Submitted:
27 January 2026
Posted:
28 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
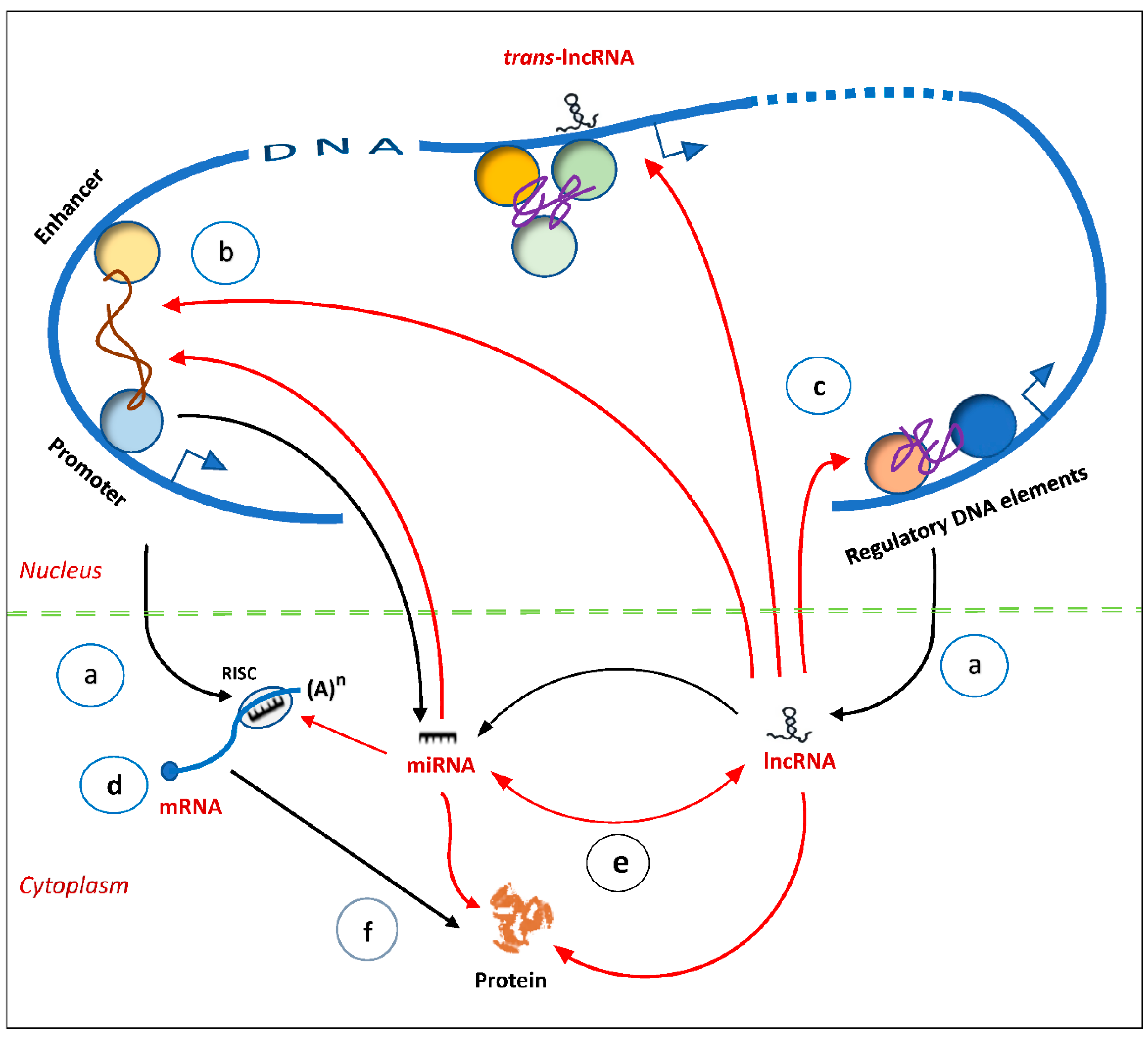
2. Non-Coding RNAs Across the Domains of Life
3. From Prokaryotes to Eukaryotes: The Hidden Layer of Non-Coding RNAs
4. The Cell from a Systems Biology Perspective
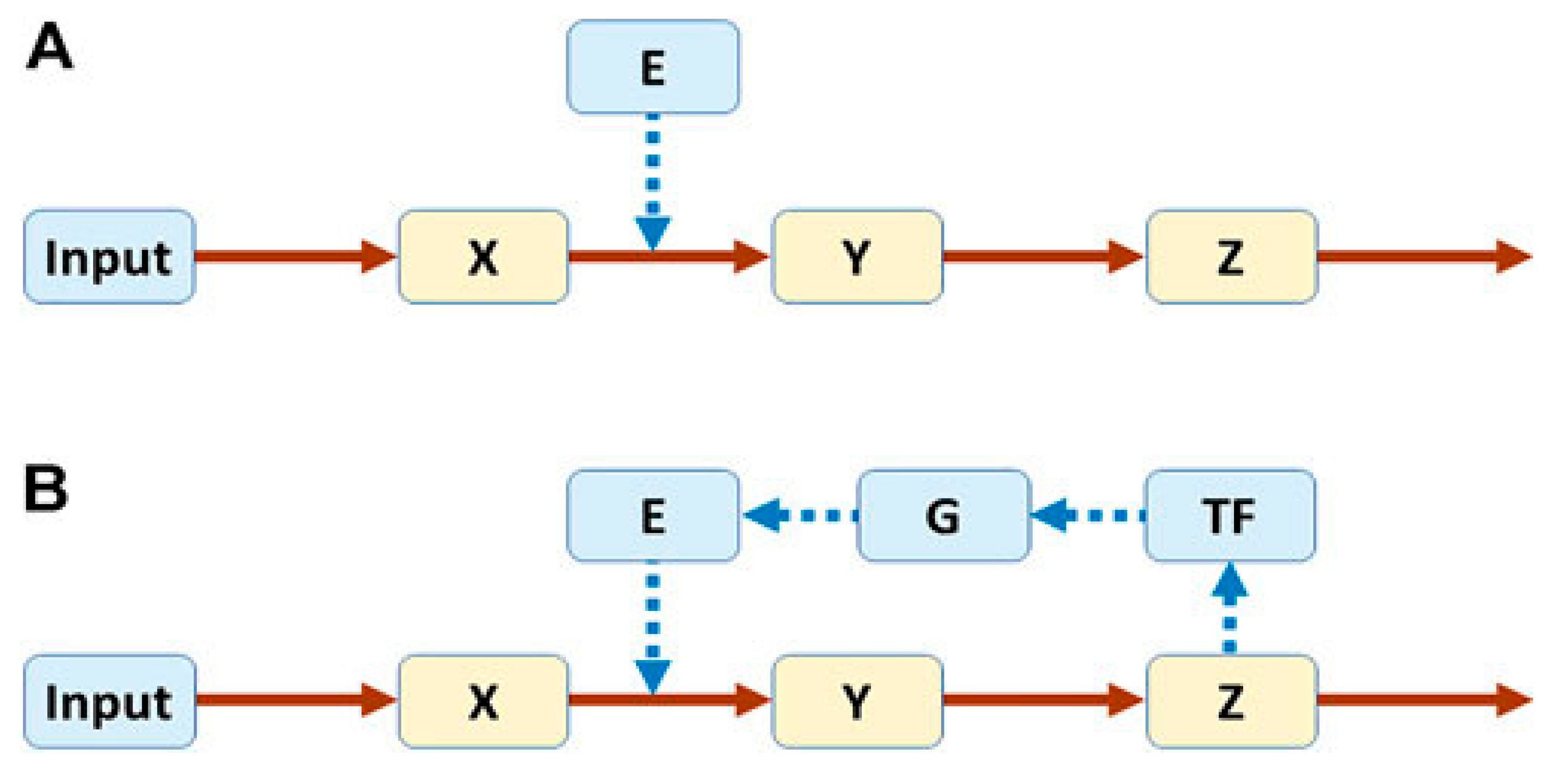
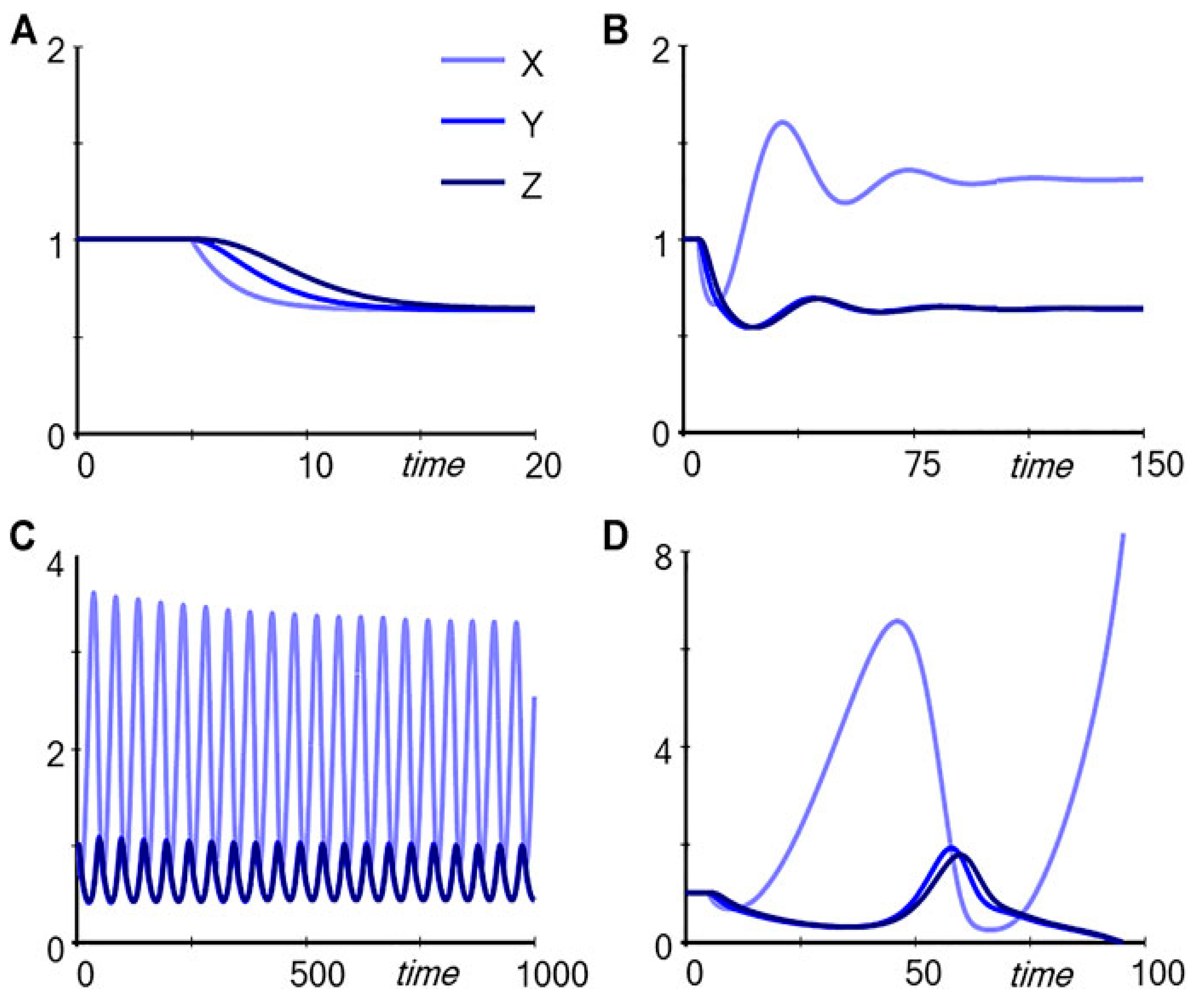
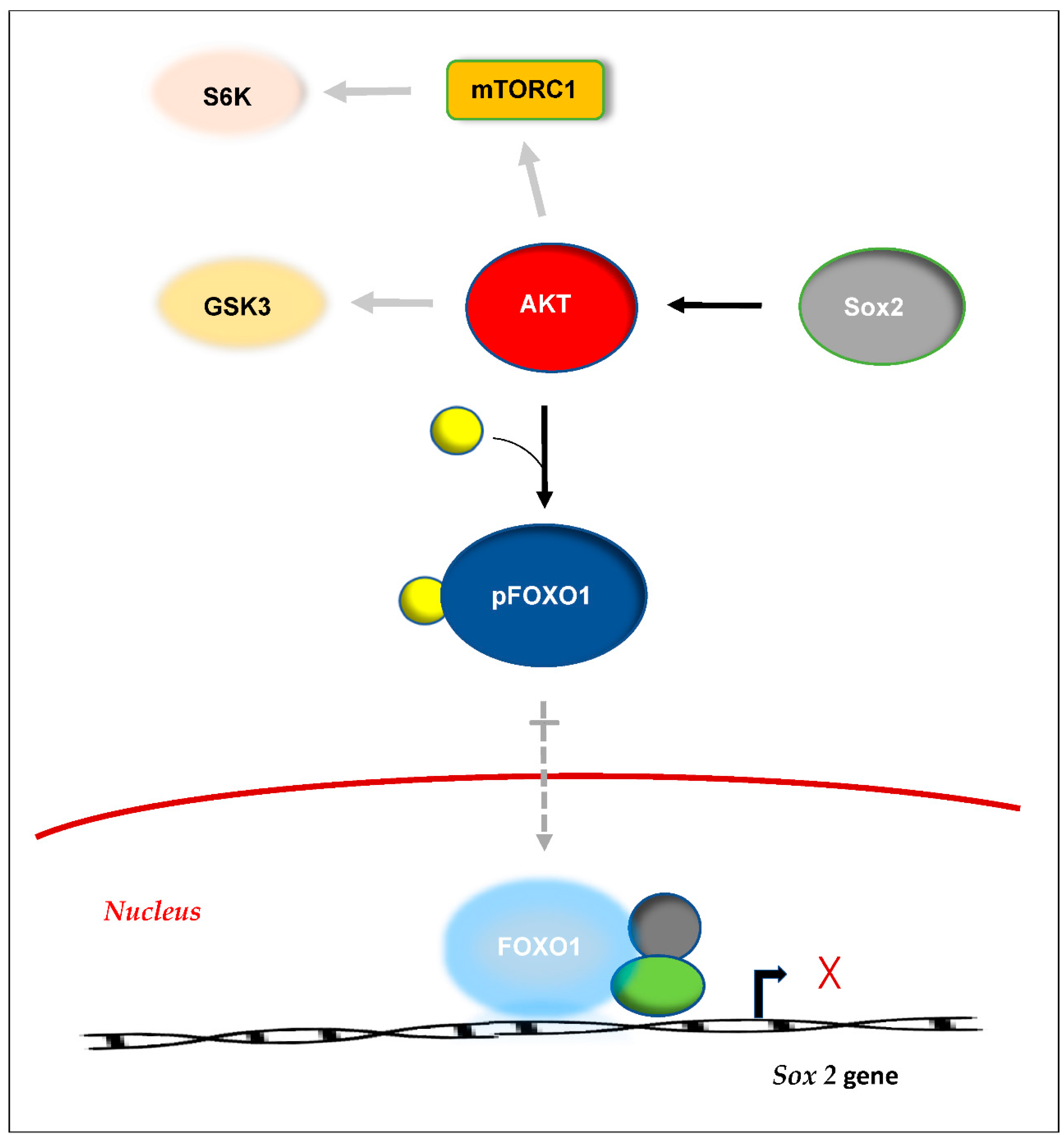
5. How Order Is Achieved: Regulatory Networks and the Origin of Attractor States
6. From Normalcy to Malignancy: Role of Non-Coding RNAs
6.1. Consequences of Disruption of Non-Coding RNAs
6.2. Clinicopathological Considerations of the Cancer Attractor State
7. Discussion
Implications for Systemic Cancer Therapy
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 3’ UTR | 3’ untranslated region |
| 5’ UTR | 5’ untranslated region |
| ceRNA | competitive endogenous RNA |
| circRNA | circular RNA |
| EMT | Epithelial-to-Mesenchymal Transition |
| GRN | genome-wide regulatory network |
| lncRNA | long non-coding RNA |
| miR | mature form of microRNA |
| miRNA | microRNA |
| mRNA | messenger RNA |
| ncRNA | non-coding RNA |
| nt | nucleotide |
| PRC2 | polycomb-repressive complex 2 |
| RBS | ribosomal binding site |
| RISC | RNA-induced silencing complex |
| rRNA | ribosomal RNA |
| snoRNA | small nucleolar RNA |
| SNV | single nucleotide variants |
| sRNA | small RNA |
| tRNA | transfer RNA |
References
- Huang, S.; Soto, A.M.; Sonnenschein, C. The end of the genetic paradigm of cancer. PLoS Biol. 2025, 23(3), e3003052. [CrossRef]
- Shah, A. The Primary Role of Non-coding RNA in the Pathogenesis of Cancer. Genes 2025, 16, 771. [CrossRef]
- The ICGC/TCGA Pan-Cancer Analysis of Whole Genomes Consortium. Pan-cancer analysis of whole genomes. Nature 2020, 578, 82–93. [CrossRef]
- de Magalhães, J.P. Every gene can (and possibly will) be associated with cancer. Trends Genet. 2022, 38(3), 216-217. [CrossRef]
- Li, Y.; Roberts, N.D.; Wala, J.A.; Shapira, O.; Schumacher, S.E.; Kumar, K.; Khurana, E.; Waszak, S.; Korbel, J.O.; Haber, J.E.; et al. PCAWG Consortium. Patterns of somatic structural variation in human cancer genomes. Nature 2020, 578, 112–121. [CrossRef]
- Ciriello, G.; Miller, M.L.; Aksoy, B.A.; Senbabaoglu, Y.; Schultz, N.; Sander, C. Emerging landscape of oncogenic signatures across human cancers. Nat Genet. 2013, 45(10), 1127-1133. [CrossRef]
- Tamborero, D.; Gonzalez-Perez, A.; Perez-Llamas, C.; Deu-Pons, J.; Kandoth, C.; Reimand, J.; Lawrence, M.S.; Getz, G.; Bader, G.D.; Ding, L.; et al. Comprehensive identification of mutational cancer driver genes across 12 tumor types. Sci Rep. 2013, 3, 2650. [CrossRef]
- Kandoth, C.; McLellan, M.D.; Vandin, F.; Ye, K.; Niu, B.; Lu, C.; Xie, M.; Zhang, Q.; McMichael, J.F.; Wyczalkowski, M.A.; et al. Mutational landscape and significance across 12 major cancer types. Nature 2013, 502(7471), 333-339. [CrossRef]
- Porta-Pardo, E.; Garcia-Alonso, L.; Hrabe, T.; Dopazo, J.; Godzik, A. A Pan-cancer catalogue of cancer driver protein interaction interfaces. PLoS Comput Biol. 2015, 11(10), e1004518. [CrossRef]
- Martínez-Jiménez, F.; Muiños, F.; Sentís, I.; Deu-Pons, J.; Reyes-Salazar, I.; Arnedo-Pac, C.; Mularoni, L.; Pich, O.; Bonet, J.; Kranas, H.; et al. A compendium of mutational cancer driver genes. Nat Rev Cancer 2020, 20, 555–572. [CrossRef]
- Kato, S.; Lippman, S.M.; Flaherty, K.T.; Kurzrock, R. The Conundrum of Genetic "Drivers" in Benign Conditions. J Natl Cancer Inst. 2016, 108(8), djw036. [CrossRef]
- Adashek, J.J.; Kato, S.; Lippman, S.M.; Kurzrock, R. The paradox of cancer genes in non-malignant conditions: implications for precision medicine. Genome Med. 2020, 12(1), 16. [CrossRef]
- Shain, A.H.; Yeh, I.; Kovalyshyn, I.; Sriharan, A.; Talevich, E.; Gagnon, A.; et al. The Genetic Evolution of Melanoma from Precursor Lesions. N Engl J Med. 2015, 373(20), 1926-1936. [CrossRef]
- Torreggiani, S.; Castellan, F.S., Aksentijevich, I.; Beck, D.B. Somatic mutations in autoinflammatory and autoimmune disease. Nat Rev Rheumatol. 2024, 20, 683–698. [CrossRef]
- Hafner, C.; van Oers, J.M.; Hartmann, A.; Landthaler, M.; Stoehr, R.; Blaszyk, H.; et al. High frequency of FGFR3 mutations in adenoid seborrheic keratoses. J Invest Dermatol. 2006, 126(11), 2404-2407. [CrossRef]
- The Cancer Genome Atlas Research Network. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature 2014, 507, 315–322. [CrossRef]
- L'Hôte C.G.; Knowles, M.A. Cell responses to FGFR3 signalling: growth, differentiation and apoptosis. Exp Cell Res. 2005, 304(2), 417-431. [CrossRef]
- Coorens, T.H.H.; Collord, G.; Jung, H.; Wang, Y.; Moore, L.; Hooks, Y.; Mahbubani, K.; Law, S.Y.K.; Yan, H.H.N.; Yuen, S.T. et al. The somatic mutation landscape of normal gastric epithelium. Nature 2025, 640, 418–426. [CrossRef]
- Machado, H.E.; Mitchell, E.; Øbro, N.F.; Kübler, K.; Davies, M.; Leongamornlert, D.; Cull, A.; Maura, F.; Sanders, M.A.; Cagan, A.T.J.; et al. Diverse mutational landscapes in human lymphocytes. Nature 2022, 608, 724–732. [CrossRef]
- Moore, L.; Leongamornlert, D.; Coorens, T.H.H.; Sanders, M.A.; Ellis, P.; Dentro, S.C.; Dawson, K.J.; Butler, T.; Rahbari, R.; Mitchell, T.J.; et al. The mutational landscape of normal human endometrial epithelium. Nature 2020, 580(7805), 640-646. [CrossRef]
- Lee-Six, H.; Olafsson, S.; Ellis, P.; Osborne, R.J.; Sanders, M.A.; Moore, L.; Georgakopoulos, N.; Torrente, F.; Noorani, A.; Goddard, M.; et al. The landscape of somatic mutation in normal colorectal epithelial cells. Nature 2019, 574(7779), 532-537. [CrossRef]
- Martincorena, I.; Fowler, J.C.; Wabik, A.; Lawson, A.R.J.; Abascal, F.; Hall, M.W.J.; Cagan, A.; Murai, K.; Mahbubani, K.; Stratton, M.R.; et al. Somatic mutant clones colonize the human esophagus with age. Science 2018, 362(6417), 911-917. [CrossRef]
- Martincorena, I.; Roshan, A.; Gerstung, M.; Ellis, P.; Van Loo, P.; McLaren, S.; Wedge, D.C.; Fullam, A.; Alexandrov, L.B.; Tubio, J.M.; et al. Tumor evolution. High burden and pervasive positive selection of somatic mutations in normal human skin. Science 2015, 348(6237), 880-886. [CrossRef]
- Yokoyama, A.; Kakiuchi, N.; Yoshizato, T.; Nannya, Y.; Suzuki, H.; Takeuchi, Y.; Shiozawa, Y.; Sato, Y.; Aoki, K.; Kim, S.K.; et al. Age-related remodelling of oesophageal epithelia by mutated cancer drivers. Nature 2019, 565(7739), 312-317. [CrossRef]
- Shlush, L.I. Age-related clonal hematopoiesis. Blood 2018, 131(5), 496-504. [CrossRef]
- Lawson, A.R.J.; Abascal, F.; Nicola, P.A.; Lensing S.V.; Roberts A.L.; Kalantzis G.; et al. Somatic mutation and selection at population scale. Nature 2025, 647, 411–420. [CrossRef]
- Carvalho Barbosa, C.; Calhoun, S.H.; Wieden, H.J. Non-coding RNAs: what are we missing? Biochem Cell Biol. 2020, 98(1), 23-30. [CrossRef]
- Beisel, C.L.; Storz, G. The Base-Pairing RNA Spot 42 Participates in a Multioutput Feedforward Loop to Help Enact Catabolite Repression in Escherichia coli. Molecular Cell 2010, 41(3), 286-297. [CrossRef]
- Majumder, R.; Ghosh, S.; Das, A.; Singh, M.K.; Samanta, S.; Saha, A.; Saha, R.P. Prokaryotic ncRNAs: Master regulators of gene expression. Current Research in Pharmacology and Drug Discovery 2022, 3, 100136. [CrossRef]
- Dutta, T.; Srivastava, S. Small RNA-mediated regulation in bacteria: A growing palette of diverse mechanisms. Gene 2018, 656, 60-72. [CrossRef]
- Thomason, M.K.; Storz, G. Bacterial antisense RNAs: how many are there, and what are they doing? Annu Rev Genet. 2010, 44, 167-188. [CrossRef]
- Gottesman, S.; Storz, G. Bacterial small RNA regulators: versatile roles and rapidly evolving variations. Cold Spring Harb Perspect Biol. 2011, 3(12), a003798. [CrossRef]
- Duss, O.; Michel, E.; Dit Konté, N. D.; Schubert, M.; Allain, F. H. T. Molecular basis for the wide range of affinity found in Csr/Rsm protein-RNA recognition. Nucleic Acids Research 2014, 42(8), 5332–5346. [CrossRef]
- Auguet J-C.; Barberan, A.; Casamayor, E.O. Global ecological patterns in uncultured Archaea. The ISME Journal 2010, 4(2), 182–190. [CrossRef]
- Bang, C.; Schmitz, R.A. Archaea associated with human surfaces: not to be underestimated. FEMS Microbiology Reviews 2015, 39(5), 631–648. [CrossRef]
- Gelsinger, D.R.; DiRuggiero, J. The Non-Coding Regulatory RNA Revolution in Archaea. Genes 2018, 9(3), 141. [CrossRef]
- Arias-Carrasco, R.; Aliaga-Tobar, V.; Abades, S.; Maracaja-Coutinho, V. The repertoire of candidate archaeal noncoding RNAs and their association with temperature adaptation. BioSystems. 2025, 254, 105519. [CrossRef]
- Galagan, J.E.; Nusbaum, C.; Roy, A.; Endrizzi, M.G.; Macdonald, P.; FitzHugh, W.; et al. The genome of M. acetivorans reveals extensive metabolic and physiological diversity. Genome Res. 2002, 12, 532-542. [CrossRef]
- Liang, Y.; Qi, W.; Dong, X.; Li, J. Archaeal RNA processing and regulation: expanding the functional landscape. Microbiol Mol Biol Rev. 2025, 89(4), e00318-24. [CrossRef]
- Chen L.L.; Kim V.N. Small and long non-coding RNAs: Past, present, and future. Cell 2024, 187, 6451–6485. [CrossRef]
- Statello, L.; Guo, C.J.; Chen, L.L.; Huarte, M. Gene regulation by long non-coding RNAs and its biological functions. Nat Rev Mol Cell Biol. 2021, 22(2), 96-118. [CrossRef]
- Kim, J. Circular RNAs: Novel Players in Cancer Mechanisms and Therapeutic Strategies. Int J Mol Sci. 2024, 25(18), 10121. [CrossRef]
- Hertel, J.; Lindemeyer, M.; Missal, K.; Fried, C.; Tanzer, A.; Flamm, C.; Hofacker, I.L.; Stadler, P.F.; et al. The expansion of the metazoan microRNA repertoire. BMC Genomics 2006, 7, 25. [CrossRef]
- Nakabachi, A.; Yamashita, A.; Toh, H.; Ishikawa, H.; Dunbar, H.E.; Moran, N.A.; et al. The 160-kilobase genome of the bacterial endosymbiont Carsonella. Science 2006, 314(5797), 267. [CrossRef]
- Binnewies, T.T.; Motro, Y.; Hallin, P.F.; Lund, O.; Dunn, D.; La, T.; et al. 2006. Ten years of bacterial genome sequencing: comparative-genomics-based discoveries. Funct Integr Genomics 2006, 6(3), 165–185. [CrossRef]
- Gottesman, S. Micros for microbes: non-coding regulatory RNAs in bacteria. Trends Genet. 2005, 21, 399–404. [CrossRef]
- Mattick, J. RNA regulation: a new genetics? Nat Rev Genet. 2004, 5, 316–323. [CrossRef]
- Taft, R.J.; Pheasant, M.; Mattick, J.S. The relationship between non-protein-coding DNA and eukaryotic complexity. Bioessays 2007, 29(3), 288-299. [CrossRef]
- Croft, L.J.; Lercher, M.J.; Gagen, M.J.; Mattick, J.S. Is prokaryotic complexity limited by accelerated growth in regulatory overhead? Genome Biol. 2003, 5, 1-26. [CrossRef]
- Gagen, M.J.; Mattick, J.S. Inherent size constraints on prokaryote gene networks due to "accelerating" growth. Theory Biosci. 2005, 123(4), 381-411. PMID: 18202872. [CrossRef]
- Frith, M.C.; Pheasant, M.; Mattick, J.S. The amazing complexity of the human transcriptome. Eur J Hum Genet. 2005, 13(8), 894-898. [CrossRef]
- Carninci, P.; Kasukawa, T.; Katayama, S.; Gough, J.; Frith, M.C.; Maeda, N.; et al. The transcriptional landscape of the mammalian genome. Science 2005, 309(5740), 1559-1563. [CrossRef]
- Mattick, J.S. Challenging the dogma: the hidden layer of non-protein-coding RNAs in complex organisms. Bioessays. 2003, 25, 930–939. [CrossRef]
- DeVeale, B.; Swindlehurst-Chan, J.; Blelloch, R. The roles of microRNAs in mouse development. Nature Reviews. Genetics 2021, 22(5), 307-323. [CrossRef]
- Sun, B.; Liu, C.; Li, H.; Zhang, L.; Luo, G.; Liang, S.; Lü, M. Research progress on the interactions between long non-coding RNAs and microRNAs in human cancer. Oncology letters. 2020, 19(1), 595-605. [CrossRef]
- Bhattacharjee, R.; Prabhakar, N.; Kumar, L.; Bhattacharjee, A.; Kar, S.; Malik, S.; et al. Crosstalk between long noncoding RNA and microRNA in Cancer. Cell Oncol. 2023, 46, 885–908. [CrossRef]
- Voit, E.O. Perspective: Systems biology beyond biology. Front. Syst. Biol. 2022, 2. [CrossRef]
- Brandman, O.; Meyer, T. Feedback loops shape cellular signals in space and time. Science 2008, 322(5900), 390-395. [CrossRef]
- Tian, X.; Zhang, X.; Liu, F.; Wang, W. Interlinking positive and negative feedback loops creates a tunable motif in gene regulatory networks. Phys. Rev. 2009, E 80, 011926. [CrossRef]
- Ormsbee Golden BD, Wuebben EL, Rizzino A. Sox2 expression is regulated by a negative feedback loop in embryonic stem cells that involves AKT signaling and FoxO1. PLoS One 2013, 8(10), e76345. [CrossRef]
- Zhao, P.; Liu, J.; Bao, T.; Huo, H.; Yuan, Y.; Fang, T. Flexible modulation of hybrid feedback loops in competitive biological oscillators. npj Syst Biol Appl. 2025, 11, 122. [CrossRef]
- Thomas, R. Laws for the dynamics of regulatory networks. Int J Dev Biol. 1998, 42, 479-485. PMID: 9654035.
- Mochizuki A. Controlling complex dynamical systems based on the structure of the networks. Biophys Physicobiol. 2023, 20(2), e200019. [CrossRef]
- Jordan, J.D.; Landau, E.M.; Iyengar, R. Signaling networks: the origins of cellular multitasking. Cell 2000, 103(2), 193-200. [CrossRef]
- Freeman M. Feedback control of intercellular signalling in development. Nature 2000, 408(6810), 313-319. [CrossRef]
- Kauffman, S. Metabolic stability and epigenesis in randomly constructed genetic nets, J. Theor. Biol. 1969, 22, 437-467. [CrossRef]
- Kauffman, S. Homeostasis and differentiation in random genetic control networks. Nature 1969, 224, 177-178. [CrossRef]
- Fink, T. M. A.; Sheldon, F. Number of Attractors in the Critical Kauffman Model Is Exponential. Physical review letters 2023, 131(26), 267402. [CrossRef]
- Li, R.; Rozum, J.C.; Quail, M.M.; Qasim, M.N.; Sindi, S.S.; Nobile, C.J.; Albert, R.; Hernday, A.D. Inferring gene regulatory networks using transcriptional profiles as dynamical attractors. PLoS Comput Biol. 2023, 19(8), e1010991. [CrossRef]
- Huang, S. On the intrinsic inevitability of cancer: From foetal to fatal attraction. Seminars in Cancer Biology 2011, 21(3), 183-199. [CrossRef]
- Milnor, J.W. On the concept of attractor. Communications in Mathematical Physics 1985, 99, 177-195. [CrossRef]
- Waddington, C.H. Canalization of development and the inheritance of acquired characters. Nature 1942, 150(3811), 563-565. [CrossRef]
- Raj, A.; van Oudenaarden, A. Nature, nurture, or chance: stochastic gene expression and its consequences. Cell 2008,135(2), 216-226. [CrossRef]
- Dong, P.; Liu, Z. Shaping development by stochasticity and dynamics in gene regulation. Open Biol. 2017, 7, 170030. dx. [CrossRef]
- Misteli, T. The Self-Organizing Genome: Principles of Genome Architecture and Function. Cell 2020, 183(1), 28-45. [CrossRef]
- Scarpa E., Mayor R. Collective cell migration in development. J Cell Biol. 2016, 212 (2), 143–155. [CrossRef]
- Greenberg M.V.C., Bourc’his D. The diverse roles of DNA methylation in mammalian development and disease. Nat. Rev. Mol. Cell Biol. 2019, 20, 590–607. [CrossRef]
- Jambhekar, A.; Dhall, A.; Shi, Y. Roles and regulation of histone methylation in animal development. Nat. Rev. Mol. Cell Biol. 2019, 20, 625–641. [CrossRef]
- Nojima T., Proudfoot N.J. Mechanisms of lncRNA biogenesis as revealed by nascent transcriptomics. Nat. Rev. Mol. Cell Biol. 2022, 23, 389–406. [CrossRef]
- Wang A. Conceptual breakthroughs of the long noncoding RNA functional system and its endogenous regulatory role in the cancerous regime. Explor. Target. Antitumor Ther. 2024, 5, 1706. [CrossRef]
- Lai, F.; Orom, U.A.; Cesaroni, M.; Beringer, M.; Taatjes, D.J.; Blobel, G.A.; et al. Activating RNAs associate with Mediator to enhance chromatin architecture and transcription. Nature 2013, 494, 497–501. [CrossRef]
- Billi, M.; De Marinis, E.; Gentile, M.; Nervi, C.; Grignani, F. Nuclear miRNAs: Gene Regulation Activities. Int. J. Mol. Sci. 2024, 25, 6066. [CrossRef]
- Hu, Q.; Kwon, Y.S.; Nunez, E.; Cardamone, M.D.; Hutt, K.R.; Ohgi, K.A.; et al. Enhancing nuclear receptor-induced transcription requires nuclear motor and LSD1-dependent gene networking in interchromatin granules. Proc Natl Acad Sci USA. 2008, 105, 19199–19204. [CrossRef]
- Nunez, E.; Fu, X.D.; Rosenfeld, M.G. Nuclear organization in the 3D space of the nucleus—cause or consequence? Curr Opin Genet Dev. 2009, 19, 424–436. [CrossRef]
- Fu, X.D. Non-coding RNA: a new frontier in regulatory biology. Natl Sci Rev. 2014, 1(2),190-204. [CrossRef]
- Huang, S.; Eichler, G.; Bar-Yam, Y.; Ingber, D.E. Cell fates as high-dimensional attractor states of a complex gene regulatory network. Phys Rev Lett. 2005, 94, 128701. [CrossRef]
- Jin, Y.; Wang, J.; Bachtiar, M.; Chong, S.S.; Lee, C.G.L. Architecture of polymorphisms in the human genome reveals functionally important and positively selected variants in immune response and drug transporter genes. Hum Genomics 2018, 12(1), 43. [CrossRef]
- Yang, Y.; Wang, D.; Miao, Y.R.; Wu, X.; Luo, H.; Cao, W.; et al. lncRNASNP v3: an updated database for functional variants in long non-coding RNAs. Nucleic Acids Res. 2023, 51(D1), D192-D198. [CrossRef]
- Preskill, C.; Weidhaas, J.B. SNPs in microRNA binding sites as prognostic and predictive cancer biomarkers. Crit Rev Oncog. 2013, 18(4), 327-340. [CrossRef]
- Park, M.S.; Jeong, S.D.; Shin, C.H.; Cha, S.; Yu, A.; Kim, E.J.; et al. LINC02257 regulates malignant phenotypes of colorectal cancer via interacting with miR-1273g-3p and YB1. Cell Death Dis. 2024, 15, 895. [CrossRef]
- Du, Z.; Sun, T.; Hacisuleyman, E.; Fei, T.; Wang, X.; Brown, M.; et al. Integrative analyses reveal a long noncoding RNA-mediated sponge regulatory network in prostate cancer. Nat Commun. 2016, 7, 10982. PMID: 26975529; PMCID: PMC4796315. [CrossRef]
- Guarnerio, J.; Bezzi, M.; Jeong, J.C.; Paffenholz, S.V.; Berry, K.; Naldini, M.M.; et al. Oncogenic role of fusion-circRNAs derived from cancer-associated chromosomal translocations. Cell 2016, 165(2), 289-302. Epub 2016 Mar 31. [CrossRef]
- Zhang, L.; Yang, C.S.; Varelas, X.; Monti, S. Altered RNA editing in 3' UTR perturbs microRNA-mediated regulation of oncogenes and tumor-suppressors. Sci Rep. 2016, 6, 23226. [CrossRef]
- Zhai, X., Zhang, Z., Chen, Y. et al. Current and future therapies for small cell lung carcinoma. J Hematol Oncol. 2025, 18, 37. [CrossRef]
- Bernards, R.; Weinberg, R. Metastasis genes: A progression puzzle. Nature. 2002, 418(6900), 823. [CrossRef]
- Lambert, A.W.; Pattabiraman, D.R.; Weinberg, R.A. Emerging Biological Principles of Metastasis. Cell. 2017, 168(4), 670-691. [CrossRef]
- Birkbak, N.J.; McGranahan, N. Cancer genome evolutionary trajectories in metastasis. Cancer Cell. 2020, 37(1), 8-19. [CrossRef]
- Hu, Z.; Ding, J.; Ma, Z.; et al. Quantitative evidence for early metastatic seeding in colorectal cancer. Nat Genet. 2019, 51(7), 1113-1122. [CrossRef]
- Roche, J. The Epithelial-to-Mesenchymal Transition in Cancer. Cancers. 2018, 10(2), 52. [CrossRef]
- Allgayer, H.; Mahapatra, S.; Mishra, B.; Swain, B.; Saha, S.; Khanra, S.; et al. Epithelial-to-mesenchymal transition (EMT) and cancer metastasis: the status quo of methods and experimental models. Mol Cancer. 2025, 24, 167. [CrossRef]
- Nieto, M.A.; Huang, R.Y-J.; Jackson, R.A.; Thiery, J.P. EMT: 2016. Cell. 2016, 166, 21–45. [CrossRef]
- Schnirman, R.E.; Kuo, S.J.; Kelly, R.C.; Yamaguchi, T.P.; Willert, K. Chapter Five - The role of Wnt s [ignaling in the development of the epiblast and axial progenitors. Current Topics in Developmental Biology. Academic Press. 2023, 153, 145-180. [CrossRef]
- Lu, W.; Kang, Y. Epithelial-Mesenchymal Plasticity in Cancer Progression and Metastasis. Dev Cell. 2019, 49(3), 361-374. [CrossRef]
- Barbeau, M.C.; Brown, B.A.; Adair, S.A.; Bauer, T.W.; Lazzara, M.J. The kinase ERK plays a conserved dominant role in the heterogeneity of epithelial-mesenchymal transition in pancreatic cancer cells. Sci. Signal. 2025, 18, eads7002. [CrossRef]
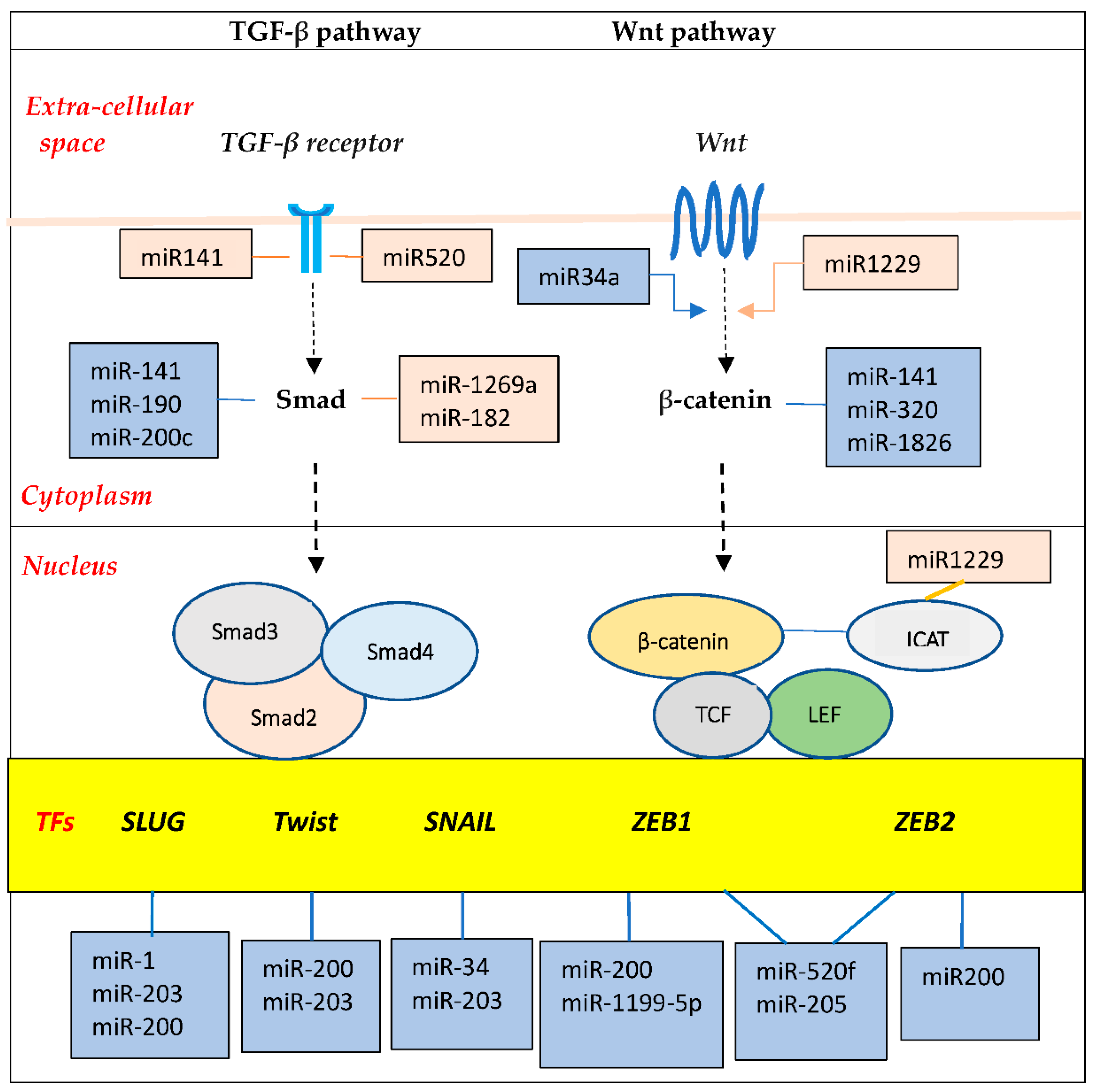
- Khanbabaei, H.; Ebrahimi, S.; García-Rodríguez, J.L.; Ghasemi, Z.; Pourghadamyari, H.; Mohammadi, M.; Kristensen, L.S. Non-coding RNAs and epithelial mesenchymal transition in cancer: molecular mechanisms and clinical implications. J Exp Clin Cancer Res. 2022, 41(1), 278. [CrossRef]
- Gómez Tejeda Zañudo, J.; Guinn, M.T.; Farquhar, K.; Szenk, M.; Steinway, S.N.; Balázsi, G.; Albert, R. Towards control of cellular decision-making networks in the epithelial-to-mesenchymal transition. Phys. Biol. 2019, 16(3), 031002. [CrossRef]
- Peng, J.; Liu, W.; Tian, J.; Shu, Y.; Zhao, R.; Wang, Y. Non-coding RNAs as key regulators of epithelial-mesenchymal transition in breast cancer. Front Cell Dev Biol. 2025, 13, 1544310. [CrossRef]
- Nadukkandy, A.S.; Blaize, B.; Kumar, C.D.; Mori, G.; Cordani, M.; Kumar. L.D. Non-coding RNAs as mediators of epithelial to mesenchymal transition in metastatic colorectal cancers. Cellular Signalling. 2025, 127, 111605. [CrossRef]
- Huang, S.; Ingber. D.E. A non-genetic basis for cancer progression and metastasis: self-organizing attractors in cell regulatory networks. Breast Dis. 2006, 26, 27-54. [CrossRef]
- Wang, W.; Poe, D.; Yang, Y.; Hyatt, T.; Xing, J. Epithelial-to-mesenchymal transition proceeds through directional destabilization of multidimensional attractor. eLife. 2022, 11, e74866. [CrossRef]
- Huang, Y.; Hong, W.; Wei, X. The molecular mechanisms and therapeutic strategies of EMT in tumor progression and metastasis. J Hematol Oncol. 2022, 15, 129. [CrossRef]
- Pastushenko, I.; Brisebarre, A.; Sifrim, A.; Fioramonti, M.; Revenco, T.; Boumahdi, S.; et al. Identification of the tumour transition states occurring during EMT. Nature. 2018, 556(7702), 463–468. [CrossRef]
- Krueger, K.E. Survey for Activating Oncogenic Mutation Variants in Metazoan Germline. Genes. J Mol Evol. 2024, 92, 930–943. [CrossRef]
- Fan, J. Revisiting the somatic mutation theory of cancer pathogenesis. Nat Rev Genet. 2026, 27, 116. [CrossRef]
- Roy, L.; Chatterjee, O.; Bose, D.; Roy, A.; Chatterjee, S. Noncoding RNA as an influential epigenetic modulator with promising roles in cancer therapeutics. Drug Discov Today. 2023, 28, 103690. [CrossRef]
- Aznaourova, M.; Schmerer, N.; Schmeck, B.; Schulte, L.N. Disease-Causing Mutations and Rearrangements in Long Non-coding RNA Gene Loci. Front Genet. 2020, 11, 527484. [CrossRef]
- Huang, S. Gene expression profiling, genetic networks, and cellular states: an integrating concept for tumorigenesis and drug discovery. Mol Med. 1999, 77, 469–480. [CrossRef]
- Rhoads, C.P. Nitrogen mustards in the treatment of neoplastic disease; official statement. J Am Med Assoc. 1946, 131, 656-658. [CrossRef]
- Min, H.Y.; Lee, H.Y. Molecular targeted therapy for anticancer treatment. Exp Mol Med. 2022, 54(10), 1670-1694. [CrossRef]
- Tregear, M.; Visco, F. Outcomes that matter to patients with cancer: living longer and living better. EClinicalMedicine. 2024 Sep 11;76:102833. [CrossRef]
- Piergentili, R.; Sechi, S. Targeting Regulatory Noncoding RNAs in Human Cancer: The State of the Art in Clinical Trials. Pharmaceutics 2025, 17, 471. [CrossRef]
- Liu, Y.; Wang, C.; Fu, X.; Ren, M. The Progress and Evolving Trends in Nucleic-Acid-Based Therapies. Biomolecules. 2025, 15(3), 376. [CrossRef]
- Das Adhikari, S.; Yang, J.; Wang, J.; Cui, Y. Recent advances in spatially variable gene detection in spatial transcriptomics. Comput Struct Biotechnol J. 2024, 23, 883-891. [CrossRef]
- Tian, L.; Xiao, J.; Yu, T. A robust statistical approach for finding informative spatially associated pathways. Brief Bioinform. 2024, 25(6), bbae543. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



