1. The Growing Spine–Anatomy and Biomechanics
The spine is a complex structure composed of multiple interdependent elements that provide remarkable stability and strength while enabling mobility. Its growth and development extend over many years until maturation and ossification are complete. Proper morphological development of the spinal column determines its strength and its ability to bear mechanical loads effectively. Any congenital or acquired defect increases the risk of degenerative changes and chronic pain syndromes. Due to their unique anatomical and biomechanical characteristics, the spines of children and adolescents are particularly vulnerable to maladaptive responses when subjected to prolonged or asymmetrical mechanical stress. This condition underscores the growing impact of modern lifestyle factors on spinal health, particularly in populations whose spines are still developing.
The development of the spine begins in the third week of gestation. It undergoes a long process of growth, formation, maturation, chondrification, and ossification, all of which are precisely controlled by genes, forces, movement, hormones, nutrients, and time. The immature spine is a mosaic of primary growth centers composed of cartilaginous tissue responsible for longitudinal spinal growth (endochondral ossification) [
1,
2,
3]. Ossification of the cartilaginous part of the vertebral endplate (CEP) begins around the age of five, leading to the formation of the ring apophysis (secondary ossification growth centres), which are essential for the structural maturation of the vertebral bodies. The ring apophysis should be fully formed by the age of 12, but fusion does not occur until skeletal maturity [
4]. Spine maturation is a complex and highly individual process. The study by Costa et al. describes ring apophysis fusion patterns that vary by age, spinal level, and sex. According to this study, ossification of the ring apophysis occurs between the ages of 9 and 15 in males and 7 and 15 in females. Fusion occurs between the ages of 14 and 19 in both sexes [
5]. Radiographic studies of the cervical spine revealed that the earliest apophysis was observed in a patient aged 3 years, and that all patients aged 14 years and older exhibited the apophysis. The inferior apophysis of a cervical vertebral body appeared before the superior apophysis; however, the superior apophysis fused first in most cases (86%). The superior apophyses have fused by the age of 18, although unfused inferior apophyses were still evident in the 20-year-old group [
6]. All maturation processes aim to strengthen the spine's resilience to the mechanical effects of growing body mass and control shifts in the body's center of mass [
4,
7]. Due to its increased elasticity and enhanced resistance to compressive forces, the paediatric spine is less susceptible to fractures following low-energy trauma [
7]. However, severe accidents often result in cervical spine injuries, which can lead to lifelong disability [
8]. The biomechanics of the growing spine differ from those of the adult spine, and certain anatomical features make the former more susceptible to stress and load (
Table 1).
2. Static Versus Dynamic Spinal Load
Forces acting on the musculoskeletal system are crucial in shaping the growing spine. These forces, which include compression, tension, shearing, flexion-extension, and hydrostatic pressure, participate in biological regulatory pathways through the principles of mechanotransduction [
9,
10]. The intervertebral disc (IVD) plays a critical role in load transmission and flexibility, thereby contributing to the overall stability of the entire vertebral column. This stability is also influenced by the morphological properties of the vertebral bodies, including their shape, structural composition, and resistance to mechanical loading [
11,
12,
13]. During maturation, the IVD gradually decreases in hydration and vascularization, and its cellular matrix composition changes [
14]. A defining feature of the young spine is its high disc hydration, which increases during the first two decades of life, due to enhanced glycosaminoglycan synthesis in the nucleus pulposus (NP) [
15,
16]. Hydration and IVD osmolality are dynamic and influenced by applied mechanical load. The NP of the intervertebral disc is a highly permeable, gelatinous tissue that adapts to vertical stresses by regulating its water content. NP cells respond to osmotic fluctuations by regulating water flow through aquaporins, which are water channel proteins [
17]. Previous studies have demonstrated the key role of aquaporins 1 (AQP1) and 3 (AQP3) in delivering nutrients and transporting metabolic substances in NP tissue. The expression of aquaporins in IVD cells is regulated by the osmotic environment; for example, a decrease in osmotic pressure reduces AQP1 expression. Notably, AQP1 also exhibits significant oxygen permeability. Under hypoxic conditions, upregulation of AQP1 expression facilitates oxygen transport across membranes, potentially alleviating hypoxia-induced damage in the disc. Results show that as the degree of degeneration of human NP tissue increases, its water content decreases, leading to a progressive reduction in AQP levels [
17].
CEPs play a crucial role in the development of (IVDs), as they maintain metabolic homeostasis and provide nutrition to the NP [
11,
18]. Disc nutrition depends on fluid flow within the NP, driven by a pressure gradient. Even under constant mechanical loading, significant pressure gradients exist within the NP, particularly during the initial loading phase and in more hydrated IVDs. Pressure reaches a maximum at the center of the nucleus, decreasing axially toward the CEP and radially toward the annulus fibrosus (AF) [
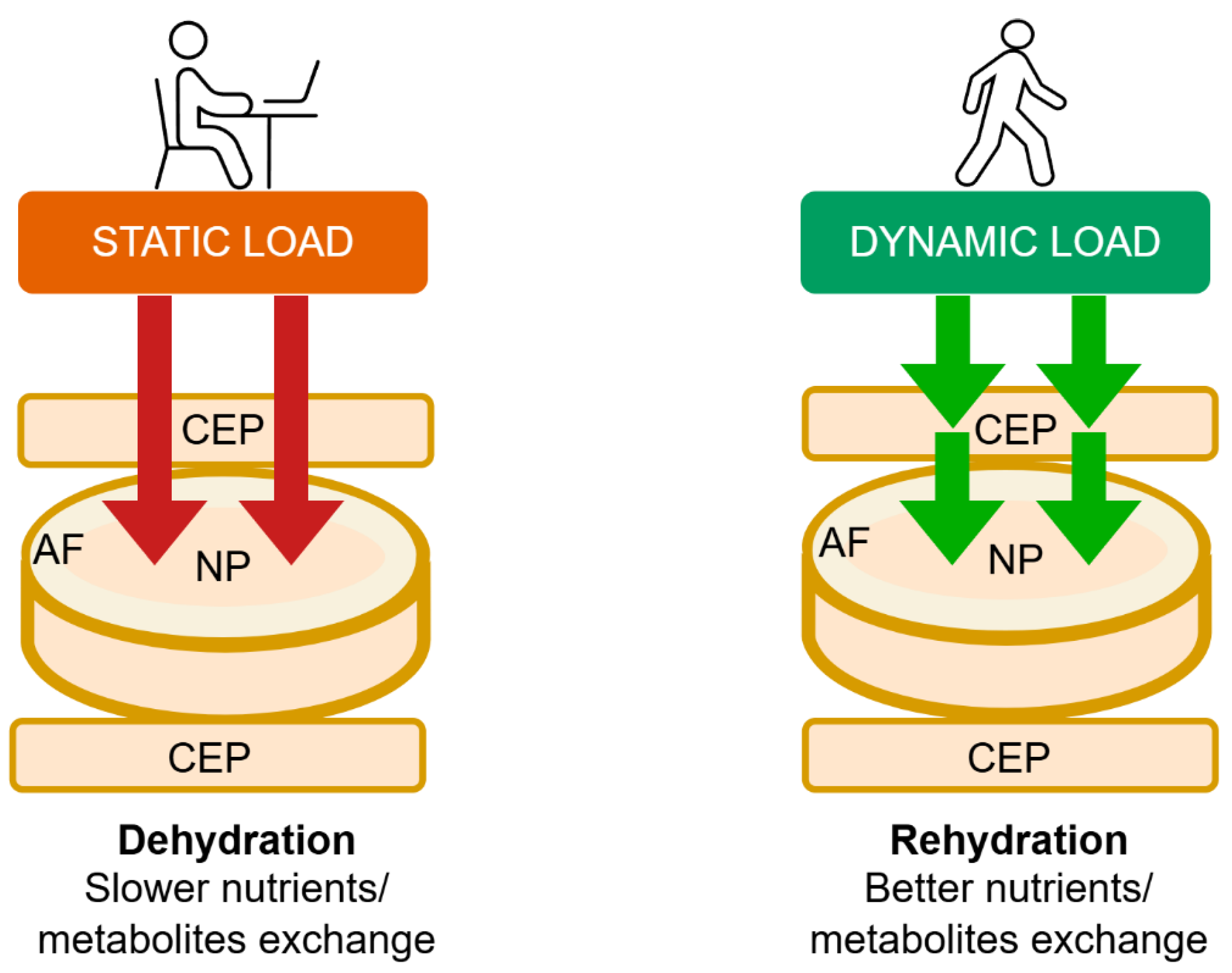
19]. As static loading and disc dehydration increase over time, circulation within the disc slows down, as does the exchange of metabolites and nutrients through the CEP (
Figure 1). Studies have shown that constant pressure gradually reduces the number of vessel buds in the CEP and significantly decreases vascular endothelial growth factor (VEGFA) concentrations [
20]. Compared to constant static loading, dynamic mechanical forces enhance the diffusion and convection of essential nutrients within the IVD, such as oxygen and glucose, while also facilitating the removal of metabolic waste products. Cyclic mechanical loading, in particular, favours the maintenance of a larger viable cell population and promotes extracellular matrix synthesis, thereby contributing to improve disc hydration and overall disc health [
21,
22]. The role of exercise in preventing degenerative disc disease remains a topic of debate. High physical strain and competitive sports can result in spinal pain and adverse changes [
23]. Studies indicate that moderate effort and an upright, bipedal posture benefit IVD health [
24,
25,
26].
3. The Importance of Sagittal Profile Development
Anatomically, the spine exhibits four natural curvatures in the sagittal plane: cervical lordosis, thoracic kyphosis, lumbar lordosis, and sacral kyphosis. These natural curvatures are an evolutionary adaptation that enables the body to maintain an upright posture with minimal energy expenditure and maximum resistance to gravity [
27,
28]. Proper formation of these curvatures prevents excessive stress on intervertebral discs, ligaments, and muscles by efficiently distributing forces and transferring mechanical loads to each moving segment [
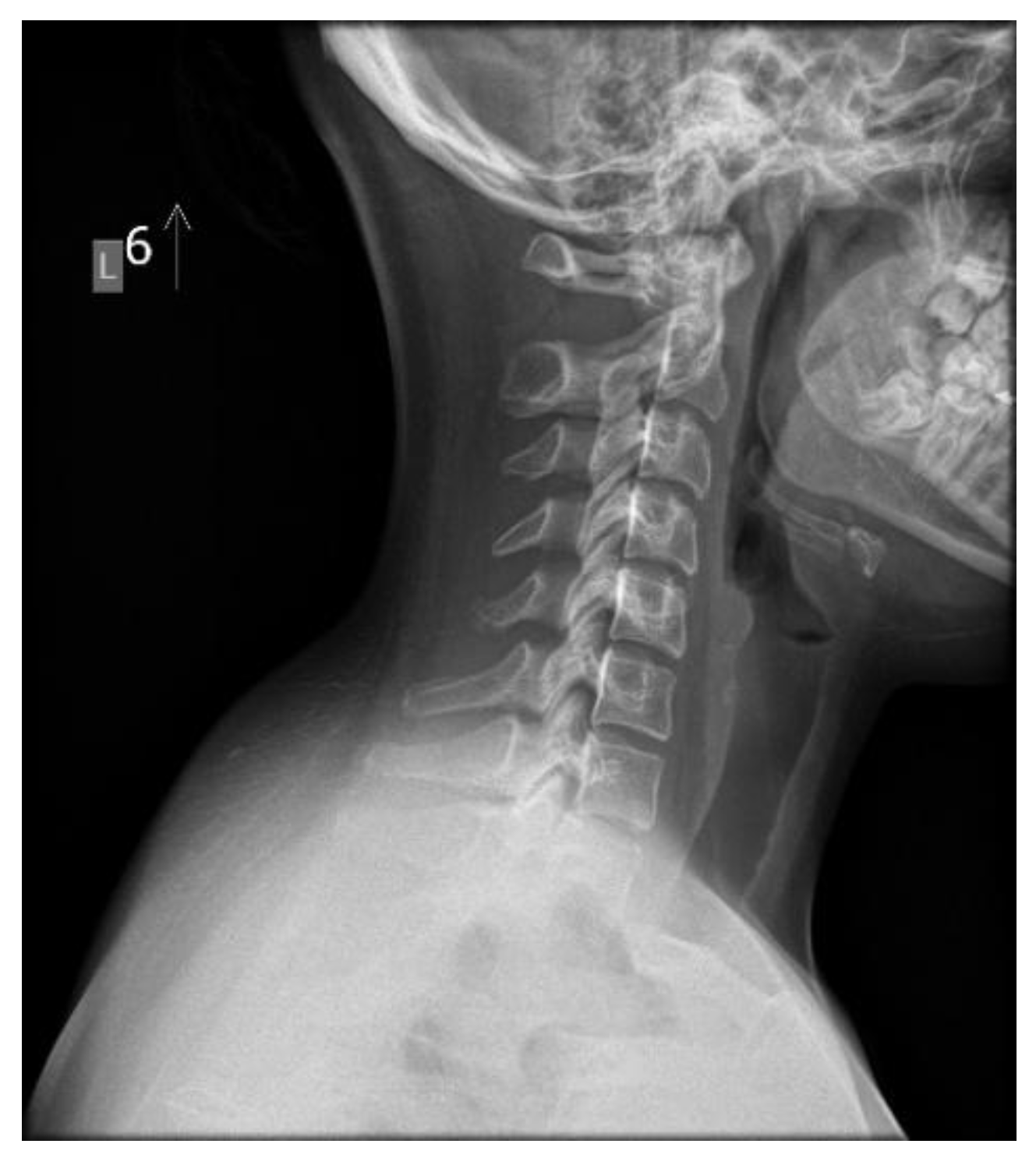
29]. The biomechanical balance of the counter curves provides optimal stabilization of the entire spine. Disturbances to this mechanical system can contribute to a gradual and often prolonged process of tissue degeneration. Clinical symptoms appear when the compensation limit is reached and the critical point of permanent tissue damage is exceeded (
Figure 2) [
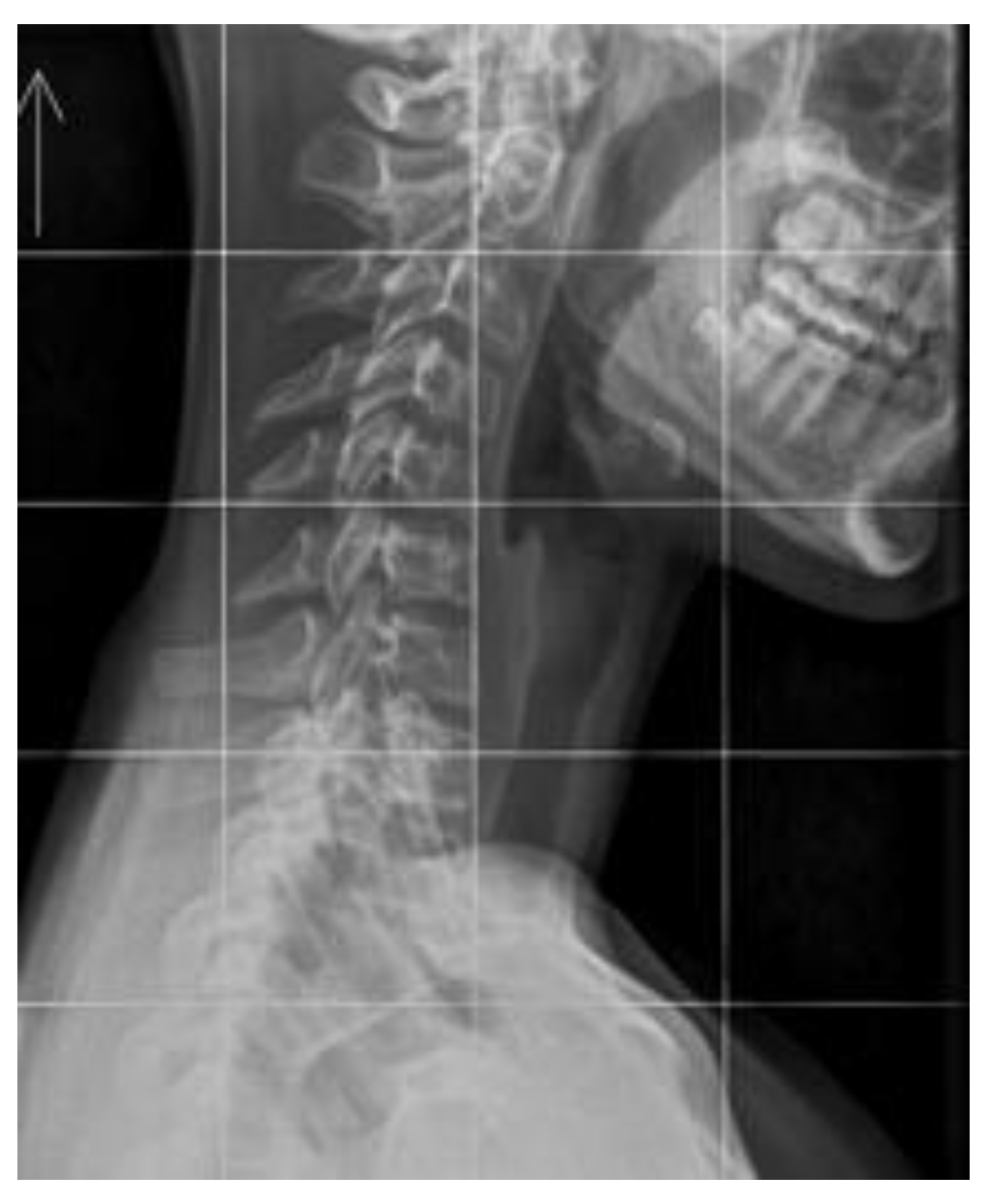
30]. The patients with abnormal physiological spinal curvatures and the absence of clinical symptoms, such as pain, do not necessarily indicate a normal state, despite the surprisingly high prevalence of such cases (
Figure 3).
Lee et al. conducted a radiographic analysis of sagittal alignment in 181 asymptomatic adolescents. They found a high prevalence of cervical hypolordosis in 13 –17-year-olds (71%) and 8–12-year-olds (65.6%). The overall prevalence of cervical kyphosis in the studied population was approximately 40% [
31]. These findings provide valuable insight into the extent of cervical lordosis maldevelopment among young populations. There is a clear need for further research focused specifically on the paediatric population, particularly studies examining the influence of lifestyle factors, such as prolonged static postures with sustained cervical flexion, as we commonly observe in children and adolescents when using mobile phones, tablets, and other screen-based devices. These studies should also encompass a broader range of symptoms other than just cervical pain, as this is not typical in children and adolescents. The spinal sagittal profile matures gradually during growth and development. Studies establishing reference values for sagittal curvatures in the growing spine are limited, particularly across different age groups and sexes. Research by Takács et al. suggests that relating normal physiological curvatures to height rather than age provides a more accurate assessment [
32]. Multicenter studies involving large populations have demonstrated that changes in thoracic kyphosis and lumbar lordosis occur during growth spurts in children and are interdependent, forming a specific, cascading phenomenon [
33]. Unfortunately, many valuable studies do not examine the cervical sagittal profile, an integral part of overall spinal alignment [
33,
34]
4. Adolescents, Technology, and Nomophobia
The time we spend in front of digital devices is increasing year after year, and each generation is becoming more dependent on the digital world. According to the Common Sense Census, 84% of teenagers own smartphones, and 70% use social media multiple times a day [
35]. During the COVID-19 pandemic, the total daily screen time increased from 4 hours and 44 minutes to 5 hours and 33 minutes for tweens, and from 7 hours and 22 minutes to 8 hours and 39 minutes for teens [
35]. This increase has been significantly faster over the past two years than it was in the previous four [
35]. A study of 384,591 15-year-olds from 55 countries found that children start using digital devices at a very early age. The results showed that 7.53% start before the age of three, 27.83% start between the ages of four and six, 35.56% start at the age of seven or nine, 21.07% start at the age of ten or twelve, and only 6.85% after the age of thirteen [
36]. These results indicate that 70.92% of children start using digital devices before the age of ten. A study involving 237 children aged 3 to 5 years who averaged nearly four hours of screen time per day observed a positive association between parental and child screen use, indicating that familial habits and behaviors significantly influence screen exposure in young children [
37]. Hantal et al. conducted a study linking discopathy in adolescents to screen time. The study involved 94 patients with disc pathology (bulging, protrusion, extrusion) aged 10-16 who spent an average of 5.16 hours per day on digital devices. This was also associated with low physical activity levels [
38]. This is the first and most significant study to highlight the impact of adolescents' lifestyles on early spine degeneration. Smartphone addiction is a significant challenge in today`s society. The term nomophobia (no mobile phone phobia) was introduced into scientific literature with the rise of excessive phone use. This refers to the fear of losing access to one`s phone, its content, and connectivity with a particular community [
39]. A meta-analysis of 52 studies involving almost 47,400 participants from 20 countries found that the overall prevalence of nomophobia across all symptom and severity categories was 93.92%. Mild nomophobia symptoms were observed in around 25.80% of individuals, moderate symptoms in approximately 52.40%, and severe symptoms in around 20.35% [
40].
Excessive or problematic screen media use can be examined in relation to various health aspects, but psychological hazards are the main focus [
41]. We believe that insufficient research has been conducted into the impact of early and long-term use of digital devices by children and adolescents on the development of the skeletal system, including posture and spine formation. This can have negative long-term health consequences [
42,
43]. Spending extended periods sitting in front of digital media also contributes to a sedentary lifestyle, which has implications for musculoskeletal development, including low physical activity [
44,
45,
46,
47]. Trends in insufficient physical activity among adolescents were examined between 2001 and 2016. Data from 298 surveys in 146 countries, covering 1.6 million students, showed that over 80% of adolescents did not meet the WHO's physical activity recommendations. This study highlights the importance of interventions to promote physical activity, particularly among girls [
48]. A cohort study of children aged 11 to 12 found that the more time spent in front of a screen, the greater the risk of experiencing moderate or severe spinal pain. Interestingly, this link was independent of physical activity levels [
49], which may be explained by the effects of constant static forces on the spine and muscles. Spending extended hours using digital media replaces an active lifestyle, raising the risk of pain and spinal maldevelopment issues even further [
51].
5. Forward Head Posture Syndrome and Cervicovagopathy
This syndrome is characterized by stress on the cervical spine tissues caused by prolonged flexed positions resulting from the use of mobile devices. Several terms describing the same anatomical and pathological issue of forward head protraction appear in the literature, including text neck syndrome, tech neck syndrome, turtle neck syndrome, and forward head posture syndrome [
50,
51,
52,
53,
54]. Earlier generations of mobile phones were primarily used for communication, and users frequently bent over to text, which led to the development of text neck syndrome. Today, smartphones offer a wider range of functions, and young people often use them for hours at a time to watch films, play games, and chat with friends. In broader terms, concepts less affected by technological change tend to be more universal. Forward head posture (FHP) refers to the position of the head and neck, regardless of the activity being performed.
The cervical spine supports the weight of the head while enabling multi-directional movement. This is achieved through the deep muscles of the cervical spine, which aid in perception and stabilize posture, along with the vestibular system and visual input. The upper cervical spine, particularly C1–C3, and the occipitoparietal junction are particularly important. It is estimated that the suboccipital muscles contain a high number of spindles (proprioceptors) [
55]. The activity of these mechanoreceptors can be affected by injury, inflammation, or degenerative changes [
56]. Changes in loads and impulses in FHP result not only from overloads during work in a head-down position but also from persistent dysfunction. The cervical lordosis loss is a structural disorder that can be seen on neck X-rays in a relaxed, habitual posture (
Figure 2 and
Figure 3). Muscles have a high capacity to adapt to altered working conditions in non-physiological positions by shortening, atrophy, and degenerative changes. This can impair muscle strength and endurance, creating a vicious cycle whereby incorrect posture leads to changes in muscle morphology and innervation. These changes then cause weakened strength and neurocontrol, which further deteriorate stability and posture. These morphological changes in muscles also cause abnormal, continuous sensory impulses from the paraspinal and suboccipital muscles (deep stabilizers) to the central systems responsible for posture, balance, and coordination, resulting in information noise [
56,
57].
Studies have demonstrated a direct correlation between these morphological alterations and symptoms, such as chronic headaches, neck pain, somatic dysfunction, and balance disorders [
58,
59]. The clinical signs of FHPS reported in the literature are diverse and often nonspecific. These include pain and limited mobility of the cervical spine, increased neck muscle tension, headaches, dizziness, balance problems, visual disturbances, and fatigue. Less common clinical issues associated with FHPS include chest pain, brain fog, postural orthostatic tachycardia syndrome, sleep disturbances, gastrointestinal problems such as gastroparesis, and tinnitus [
57,
58,
59,
60,
61]. The variety and nonspecific nature of symptoms associated with FHPS can be attributed to cervicovagopathy, a term introduced by Hauser [
60,
61]. The vagus nerve (VN) is the longest cranial nerve and carries sensory, motor, and parasympathetic fibers to organs in the chest and abdomen. Anatomically, the VN is closely associated with the C1 and C2 vertebrae. The superior vagal ganglion is located in the lateral part of the jugular foramen, and the inferior ganglion is situated below and in front of the transverse processes of the C1 and C2 vertebrae, where compression and/or stretching of the nerve structures most often occur. Abnormal spinal anatomy or slight instability of the upper cervical spine can cause conflict between the vertebrae and the vagus nerve and/or its ganglia (cervicovagopathy). Due to the complex functions of the X nerve (autonomic, sensory, and motor), its dysfunction can manifest as diverse symptoms affecting many organs and systems [
60,
61].
6. Highly Processed Food Consumption, Obesity, and Myosteatosis
Proper nutrition and physical activity are essential for a child`s growth and development. However, spending many hours in the digital world is often associated with increased snacking and the replacement of full meals with ultra-processed foods (UPF). This trend is driven by the widespread availability and easy preparation of these products [
62,
63]. Unfortunately, analyses of evolving dietary habits show that UPF consumption is increasing among young people [
64,
65]. There is a strong connection between prolonged screen time and UPF consumption. A cross-sectional study of four-year-old children (n = 362) found that screen time exceeding 60 minutes was associated with lower developmental scores, greater screen-based eating, and higher UPF consumption [
66]. Studies conducted among adolescents suggest that sedentary behaviours related to screen time may lead to impulsive eating and a preference for convenience foods, which are often UPF [
67]. These findings could also pose risks to the proper development of the adolescent skeletal system tissues. The impact of UPF on children's health is now being discussed not only among specialists but also within highly developed societies, where the adverse health effects of high consumption are evident.
The impact of UPF on skeletal and muscular development remains unclear. An experimental study in rats showed that UPF can disrupt endochondral ossification in the skeleton. Young rats fed UPF exhibited skeletal pathologies, including growth retardation, decreased bone mineral density, structural deterioration, and poor skeletal tissue quality [
68]. Studies on mice have shown that UPF negatively affects bone quality, increases marrow fat, alters growth patterns, and modifies the gut microbiome [
69]. UPFs also harm muscle mass. Studies in adults have revealed a significant correlation between increased UPF intake and a higher risk of reduced muscle mass and grip strength [
70,
71]. An inadequate diet is a well-known risk for developing metabolic syndrome (MS), which is an increasing concern in the pediatric population [
72]. The effect of metabolic disorders on osteoarthritis and intervertebral disc diseases (IVDD) development has been widely studied, primarily in adults. A study involving 928 participants showed that individuals with one or more components of MS had a higher risk of IVDD compared to those without MS components, suggesting that the accumulation of metabolic abnormalities increases the likelihood of IVDD [
73]. Hyperglycemia and dyslipidemia are also recognised as risk factors for IVDD severity [
74,
75].
Obesity in children and adolescents is an increasingly prevalent issue with complex effects on health and development. When considering how lifestyle influences the musculoskeletal system, obesity should not be overlooked. In children, obesity affects musculoskeletal development through changes in hormonal environments, including shifts in growth and pubertal hormones, as well as alterations in adipokine release. Furthermore, different fat compartments, particularly visceral fat, can negatively impact bone quality and strength [
76,
77]. Obesity has also been linked to myosteatosis, characterized by increased fat infiltration into muscles and a negative impact on strength and metabolic function [
78,
79].
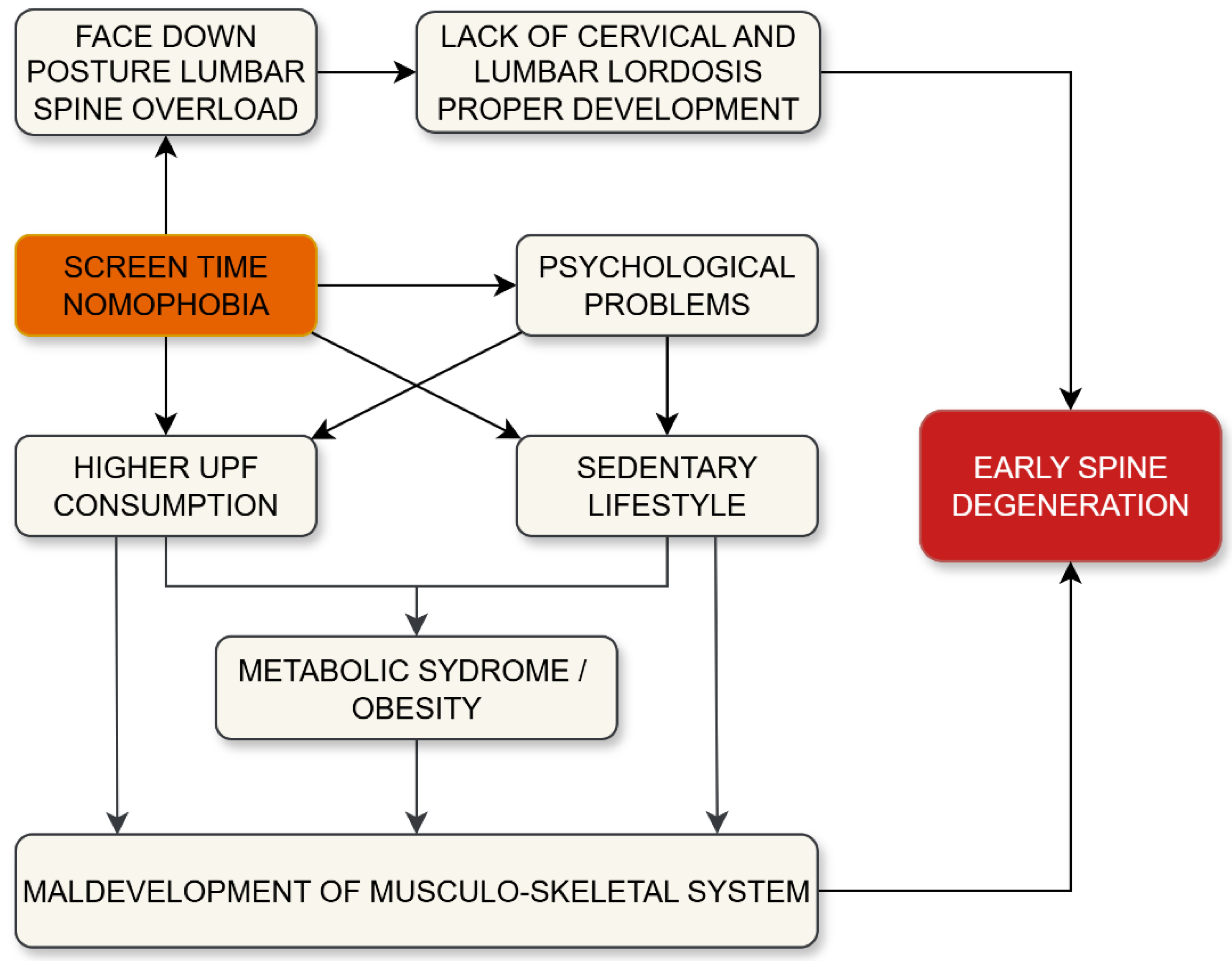
Studies examining the impact of metabolic disorders on the spinal disc and muscles provide valuable insights into the causes of spinal degeneration in developed countries. Biomechanical theories remain prevalent among researchers and clinicians. When considering child and adolescent development, it is important to address all risk factors for early degenerative spinal changes caused by modern lifestyles (
Figure 4).
Data Availability Statement:
Not applicable.
Funding
The work was funded by the Minister of Science under “the Regional Initiative for Excellence Program.”.
Author Contributions:
K.Z.S.: P.T.W., A.So., and M.K. wrote the main manuscript text; P.T.W. prepared the figures. A.Sk. provided critical edits, outline review, and contextual relevance. All authors reviewed the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Dimeglio, A.; Canavese, F. The growing spine: how spinal deformities influence normal spine and thoracic cage growth. Eur. Spine J. 2012, 21, 64–70. [Google Scholar] [CrossRef]
- Labrom, R. D. Growth and maturation of the spine from birth to adolescence. J. Bone Joint Surg. Am. 2007, 89, 3–7. [Google Scholar] [CrossRef]
- Sanders, J. O. Normal growth of the spine and skeletal maturation. Semin. Spine Surg. 2015, 27, 16–20. [Google Scholar] [CrossRef]
- Srinivasan, V.; Jea, A. Pediatric thoracolumbar spine trauma. Neurosurg. Clin. N. Am. 2017, 28, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Costa, L.; de Reuver, S.; Kan, L. Ossification and fusion of the vertebral ring apophysis as an important part of spinal maturation. J. Clin. Med. 2021, 10, 3217. [Google Scholar] [CrossRef]
- Woo, T. D.; Tony, G.; Charran, A. Radiographic morphology of normal ring apophyses in the immature cervical spine. Skeletal Radiol. 2018, 47, 1221–1228. [Google Scholar] [CrossRef]
- Daniels, A. H.; Sobel, A. D.; Eberson, C. P. Pediatric thoracolumbar spine trauma. J. Am. Acad. Orthop. Surg. 2013, 21, 707–716. [Google Scholar] [CrossRef]
- Patel, J. C.; Tepas, J. J.; Mollitt, D. L.; Pieper, P. Pediatric cervical spine injuries: defining the disease. J. Pediatr. Surg. 2001, 36, 373–376. [Google Scholar] [CrossRef]
- Tsai, T. T.; Cheng, C. M.; Chen, C. F.; Lai, P. L. Mechanotransduction in intervertebral discs. J. Cell. Mol. Med. 2014, 18, 2351–2360. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Kamm, R.; Lee, R. Cell mechanics and mechanotransduction: pathways, probes, and physiology. Am. J. Physiol. Cell Physiol. 2004, 287, C1–C11. [Google Scholar] [CrossRef]
- Gersing, A. S.; Woertler, K.; Jungmann, P. M.; Bollwein, C.; Schwaiger, B. J. Vertebrae, vertebral end plates, and disks: concepts and specific pathologies. Semin. Musculoskelet. Radiol. 2019, 23, 489–496. [Google Scholar] [CrossRef]
- Pickering, E.; Pivonka, P.; Little, J. P. Toward patient specific models of pediatric IVDs: a parametric study of IVD mechanical properties. Front. Bioeng. Biotechnol. 2021, 9, 632408. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Zhu, Q.; Gu, W. An anisotropic multiphysics model for intervertebral disk. J. Appl. Mech. 2016, 83, 021011. [Google Scholar] [CrossRef] [PubMed]
- Moens, A. J. B. W. D.; Magré, J.; Kruyt, M. C.; Castelein, R. M.; de Reuver, S. Morphologic changes of the intervertebral disk during growth. Spine 2024, 49, 956–963. [Google Scholar] [CrossRef]
- Antoniou, J.; Steffen, T.; Nelson, F. The human lumbar intervertebral disc: evidence for changes in the biosynthesis and denaturation of the extracellular matrix with growth, maturation, ageing, and degeneration. J. Clin. Invest. 1996, 98, 996–1003. [Google Scholar] [CrossRef]
- Bolzinger, M.; Estivalèzes, E.; Gallini, A.; Polirsztok, E.; Abelin-Genevois, K. MRI evaluation of the hydration status of non-pathological lumbar intervertebral discs in a pediatric population. Orthop. Traumatol. Surg. Res. 2020, 106, 1281–1285. [Google Scholar] [CrossRef]
- Snuggs, J. W.; Day, R. E.; Bach, F. C. Aquaporin expression in the human and canine intervertebral disc during maturation and degeneration. JOR Spine 2019, 2, e1049. [Google Scholar] [CrossRef]
- Moore, R. J. The vertebral end-plate: what do we know? Eur. Spine J. 2000, 9, 92–96. [Google Scholar] [CrossRef]
- Ranganathan, A.; Freeman, B.; Scammell, B.; McNally, D. S.; Cox, E.; Gowland, P. What influence does sustained mechanical load have on diffusion in the human intervertebral disc? An in vivo study using serial postcontrast magnetic resonance imaging. Spine 2009, 34, 2324–2337. [Google Scholar] [CrossRef]
- Zhan, J.-W.; Wang, S.-Q.; Feng, M.-S.; Wei, X.; Yu, J.; Yin, X.-L. Constant compression decreases vascular bud and VEGFA expression in a rabbit vertebral endplate ex vivo culture model. PLoS One 2020, 15, e0234747. [Google Scholar] [CrossRef]
- Schmidt, H.; Galbusera, F.; Rohlmann, A.; Shirazi-Adl, A. What have we learned from finite element model studies of lumbar intervertebral discs in the past four decades? J. Biomech. 2013, 46, 2342–2355. [Google Scholar] [CrossRef]
- Zhu, Q.; Jackson, A. R.; Gu, W. Y. Cell viability in intervertebral disc under various nutritional and dynamic loading conditions: 3d finite element analysis. J. Biomech. 2012, 45, 2769–2777. [Google Scholar] [CrossRef]
- Ogurkowska, M. B.; Kawalek, K. Pathological changes in the lumbar intervertebral discs among professional field hockey players. J. Sports Med. Phys. Fitness 2016, 56, 85–91. [Google Scholar]
- Chodkowski, J.; Korotko, U. The association between physical activity and the development of degenerative spine disease: a review. Qual. Sport 2025, 37, 56898. [Google Scholar] [CrossRef]
- Samanna, C. L.; Owen, P.; Mitchell, U.; Ehrenbrusthoff, K.; Saueressig, T.; Moreira, E.; Arora, N. K.; Mundell, N. L.; Tait, J. L.; Donath, L.; Karner, V.; Belavy, D. L. Upright bipedal exercise training improves intervertebral disc health: a systematic review and meta-analysis. J. Clin. Exerc. Physiol. 2024, 13(2), 391–391. [Google Scholar] [CrossRef]
- Belavy, D. L.; Albracht, K.; Bruggemann, G. P. Can exercise positively influence the intervertebral disc? Sports Med. 2016, 46, 473–485. [Google Scholar] [CrossRef]
- Kariman, A.-G. Sagittal balance of the spine. Orthop. Traumatol. Surg. Res. 2021, 107, 102769. [Google Scholar] [CrossRef]
- Le Huec, J. C.; Thompson, W.; Mohsinaly, Y. Sagittal balance of the spine. Eur. Spine J. 2019, 28, 1889–1905. [Google Scholar] [CrossRef]
- Roussouly, P.; Pinheiro-Franco, J. L. Sagittal parameters of the spine: biomechanical approach. Eur. Spine J. 2011, 20, 578. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Lemans, J. V. C.; Zavatsky, J. M.; Agarwal, A.; Kruyt, M. C.; Matsumoto, K.; Serhan, H.; Agarwal, A. K.; Goel, V. K. Spinal balance/alignment - clinical relevance and biomechanics. J. Biomech. Eng. 2019, 141, 070805. [Google Scholar] [CrossRef]
- Lee, C. S.; Noh, H.; Lee, D. H.; Hwang, C. J.; Kim, H.; Cho, S. K. Analysis of sagittal spinal alignment in 181 asymptomatic children. J. Spinal Disord. Tech. 2012, 25, E259–E263. [Google Scholar] [CrossRef] [PubMed]
- Takacs, M.; Rudner, E.; Kovacs, A. The assessment of the spinal curvatures in the sagittal plane of children using an ultrasound-based motion analysing system. Ann. Biomed. Eng. 2015, 43, 348–362. [Google Scholar] [CrossRef]
- Pesenti, S.; Charles, Y. P.; Prost, S.; Solla, F.; Blondel, B.; Ilharreborde, B. Spinal sagittal alignment changes during childhood: results of a national cohort analysis of 1,059 healthy children. J. Bone Joint Surg. 2023, 105, 676–686. [Google Scholar] [CrossRef]
- Sainz de Baranda, P.; Andujar, P.; Collazo-Dieguez, M.; Pastor, A.; Santonja-Renedo, F.; Martinez-Romero, M. T.; Aparicio-Sarmiento, A.; Cejudo, A.; Rodriguez-Ferran, O.; Santonja-Medina, F. Sagittal standing spinal alignment and back pain in 8 to 12-year-old children from the Region of Murcia, Spain: The ISQUIOS Program. J. Back Musculoskelet. Rehabil. 2020, 33, 1003–1014. [Google Scholar] [CrossRef]
- Rideout, V.; Peebles, A.; Mann, S.; Robb, M. B. Common Sense Census: media use by tweens and teens, 2021; Common Sense: San Francisco, CA, 2022. [Google Scholar]
- Slater, S. O.; Arundell, L.; Grontved, A.; Salmon, J. Age of first digital device use and screen media use at age 15: a cross-sectional analysis of 384,591 participants from 55 countries. Public Health Pract. 2025, 9, 100596. [Google Scholar] [CrossRef]
- Frata, B.; Souza, J. M.; Montemezzo, D. Predictors for Screen Time Exposure among Children between 3 and 5 Years Old in Southern Brazil. J. Trop. Pediatr. 2021, 67, fmab092. [Google Scholar] [CrossRef]
- Hantal, A. O.; Iptec, M. The relationship between intervertebral disc pathologies and the use of digital devices and lack of physical activity in adolescents. J. Surg. Med. 2024, 8, 117–122. [Google Scholar] [CrossRef]
- Vagka, E.; Gnardellis, C.; Lagiou, A.; Notara, V. Prevalence and factors related to nomophobia: arising issues among young adults. Eur. J. Invest. Health Psychol. Educ. 2023, 13, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Jahrami, H.; Trabelsi, K.; Boukhris, O.; Hussain, J. H.; Alenezi, A. F.; Humood, A.; Saif, Z.; Pandi-Perumal, S. R.; Seeman, M. V. The prevalence of mild, moderate, and severe nomophobia symptoms: a systematic review, meta-analysis, and meta-regression. Behav. Sci. 2023, 13, 35. [Google Scholar] [CrossRef]
- Dong, N.; Zhou, Y.; Lei, L.; Lee, T. M. C.; Lam, C. L. M. The longitudinal impact of screen media activities on brain function, architecture and mental health in early adolescence. Int. J. Clin. Health Psychol. 2025, 25, 100589. [Google Scholar] [CrossRef] [PubMed]
- Faeze, S.; Sakineh, V. Comparing the effect of the posture of using smartphones on head and neck angles among college students. Ergonomics 2022, 65, 1631–1638. [Google Scholar] [CrossRef]
- In, T. S.; Jung, J. H.; Jung, K. S.; Cho, H. Y. Spinal and pelvic alignment of sitting posture associated with smartphone use in adolescents with low back pain. Int. J. Environ. Res. Public Health 2021, 18, 8369. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A. J.; Bergouignan, A.; Dempsey, P. C.; Roschel, H.; Owen, N.; Gualano, B.; Dunstan, D. W. The physiology of sedentary behavior. Physiol. Rev. 2023, 103(4), 2561–2622. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Yang, Y.; Yue, R.; Su, C. Potential causal association between leisure sedentary behaviors, physical activity and musculoskeletal health: a Mendelian randomization study. PLoS One 2023, 18, e0283014. [Google Scholar] [CrossRef]
- Oh, K. H.; Min, J. Y.; Seo, K.; Min, K. B. Association of sedentary lifestyle with skeletal muscle strength and mass in US adolescents: results from the National Health and Nutrition Examination Survey (2011-2014). J. Prev. Med. Public Health 2025, 58, 278–288. [Google Scholar] [CrossRef]
- Nunes, E. A.; Stokes, T.; McKendry, J.; Currier, B. S.; Phillips, M. Disuse-induced skeletal muscle atrophy in disease and non-disease states in humans: mechanisms, prevention, and recovery strategies. Am. J. Physiol. Cell Physiol. 2022, 322, C1068–C1084. [Google Scholar] [CrossRef]
- Guthold, R. Global trends in insufficient physical activity among adolescents: a pooled analysis of 298 population-based surveys with 1.6 million participants. Lancet Child Adolesc. Health 2020, 4, 23–35. [Google Scholar] [CrossRef]
- Joergensen, A. C.; Strandberg-Larsen, K.; Andersen, P. K. Spinal pain in pre-adolescence and the relation with screen time and physical activity behavior. BMC Musculoskelet. Disord. 2021, 22, 393. [Google Scholar] [CrossRef] [PubMed]
- Seyedahmadi, M.; Rostami, J.; Khalaghi, K. The relationship between mobile phone addiction and changes in spinal column angles of male high school students with endomorphic body type. Int. J. Prev. Med. 2025, 16, 30. [Google Scholar] [CrossRef]
- Chu, E.; Mok, T.; Ng, G. Pediatric text neck syndrome. Cureus 2023, 15, e38034. [Google Scholar] [CrossRef]
- Ahn, M. E.; Park, S. M. Prevention and treatment device of turtle neck syndrome. Republic of Korea Patent No. 10-0869693, 2020. [Google Scholar]
- David, D.; Giannini, C.; Chiarelli, F.; Mohn, A. Text neck syndrome in children and adolescents. Int. J. Environ. Res. Public Health 2021, 18, 1565. [Google Scholar] [CrossRef]
- Yong, M. S.; Lee, H. Y. Does forward head posture influence muscle tone, stiffness, and elasticity in university students? J. Clin. Med. 2025, 14, 1888. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, V.; Chandy, M. J.; Babu, K. S. Quantitative study of muscle spindles in suboccipital muscles of human foetuses. Neurol. India 2001, 49, 355–359. [Google Scholar] [PubMed]
- Martinez-Merinero, P.; Nunez-Nagy, S.; Achalandabaso-Ochoa, A.; Fernandez-Matias, R.; Pecos-Martin, D.; Gallego-Izquierdo, T. J. Relationship between forward head posture and tissue mechanosensitivity: a cross-sectional study. J. Clin. Med. 2020, 9, 634. [Google Scholar] [CrossRef] [PubMed]
- Sung, Y. H. Upper cervical spine dysfunction and dizziness. J. Exerc. Rehabil. 2020, 16, 385–391. [Google Scholar] [CrossRef]
- Li, Y.; Yang, L.; Dai, C.; Peng, B. Proprioceptive cervicogenic dizziness: a narrative review of pathogenesis, diagnosis, and treatment. J. Clin. Med. 2022, 11, 6293. [Google Scholar] [CrossRef]
- Chu, Chun; Pu, E. Cervical radiculopathy as a hidden cause of angina: cervicogenic angina. J. Med. Cases 2022, 13, 545–550. [Google Scholar] [CrossRef]
- Hauser, R. A.; Matias, D.; Rawlings, B. The ligamentous cervical instability etiology of human disease from the forward head-facedown lifestyle: emphasis on obstruction of fluid flow into and out of the brain. Front. Neurol. 2024, 15, 1430390. [Google Scholar] [CrossRef]
- Hauser, R. A. Hauser’s laws on the ligamentous structural causes of chronic disabling symptoms of human diseases. Open J. Neurol. Brain Disord. 2024, 7, 000254. [Google Scholar] [CrossRef]
- Monteiro, C. A.; Moubarac, J.-C.; Cannon, G.; Ng, S. W.; Popkin, B. M. Ultra-processed products are becoming dominant in the global food system. Obes. Rev. 2013, 14, 21–28. [Google Scholar] [CrossRef]
- Monteiro, C. A.; Levy, R. B.; Claro, R. M.; Ribeiro de Castro, I. R.; Cannon, G. Increasing consumption of ultra-processed foods and likely impact on human health: evidence from Brazil. Public Health Nutr. 2010, 14, 5–13. [Google Scholar] [CrossRef]
- Al-Jawaldeh, A.; Taktouk, M.; Nasreddine, L. Food consumption patterns and nutrient intakes of children and adolescents in the Eastern Mediterranean Region: a call for policy action. Nutrients 2020, 12, 3345. [Google Scholar] [CrossRef]
- Morais, R.; Rodrigues, M.; Ferreira, F.; Barros, R.; Padrao, P.; Ortigao, M.; Tavares, M.; Moreira, P. Ultra-processed foods and nutritional intake of children and adolescents from Cantagalo, São Tomé and Príncipe. Children 2024, 11(9), 1089. [Google Scholar] [CrossRef] [PubMed]
- Gomes, G. M.; Souza, R. C. V.; dos Santos, T. N.; dos Santos, L. C. Screen exposure in 4-year-old children: association with development, daily habits, and ultra-processed food consumption. Int. J. Environ. Res. Public Health 2024, 21, 1504. [Google Scholar] [CrossRef]
- Rodriguez-Barniol, M.; Pujol-Busquets, G.; Bach-Faig, A. Screen time use and ultra-processed food consumption in adolescents: a focus group qualitative study. J. Acad. Nutr. Diet. 2024, 124(10), 1336–1346. [Google Scholar] [CrossRef] [PubMed]
- Zaretsky, J.; Griess-Fishheimer, S.; Carmi, A.; Travinsky Shmul, T.; Ofer, L.; Sinai, T.; Penn, S.; Shahar, R.; Monsonego-Ornan, E. Ultra-processed food targets bone quality via endochondral ossification. Bone Res. 2021, 9, 14. [Google Scholar] [CrossRef] [PubMed]
- Travinsky-Shmul, T.; Beresh, O.; Zaretsky, J.; Griess-Fishheimer, S.; Rozner, R.; Kalev-Altman, R.; Penn, S.; Shahar, R.; Monsonego-Ornan, E. Ultra-processed food impairs bone quality, increases marrow adiposity and alters gut microbiome in mice. Foods 2021, 10, 3107. [Google Scholar] [CrossRef]
- Kong, W.; Xie, Y.; Hu, J.; Ding, W.; Cao, C. Higher ultra processed foods intake is associated with low muscle mass in young to middle-aged adults: a cross-sectional NHANES study. Front. Nutr. 2024, 11, 1280665. [Google Scholar] [CrossRef]
- Zhang, S.; Zhang, S.; Gu, Y.; Rayamajhi, S.; Thapa, A.; Meng, G.; Zhang, Q.; Liu, L.; Wu, H.; Zhang, T.; Wang, X.; Dong, J.; Zheng, X.; Cao, Z.; Zhang, X.; Dong, X.; Sun, S.; Wang, X.; Zhou, M.; Niu, K. Ultra-processed food intake is associated with grip strength decline in middle-aged and older adults: a prospective analysis of the TCLSIH study. Eur. J. Nutr. 2021, 61(3), 1331–1341. [Google Scholar] [CrossRef]
- Zhang, B.; Shi, H.; Cai, W.; Yang, B.; Xiu, W. Metabolic syndrome in children and adolescents: definitions, epidemiology, pathophysiology, interventions, and challenges. Front. Endocrinol. 2025, 16, 1512642. [Google Scholar] [CrossRef]
- Teraguchi, M.; Yoshimura, N.; Hashizume, H.; Muraki, S.; Yamada, H.; Oka, H.; Minamide, A.; Ishimoto, Y.; Nagata, K.; Kagotani, R.; Tanaka, S.; Kawaguchi, H.; Nakamura, K.; Akune, T.; Yoshida, M. Metabolic syndrome components are associated with intervertebral disc degeneration: the Wakayama Spine Study. PLoS One 2016, 11, e0147565. [Google Scholar] [CrossRef]
- Li, S.; Du, J.; Huang, Y.; Gao, S.; Zhao, Z.; Chang, Z.; Zhang, X.; He, B. From hyperglycemia to intervertebral disc damage: exploring diabetic-induced disc degeneration. Front. Immunol. 2024, 15, 1355503. [Google Scholar] [CrossRef]
- Huang, Z.; Chen, J. J.; Su, Y.-C.; Guo, M.; Chen, Y.; Zhu, Y.; Nie, G.; Ke, R.; Chen, H.; Hu, J. Impact of dyslipidemia on the severity of symptomatic lumbar spine degeneration: a retrospective clinical study. Front. Nutr. 2022, 9, 1033375. [Google Scholar] [CrossRef]
- Dimitri, P. The impact of childhood obesity on skeletal health and development. J. Obes. Metab. Syndr. 2019, 28, 4–17. [Google Scholar] [CrossRef]
- Hasan, S.; Naseer, S.; Zamzam, M.; Mohilldean, H.; Wagoner, C.; Hasan, A. I.; Saleh, E. S.; Uhley, V.; Kamel-ElSayed, S. Nutrient and hormonal effects on long bone growth in healthy and obese children: a literature review. Children 2024, 11, 817. [Google Scholar] [CrossRef]
- Sinha, R.; Dufour, S.; Petersen, K. F.; LeBon, V.; Enoksson, S.; Ma, Y.-Z.; Savoye, M.; Rothman, D. L.; Shulman, G. I.; Caprio, S. Assessment of skeletal muscle triglyceride content by 1H nuclear magnetic resonance spectroscopy in lean and obese adolescents: relationships to insulin sensitivity, total body fat, and central adiposity. Diabetes 2002, 51, 1022–1027. [Google Scholar] [CrossRef]
- Vieira, F. T.; Cai, Y.; Gonzalez, M. C.; Goodpaster, B. H.; Prado, C. M.; Haqq, A. M. Poor muscle quality: a hidden and detrimental health condition in obesity. Rev. Endocr. Metab. Disord. 2025, 26(5), 723–744. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).