Submitted:
29 January 2026
Posted:
30 January 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Results
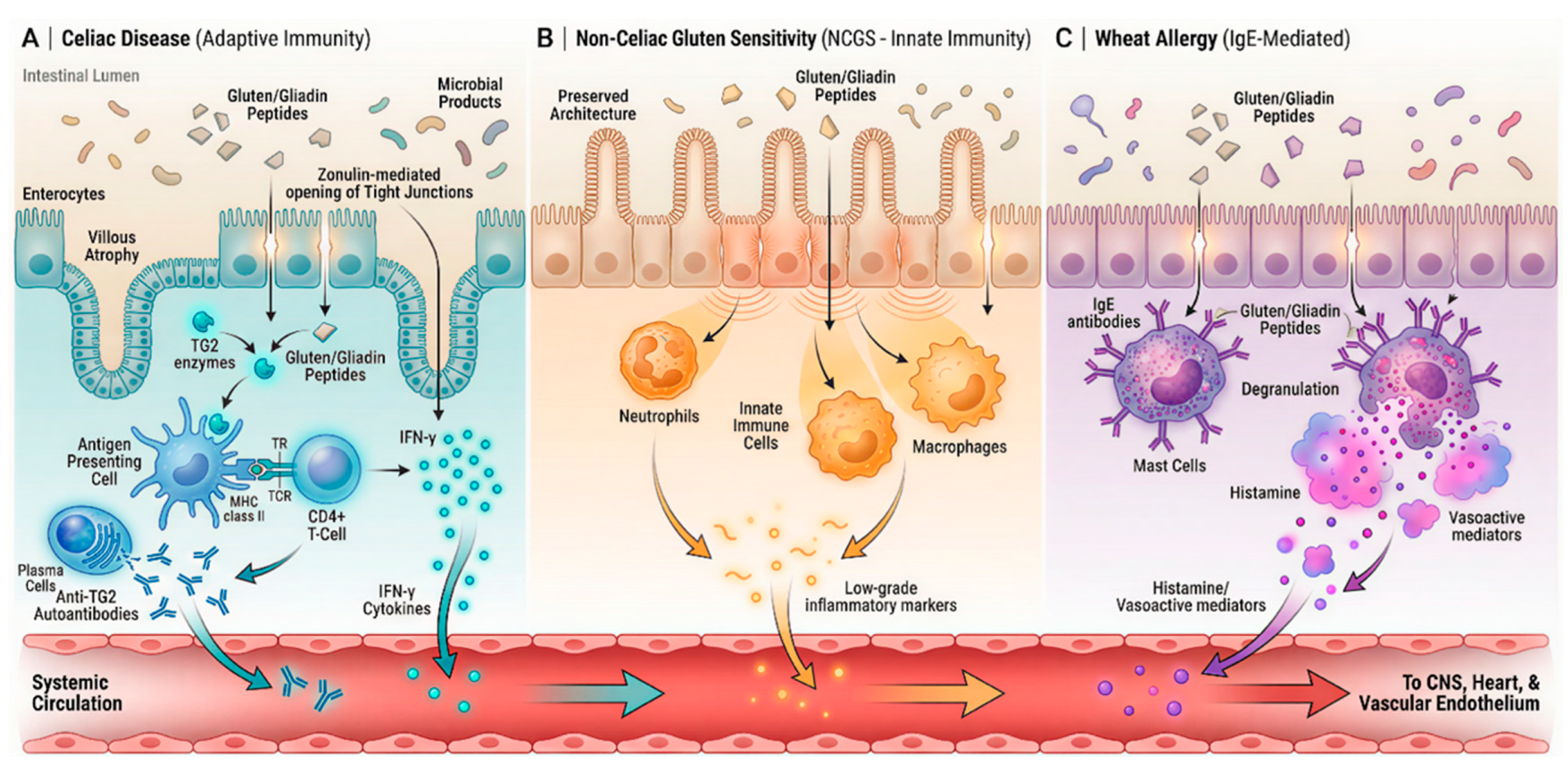
Immuno-Pathophysiology of Gluten-Related Disorders
- Adaptive and innate immune activation
- 2.
- Intestinal barrier dysfunction and systemic immune signaling
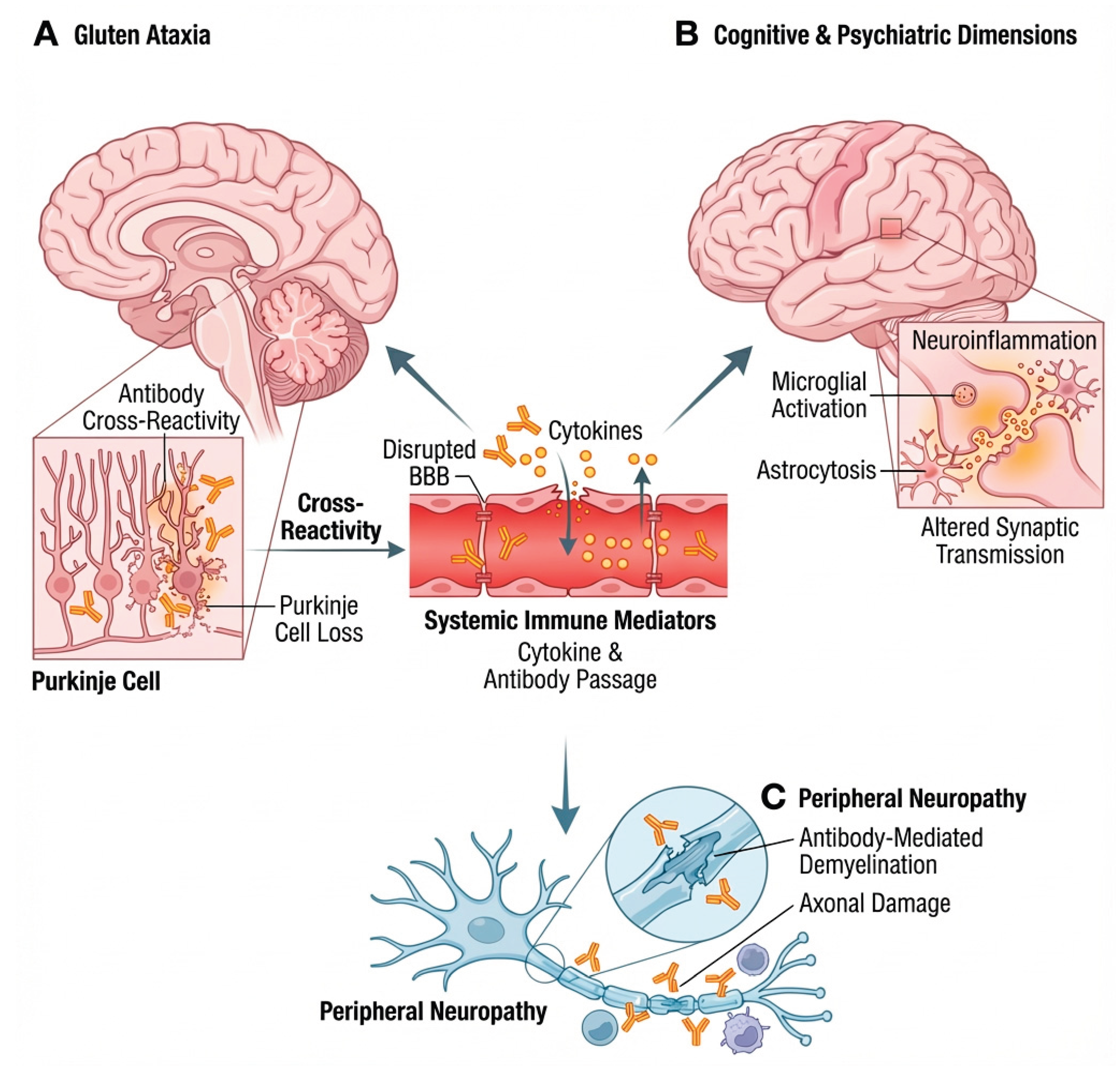
Central Nervous System Involvement in Gluten-Related Disorders
- Neurological manifestations
- 2.
- Psychiatric and affective dimensions
- 3.
- Blood–brain barrier dysfunction and neuroimmune interactions
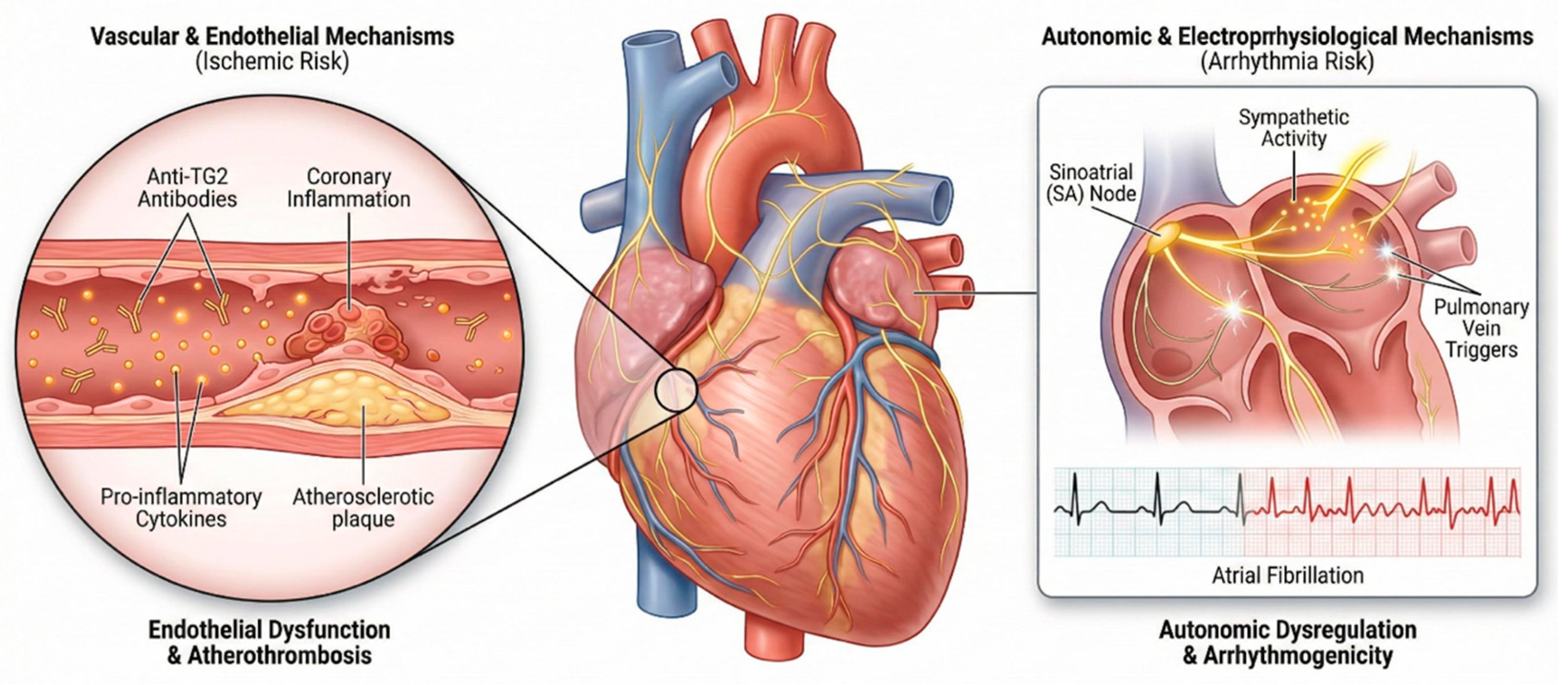
Cardiovascular Involvement in GRDs
- Epidemiological associations
- 2.
- Inflammation, endothelial dysfunction, and thrombosis
- 3.
- Autonomic dysregulation and cardiac electrophysiology
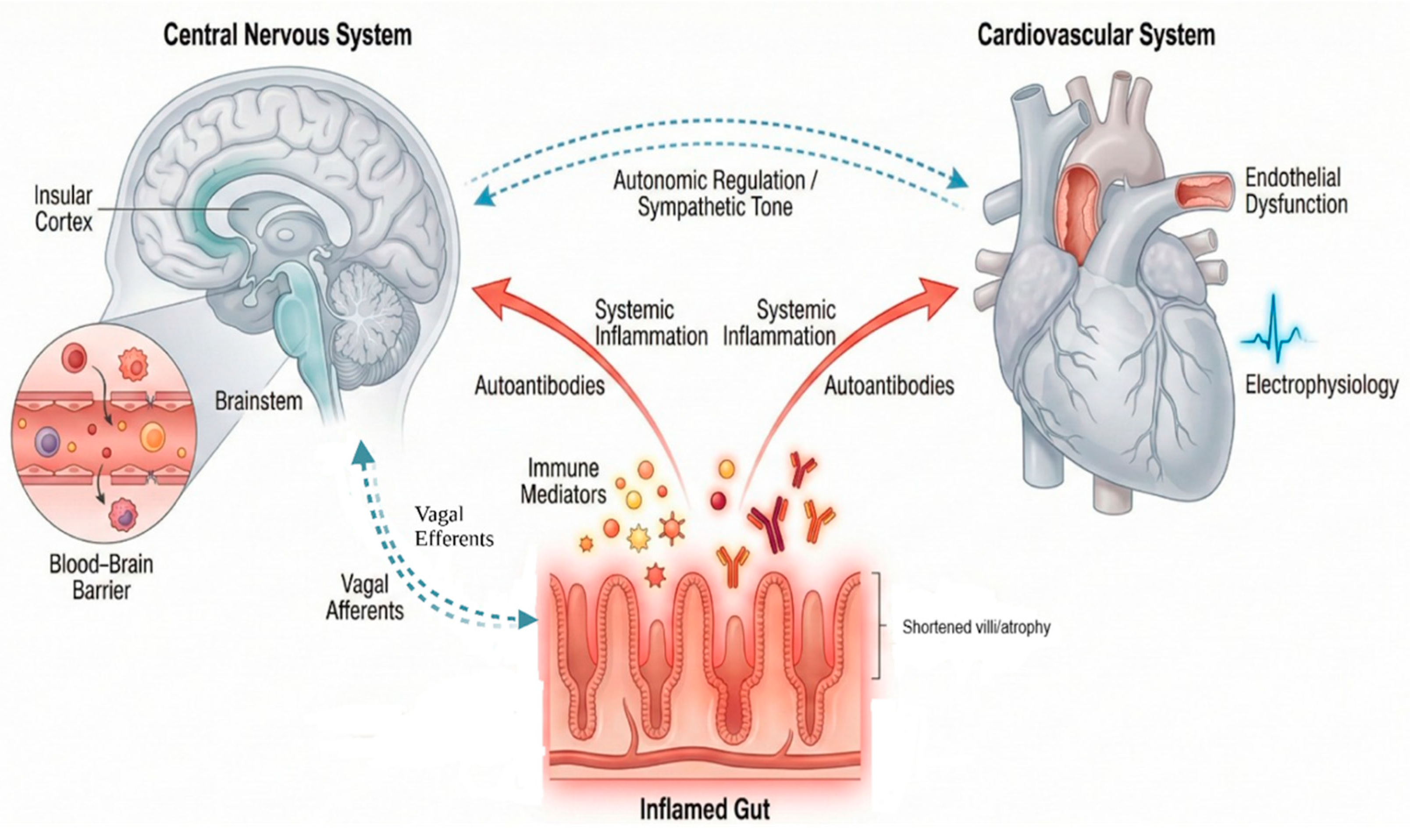
The Gut-Brain-Heart Axis in GRDs
Therapeutic Implications and Reversibility of Brain-Heart Effects
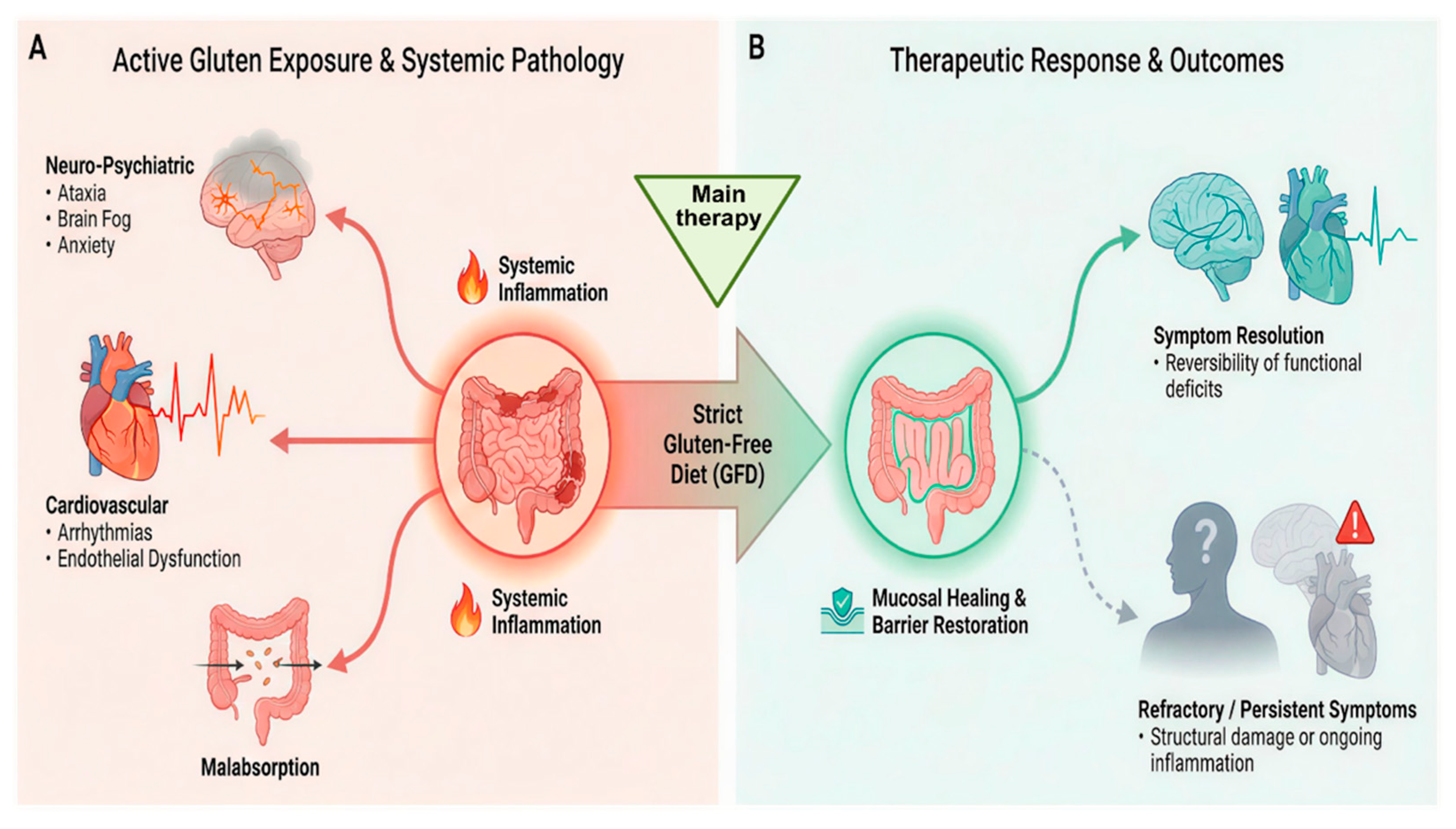
Knowledge Gaps and Future Directions
Limitations
Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BBB | Blood-brain barrier |
| CD4 | The cluster of differentiation marker CD4 |
| GFD | Gluten free diet |
| GRDs | Gluten-related disorders |
| HLA-DQ2 | Human leukocyte antigen (HLA) class II molecule (DQ2) |
| HLA-DQ8 | Human leukocyte antigen (HLA) class II molecule (DQ8) |
| HLA | Human leukocyte antigen |
| IgE | Immunoglobulin E |
| SANRA | Scale for the assessment of narrative review articles |
| Th1 | T helper 1 cells |
| TG2 | Tissue transglutaminase 2 |
References
- Taraghikhah, N; Ashtari, S; Asri, N; Shahbazkhani, B; Al-Dulaimi, D; Rostami-Nejad, M; Rezaei-Tavirani, M; Razzaghi, MR; Zali, MR. An updated overview of spectrum of gluten-related disorders: clinical and diagnostic aspects. BMC Gastroenterol 2020, 20(1), 258. [Google Scholar] [CrossRef] [PubMed]
- Giuffrè, M; Gazzin, S; Zoratti, C; Llido, JP; Lanza, G; Tiribelli, C; Moretti, R. Celiac Disease and Neurological Manifestations: From Gluten to Neuroinflammation. Int J Mol Sci. 2022, 23(24), 15564. [Google Scholar] [CrossRef]
- Singh, P; Arora, A; Strand, TA; Leffler, DA; Catassi, C; Green, PH; Kelly, CP; Ahuja, V; Makharia, GK. Global Prevalence of Celiac Disease: Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol 2018, 16(6), 823–836.e2. [Google Scholar] [CrossRef]
- Santonicola, A; Wieser, H; Gizzi, C; Soldaini, C; Ciacci, C. Associations between Celiac Disease, Extra-Gastrointestinal Manifestations, and Gluten-Free Diet: A Narrative Overview. Nutrients 2024, 16(12), 1814. [Google Scholar] [CrossRef] [PubMed]
- Rodrigo, L. Celiac Disease and Associated Extraintestinal Manifestations, with Special Reference to Neurological Disorders. OBM Neurobiology 2020, 4(4), 074. [Google Scholar] [CrossRef]
- Eng, S; Gabr, A; Raghav, S; Frishman, WH; Aronow, WS. The Gut-Heart Connection: Unraveling Cardiovascular Risks in Celiac Disease. Cardiol Rev 2025. [Google Scholar] [CrossRef] [PubMed]
- Baethge, C; Goldbeck-Wood, S; Mertens, S. SANRA-a scale for the quality assessment of narrative review articles. Res Integr Peer Rev 2019, 4, 5. [Google Scholar] [CrossRef]
- Aboulaghras, S; Piancatelli, D; Taghzouti, K; Balahbib, A; Alshahrani, MM; Al Awadh, AA; Goh, KW; Ming, LC; Bouyahya, A; Oumhani, K. Meta-Analysis and Systematic Review of HLA DQ2/DQ8 in Adults with Celiac Disease. Int J Mol Sci. 2023, 24(2), 1188. [Google Scholar] [CrossRef]
- Sollid, LM. Coeliac disease: dissecting a complex inflammatory disorder. Nat Rev Immunol 2002, 2(9), 647–55. [Google Scholar] [CrossRef]
- Sapone, A; Bai, JC; Ciacci, C; Dolinsek, J; Green, PH; Hadjivassiliou, M; Kaukinen, K; Rostami, K; Sanders, DS; Schumann, M; Ullrich, R; Villalta, D; Volta, U; Catassi, C; Fasano, A. Spectrum of gluten-related disorders: consensus on new nomenclature and classification. BMC Med. 2012, 10, 13. [Google Scholar] [CrossRef]
- Sicherer, SH; Sampson, HA. Food allergy: Epidemiology, pathogenesis, diagnosis, and treatment. J Allergy Clin Immunol. 2014, 133(2), 291–307; quiz 308. [Google Scholar] [CrossRef]
- Fasano, A. Zonulin and its regulation of intestinal barrier function: the biological door to inflammation, autoimmunity, and cancer. Physiol Rev. 2011, 91(1), 151–75. [Google Scholar] [CrossRef]
- Voisine, J; Abadie, V. Interplay Between Gluten, HLA, Innate and Adaptive Immunity Orchestrates the Development of Coeliac Disease. Front Immunol 2021, 12, 674313. [Google Scholar] [CrossRef]
- Serena, G; D’Avino, P; Fasano, A. Celiac Disease and Non-celiac Wheat Sensitivity: State of Art of Non-dietary Therapies. Front Nutr 2020, 7, 152. [Google Scholar] [CrossRef]
- Patel, N; Samant, H. Wheat Allergy. In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), 25 Jun 2023. [Google Scholar] [PubMed]
- Fasano, A. All disease begins in the (leaky) gut: role of zonulin-mediated gut permeability in the pathogenesis of some chronic inflammatory diseases. F1000Res 2020, 9, F1000 Faculty Rev–69. [Google Scholar] [CrossRef]
- Varrassi, G; Leoni, MLG; Farì, G; Al-Alwany, AA; Al-Sharie, S; Fornasari, D. Neuromodulatory Signaling in Chronic Pain Patients: A Narrative Review. Cells 2025, 14(17), 1320. [Google Scholar] [CrossRef] [PubMed]
- Leoni, MLG; Mercieri, M; Viswanath, O; Cascella, M; Rekatsina, M; Pasqualucci, A; Caruso, A; Varrassi, G. Neuropathic Pain: A Comprehensive Bibliometric Analysis of Research Trends, Contributions, and Future Directions. Curr Pain Headache Rep. 2025, 29(1), 73. [Google Scholar] [CrossRef]
- Therrien, A; Kelly, CP; Silvester, JA. Celiac Disease: Extraintestinal Manifestations and Associated Conditions. J Clin Gastroenterol. 2020, 54(1), 8–21. [Google Scholar] [CrossRef] [PubMed]
- Hadjivassiliou, M; Sanders, DD; Aeschlimann, DP. Gluten-related disorders: gluten ataxia. Dig Dis. 2015, 33(2), 264–268. [Google Scholar] [CrossRef]
- Yelland, GW. Gluten-induced cognitive impairment (“brain fog”) in coeliac disease. J Gastroenterol Hepatol. 2017, 32 Suppl 1, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Addolorato, G; Leggio, L; D’Angelo, C; Mirijello, A; Ferrulli, A; Cardone, S; Vonghia, L; Abenavoli, L; Leso, V; Nesci, A; Piano, S; Capristo, E; Gasbarrini, G. Affective and psychiatric disorders in celiac disease. Dig Dis. 2008, 26(2), 140–8. [Google Scholar] [CrossRef]
- Hadjivassiliou, M; Sanders, DS; Grünewald, RA; Woodroofe, N; Boscolo, S; Aeschlimann, D. Gluten sensitivity: from gut to brain. Lancet Neurol 2010, 9(3), 318–30. [Google Scholar] [CrossRef]
- Huang, J. Assessment of the causal association between celiac disease and cardiovascular diseases. Front Cardiovasc Med 2022, 9, 1017209. [Google Scholar] [CrossRef]
- Ludvigsson, JF; James, S; Askling, J; Stenestrand, U; Ingelsson, E. Nationwide cohort study of risk of ischemic heart disease in patients with celiac disease. Circulation 2011, 123(5), 483–90. [Google Scholar] [CrossRef]
- Emilsson, L; Smith, JG; West, J; Melander, O; Ludvigsson, JF. Increased risk of atrial fibrillation in patients with coeliac disease: a nationwide cohort study. Eur Heart J 2011, 32(19), 2430–7. [Google Scholar] [CrossRef] [PubMed]
- Batty, M; Bennett, MR; Yu, E. The Role of Oxidative Stress in Atherosclerosis. Cells 2022, 11(23), 3843. [Google Scholar] [CrossRef] [PubMed]
- Hadithi, M; Mulder, CJ; Stam, F; et al. Effect of B vitamin supplementation on plasma homocysteine levels in celiac disease. World J Gastroenterol. 2009, 15(8), 955–960. [Google Scholar] [CrossRef] [PubMed]
- Furgala, A; Przybylska-Felus, M; Ciesielczyk, K; Gil, K; Zwolinska-Wcislo, M. Effects of water ingestion on autonomic activity and gastric motility in patients with celiac disease. J Physiol Pharmacol. 2023, 74(2). [Google Scholar] [CrossRef]
- Giorgetti, GM; Tursi, A; Iani, C; Arciprete, F; Brandimarte, G; Capria, A; Fontana, L. Assessment of autonomic function in untreated adult coeliac disease. World J Gastroenterol 2004, 10(18), 2715–8. [Google Scholar] [CrossRef]
- Calvillo, L; Vanoli, E; Ferrara, F; Caradonna, E. Interplay Among Gut Microbiota-Derived TMAO, Autonomic Nervous System Dysfunction, and Heart Failure Progression. Int J Mol Sci 2025, 27(1), 203. [Google Scholar] [CrossRef]
- Banks, WA. Blood-brain barrier transport of cytokines: a mechanism for neuropathology. Curr Pharm Des. 2005, 11(8), 973–84. [Google Scholar] [CrossRef]
- Inayat, N; Zahir, A; Hashmat, AJ; Khan, A; Ahmad, A; Sikander, M; Zakir, S; Ahmad, S; Awan, SK; Raza, SS; Varrassi, G. Gut Microbiota as a Key Modulator of Chronic Disease: Implications for Diabetes, Autoimmunity, and Cancer. Cureus 2025, 17(5), e84687. [Google Scholar] [CrossRef]
- Wacklin, P; Laurikka, P; Lindfors, K; Collin, P; Salmi, T; Lähdeaho, ML; Saavalainen, P; Mäki, M; Mättö, J; Kurppa, K; Kaukinen, K. Altered duodenal microbiota composition in celiac disease patients suffering from persistent symptoms on a long-term gluten-free diet. Am J Gastroenterol 2014, 109(12), 1933–41. [Google Scholar] [CrossRef]
- Benarroch, EE. The central autonomic network: functional organization, dysfunction, and perspective. Mayo Clin Proc. 1993, 68(10), 988–1001. [Google Scholar] [CrossRef]
- Varrassi, G.; Paladini, A.; Tran, Y. V.; Pham, V. P.; Al Alwany, A. A.; Farì, G.; Caruso, A.; Mercieri, M.; Pergolizzi, J. V.; Kaye, A. D.; Breve, F.; Corriero, A.; Gharibo, C.; Leoni, M. L. G. Advances in the Pathophysiology and Management of Cancer Pain: A Scoping Review. Cancers 2026, 18(2), 259. [Google Scholar] [CrossRef] [PubMed]
- Thayer, JF; Lane, RD. A model of neurovisceral integration in emotion regulation and dysregulation. J Affect Disord. 2000, 61(3), 201–16. [Google Scholar] [CrossRef] [PubMed]
- Tracey, KJ. The inflammatory reflex. Nature 2002, 420(6917), 853–9. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B; Rubio-Tapia, A. Epidemiology, Presentation, and Diagnosis of Celiac Disease. Gastroenterology 2021, 160(1), 63–75. [Google Scholar] [CrossRef]
- Hall, NJ; Rubin, G; Charnock, A. Systematic review: adherence to a gluten-free diet in adult patients with coeliac disease. Aliment Pharmacol Ther. 2009, 30(4), 315–330. [Google Scholar] [CrossRef]
- Varrassi, G; Cereda, E; Ruoppolo, GA; Mercieri, M; Muscaritoli, M. The drug called food and its role as a therapy for chronic patients: a comprehensive narrative review. Adv Health Res. cited. 2024, 1 1(1). [Google Scholar] [CrossRef]
- Schmucker, C; Eisele-Metzger, A; Meerpohl, JJ; Lehane, C; Kuellenberg de Gaudry, D; Lohner, S; Schwingshackl, L. Effects of a gluten-reduced or gluten-free diet for the primary prevention of cardiovascular disease. Cochrane Database Syst Rev 2022, 2(2), CD013556. [Google Scholar] [CrossRef] [PubMed]
- Rubio-Tapia, A; Hill, ID; Semrad, C; Kelly, CP; Greer, KB; Limketkai, BN; Lebwohl, B. American College of Gastroenterology Guidelines Update: Diagnosis and Management of Celiac Disease. Am J Gastroenterol 2023, 118(1), 59–76. [Google Scholar] [CrossRef] [PubMed]
| Topics | Sub-topics | References | Results |
| Immuno-pathophysiology of gluten-related disorders |
|
[8,9,10,11,12,13,14,15,16] | Celiac disease: autoimmune gluten response affecting gut, nerves, heart. Non-celiac sensitivity/allergy: innate/IgE pathways causing systemic low-level inflammation. |
| Central nervous system involvement in gluten-related disorders |
|
[17,18,19,20,21,22,23] | Gluten-induced autoimmunity can cause cerebellar ataxia, peripheral neuropathy, epilepsy, headaches and cognitive/psychiatric symptoms through blood-brain-barrier disruption and anti-tTG antibodies, even without gut disease. |
| Cardiovascular involvement in gluten-related disorders |
|
[24,25,26,27,28,29,30] | Studies link celiac disease to higher ischemic heart disease, atrial fibrillation and stroke risk, independent of traditional factors, likely via inflammation, endothelial dysfunction and autonomic dysregulation; non-celiac evidence remains preliminary. |
| The gut-brain-heart axis in gluten-related disorders | [31,32,33,34,35,36,37,38] | The gut-brain-heart axis describes bidirectional immune and neural communication from gut inflammation to brain and heart, influencing autonomic control and symptoms like fatigue, palpitations. | |
| Therapeutic implications and reversibility of brain-heart effects | [39,40,41,42,43] | Gluten-free diet is main therapy; adherence restores barrier, reduces inflammation and extra-intestinal symptoms; however, some patients need adjunctive measures. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




