Submitted:
02 February 2026
Posted:
02 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
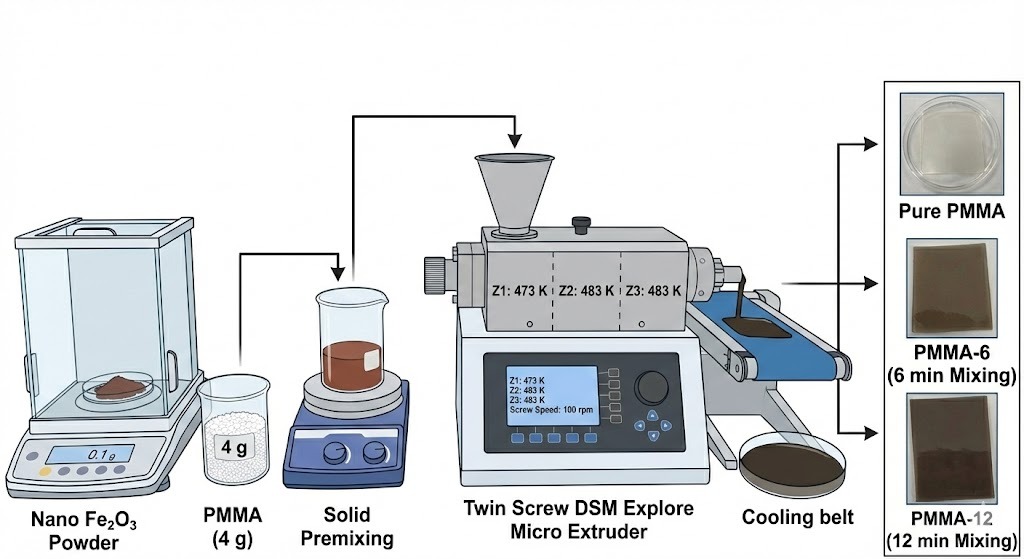
2. Materials and Methods
3. Results And Discussions
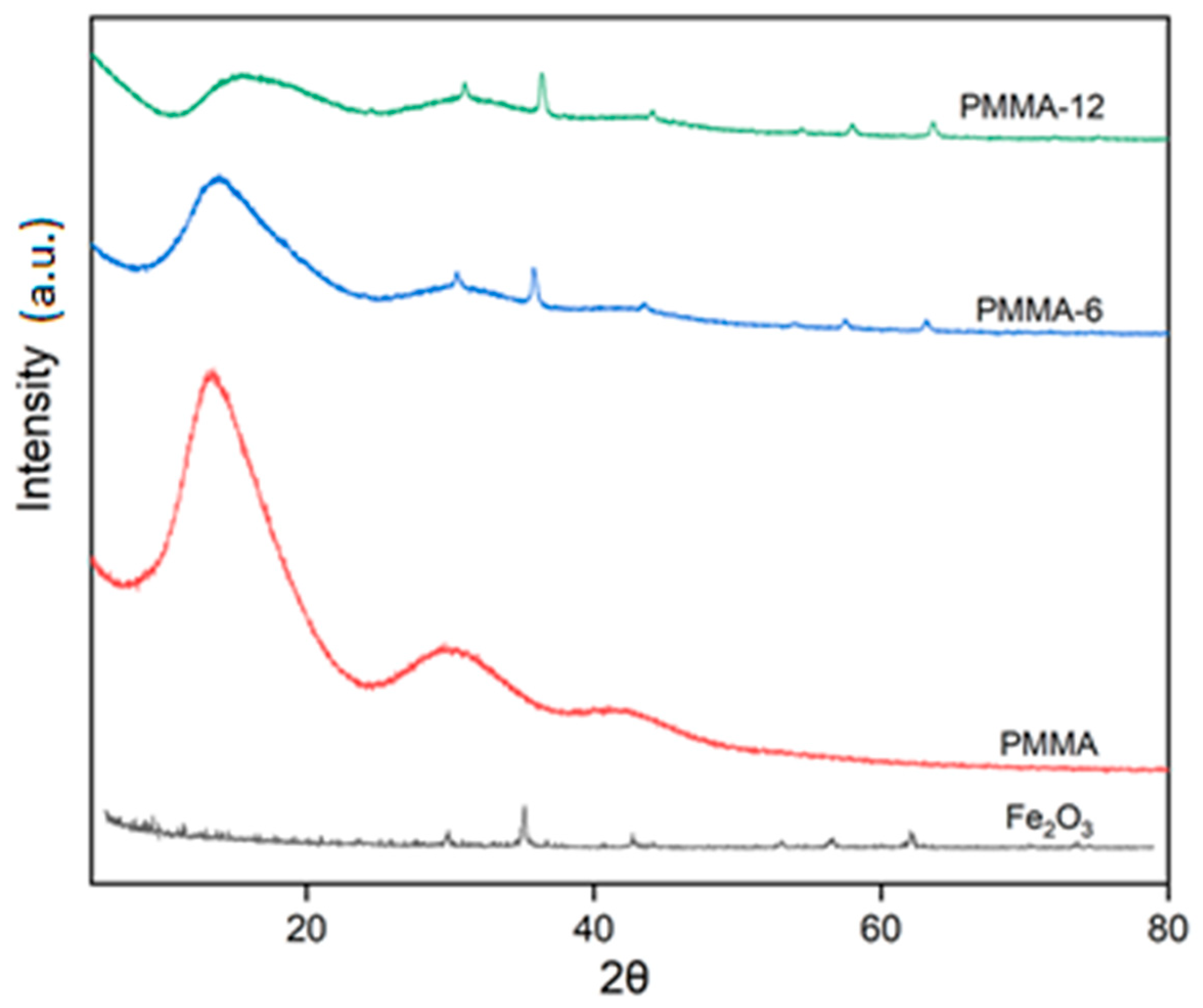
3.1. XRD Characterization
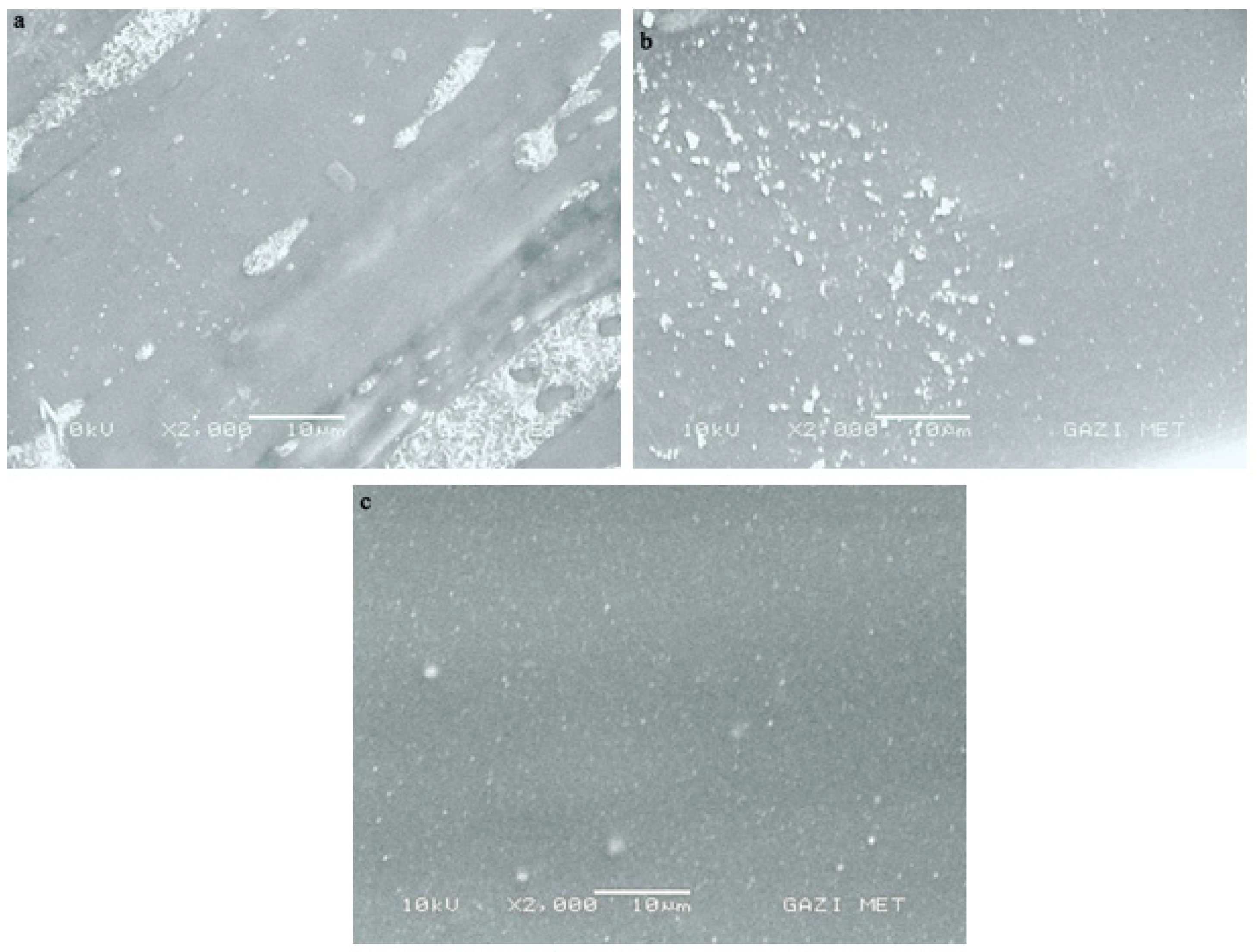
3.2. Morphology Characterization
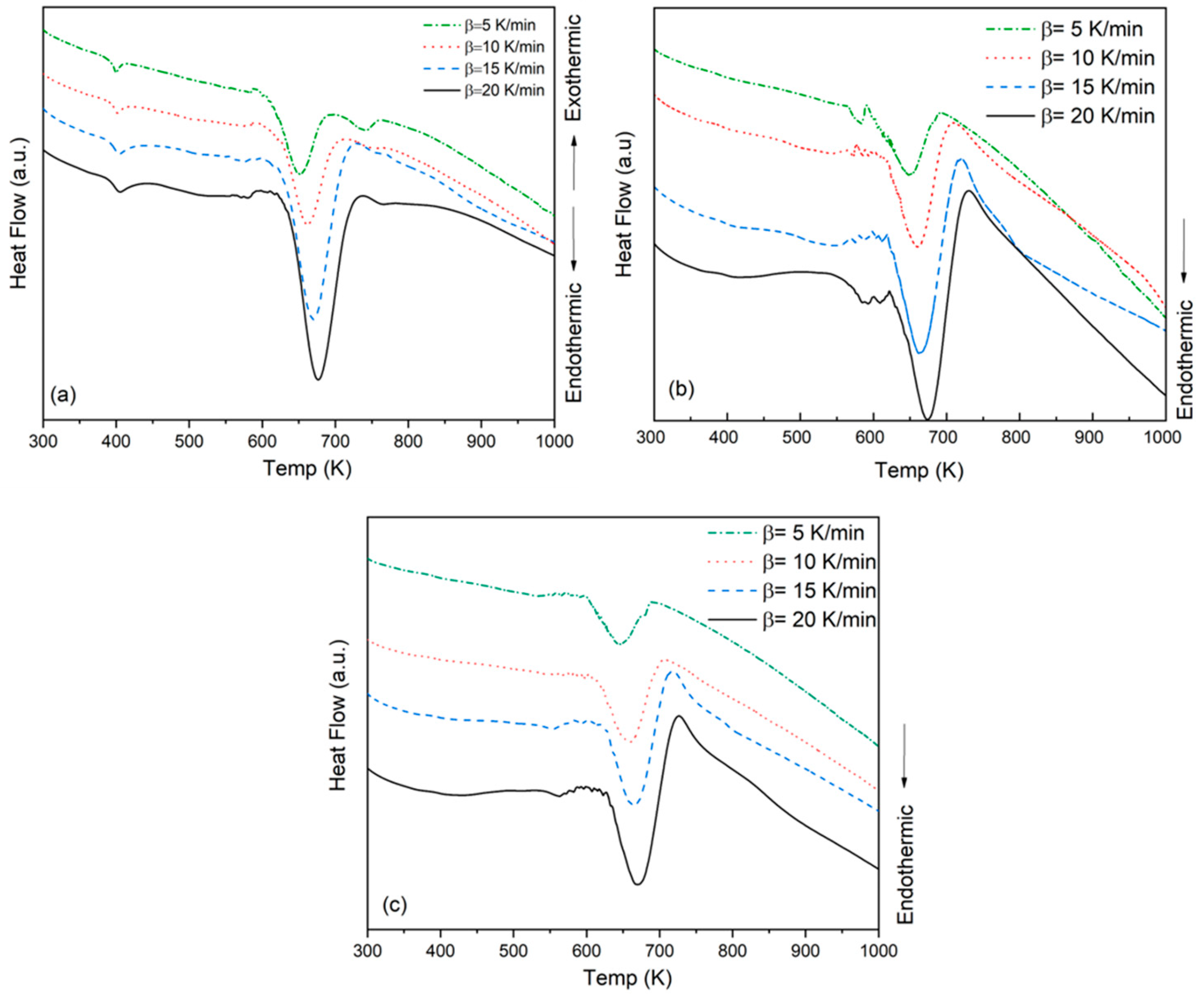
3.3. Differential Thermal Analysis
3.4. Thermal Activation Energies
3.5. Thermogravimetric Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Symbol | Meaning |
| APTS | (3-Aminopropyl) triethoxysilane |
| DTA | Differential thermal analysis |
| JCPDS | Joint Committee on Powder Diffraction Standards Card No |
| K | The amount of substance remaining in the K region |
| L | The amount of substance remaining in the L region |
| M.W. | Molecular Weight |
| PMMA | Poly(methyl methacrylate) |
| PMMA-6 | Nanocomposite with 2.5% reinforcement particle ratio prepared by mixing for 6 minutes |
| PMMA-12 | Nanocomposite with 2.5% reinforcement particle ratio prepared by mixing for 12 minutes |
| Residue | Remaining substance as a percentage |
| SEM | Scanning electron microscope |
| TG | Initial amount of substance |
| XRD | X-ray diffractions |
| A | Calculation of the area under the thermal graph training |
| β | Heating Rate |
| Tx | The maximum mass loss temperatures |
References
- Hazim, A.; Abduljalil, H.M.; Hashim, A. Design of PMMA doped with inorganic materials as promising structures for optoelectronics applications. Trans. Electr. Electron. Mater. 2021, 22, 851–868.
- Çankaya, N.; Beşci, G. Synthesis, characterization, thermal properties and reactivity ratios of methacrylate copolymers including methoxy group. J. Fac. Eng. Archit. Gazi Univ. 2018, 33(3), 1155–1170.
- Ul-Haq, Y.; Murtaza, I.; Mazhar, S.; Ullah, R.; Iqbal, M.; Ul-Huq, Z.; Qarni, A.A.; Amin, S. Dielectric, thermal and mechanical properties of hybrid PMMA/RGO/Fe₂O₃ nanocomposites fabricated by in-situ polymerization. Ceram. Int. 2020, 46, 5828–5840.
- Khokhar, D.; Jadoun, S.; Arif, R.; Jabin, S. Functionalization of conducting polymers and their applications in optoelectronics. Polym. Plast. Technol. Mater. 2021, 60, 465–487.
- Alsaad, A.M.; Al-Bataineh, Q.M.; Ahmad, A.A.; Jum’h, I.; Alaqtash, N.; Bani-Salameh, A.A. Optical properties of transparent PMMA-PS/ZnO NPs polymeric nanocomposite films: UV-shielding applications. Mater. Res. Express 2019, 6, 126446.
- Lee, J.K.Y.; Chen, N.; Peng, S.; Li, L.; Tian, L.; Thakor, N.; Ramakrishna, S. Polymer-based composites by electrospinning: Preparation & functionalization with nanocarbons. Prog. Polym. Sci. 2018, 86, 40–84.
- Bafna, M.; Sain, N.; Khandelwal, A.; Deeba, F.; Gupta, A.K. Study of refractive index and dispersion behavior of KMnO₄ doped poly(methyl methacrylate) (PMMA) composites. Mater. Today Proc. 2022, 66, 3481–3486.
- Agarwal, K.; Prasad, M.; Katiyar, M.; Jaiswal, R.; Kumar, S.; Prasad, N.E. Study of electromagnetic and thermal properties of fabricated NiFe₂O₄/polyurethane nanocomposites. J. Appl. Polym. Sci. 2020, 137(27),.
- Mosleh, A.T.; Yousef, T.A.; Khairy, M.; Ferjani, H.; Almuhana, A.R.Y.; Zahran, H.Y.; Rahman, A.E.; Essam, O.A.; Abdelnasser, M.I.; Abdelbaset, S.A.; Yahi, I.S.; Kamoun, E.A. Multifunctional prospects of PMMA/Fe₂O₃@NiO nanocomposite membranes: Advanced optical, dielectric, and photocatalytic properties for electronic, optoelectronic devices, and environmental applications. J. Sol-Gel Sci. Technol. 2025, 116, 778–797.
- Rocha, M.V.J.; de Carvalho, H.W.P.; Sarmento, V.H.; Craievich, A.F.; Ramalho, T.C. Thermal and structural characterization of maghemite-PMMA nanocomposite obtained by in-situ polymerization. Polym. Compos. 2016, 37, 51–58.
- Dallas, P.; Georgakilas, V.; Niarchos, D.; Komninou, P.; Kehagias, T.; Petridis, D. Thermal properties of polymer/magnetic nanoparticle composite films. Nanotechnology 2006, 17, 2046–2054.
- Singh, B.P.; Kumar, A.; Areizaga-Martinez, H.I.; Vega-Olivencia, C.A.; Tomar, M.S. Structural, optical, and dielectric studies of NiO/PMMA nanocomposite films prepared by solution casting. Indian J. Pure Appl. Phys. 2017, 55, 722–729.
- Laachachi, A.; Cochez, M.; Ferriol, M.; Lopez-Cuesta, J.M.; Leroy, E. Mechanical and thermal properties of polypropylene/metal oxide composite materials. Mater. Lett. 2005, 59, 36–39.
- Barandiaran, I.; Cappelletti, A.; Strumia, M.; Eceiza, A.; Kortaberria, G. Generation of nanocomposites based on (PMMA-b-PCL)-grafted Fe₂O₃ nanoparticles and PS-b-PCL block copolymer. Eur. Polym. J. 2014, 58, 226–232.
- Aymonier, C.; Bortzmeyer, D.; Thomann, R.; Mülhaupt, R. Poly(methyl methacrylate)/palladium nanocomposites: Synthesis and characterization of morphological, thermomechanical and thermal properties. Chem. Mater. 2003, 15, 4874–4878.
- Ulutas, A. NiO/PMMA Nanokompozitlerinin Termal Kararlılığı ve Aktivasyon Enerjileri Üzerine Karıştırma Süresinin Etkisi. J. Compos. Sci. 2025, 9(10), 557.
- Rahman, M.M.; Khan, K.H.; Parvez, M.M.H.; Irizarry, N.; Uddin, M.N. Polymer Nanocomposites with Optimized Nanoparticle Dispersion and Enhanced Functionalities for Industrial Applications. Processes 2025, 13(4), 994.
- Rishi, K.; Narayanan, V.; Beaucage, G.; McGlasson, A.; Kuppa, V.; Ilavsky, J.; Rackaitis, M. A thermal model to describe kinetic dispersion in rubber nanocomposites: The effect of mixing time on dispersion. Polymer 2019, 175, 272–282.
- Sun, T.; Fan, H.; Wang, Z.; Liu, X.; Wu, Z. Modified nano Fe2O3-epoxy composite with enhanced mechanical properties. Mater. Des. 2015, 87, 10–16.
- Naguib, H.M.; Ahmed, M.A.; Abo-Shanab, Z.L. Studying the loading impact of silane grafted Fe₂O₃ nanoparticles on mechanical characteristics of epoxy matrix. Egypt. J. Petrol. 2019, 28, 27–34.
- Khosravi Maleki, F.; Nasution, M.K.M.; Gok, M.S.; Arab Maleki, V. An experimental investigation on mechanical properties of Fe₂O₃ microparticles reinforced polypropylene. J. Mater. Res. Technol. 2022, 16, 229–237.
- Katea, M.B.; Aboiad, A.K.; Al-Alawy, I.T. Synthesis and characterization of PMMA-based nanocomposites doped with Bi₂O₃, MoO₃, B₂O₃, SiO₂, Na₂O and Fe₂O₃ for gamma radiation shielding. Appl. Radiat. Isot. 2025, 226, 11252.
- Cho, Y.-M.; Kim, J.-H.; Choi, J.-H.; Kim, J.-C.; Cho, S.-M.; Park, S.-W.; Kwak, H.W.; Choi, I.-G. Physicochemical characteristics of lignin-g-PMMA/PLA blend via atom transfer radical polymerization depending on the structural difference of organosolv lignin. Int. J. Biol. Macromol. 2023, 226, 279–290.
- Li, S.; Meng Lin, M.; Toprak, M.S.; Kim, D.K.; Muhammed, M. Nanocomposites of Polymer and Inorganic Nanoparticles for Optical and Magnetic Applications. Nano Rev. 2010, 1, 5214.
- Hashem, M.; Rez, M.F.; Fouad, H.; Elsarnagawy, T.; Elsharawy, M.A.; Umar, A.; Assery, M.; Ansari, S.G. Influence of titanium oxide nanoparticles on the physical and thermomechanical behavior of poly(methyl methacrylate) (PMMA): A denture base resin. Sci. Adv. Mater. 2017, 9(6), 938–944.
- Ayanoğlu, Z.G.; Doğan, M. Characterization and Thermal Kinetic Analysis of PMMA/Modified-MWCNT Nanocomposites. Diam. Relat. Mater. 2020, 108, 107950.
- Campo, E.A. Polymeric Materials and Properties. In Selection of Polymeric Materials; Elsevier: Amsterdam, The Netherlands, 2008; pp. 1–39.
- C. E. Porter and F.D. Blum, Thermal Characterization of PMMA Thin Films Using Modulated Differential Scanning Calorimetry, Macromolecules, 2000 33, 7016-7020.
- Ozawa, T. Kinetic Analysis of Derivative Curves in Thermal Analysis. J. Therm. Anal. 1970, 2, 301–324.
- Kissinger, H.E. Variation of Peak Temperature with Heating Rate in Differential Thermal Analysis. J. Res. Natl. Bur. Stand. (1934) 1956, 57, 217.
- Ul-Haq, Y.; Murtaza, I.; Mazhar, S.; Ullah, R.; Iqbal, M.; Zeeshan-ul-Huq; Qarni, A.A.; Amin, S. Dielectric, Thermal and Mechanical Properties of Hybrid PMMA/RGO/Fe2O3 Nanocomposites Fabricated by in-Situ Polymerization. Ceram. Int. 2020, 46, 5828–5840.
- Aygahoğlu, A.; Döner, N.; Pargan, A.R. Hematite particles’ radiative properties within the ultraviolet–visible spectrum and thermal properties at low temperatures. J. Fac. Eng. Archit. Gazi Univ. 2020, 36(1), 191–200.
- Yanmaz, E.; Doğan, M.; Turhan, Y. Effect of sodium dodecyl sulfate on thermal properties of polyvinyl alcohol (PVA)/modified single-walled carbon nanotube (SWCNT) nanocomposites. Diam. Relat. Mater. 2021, 115, 108359.
- Hu, Y.-H.; Chen, C.-Y.; Wang, C.-C. Viscoelastic properties and thermal degradation kinetics of silica/PMMA nanocomposites. Polym. Degrad. Stab. 2004, 84(3), 545–553.
- Imai, T.; Matsui, T.; Fujii, Y.; Nakai, T.; Tanaka, S. Oxidation catalyst of iron oxide suppressing dioxin formation in polyethylene combustion. J. Mater. Cycles Waste Manage. 2001, 3, 103–109.

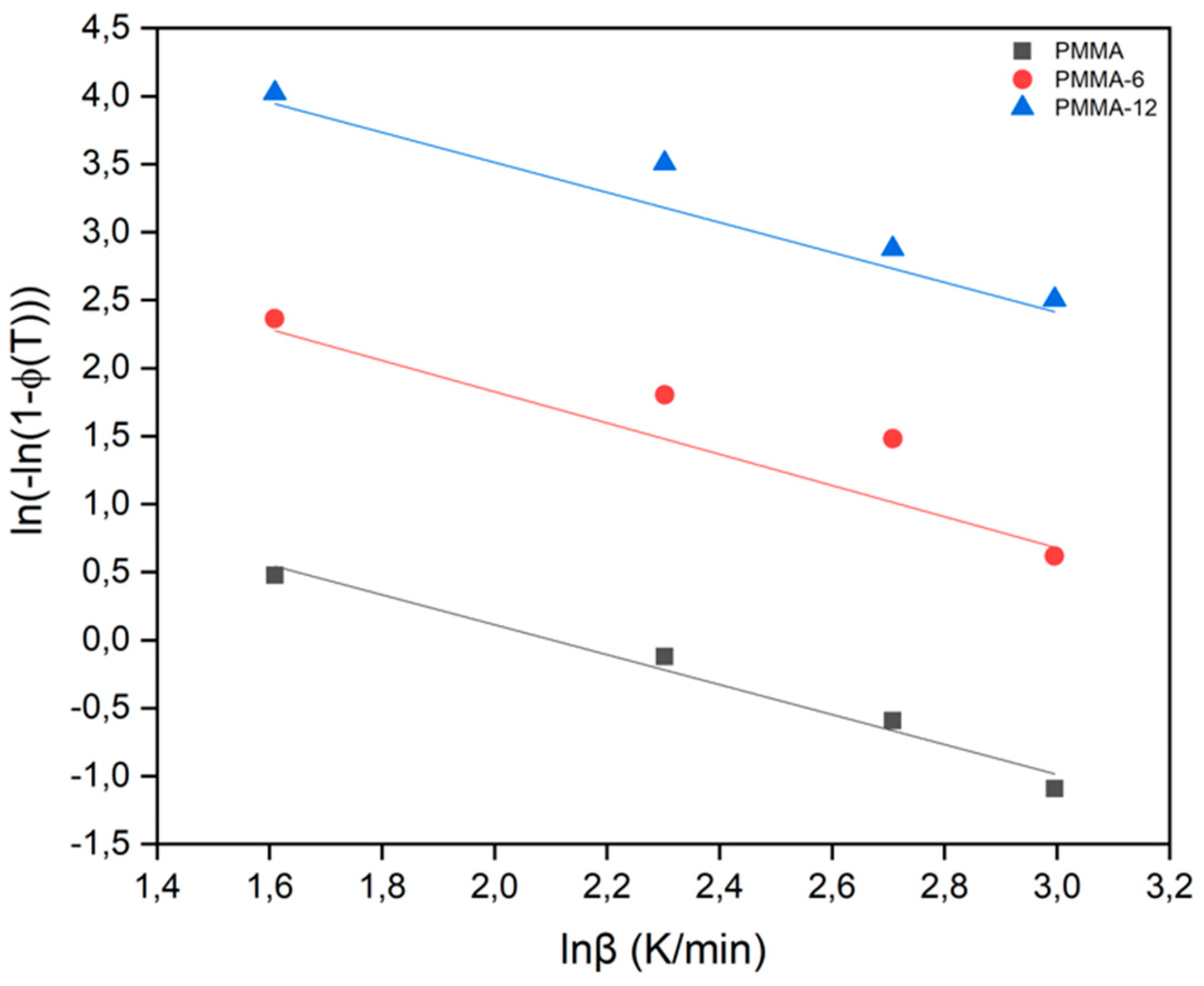
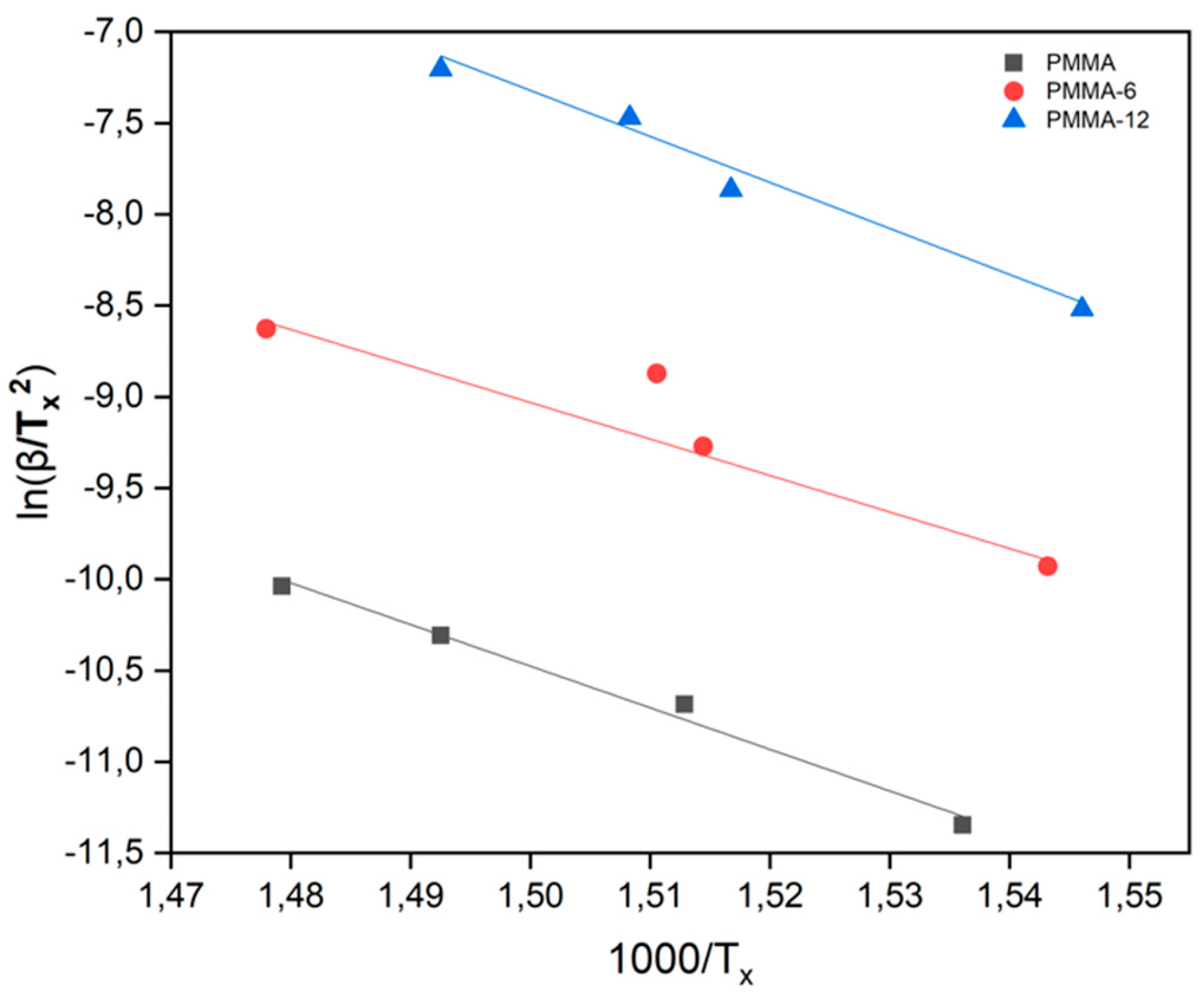
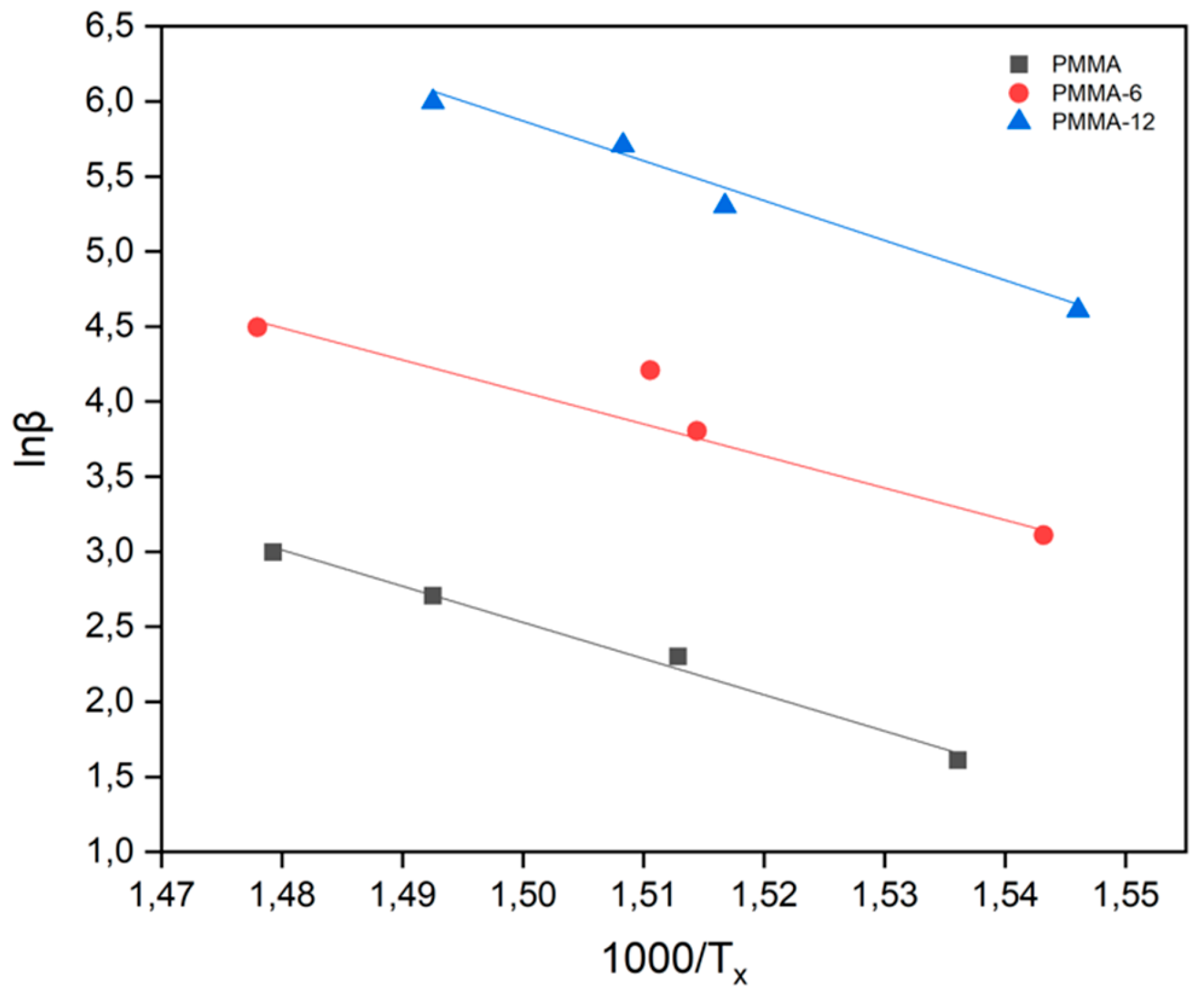
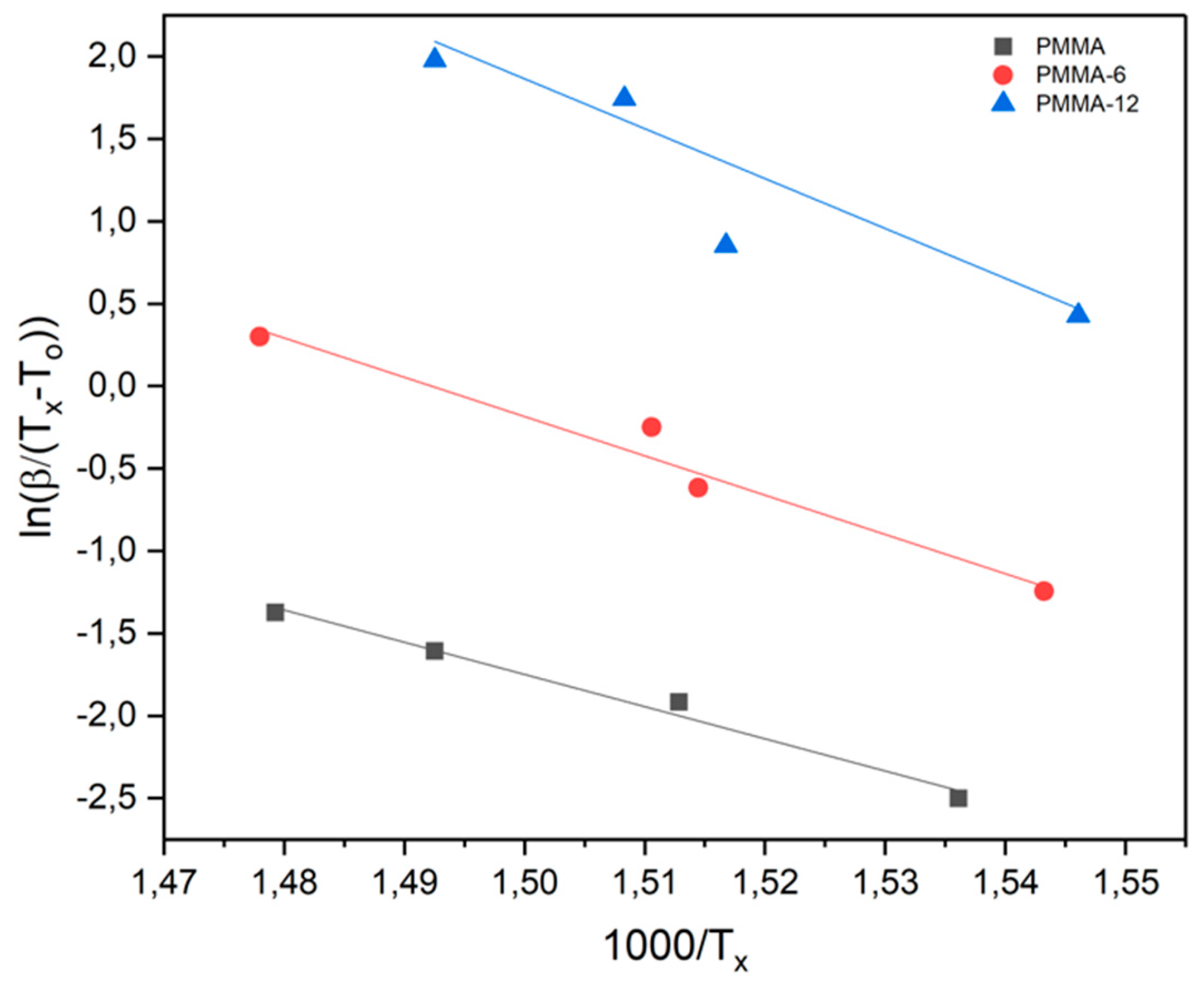
| SAMPLES |
β (K/min) |
TO (K) |
TX (K) |
A |
| PMMA | 5 | 590 | 651 | 266 |
| 10 | 593 | 661 | 279 | |
| 15 | 595 | 670 | 311 | |
| 20 | 597 | 676 | 281 | |
| PMMA-6 | 5 | 556 | 648 | 372 |
| 10 | 562 | 660 | 323 | |
| 15 | 560 | 662 | 323 | |
| 20 | 598 | 676 | 289 | |
| PMMA-12 | 5 | 594 | 646 | 337 |
| 10 | 590 | 659 | 306 | |
| 15 | 620 | 663 | 274 | |
| 20 | 625 | 670 | 249 |
| Samples | Avrami Parameter | Kissinger Method (Kj/Mol) |
Takhor Method (Kj/Mol) |
Augis-Bennett Method (Kj/Mol) |
| PMMA | 1 | 189 | 200 | 162 |
| PMMA-6 | 1 | 166 | 177 | 198 |
| PMMA-12 | 1 | 209 | 220 | 251 |
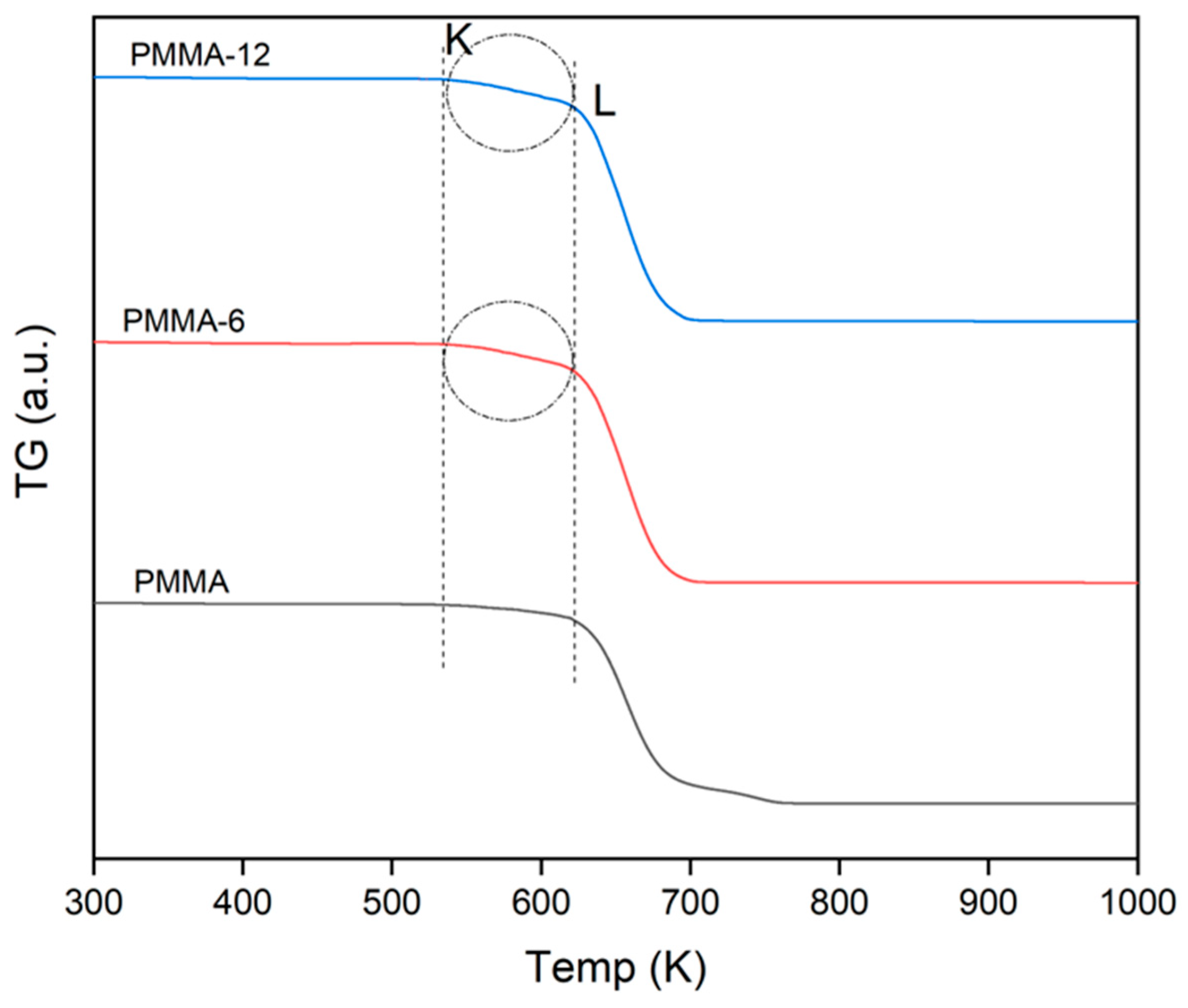
| Samples |
TG (µg) |
K (µg) |
L (µg) |
T5 (K) |
T30 (K) |
T50 (K) |
T80 (K) |
TX |
Residue % |
| PMMA | 11.19 | 11.08 | 10.26 | 601 | 647 | 658 | 682 | 661 | 0.42 |
| PMMA-6 | 13.01 | 12.89 | 11.45 | 582 | 641 | 653 | 671 | 660 | 0.90 |
| PMMA-12 | 13.32 | 13.19 | 11.68 | 578 | 640 | 653 | 671 | 659 | 1.43 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




