circuits: models and reality. Neurophysiology 2021, 53, 142–222. [Google Scholar] [CrossRef]
- Windhorst, U; Dibaj, P. Plastic spinal motor circuits in health and disease. J Integr Neurosci 2023, 22, 167. [Google Scholar] [CrossRef]
- Windhorst, U; Dibaj, P. Nociception and acute pain: ascending functional structure and descending modulation. Preprints 2025a, 2025050743. [Google Scholar]
- Windhorst, U; Dibaj, P. Nociception and acute pain: Neurotransmitters and neuromodulators. Preprints 2025b, 2025080487. [Google Scholar]
- Windhorst, U; Meyer-Lohmann, J; Kirmayer, D; Zochodne, D. Renshaw cell responses to intra-arterial injection of muscle metabolites into cat calf muscles. Neurosci Res 1997, 27, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Witter, MP; Doan, TP; Jacobsen, B; Nilssen, ES; Ohara, S. Architecture of the entorhinal cortex. A review of entorhinal anatomy in rodents with some comparative notes. Front Syst Neurosci 2017, 11, 46. [Google Scholar] [CrossRef]
- Woo, S-H; Lukacs, V; de Nooij, JC; Zaytseva, D; Criddle, CR; Francisco, A; Jessell, TM; Wilkinson, KA; Patapoutian, A. Piezo2 is the principal mechanotransduction channel for proprioception. Nat Neurosci 2015, 18, 1756–1762. [Google Scholar] [CrossRef]
- Woodbury, CJ; Kullmann, FA; McIlwrath, SL; Koerber, HR. Identity of myelinated cutaneous sensory neurons projecting to nocireceptive laminae following nerve injury in adult mice. J Comp Neurol 2008, 508, 500–509. [Google Scholar] [CrossRef] [PubMed]
- Woolf, CJ. Central sensitization: implications for the diagnosis and treatment of pain. Pain 2011, 152, S2–S15. [Google Scholar] [CrossRef]
- Woolf, CJ; Salter, MW. Neuronal plasticity: increasing the gain in pain. Science 2000, 288, 1765–1768. [Google Scholar] [CrossRef]
- Xiao, X; Zhang, Y-Q. A new perspective on the anterior cingulate cortex and affective pain. Neurosci Biobehav Rev 2018, 90, 200–211. [Google Scholar] [CrossRef]
- Xie, W; Strong, JA; Zhang, J-M. Increased excitability and spontaneous activity of rat sensory neurons following in vitro stimulation of sympathetic fiber sprouts in the isolated dorsal root ganglion. Pain 2010, 151, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y; Lin, Y; Yu, M; Zhou, K. The nucleus accumbens in reward and aversion processing: insights and implications. Front Behav Neurosci 2024, 18, 1420028. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y; Zhu, X; Chen, Y; Chen, Y; Zhu, Y; Xiao, S; Wu, M; Wang, Y; Zhang, C; Wu, Z; He, X; Liu, B; Shen, Z; drn, X; Fang, J. Electroacupuncture alleviates mechanical allodynia and anxiety-like behaviors induced by chronic neuropathic pain via regulating rostral anterior cingulate cortex-dorsal raphe nucleus neural circuit. CNS Neurosci Ther 2023, 29, 4043–4058. [Google Scholar] [CrossRef]
- Yalcin, I; Barthas, F; Barrot, M. Emotional consequences of neuropathic pain: insight from preclinical studies. Neurosci Biobehav Rev 2014, 47, 154–164. [Google Scholar] [CrossRef]
- Yamauchi, N; Sato, K; Sato, K; Murakawa, S; Hamasaki, Y; Nomura, H; Amano, T; Minami, M. Chronic pain-induced neuronal plasticity in the bed nucleus of the stria terminalis causes maladaptive anxiety. Sci Adv 2022, 8, eabj5586. [Google Scholar] [CrossRef]
- Yang, S; Chang, MC. Chronic pain: Structural and functional changes in brain structures and associated negative affective states. Int J Mol Sci 2019, 20, 3130. [Google Scholar] [CrossRef]
- Yang, X; Xu, W; Hou, X; Ding, Z; Zhang, C; Guo, Q; Zou, W; Wang, J. Chronic stress enhances neuropathic pain through activation of the anterior cingulate cortex-spinal dorsal horn pathway in rats. Prog Neuropsychopharmacol Biol Psychiatry 2025, 138, 111352. [Google Scholar] [CrossRef]
- Yarmolinsky, DA; Zeng, X; MacKinnon-Booth, N; Greene, CA; Kim, C; Cheng, Y-T; Lenders Turnes, B; Woolf, CJ. Differential modification of ascending spinal outputs in acute and chronic pain states. Neuron 2025, 113, 1223–1239.e5. [Google Scholar] [CrossRef] [PubMed]
- Yetnikoff, L; Laezzi, HN; Reichard, RA; Zahm, DS. An update on the connections of the ventral mesencephalic dopaminergic complex. Neuroscience 2014, 282, 23–48. [Google Scholar] [CrossRef] [PubMed]
- Yin, C; Jia, T; Luan, Y; Zhang, X; Xiao, C; Zhou, C. A nigra-subthalamic circuit is involved in acute and chronic pain states. Pain 2022, 163, 1952–1966. [Google Scholar] [CrossRef] [PubMed]
- Yin, J-B; Liang, S-H; Li, F; Zhao, W-J; Bai, Y; Sun, Y; Wu, Z-Y; Ding, T; Sun, Y; Liu, H-X; Lu, Y-C; Zhang, T; Huang, J; Chen, T; Li, H; Chen, Z-F; Cao, J; Ren, R; Peng, Y-N; Yang, J; Zang, W-D; Li, X; Dong, Y-L; Li, Y-Q. dmPFC-vlPAG projection neurons contribute to pain threshold maintenance and antianxiety behaviors. J Clin Invest 2020, 130, 6555–6570. [Google Scholar] [CrossRef]
- You, H-J; Lei, J; Pertovaara, A. Thalamus: The 'promoter' of endogenous modulation of pain and potential therapeutic target in pathological pain. Neurosci Biobehav Rev 2022, 139, 104745. [Google Scholar] [CrossRef]
- Yu, R; Gollub, RL; Spaeth, R; Napadow, V; Wasan, A; Kong, J. Disrupted functional connectivity of the periaqueductal gray in chronic low back pain. Neuroimage Clin 2014, 23(6), 100–108. [Google Scholar] [CrossRef]
- Zeilhofer, HU; Werynska, K; Gingras, J; Yévenes, GE. Glycine receptors in spinal nociceptive control – An update. Biomolecules 2021, 11, 846. [Google Scholar] [CrossRef] [PubMed]
- Zeilhofer, HU; Wildner, H; Yévenes, GE. Fast synaptic inhibition in spinal sensory processing and pain control. Physiol Rev 2012, 92, 193–235. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H; Li, L; Zhang, X; Ru, G; Zang, W. Role of the dorsal raphe nucleus in pain processing. Brain Sci 2024, 14, 982. [Google Scholar] [CrossRef]
- Zhang, Y; Chen, Y; Liedtke, W; Wang, F. Lack of evidence for ectopic sprouting of genetically labeled Aβ touch afferents in inflammatory and neuropathic trigeminal pain. Mol Pain 2015, 11, 18. [Google Scholar] [CrossRef]
- Zhou, H; Martinez, E; Lin, HH; Yang, R; Dale, JA; Liu, K; Huang, D; Wang, J. Inhibition of the prefrontal projection to the nucleus accumbens enhances pain sensitivity and affect. Front Cell Neurosci 2018, 12, 240. [Google Scholar] [CrossRef]
- Zhuo, M. Descending facilitation: From basic science to the treatment of chronic pain. Mol Pain 2017, 13, 1–12. [Google Scholar] [CrossRef]
- Zhou, XF; Deng, YS; Chie, E; Xue, Q; Zhong, JH; McLachlan, EM; Rush, RA; Xian, CJ. Satellite-cell-derived nerve growth factor and neurotrophin-3 are involved in noradrenergic sprouting in the dorsal root ganglia following peripheral nerve injury in the rat. Eur J Neurosci 1999, 11, 1711–1722. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, M. Long-term plasticity of NMDA GluN2B (NR2B) receptor in anterior cingulate cortical synapses. Mol Pain 2024, 20, 17448069241230258. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X; Xu, Y; Shen, Z; Zhang, H; Xiao, S; Zhu, Y; Wu, M; Chen, Y; Wu, Z; Xu, Y; He, X; Liu, B; Liu, J; Liu, Y; Du, J; Sun, J; Fang, J; Shao, X. Rostral anterior cingulate cortex-ventrolateral periaqueductal gray circuit underlies electroacupuncture to alleviate hyperalgesia but not anxiety-like behaviors in mice with spared nerve injury. Front Neurosci 2022, 15, 757628. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, AL; Kovatsis, EM; Pozsgai, RY; Tasnim, A; Zhang, Q; Ginty, DD. Distinct modes of presynaptic inhibition of cutaneous afferents and their functions in behavior. Neuron 2019, 102, 420–434.e8. [Google Scholar] [CrossRef]
- Zimmermann, M. Pathobiology of neuropathic pain. Eur J Pharmacol 2001, 429, 23–37. [Google Scholar] [CrossRef]
- Zouikr, I; Bartholomeusz, MD; Hodgson, DM. Early life programming of pain: focus on neuroimmune to endocrine communication. J Transl Med 2016, 14, 123. [Google Scholar] [CrossRef]
- Zyuzin, J; Jendzjowsky, N. Neuroanatomical and neurophysiological evidence of pulmonary nociceptor and carotid chemoreceptor convergence in the nucleus tractus solitarius and nucleus ambiguus. J Neurophysiol 2022, 127, 1511–1518. [Google Scholar] [CrossRef]
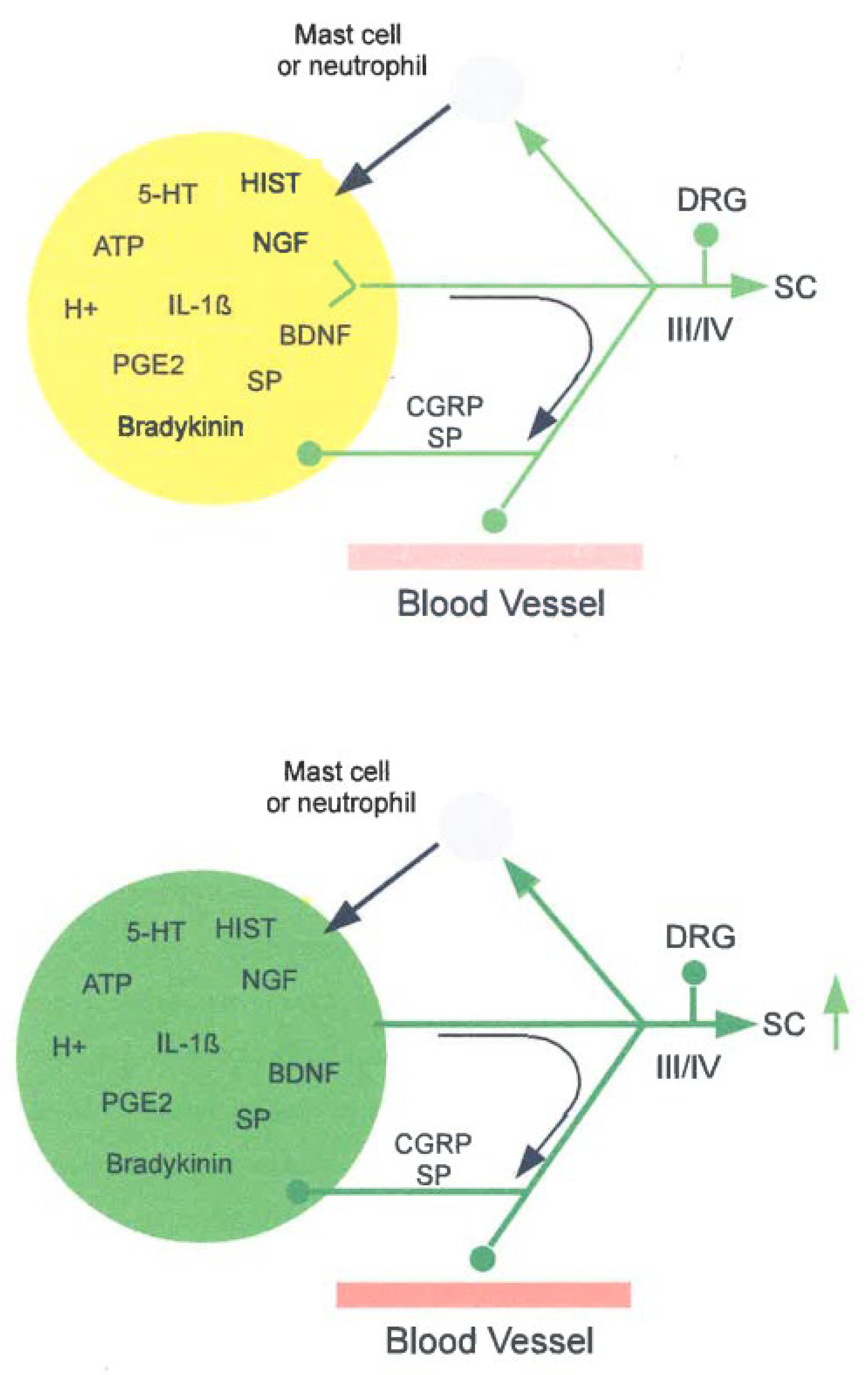
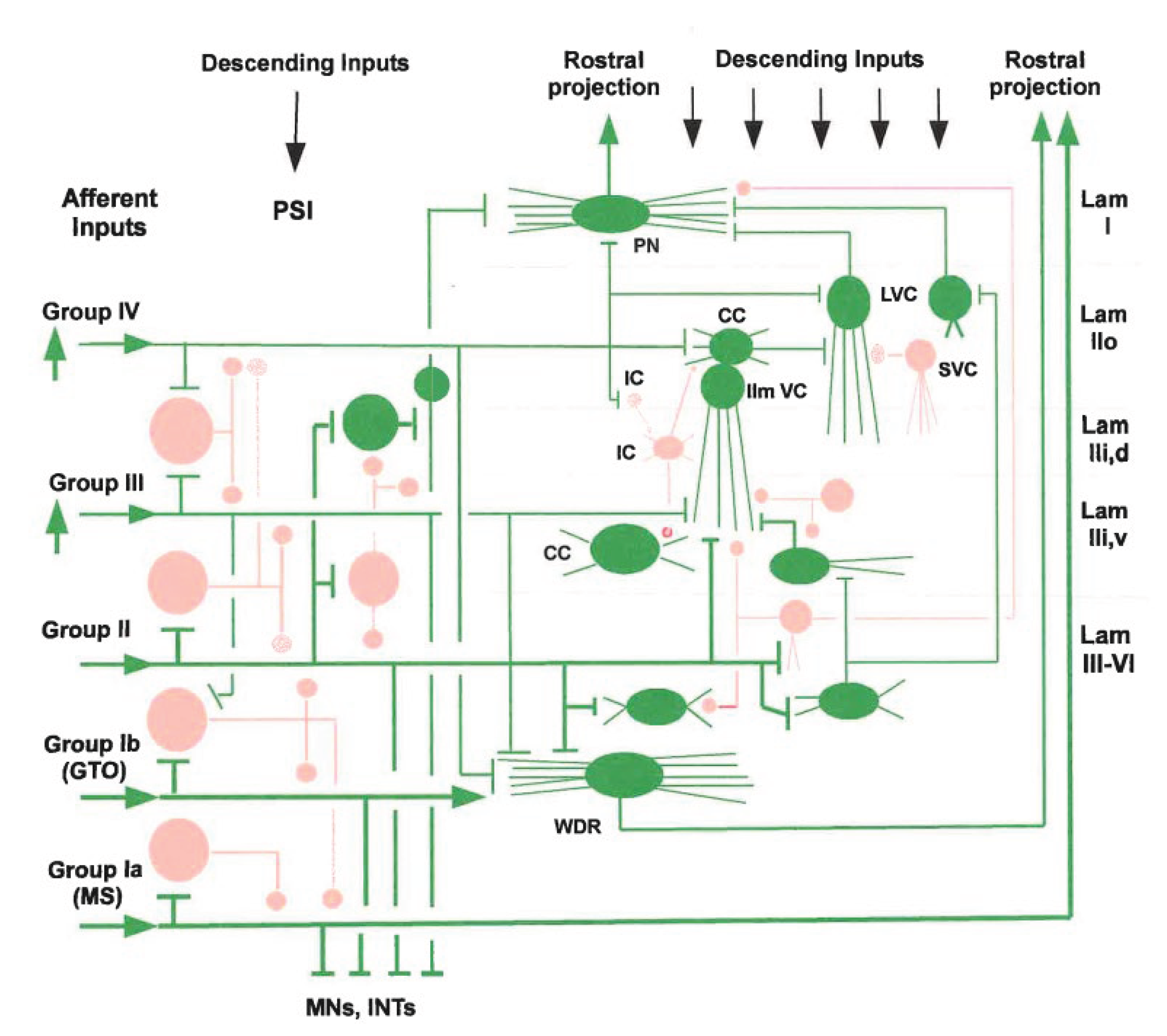
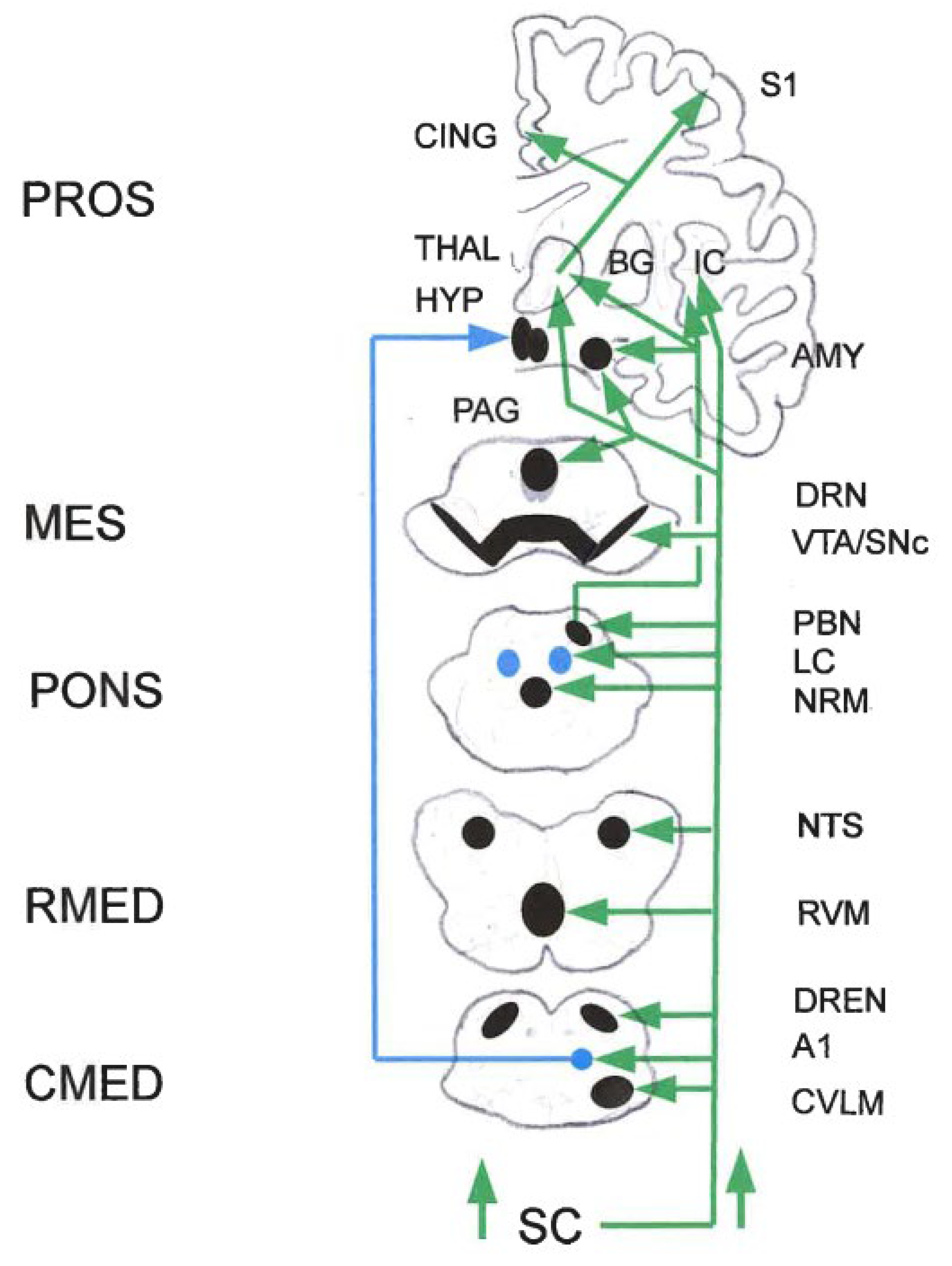
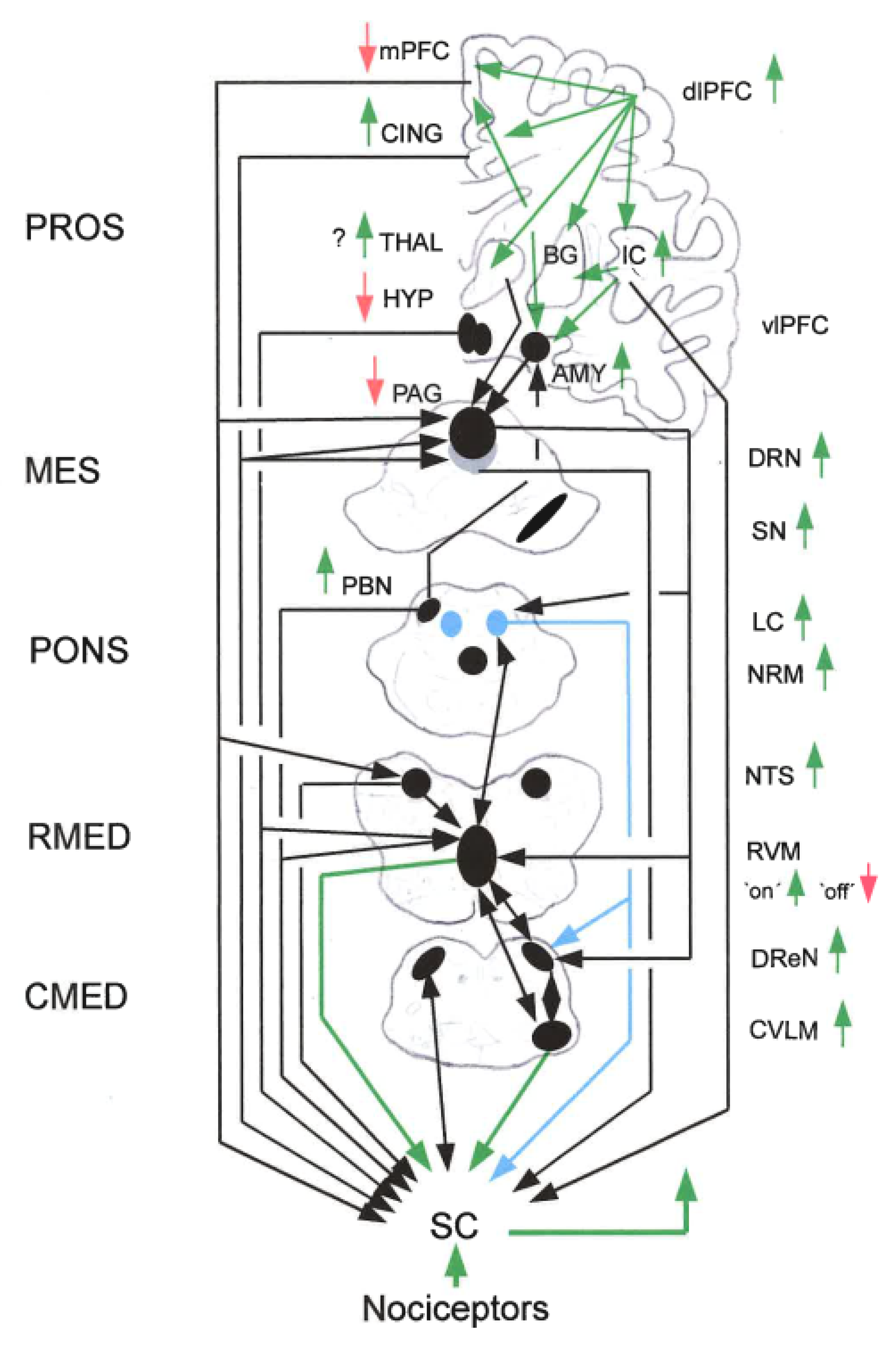
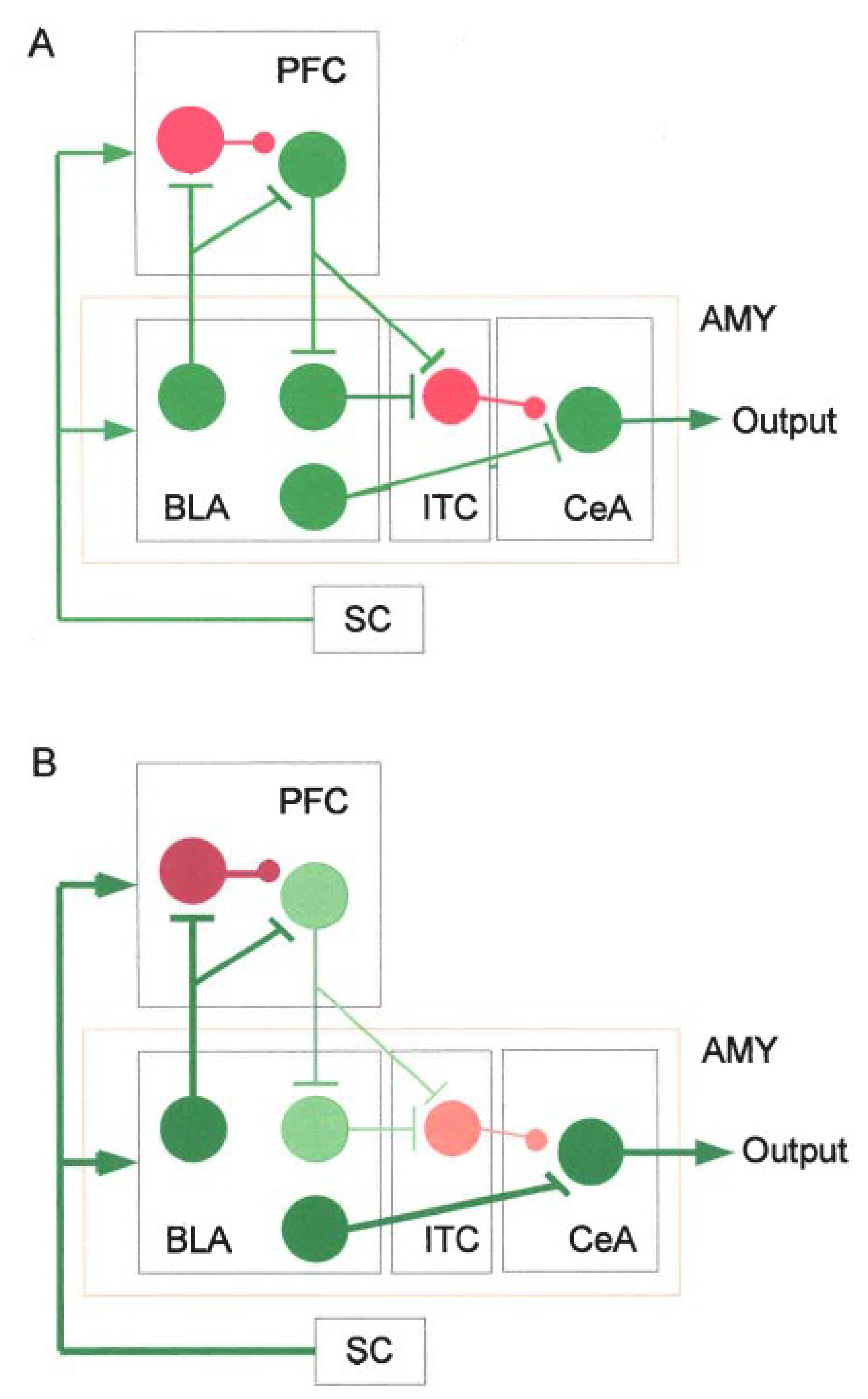
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



