Submitted:
28 January 2026
Posted:
28 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethical Approval
2.2. Participant Selection
2.3. Data Collection and Measurement Techniques
2.4. Statistical Analysis
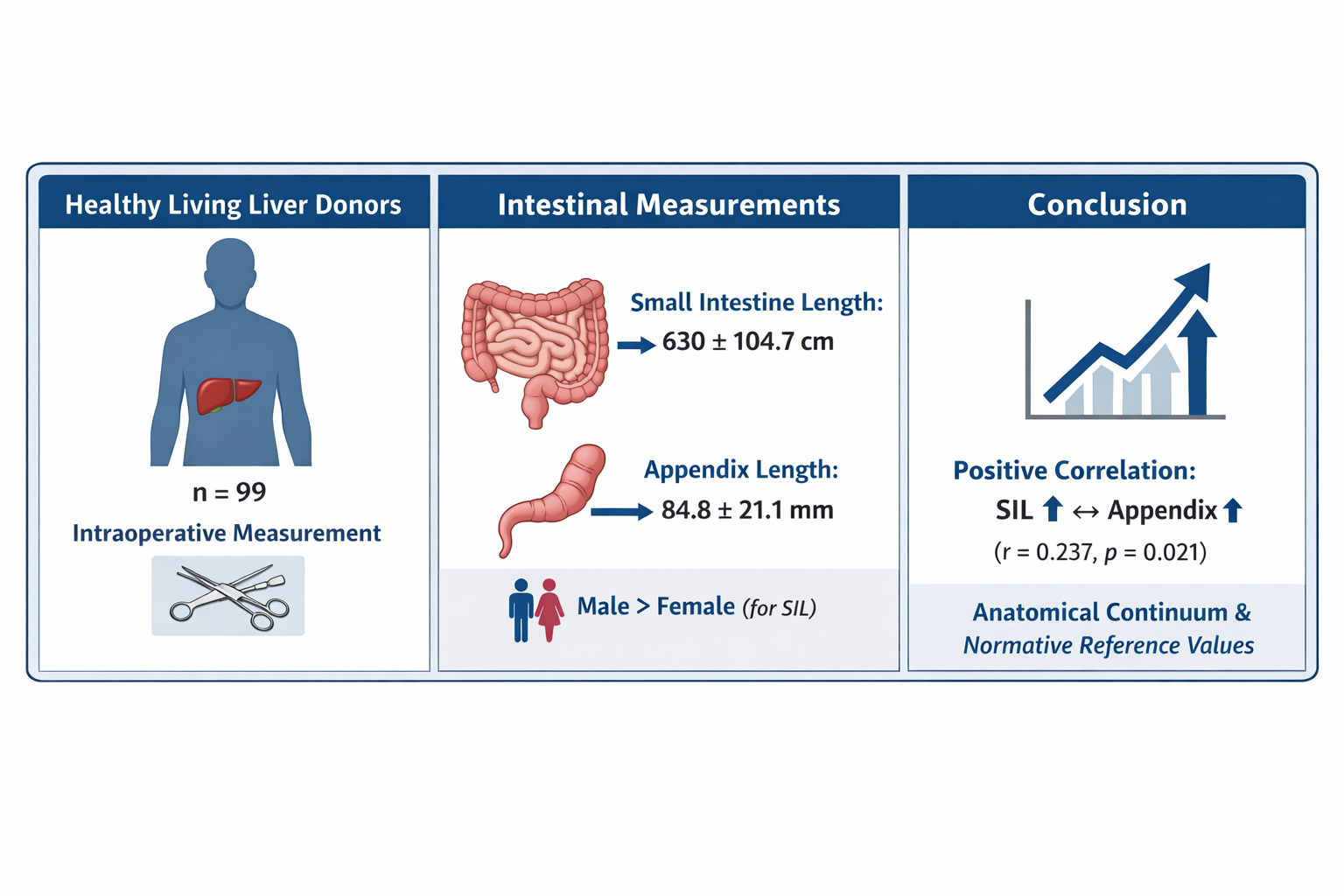
3. Results
3.1. Demographic and Anthropometric Characteristics
3.2. Small Intestine and Appendix Morphometry
3.3. Geographical Distribution of the Cohort
3.4. Anatomical Variations
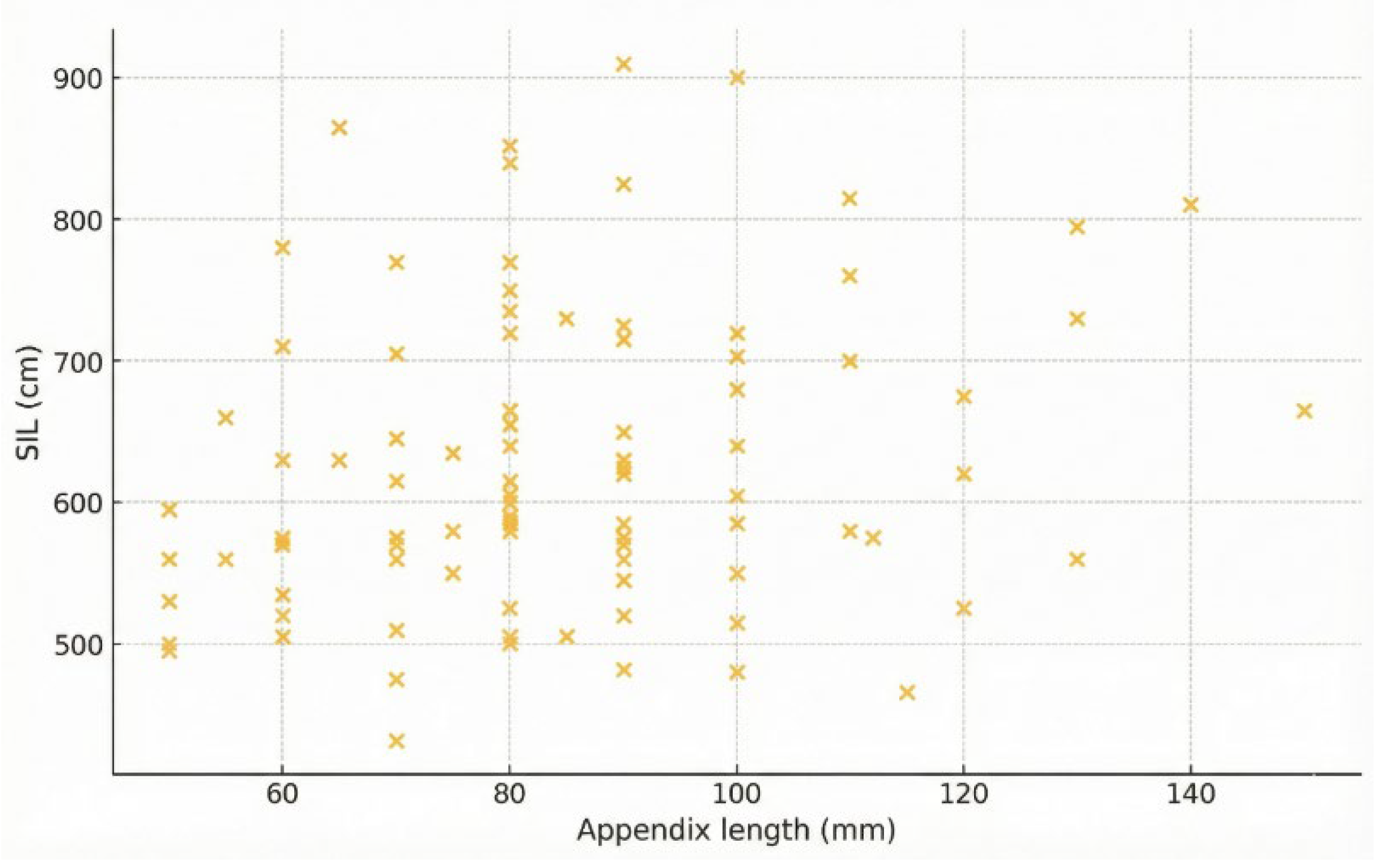
3.5. Correlation Analysis
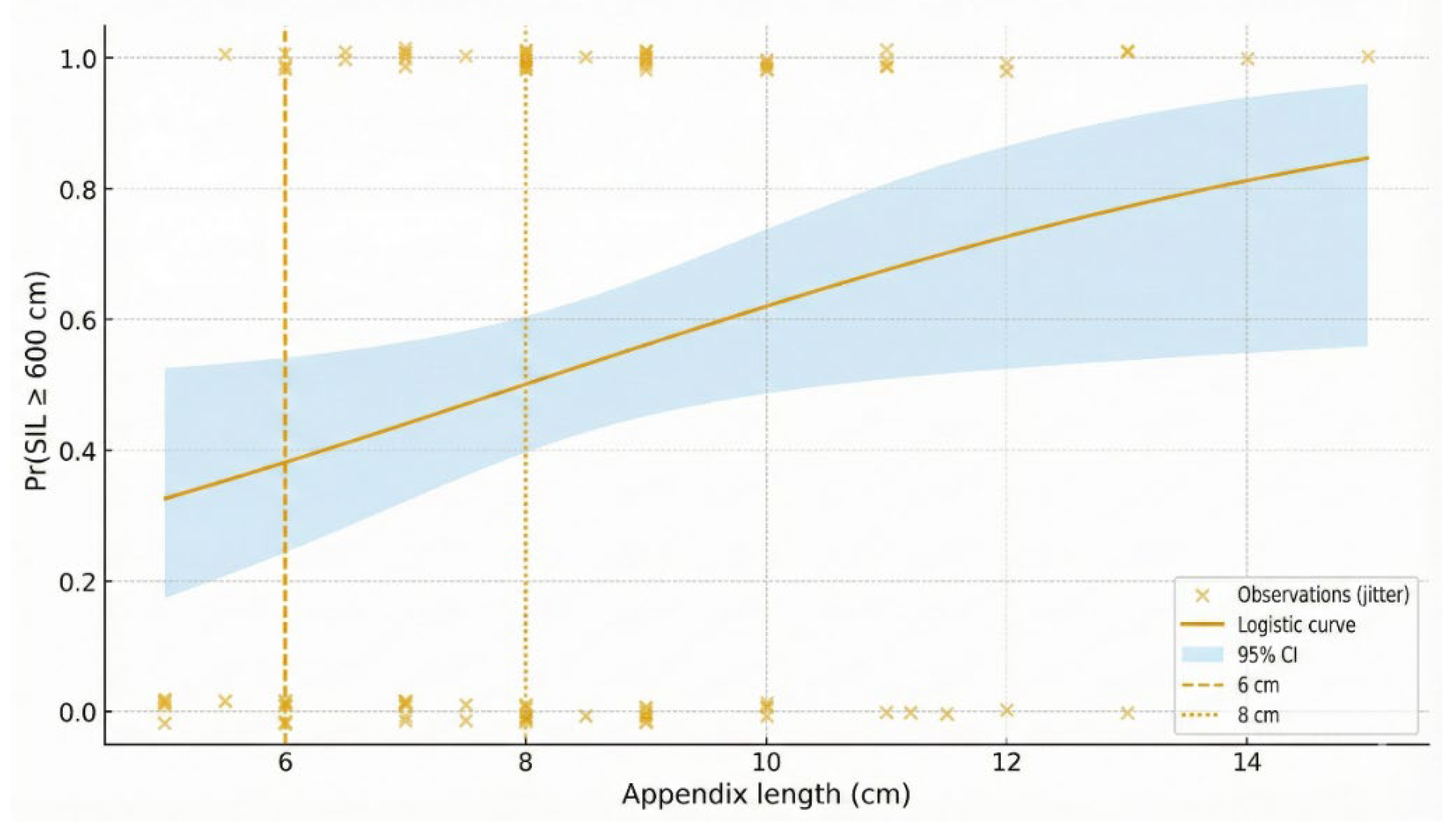
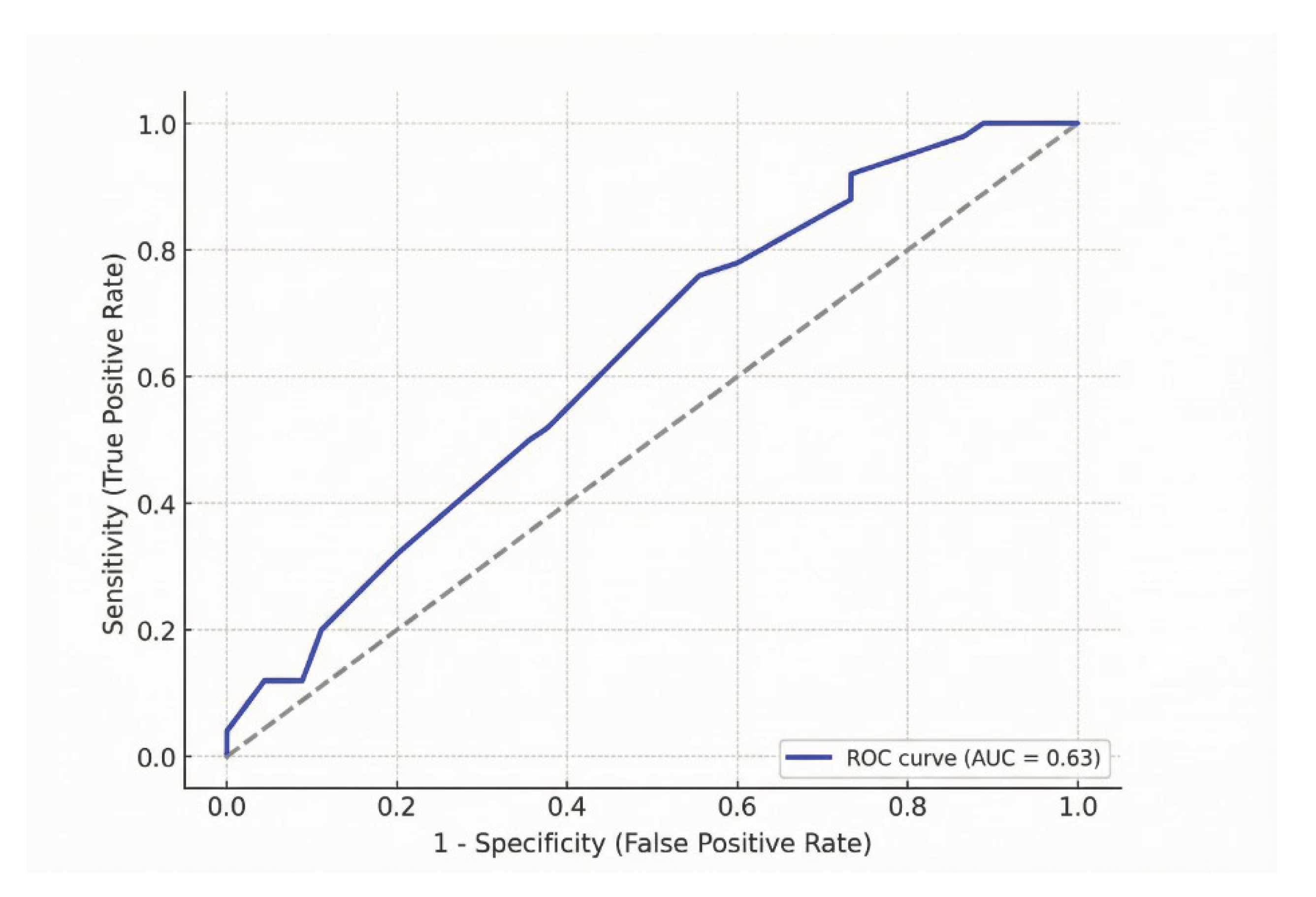
3.6. Predictive Value of Appendix Length
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hounnou, G.; Destrieux, C.; Desmé, J.; Bertrand, P.; Velut, S. Anatomical study of the length of the human intestine. Surg. Radiol. Anat. 2002, 24, 290–294. [Google Scholar] [CrossRef]
- Underhill, B.M. Intestinal length in man. Br. Med. J. 1955, 2, 1243–1246. [Google Scholar] [CrossRef]
- Parrish, C.R. The clinician’s guide to short bowel syndrome. Gut 2005, 54, iv1–iv12. [Google Scholar]
- Mahawar, K.K.; Kumar, P.; Parmar, C.; Graham, Y.; Carr, W.R.; Jennings, N.; Schroeder, N.; Small, P.K. Small bowel limb lengths and Roux-en-Y gastric bypass: a systematic review. Obes. Surg. 2016, 26, 660–671. [Google Scholar] [CrossRef]
- Felsenreich, D.M.; Langer, F.B.; Eichelter, J.; Jedamzik, J.; Kristo, I.; Prager, G. Bariatric surgery—how much malabsorption do we need? A review of various limb lengths in different gastric bypass procedures. J. Clin. Med. 2021, 10, 674. [Google Scholar] [CrossRef]
- Akbulut, S.; Tas, M.; Sogutcu, N.; Arikanoglu, Z.; Basbug, M.; Ulku, A.; Yagmur, Y. Unusual histopathological findings in appendectomy specimens: a retrospective analysis and literature review. World J. Gastroenterol. 2011, 17, 1961–1970. [Google Scholar] [CrossRef] [PubMed]
- Mowat, A.M.; Agace, W.W. Regional specialization within the intestinal immune system. Nat. Rev. Immunol. 2014, 14, 667–685. [Google Scholar] [CrossRef]
- Girard-Madoux, M.J.H.; Gómez de Agüero, M.; Ganal-Vonarburg, S.C.; Mooser, C.; Belz, G.T.; Macpherson, A.J.; Vivier, E. The immunological functions of the appendix: An example of redundancy? Semin. Immunol. 2018, 36, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Vitetta, L.; Chen, J.; Clarke, S. The vermiform appendix: an immunological organ sustaining a microbiome inoculum. Clin. Sci. 2019, 133, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gondolesi, G.; Ramisch, D.; Padin, J.; Rumbo, C.; Alvarez, G.; Pekolj, J. What is the normal small bowel length in humans? First donor-based cohort analysis. Am. J. Transplant. 2012, 12, S49–S54. [Google Scholar] [CrossRef]
- Shatari, T.; Clark, M.A.; Yamane, T.; Engel, A.F. Reliability of radiographic measurement of small intestinal length. Colorectal Dis. 2004, 6, 327–329. [Google Scholar] [CrossRef] [PubMed]
- Muise, E.D.; Tackett, J.J.; Callender, K.A.; et al. Accurate assessment of bowel length: the method of measurement matters. J. Surg. Res. 2016, 206, 146–150. [Google Scholar] [CrossRef]
- Karagül, S.; Kayaalp, C.; Kirmizi, S.; Tamer, D.; Ersan, V. Influence of repeated measurements on small bowel length. SpringerPlus 2016, 5, 1649. [Google Scholar] [CrossRef] [PubMed]
- Karagül, S.; Kayaalp, C. Repeated stretched or non-stretched small bowel length measurements in healthy individuals. Turk. J. Surg. 2019, 35, 1–5. [Google Scholar] [CrossRef]
- Hosseinpour, M.; Behdad, A. Evaluation of small bowel measurement in alive patients. Surg. Radiol. Anat. 2008, 30, 653–655. [Google Scholar] [CrossRef]
- Teitelbaum, E.N.; Vaziri, K.; Zettervall, S.; Amdur, R.L.; Orkin, B.A. Intraoperative small bowel length measurements and analysis of demographic predictors of increased length. Clin. Anat. 2013, 26, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Sayadishahraki, M.; Mahmoudieh, M.; Keleidari, B.; Jamshidi, K.; Shahraki, A.D. Using anthropometric and demographic factors to predict small bowel length to prevent malnutrition in bariatric surgery. Adv. Biomed. Res. 2023, 12, 209. [Google Scholar] [CrossRef]
- Kacprzyk, A.; Droś, J.; Stefura, T.; Krzysztofik, M.; Jasińska, K.; Pędzisz, P.; Mizia, E.; Major, P. Variations and morphometric features of the vermiform appendix: a systematic review and meta-analysis of 114,080 subjects with clinical implications. Clin. Anat. 2020, 33, 85–98. [Google Scholar] [CrossRef]
- Raschka, S.; Raschka, C. On the relationship between body dimensions and appendix length. Anthropol. Anz. 2008, 66, 67–72. [Google Scholar] [CrossRef]
- Sachdev, R.; Ackerman, N.B. Comparison of changes in intestinal length and circumference after 90 per cent bypass and 90 per cent resection. Am. J. Surg. 1979, 137, 208–212. [Google Scholar] [CrossRef]
- Wakeley, C.P.G. The position of the vermiform appendix as ascertained by an analysis of 10,000 cases. J. Anat. 1933, 67, 277–283. [Google Scholar]
- Ghorbani, A.; Forouzesh, M.; Kazemifar, A.M. Variation in anatomical position of vermiform appendix among Iranian population: an old issue which has not lost its importance. Anat. Res. Int. 2014, 2014, 313575. [Google Scholar] [CrossRef]
- Hsieh, C.H.; Wang, Y.C.; Yang, H.R.; Chung, P.K.; Jeng, L.B.; Chen, R.J. Retroperitoneal abscess resulting from perforated acute appendicitis: analysis of its management and outcome. Surg. Today 2007, 37, 762–767. [Google Scholar] [CrossRef] [PubMed]
- Sagar, J.; Kumar, V.; Shah, D.K. Meckel’s diverticulum: a systematic review. J. R. Soc. Med. 2006, 99, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Kuru, S.; Kismet, K. Meckel’s diverticulum: clinical features, diagnosis and management. Rev. Esp. Enferm. Dig. 2018, 110, 726–732. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Mean ± SD / n (%) | Range (min-max) |
|---|---|---|
| Age (years) | 33.6 ± 9.6 | 18–55 |
| Sex | Female 46 (46.5%) Male 53 (53.5%) |
– |
| Height (cm) | 170.7 ± 9.6 | 147–192 |
| Weight (kg) | 70.1 ± 12.8 | 46–106 |
| BMI (kg/m²) | 24.0 ± 3.5 | 16.5–34.4 |
| Parameter | Entire Cohort | Female | Male | p value* |
|---|---|---|---|---|
| SIL (cm) | 630.0 ± 104.7 | 601.5 ± 91.1 | 654.8 ± 110.2 | 0.011 |
| Appendix (mm) | 84.8 ± 21.1 | 82.8 ± 21.4 | 86.5 ± 21.0 | 0.389 |
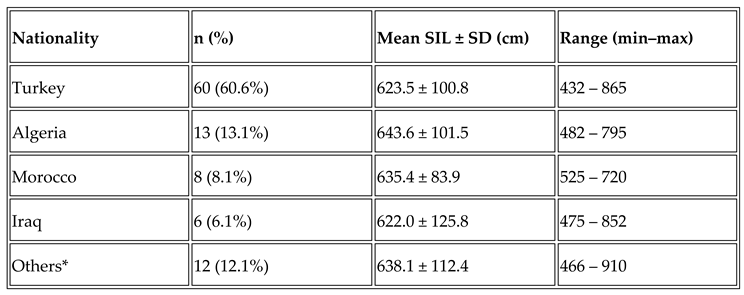 |
| Parameter | Height | BMI | Age |
|---|---|---|---|
| SIL | 0.256 (p=0.011) | 0.261 (p=0.009) | -0.009 (p=0.927) |
| Appendix | 0.214 (p=0.037) | 0.292 (p=0.004) | -0.072 (p=0.486) |
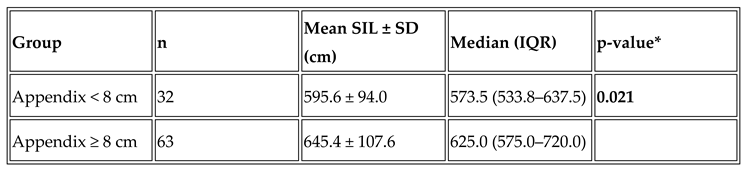 |
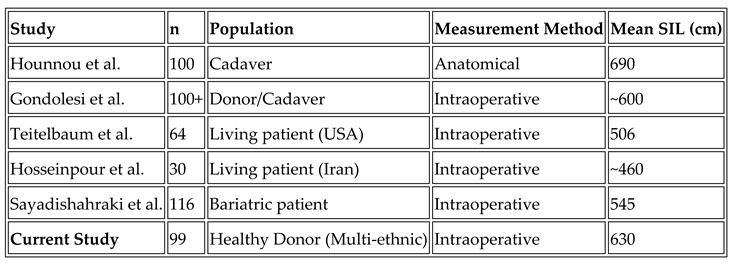 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




