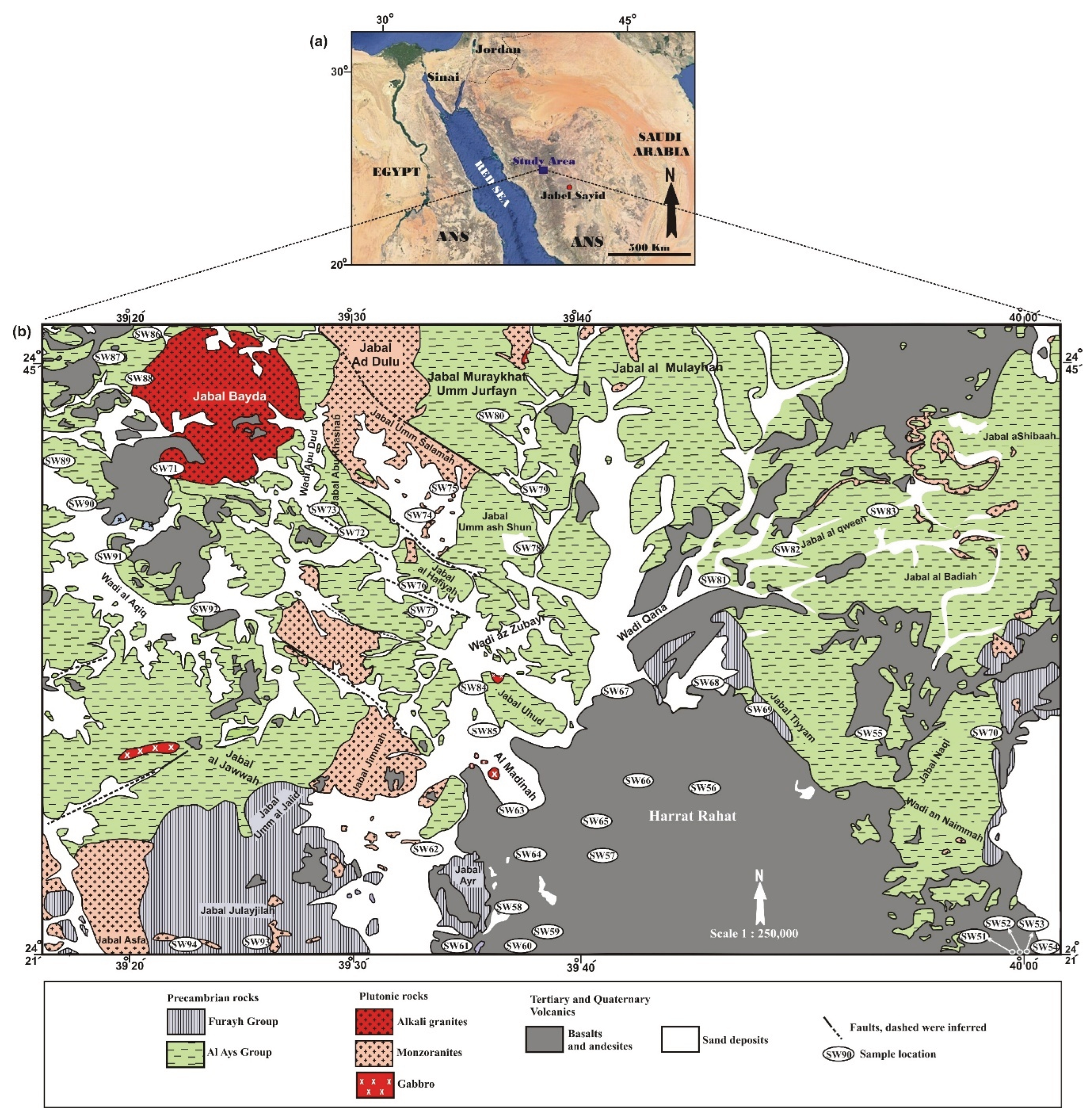
5.1. Physicochemical Parameters and Major Ions Concentrations of Groundwater
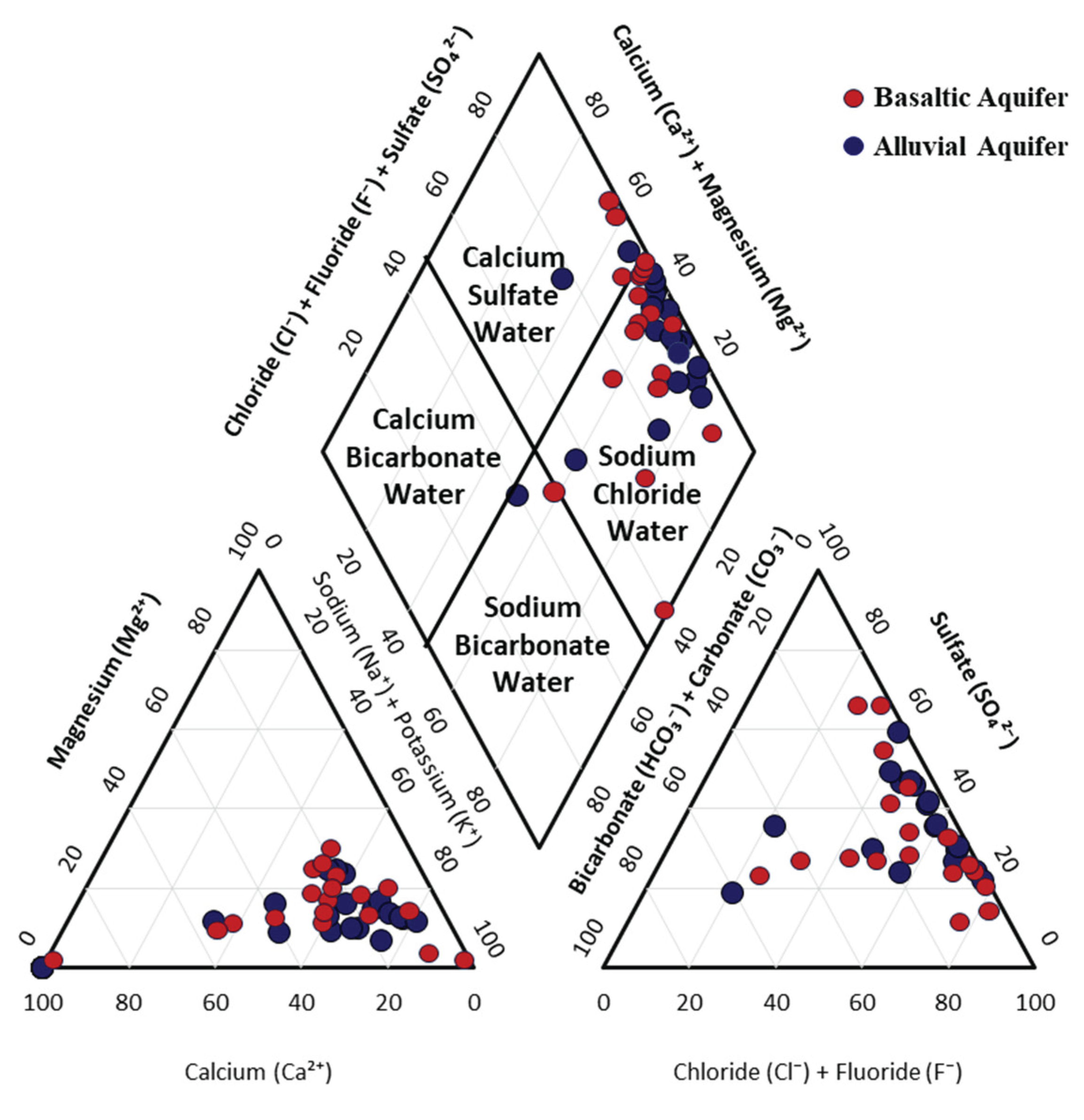
Piper’s trilinear diagram was used to classify the hydrochemical facies of groundwater based on dominant ion composition [
29]. The majority of the analyzed samples are located within the sodium-chloride and calcium-sulfate water types (
Figure 4), indicating a strong influence of evaporite mineral dissolution, particularly halite and gypsum, which contributes to elevated groundwater salinity. The dominance of sodium and chloride ions, together with calcium and sulfate, reflects the high evaporation rates typical of arid regions. The high concentrations of these ions, combined with the limited occurrence of bicarbonate-rich facies, suggests minimal recent freshwater recharge and reduced interaction with carbonate minerals. Overall, the Piper diagram indicates that shallow groundwater in the study area is primarily controlled by evaporation, evaporite dissolution, and ion exchange processes, resulting in chemically evolved water with high salinity. This hydrochemical classification highlights potential limitations for the use of this groundwater for drinking and irrigation purposes.
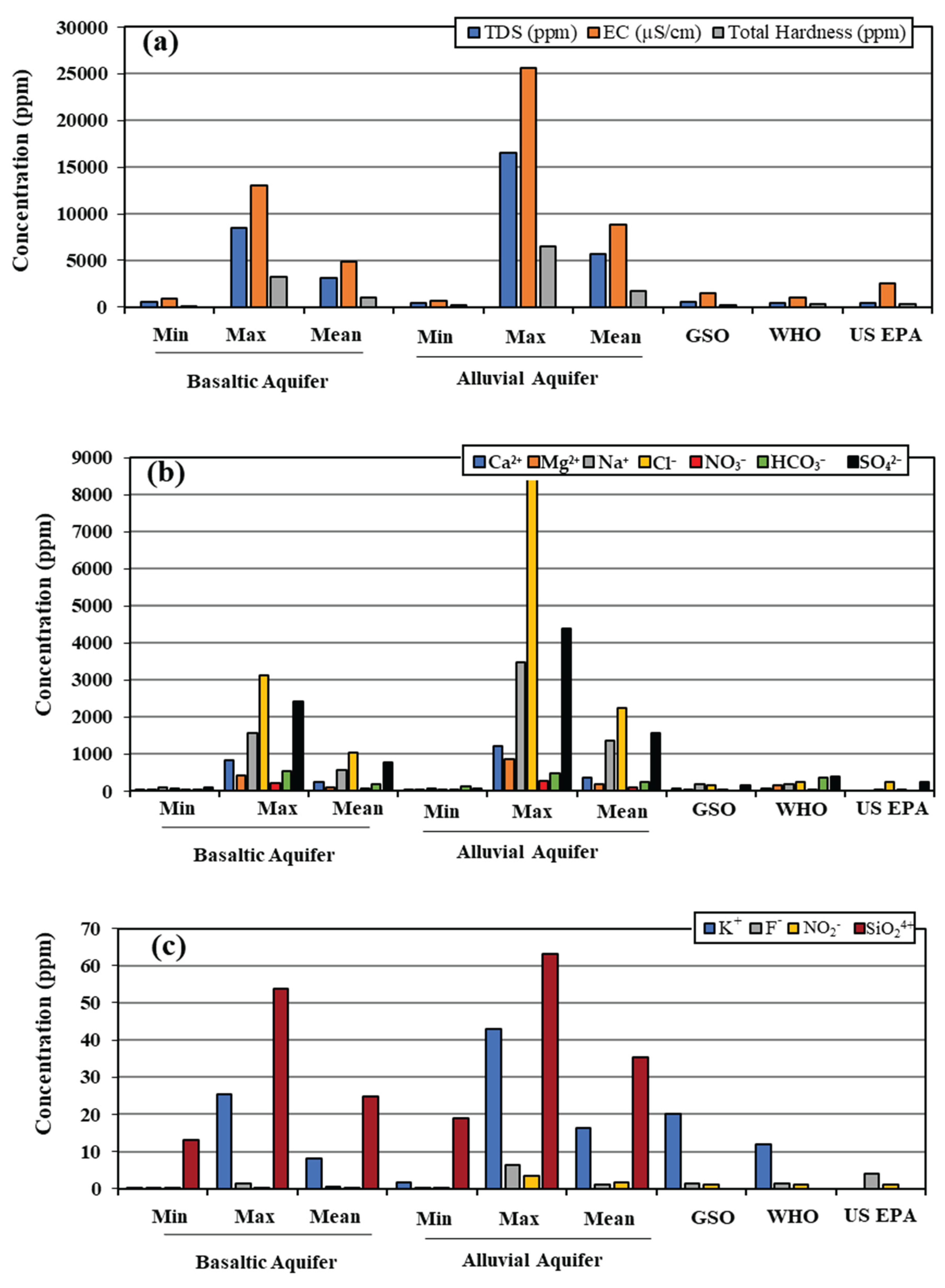
The total dissolved solids (TDS), electrical conductivity (EC), and total hardness (TH) values show a wide range in both basaltic and alluvial aquifers. However, the alluvial aquifer exhibits higher values, exceeding the maximum salinity limits recommended by international standards, including GSO [
30], WHO [
31], and the US EPA [
32] (
Figure 5a). Elevated TDS and EC levels indicate increased salinity, which may adversely affect agricultural activities and reduce the suitability of groundwater for drinking purposes. Such high concentrations may reflect natural geochemical processes, such as mineral leaching in geologically heterogeneous formations, or anthropogenic influences including agricultural runoff, industrial effluents, and urban contamination, ultimately leading to groundwater quality deterioration. Furthermore, elevated TH values typically indicate higher concentrations of calcium and magnesium ions, commonly associated with groundwater interaction with limestone or other mineral-rich lithologies.
The chemical analysis of major ions (Ca
2+, Mg
2+, Na
+, Cl
−, NO
3−, HCO
3−, and SO
42−) (
Table 1;
Figure 5b) indicates that the alluvial aquifer contains significantly higher ion concentrations, exceeding the permissible limits established by GSO, WHO, and the US EPA. In addition, chloride concentrations in both the basaltic and alluvial aquifers exceed international guideline values. Potassium (K
+) and fluoride (F
−) concentrations in both aquifers (
Table 1;
Figure 5c) remain below the maximum limits recommended by GSO. Nitrate (NO
3−) concentrations in the basaltic aquifer fall within the acceptable limits defined by GSO, WHO, and the US EPA, whereas elevated nitrate levels are observed in the alluvial aquifer. The physical parameters of groundwater samples from both aquifers show that pH values fall within the recommended range (6.0–8.5), indicating that the groundwater is generally neutral to slightly alkaline. According to GSO, WHO, and US EPA standards, this pH range suggests that the groundwater is suitable for human consumption. However, several major ions, including Ca
2+, Mg
2+, Na
+, Cl
−, HCO
3−, SO
42−, and NO
3−, exceed the maximum acceptable limits in both aquifers. These elevated concentrations raise concerns regarding groundwater quality and highlight the need for further assessment and management to mitigate potential health risks. It can be concluded that while the pH is acceptable, the elevated levels of specific ions warrant caution regarding the overall safety and suitability of the groundwater for consumption.
Figure 5.
(a) Variation in electrical conductivity (EC), total dissolved solids (TDS), and total hardness (TH) between the basaltic and alluvial aquifers. (b) Variation in major ion concentrations across the two aquifers. (c) Variation in minor ion concentrations in the two aquifers, compared to Gulf and international standards.
Figure 5.
(a) Variation in electrical conductivity (EC), total dissolved solids (TDS), and total hardness (TH) between the basaltic and alluvial aquifers. (b) Variation in major ion concentrations across the two aquifers. (c) Variation in minor ion concentrations in the two aquifers, compared to Gulf and international standards.
Elevated calcium concentrations can adversely affect renal function, leading to hypercalciuria and alkaline urine, which may promote kidney stone formation. Crystalline calcium carbonate can also increase gastric alkalinity and stimulate acidic gastric secretions, potentially counteracting elevated stomach pH. Excess sodium (Na+) in blood plasma may cause dehydration, mild hand tremors, excessive nervousness, involuntary muscle movements, anxiety, impaired concentration, and in severe cases, coma. High chloride (Cl−) concentrations in drinking water can be associated with various health issues, including cardiovascular disorders, respiratory problems, skin and hair conditions, dental weakness, miscarriages, and an increased risk of cancer. Elevated sulfate (SO42−) levels may induce dehydration through repeated diarrhea, particularly in children. Additionally, the activity of sulfate-reducing bacteria can produce hydrogen sulfide gas (H2S), which may cause dizziness, headaches, and, in extreme cases, can be fatal at very high concentrations. If sulfate levels exceed permissible limits, alternative water sources or treatment methods such as distillation or reverse osmosis are recommended. Finally, high nitrate (NO3−) concentrations in drinking water can lead to methemoglobinemia in infants, a condition characterized by cyanosis, and in severe cases may result in brain damage or death due to oxygen deprivation.
5.2. Heavy Metals Concentrations
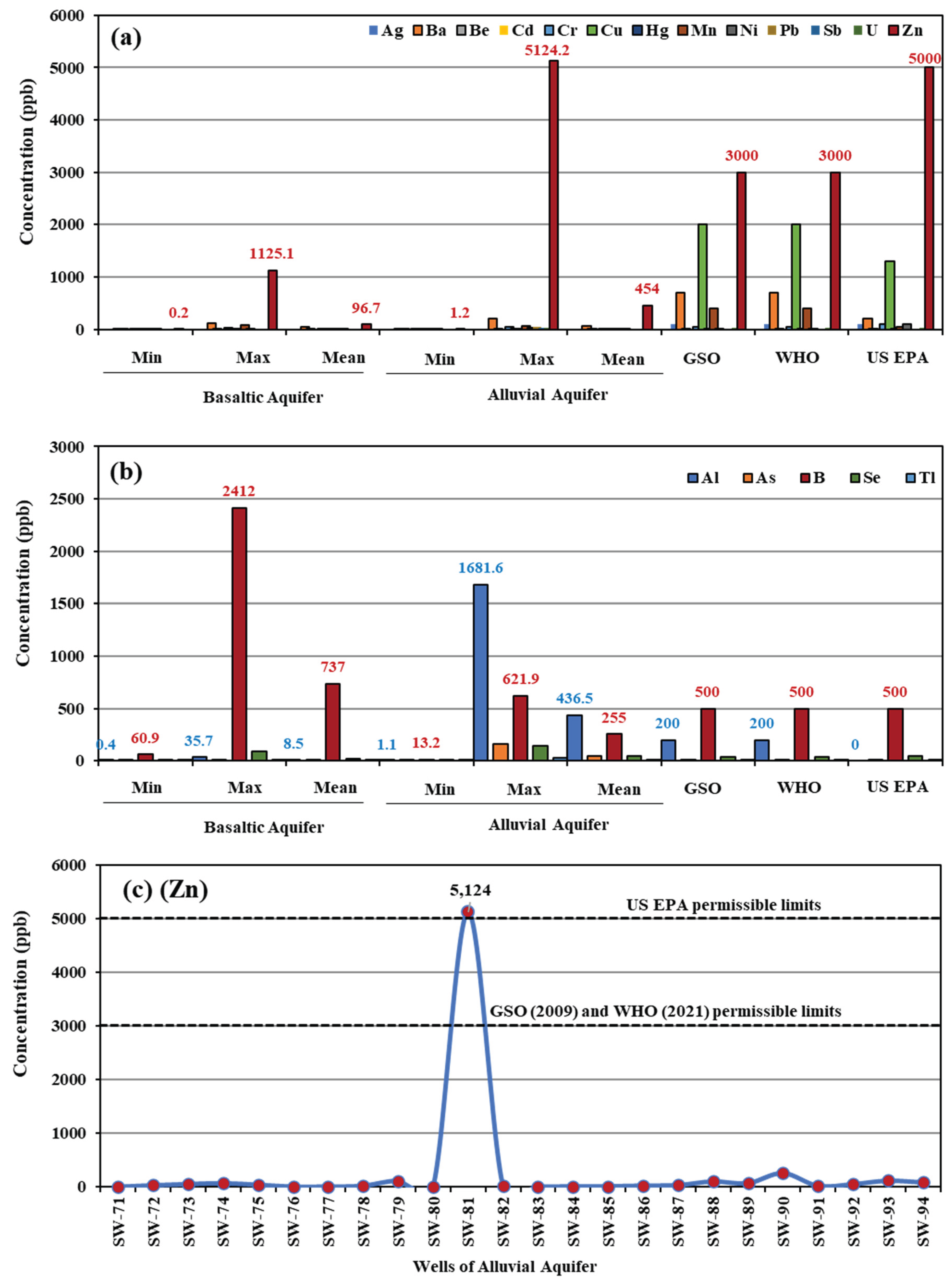
A total of 27 heavy metals (HMs) were analyzed in groundwater, with detailed results presented in the
Supplementary (2).
Table 2 offers a statistical summary of the HM concentrations in both basaltic and alluvial aquifers, compared to the permissible limits established by GSO, WHO, and US EPA.
Figure 6 illustrates the variations in HM concentrations across the two aquifers. Some HM concentrations from the two aquifers, including Ag, Ba, Be, Cd, Cr, Cu, Hg, Mn, Ni, Pb, Sb, U, and Zn, were found to be within safe limits for groundwater use (
Figure 6a). However, certain metals exceeded the permissible limits; for instance, zinc (Zn) in the alluvial well (SW81), surpassed the thresholds set by GSO, WHO, and US EPA (
Figure 6c).
Data in
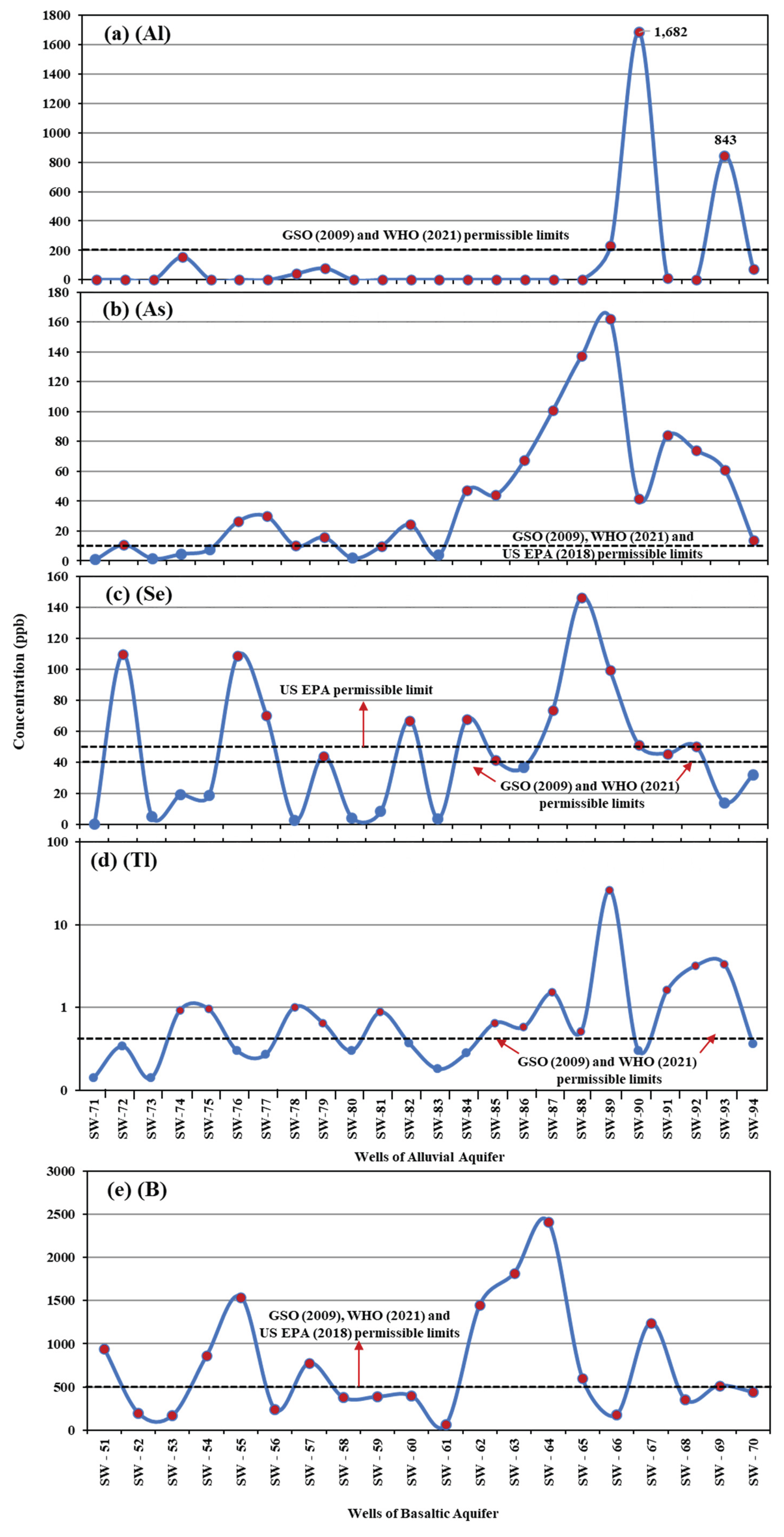
Figure 6b indicate that the alluvial aquifer exhibits relatively higher aluminum (Al) concentrations, particularly in wells SW90 and SW93, which exceed the permissible limits set by GSO and WHO (
Figure 7a). All basaltic wells show arsenic (As) concentrations below the permissible limits established by GSO, WHO and US EPA. Conversely, several alluvial wells display elevated concentrations of arsenic (As), selenium (Se), and thallium (Tl) exceeding the permissible limits of GSO and WHO (
Figure 7b–d), likely due to the interaction of groundwater with various rock and mineral types composing the alluvial deposits, whereas the basaltic aquifer is derived from a more uniform rock type. Furthermore, all alluvial wells report boron (B) levels within the permissible limits of GSO, WHO, and US EPA, while the majority of basaltic wells exceed these international boron limits (
Figure 7e).
Heavy metals (HMs) in the study area can be broadly classified into two categories: 1) Metals with concentrations below permissible limits, including Ag, Ba, Be, Cd, Cr, Cu, Hg, Mn, Ni, Pb, Sb and U; 2) Metals with concentrations exceeding permissible limits in at least one sample, including Zn, Al, As, Se and Tl. Elevated concentrations are primarily attributed to local interactions between water and specific rocks, such as granite and felsite, which are naturally rich in these elements. Rainfall and groundwater percolation facilitate the leaching of these elements into the aquifer. Increases in arsenic and zinc concentrations may also be linked to agricultural activities, as numerous farms in the area use fertilizers; phosphate fertilizers may contain arsenic up to 1200 mg/kg and nitrogenous fertilizers up to 120 mg/kg [
33]. Continuous irrigation processes wash soil particles, increasing the concentration of these metals in groundwater. Many basaltic aquifer wells show elevated boron concentrations, likely resulting from boron released from the Harrat basalts formed during past volcanic activity.
From a health perspective, high arsenic (As) concentrations can cause severe human health issues, affecting the heart, nervous system, skin, lungs, and kidneys. Other organisms interact with arsenic in various ways; some die, while others experience growth difficulties or inability to reproduce. Environmental areas affected by arsenic have shown a marked reduction in biodiversity. High levels of aluminum are linked to neurotoxicity and may contribute to Alzheimer’s disease and other cognitive impairments. Elevated Se concentrations can result in selenosis, with symptoms including gastrointestinal disturbances, hair loss, and neurological problems. Exposure to Tl can result in serious health effects, including hair loss, nerve damage, and potential damage to organs like the kidneys and liver. Elevated boron levels can be toxic to plants, affecting growth and crop yields, particularly in sensitive species.
Table 2.
Statistical summary of heavy metal concentrations in groundwater samples (ppb).
Table 2.
Statistical summary of heavy metal concentrations in groundwater samples (ppb).
Elements |
Basaltic Aquifer
(20 samples) |
Alluvial Aquifer
(24 samples) |
Standards |
| Min |
Max |
Mean |
Min |
Max |
Mean |
GSO
(2009) |
WHO
(2021) |
US EPA
(2018) |
| Ag |
0.2 |
0.5 |
0.3 |
0.1 |
1.2 |
0.4 |
100 |
100 |
100 |
| Al |
0.4 |
36 |
8.5 |
1.1 |
1682 |
437 |
200 |
200 |
* |
| As |
0.4 |
7.5 |
1.7 |
1.7 |
162 |
46 |
10 |
10 |
10 |
| B |
60.9 |
2412 |
737 |
13 |
622 |
255 |
500 |
500 |
500 |
| Ba |
2.3 |
125 |
38 |
11 |
210 |
66 |
700 |
700 |
200 |
| Be |
<0.5 |
<0.5 |
<0.5 |
0.5 |
8.7 |
1.66 |
4 |
4 |
4 |
| Bi |
<0.1 |
<0.1 |
<0.1 |
0.1 |
0.24 |
0.11 |
* |
* |
* |
| Br |
50 |
6887 |
2522 |
231 |
19,687 |
5719 |
* |
* |
* |
| Cd |
0.1 |
0.3 |
0.12 |
0.1 |
1.99 |
0.45 |
3 |
3 |
5 |
| Cr |
0.3 |
33.7 |
7.0 |
0.3 |
39 |
10.8 |
50 |
50 |
100 |
| Cs |
0.1 |
5.96 |
0.83 |
0.1 |
22 |
2.47 |
* |
* |
* |
| Cu |
0.2 |
17.8 |
6.9 |
0.4 |
13 |
5.7 |
2000 |
2000 |
1300 |
| Hg |
0.2 |
7.5 |
1.8 |
0.1 |
4.7 |
1.2 |
6 |
6 |
2 |
| I |
16 |
826 |
236 |
99 |
1455 |
400 |
* |
* |
* |
| Li |
0.4 |
490 |
94 |
1.7 |
251 |
65 |
* |
* |
* |
| Mn |
0.1 |
90.3 |
16 |
0.6 |
65 |
13 |
400 |
400 |
50 |
| Ni |
1.0 |
5.3 |
1.29 |
0.1 |
375 |
5.4 |
20 |
20 |
100 |
| Pb |
<0.1 |
<0.1 |
<0.1 |
0.26 |
40 |
4.8 |
10 |
10 |
15 |
| Rb |
0.1 |
18.7 |
6.4 |
0.3 |
16 |
7.2 |
* |
* |
* |
| Sb |
0.5 |
1.23 |
0.57 |
0.5 |
26 |
3.7 |
5 |
5 |
6 |
| Se |
0.4 |
90.0 |
18.7 |
2.9 |
147 |
51 |
40 |
40 |
50 |
| Sn |
<0.1 |
<0.1 |
<0.1 |
0.1 |
11 |
1.2 |
* |
* |
* |
| Sr |
61 |
18,563 |
5458 |
584 |
25,275 |
7713 |
* |
* |
* |
| Ta |
0.1 |
0.27 |
0.12 |
0.1 |
3.6 |
0.53 |
* |
* |
* |
| Tl |
0.1 |
0.4 |
0.2 |
0.1 |
26 |
2.9 |
0.5 |
0.5 |
0.5 |
| U |
0.2 |
12.4 |
1.9 |
0.6 |
37 |
6.9 |
30 |
30 |
30 |
| Zn |
0.2 |
1125 |
97 |
1.2 |
5124 |
454 |
3000 |
3000 |
5000 |
Figure 6.
(a, b) Variations in heavy metal (HM) concentrations in the two groundwater aquifers. (c) Distribution of zinc concentrations in wells of the alluvial aquifer.
Figure 6.
(a, b) Variations in heavy metal (HM) concentrations in the two groundwater aquifers. (c) Distribution of zinc concentrations in wells of the alluvial aquifer.
Figure 7.
Variations in the concentrations of (a) aluminum, (b) arsenic, (c) selenium, (d) thallium in wells of the alluvial aquifer, and (e) boron in wells of the basaltic aquifer.
Figure 7.
Variations in the concentrations of (a) aluminum, (b) arsenic, (c) selenium, (d) thallium in wells of the alluvial aquifer, and (e) boron in wells of the basaltic aquifer.
5.3. Sources and Processes Affecting Groundwater Quality
5.3.1. Dissolution and Precipitation of Minerals
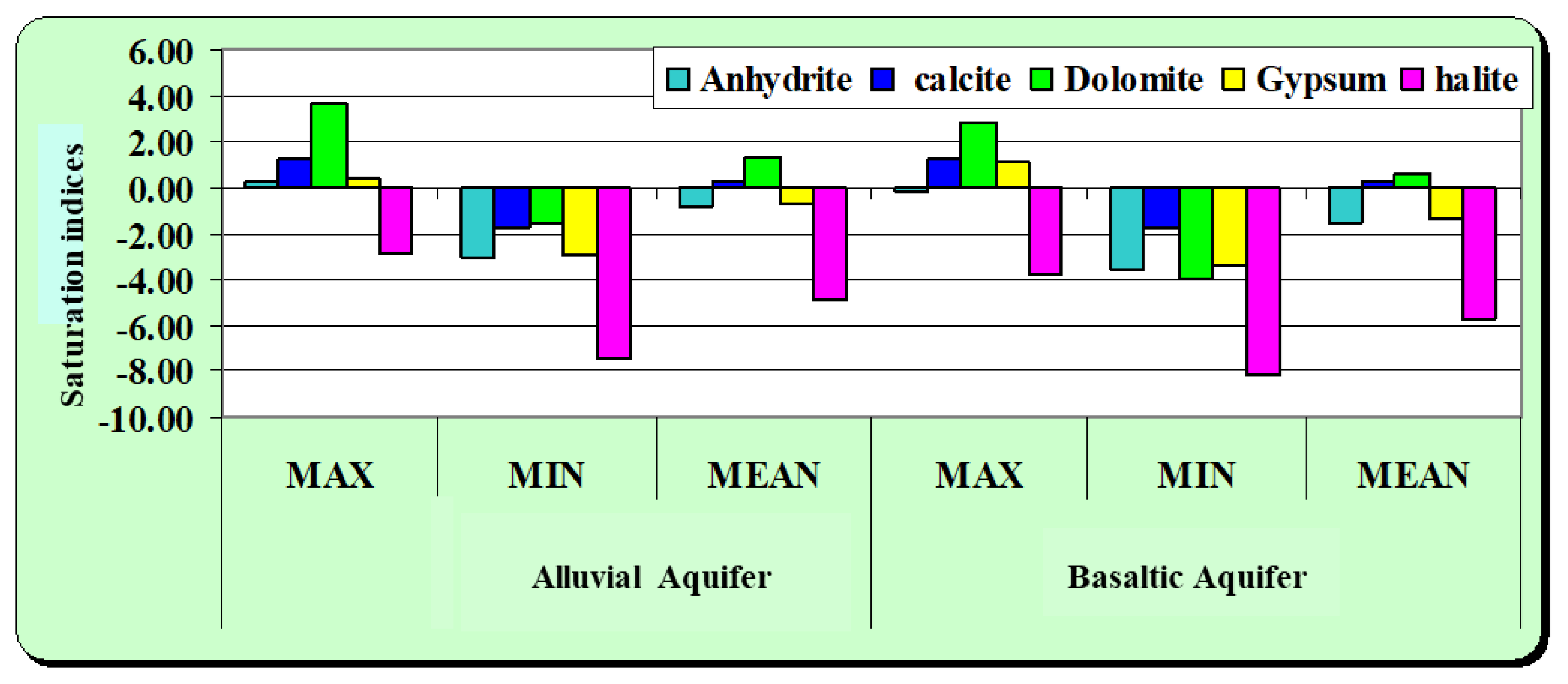
Table 1 shows the saturation indices of anhydrite, gypsum, calcite, dolomite, and halite. These indices indicate that most waters are supersaturated with respect to calcite (CaCO
3) and dolomite [CaMg(CO
3)
2], while only a few waters are supersaturated with gypsum (CaSO
4·2H
2O) and anhydrite (CaSO
4), suggesting a potential for the precipitation of these minerals (
Figure 8). In contrast, the groundwater is undersaturated with respect to halite (NaCl), suggesting that it still has the capacity to dissolve this salt if present. Most data points plot below the zero saturation index line, indicating that groundwater does not significantly precipitate or dissolve these minerals. Therefore, the minerals calcite, dolomite, gypsum, and anhydrite are secondary minerals whose dissolution and precipitation processes influence groundwater chemistry by adding or reducing ions like Ca
2+, Mg
2+, SO
42−, and HCO
3−, alongside contributions from the chemical weathering of silicate minerals, collectively contributing to the overall hydrogeochemical evolution of the groundwater. These saturation index results are consistent with the observed major ion chemistry and confirm that carbonate and sulfate mineral equilibria play a central role in controlling groundwater composition.
5.3.2. Contribution of Nitrate
The main sources of nitrate in groundwater include: (1) application of chemical and animal fertilizers rich in nitrates, (2) atmospheric nitrate deposition due to the oxidation of nitrogen gas by oxygen in the atmosphere, and (3) oxidation of ammonia (NH4) and nitrite (NO2) into nitrate. These oxidation processes can be represented by two equations:
NH4 + 1.5 O2 → NO2 + H2O + 2H+ (1)
NO2 + 0.5 O2 → NO3 (2)
The first source is considered a major contributor to the increase in nitrate content in groundwater owing to continuous use of nitrate-rich chemical fertilizers and animal manure in the agricultural areas of western and northwestern Al Madinah. In contrast, the contributions of sources (2) and (3) are limited due to the generally low concentrations of ammonia and nitrite in groundwater (
Table 1).
Chemical analyses indicate that nitrate concentrations in groundwater from the basaltic aquifer range from 4 to 228 ppm, with an average value of 77 ppm. In the alluvial aquifer, nitrate concentrations range from 4 to 270 ppm, with an average of 99 ppm (
Table 1). These values exceed the maximum permissible limits of 50 ppm set by GSO (2009) and 10 ppm established by WHO and the US EPA.
To assess potential influence of sewage leakage on groundwater nitrate levels, the U.S. Environmental Protection Agency [
34] identifies chloride and nitrate as key indicators of sewage contamination. In the basaltic aquifer, a moderate positive correlation (r = 0.298,
Table 3) suggests a partial contribution from sewage sources. In contrast, the alluvial aquifer exhibits a weak negative correlation (r = -0.023,
Table 4), indicating a limited influence from sewage contamination. On the other hand, moderate to strong significant positive correlations between Cl
− and SO
42− in both basaltic and alluvial aquifers (r = 0.422 and 0.659, respectively) may reflect contributions from sewage effluents, industrial discharges, and natural geochemical processes such as mineral weathering.
Additionally, positive correlations between K+, Na+, Mg2+, and NO3− in the basaltic aquifer suggest the influence of agricultural fertilizers, whereas weaker correlations observed in the alluvial aquifer imply comparatively lower agricultural impacts. The correlations between TDS and various ions (Ca2+, Mg2+, Na+, K+, Cl−, HCO3−, NO3−, SO42−, F−, and TH) provide valuable insights into groundwater geochemical evolution. Elevated TDS values likely result from a combination of mineral dissolution, agricultural runoff, industrial discharges, and possible sewage contamination. Finally, positive correlations between SO42− and Ca2+, Mg2+, Na+, and K+ highlight influences from both natural geological processes and anthropogenic activities.
5.3.3. Rock-Water Interactions
The sources of major and trace elements in groundwater, including heavy metals, are diverse. Rock–water interaction represents a primary mechanism controlling the release of these elements, as rocks undergo prolonged chemical weathering driven by factors such as acidic precipitation, oxygenated surface waters, and chemically active hydrothermal fluids. These processes promote the mobilization of elements from rocks into groundwater and, in some cases, the transfer of elements from water into rocks through adsorption and mineral precipitation. The interaction between rocks and water depends on the physicochemical conditions available at the rock–water interface, such as acidity, temperature, oxygen levels, and the chemical composition of the rocks and groundwater.
In addition to geogenic sources, groundwater chemistry may also be influenced by anthropogenic inputs, primarily associated with industrial, agricultural, and domestic discharges, as well as mining activities and mineral processing. To evaluate the contribution of rock–water interaction as a source of elements in the groundwater of the study area, representative rock samples were collected in the vicinity of selected groundwater wells (
Supplementary 3). These samples were analyzed and compared with the chemical composition of their associated groundwater.
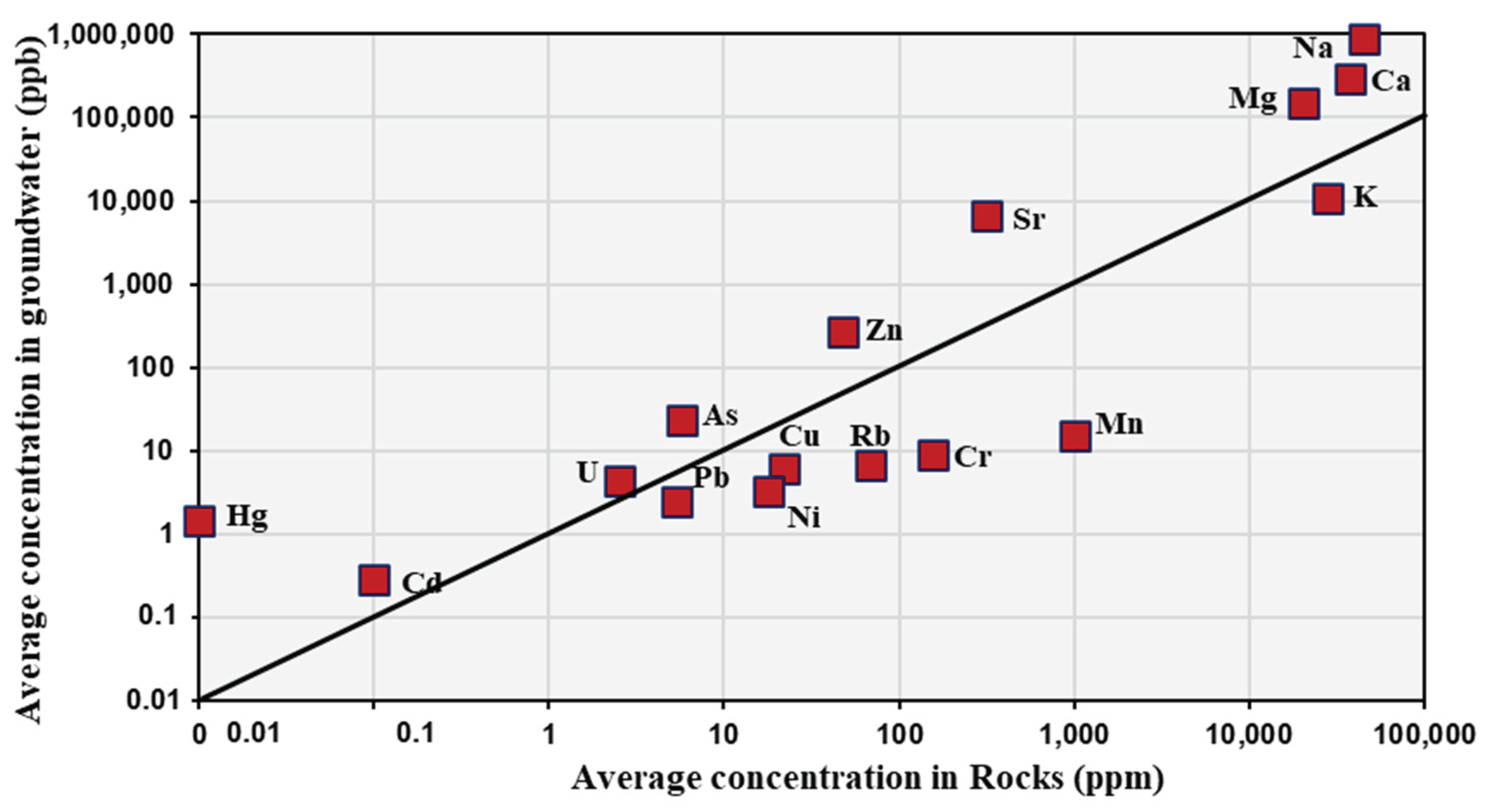
Table 5 shows the average concentrations of selected elements and heavy metals in both rock samples and their associated waters.
Figure 9 illustrates the relationship between these average concentrations. The observed positive correlations indicate that increases in elemental concentrations in the rocks are accompanied by corresponding increases in groundwater, and vice versa. This relationship strongly suggests that chemical weathering of local rocks constitutes the dominant source of dissolved elements in groundwater within the study area. It is noteworthy that if external sources such as wastewater or industrial effluents exerted a significant influence, the elemental relationships would be expected to deviate from this trend, potentially displaying inverse or anomalously elevated concentrations in groundwater relative to the host rock
The chemical evolution of groundwater quality in crystalline igneous rocks is primarily controlled by the chemical weathering of minerals within the rock matrix and is strongly influenced by water acidity and the presence of carbon dioxide gas. Mineral weathering is largely governed by the activity of hydrogen ions (H+), which are generated through the dissociation of carbonic acid (H2CO3), as illustrated by the following reactions:
H2O + CO2(g) ↔ CO2(aq) + H2O
H2O + CO2(aq) ↔ H2CO3
H2CO3 ↔ H+ + HCO3
The hydrogen ions (H+) play a key role in the chemical weathering of silicate minerals by enhancing the ability of groundwater to dissolve and mobilize components from minerals present in the rock matrix and alluvial deposits. In the Harrat Rahat and Al Madinah region, the dominant rock type is olivine basalt, composed mainly of forsterite (Mg2SiO4), diopside (MgCaSi2O6), augite (CaMgFe(SiO3)2), and calcic plagioclase (CaAl2Si3O8), the chemical weathering of silicate minerals such as forsterite, diopside, and augite can be represented by the following equations:
Mg2SiO4 + 4CO2 + 4H2O ⇌ 2Mg2+ + 4HCO3− + H4SiO4
MgCaSi2O6 + 4CO2 + 6H2O ⇌ 2Mg2+ + 2Ca2+ + 4HCO3− + 2H4SiO4
CaMgFe(SiO3)2 + 4CO2 + 6H2O ⇌ Ca2+ + Mg2+ + Fe2+ + 4HCO3− + 2H4SiO4
These reactions demonstrate that the chemical weathering of basaltic minerals releases significant amounts of Ca2+, Mg2+, Fe2+, and HCO3− into groundwater, thereby modifying its chemical composition and influencing overall groundwater quality.
5.4. Conceptual Hydrogeochemical Model
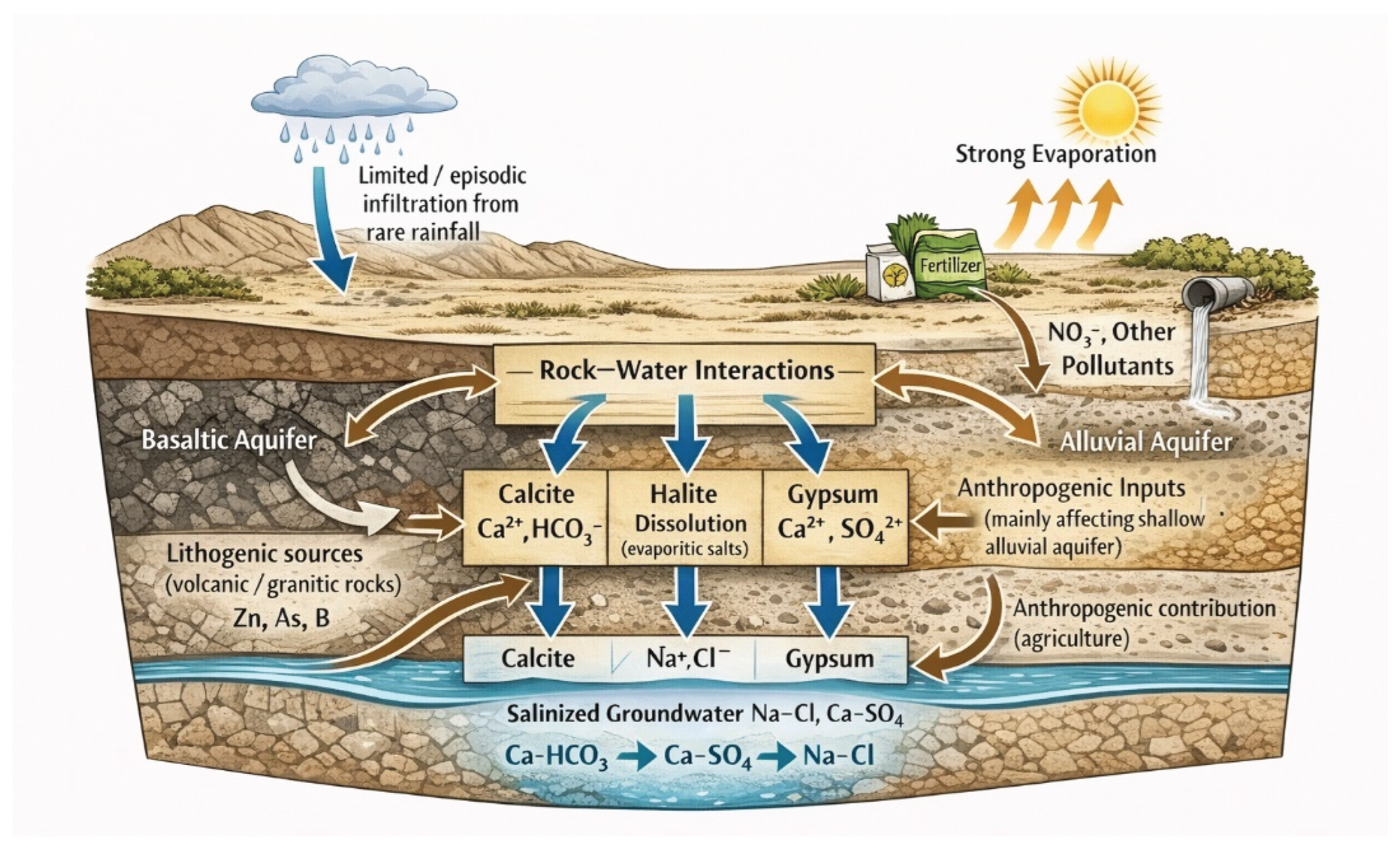
Based on the integration of geological setting, hydrochemical data, saturation index calculations, correlation analysis, and rock–water interaction results, a conceptual hydrogeochemical model is proposed to explain the evolution and quality of groundwater in the Al Madinah region (
Figure 10).
Groundwater recharge in the study area is primarily derived from limited rainfall and surface runoff infiltrating through fractured basaltic lava flows and Quaternary alluvial deposits. The study area is characterized by arid climatic conditions, where rainfall is infrequent, short-lived, and highly episodic. As a result, sustained surface water flow is absent, and runoff events occur only sporadically during rare intense storms, without forming permanent drainage networks. Additionally, the high permeability of fractured basaltic lava flows and coarse alluvial deposits promotes rapid infiltration of precipitation, limiting surface runoff and enhancing diffuse recharge mechanisms. Consequently, groundwater recharge is dominated by limited, episodic infiltration rather than continuous surface water flow, which explains the absence of surface hydrological features and reinforces the dominance of subsurface-controlled hydrogeochemical processes in the study area. In the basaltic aquifer, groundwater movement is mainly controlled by vesicles, fractures, weathered zones, and interflow boundaries between successive lava flows. In contrast, groundwater within the alluvial aquifer flows through interbedded layers of sand, gravel, and clay, resulting in longer residence times and enhanced hydrogeochemical evolution.
As recharge water percolates through the subsurface, intensive rock–water interactions occur. The chemical weathering of silicate minerals within basaltic rocks contributes Ca
2+, Mg
2+, Na
+, HCO
3−, and SiO
24+ to groundwater. However, the hydrochemical composition is strongly governed by carbonate and sulfate mineral equilibria, which play a central role in controlling groundwater chemistry. Saturation index calculations indicate that groundwater is predominantly supersaturated with respect to calcite and dolomite, suggesting active precipitation and buffering of Ca
2+, Mg
2+, and HCO
3− concentrations. In contrast, partial saturation or near-equilibrium conditions with respect to gypsum and anhydrite indicate ongoing dissolution–precipitation processes that regulate sulfate and calcium levels in the groundwater system. The hydrochemical evolution from Ca–HCO
3 to Ca–SO
4 and finally to Na–Cl facies reflect increasing groundwater residence time, enhanced rock–water interaction, and evaporation effects, consistent with Piper diagram interpretations (
Figure 10).
Groundwater remains undersaturated with respect to halite, indicating continued dissolution potential and explaining the widespread enrichment of Na+ and Cl− ions, particularly in the alluvial aquifer. These processes, combined with strong evaporation under arid climatic conditions, lead to progressive salinization and the development of chemically evolved groundwater dominated by sodium–chloride and calcium–sulfate facies.
Anthropogenic inputs further modify groundwater chemistry, especially in agricultural areas. Elevated concentrations of nitrate, potassium, zinc, and arsenic reflect the influence of fertilizer application, irrigation return flow, and localized sewage leakage. These impacts are more pronounced in the alluvial aquifer due to its higher permeability, longer groundwater residence time, and greater exposure to surface-derived contaminants.
Heavy metal concentrations are largely controlled by lithological composition and mineral dissolution, particularly from granitic, felsitic, and volcanic rocks, with additional contributions from agricultural activities. Volcanic rocks in the Harrat region contribute to elevated boron levels in the basaltic aquifer, while alluvial sediments facilitate the accumulation of trace metals through adsorption and prolonged water–sediment interaction.
Overall, the conceptual model illustrates that groundwater quality in the Harrat Rahat–Al Madinah region results from the combined effects of climate-driven evaporation, carbonate and sulfate mineral equilibria, silicate weathering, and anthropogenic activities. These interacting processes explain the observed spatial variations in groundwater chemistry and provide a robust framework for understanding groundwater evolution, vulnerability, and management challenges in arid volcanic terrains.
Figure 10.
Conceptual hydrogeochemical model illustrating evolution and quality of groundwater in the Harrat Rahat–Al Madinah region.
Figure 10.
Conceptual hydrogeochemical model illustrating evolution and quality of groundwater in the Harrat Rahat–Al Madinah region.