Submitted:
23 January 2026
Posted:
26 January 2026
You are already at the latest version
Abstract
Keywords:
Introduction
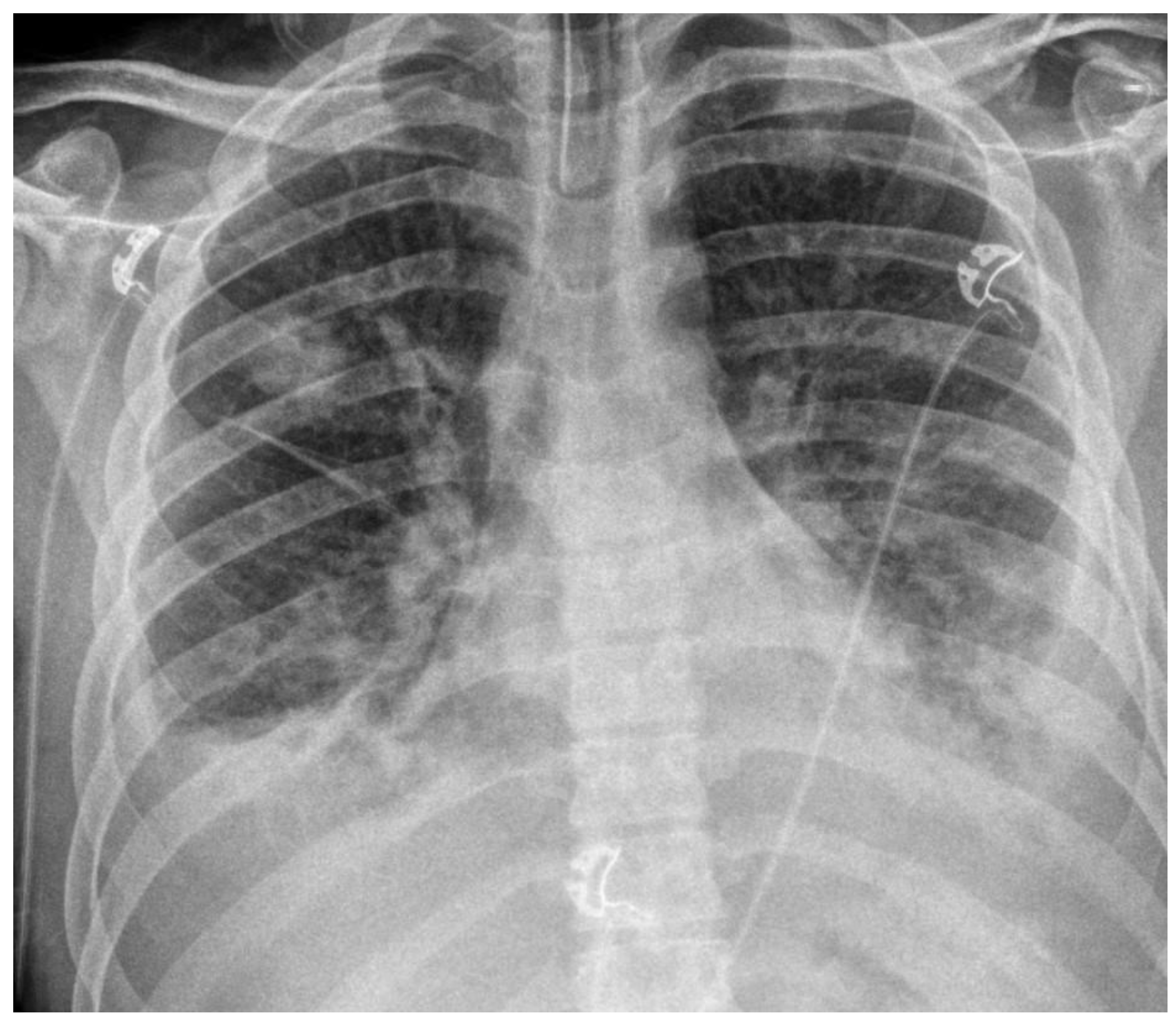
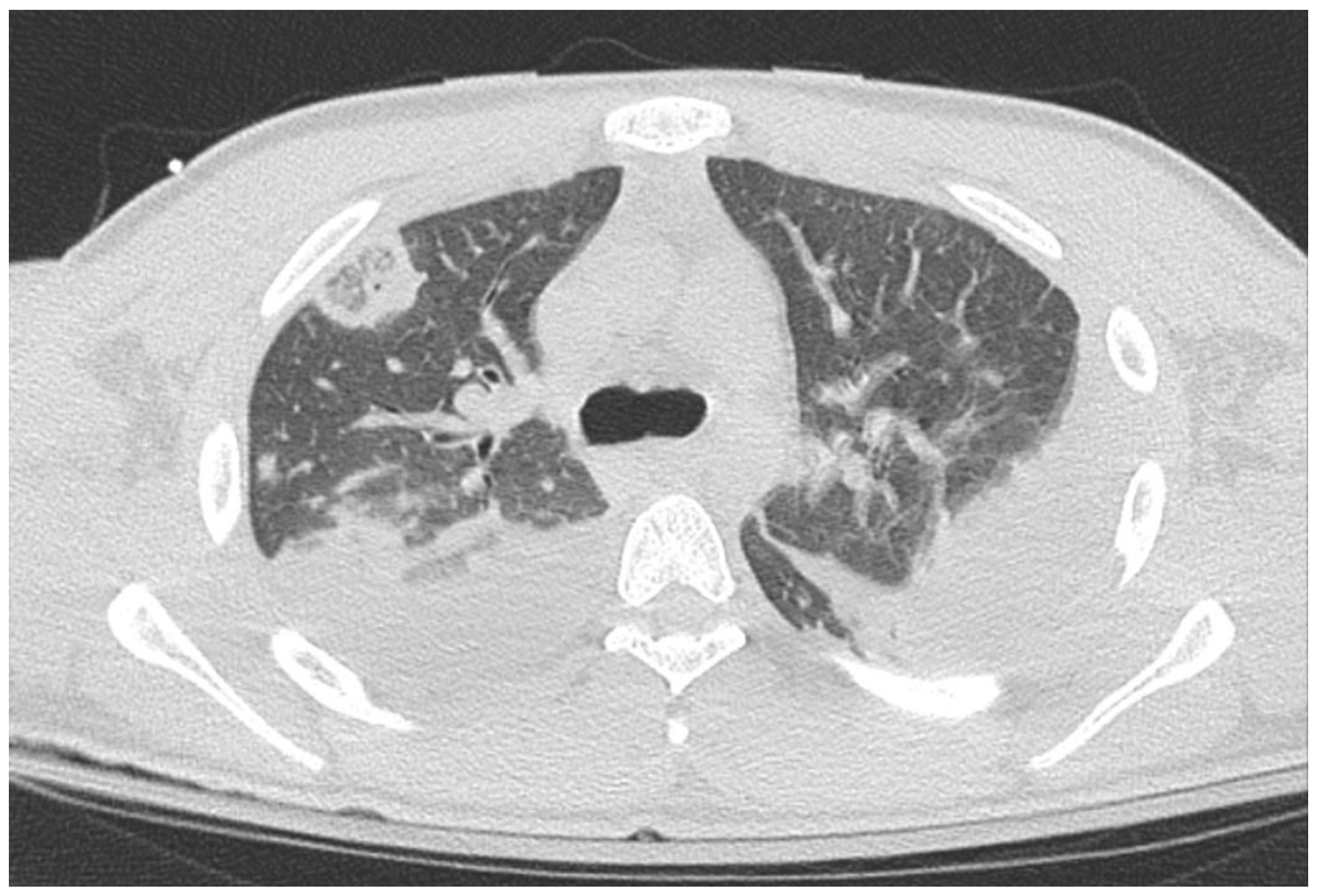
Case Presentation:
References
- Sen, Lee W; Jean, SS; Chen, FL; Hsieh, SM; Hsueh, PR. Lemierre’s syndrome: A forgotten and re-emerging infection. In Journal of Microbiology, Immunology and Infection; Elsevier Ltd, 2020; Vol. 53, pp. 513–7. [Google Scholar]
- Eilbert, W; Singla, N. Lemierre's syndrome. Int J Emerg Med 2013, 6(1), 40. [Google Scholar] [CrossRef]
- Karkos, PD; Asrani, S; Karkos, CD; Leong, SC; Theochari, EG; Alexopoulou, TD; et al. Lemierre’s syndrome: A systematic review. Laryngoscope 2009, 119(8), 1552–9. [Google Scholar] [CrossRef]
- Valerio, L; Corsi, G; Sebastian, T; Barco, S. Lemierre syndrome: Current evidence and rationale of the Bacteria-Associated Thrombosis, Thrombophlebitis and LEmierre syndrome (BATTLE) registry. In Thrombosis Research; Elsevier Ltd, 2020; Vol. 196, pp. 494–9. [Google Scholar]
- Huggan, PJ; Murdoch, DR. Fusobacterial infections: Clinical spectrum and incidence of invasive disease. Journal of Infection 2008, 57(4), 283–9. [Google Scholar] [CrossRef] [PubMed]
- Saar, M; Vaikjärv, R; Parm, Ü; Kasenõmm, P; Kõljalg, S; Sepp, E; et al. Unveiling the etiology of peritonsillar abscess using next generation sequencing. Ann Clin Microbiol Antimicrob 2023, 22(1). [Google Scholar] [CrossRef] [PubMed]
- Afra, K; Laupland, K; Leal, J; Lloyd, T; Gregson, D. Incidence, risk factors, and outcomes of Fusobacterium species bacteremia. BMC Infect Dis 2013, 13(1). [Google Scholar] [CrossRef] [PubMed]
- Liu, Y; Li, Z; Fu, H; Ruan, W; Wang, H; Ding, Y; et al. The first case report: diagnosis and management of necrotizing fusobacterium lung abscess via BALF next-generation sequencing. BMC Infect Dis 2024, 24(1). [Google Scholar] [CrossRef] [PubMed]
- Bank, S; Nielsen, HM; Hoyer Mathiasen, B; Christiansen Leth, D; Hagelskjær Kristensen, L; Prag, J. Fusobacterium necrophorum- detection and identification on a selective agar. APMIS 2010, 118(12), 994–9. [Google Scholar] [CrossRef] [PubMed]
- Shiraishi, Y; Kryukov, K; Tomomatsu, K; Sakamaki, F; Inoue, S; Nakagawa, S; et al. Diagnosis of pleural empyema/parapneumonic effusion by next-generation sequencing. Infect Dis. 2021, 53(6), 450–9. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M; Patel, S; Sadaka, AS; Bedawi, EO; Corcoran, JP; Porcel, JM. Recent insights into the management of pleural infection. In International Journal of General Medicine; Dove Medical Press Ltd, 2021; Vol. 14, pp. 3415–29. [Google Scholar]
- Janda, JM; Abbott, SL. 16S rRNA gene sequencing for bacterial identification in the diagnostic laboratory: Pluses, perils, and pitfalls. Journal of Clinical Microbiology 2007, Vol. 45, 2761–4. [Google Scholar] [CrossRef]
- Hjertman, J; Bläckberg, J; Ljungquist, O. 16S rRNA is a valuable tool in finding bacterial aetiology of community-acquired pleural empyema–a population-based observational study in South Sweden. Infect Dis. 2022, 54(3), 163–9. [Google Scholar] [CrossRef]
- Seiler-Ramadas, R; Sandner, I; Haider, S; Grabovac, I; Dorner, TE. Health effects of electronic cigarette (e-cigarette) use on organ systems and its implications for public health. In Wiener Klinische Wochenschrift; Springer, 2021; Vol. 133, pp. 1020–7. [Google Scholar]
- Goldberg, EA; Venkat-Ramani, T; Hewit, M; Bonilla, HF. Epidemiology and clinical outcomes of patients with Fusobacterium bacteraemia. Epidemiol Infect. 2013, 141(2), 325–9. [Google Scholar] [CrossRef]
- El Chebib, H; McArthur, K; Gorbonosov, M; Domachowske, JB. Anaerobic necrotizing pneumonia: Another Potential Life-threatening Complication of Vaping? Pediatrics 2020, 145(4). [Google Scholar] [CrossRef] [PubMed]
- Venditto, L; Ferrante, G; Caccin, A; Franchini, G; Zaffanello, M; Tenero, L; et al. Lung abscess as a complication of Lemierre Syndrome in adolescents: a single center case reports and review of the literature. Ital J Pediatr 2023, 49(1). [Google Scholar] [CrossRef] [PubMed]
- Sussan, TE; Gajghate, S; Thimmulappa, RK; Ma, J; Kim, JH; Sudini, K; et al. Exposure to electronic cigarettes impairs pulmonary anti-bacterial and anti-viral defenses in a mouse model. PLoS One 2015, 10(2). [Google Scholar] [CrossRef] [PubMed]
- Laube, BL; Afshar-Mohajer, N; Koehler, K; Chen, G; Lazarus, P; Collaco, JM; et al. Acute and chronic in vivo effects of exposure to nicotine and propylene glycol from an E-cigarette on mucociliary clearance in a murine model. Inhal Toxicol 2017, 29(5), 197–205. [Google Scholar] [CrossRef] [PubMed]
- Hwang, JH; Lyes, M; Sladewski, K; Enany, S; McEachern, E; Mathew, DP; et al. Electronic cigarette inhalation alters innate immunity and airway cytokines while increasing the virulence of colonizing bacteria. J Mol Med. 2016, 94(6), 667–79. [Google Scholar] [CrossRef] [PubMed]
| Test | Patient Result (reference range) |
| Serology | |
| Initial Leukocytes | 26 x109/L |
| Initial Platelet | 104 x109/L |
| Initial Hemoglobin | 102 x109/L |
| Initial Creatinine | 180 mmol/L |
| Initial Venous Lactate | 3.0 mmol/L |
| Immunoglobulins | |
| IgG | 19.4g/L (6.35-16.65g/L) |
| IgA | 4.97g/L (0.7-3.5g/L) |
| IgM | 2.33g/L (0.41-2.07g/L) |
| Pleural Analysis (exudative) | |
| Cell Count | 118,760 x106/L |
| Leukocytes | 117,760 x106/L |
| LDH | >10,000 u/L (upper limit of assay detection) |
| Protein | 44g/L |
| Gram Stain + Culture | No Growth |
| Tuberculosis Culture | No Growth |
| Fungal Culture | No Growth |
| 16s PCR | Positive – presence of Fusibacterium Necrophorum |
| Bronchoscopy | |
| Bacterial Culture | Negative |
| Fungal | No fungal elements |
| Aspergillus Galactomannan Antigen | 0.49 (<0.5) |
| Pneumocystis DFA | Not Detected |
| Brucella AB | Non Reactive (<1:20) |
| Viral Panel* | Negative |
| Tuberculosis Culture | Negative |
| Myobacterium PCR | Not Detected |
| Legionella DNA | Not Detected |
| Histoplasmosis AB | Negative |
| Right Upper Lobe Biopsy | Inflammatory Cells, No microorganisms |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





