Submitted:
23 January 2026
Posted:
26 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Sampling Data and Morphological Analysis
2.3. Molecular Analysis
2.4. Reproductive Analysis
2.5. Spanish Tetraodontiform Species and Vulnerability
2.6. Marine Biotoxins
3. Results
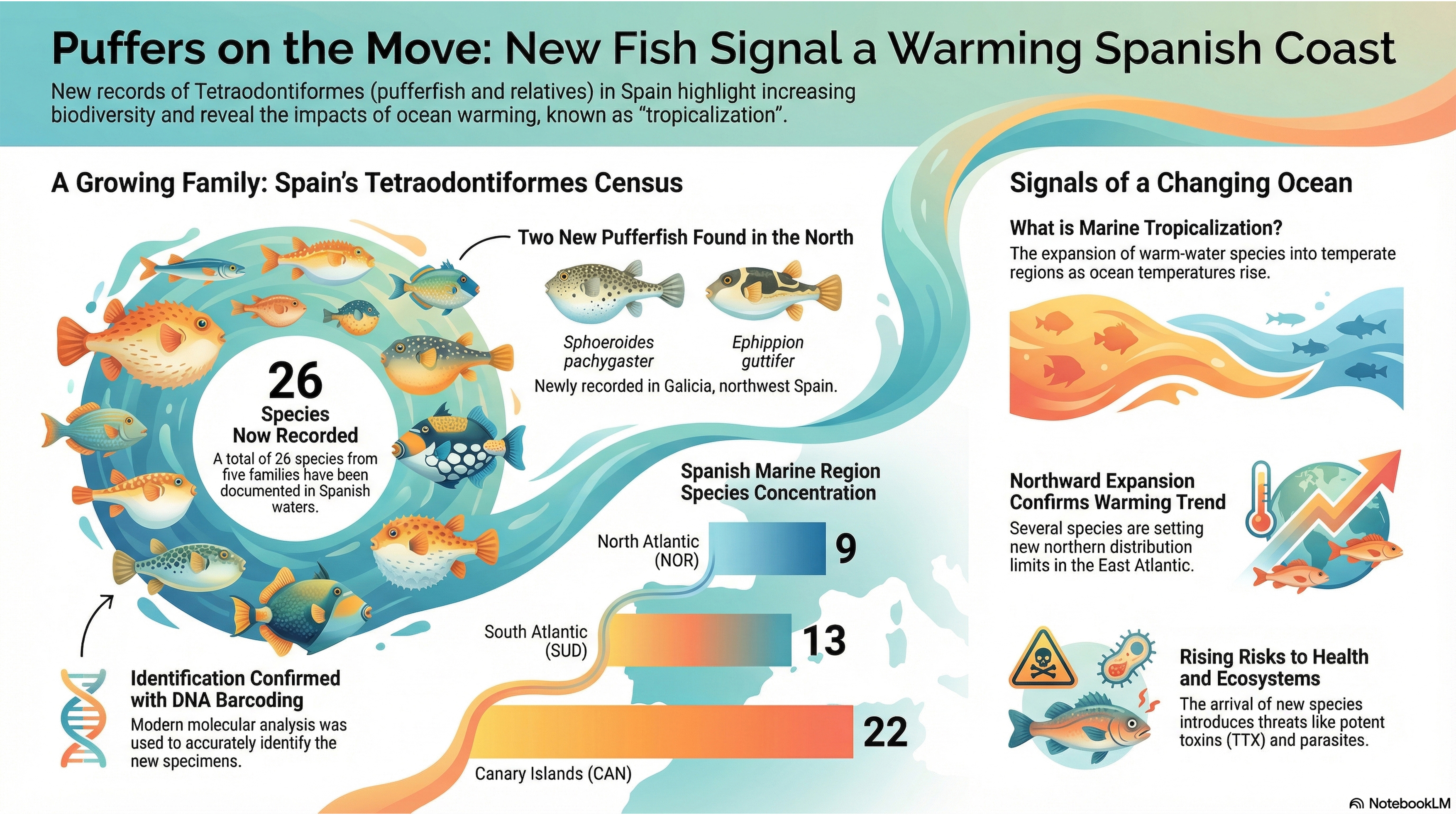
3.1. New Tetraodontidae Records
3.1.1. Molecular Analysis
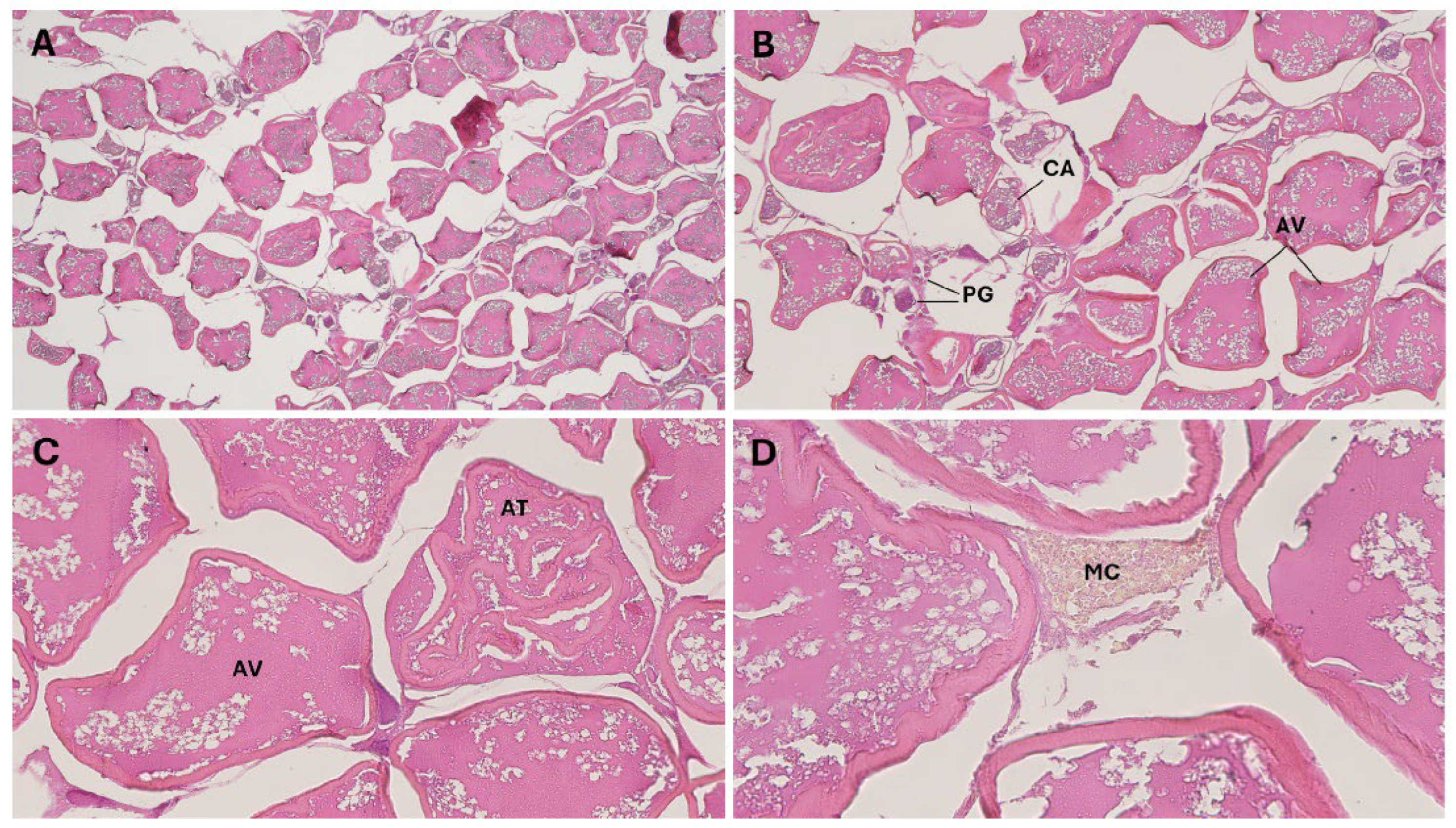
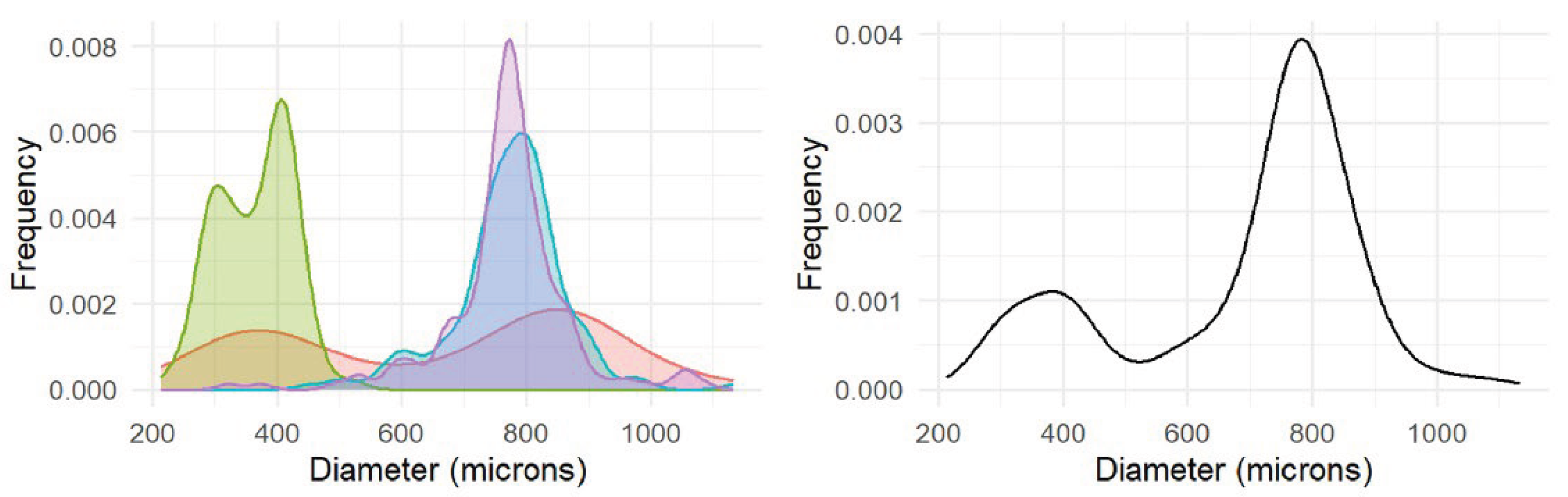
3.1.2. Reproductive Analysis
3.2. Spanish Tetraodontiform Species
3.2.1. Vulnerability Status
3.2.2. Report of TTXs and/or PSTs in the Spanish Tetraodontiform Species
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Near, T.J.; Thacker, C.E. Phylogenetic classification of living and fossil ray-finned fishes (Actinopterygii). Bull. Peabody Mus. Nat. Hist. 2024, 65, 3–302. [Google Scholar] [CrossRef]
- Matsuura, K. Taxonomy and systematics of tetraodontiform fishes: a review focusing primarily on progress in the period from 1980 to 2014. Ichthyol. Res. 2015, 62, 72–113. [Google Scholar] [CrossRef]
- Fricke, R.; Eschmeyer, W.N.; Fong, J.D. Eschmeyer’s catalog of fishes: genera/species by family/subfamily. Electronic version. http://researcharchive.calacademy.org/research/ichthyology/catalog/SpeciesByFamily.asp (accessed on 12 December 2025).
- Neafsey, D.E.; Palumbi, S.R. Genome size evolution in pufferfish: a comparative analysis of diodontid and tetraodontid pufferfish genomes. Genome Res. 2003, 13, 821–830. [Google Scholar] [CrossRef]
- Noguchi, T.; Ebesu, J.S.M. Puffer poisoning: epidemiology and treatment. J. Toxicol. Toxin Rev. 2001, 20, 1–10. [Google Scholar] [CrossRef]
- Sabatino, L.; Esposito, G.; Meloni, D. Occurrence of silver-cheeked toadfish Lagocephalus sceleratus (Tetraodontiformes: Tetraodontidae) and tetrodotoxins in the Mediterranean Sea: Implications for the Italian fishing sector? Reg. Stud. Mar. Sci. 2024, 75, 103562. [Google Scholar] [CrossRef]
- Liu, K.; Sun, H.; Zhao, X.; Wang, C.; An, C.; Li, A.; Liu, S.; Zhuang, Z. DNA barcoding, identification, and validation of the pufferfish (Order: Tetraodontiformes) in China coastal waters. Ecol. Evol. 2024, 14, e10944. [Google Scholar] [CrossRef]
- Santini, F.; Sorenson, L.; Alfaro, M.E. A new phylogeny of tetraodontiform fishes (Tetraodontiformes, Acanthomorpha) based on 22 loci. Mol. Phylogenet. Evol. 2013, 69, 177–187. [Google Scholar] [CrossRef]
- Masuda, Y.; Takeoka, S.; Tabeta, O.; Dotsa, Y. Genetic differences between two newly described swellfishes of the genus Lagocephalus from southern Kyushu, Japan. Nippon Suisan Gakk 1987, 53, 767–772. [Google Scholar] [CrossRef]
- Turan, C.; Gürlek, M.; Ergüden, D.; Uyan, A.; Karan, S.; Doğdu, S.A. Assessing DNA barcodes for identification of pufferfish species (Tetraodontidae) in Turkish marine waters. Nat. Eng. Sci. 2017, 2, 48–59. [Google Scholar] [CrossRef]
- Bañón, R.; Alonso-Fernández, A.; Barros-García, D.; Rios, M.; de Carlos, A. Geographic range expansion of Ephippion guttifer (Tetraodontidae) in the northeastern Atlantic. J. Fish Biol. 2018, 93, 733–737. [Google Scholar] [CrossRef]
- Virgili, R.; Fasciglione, P.; Tiralongo, F.; Pannone, R.; Palomba, M.; Santoro, M.; De Luca, P.; Crocetta, F. Interdisciplinary approaches at early invasion stages maximise the evidence on human-induced and climate-driven fish dispersal. Hydrobiologia 2025, 852, 2241–2261. [Google Scholar] [CrossRef]
- Essl, F.; Dullinger, S.; Genovesi, P.; Hulme, P.E.; Jeschke, J.M.; Katsanevakis, S.; Kühn, I.; Lenzner, B.; Pauchard, A.; Pysek, P.; Rabitsch, W.; Richardson, D.M.; Seebens, H.; Van Kleunen, M.; Van Der Putten, W.H.; Vila, M.; Bacher, S. A conceptual framework for range-expanding species that track human-induced environmental change. BioScience 2019, 69, 908–919. [Google Scholar] [CrossRef]
- Ulman, A.; Abd Rabou, A.F.N.; Al Mabruk, S.; Bariche, M.; Bilecenoğlu, M.; Demirel, N.; Galil, B.S.; Hüseyinoglu, M.F.; Jimenez, C.; Hadjioannou, L.; Kosker, A.R.; Peristeraki, P.; Saad, A.; Samaha, Z.; Stoumboudi, M.Th.; Temraz, T.A.; Karachle, P.K. Assessment of human health impacts from invasive pufferfish (attacks, poisonings and fatalities) across the Eastern Mediterranean. Biology 2024, 13, 208. [Google Scholar] [CrossRef] [PubMed]
- Kawase, H.; Okata, Y.; Ito, K. Role of huge geometric circular structures in the reproduction of a marine pufferfish. Sci. Rep. 2013, 3, 2106. [Google Scholar] [CrossRef]
- Kawase, H.; Okata, Y.; Ito, K. Spawning behavior and paternal egg care in a circular structure constructed by pufferfish, Torquigener albomaculosus (Pisces: Tetraodontidae). Bull. Mar. Sci. 2014, 91, 33–43. [Google Scholar] [CrossRef]
- Sánchez-Cárdenas, R.; Arellano-Martínez, M.; Valdez-Pineda, M.C.; Morán-Angulo, R.E.; Ceballos-Vázquez, B.P. Reproductive cycle and sexual maturity of Sphoeroides annulatus (Jenyns, 1842) (Tetraodontiformes, Tetraodontidae) from the coast of Mazatlan, Sinaloa, Mexico. J. Appl. Ichthyol. 2011, 27, 1190–1196. [Google Scholar] [CrossRef]
- Valdez-Pineda, M.; Morán-Angulo, R.; Voltolina, D.; Castillo-Vargasmachuca, S. Population structure and reproductive aspects of puffer fish Sphoeroides annulatus (Jenyns, 1842) (Osteichthyes: Tetraodontidae), landed in Teacapán, Sinaloa, Mexico. Lat. Am. J. Aquat. Res. 2017, 42, 121–126. [Google Scholar] [CrossRef]
- Peniche-Pérez, J.; González-Salas, C.; Villegas-Hernández, H.; Díaz-Gamboa, R.; Aguilar-Perera, A.; Guillen-Hernández, S.; Poot-López, G. Reproductive biology of the southern pufferfish, Sphoeroides nephelus (Actinopterygii: Tetraodontiformes: Tetraodontidae), in the northern coast off the Yucatan Peninsula, Mexico. Acta Ichthyol. Pisc 2019, 49, 133–146. [Google Scholar] [CrossRef]
- Anupama, K.M.; Hari Sankar, H.S.; Rithin Raj, M.; Harikrishnan, M. Reproductive biology of malabar pufferfish Carinotetraodon travancoricus (Tetraodontidae). J. Ichthyol. 2019, 59, 545–554. [Google Scholar] [CrossRef]
- Momota, K.; Doi, H.; Obata, H.; Sakai, H. Tandem spawning and development in the African freshwater pufferfish Tetraodon schoutedeni. Zoo Biol. 2023, 42, 357–363. [Google Scholar] [CrossRef]
- Chandana, B.; Sanal, A.; Raghavan, R.; Varghese, B. Embryonic development of the world’s smallest puffer fish, Carinotetraodon travancoricus – a threatened freshwater fish of the Western Ghats Biodiversity Hotspot. Zygote 2024, 32, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Sonoyama, T.; Yamada, M.; Gao, W.; Tatsuno, R.; Takatani, T.; Arakawa, O. Co-occurrence of tetrodotoxin and saxitoxins and their intra-body distribution in the pufferfish Canthigaster valentini. Toxins 2020, 12, 436. [Google Scholar] [CrossRef]
- Barrientos, R.G.; Hernández-Mora, G.; Alegre, F.; Field, T.; Flewelling, L.; McGrath, S.; Deeds, J.; Chacón, Y.S.; Rojas Arrieta, K.; Vargas, E.C.; Artavia, K.B.; Stacy, B.A. Saxitoxin poisoning in green turtles (Chelonia mydas) linked to scavenging on mass mortality of Caribbean sharpnose puffer fish (Canthigaster rostrata-Tetraodontidae). Front. Vet. Sci. 2019, 6, 466. [Google Scholar] [CrossRef]
- Rey, V.; Rossignoli, A. E.; Rodríguez, F.; Blanco, J.; Garrido, S.; Ben-Gigirey, B. Evaluation of the prevalence of Paralytic Shellfish Toxins in non-traditional vectors and potential health risks associated to their consumption. Food Control 2025, 176, 111351. [Google Scholar] [CrossRef]
- Ben-Gigirey, B.; Rossignoli, A.E.; Riobó, P.; Rodríguez, F. First report of paralytic shellfish toxins in marine invertebrates and fish in Spain. Toxins 2020, 12, 723. [Google Scholar] [CrossRef]
- Ben-Gigirey, B.; Soliño, L.; Bravo, I.; Rodríguez, F.; Casero, M.V. Paralytic and amnesic shellfish toxins impacts on seabirds, analyses and management. Toxins 2021, 13, 454. [Google Scholar] [CrossRef]
- Katikou, P.; Gokbulut, C.; Kosker, A.R.; Campàs, M.; Ozogul, F. An updated review of tetrodotoxin and its peculiarities. Mar. Drugs 2022, 20, 47. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, T.; Onuki, K.; Arakawa, O. Tetrodotoxin poisoning due to pufferfish and gastropods, and their intoxication mechanism. Int. Sch. Res. Notices 2011, 2011, 276939. [Google Scholar] [CrossRef] [PubMed]
- Martínez, A.; Garrido-Maestu, A.; Ben-Gigirey, B.; Chapela, M.J.; González, V.; Vieites, J.M.; Cabado, A.G. Marine Biotoxins. In Emerging Marine Biotoxins; Kim, S.K., Ed.; Springer Handbook of Marine Biotechnology; Springer Handbooks. Springer: Berlin, Heidelberg, Germany, 2015; pp. 869–904. [Google Scholar] [CrossRef]
- Almón, B.; Bañón, R. Research trends and state of knowledge of decapod crustaceans in Spain: A bibliometric analysis. Ecologies 2025, 6, 63. [Google Scholar] [CrossRef]
- Shipp, R.L. The pufferfishes (Tetraodontidae) of the Atlantic Ocean. GCRL Publications 1974, 41, 1–162. [Google Scholar]
- Ivanova, N.V.; Zemlak, T.S.; Hanner, R.H.; Hebert, P.D.N. Universal primer cocktails for fish DNA barcoding. Mol. Ecol. Notes 2007, 7, 544–548. [Google Scholar] [CrossRef]
- Sanger, F.; Nicklen, S.; Coulson, A.R. DNA sequencing with chain-terminating inhibitors. PNAS 1977, 74, 5463–5467. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular evolutionary genetics analysis version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef]
- Bagenal, T.B.; Braum, E. Eggs and early life history. In Methods for Assessment of Fish Production in Fresh Waters; Bagenal, T.B., Ed.; Blackwell Scientific: Oxford, 1978; pp. 165–201. [Google Scholar]
- Witthames, P.R.; Thorsen, A.; Murua, H.; Saborido-Rey, F.; Greenwood, L.N.; Dominguez, R.; Korta, M.; Kjesbu, O.S. Advances in methods for determining fecundity: application of the new methods to some marine fishes. Fish. Bull. 2009, 107, 148–164. [Google Scholar]
- Lowerre-Barbieri, S.K.; Barbieri, L.R. A new method of oocyte separation and preservation for fish reproduction studies. Fish. Bull. 1993, 91, 159–164. [Google Scholar]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; Tinevez, J.Y.; White, D.J.; Hartenstein, V.; Eliceiri, K.; Tomancak, P.; Cardona, A. Fiji: an open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A language and environment for statistical computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; https://www.R-project.org/.
- Posit; team. RStudio: Integrated Development Environment for R. Posit Software, PBC; Boston, MA, 2025; http://www.posit.co/.
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer-Verlag New York, 2016; https://ggplot2.tidyverse.org[R1ISBN 978-3-319-24277-4.
- GBIF Occurrence Download. Available online: https://www.gbif.org/es/ (accessed on 12 November 2025). [CrossRef]
- Báez, C.; Rodríguez-Cabello, C.; Bañón, R.; Brito, A.; Falcón, J.M.; Maño, T.; Baro, J.; Macías, D.; Meléndez, M.J.; Camiñas, J.A.; Arias-García, A.; Gil, J.; Farias, C.; Artexe, I.; Sánchez, F. Updating the national checklist of marine fishes in Spanish waters: An approach to priority hotspots and lessons for conservation. Mediterr. Mar. Sci. 2019, 20, 260–270. [Google Scholar] [CrossRef]
- Guallart, J.; Vicent, J.J. First record of the unicorn leatherjacket Aluterus monoceros (Pisces: Monacanthidae) from the Mediterranean Sea. Mar. Biodivers. Rec. 2009, 2, e103. [Google Scholar] [CrossRef]
- Azzurro, E.; Bariche, M.; Cerri, J.; Garrabou, J. The long reach of the Suez Canal: Lagocephalus sceleratus (Gmelin, 1789) an unwanted Indo-Pacific pest at the Atlantic gates. BioInvasions Rec. 2020, 9, 204–208. [Google Scholar] [CrossRef]
- The IUCN Red List of Threatened Species. 1964 - onwards. Version 2025-1. Available online: https://www.iucnredlist.org (accessed on 23 November 2025).
- Nieto, A.; Ralph, G.M.; Comeros-Raynal, M.T.; Kemp, J.; García-Criado, M.; Allen, D.J.; Dulvy, N.K.; Walls, R.H.L.; Russell, B.; Pollard, D.; et al. European Red List of marine fishes; Publications Office of the European Union: Luxembourg, 2015; pp. 1–81. [Google Scholar]
- Silva, M.; Rodríguez, I.; Barreiro, A.; Kaufmann, M.; Neto, A.I.; Hassouani, M.; Sabour, B.; Alfonso, A.; Botana, L.M.; Vasconcelos, V. Tetrodotoxins occurrence in non-traditional vectors of the north atlantic waters (Portuguese maritime territory, and morocco coast). Toxins 2019, 11, 306. [Google Scholar] [CrossRef]
- Pinto, E.P.; Rodrigues, S.M.; Gouveia, N.; Timóteo, V.; Costa, P.R. Tetrodotoxin and saxitoxin in two native species of puffer fish, Sphoeroides marmoratus and Lagocephalus lagocephalus, from NE Atlantic Ocean (Madeira Island, Portugal). Mar. Environ. Res. 2019, 151, 104780. [Google Scholar] [CrossRef]
- Rambla-Alegre, M.; Reverté, L.; Del Río, V.; de la Iglesia, P.; Palacios, O.; Flores, C.; Caixach, J.; Campbell, K.; Elliott, C.T.; Izquierdo-Muñoz, A.; Campàsa, M.; Diogène, J. Evaluation of tetrodotoxins in puffer fish caught along the Mediterranean coast of Spain. Toxin profile of Lagocephalus sceleratus. Environ. Res. 2017, 158, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Malloggi, C.; Rizzo, B.; Giusti, A.; Guardone, L.; Gasperetti, L.; Dall’Ara, S.; Armani, A. First toxicological analysis of the pufferfish Sphoeroides pachygaster collected in Italian waters (Strait of Sicily): Role of citizens science in monitoring toxic marine species. Animals 2023, 13, 1873. [Google Scholar] [CrossRef] [PubMed]
- Alkassar, M.; Tudó, À.; Rambla-Alegre, M.; Ferreres, L.; Diogène, J.; Sureda, F.X.; Campàs, M. First record of paralytic shellfish toxins in marine pufferfish from the Spanish Mediterranean coast using cell-based assay, automated patch clamp and HPLC-FLD. Chemosphere 2024, 364, 143053. [Google Scholar] [CrossRef]
- Oliveira, J.S.; Pires Junior, O.R.; Morales, R.A.V. Toxicity of Puffer fish-two species (Lagocephalus laevigatus, Linaeus 1766 and Sphoeroides spengleri, Bloch 1785) from the southeren Brazilian coast. J. Venom. Anim. Toxins Incl. Trop. 2003, 9, 76–82. [Google Scholar] [CrossRef]
- Rodríguez, P.; Alfonso, A.; Otero, P.; Katikou, P.; Georgantelis, D.; Botana, L.M. Liquid chromatography–mass spectrometry method to detect Tetrodotoxin and Its analogues in the puffer fish Lagocephalus sceleratus (Gmelin, 1789) from European waters. Food Chem. 2012, 132, 1103–1111. [Google Scholar] [CrossRef]
- Saito, T.; Noguchi, T.; Shida, Y.; Abe, T.; Hashimoto, K. Screening of Tetrodotoxin and Its Derivatives in Puffer-related Species. Nippon Suisan Gakk 1991, 57, 1573–1577. [Google Scholar] [CrossRef]
- Yu, C.; Yu, P.H. A preliminary study of puffer fishes and their toxins found in Hong Kong waters. J. Food Hyg. Soc. Japan 1997, 7(38), 460–463. [Google Scholar] [CrossRef]
- Tamele, I.J.; Timba, I.; Costa, P.R.; Vasconcelos, V. Tetrodotoxin and analogues in two local pufferfish species from Inhaca Island–South of Mozambique: First report in the Mozambican coast. Toxicon 2022, 216, 88–91. [Google Scholar] [CrossRef]
- Elshaer, F.M. Comparative histopathological studies on kidney and liver of rat treated by tetrodotoxin (TTX) extracted from gonads and muscles of porcupine fish species. Int. J. Fish. Aquat. Stud. 2016, 4, 355–360. [Google Scholar]
- Baptista, M.; Braga, A.C.; Rosa, R.; Costa, P.R. Does ocean sunfish Mola spp. (Tetraodontiformes: Molidae) represent a risk for tetrodotoxin poisoning in the Portuguese coast? Mar. Drugs 2022, 20, 594. [Google Scholar] [CrossRef]
- Giusti, A.; Guarducci, M.; Stern, N.; Davidovich, N.; Golani, D.; Armani, A. The importance of distinguishing pufferfish species (Lagocephalus spp.) in the Mediterranean Sea for ensuring public health: Evaluation of the genetic databases reliability in supporting species identification. Fish. Res. 2019, 210, 14–21. [Google Scholar] [CrossRef]
- ICES. Report of the Working Group on the Application of Genetics in Fisheries and Mariculture (WGAGFM). ICES WGAGFM Report, Reykjavik, 7–9 May 2013, Iceland. ICES, Copenhagen, Denmark, 2013; pp. 1–52. [Google Scholar]
- Muhammad Tahir, H.; Akhtar, S. Services of DNA barcoding in different fields. Mitochondrial DNA A 2016, 27, 4463–4474. [Google Scholar] [CrossRef]
- Lowerre-Barbieri, S.K.; Brown-Peterson, N.J.; Wyanski, D.M.; Moncrief-Cox, H.E.; Kolmos, K.J.; Menendez, H.S.; Barnett, B.K.; Friess, C. A unified framework and terminology for reproductive traits integral to understanding fish population productivity. Mar. coast. fish. 2023, 15, e210276. [Google Scholar] [CrossRef]
- Sirisha, R.; Imandi; Rao, P.Y. Maturation and spawning of green – rough – back pufferfish, Lagocephalus lunaris. NVBS 2021, 4, 8–18. [Google Scholar] [CrossRef]
- Mat Piah, R.; Bucher, D.J. Reproductive Biology of Estuarine Pufferfish, Marilyna pleurosticta and Tetractenos hamiltoni (Teleostei: Tetraodontidae) in Northern New South Wales: Implications for Biomonitoring. Proc. Linn. Soc. N.S.W 2014, 136, 219–229. [Google Scholar]
- Ramadan, A.M.; El-Halfawy, M.M. Reproductive biology of the yellow-spotted puffer Torquigener flavimaculosus (Osteichthyes: Tetraodontidae) from Gulf of Suez, Egypt. EJABF 2019, 23, 503–511. [Google Scholar] [CrossRef]
- Rocha, C.; Favaro, L.F.; Spach, H.L. Reproductive biology of Sphoeroides testudineus (Linneus) (Pisces, Osteichthyes, Tetraodontidae) of the gamboa do Baguaçu, bay of Paranaguá, State of Paraná, Brazil. Rev. Bras. Zool 2002, 19, 57–63. [Google Scholar] [CrossRef]
- Carbonara, P.; Kolitari, J.; Đurović, M.; Gaudio, P.; Ikica, Z.; Kroqi, G.; Milonbe, N.; Spedicato, M.T. The presence of Tetraodontidae species in the Central Mediterranean: an update from the southern Adriatic Sea. Acta Adriat. 2017, 58, 325–338. [Google Scholar] [CrossRef]
- Cornide, J. Ensayo de una historia de los peces y otras producciones marinas de la costa de Galicia. Edición facsmil; Edicións do Castro: O Castro-Sada, Spain, 1788; pp. 1–263. [Google Scholar]
- Arias, A.M.; Romero, P.; De la Torre, M.; Bañón, R. La ictiología de Andalucía. Catálogo crítico e ilustrado de las especies marinas estudiadas por Antonio Cabrera y Corro (1762-1827), canónigo magistral de la catedral de Cádiz; Editorial UCA, Servicio de Publicaciones de la Universidad de Cádiz: Cádiz, Spain, 2025; pp. 1–636. [Google Scholar]
- Pérez-Arcas, L. Sobre T. guttifer en la costa de Málaga. Anales Soc. Esp. Hist. Nat. 1872, 1, 4–7. [Google Scholar]
- De Buen, F. Fauna ictiológica. Catálogo de los peces ibéricos de la planicie continental, aguas dulces, pelágicos y de los abismos próximos. II parte. Not. y Res. Inst. Esp. Ocean. 1935, 82, 89–148. [Google Scholar]
- Lozano-Rey, L. Peces Fisoclistos: O. Bericiformes, Zeiformes, Perciformes, Escorpeniformes y Balistiformes; Memorias de la Real Academia de Ciencias Exactas, Fisicas y Naturales de Madrid: Madrid, España, 1952; Primera Parte; pp. 1–378. [Google Scholar]
- Triay-Portella, R.; Pajuelo, J.G.; Manen, T.P.; Espino, F.; Ruiz-Díaz, R.; Lorenzo, J.M.; González-Pérez, J.A. New records of non-indigenous fishes (Perciformes and Tetraodontiformes) from the Canary Islands (north-eastern Atlantic). Cybium 2015, 39, 163–174. [Google Scholar] [CrossRef]
- Wirtz, P.; Biscoito, M. The distribution of Mola alexandrini in the subtropical eastern Atlantic, with a note on Mola mola. Bocagiana 2019, 245, 1–6. [Google Scholar]
- Falcón, J.M.; Brito, A.; Herrera, R.; Ayza, O.; Moro, L; Caro, M.B. Peces marinos tropicales exóticos de Canarias; Consejería de Transición Ecológica, Lucha contra el Cambio Climático y Planificación Territorial del Gobierno de Canarias, 2023; pp. 1–247. [Google Scholar]
- Bañón, R.; Conde-Pardo, P.; Álvarez-Salgado, X.A.; de Carlos, A.; Arronte, J.C.; Piedracoba, S. Tropicalization of fish fauna of Galician coastal waters, in the NW Iberian upwelling system. Reg. Stud. Mar. Sci. 2024, 70, 103369. [Google Scholar] [CrossRef]
- González, J.A.; Espino, F.; González-Lorenzo, J.G. Changes in biogeographic patterns of coastal fishes: Indicators of tropicalization in the Canary Islands over the last 40 years. Mar. Environ. Res. 2025, 205, 107002. [Google Scholar] [CrossRef] [PubMed]
- Booth, D.J.; Bond, N.; Macreadie, P. Detecting range shifts among Australian fishes in response to climate change. Mar. Freshw. Res. 2011, 62, 1027–1042. [Google Scholar] [CrossRef]
- de Carvalho-Souza, G.F.; Lobato, C.; Mesa, D.A.; González-Ortegón, E. Ongoing tropicalization of marine fishes: is range expansion and establishment occurring in the Gulf of Cadiz (southernmost Europe)? J. Mar. Biol. Assoc. U.K. 2025, 105, e61. [Google Scholar] [CrossRef]
- Torreblanca, D.; Báez, J.C. Tropicalization of the Mediterranean Sea Reflected in Fish Diversity Changes: A Case Study from Spanish Waters. J. Mar. Sci. Eng. 2025, 13, 1119. [Google Scholar] [CrossRef]
- Quéro, J.C.; Du Buit, M.H.; Vayne, J.J. Les captures de poissons à affinités tropicales le long des côtes Atlantiques Européennes. Ann. Soc. Sci. Nat. Charente-Marit. 1996, 8, 651–673. [Google Scholar]
- Quéro, J.C. Changes in the Euro-Atlantic fish species composition resulting from fishing and ocean warming. Ital. J. Zool. 1998, 65, 493–499. [Google Scholar] [CrossRef]
- Quéro, J.C.; Spitz, J.; Vayne, J.J. Faune française de l’Atlantique. Poissons Tetraodontiformes. Ann. Soc. Sci. Nat. Charente-Marit. 2008, 9, 815–832. [Google Scholar]
- Bañón, R.; Santás, V. First record of Lagocephalus laevigatus (Tetraodontiformes, Tetraodontidae) from Galician waters (northwest Spain), a northernmost occurrence in the north-east Atlantic. J. Fish Biol. 2011, 78, 1574–1578. [Google Scholar] [CrossRef] [PubMed]
- Azzurro, E.; Bariche, M.; Cerri, J.; Garrabou, J. The long reach of the Suez Canal: Lagocephalus sceleratus (Gmelin, 1789) an unwanted Indo-Pacific pest at the Atlantic gates. Bioinvasions Rec. 2020, 9, 204–208. [Google Scholar] [CrossRef]
- Azzurro, E.; D’Amen, M. Climate change paves the way for a new inter-ocean fish interchange. Front. Ecol. Environ 2022, 20, 558–563. [Google Scholar] [CrossRef]
- Png-Gonzalez, L.; Comas-González, R.; Calvo-Manazza, M.; Follana-Berná, G.; Ballesteros, E.; Díaz-Tapia, P.; Falcón, J.M.; García Raso, J.E.; Gofas, S.; González-Porto, M.; López, E.; Ramos-Esplá, A.A.; Velasco, E.; Carbonell, A. Updating the national baseline of non-indigenous species in Spanish marine waters. Diversity 2023, 15, 630. [Google Scholar] [CrossRef]
- Espino, F.; Tuya, F.; del Rosario, A.; Bosch, N.E.; Coca, J.; González-Ramos, A.J.; del Rosario, F.; Otero-Ferrer, F.J.; Moreno, A.C.; Haroun, R. Geographical range extension of the spotfin burrfish, Chilomycterus reticulatus (L. 1758), in the Canary Islands: a response to ocean warming? Diversity 2019, 11, 230. [Google Scholar] [CrossRef]
- Stump, E.; Ralph, G.M.; Comeros-Raynal, M.T.; Matsuura, K.; Carpenter, K.E. Global conservation status of marine pufferfishes (Tetraodontiformes: Tetraodontidae). Glob. Ecol. Conserv. 2018, 14, e00388. [Google Scholar] [CrossRef]
- Eduardo, L.N.; Bertrand, A.; Frédou, T.; Lira, A.S.; Lima, R.S.; Ferreira, B.P.; Menard, F.; Lucena-Frédou, F. Biodiversity, ecology, fisheries, and use and trade of Tetraodontiformes fishes reveal their socio-ecological significance along the tropical Brazilian continental shelf. Aquat. Conserv.: Mar. Freshw. Ecosyst. 2020, 30, 761–774. [Google Scholar] [CrossRef]
- Phillips, N.; Nyegaard, M.; Sawai, E.; Chang, C.T.; Baptista, M.; Thys, T. The ocean sunfishes (family Molidae): Recommendations from the IUCN molidae review panel. Mar. Pol. 2023, 155, 105760. [Google Scholar] [CrossRef]
- Ulman, A.; Harris, H.E.; Doumpas, N.; Deniz Akbora, H.; Al Mabruk, S.A.A.; Azzurro, E.; Bariche, M.; Çiçek, B.A.; Deidun, A.; Demirel, N.; Fogg, A.Q.; Katsavenakis, S.; Kletou, D.; Kleitou, P.; Papadopoulou, A.; Ben Souissi, J.; Hall-Spencer, J.M.; Tiralongo, F.; Yildiz, T. Low pufferfish and lionfish predation in their native and invaded ranges suggests human control mechanisms may be necessary to control their Mediterranean abundances. Front. Mar. Sci. 2021, 8, 670413. [Google Scholar] [CrossRef]
- Rodríguez, H.; Bañón, R.; Ramilo, A. The hidden companion of non-native fishes in north-east Atlantic waters. J. Fish Dis. 2019, 42, 1013–1021. [Google Scholar] [CrossRef] [PubMed]
- Azzurro, E. Unusual occurrences of fish in the Mediterranean Sea: an insight into early detection. In Fish Invasions of the Mediterranean Sea: Change and Renewal; Golani, D., Appelbaum-Golani, B., Eds.; Pensoft Publishers: Sofia-Moscow, 2010; pp. 99–126. [Google Scholar]
- Reglamento (CE) Nº 853/2004 del parlamento europeo y del consejo de 29 de abril de 2004 por el que se establecen normas específicas de higiene de los alimentos de origen animal. Diario Oficial de la Unión Europea L 139/55.
- Nakatani, T.; Shimizu, M.; Yamano, T. The contents and composition of tetrodotoxin and paralytic shellfish poisoning toxins in marine pufferfish Canthigaster rivulata. J. Food Hyg. Soc. Japan 2016, 57, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Guardone, L.; Maneschi, A.; Meucci, V.; Gasperetti, L.; Nucera, D.; Armani, A. A global retrospective study on human cases of tetrodotoxin (TTX) poisoning after seafood consumption. Food Rev. Int. 2020, 36, 645–667. [Google Scholar] [CrossRef]
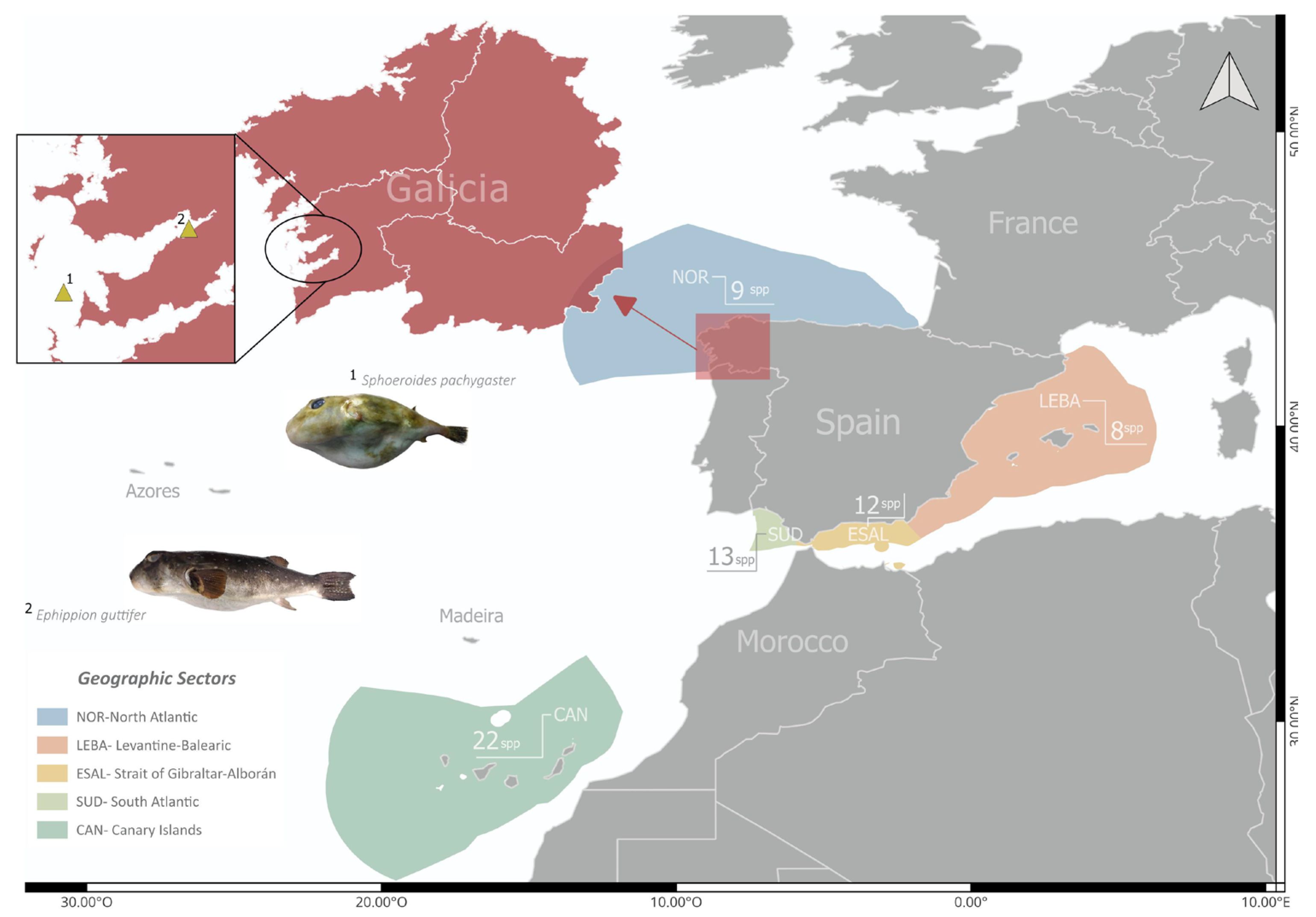


|
Ephippion guttifer MHN USC 25103-1 |
Sphoeroides pachygaster MHN USC 25234 |
|
|---|---|---|
| Total length (mm) | 567 | 220 |
| Standard length (mm) | 462 | 187 |
| As % Standard length | ||
| Head length | 29.6 | 28.9 |
| Pre-orbital length | 14 | 19.7 |
| Post-orbital length | 14.9 | 13.5 |
| Horizontal eye diameter | 5.8 | 8.9 |
| Interorbital width | 13.5 | 17.8 |
| Length of gill opening | 10.2 | 9.8 |
| Pre-dorsal-fin length | 68.4 | 75.5 |
| Dorsal-fin base length | 7.2 | 11.3 |
| Pre-anal-fin length | 71.2 | 81.3 |
| Pre-pectoral-fin length | 31.8 | 43.9 |
| Anal-fin base | 5.3 | 3.9 |
| Pectoral-fin length | 14.8 | 15.3 |
| Caudal peduncle depth | 10.2 | 9.8 |
| Body depth | 28.4 | 29.1 |
| Body width | 28.8 | 24.9 |
| Meristic | ||
| Dorsal fin rays | 10 | 8 |
| Anal fin rays | 9 | 8 |
| Pectoral fin rays | 19 | 14 |
| Caudal-fin rays | 10 | 10 |
| SW | NO | OD | |
|---|---|---|---|
| Subsample1 | 0.046 | 87 | 1891 |
| Subsample2 | 0.042 | 85 | 2024 |
| Subsample3 | 0.02 | 197 | 9850 |
| Subsample4 | 0.029 | 194 | 6690 |
| Family | Species | NOR | SUD | ESAL | LEBA | CAN |
|---|---|---|---|---|---|---|
| Tetraodontidae | Sphoeroides marmoratus (Lowe, 1838) | P | P | P* | P | |
| Tetraodontidae | Sphoeroides pachygaster (Müller & Troschel, 1848) | P | P | P | P* | P |
| Tetraodontidae | Canthigaster capistrata (Lowe, 1839) | P | P* | P | ||
| Tetraodontidae | Lagocephalus laevigatus (Linnaeus, 1766) | P | P* | |||
| Tetraodontidae | Lagocephalus lagocephalus (Linnaeus, 1758) | P | P | P | P | P |
| Tetraodontidae | Lagocephalus sceleratus (Gmelin, 1789) | P* | P | |||
| Tetraodontidae | Ephippion guttifer (Bennett, 1831) | P* | P* | P | ||
| Monacanthidae | Aluterus monoceros (Linnaeus, 1758) | P | P | P* | P | |
| Monacanthidae | Aluterus scriptus (Osbeck, 1765) | P | ||||
| Monacanthidae | Cantherhines pullus (Ranzani, 1842)* | P* | ||||
| Monacanthidae | Cantherhines macrocerus (Hollard, 1853)* | P* | ||||
| Monacanthidae | Stephanolepis hispida (Linnaeus, 1766) | P | ||||
| Diodontidae | Chilomycterus reticulatus (Linnaeus, 1758) | P | ||||
| Diodontidae | Chilomycterus mauretanicus (Le Danois, 1954)* | P* | P* | |||
| Diodontidae | Diodon eydouxii Brisout de Barneville, 1846 | P | P | P | ||
| Diodontidae | Diodon holocanthus Linnaeus, 1758 | P* | P | |||
| Diodontidae | Diodon hystrix Linnaeus, 1758 | P | P* | P | ||
| Molidae | Masturus lanceolatus (Liénard, 1840) | P | ||||
| Molidae | Mola mola (Linnaeus, 1758) | P | P | P | P | P |
| Molidae | Mola alexandrine (Ranzani 1834)* | P* | ||||
| Molidae | Ranzania laevis (Pennant, 1776) | P* | P | P | P | P |
| Balistidae | Canthidermis sufflamen (Mitchill, 1815) | P | ||||
| Balistidae | Canthidermis maculata (Bloch, 1786) | P | ||||
| Balistidae | Melichthys niger (Bloch, 1786)* | P* | ||||
| Balistidae | Balistes capriscus Gmelin, 1789 | P | P | P | P | P |
| Balistidae | Balistes punctatus Gmelin, 1789 | P |
| Species | IUCN Global | IUCN Europe |
|---|---|---|
| Sphoeroides marmoratus | LC | LC |
| Sphoeroides pachygaster | LC | LC |
| Canthigaster capistrata | LC | LC |
| Lagocephalus laevigatus | LC | NE |
| Lagocephalus lagocephalus | LC | LC |
| Lagocephalus sceleratus | LC | NE |
| Ephippion guttifer | LC | DD |
| Aluterus monoceros | LC | DD |
| Aluterus scriptus | LC | DD |
| Cantherhines pullus | LC | NE |
| Cantherhines macrocerus | LC | NE |
| Stephanolepis hispida | LC | LC |
| Chilomycterus reticulatus | LC | LC |
| Chilomycterus mauretanicus | LC | NA |
| Diodon eydouxii | LC | NA |
| Diodon holocanthus | LC | NE |
| Diodon hystrix | LC | DD |
| Masturus lanceolatus | LC | NA |
| Mola mola | VU | DD |
| Mola alexandrini | NE | NE |
| Ranzania laevis | LC | DD |
| Canthidermis sufflamen | LC | NA |
| Canthidermis maculata | LC | LC |
| Melichthys niger | LC | NE |
| Balistes capriscus | VU | DD |
| Balistes punctatus | VU | NE |
| Species | TTX | PST | Source |
|---|---|---|---|
| Sphoeroides marmoratus | D | NE | [49] |
| D | ND | [50] | |
| Sphoeroides pachygaster | ND | NE | [51] |
| ND | ND | [52] | |
| NE | ND | [53] | |
| Canthigaster capistrata | NE | NE | — |
| Lagocephalus laevigatus | D | NE | [54] |
| Lagocephalus lagocephalus | ND | NE | [51] |
| ND | D | [50] | |
| NE | D | [53] | |
| Lagocephalus sceleratus | D | NE | [55] |
| D | NE | [51] | |
| Ephippion guttifer | NE | NE | — |
| Aluterus monoceros | NE | NE | — |
| Aluterus scriptus | NE | NE | — |
| Cantherhines pullus | NE | NE | — |
| Cantherhines macrocerus | NE | NE | — |
| Stephanolepis hispida | NE | NE | — |
| Chilomycterus reticulatus | NE | NE | — |
| Chilomycterus mauretanicus | NE | NE | — |
| Diodon eydouxii | NE | NE | — |
| Diodon holocanthus | D | NE | [56] |
| ND | NE | [57] | |
| Diodon hystrix | D | NE | [58] |
| D | NE | [59] | |
| Masturus lanceolatus | NE | NE | — |
| Mola mola | ND | NE | [56] |
| ND | NE | [60] | |
| Mola alexandrini | ND | NE | [60] |
| Ranzania laevis | NE | NE | — |
| Canthidermis sufflamen | NE | NE | — |
| Canthidermis maculata | NE | NE | — |
| Melichthys niger | NE | NE | — |
| Balistes capriscus | NE | NE | — |
| Balistes punctatus | NE | NE | — |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




