Submitted:
26 January 2026
Posted:
27 January 2026
You are already at the latest version
Abstract
Many cannabinoids are derived from Cannabis and exhibit a diverse range of pharmacological properties. Predictions of bioactivities of these compounds were conducted by molecular docking computation on two transient receptor potential (TRP) receptors (TRPV1 and TRPC5) found on human glioma (U-87 MG) cells. These predictions were experimentally confirmed by monitoring changes in intracellular calcium concentration in U-87 MG cells treated with cannabinol (CBN), cannabichromene (CBC), and cannabicyclol (CBL), as measured using a fluorescence microplate reader. The results indicate that CBN and CBC are bioactive, whereas CBL exhibits minimal activity. These findings are consistent with predictions obtained from molecular docking computation based on AutoDock Vina.
Keywords:
Introduction
Experimental Section
Reagents
Cell Samples
Instrument

Cell Calcium Bulk Analysis
3.2. Loading Cells on a 96-Well Plate
3.3. Fluo-4 AM Dye Loading
3.4. Fluorescence Assay on a 96-Well Plate
Data Analysis and Results
Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pisanti, S; Malfitano, AM; Ciaglia, E; Lamberti, A; Ranieri, R; Cuomo, G; et al. Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacology & therapeutics 2017, 175, 133–50. [Google Scholar]
- Nabissi, M; Morelli, MB; Santoni, M; Santoni, G. Triggering of the TRPV2 channel by cannabidiol sensitizes glioblastoma cells to cytotoxic chemotherapeutic agents. Carcinogenesis 2013, 34(1), 48–57. [Google Scholar] [PubMed]
- Nabissi, M; Morelli, MB; Amantini, C; Liberati, S; Santoni, M; Ricci-Vitiani, L; et al. Cannabidiol stimulates Aml-1a-dependent glial differentiation and inhibits glioma stem-like cells proliferation by inducing autophagy in a TRPV2-dependent manner. International journal of cancer 2015, 137(8), 1855–69. [Google Scholar] [CrossRef] [PubMed]
- Rice, T; Lachance, DH; Molinaro, AM; Eckel-Passow, JE; Walsh, KM; Barnholtz-Sloan, J; et al. Understanding inherited genetic risk of adult glioma – a review. Neuro-Oncology Practice 2015, 3(1), 10–6. [Google Scholar] [CrossRef]
- Dumitru, CA; Sandalcioglu, IE; Karsak, M. Cannabinoids in Glioblastoma Therapy: New Applications for Old Drugs. Frontiers in molecular neuroscience 2018, 11, 159. [Google Scholar]
- Velasco, G; Carracedo, A; Blázquez, C; Lorente, M; Aguado, T; Haro, A; et al. Cannabinoids and gliomas. Molecular neurobiology 2007, 36(1), 60–7. [Google Scholar] [CrossRef]
- Nimmrich, V; Gross, G. P/Q-type calcium channel modulators. Br J Pharmacol. 2012, 167(4), 741–59. [Google Scholar]
- Montana, V; Sontheimer, H. Bradykinin promotes the chemotactic invasion of primary brain tumors. The Journal of neuroscience: the official journal of the Society for Neuroscience 2011, 31(13), 4858–67. [Google Scholar] [CrossRef]
- Watkins, S; Sontheimer, H. Unique biology of gliomas: challenges and opportunities. Trends in neurosciences 2012, 35(9), 546–56. [Google Scholar] [CrossRef]
- Bomben, VC; Sontheimer, HW. Inhibition of transient receptor potential canonical channels impairs cytokinesis in human malignant gliomas. Cell proliferation 2008, 41(1), 98–121. [Google Scholar] [CrossRef]
- Cuddapah, VA; Turner, KL; Sontheimer, H. Calcium entry via TRPC1 channels activates chloride currents in human glioma cells. Cell calcium 2013, 53(3), 187–94. [Google Scholar] [CrossRef]
- Alptekin, M; Eroglu, S; Tutar, E; Sencan, S; Geyik, MA; Ulasli, M; et al. Gene expressions of TRP channels in glioblastoma multiforme and relation with survival. Tumour biology: the journal of the International Society for Oncodevelopmental Biology and Medicine 2015, 36(12), 9209–13. [Google Scholar] [CrossRef] [PubMed]
- Duncan, RS; Goad, DL; Grillo, MA; Kaja, S; Payne, AJ; Koulen, P. Control of intracellular calcium signaling as a neuroprotective strategy. Molecules (Basel, Switzerland) 2010, 15(3), 1168–95. [Google Scholar] [CrossRef] [PubMed]
- Li, X; Li, PC. Microfluidic selection and retention of a single cardiac myocyte, on-chip dye loading, cell contraction by chemical stimulation, and quantitative fluorescent analysis of intracellular calcium. Analytical chemistry 2005, 77(14), 4315–22. [Google Scholar] [CrossRef]
- Li, X; Xue, X; Li, PCH. Real-time detection of the early event of cytotoxicity of herbal ingredients on single leukemia cells studied in a microfluidic biochip. Integrative Biology 2009, 1(1), 90–8. [Google Scholar] [CrossRef]
- Huang, H. C.; Chang, P.; Lu, S. Y.; Zheng, B. W.; Jiang, Z. F. Protection of curcumin against amyloid-beta-induced cell damage and death involves the prevention from NMDA receptor-mediated intracellular Ca2+ elevation. Journal of receptor and signal transduction research 2015, 35(5), 450–7. [Google Scholar] [CrossRef]
- Takahashi, A.; Camacho, P.; Lechleiter, J. D.; Herman, B. Measurement of intracellular calcium. Physiological Reviews 1999, 79(4), 1089–125. [Google Scholar] [CrossRef] [PubMed]
- Gee, KR; Brown, KA; Chen, WN; Bishop-Stewart, J; Gray, D; Johnson, I. Chemical and physiological characterization of fluo-4 Ca(2+)-indicator dyes. Cell calcium 2000, 27(2), 97–106. [Google Scholar] [CrossRef]
- Fraguas-Sánchez, AI; Martín-Sabroso, C; Torres-Suárez, AI. Insights into the effects of the endocannabinoid system in cancer: a review. Br J Pharmacol. 2018, 175(13), 2566–80. [Google Scholar] [CrossRef]
- Hinz, B; Ramer, R. Anti-tumour actions of cannabinoids. Br J Pharmacol. 2019, 176(10), 1384–94. [Google Scholar] [CrossRef]
- Ramer, R; Hinz, B. Cannabinoids as Anticancer Drugs. Advances in pharmacology (San Diego, Calif) 2017, 80, 397–436. [Google Scholar] [PubMed]
- Ligresti, A; Moriello, AS; Starowicz, K; Matias, I; Pisanti, S; De Petrocellis, L; et al. Antitumor activity of plant cannabinoids with emphasis on the effect of cannabidiol on human breast carcinoma. The Journal of pharmacology and experimental therapeutics 2006, 318(3), 1375–87. [Google Scholar] [CrossRef] [PubMed]
- Shrivastava, A; Kuzontkoski, PM; Groopman, JE; Prasad, A. Cannabidiol induces programmed cell death in breast cancer cells by coordinating the cross-talk between apoptosis and autophagy. Molecular cancer therapeutics 2011, 10(7), 1161–72. [Google Scholar] [CrossRef] [PubMed]
- Sultan, AS; Marie, MA; Sheweita, SA. Novel mechanism of cannabidiol-induced apoptosis in breast cancer cell lines. Breast (Edinburgh, Scotland) 2018, 41, 34–41. [Google Scholar] [CrossRef]
- Massi, P; Vaccani, A; Ceruti, S; Colombo, A; Abbracchio, MP; Parolaro, D. Antitumor effects of cannabidiol, a nonpsychoactive cannabinoid, on human glioma cell lines. The Journal of pharmacology and experimental therapeutics 2004, 308(3), 838–45. [Google Scholar] [CrossRef]
- Vaccani, A; Massi, P; Colombo, A; Rubino, T; Parolaro, D. Cannabidiol inhibits human glioma cell migration through a cannabinoid receptor-independent mechanism. Br J Pharmacol. 2005, 144(8), 1032–6. [Google Scholar] [CrossRef]
- Massi, P; Valenti, M; Vaccani, A; Gasperi, V; Perletti, G; Marras, E; et al. 5-Lipoxygenase and anandamide hydrolase (FAAH) mediate the antitumor activity of cannabidiol, a non-psychoactive cannabinoid. Journal of neurochemistry 2008, 104(4), 1091–100. [Google Scholar] [CrossRef]
- Solinas, M; Massi, P; Cinquina, V; Valenti, M; Bolognini, D; Gariboldi, M; et al. Cannabidiol, a non-psychoactive cannabinoid compound, inhibits proliferation and invasion in U87-MG and T98G glioma cells through a multitarget effect. PloS one 2013, 8(10), e76918. [Google Scholar] [CrossRef]
- De Petrocellis, L; Ligresti, A; Moriello, AS; Allarà, M; Bisogno, T; Petrosino, S; et al. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. Br J Pharmacol. 2011, 163(7), 1479–94. [Google Scholar] [CrossRef]
- Bisogno, T; Hanus, L; De Petrocellis, L; Tchilibon, S; Ponde, DE; Brandi, I; et al. Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br J Pharmacol. 2001, 134(4), 845–52. [Google Scholar] [CrossRef]
- Lu, J; Ju, YT; Li, C; Hua, FZ; Xu, GH; Hu, YH. Effect of TRPV1 combined with lidocaine on cell state and apoptosis of U87-MG glioma cell lines. Asian Pacific journal of tropical medicine 2016, 9(3), 288–92. [Google Scholar] [CrossRef]
- Kárai, LJ; Russell, JT; Iadarola, MJ; Oláh, Z. Vanilloid receptor 1 regulates multiple calcium compartments and contributes to Ca2+-induced Ca2+ release in sensory neurons. The Journal of biological chemistry 2004, 279(16), 16377–87. [Google Scholar] [CrossRef] [PubMed]
- Noghabi, Hamideh Sharifi; Ahmed, Abdul Q.; Li, Paul C. H. Intracellular Calcium Increases due to Curcumin Measured using a Single-Cell Biochip. Anal. Lett. 2021, 54, 2769–2776. [Google Scholar] [CrossRef]
- Pflaum-Jaeger, Natali; Shahbod, Bardia; Rahimi, Abolfazl; Li, Paul C.H. Responses to herbal compounds in brain cancer cells: two cell-calcium assays and a molecular docking computation study. Anal. Lett. 2025, 58, 2338–2350. [Google Scholar] [CrossRef]

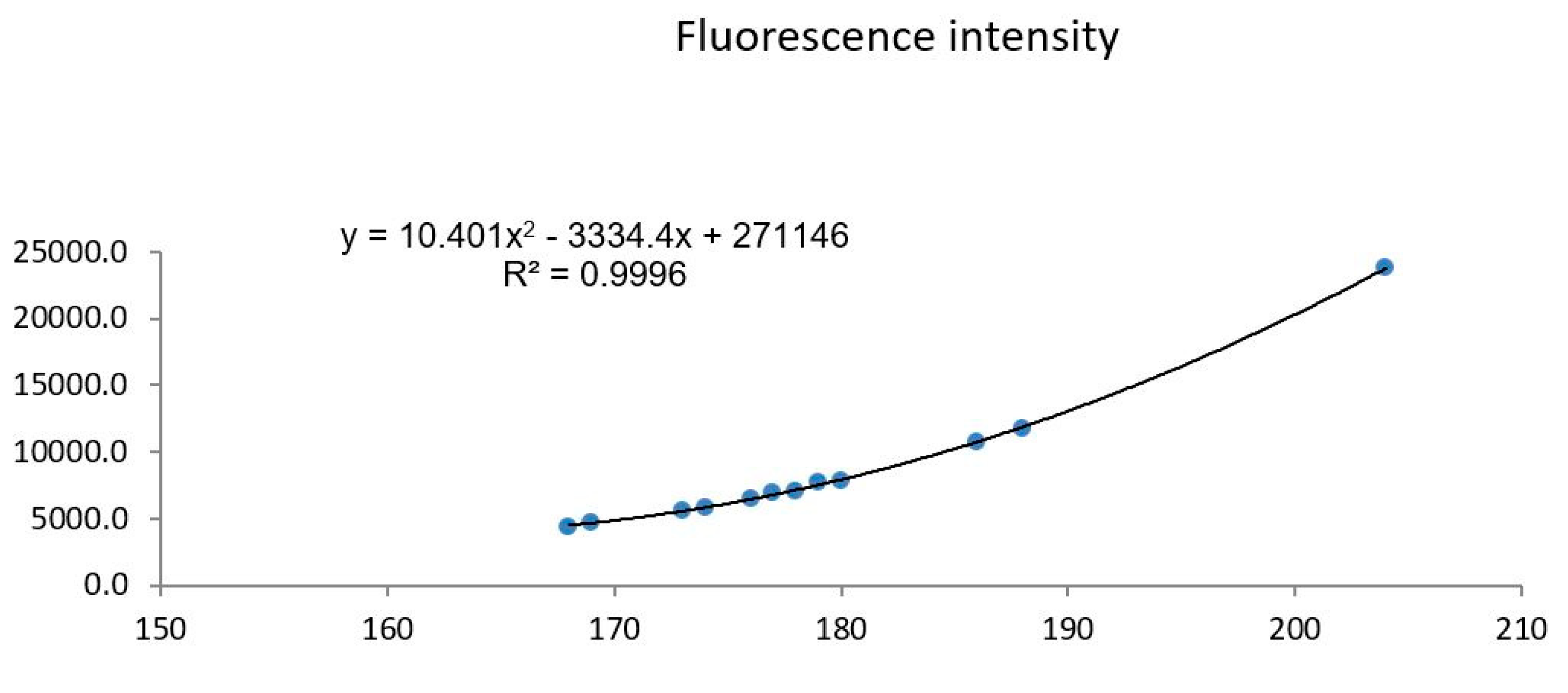
| Compound | Resting [Ca²⁺] (nM) | After Treatment [Ca²⁺] (nM) | Fold Increase |
|---|---|---|---|
| CBN | 445 ± 8 | 2144 ± 47 | 4.8 |
| CBC | 463 ± 6 | 2295 ± 31 | 4.9 |
| CBL | 311 ± 40 | 361 ± 15 | 1.2 |
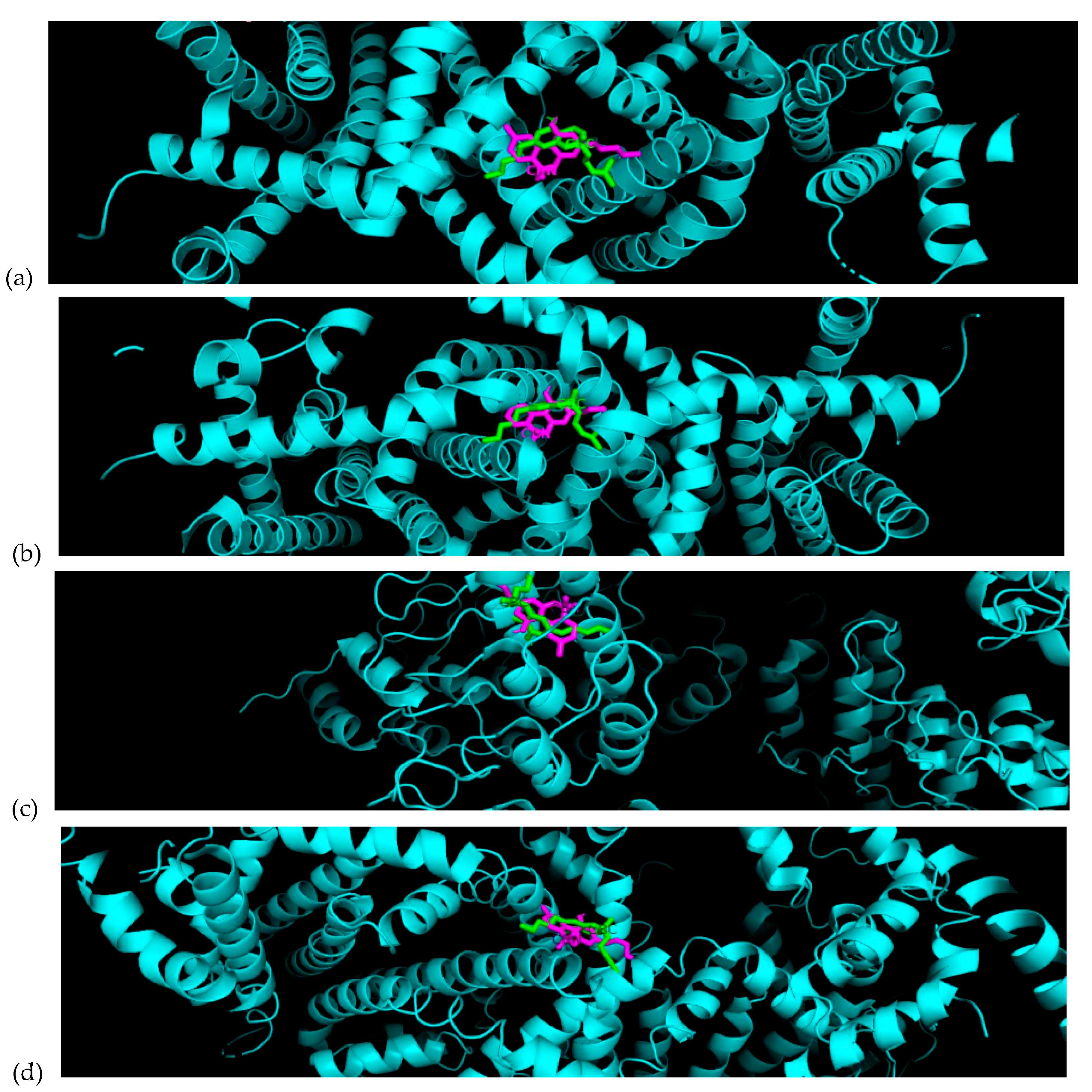
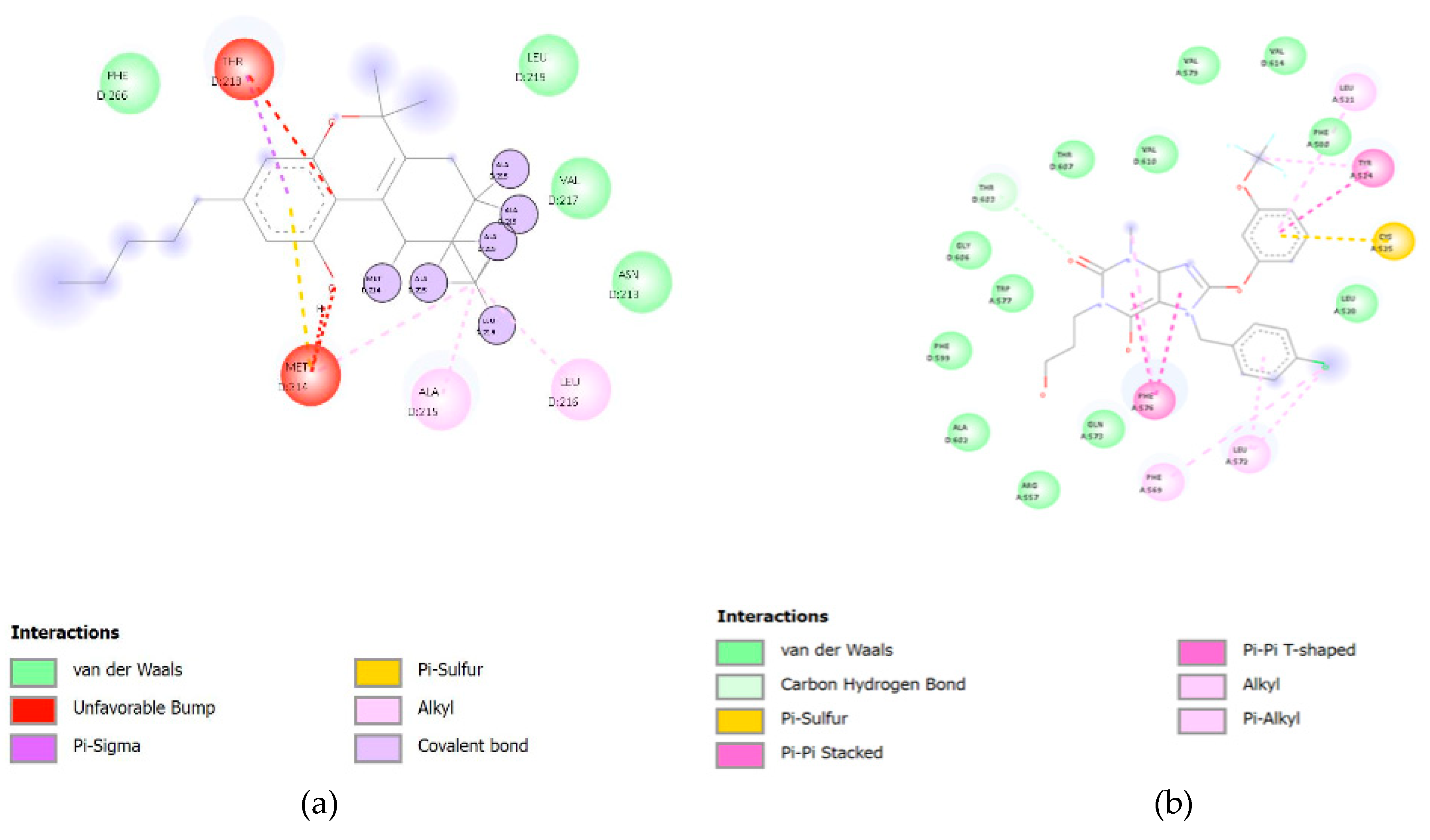
| Ion Channel | PDB ID | CBC (kcal/mol) | CBL (kcal/mol) | CBN (kcal/mol) |
|---|---|---|---|---|
| Rat TRPV1 | 3J5Q | −5.7 | −6.0 | −5.2 |
| Rat TRPV1 | 3J5R | −7.6 | −8.3 | −8.3 |
| Human TRPV1 | 6L93 | −7.4 | −7.9 | −8.0 |
| Human TRPC5 | 6YSN | −4.2 | −4.5 | −5.0± |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




