Submitted:
26 January 2026
Posted:
27 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Synthesis and Functionalization of Cucurbiturils for Gel Design
2.1. Classical Acid-Catalyzed Synthesis
2.2. Green and Improved Synthetic Routes
2.3. Functionalization Strategies
3. Design Principles of Cucurbituril-Based Supramolecular Polymer Gels
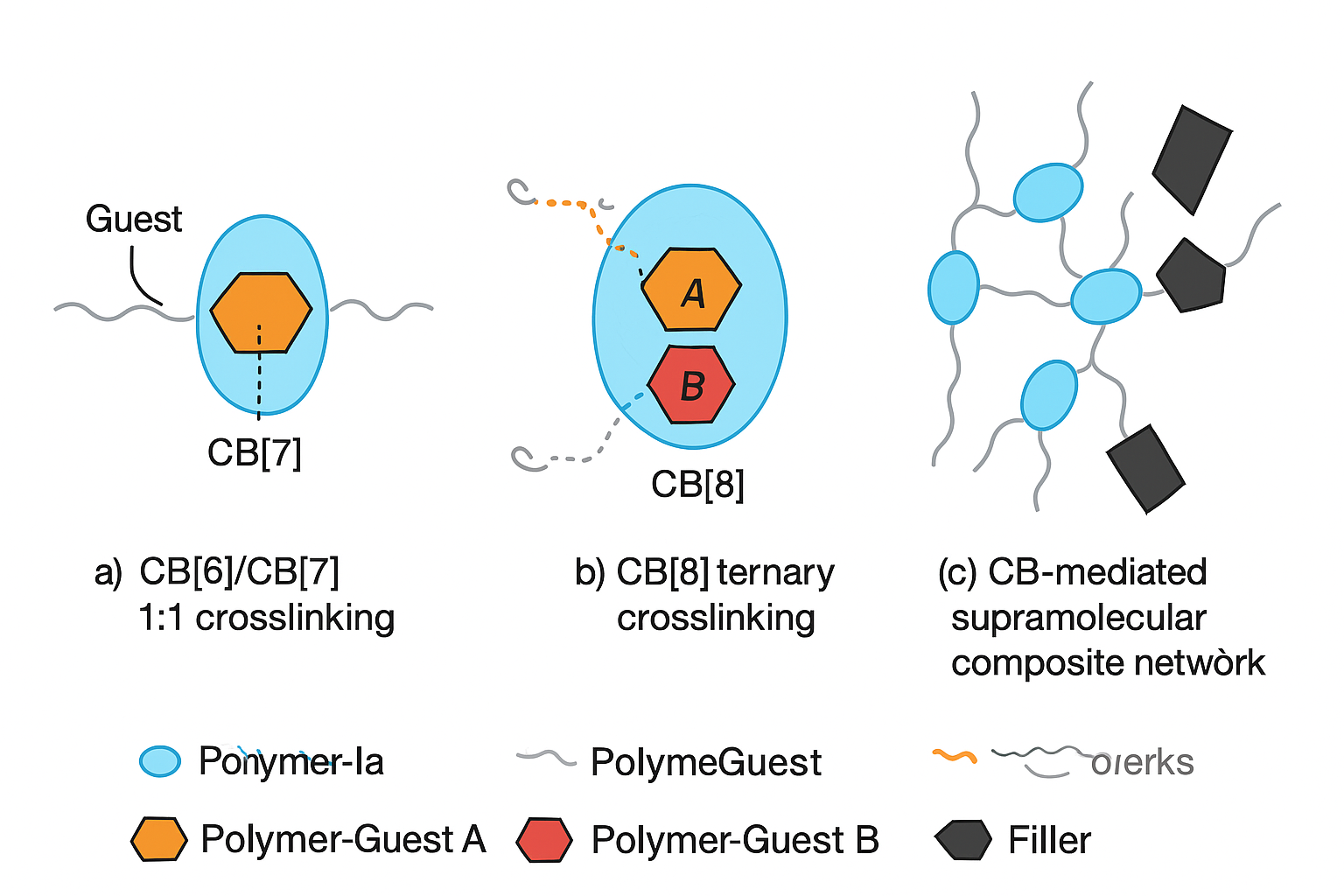
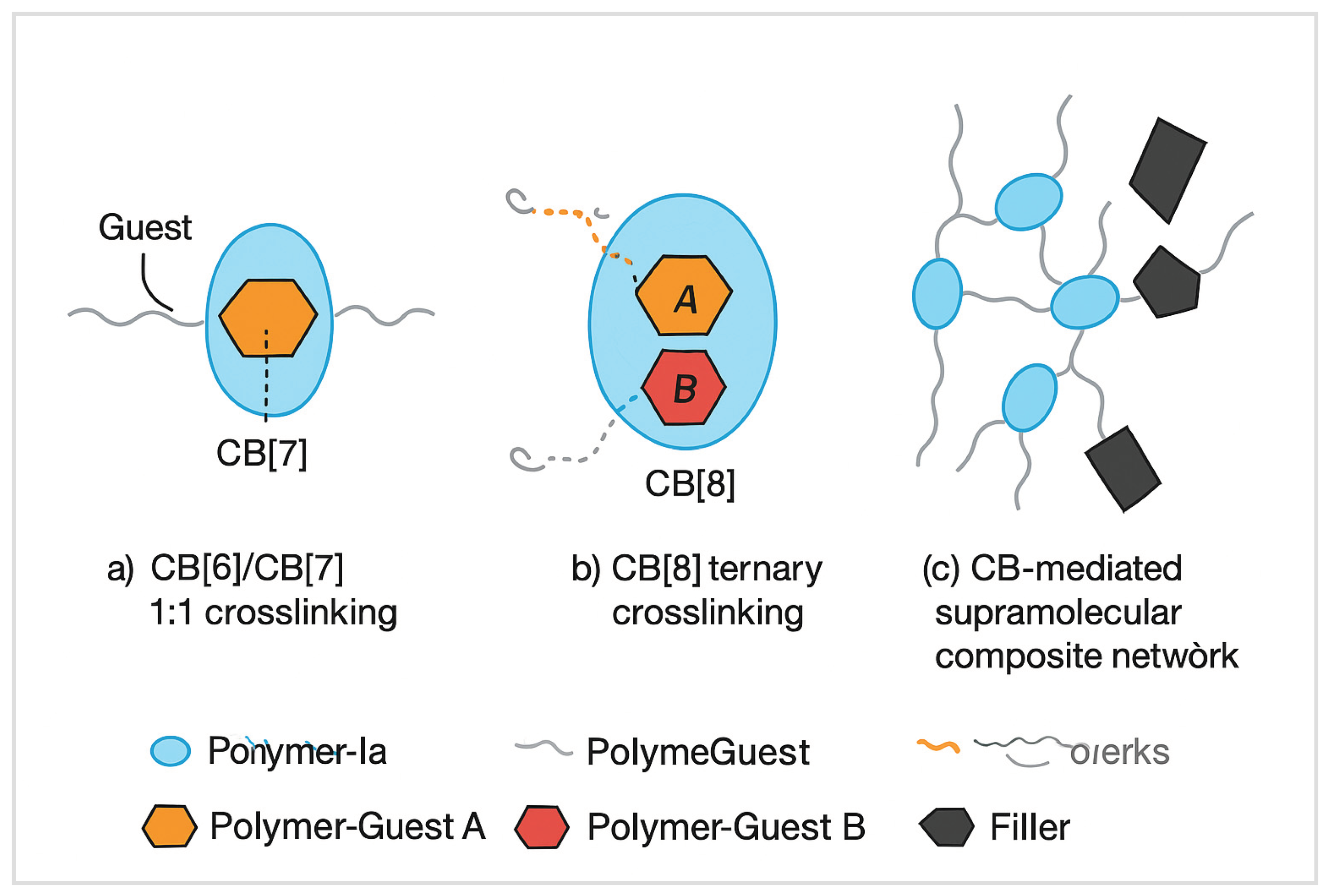
3.1. Network Architecture via Host–Guest Crosslinking
3.2. Dynamic Bonding and Self-Healing
3.3. Multistimuli Responsiveness
3.4. Polymer Backbone Selection and Topology
3.5. Composite Design: Integration with Secondary Fillers
4. Structure–Property Relationships in CB-Based Supramolecular Gels
4.1. Rheological Characterization
4.2. Mechanical Properties and Failure Behavior
4.3. Swelling and Solvent Uptake
5. Applications of Cucurbituril-Based Polymer Gel Composites
5.1. Biomedical Engineering and Drug Delivery
5.2. Soft Robotics and Actuators
5.3. Soft Electronics and Sensors
5.4. Separation and Environmental Remediation
6. Outlook and Future Directions
6.1. Scalable Green Synthesis
6.2. Advanced Processing and 4D Printing
6.3. Multi-Component Adaptive Composites
6.4. Mechanistic Understanding and Modeling
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peppas, N.A.; Hilt, J.Z.; Khademhosseini, A.; Langer, R. Hydrogels in biology and medicine: From molecular principles to bionanotechnology. Adv. Mater. 2006, 18, 1345–1360. [Google Scholar] [CrossRef]
- Appel, E.A.; del Barrio, J.; Loh, X.J.; Scherman, O.A. Supramolecular polymeric hydrogels. Chem. Soc. Rev. 2012, 41, 6195–6214. [Google Scholar] [CrossRef]
- Aida, T.; Meijer, E.W.; Stupp, S.I. Functional supramolecular polymers. Science 2012, 335, 813–817. [Google Scholar] [CrossRef]
- Assaf, K.I.; Nau, W.M. The aqueous supramolecular chemistry of cucurbiturils, pillararenes and deep cavity cavitands. Chem. Soc. Rev. 2015, 44, 394–418. [Google Scholar] [CrossRef]
- Kim, K.; Selvapalam, N.; Oh, D.H. Cucurbiturils—A New Family of Host Molecules. J. Incl. Phenom. Macrocycl. Chem. 2004, 50, 31–36. [Google Scholar] [CrossRef]
- Mock, W.L.; Shih, N.Y. Structure and selectivity in the complexation of neutral substrates by cucurbituril. J. Org. Chem. 1986, 51, 4440–4446. [Google Scholar] [CrossRef]
- Lagona, L.; Mukhopadhyay, P.; Chakrabarti, S.; Isaacs, L. The cucurbit[n]uril family. Angew. Chem. Int. Ed. 2005, 44, 4844–4870. [Google Scholar] [CrossRef]
- Barrow, S.J.; Kasera, S.; Rowland, M.J.; del Barrio, J.; Scherman, O.A. Cucurbituril-based molecular recognition. Chem. Rev. 2015, 115, 12320–12406. [Google Scholar] [CrossRef]
- Zhou, W.-L.; Lin, W.; Chen, Y.; Dai, X.-Y.; Liu, Z.; Liu, Y. Multivalent supramolecular assembly with ultralong organic room temperature phosphorescence, high transfer efficiency and ultrahigh antenna effect in water. Chem. Sci. 2022, 13, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Rodell, C.B.; Kaminski, A.L.; Burdick, J.A. Rational design of network properties in guest–host assembled and shear-thinning hyaluronic acid hydrogels. Biomacromolecules 2013, 14, 4125–4134. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer 2008, 49, 1993–2007. [Google Scholar] [CrossRef]
- Day, A.I.; Arnold, A.P.; Blanch, R.J. A Method for Synthesizing Partially Substituted Cucurbit[n]uril. Molecules 2003, 8, 74–84. [Google Scholar] [CrossRef]
- Cox, C.J.T.; Hale, J.; Molińska, P.; Lewis, J.E.M. Supramolecular and molecular capsules, cages and containers. Chem. Soc. Rev. 2024, 53, 10380–10408. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Šekutor, M.; Zavalij, P.Y.; Mlinarić-Majerski, K.; Ghiviriga, I.; Isaacs, L. Cucurbit [7]uril·guest inclusion complexes. Angew. Chem. Int. Ed. 2014, 53, 988–993. [Google Scholar] [CrossRef]
- Assaf, K.I.; Nau, W.M. Cucurbiturils: From synthesis to high-affinity binding and catalysis. Chem. Soc. Rev. 2015, 44, 394–418. [Google Scholar] [CrossRef]
- Kim, J.; Jung, I.S.; Kim, S.Y.; Lee, E.; Kang, J.K.; Sakamoto, S.; Yamaguchi, K.; Kim, K. New cucurbituril homologues: Syntheses, isolation, and X-ray crystallographic structures. J. Am. Chem. Soc. 2000, 122, 540–541. [Google Scholar] [CrossRef]
- Isaacs, L.; Witt, D.; Lagona, L.; Mukhopadhyay, P.; Chakrabarti, S. Click chemistry and cucurbiturils. In Functional Supramolecular Architectures; Schmitt, J.-L., Strable, E., Eds.; Wiley-VCH: Weinheim, Germany, 2003; pp. 233–259. [Google Scholar]
- Lee, J.W.; Samal, S.; Selvapalam, N.; Kim, H.-J.; Kim, K. Cucurbituril homologues and derivatives: Synthesis, characterization, and applications. Acc. Chem. Res. 2003, 36, 621–630. [Google Scholar] [CrossRef] [PubMed]
- Lecorché, P.; Jacquot, A.; Bardelang, D.; Tóth, G.; Parella, T.; Ricoux, R.; Roux, G.; Milet, A.; Herson, P.; Fensterbank, L.; et al. Challenges and opportunities for functionalized cucurbiturils for biomedical applications. JACS Au 2023, 3, 1923–1946. [Google Scholar] [CrossRef]
- Kim, K.; Selvapalam, N.; Ko, Y. H.; Park, K. M.; Kim, D.; Kim, J. Functionalized cucurbiturils and their applications. Chem. Soc. Rev. 2007, 36, 267–279. [Google Scholar] [CrossRef]
- Kégl, T.R.; Kégl, T. Cavitands: Structure, Recognition, and Function in Supramolecular Chemistry. ChemTexts 2025, 11, 16. [Google Scholar] [CrossRef]
- Scherman, O.A.; Rowland, M.J.; Martinez-Martinez, A.J.; Walker, S.A.; Tsai, H.C. Supramolecular polymers built by cucurbituril-mediated host–guest interactions. In Supramolecular Polymer Chemistry; Harada, A., Takashima, Y., Eds.; Wiley-VCH: Weinheim, Germany, 2012; pp. 145–176. [Google Scholar]
- Appel, E.A.; Biedermann, F.; Rauwald, U.; Jones, S.T.; Zayed, J.M.; Scherman, O.A. Supramolecular cross-linked networks via host–guest complexation with cucurbit [8]uril. J. Am. Chem. Soc. 2010, 132, 14251–14260. [Google Scholar] [CrossRef]
- Harada, A.; Takashima, Y.; Nakahata, M. Supramolecular polymeric materials via cyclodextrin-guest interactions. Acc. Chem. Res. 2014, 47, 2128–2140. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Wang, J.; Du, X.; Cao, Y.; Ping, K.; Liu, D. Cucurbit [8]uril-based supramolecular theranostics. J. Nanobiotechnol. 2024, 22, 235. [Google Scholar] [CrossRef]
- Cordier, P.; Tournilhac, F.; Soulié-Ziakovic, C.; Leibler, L. Self-healing and thermoreversible rubber from supramolecular assembly. Nature 2008, 451, 977–980. [Google Scholar] [CrossRef] [PubMed]
- Yin, H.; Liu, F.; Abdiryim, T.; Liu, X. Self-Healing Hydrogels: From Synthesis to Multiple Applications. ACS Mater. Lett. 2023, 5, 1787–1830. [Google Scholar] [CrossRef]
- Drozdov, A.D.; Christiansen, J.D. Structure–Property Relations in Linear Viscoelasticity of Supramolecular Hydrogels. RSC Adv. 2021, 11, 16860–16880. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Hao, J.; Lu, Y.; Hu, W.; Jiang, H.; Zhou, B.; Yang, H.; Kang, W.; Sarsenbekuly, B. Tunable macroscopic self-healing of supramolecular gel through host–guest inclusion. Phys. Fluids 2024, 36, 113126. [Google Scholar] [CrossRef]
- Appel, E.A.; Loh, X.J.; Jones, S.T.; Biedermann, F.; Dreiss, C.A.; Scherman, O.A. Ultrahigh-Water-Content Supramolecular Hydrogels Exhibiting Multistimuli Responsiveness. J. Am. Chem. Soc. 2012, 134, 11767–11773. [Google Scholar] [CrossRef]
- Ooi, H.W.; Kocken, J.M.M.; Morgan, F.L.C.; Malheiro, A.; Zoetebier, B.; Karperien, M.; Wieringa, P.A.; Dijkstra, P.J.; Moroni, L.; Baker, M.B. Multivalency Enables Dynamic Supramolecular Host–Guest Hydrogel Formation. Biomacromolecules 2020, 21, 2208–2217. [Google Scholar] [CrossRef]
- Appel, E.A.; Biedermann, F.; Rauwald, U.; Jones, S.T.; Zayed, J.M.; Scherman, O.A. Light-Induced Transformation from Covalent to Supramolecular Polymer Networks. ACS Macro Lett. 2025, 14, 169–175. [Google Scholar] [CrossRef]
- Cardellini, A.; Caruso, C.; Rijns, L.; Dankers, P. Y. W.; Pavan, G. M.; Perego, C. Monomer exchange dynamics in ureido–pyrimidinone supramolecular polymers via molecular simulations. J. Mater. Chem. B 2025, 13, 14326–14337. [Google Scholar] [CrossRef]
- Baghali, M.; Ziyadi, H.; Di Martino, A. Engineering Smart Hydrogels for Intelligent Drug Delivery. Polym. Bull. 2025, 82, 2287–2328. [Google Scholar] [CrossRef]
- Dong, R.; Pang, Y.; Su, Y.; Zhu, X. Supramolecular Hydrogels: Synthesis, Properties and Their Biomedical Applications. Biomater. Sci. 2015, 3, 937–954. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhang, G.; Liu, S. Enzyme-responsive polymeric assemblies, nanoparticles and hydrogels. Chem. Soc. Rev. 2012, 41, 5933–5949. [Google Scholar] [CrossRef]
- Winter, H.H.; Chambon, F. Analysis of linear viscoelasticity of a crosslinking polymer at the gel point. J. Rheol. 1986, 30, 367–382. [Google Scholar] [CrossRef]
- Rubinstein, M.; Colby, R.H. Polymer Physics; Oxford University Press: New York, NY, USA, 2003. [Google Scholar]
- Kadic, M.; Bückmann, T.; Schittny, R.; Gumbsch, P.; Wegener, M. Pentamode Metamaterials with Independently Tailored Bulk Modulus and Mass Density. Phys. Rev. Appl. 2014, 2, 054007. [Google Scholar] [CrossRef]
- Ducrot, E.; Chen, Y.; Bulters, M.J.H.; Sijbesma, R.P.; Creton, C. Toughening Elastomers with Sacrificial Bonds and Watching Them Break. Science 2014, 344, 186–189. [Google Scholar] [CrossRef]
| CB[n] | Host–Guest Binding Mode | Typical Guest Units | Polymer Matrix | Mechanical properties (G′ / E) | Key Composite Features | Ref. |
|---|---|---|---|---|---|---|
| CB [6] | 1:1 inclusion complex | Alkylammonium, diammonium | PEG, polyacrylamide | G′ ≈ 10–10³ Pa | Stable but relatively slow relaxation; good dimensional stability | [8,23] |
| CB [7] | 1:1 inclusion complex | Adamantyl, ammonium, aromatic cations | PEG, gelatin, hyaluronic acid | G′ ≈ 10²–10⁴ Pa | High binding affinity, enhanced toughness, moderate self-healing | [9,14,30] |
| CB [8] | Ternary complex (1:2) | Donor–acceptor aromatic pairs | PEG, chitosan, hybrid matrices | G′ ≈ 10³–10⁵ Pa | Dense reversible crosslinking; excellent self-healing and energy dissipation | [24,25,28] |
| CB [8] + fillers | Host–guest + physical filler interaction | Aromatic-functional fillers | Nanocomposites (NFC, CNTs, graphene) | E ≈ 10–500 kPa | Improved load transfer, crack resistance, multifunctionality | [28,36] |
| Application Area | Composite Components | CB[n] Role | Stimulus / Function | Composite Advantage | Ref. |
|---|---|---|---|---|---|
| Biomedical scaffolds | Biopolymer + CB[n] | Dynamic crosslinker | pH, redox | Self-healing, injectability, tissue-matching mechanics | [35] |
| Injectable drug carriers | PEG/CB [8] networks | Ternary junctions | Shear, pH | Shear-thinning, rapid recovery, controlled release | [28,30] |
| Soft robotics | Polymer + CB [8] + redox guest | Switchable crosslinks | Electric potential | Reversible stiffness modulation | [31] |
| Soft electronics | Polymer + CNT/graphene + CB[n] | Interfacial binder | Mechanical damage | Self-healing conductivity | [36] |
| Environmental remediation | Gel + CB[n] | Selective adsorption sites | pH, competing guests | Regenerable composite sorbents | [4,8] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




