Submitted:
30 January 2026
Posted:
02 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Material
2.2.1. Curing Kinetics Evolution
2.2.2. Gelation
2.2.3. Glass Transition Temperatures
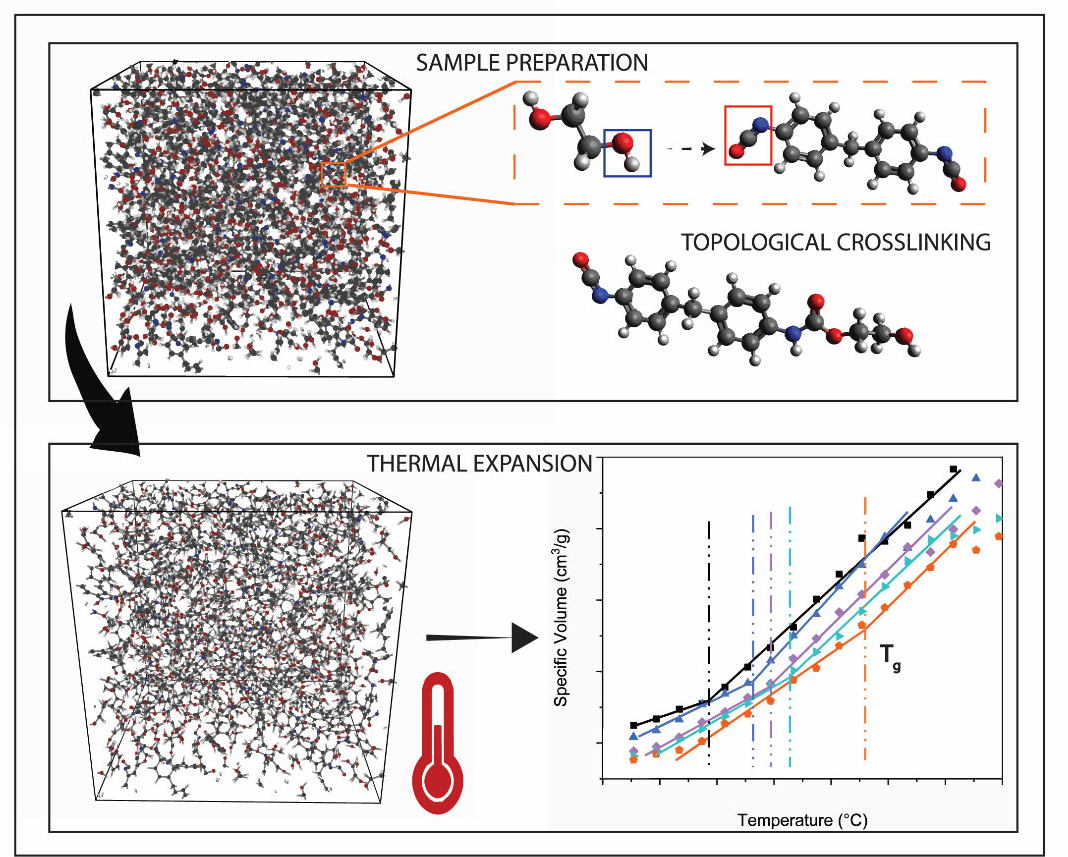
2.2.4. Molecular Dynamic Simulation capturing Tg
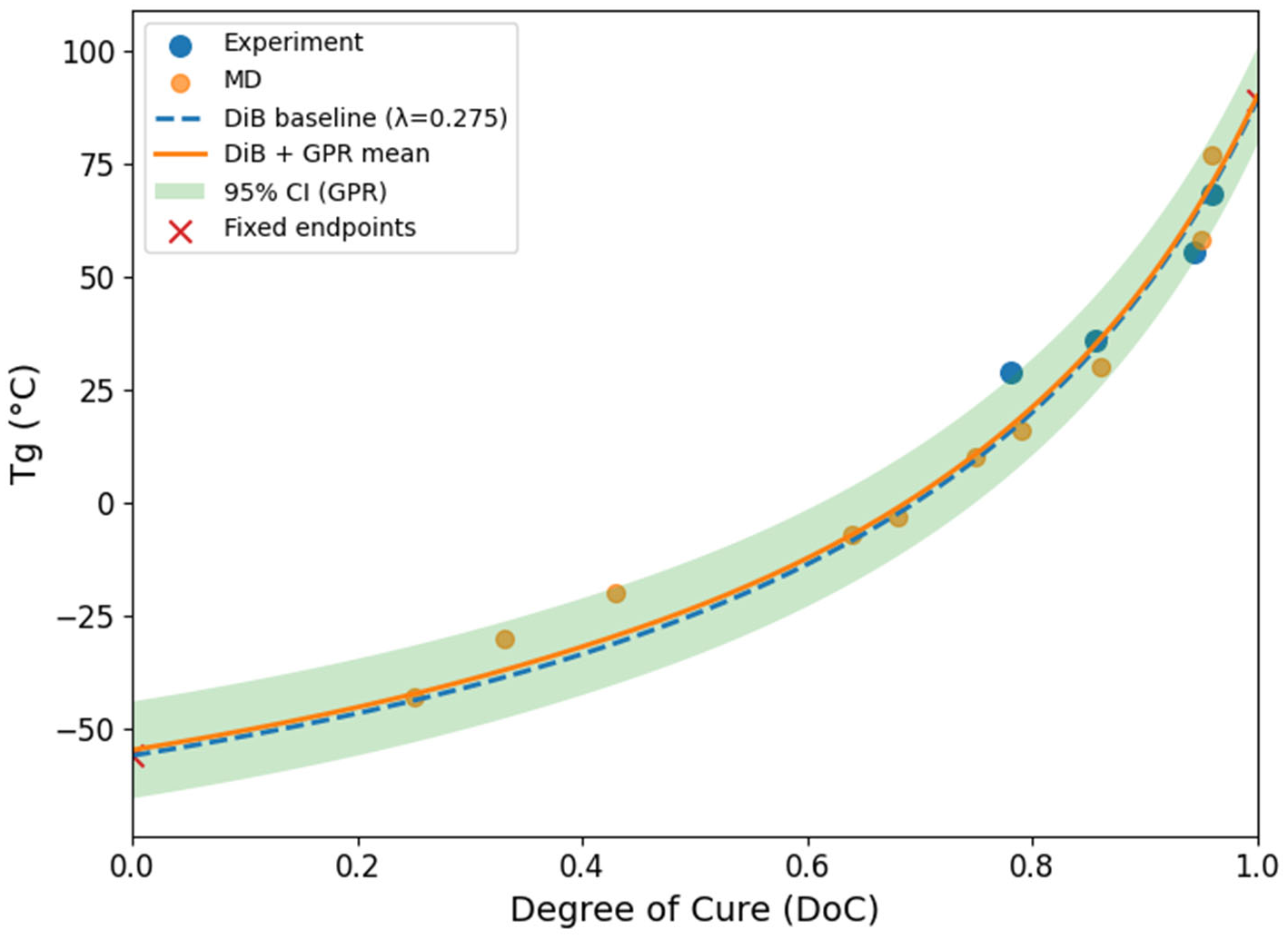
2.3. Physics-Informed Prediction
3. Results
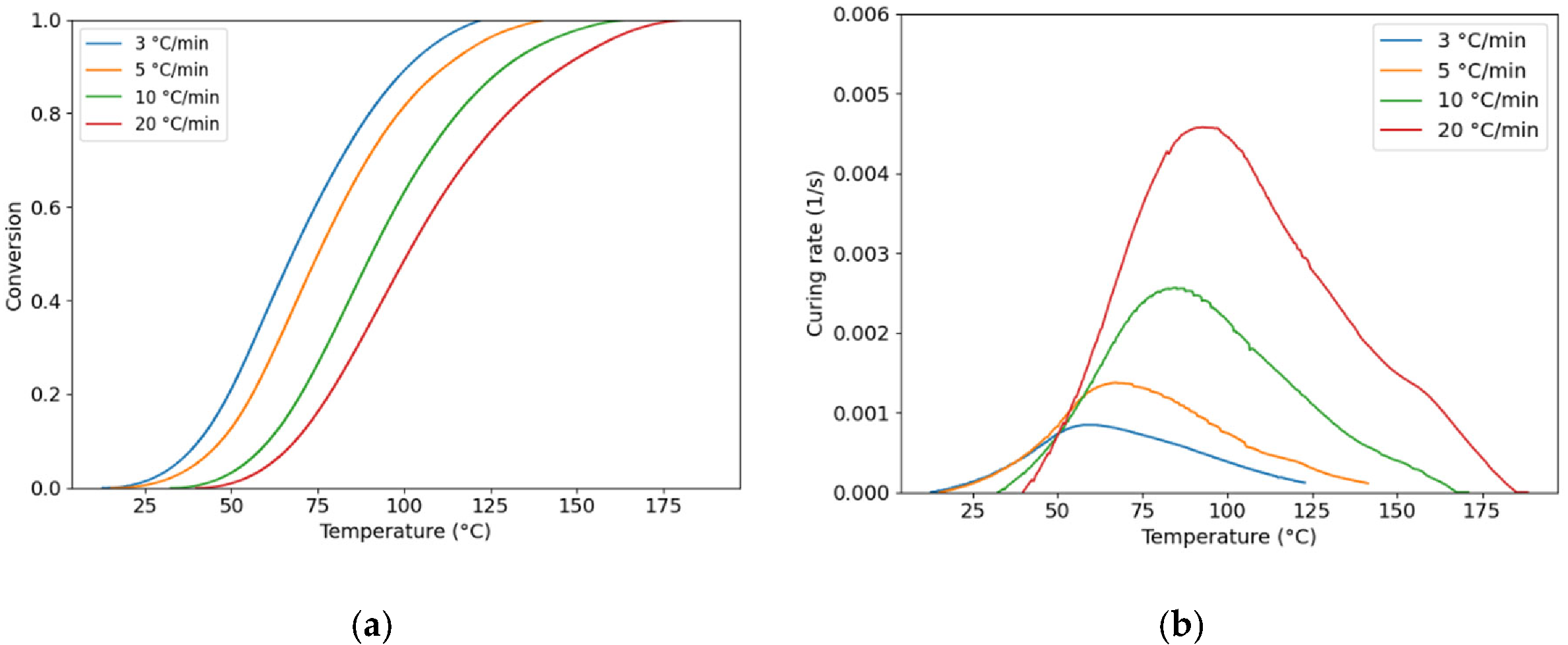
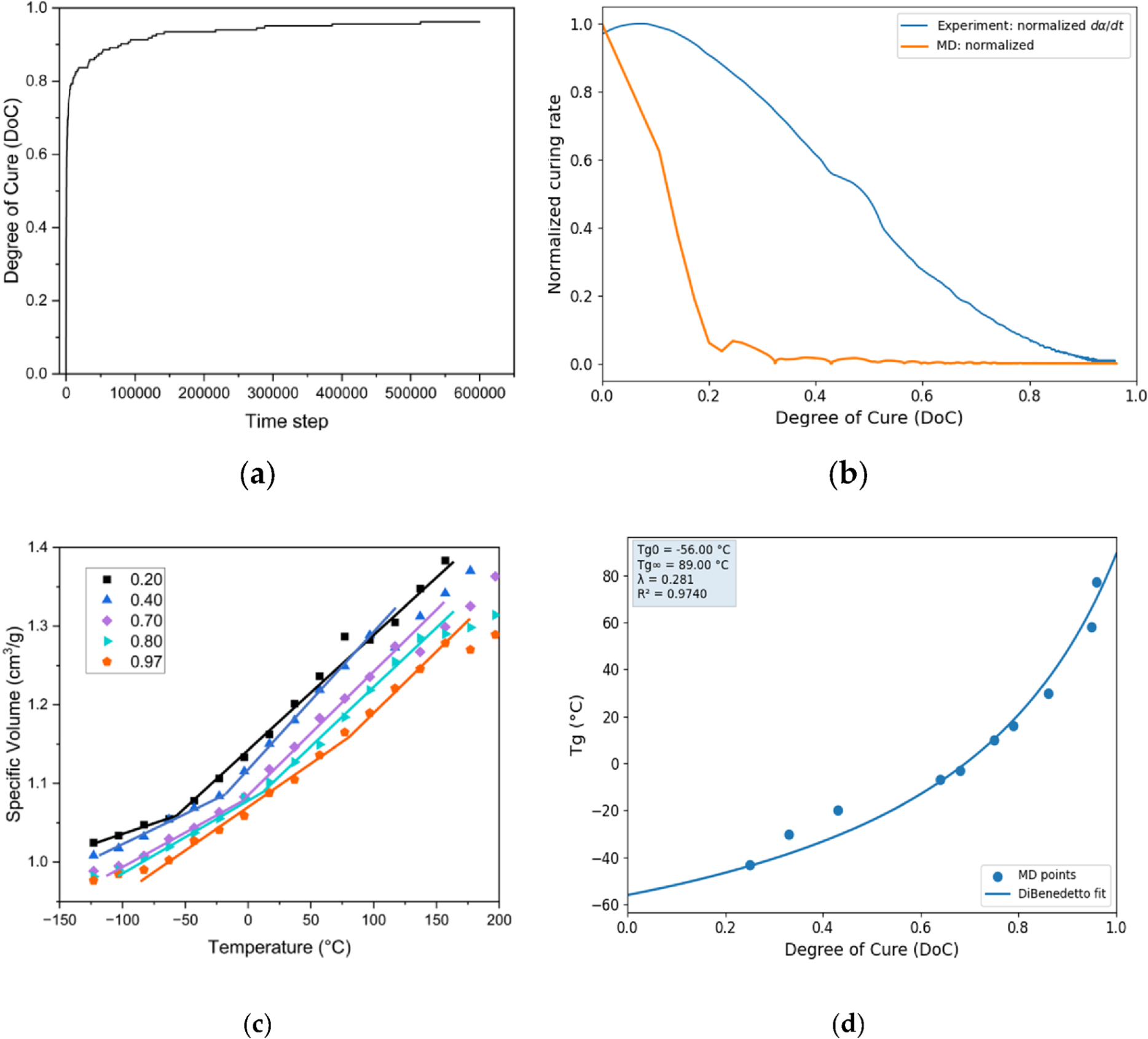
3.1. Curing Kinetics Evolution
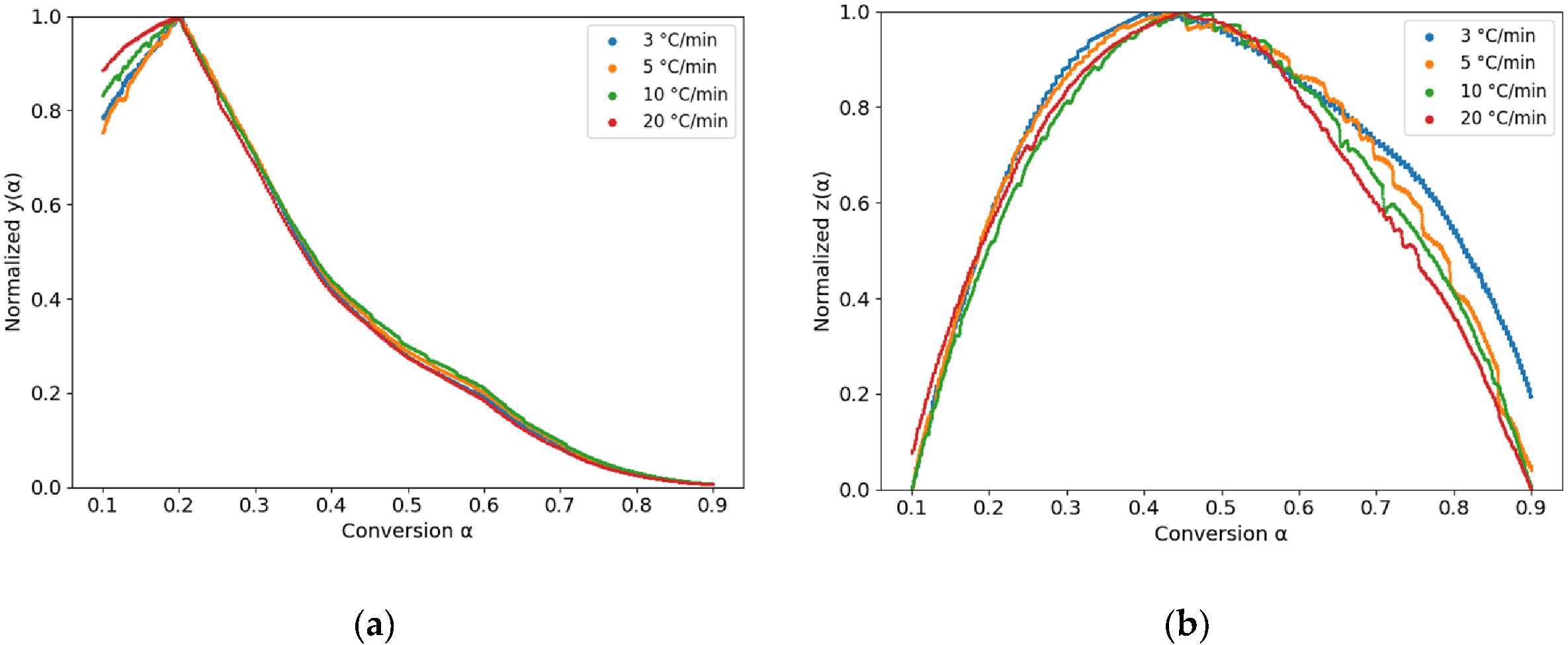
| Heating rate | αp | αM | αp∞ |
|---|---|---|---|
| 3 °C/min | 0.325 | 0.201 | 0.421 |
| 5 °C/min | 0.381 | 0.200 | 0.448 |
| 10 °C/min | 0.407 | 0.200 | 0.450 |
| 20 °C/min | 0.389 | 0.198 | 0.450 |
| Heating rate | R2 | |
| DOC | Curing rate | |
| 3 °C/min | 0.997 | 0.953 |
| 5 °C/min | 0.999 | 0.972 |
| 10 °C/min | 0.999 | 0.981 |
3.2. Gelation
3.3. Glass Transition Temperatures
3.4. Molecular Dynamic Simulation capturing Tg
3.5. Gaussian Process Regression Prediction
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PU | Polyurethane |
| Tg | Glass transition temperature |
| DSC | Differential scanning calorimetry |
| TMA | Thermomechanical analysis |
| DoC | Degree of cure |
| MD | Molecular dynamics |
| EMC | Enhanced Monte Carlo |
| ML | Machine learning |
| GPR | Gaussian Process Regression |
| CTE | Coefficient of thermal expansion |
| OPLS-AA | Optimized potentials for liquid simulation all atom |
| COMPASS | Condensed-phase Optimized Molecular Potential for Atomistic Simulation Studies |
| PCFF | Polymer consistent force field |
| NPT | Constant Number of particles, Pressure, and Temperature |
| RMSE | Root mean square error |
| KS | Kamal–Sourour |
| TTT | Time–temperature–transformation |
References
- De Souza, Felipe M; Kahol, Pawan K; Gupta, Ram K. Introduction to polyurethane chemistry. In Polyurethane chemistry: Renewable polyols and isocyanates; ACS Publications, 2021; pp. pages 1–24. [Google Scholar]
- Heath, Daniel E.; Cooper, Stuart L. A - polyurethanes. In Biomaterials Science (Third Edition), third edition edition; Ratner, Buddy D., Hoffman, Allan S., Schoen, Frederick J., Lemons, Jack E., Eds.; Academic Press, 2013; pp. pages 79–82. [Google Scholar]
- Wienen, David; Gries, Thomas; Cooper, Stuart L; Heath, Daniel E. An overview of polyurethane biomaterials and their use in drug delivery. Journal of Controlled Release 2023, 363, 376–388. [Google Scholar] [CrossRef]
- Das, Abhijit; Mahanwar, Prakash. A brief discussion on advances in polyurethane applications. Advanced Industrial and Engineering Polymer Research 2020, 3(3), 93–101. [Google Scholar] [CrossRef]
- Mukherji, Arindam; Njuguna, James. An assessment on effect of process parameters on pull force during pultrusion. The International Journal of Advanced Manufacturing Technology 2022, 121(5), 3419–3438. [Google Scholar] [CrossRef]
- Irfan, Muhammad S; Shotton-Gale, Nicholas; Paget, Mark A; Machavaram, Venkata R; Leek, Colin; Wootton, Shane; Hudson, Mark; Helsmans, Stefan; Bogonez, Francisco N; Pandita, Surya D; et al. A modified pultrusion process. Journal of Composite Materials 2017, 51(13), 1925–1941. [Google Scholar] [CrossRef]
- Baran, Ismet; Tutum, Cem C; Nielsen, Michael W; Hattel, Jesper H. Process induced residual stresses and distortions in pultrusion. Composites Part B: Engineering 2013, 51, 148–161. [Google Scholar] [CrossRef]
- Venditti, RA; Gillham, JK. A relationship between the glass transition temperature (tg) and fractional conversion for thermosetting systems. Journal of Applied Polymer Science 1997, 64(1), 3–14. [Google Scholar] [CrossRef]
- Ramis, X; Cadenato, A; Morancho, JM; Salla, JM. Curing of a thermosetting powder coating by means of dmta, tma and dsc. Polymer 2003, 44(7), 2067–2079. [Google Scholar] [CrossRef]
- Teil, H; Page, SA; Michaud, V; Månson, J-AE. Ttt-cure diagram of an anhydridecured epoxy system including gelation, vitrification, curing kinetics model, and monitoring of the glass transition temperature. Journal of Applied Polymer Science 2004, 93(4), 1774–1787. [Google Scholar] [CrossRef]
- Gu, Yuwei; Zhao, Julia; Johnson, Jeremiah A. Polymer networks: from plastics and gels to porous frameworks. Angewandte Chemie International Edition 2020, 59(13), 5022–5049. [Google Scholar] [CrossRef] [PubMed]
- Imrie, Patrick; Jin, Jianyong. Gelation of raft polymer networks analyzed by rheology. Polymer 2025, 317, 127956. [Google Scholar] [CrossRef]
- Karim Boulkadid, Moulai; Touidjine, Sabri; Tra che, Djalal; Belkhiri, Samir. Analytical methods for the assessment of curing kinetics of polyurethane binders for high-energy composites. Critical Reviews in Analytical Chemistry 2022, 52(5), 1112–1121. [Google Scholar] [CrossRef]
- Jennrich, Rebecca; Lion, Alexander; Johlitz, Michael; Ernst, Sarah; Dilger, Klaus; Stammen, Elisabeth. Thermomechanical characterization and modeling of fast-curing polyurethane adhesives. Continuum Mechanics and Thermodynamics 2020, 32(2), 421–432. [Google Scholar] [CrossRef]
- Han, Jie; Gee, Richard H; Boyd, Richard H. Glass transition temperatures of polymers from molecular dynamics simulations. Macromolecules 1994, 27(26), 7781–7784. [Google Scholar] [CrossRef]
- in’t Veld, Pieter J; Rutledge, Gregory C. Temperature-dependent elasticity of a semicrystalline interphase composed of freely rotating chains. Macromolecules 2003, 36(19), 7358–7365. [Google Scholar] [CrossRef]
- Seeger, Matthias. Gaussian processes for machine learning. International journal of neural systems 2004, 14(02), 69–106. [Google Scholar] [CrossRef]
- Wang, Jie. An intuitive tutorial to gaussian process regression. Computing in Science & Engineering 2023, 25(4), 4–11. [Google Scholar] [CrossRef]
- Flory, Paul J. Molecular size distribution in three dimensional polymers. v. postgelation relationships. Journal of the American Chemical Society 1947, 69(1), 30–35. [Google Scholar] [CrossRef]
- Lang, Michael; Muller, T. Analysis of the gel point of polymer model networks by computer simulations. Macromolecules 2020, 53(2), 498–512. [Google Scholar] [CrossRef]
- Li, X; Nakagawa, S; Tsuji, Y; Watanabe, N; Shibayama, M. Polymer gel with a flexible and highly ordered three-dimensional network synthesized via bond percolation. Science advances 2019, 5(12), eaax8647. [Google Scholar] [CrossRef]
- Zhang, Chao; Li, Yi; Wu, Yongshen; Wang, Cuixia; Liang, Jian; Xu, Zihan; Zhao, Peng; Wang, Jing. Key role of cross-linking homogeneity in polyurethane mechanical properties: Insights from molecular dynamics. The Journal of Physical Chemistry B 2024, 128(50), 12612–12627. [Google Scholar] [CrossRef]
- Hadipeykani, Majid; Aghadavoudi, Farshid; Toghraie, Davood. A molecular dynamics simulation of the glass transition temperature and volumetric thermal expansion coefficient of thermoset polymer based epoxy nanocomposite reinforced by cnt: a statistical study. Physica A: Statistical Mechanics and its Applications 2020, 546, 123995. [Google Scholar] [CrossRef]
- Zheng, Tianze; Li, Ting; Shi, Jiaxin; Wu, Tianyu; Zhuang, Zhuo; Xu, Jun; Guo, Baohua. Molecular insight into the toughness of polyureas: A hybrid all atom/coarse-grained molecular dynamics study. Macromolecules 2022, 55(8), 3020–3029. [Google Scholar] [CrossRef]
- Olejnik, Adrian; Gosz, Kamila; Piszczyk, Łukasz. Kinetics of cross-linking processes of fast-curing polyurethane system. Thermochimica Acta 2020, 683, 178435. [Google Scholar] [CrossRef]
- Molecular Massively Parallel Simulator. Lammps. 2013. Available online: http://lammps.sandia.gov.
- Fried, J.R.; Li, B. Atomistic simulation of the glass transition of di-substituted polysilanes. Computational and Theoretical Polymer Science 2001, 11(4), 273–281. [Google Scholar] [CrossRef]
- Sun, H.; Ren, P.; Fried, J.R. The com-pass force field: parameterization and validation for phosphazenes. Computational and Theoretical Polymer Science 1998, 8(1), 229–246. [Google Scholar] [CrossRef]
- DiBenedetto, AT. Prediction of the glass transition temperature of polymers: a model based on the principle of corresponding states. Journal of Polymer Science Part B: Polymer Physics 1987, 25(9), 1949–1969. [Google Scholar] [CrossRef]
- Duvenaud, David. The kernel cookbook: Advice on covariance functions. 2014. Available online: https://www.
- Ameli, Siavash; Shadden, Shawn C. Noise estimation in gaussian process regression. arXiv 2022, arXiv:2206.09976. [Google Scholar] [CrossRef]
- Anguita, Davide; Ghelardoni, Luca; Ghio, Alessandro; Oneto, Luca; Ridella, Sandro; et al. The’k’in k-fold cross validation. Esann 2012, volume 102, 441–446. [Google Scholar]
- Qiu, Chunfu. Nonparametric regression and generalized linear models: a roughness, penalty approach; 1995. [Google Scholar]
- Bishop, Christopher M; Nasrabadi, Nasser M. Pattern recognition and machine learning; Springer, 2006; volume 4. [Google Scholar]
- Navarro-García, Manuel; Guerrero, Vanesa; Durban, María. On constrained smoothing and out-of-range prediction using p-splines: A conic optimization approach. Applied Mathematics and Computation 2023, 441, 127679. [Google Scholar] [CrossRef]
- Yousefi, A; Lafleur, Pierre G; Gauvin, Raymond. Kinetic studies of thermoset cure reactions: a review. Polymer Composites 1997, 18(2), 157–168. [Google Scholar] [CrossRef]
- D Ro¸su, CN Ca¸scaval; Mustatˇa, F.; Ciobanu, C. Cure kinetics of epoxy resins studied by non-isothermal dsc data. Thermochimica Acta 2002, 383(1-2), 119–127. [Google Scholar] [CrossRef]
- Málek, Jiří. Kinetic analysis of non-isothermal calorimetric data; 1996. [Google Scholar]
- Málek, Jiří. Kinetic analysis of crystallization processes in amorphous materials. Thermochimica Acta 2000, 355(1-2), 239–253. [Google Scholar] [CrossRef]
- Senum, GI; Yang, RT. Rational approximations of the integral of the arrhenius function. Journal of thermal analysis 1977, 11(3), 445–447. [Google Scholar] [CrossRef]
- Vyazovkin, Sergey. Isoconversional kinetics of thermally stimulated processes; Springer, 2015. [Google Scholar]
- Kamal, MR; Sourour, S. Kinetics and thermal characterization of thermoset cure. Polymer Engineering & Science 1973, 13(1), 59–64. [Google Scholar] [CrossRef]
- Dennis, John E, Jr.; Welsch, Roy E. Techniques for nonlinear least squares and robust regression. Communications in Statistics-simulation and Computation 1978, 7(4), 345–359. [Google Scholar] [CrossRef]
- Gazo Hanna, Eddie; Younes, Khaled; Amine, Semaan; Roufayel, Rabih. Exploring gel-point identification in epoxy resin using rheology and unsupervised learning. Gels 2023, 9(10), 828. [Google Scholar] [CrossRef] [PubMed]
- Lange, J; Altmann, N; Kelly, CT; Halley, PJ. Understanding vitrification during cure of epoxy resins using dynamic scanning calorimetry and rheological techniques. Polymer 2000, 41(15), 5949–5955. [Google Scholar] [CrossRef]
- Cai, Jianfen J; Salovey, Ronald. Chemorheology of model filled rubber compounds during curing. Polymer Engineering & Science 2001, 41(11), 1853–1858. [Google Scholar] [CrossRef]
- Winter, H Henning; Chambon, Francois. Analysis of linear viscoelasticity of a crosslinking polymer at the gel point. Journal of rheology 1986, 30(2), 367–382. [Google Scholar] [CrossRef]
- Ionescu, Mihail. Chemistry and technology of polyols for polyurethanes; iSmithers Rapra Publishing, 2005. [Google Scholar]
- Gertig, Christoph; Erdkamp, Eric; Ernst, Andreas; Hemprich, Carl; Kr¨oger, Leif C; Langanke, Jens; Bardow, Andr´e; Leonhard, Kai. Reaction mechanisms and rate constants of auto-catalytic urethane formation and cleavage reactions. ChemistryOpen 2021, 10(5), 534–544. [Google Scholar] [CrossRef] [PubMed]
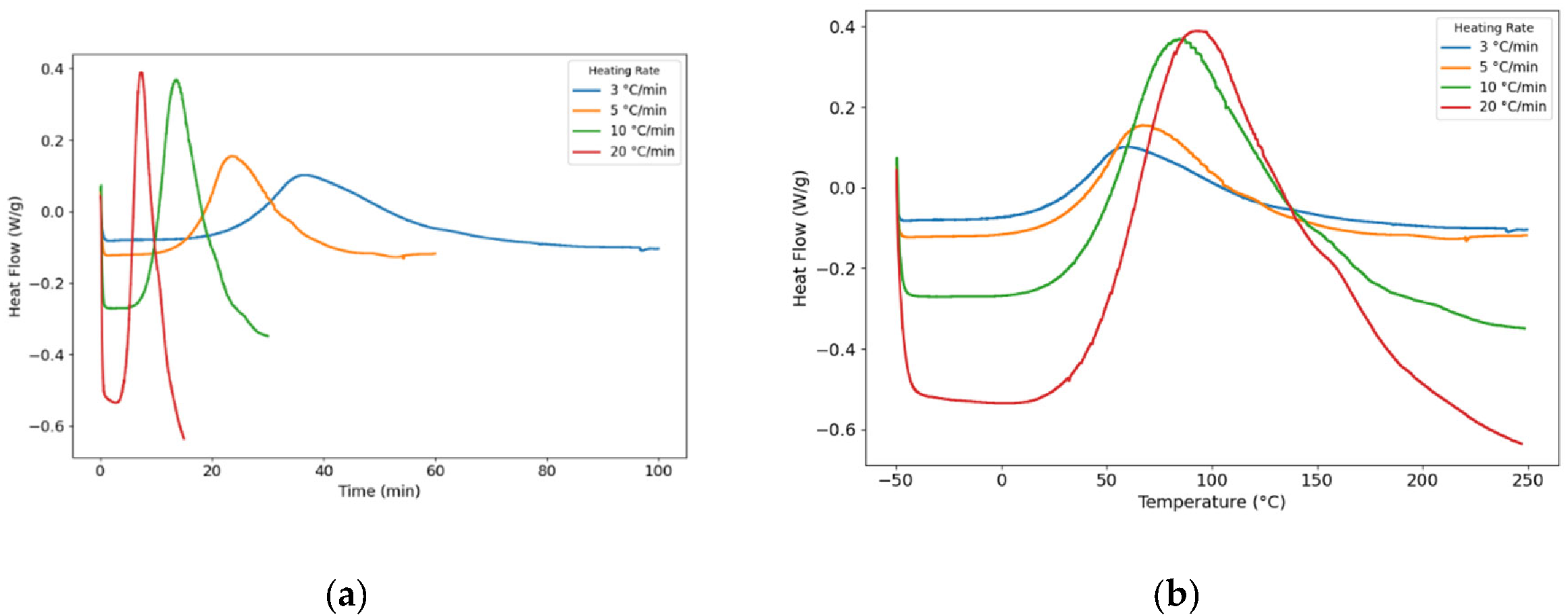
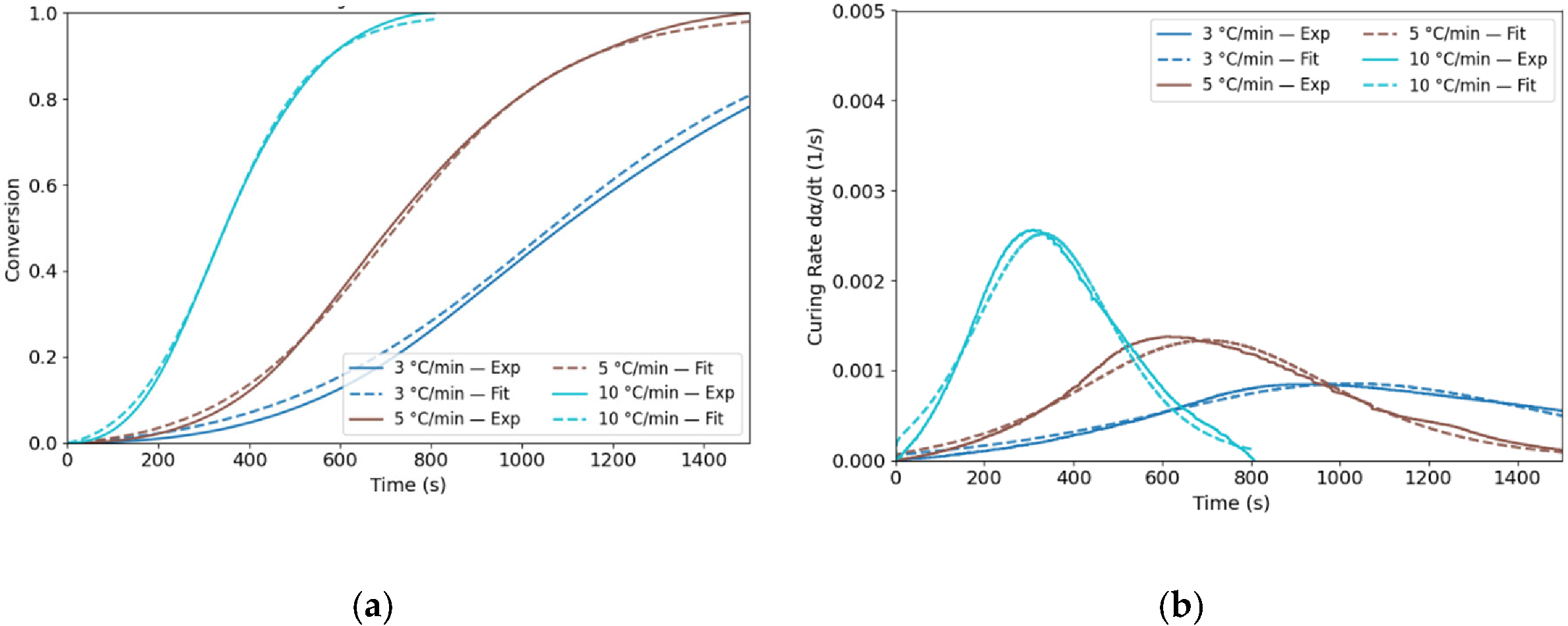
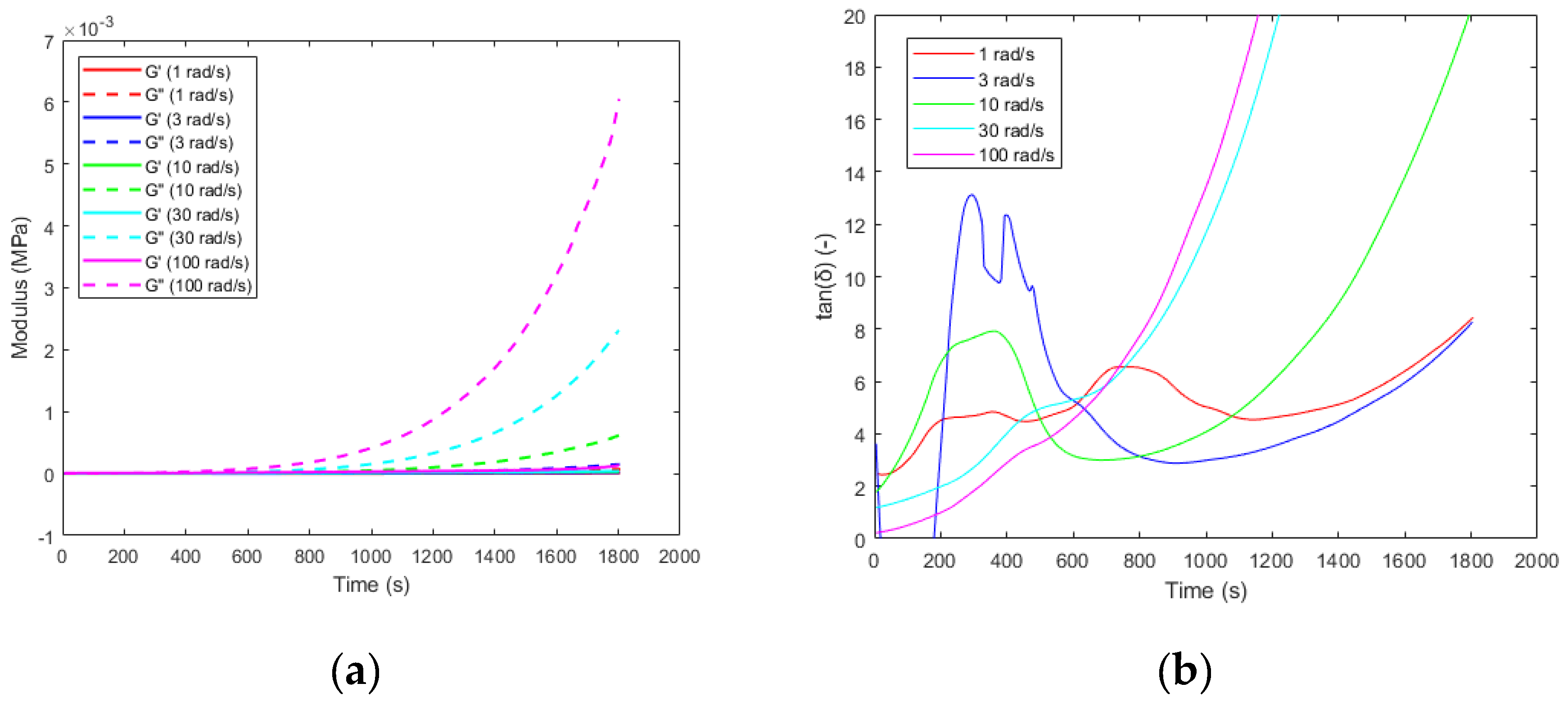
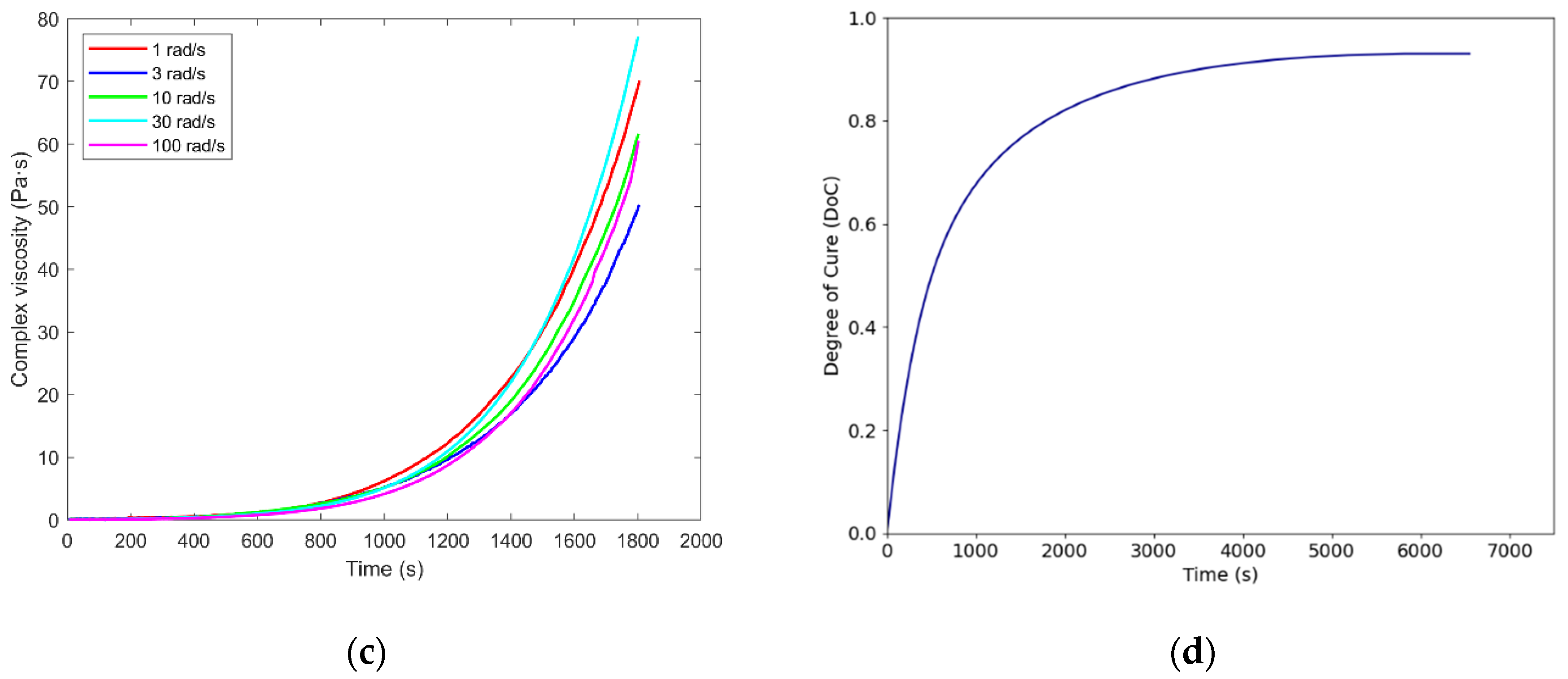
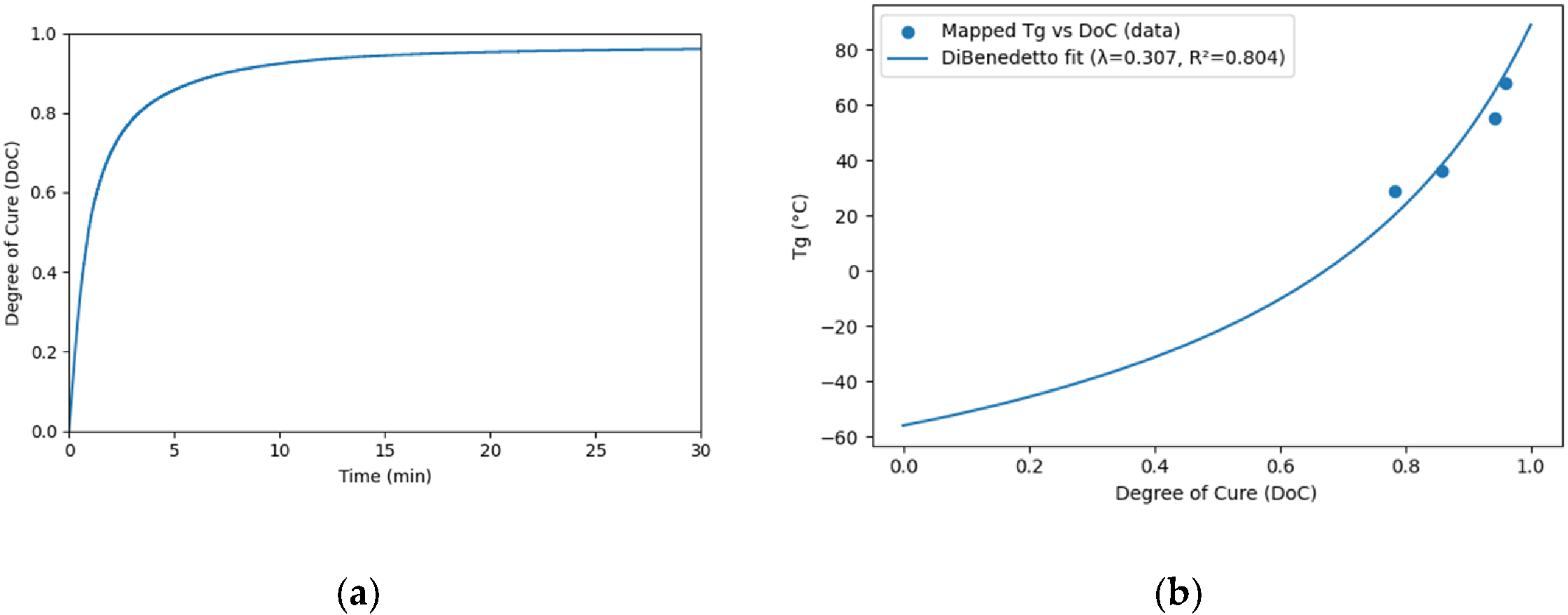
| Tonset (°C) | Tp (°C) | Tendset (°C) | Curing duration (min) | ∆H (J/g) | |
|---|---|---|---|---|---|
| 3 °C/min | 12.70 | 59.12 | 122.81 | 35.12 | 108.34 |
| 5 °C/min | 20.39 | 67.50 | 183.53 | 25.17 | 112.28 |
| 10 °C/min | 40.74 | 84.60 | 169.89 | 13.81 | 149.64 |
| 20 °C/min | 37.37 | 93.80 | 142.42 | 7.40 | 119.63 |
| Resin | A1 | A2 | m | n | Ea (kJ/mol) | R2 |
| PU | 1.002e-2 | 3.516e4 | 0.110 | 1.6384 | 46.12 | 0.97 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




