Submitted:
26 January 2026
Posted:
27 January 2026
You are already at the latest version
Abstract
Keywords:
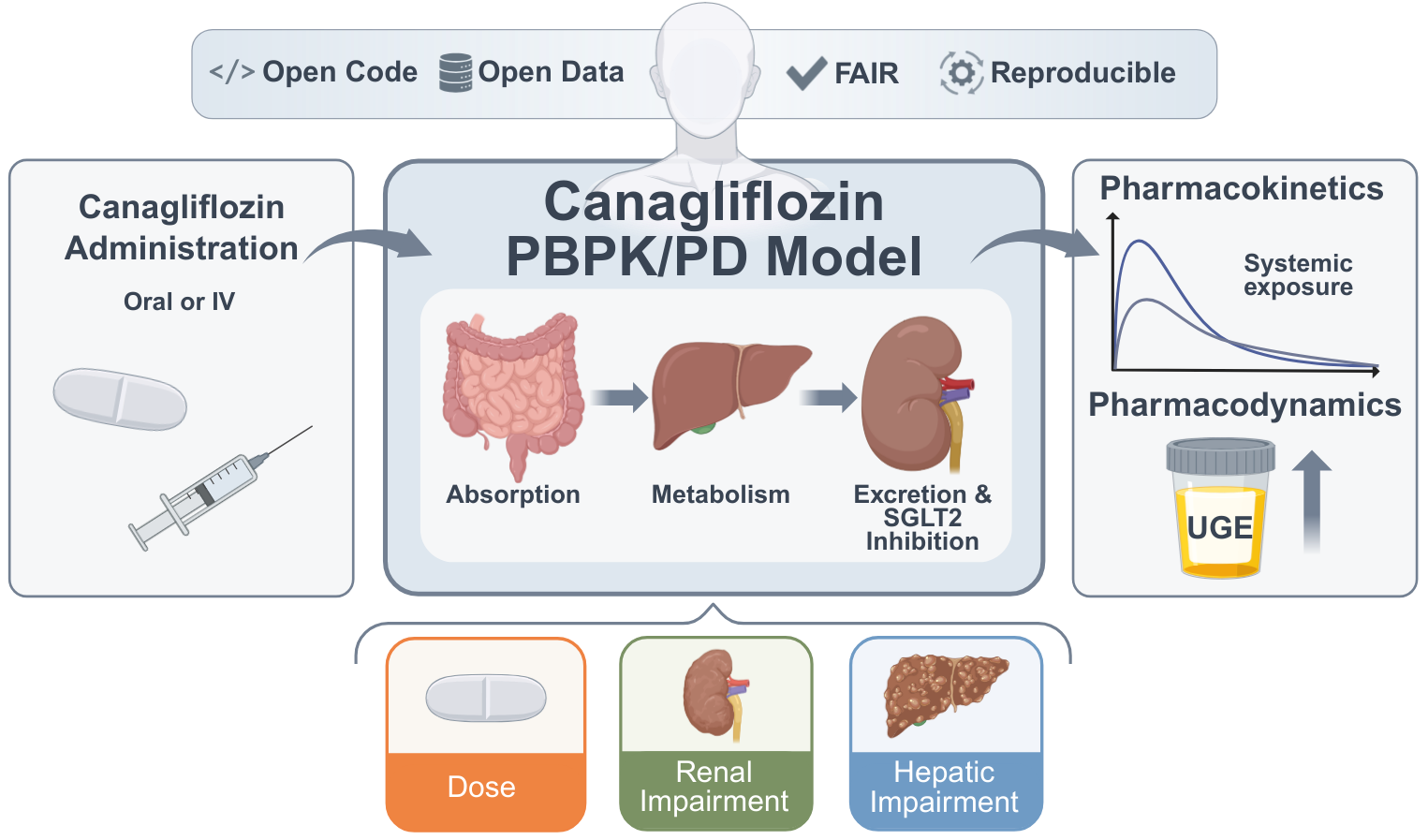
1. Introduction
2. Materials and Methods
2.1. Systematic Literature Research and Data Curation
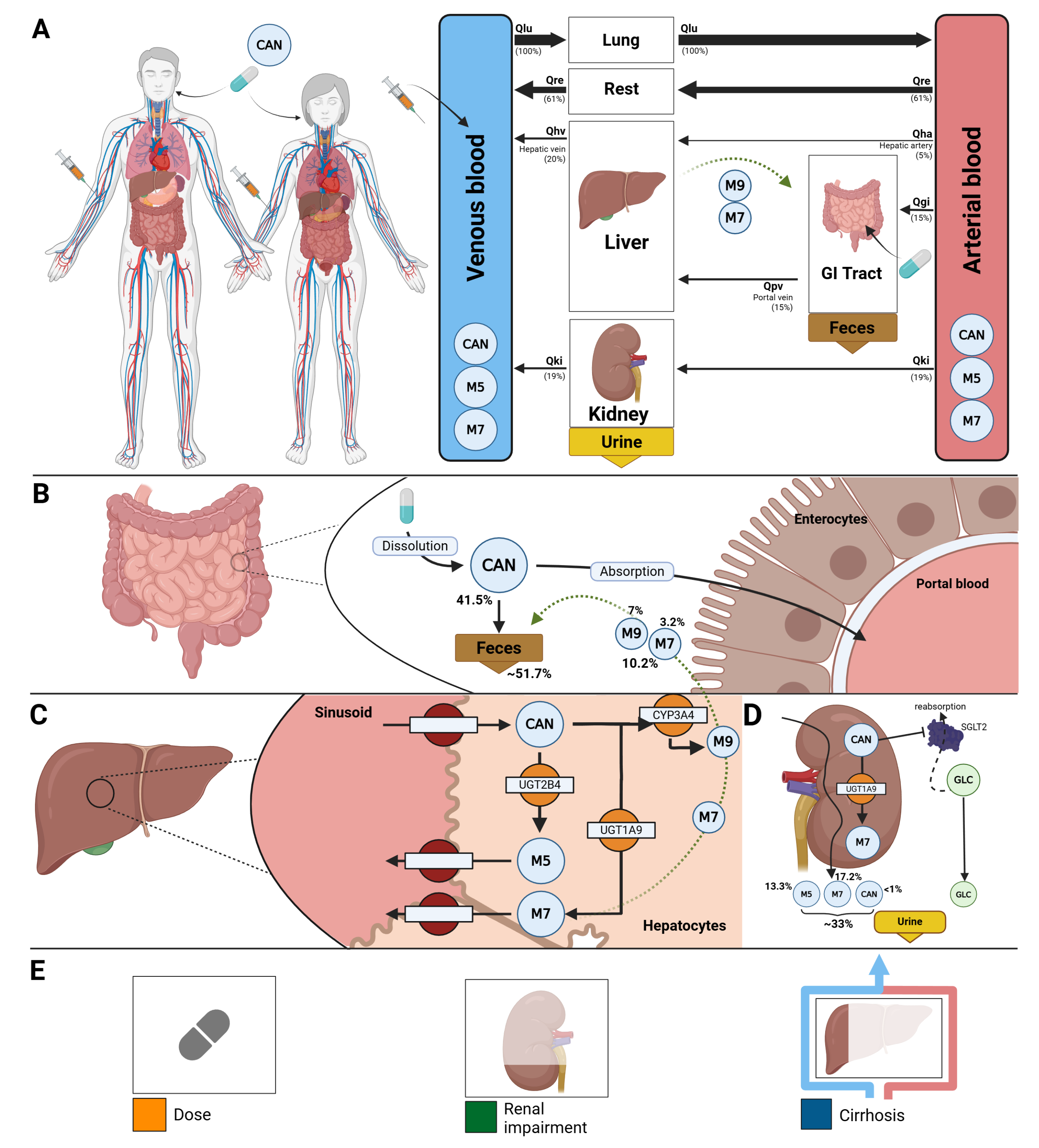
2.2. Computational Model
2.3. Model Assumptions
- Canagliflozin absorption was modeled as a first-order process.
- Diurnal variation in plasma glucose concentrations was not modeled explicitly. Instead, a constant fasting plasma glucose (FPG) concentration was assumed and used for the calculation of UGE. When reported, study-specific FPG values were used. Otherwise, FPG values of 5 mM for healthy subjects, 8.0 mM for subjects with T1DM, and 8.0 mM for subjects with T2DM were assumed.
- The RTG was parameterized using parameter optimization, with optimized values reported in Supplementary Table S3.
- Renal filtration and tubular glucose reabsorption were not modelled explicitly. Renal elimination of canagliflozin (CAN) and its metabolites was instead described using first-order processes, depending on kidney volume, renal function (GFR), and compound-specific excretion rate constants. The parameters KI__CANEX_k, KI__M5EX_k and KI__M7EX_k were estimated via parameter optimization.
- The conversion of CAN to M5 by UGT2B4 and to M7 by UGT1A9 in the liver and kidneys was modelled using irreversible Michaelis–Menten kinetics.
2.4. Model Parameterization
2.5. Simulations
2.6. Pharmacokinetic and Pharmacodynamic Parameters
2.7. Sensitivity Analysis
2.7.1. Sampling-Based Sensitivity Analysis
2.7.2. Local Sensitivity Analysis
2.7.3. Global Sensitivity Analysis
3. Results
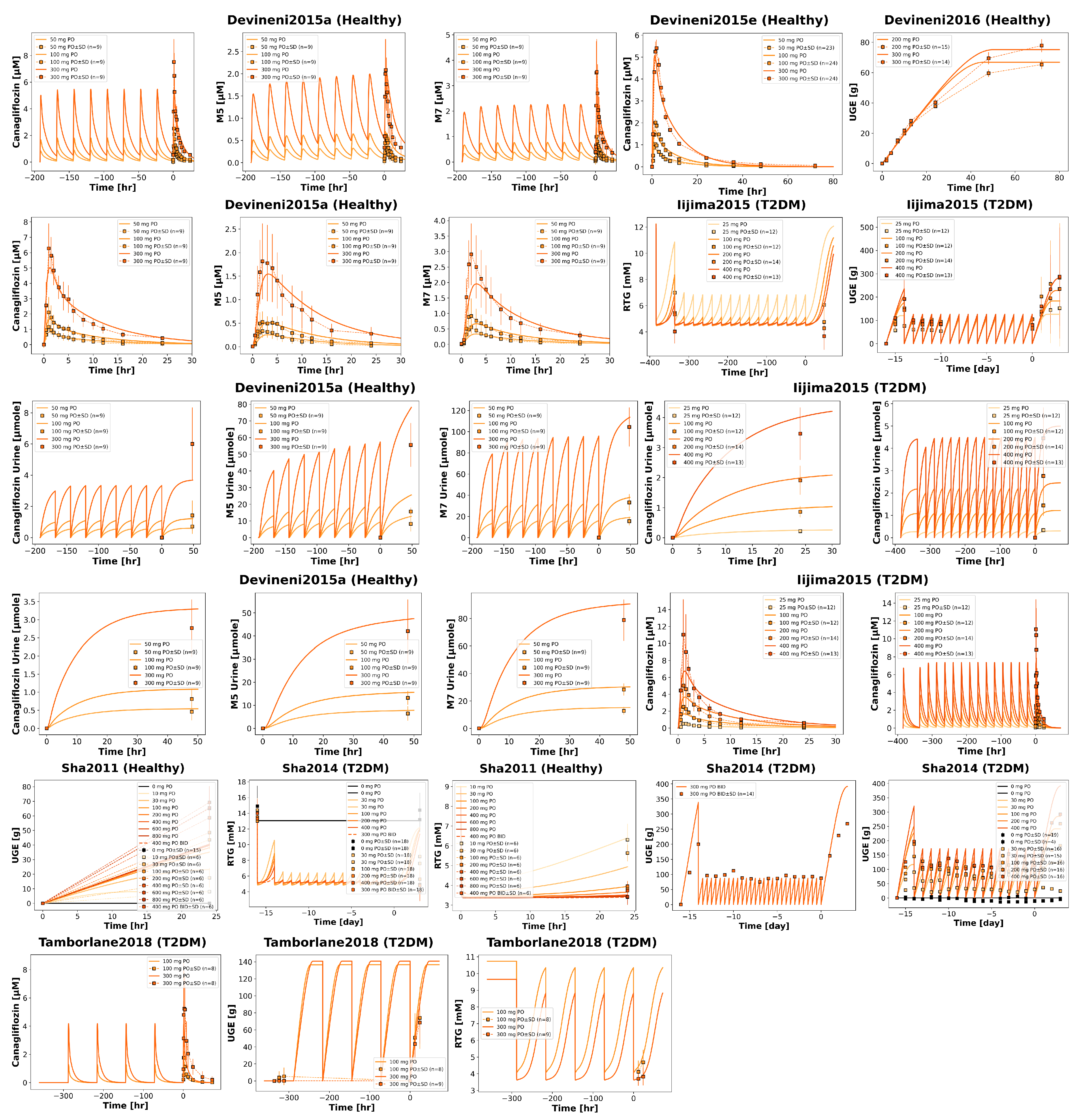
3.1. Canagliflozin Database
3.2. Computational Model
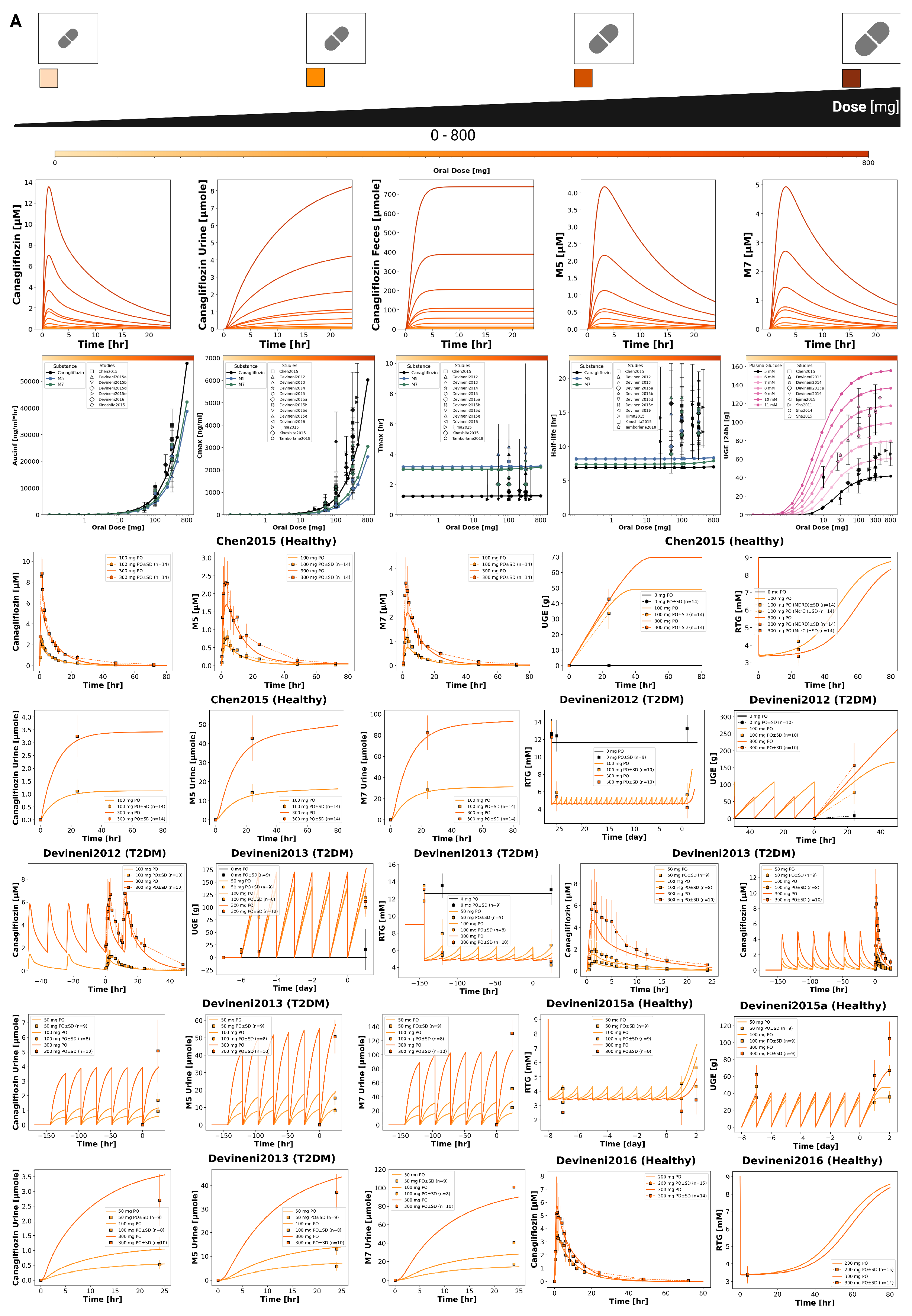
3.3. Dose Dependency
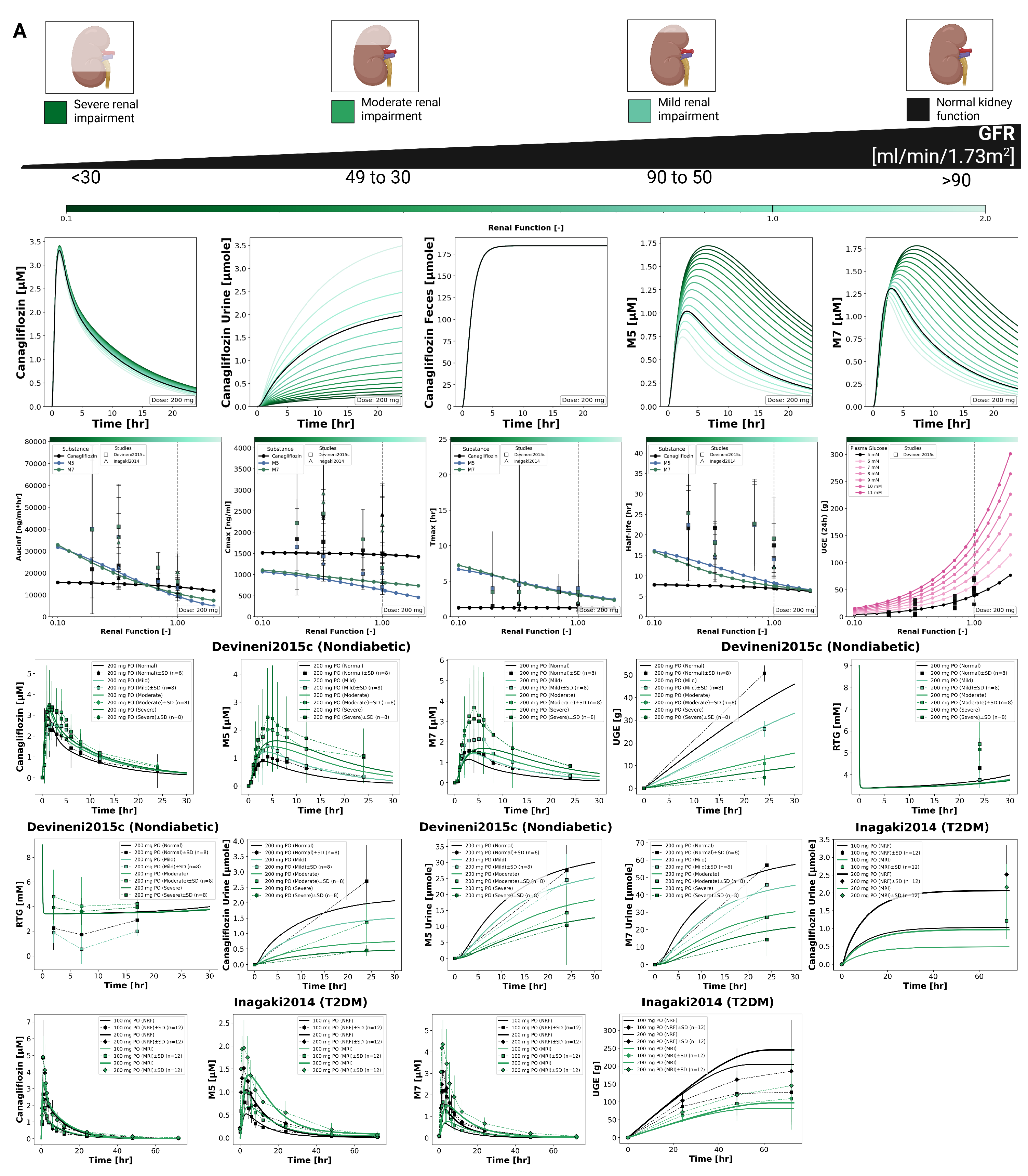
3.4. Renal Impairment
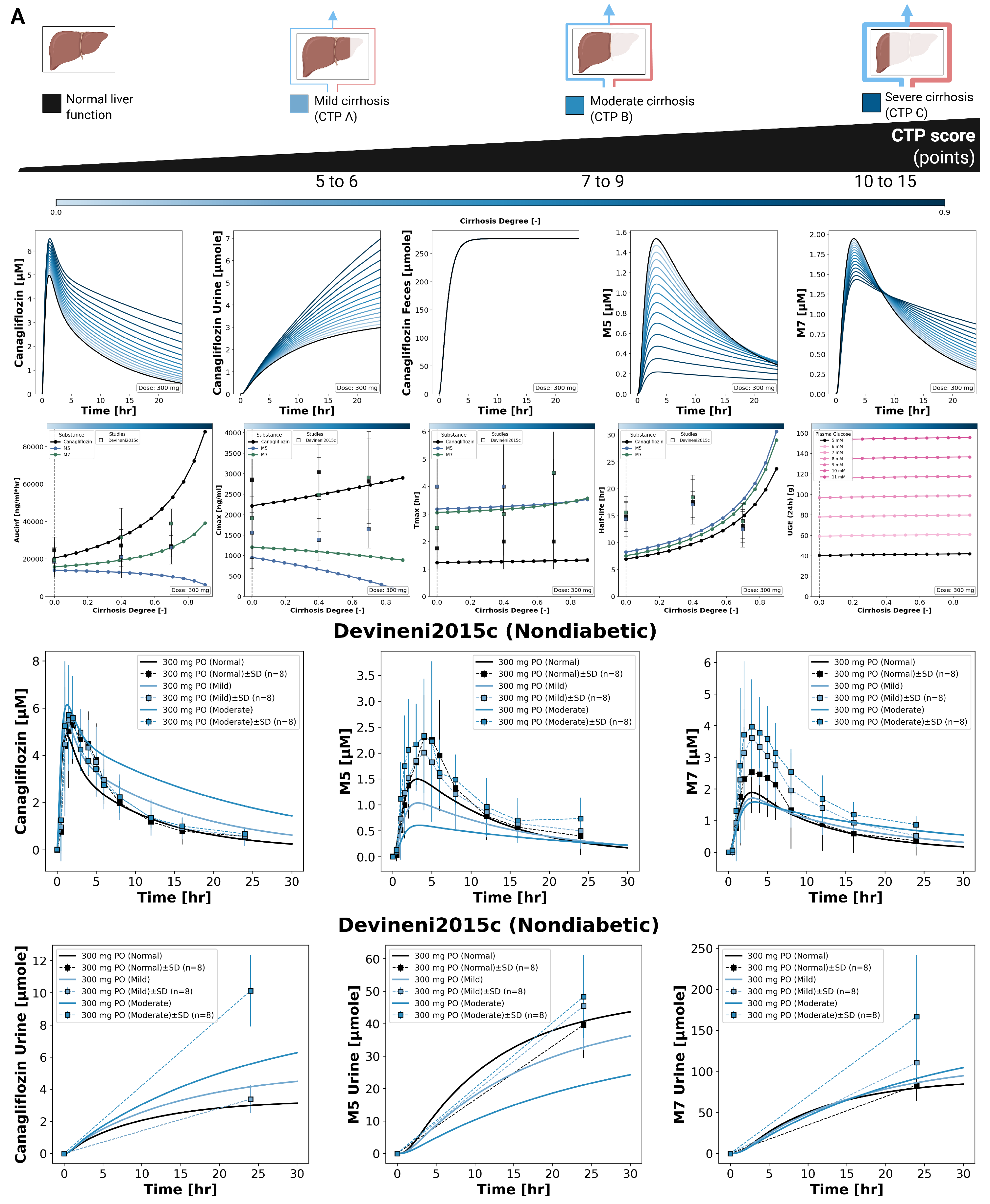
3.5. Hepatic Impairment
| Study | PK-DB | PMID | Route | Dosing | Dose [mg] | Healthy | Renal impairment | Hepatic impairment | T2DM |
|---|---|---|---|---|---|---|---|---|---|
| Chen2015 [46] | PKDB00858 | 26048186 | PO | single | 100, 300 | ✓ | |||
| Devineni2012 [47] | PKDB00874 | 22226086 | PO | single, multi | 100, 300 | ✓ | |||
| Devineni2013 [48] | PKDB00875 | 23670707 | PO | single, multi | 50, 100, 300 | ✓ | |||
| Devineni2014 [56] | PKDB00879 | 24726680 | PO | multi | 300 | ✓ | |||
| Devineni2015 [57] | PKDB00880 | 27140803 | PO | single | 300 | ✓ | |||
| Devineni2015a [49] | PKDB00876 | 25500487 | PO | single, multi | 50, 100, 300 | ✓ | |||
| Devineni2015b [58] | PKDB00877 | 27136910 | PO, IV | single | 0.010, 300 | ✓ | |||
| Devineni2015c [9] | PKDB00881 | 25659911 | PO | single | 300 | ✓ | ✓ | ✓ | |
| Devineni2015d [59] | PKDB00882 | 25407255 | PO | single, multi | 300 | ✓ | |||
| Devineni2015e [60] | PKDB00883 | 27136908 | PO | single | 50, 100, 300 | ✓ | |||
| Devineni2016 [50] | PKDB00884 | 26687552 | PO | single | 200, 300 | ✓ | |||
| Iijima2015 [51] | PKDB00885 | 26280756 | PO | single, multi | 25, 100, 200, 400 | ✓ | |||
| Inagaki2014 [55] | PKDB00878 | 25200141 | PO | single | 100, 200 | ✓ | ✓ | ||
| Kinoshita2015 [61] | PKDB00886 | 25424014 | PO | single, multi | 200 | ✓ | |||
| Mamidi2014 [62] | PKDB00887 | 24568888 | PO | single | 188 | ✓ | |||
| Mohamed2019 [63] | PKDB00888 | 31384829 | PO | single | 50 | ✓ | |||
| Murphy2015 [64] | PKDB00857 | 25546166 | PO | single | 150 | ✓ | |||
| Sha2011 [52] | PKDB00889 | 21457428 | PO | single, multi | 10, 30, 100, 200, 400, 600, 800 | ✓ | |||
| Sha2014 [53] | PKDB00890 | 25166023 | PO | single, multi | 30, 100, 200, 300, 400 | ✓ | |||
| Sha2015 [65] | PKDB00891 | 25421015 | PO | multi | 300 | ✓ | |||
| Tamborlane2018 [54] | PKDB00892 | 29271103 | PO | multi | 100, 300 | ✓ | |||
| Wattamwar2020 [66] | PKDB01127 | 32763846 | PO | multi | 150 | ✓ |
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- American Diabetes Association Professional Practice Committee. 2. Diagnosis and Classification of Diabetes: Standards of Care in Diabetes-2024. Diabetes Care 2024, 47, S20–S42. [Google Scholar] [CrossRef]
- Institute for Health Metrics and Evaluation (IHME). Global Burden of Disease 2021: Findings from the GBD 2021 Study; Technical report; Institute for Health Metrics and Evaluation, University of Washington, 2021. [Google Scholar]
- Vallon, V. Glucose Transporters in the Kidney in Health and Disease. Pflugers Archiv: European Journal of Physiology 2020, 472, 1345–1370. [Google Scholar] [CrossRef]
- DeFronzo, R.A.; Hompesch, M.; Kasichayanula, S.; Liu, X.; Hong, Y.; Pfister, M.; Morrow, L.A.; Leslie, B.R.; Boulton, D.W.; Ching, A.; et al. Characterization of Renal Glucose Reabsorption in Response to Dapagliflozin in Healthy Subjects and Subjects with Type 2 Diabetes. Diabetes Care 2013, 36, 3169–3176. [Google Scholar] [CrossRef]
- Nisly, S.A.; Kolanczyk, D.M.; Walton, A.M. Canagliflozin, a New Sodium-Glucose Cotransporter 2 Inhibitor, in the Treatment of Diabetes. American journal of health-system pharmacy: AJHP: official journal of the American Society of Health-System Pharmacists 2013, 70, 311–319. [Google Scholar] [CrossRef]
- Deeks, E.D.; Scheen, A.J. Canagliflozin: A Review in Type 2 Diabetes. Drugs 2017, 77, 1577–1592. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. Invokana (Canagliflozin) Tablets: Full Prescribing Information. 2024. [Google Scholar]
- Perkovic, V.; Jardine, M.; Vijapurkar, U.; Meininger, G. Renal Effects of Canagliflozin in Type 2 Diabetes Mellitus. Current medical research and opinion 2015, 31, 2219–2231. [Google Scholar] [CrossRef]
- Devineni, D.; Curtin, C.R.; Marbury, T.C.; Smith, W.; Vaccaro, N.; Wexler, D.; Vandebosch, A.; Rusch, S.; Stieltjes, H.; Wajs, E. Effect of Hepatic or Renal Impairment on the Pharmacokinetics of Canagliflozin, a Sodium Glucose Co-Transporter 2 Inhibitor. Clinical therapeutics 2015, 37, 610–628.e4. [Google Scholar] [CrossRef]
- Hoeben, E.; De Winter, W.; Neyens, M.; Devineni, D.; Vermeulen, A.; Dunne, A. Population Pharmacokinetic Modeling of Canagliflozin in Healthy Volunteers and Patients with Type 2 Diabetes Mellitus. Clinical pharmacokinetics 2016, 55, 209–223. [Google Scholar] [CrossRef]
- de Winter, W.; Dunne, A.; de Trixhe, X.W.; Devineni, D.; Hsu, C.H.; Pinheiro, J.; Polidori, D. Dynamic Population Pharmacokinetic-Pharmacodynamic Modelling and Simulation Supports Similar Efficacy in Glycosylated Haemoglobin Response with Once or Twice-Daily Dosing of Canagliflozin. British journal of clinical pharmacology 2017, 83, 1072–1081. [Google Scholar] [CrossRef]
- Dunne, A.; de Winter, W.; Hsu, C.H.; Mariam, S.; Neyens, M.; Pinheiro, J.; Woot de Trixhe, X. The Method of Averaging Applied to Pharmacokinetic/Pharmacodynamic Indirect Response Models. Journal of pharmacokinetics and pharmacodynamics 2015, 42, 417–426. [Google Scholar] [CrossRef]
- Mori, K.; Saito, R.; Nakamaru, Y.; Shimizu, M.; Yamazaki, H. Physiologically Based Pharmacokinetic-Pharmacodynamic Modeling to Predict Concentrations and Actions of Sodium-Dependent Glucose Transporter 2 Inhibitor Canagliflozin in Human Intestines and Renal Tubules. Biopharmaceutics & drug disposition 2016, 37, 491–506. [Google Scholar] [CrossRef]
- Mori-Anai, K.; Tashima, Y.; Nakada, T.; Nakamaru, Y.; Takahata, T.; Saito, R. Mechanistic Evaluation of the Effect of Sodium-Dependent Glucose Transporter 2 Inhibitors on Delayed Glucose Absorption in Patients with Type 2 Diabetes Mellitus Using a Quantitative Systems Pharmacology Model of Human Systemic Glucose Dynamics. Biopharmaceutics & drug disposition 2020, 41, 352–366. [Google Scholar] [CrossRef]
- Mamidi, R.N.V.S.; Dallas, S.; Sensenhauser, C.; Lim, H.K.; Scheers, E.; Verboven, P.; Cuyckens, F.; Leclercq, L.; Evans, D.C.; Kelley, M.F.; et al. In Vitro and Physiologically-Based Pharmacokinetic Based Assessment of Drug-Drug Interaction Potential of Canagliflozin. British journal of clinical pharmacology 2017, 83, 1082–1096. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Zhou, J.; Song, L.; Ren, Y.; Hu, P.; Liu, D. A Model-Based Meta Analysis Study of Sodium Glucose Co-Transporter-2 Inhibitors. CPT: pharmacometrics & systems pharmacology 2023, 12, 487–499. [Google Scholar] [CrossRef]
- Tiwari, K.; Kananathan, S.; Roberts, M.G.; Meyer, J.P.; Sharif Shohan, M.U.; Xavier, A.; Maire, M.; Zyoud, A.; Men, J.; Ng, S.; et al. Reproducibility in Systems Biology Modelling. Molecular Systems Biology 2021, 17, e9982. [Google Scholar] [CrossRef]
- Domínguez-Romero, E.; Mazurenko, S.; Scheringer, M.; Martins Dos Santos, V.A.P.; Evelo, C.T.; Anton, M.; Hancock, J.M.; Županič, A.; Suarez-Diez, M. Making PBPK Models More Reproducible in Practice. Briefings in Bioinformatics 2024, 25, bbae569. [Google Scholar] [CrossRef]
- Wilkinson, M.D.; Dumontier, M.; Aalbersberg, I.J.J.; Appleton, G.; Axton, M.; Baak, A.; Blomberg, N.; Boiten, J.W.; da Silva Santos, L.B.; Bourne, P.E.; et al. The FAIR Guiding Principles for Scientific Data Management and Stewardship. Scientific Data 2016, 3, 160018. [Google Scholar] [CrossRef]
- Jones, H.; Rowland-Yeo, K. Basic Concepts in Physiologically Based Pharmacokinetic Modeling in Drug Discovery and Development. CPT: Pharmacometrics & Systems Pharmacology 2013, 2, 1–12. [Google Scholar] [CrossRef]
- Sager, J.E.; Yu, J.; Ragueneau-Majlessi, I.; Isoherranen, N. Physiologically Based Pharmacokinetic (PBPK) Modeling and Simulation Approaches: A Systematic Review of Published Models, Applications, and Model Verification. Drug Metabolism and Disposition 2015, 43, 1823–1837. [Google Scholar] [CrossRef]
- Hartmanshenn, C.; Scherholz, M.; Androulakis, I.P. Physiologically-Based Pharmacokinetic Models: Approaches for Enabling Personalized Medicine. Journal of Pharmacokinetics and Pharmacodynamics 2016, 43, 481–504. [Google Scholar] [CrossRef]
- Hucka, M.; Bergmann, F.T.; Chaouiya, C.; Dräger, A.; Hoops, S.; Keating, S.M.; König, M.; Novère, N.L.; Myers, C.J.; Olivier, B.G.; et al. The Systems Biology Markup Language (SBML): Language Specification for Level 3 Version 2 Core Release 2. Journal of Integrative Bioinformatics 2019, 16, 20190021. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez Hernandez, F.; Carter, S.J.; Iso-Sipilä, J.; Goldsmith, P.; Almousa, A.A.; Gastine, S.; Lilaonitkul, W.; Kloprogge, F.; Standing, J.F. An Automated Approach to Identify Scientific Publications Reporting Pharmacokinetic Parameters. Wellcome Open Research 2021, 6, 88. [Google Scholar] [CrossRef]
- Grzegorzewski, J.; Brandhorst, J.; Green, K.; Eleftheriadou, D.; Duport, Y.; Barthorscht, F.; Köller, A.; Ke, D.Y.J.; De Angelis, S.; König, M. PK-DB: Pharmacokinetics Database for Individualized and Stratified Computational Modeling. Nucleic Acids Research 2021, 49, D1358–D1364, [33151297. [Google Scholar] [CrossRef] [PubMed]
- Rohatgi, A. WebPlotDigitizer. 2024. [Google Scholar]
- Keating, S.M.; Waltemath, D.; König, M.; Zhang, F.; Dräger, A.; Chaouiya, C.; Bergmann, F.T.; Finney, A.; Gillespie, C.S.; Helikar, T.; et al. SBML Level 3: An Extensible Format for the Exchange and Reuse of Biological Models. Molecular Systems Biology 2020, 16, e9110. [Google Scholar] [CrossRef]
- König, M. Sbmlutils: Python Utilities for SBML; Zenodo, 2024. [Google Scholar] [CrossRef]
- König, M.; Dräger, A.; Holzhütter, H.G. CySBML: A Cytoscape Plugin for SBML. Bioinformatics (Oxford, England) 2012, 28, 2402–2403, [22772946]. [Google Scholar] [CrossRef]
- König, M. Cy3sbml - SBML for Cytoscape; Zenodo, 2025. [Google Scholar] [CrossRef]
- König, M. Sbmlsim: SBML Simulation Made Easy. [object Object]. 2021. [Google Scholar] [CrossRef]
- Somogyi, E.T.; Bouteiller, J.M.; Glazier, J.A.; König, M.; Medley, J.K.; Swat, M.H.; Sauro, H.M. libRoadRunner: A High Performance SBML Simulation and Analysis Library. Bioinformatics (Oxford, England) 2015, 31, 3315–3321. [Google Scholar] [CrossRef]
- Welsh, C.; Xu, J.; Smith, L.; König, M.; Choi, K.; Sauro, H.M. libRoadRunner 2.0: A High Performance SBML Simulation and Analysis Library. Bioinformatics 2023, 39, btac770. [Google Scholar] [CrossRef]
- Tereshchuk, V.; Elias, M.; König, M. Physiologically Based Pharmacokinetic/ Pharmacodynamic (PBPK/PD) Model of Canagliflozin. Zenodo 2026. [Google Scholar] [CrossRef]
- Stevens, P.E.; Ahmed, S.B.; Carrero, J.J.; Foster, B.; Francis, A.; Hall, R.K.; Herrington, W.G.; Hill, G.; Inker, L.A.; Kazancıoğlu, R.; et al. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney International 2024, 105, S117–S314. [Google Scholar] [CrossRef] [PubMed]
- Mallol, B.S.; Grzegorzewski, J.; Tautenhahn, H.M.; König, M. Insights into Intestinal P-glycoprotein Function Using Talinolol: A PBPK Modeling Approach. 2023. [Google Scholar] [CrossRef]
- Child, C.G.; Turcotte, J.G. Surgery and Portal Hypertension. Major Problems in Clinical Surgery 1964, 1, 1–85. [Google Scholar]
- Pugh, R.N.; Murray-Lyon, I.M.; Dawson, J.L.; Pietroni, M.C.; Williams, R. Transection of the Oesophagus for Bleeding Oesophageal Varices. The British Journal of Surgery 1973, 60, 646–649. [Google Scholar] [CrossRef] [PubMed]
- Köller, A.; Grzegorzewski, J.; König, M. Physiologically Based Modeling of the Effect of Physiological and Anthropometric Variability on Indocyanine Green Based Liver Function Tests. Frontiers in Physiology 2021, 2043. [Google Scholar] [CrossRef]
- Köller, A.; Grzegorzewski, J.; Tautenhahn, H.M.; König, M. Prediction of Survival After Partial Hepatectomy Using a Physiologically Based Pharmacokinetic Model of Indocyanine Green Liver Function Tests. Frontiers in physiology 2021, 12, 730418. [Google Scholar] [CrossRef]
- Sobol′, I. Global Sensitivity Indices for Nonlinear Mathematical Models and Their Monte Carlo Estimates. Mathematics and Computers in Simulation 2001, 55, 271–280. [Google Scholar] [CrossRef]
- Saltelli, A. Making Best Use of Model Evaluations to Compute Sensitivity Indices. Computer Physics Communications 2002, 145, 280–297. [Google Scholar] [CrossRef]
- Saltelli, A.; Annoni, P.; Azzini, I.; Campolongo, F.; Ratto, M.; Tarantola, S. Variance Based Sensitivity Analysis of Model Output. Design and Estimator for the Total Sensitivity Index. Computer Physics Communications 2010, 181, 259–270. [Google Scholar] [CrossRef]
- Herman, J.; Usher, W. SALib: An Open-Source Python Library for Sensitivity Analysis. The Journal of Open Source Software 2017, 2. [Google Scholar] [CrossRef]
- Iwanaga, T.; Usher, W.; Herman, J. Toward SALib 2.0: Advancing the Accessibility and Interpretability of Global Sensitivity Analyses. Socio-Environmental Systems Modelling 2022, 4, 18155. [Google Scholar] [CrossRef]
- Chen, X.; Hu, P.; Vaccaro, N.; Polidori, D.; Curtin, C.R.; Stieltjes, H.; Sha, S.; Weiner, S.; Devineni, D. Pharmacokinetics, Pharmacodynamics, and Safety of Single-Dose Canagliflozin in Healthy Chinese Subjects. Clinical therapeutics 2015, 37, 1483–1492.e1. [Google Scholar] [CrossRef] [PubMed]
- Devineni, D.; Morrow, L.; Hompesch, M.; Skee, D.; Vandebosch, A.; Murphy, J.; Ways, K.; Schwartz, S. Canagliflozin Improves Glycaemic Control over 28 Days in Subjects with Type 2 Diabetes Not Optimally Controlled on Insulin. Diabetes, obesity & metabolism 2012, 14, 539–545. [Google Scholar] [CrossRef]
- Devineni, D.; Curtin, C.R.; Polidori, D.; Gutierrez, M.J.; Murphy, J.; Rusch, S.; Rothenberg, P.L. Pharmacokinetics and Pharmacodynamics of Canagliflozin, a Sodium Glucose Co-Transporter 2 Inhibitor, in Subjects with Type 2 Diabetes Mellitus. Journal of clinical pharmacology 2013, 53, 601–610. [Google Scholar] [CrossRef]
- Devineni, D.; Vaccaro, N.; Polidori, D.; Stieltjes, H.; Wajs, E. Single- and Multiple-Dose Pharmacokinetics and Pharmacodynamics of Canagliflozin, a Selective Inhibitor of Sodium Glucose Co-Transporter 2, in Healthy Participants. International journal of clinical pharmacology and therapeutics 2015, 53, 129–138. [Google Scholar] [CrossRef]
- Devineni, D.; Polidori, D.; Curtin, C.; Stieltjes, H.; Tian, H.; Wajs, E. Single-Dose Pharmacokinetics and Pharmacodynamics of Canagliflozin, a Selective Inhibitor of Sodium Glucose Cotransporter 2, in Healthy Indian Participants. Clinical therapeutics 2016, 38, 89–98.e1. [Google Scholar] [CrossRef] [PubMed]
- Iijima, H.; Kifuji, T.; Maruyama, N.; Inagaki, N. Pharmacokinetics, Pharmacodynamics, and Safety of Canagliflozin in Japanese Patients with Type 2 Diabetes Mellitus. Advances in therapy 2015, 32, 768–782. [Google Scholar] [CrossRef] [PubMed]
- Sha, S.; Devineni, D.; Ghosh, A.; Polidori, D.; Chien, S.; Wexler, D.; Shalayda, K.; Demarest, K.; Rothenberg, P. Canagliflozin, a Novel Inhibitor of Sodium Glucose Co-Transporter 2, Dose Dependently Reduces Calculated Renal Threshold for Glucose Excretion and Increases Urinary Glucose Excretion in Healthy Subjects. Diabetes, Obesity & Metabolism 2011, 13, 669–672. [Google Scholar] [CrossRef]
- Sha, S.; Devineni, D.; Ghosh, A.; Polidori, D.; Hompesch, M.; Arnolds, S.; Morrow, L.; Spitzer, H.; Demarest, K.; Rothenberg, P. Pharmacodynamic Effects of Canagliflozin, a Sodium Glucose Co-Transporter 2 Inhibitor, from a Randomized Study in Patients with Type 2 Diabetes. PloS one 2014, 9, e110069. [Google Scholar] [CrossRef]
- Tamborlane, W.V.; Polidori, D.; Argenti, D.; Di Prospero, N.A. Pharmacokinetics and Pharmacodynamics of Canagliflozin in Pediatric Patients with Type 2 Diabetes. Pediatric diabetes 2018, 19, 649–655. [Google Scholar] [CrossRef]
- Inagaki, N.; Kondo, K.; Yoshinari, T.; Ishii, M.; Sakai, M.; Kuki, H.; Furihata, K. Pharmacokinetic and Pharmacodynamic Profiles of Canagliflozin in Japanese Patients with Type 2 Diabetes Mellitus and Moderate Renal Impairment. Clinical drug investigation 2014, 34, 731–742. [Google Scholar] [CrossRef]
- Devineni, D.; Vaccaro, N.; Polidori, D.; Rusch, S.; Wajs, E. Effects of Hydrochlorothiazide on the Pharmacokinetics, Pharmacodynamics, and Tolerability of Canagliflozin, a Sodium Glucose Co-Transporter 2 Inhibitor, in Healthy Participants. Clinical therapeutics 2014, 36, 698–710. [Google Scholar] [CrossRef] [PubMed]
- Devineni, D.; Manitpisitkul, P.; Murphy, J.; Skee, D.; Wajs, E.; Mamidi, R.N.V.S.; Tian, H.; Vandebosch, A.; Wang, S.S.; Verhaeghe, T.; et al. Effect of Canagliflozin on the Pharmacokinetics of Glyburide, Metformin, and Simvastatin in Healthy Participants. Clinical pharmacology in drug development 2015, 4, 226–236. [Google Scholar] [CrossRef]
- Devineni, D.; Murphy, J.; Wang, S.S.; Stieltjes, H.; Rothenberg, P.; Scheers, E.; Mamidi, R.N.V.S. Absolute Oral Bioavailability and Pharmacokinetics of Canagliflozin: A Microdose Study in Healthy Participants. Clinical pharmacology in drug development 2015, 4, 295–304. [Google Scholar] [CrossRef]
- Devineni, D.; Vaccaro, N.; Murphy, J.; Curtin, C.; Mamidi, R.N.V.S.; Weiner, S.; Wang, S.S.; Ariyawansa, J.; Stieltjes, H.; Wajs, E.; et al. Effects of Rifampin, Cyclosporine A, and Probenecid on the Pharmacokinetic Profile of Canagliflozin, a Sodium Glucose Co-Transporter 2 Inhibitor, in Healthy Participants. International journal of clinical pharmacology and therapeutics 2015, 53, 115–128. [Google Scholar] [CrossRef]
- Devineni, D.; Manitpisitkul, P.; Murphy, J.; Stieltjes, H.; Ariyawansa, J.; Di Prospero, N.A.; Rothenberg, P. Effect of Food on the Pharmacokinetics of Canagliflozin, a Sodium Glucose Co-Transporter 2 Inhibitor, and Assessment of Dose Proportionality in Healthy Participants. Clinical pharmacology in drug development 2015, 4, 279–286. [Google Scholar] [CrossRef]
- Kinoshita, S.; Kondo, K. Evaluation of Pharmacokinetic and Pharmacodynamic Interactions of Canagliflozin and Teneligliptin in Japanese Healthy Male Volunteers. Expert opinion on drug metabolism & toxicology 2015, 11, 7–14. [Google Scholar] [CrossRef]
- Mamidi, R.N.V.S.; Cuyckens, F.; Chen, J.; Scheers, E.; Kalamaridis, D.; Lin, R.; Silva, J.; Sha, S.; Evans, D.C.; Kelley, M.F.; et al. Metabolism and Excretion of Canagliflozin in Mice, Rats, Dogs, and Humans. Drug metabolism and disposition: the biological fate of chemicals 2014, 42, 903–916. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, D.; Elshahed, M.S.; Nasr, T.; Aboutaleb, N.; Zakaria, O. Novel LC-MS/MS Method for Analysis of Metformin and Canagliflozin in Human Plasma: Application to a Pharmacokinetic Study. BMC chemistry 2019, 13, 82. [Google Scholar] [CrossRef]
- Murphy, J.; Wang, S.S.; Stieltjes, H.; Wajs, E.; Devineni, D. Effect of Food on the Pharmacokinetics of Canagliflozin/Metformin (150/1,000 Mg) Immediate-Release Fixed-Dose Combination Tablet in Healthy Participants. International journal of clinical pharmacology and therapeutics 2015, 53, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Sha, S.; Polidori, D.; Farrell, K.; Ghosh, A.; Natarajan, J.; Vaccaro, N.; Pinheiro, J.; Rothenberg, P.; Plum-Mörschel, L. Pharmacodynamic Differences between Canagliflozin and Dapagliflozin: Results of a Randomized, Double-Blind, Crossover Study. Diabetes, obesity & metabolism 2015, 17, 188–197. [Google Scholar] [CrossRef]
- Wattamwar, T.; Mungantiwar, A.; Gujar, S.; Pandita, N. Development of LC-MS/MS Method for Simultaneous Determination of Canagliflozin and Metformin in Human Plasma and Its Pharmacokinetic Application in Indian Population under Fast and Fed Conditions. Journal of chromatography. B, Analytical technologies in the biomedical and life sciences 2020, 1154, 122281. [Google Scholar] [CrossRef] [PubMed]
- Francke, S.; Mamidi, R.N.V.S.; Solanki, B.; Scheers, E.; Jadwin, A.; Favis, R.; Devineni, D. In Vitro Metabolism of Canagliflozin in Human Liver, Kidney, Intestine Microsomes, and Recombinant Uridine Diphosphate Glucuronosyltransferases (UGT) and the Effect of Genetic Variability of UGT Enzymes on the Pharmacokinetics of Canagliflozin in Humans. Journal of clinical pharmacology 2015, 55, 1061–1072. [Google Scholar] [CrossRef] [PubMed]
- Balaur, I.; Nickerson, D.P.; Welter, D.; Wodke, J.A.; Ancien, F.; Gebhardt, T.; Grouès, V.; Hermjakob, H.; König, M.; Radde, N.; et al. FAIRification of Computational Models in Biology. 2025. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




