Submitted:
15 February 2026
Posted:
27 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Three assumptions and Two Applications of Our 4G Model of Final Unification
- 1) There exists a characteristic electroweak fermion of rest energy, . It can be considered as the zygote of all elementary particles.
- 2) There exists a nuclear elementary charge in such a way that, = Strong coupling constant and .
- 3) Each atomic interaction is associated with a characteristic large gravitational coupling constant. Their fitted magnitudes are,

3. Black Hole Radius Formula in the 4G Model Context
4. Overview of Atomic Radii
- 1) Number of Electron Shells: Additional electron shells increase the atomic size by placing electrons further from the nucleus.
- 2) Effective Nuclear Charge (Zeff): A greater nuclear charge pulls electrons closer, decreasing atomic radius. Increased shielding by inner electrons allows the radius to expand.
- 3) Bonding and Coordination: Whether atoms are isolated, bonded, or in a crystal lattice influences their measured radii.
- 4) Ionic Charge: Cations (positive ions) have smaller radii than their neutral atoms; anions (negative ions) are larger due to changes in electron repulsion and attraction.
- 5)Spin State and Coordination Number: The ionic radius is affected by electron spin states and how many adjacent atoms (coordination number) are present in a crystal structure.
- 6) Types of Atomic Radii
| Type | Definition of various atomic radii |
| Covalent Radius |
Half the bond length between two identical covalently bonded atoms; common in molecules |
| Ionic Radius | The size of an ion in a crystal lattice; varies by charge and coordination number. |
| Metallic Radius | Half the distance between nuclei of adjacent atoms in a metallic lattice. |
| Van der Waals Radius | Half the minimum distance between non-bonded atoms, often used for noble gases |
| Bohr Radius |
Physical constant representing the ground-state average distance of an electron from the nucleus in hydrogen atom (53pm) |
- 1) X-ray Crystallography: Measures distances between nuclei in crystals to determine atomic and ionic radii.
- 2) Electron Diffraction: Uses electron scattering patterns from molecules for bond length and radius estimation.
- 3) Spectroscopic Techniques: Analyse atomic spectra to deduce electron cloud extents and radii.
- 4) Theoretical Calculations: Quantum mechanical models, such as the Heisenberg Uncertainty Principle, estimate the probability boundary for the outer electrons.
5. Generalized Scaling Law for Atomic Radii
- a) Without the correction factor f(QC), for Z=1 to 118, atomic radii seem to have a range of 33 pm to 227 pm.
- b) For hydrogen atom, modern theoretical values use statistical averages from vast crystal structure data, confirming the value of (31 to 37) pm with slight variation due to chemical environments. Estimated value is twice of the actual value.
- c) Atoms as composite entities formed by nuclear and electromagnetic forces unified in a gravitational analogue framework,
- d) Electronic shell filling modulates atomic radii by altering effective nuclear charge and electron cloud distribution.
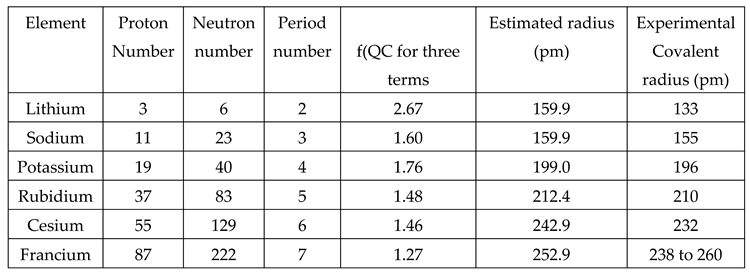
- e) Starting from the second period, estimated radii of the first element of the period is in good agreement with the experimental covalent radii. See the following Figure 1 and Table 4.
- f) Variations and anomalies in periodic atomic radii trends arise naturally from f(QC).
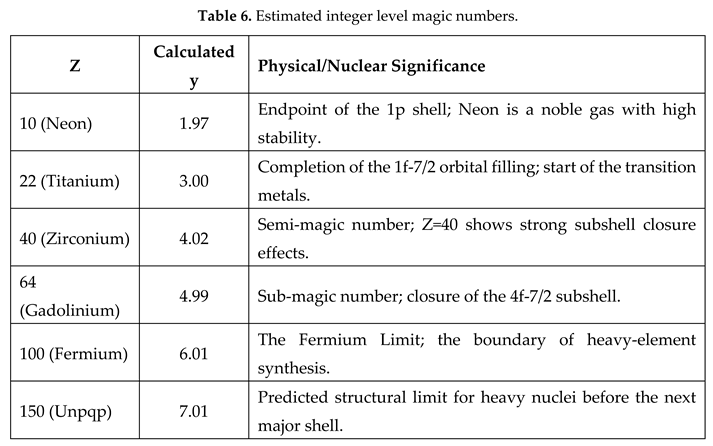
- g) Considering the stable mass number of the first element of the period and by introducing a fourth correction factor , it seems possible to give an approximate varying trend of the radii of that period.
6. Implications and Future Directions
7. String Theory Landscape Challenge
8. Are Atoms Quantum Gravitational Compact Objects?
- 1)
- Nuclear magic numbers (empirically confirmed: 28, 50, 82, 114)
- 2)
- Atomic shell architecture (universal 7-shell limit)
- 3)
- Nuclear viability threshold (Z²/A < 50)
- 4)
- Visible spectrum progression (Red 700nm → Violet 400 nm)
- 5)
- String theory compactification [40] (Calabi-Yau h{1,1}=7)
9. Dual Discreteness Formalism
9.1. Mass Channel (Gravitational)
9.2. Charge Channel (Electromagnetic)
9.3. Vector Synthesis
10. Fixed 7-Shell Atomic Template
10.1. Master Formula
- 1)
- Z ∝ n_charge: Linear shell-driving from proton number
- 2)
- √A ∝ √n_mass: Quadratic mass tempering via nuclear density
- 3)
- 7-limit: Relativistic/QED breakdown prevents higher stability
11. Nuclear Stability Constraints
11.1. Fission Barrier (Z²/A < 50)
- 1)
- Stable: Fe-56 (12.0), O-16 (4.0)
- 2)
- Marginal: U-235 (36.0), Pu-239 (38.5)
- 3)
- Fissionable: Cf-252 (51.0), Fm-256 (53.5)
11.2. Island of Stability
11.3. Unified Nuclear Density Framework
11.4. Half-Integer Stability
11.5. Harmonic Resonance in Nuclear Stability
12. The Seven Lights: Rainbow as Nuclear Fingerprint
- Shell 1.5 → 2.5 (Red → Orange): nm
- Shell 2.5 → 3.5 (Orange → Yellow): nm
- Shell 3.5 → 4.5 (Yellow → Green): nm (matches 530 nm Tin green peak)
13. String Theory Embedding
13.1. Type IIB Framework
- 1)
- Electron gravity: Ge-coupled mode (high tension)
- 2)
- Nucleon gravity: Gn-coupled mode (low tension)
- 3)
- Composite proton: n-wound string vortex with Gn ∝ n
- ➢ GeGn ~ 1086 GN², exactly the hierarchy string theory needs.
- 1)
- Shells: K=1, L=2, M=3, N=4, O=5, P=6, Q=7
- 2)
- CY moduli: t_1, t_2, t_3, t_4, t_5, t_6, t_7
- 3)
- Shell filling: Z/√A = n_D3/√(nwinding) as D3-brane charge/winding ratio
- 1)
- Z²/A < 50 → stable string vacuum
- 2)
- Z²/A > 50 → tachyonic instability (fission)
- 3)
- Z/√A ≤ 7 → CY moduli stabilization
14. Atomic and Nuclear Quantum Index (ANQI)
15. Weak Interaction Based Nuclear Stability and Transition from Integer to Half Integer
- Integer Phase (): Symmetric nucleon pairing dominates light nuclei stability, akin to compact object cohesion in 4G’s large gravitational constants.
- Half-Integer Phase (): The term—a tuned weak-Coulomb hybrid from 4G binding formulae—induces shell expansion, mimicking field "saturation" and reducing per-nucleon binding for .
16. Strong and Electroweak Mass Formula for Nuclear Binding Energy
17. Conclusions
Data availability statement
Acknowledgments
Conflicts of Interest
References
- Rutherford, E. The scattering of α and β particles by matter and the structure of the atom. Philosophical Magazine 1911, 21(125), 669–688. [Google Scholar] [CrossRef]
- Slater, C. Atomic Radii in Crystals. The Journal of Chemical Physics 1964, 41(10), 3199–3204. [Google Scholar] [CrossRef]
- Bondi. van der Waals Volumes and Radii. The Journal of Physical Chemistry 1964, 68(3), 441–451. [Google Scholar] [CrossRef]
- Clementi; Raimondi, D.L.; Reinhardt, W.P. Atomic Screening Constants from SCF Functions. II. Atoms with 37 to 86 Electrons. The Journal of Chemical Physics 1967, 47(4), 1300–1307. [Google Scholar] [CrossRef]
- Ghosh, D. C.; Biswas, R. Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 1. The Atomic Radii. Int. J. Mol. Sci. 2002, 3, 87–113. [Google Scholar] [CrossRef]
- Ganguly, P. Atomic sizes and atomic properties. J. Phys. B At. Mol. Opt. Phys. 2008, 41(10), 105002. [Google Scholar] [CrossRef]
- Mantina, Manjeera; Chamberlin, Adam C.; Valero, Rosendo; Cramer, Christopher J.; Truhlar, Donald G. Consistent van der Waals Radii for the Whole Main Group. The Journal of Physical Chemistry A. American Chemical Society (ACS) 2009, 113(19), 5806–5812. [Google Scholar] [CrossRef]
- Rahm, Martin; Hoffmann, Roald; Ashcroft, N. W. Atomic and Ionic Radii of Elements. 1–96. Chemistry (Weinheim an der Bergstrasse, Germany) 2016, 22(41), 14625–14632. [Google Scholar] [CrossRef]
- Yadav, P.; Tandon, H.; Malik, B.; et al. A quest for the universal atomic radii. Struct. Chem. 2022, 33, 389–394. [Google Scholar] [CrossRef]
- Owolabi, T.O.; Akande, K.O.; Olatunji, S.O. Estimation Of The Atomic Radii Of Periodic Elements Using Support Vector Machine. International Journal of Advanced Information Science and Technology 2014, (8), 105–113. [Google Scholar]
- Griffiths, D. J. Introduction to Quantum Mechanics, 2nd ed.; Pearson Prentice Hall, 2005. [Google Scholar]
- Pyykkö, P. Relativistic effects in chemistry: more common than you thought. Annual Review of Physical Chemistry 2008, 63, 45–64. [Google Scholar] [CrossRef]
- Gunavardhana Naidu, Seshavatharam U. V. S; Lakshminarayana, T.S. Nuclear evidences for confirming the physical existence of 585 GeV weak fermion and galactic observations of TeV radiation. International Journal of Advanced Astronomy 2025, 13(1), 1–17. [Google Scholar]
- Seshavatharam, U.V.S.; Gunavardhana Naidu, T.; Lakshminarayana, S. To confirm the existence of heavy weak fermion of rest energy 585 GeV. AIP Conf. Proc. 2022, 2451, 020003. [Google Scholar]
- Seshavatharam, U V S; Lakshminarayana, S. 4G model of final unification – A brief report Journal of Physics: Conference Series. 2022; 2197. [Google Scholar]
- Seshavatharam U. V. S, Gunavardhana Naidu T and Lakshminarayana S. 4G Model of Heavy Electroweak Charged 585 GeV Fermions as the Supposed Microscopic Origin of the 1.17 TeV All-Electron Spectral Break. International Journal of Advance Research and Innovative Ideas in Education 2025, 11(6), 2116–2140.
- Seshavatharam, U.V.S; Lakshminarayana, S. Inferring and confirming the rest mass of electron neutrino with neutron life time and strong coupling constant via 4G model of final unification. World Scientific News 2024, 191, 127–156. [Google Scholar]
- Romero, Gustavo E. Introduction to black holes. arXiv 2008, arXiv:0805.2082. [Google Scholar] [CrossRef]
- Lupsasca, Alexandru; Mayerson, Daniel R.; Ripperda, Bart; Staelens, Seppe. A Beginner’s Guide to Black Hole Imaging and Associated Tests of General Relativity. arXiv 2024, arXiv:2402.01290v1. [Google Scholar]
- Seshavatharam, U.V.S; Gunavardhana, T. N.; Lakshminarayana, S. Avogadro’s Number: History, Scientific Role, State-of-the-Art, and Frontier Computational Perspectives. Curr. Trends. Mass. Comm Preprints2025, 2025080338. 2025, 4(3), 01–10. [Google Scholar] [CrossRef]
- Seshavatharam, U.V.S; Gunavardhana, T. N.; Lakshminarayana, S. Quarks-Higgs Resonances in the 4G Model of Final Unification: Precision Mass Predictions and Observational Targets. Zenodo 2026. [Google Scholar] [CrossRef]
- d’Enterria, D; et al. The strong coupling constant: state of the art and the decade ahead. J. Phys. G: Nucl. Part. Phys. 2024, 51 090501. [Google Scholar] [CrossRef]
- Seshavatharam, U.V.S.; Lakshminarayana, S. Understanding the Origins of Quark Charges, Quantum of Magnetic Flux, Planck’s Radiation Constant and Celestial Magnetic Moments with the 4G Model of Nuclear Charge. Current Physics 2024, 1(e090524229812), 122–147. [Google Scholar] [CrossRef]
- Patel, Apoorva D. EPR Paradox, Bell Inequalities and Peculiarities of Quantum Correlations. arXiv 2025, arXiv:2502.06791v1. [Google Scholar] [CrossRef]
- Cheung, Clifford; Hillman, Aaron; Remmen, Grant N. String Theory May Be Inevitable as a Unified Theory of Physics. In Physics World; 2025. [Google Scholar]
- Seshavatharam, U.V.S; Gunavardhana, T. N.; Lakshminarayana, S. Advancing String Theory with 4G Model of Final Unification. J. Phys.: Theor. Appl. 2025, 9(2), 158–197. [Google Scholar]
- Abokhalil, Ahmed. The Higgs Mechanism and Higgs Boson: Unveiling the Symmetry of the Universe. arXiv arXiv:2306.01019. [CrossRef]
- Pyykkö, P; Atsumi, M. Molecular single-bond covalent radii for elements 1-118. Chemistry 2009, 15(1), 186–97. [Google Scholar] [CrossRef]
- CRC Handbook of Chemistry and Physics, 97th ed.; Haynes, W. M., Ed.; CRC Press, 2016. [Google Scholar]
- Susskind, Leonard. The Anthropic Landscape of String Theory. arXiv 2003, arXiv:hep-th/0302219. [Google Scholar] [CrossRef]
- Mukhi, Sunil. String theory: a perspective over the last 25 years. Class. Quantum Grav. 2011, 28 153001. [Google Scholar] [CrossRef]
- Sachdev, Subir. Strange and stringy. Scientific American 2013, 308(44), 44–51. [Google Scholar] [CrossRef]
- Blumenhagen, R.; Lüst, D.; Theisen, S. Basic Concepts of String Theory. In Theoretical and Mathematical Physics Springer Heidelberg, Germany; 2013. [Google Scholar]
- Saha, Arnab Priya; Phys, Aninda Sinha. Field Theory Expansions of String Theory Amplitudes. Rev. Lett. 132, 221601,2024. [CrossRef] [PubMed]
- Seshavatharam, U.V.S.; Lakshminarayana, S. On the compactification and reformation of string theory with three large atomic gravitational constants. International Journal of Physical Research 2021, 9(1), 42–48. [Google Scholar] [CrossRef]
- Barley, Kamal K.; Ruffing, Andreas; Suslov, Sergei K. Old Quantum Mechanics by Bohr and Sommerfeld from a Modern Perspective. arXiv 2025, arXiv:2506.00408. [Google Scholar] [CrossRef]
- Sticker, Harry. The Fine-Structure Constant as a Scaled Quantity. arXiv 2025, arXiv:2512.07027. [Google Scholar] [CrossRef]
- Brown, L.M. Paul A.M. Dirac’s The Principles of Quantum Mechanics. Perspectives in Physics 2006, 8, 381–407. [Google Scholar] [CrossRef]
- Baily, Charles. Early Atomic Models - From Mechanical to Quantum (1904-1913). European Physical Journal H 2013, 38, 1–38. [Google Scholar] [CrossRef]
- Green, MB; Schwarz, JH; Witten, E. Superstring Theory: 25th Anniversary Edition; Cambridge University Press, 2012. [Google Scholar]
- Boya, Luis J. The Thermal Radiation Formula of Planck (1900). Rev. Academia de Ciencias, Zaragoza. 2003, 58, 91–114. [Google Scholar]
- Kaplan, I.G. Pauli Exclusion Principle and its theoretical foundation. arXiv 2019, arXiv:1902.00499. [Google Scholar] [CrossRef]
- Neergård, K. Pairing Theory of the Wigner Cusp. arXiv 2017, arXiv:1707.01474. [Google Scholar] [CrossRef]
- Van Isacker, P.; Heyde, K. Exactly solvable models of nuclei. arXiv 2014, arXiv:1401.7512v1. [Google Scholar] [CrossRef]
- Chowdhury, P.R.; Samanta, C.; Basu, D.N. Modified Bethe– Weizsacker mass formula with isotonic shift and new driplines. Mod. Phys. Lett. A 2005, 20, 1605–1618. [Google Scholar] [CrossRef]
- Myers, W.D.; Swiatecki, W.J. Nuclear masses and deformations. Nuclear Physics A 1966, 81(1), 1–60. [Google Scholar] [CrossRef]
- Tran, D.T.; Ong, H.J.; Hagen, G.; et al. Evidence for prevalent Z = 6 magic number in neutron-rich carbon isotopes. Nat Commun. 2018, 9, 1594. [Google Scholar] [CrossRef]
- Xing, Y. M. =14 Magicity Revealed by the Mass of the Proton Dripline Nucleus 22Si. Phys. Rev. Lett. 135, 012501. [CrossRef]
- Pan, C; Wu, X-H. Examination of Possible Proton Magic Number Z = 126 with the Deformed Relativistic Hartree-Bogoliubov Theory in Continuum. Particles 2025, 8(1), 2. [Google Scholar] [CrossRef]
- Mehana, P.; Rajeswari, N. S. Occurrence of new magic number at Z, N = 14, 16. In Proceedings of the DAE Symp. on Nucl. Phys., 2022; 66. [Google Scholar]
- Togano, Y.; et al. Hindered proton collectivity in 28S: Possible magic number at Z=16. Phys. Rev. Lett. 2012, 108, 222501. [Google Scholar] [CrossRef]
- Kanungo, Rituparna; Tanihata, Isao; Ozawa, Akira. Observation of new proton and neutron magic numbers. AIP Conf. Proc. 2002, 610, 741–745. [Google Scholar] [CrossRef]
- Sliney, DH. What is light? The visible spectrum and beyond. Eye (Lond) 2016, 30(2), 222–9. [Google Scholar] [CrossRef] [PubMed]
- Frauendorf, S.; Macchiavelli, A.O. Overview of neutron–proton pairing. Progress in Particle and Nuclear Physics 2014, 8, 24–90. [Google Scholar] [CrossRef]
- Simkovic, F.; Moustakidis, Ch.C.; Pacearescu, L.; Faessler, Amand. Proton-neutron pairing in the deformed BCS approach. Phys. Rev. C. 2023, 68, 054319. [Google Scholar] [CrossRef]
- Seshavatharam, U. V. S.; Lakshminarayana, S. A Unified 6-Term Formula for Nuclear Binding Energy with a Single Set of Energy Coefficients for Z = 1–140. International Journal of Advance Research and Innovative Ideas in Education 2025, 2(6), 1716–1731. [Google Scholar]
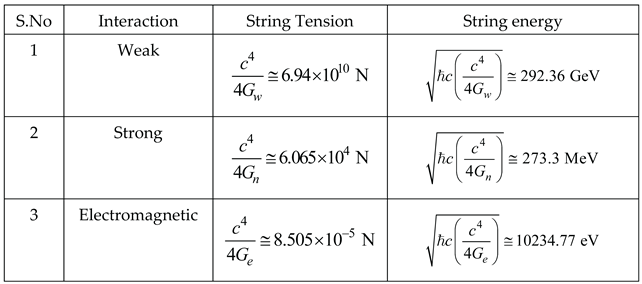
| S.No | Interaction | String Tension | String energy |
|
1 |
Weak |
||
|
2 |
Strong |
||
|
3 |
Electromagnetic |
 |
 |
| Z (Magic) | Element |
Mass number (A) |
Z/√A | Deviation from Integer | Deviation from Half-Integer | Regime | Quantized Shell Index |
| 2 | Helium | 4 | 1.00 | 0.00 (Perfect 1.0) | 0.50 | Integer | 1 |
| 8 | Oxygen | 16 | 2.00 | 0.00 (Perfect 2.0) | 0.50 | Integer | 2 |
| 20 | Calcium | 40 | 3.16 | +0.16 (Near 3.0) | -0.34 | Integer | 3 |
| 28 | Nickel | 58 | 3.67 | +0.67 | +0.17 (Near 3.5) | Half-Integer | 3.5 |
| 50 | Tin | 120 | 4.58 | +0.58 | +0.08 (Near 4.5) | Half-Integer | 4.5 |
| 82 | Lead | 208 | 5.69 | +0.69 | +0.19 (Near 5.5) | Half-Integer | 5.5 |
| 114 | Flerovium | 298 | 6.60 | +0.60 | +0.10 (Near 6.5) | Half-Integer | 6.5 |
| Target y |
Estimated Magic Z |
Z ± 4 | Notes History |
| 1.5 | 6 | 2 – 10 | Light shell; exact fit for Z=8 (Oxygen) within the stability margin. |
| 2.5 | 16 | 12 – 20 | Transition zone; captures Z=14 (Silicon) and the major closure at Z=20 (Calcium). |
| 3.5 | 30 | 26 – 34 | Iron-peak region; covers the major magic number Z=28 (Nickel). |
| 4.5 | 50 | 46 – 54 | Exact fit for Tin (Z=50); identifies the maximum number of stable isotopes in the periodic table. |
| 5.5 | 80 | 76 – 84 | Heavy closure zone; encompasses Z=82 (Lead), the heaviest stable magic nucleus. |
| 6.5 | 122 | 118 – 126 | Island of Stability; sits between the y=6.0 anchor (Z=100) and theoretical Z=126 resonances. |
| 7.5 | 184 | 180 – 188 | Superheavy limit; aligns with major theoretical neutron and proton shell closures. |
| Shell Transition | s = ceil(Z/√A)−½ | Magic Z | Element | Mass number | Colour | λ (nm) |
| K→L (1→2) | 1.5 | 8 | Oxygen | 16 | Red | 700 |
| L→M (2→3) | 2.5 | 20 | Calcium | 48 | Orange | 620 |
| M→N (3→4) | 3.5 | 28 | Nickel | 58 | Yellow | 580 |
| N→O (4→5) | 4.5 | 50 | Tin | 120 | Green | 530 |
| O→P (5→6) | 5.5 | 82 | Lead | 208 | Blue | 470 |
| P→Q (6→7) | 6.5 | 114 | Flerovium | 298 | Indigo | 420 |
| Q→∞ (7→∞) | 7.0 | — | (Vacuum) | Violet | 400 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




