Submitted:
20 January 2026
Posted:
23 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Data Collection and Management
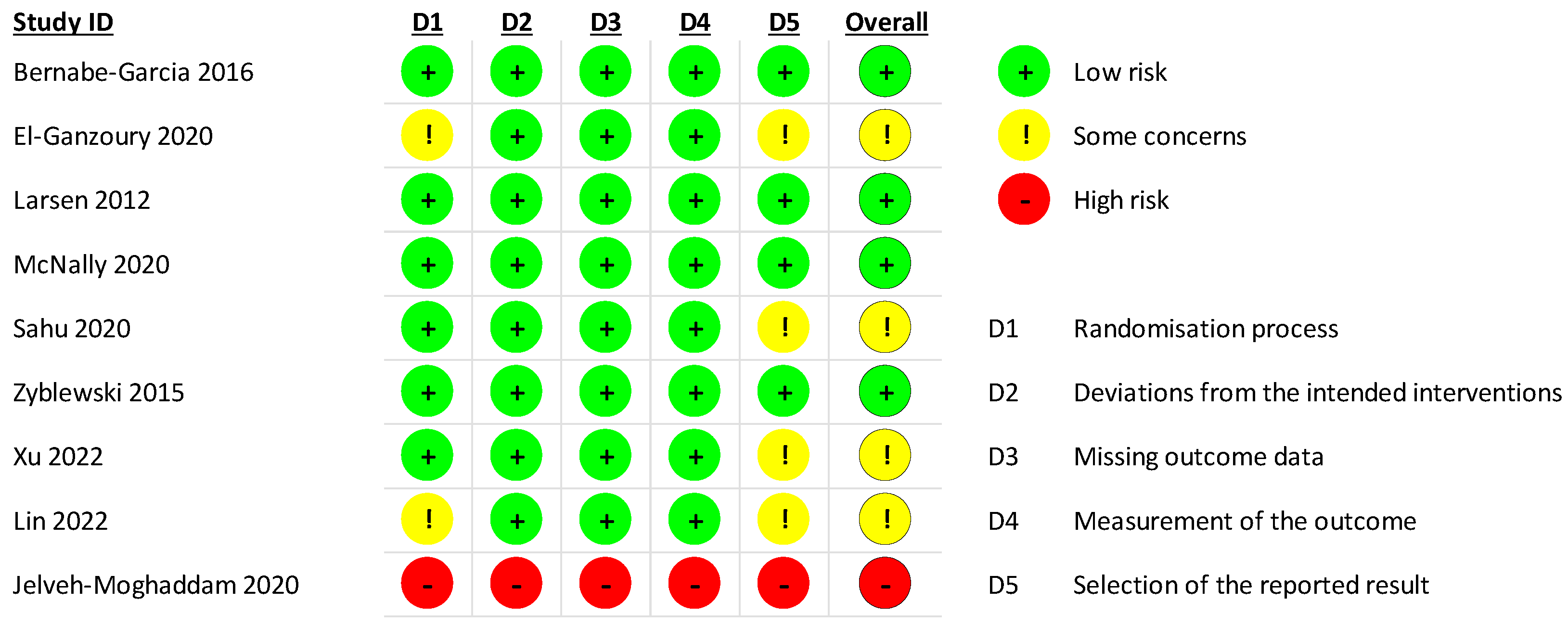
2.4. Risk of Bias Assessment
2.5. Data Analysis
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.2.1. Interventional Studies
3.2.2. Observational Studies
3.3. Risk of Bias
3.4. Effects of Any Modified Fatty Acids Emulsion
3.4.1. Enteral DHA in Sunflower Oil vs. Sunflower Oil Only
3.4.1.1. Efficacy
3.4.1.2. Safety
- postoperative sepsis and severe sepsis, in both the intention-to-treat (ITT; RR=0.67, 95%CI, 0.34 to 1.30; and RR=0.89. 95% CI, 0.51 to 1.56, respectively; n=55) and per-protocol (PP; RR=0.38, 95% CI, 0.12 to 1.15; RR=0.28, 95% CI, 0.03 to 2.26, respectively; n=34) analyses;
- any postoperative organ dysfunction, including: respiratory (RR=0.19; 95% CI, 0.03 to 1.4), cardiovascular (RR=0.28; 95% CI, 0.03 to 2.26), hematological (RR=0.16; 95% CI, 0.01 to 2.87), hepatic (RR=0.37; 95% CI, 0.02 to 8.55), and renal dysfunctions (RR=0.16; 95% CI, 0.01 to 2.87);
- vomiting events in the ICU (RR=1.13, 95% CI, 0.18 to 7.09; n=34);
- mortality in either the ITT analysis (RR=0.21; 0.03 to 1.66; n=55) or in the PP analysis (RR=0.37; 0.02 to 8.55; n=34); none of the deaths were related to DHA administration (21).
3.4.2. Intravenous 50% MCT and 40% LCT and 10% of Fish Oil Emulsion (MCT/LCT/fish oil) vs. Fully LCT Emulsion
3.4.2.1. Efficacy
- mean duration of the ICU stay (MD = -7.4 days, 95% CI, -10.86 to -3.94);
- mean duration of mechanical ventilation (MD=-2.1 days, 95% CI, -2.86 to -1.34);
- and mean length of hospital stay (MD=-5.1 days, 95% CI, -8.77 to -1.43) (22).
3.4.2.2. Safety
3.4.3. High-Dose Cholecalciferol vs Usual Care/No Intervention
3.4.3.1. Efficacy
3.4.3.2. Safety
3.4.3.3. Compliance
3.4.4. Effect of human milk fortifier vs. placebo in breastfed infants
3.4.4.1. Efficacy
3.4.4.2. Safety
3.4.5. Effects of Preoperative Nutrition-Based Protocols
3.4.5.1. Efficacy of Preoperative Trophic Breast Milk Feeds
3.4.5.1.1. Efficacy
3.4.5.1.2. Safety
3.4.5.2. A 2-week vs. 1-week preoperative nutrition support
3.4.5.2.1. Efficacy of the 2-week vs. 1-week preoperative nutrition support
- a shorter mean length of ICU stay (MD=36.5 hours, 95% CI, -44.61 to -28.39) (16);
- a shorter mean hospital length of stay (MD=-40.9 days, 95%CI, -65.26 to -16.54) (16);
- a shorter mean duration of postoperative mechanical ventilation was found (MD=-14.0 hours, 95% CI, -17.95 to -10.05) (16);
- and higher mean postoperative feeding volume intake, measured before discharge (MD = 7.53 ml/feed, 95%CI, 0.99 to 14.07) (16).
- other feeding-related outcomes, including: day of enteral feeding initiation (RR = 1.33, 95% CI, 0.88 to 2.03, and RR=1.00, 95% CI, 0.34 to 2.93, for day 1 and 2 respectively), route of feeding (oral: RR = 1.42, 95% CI, 0.95 to 2.12, vs oral and nasogastric tube: RR = 0.38, 95% CI, 0.12 to 1.21), and feeding frequency (every 2 hours: RR = 1.19, 95% CI, 0.93 to 1.51, vs every 3 hours: RR = 0.25, 95% CI, 0.03 to 2.05) (16);
- and the proportion of children with successful extubation (RR=1.05, 95% CI, 0.92 to 1.2), early extubation (≤48 h) (RR=1.2, 95% CI, 0.9 to 1.61), late extubation (>48 h) (RR=0.33, 95% CI, 0.04 to 2.94), and reintubation (RR=0.5, 95%CI, 0.05 to 5.08) (16).
3.4.5.2.2. Safety of the 2-week vs 1-week preoperative nutrition support
3.4.5.3. Efficacy of a 1-month preoperative nutrition support vs no support
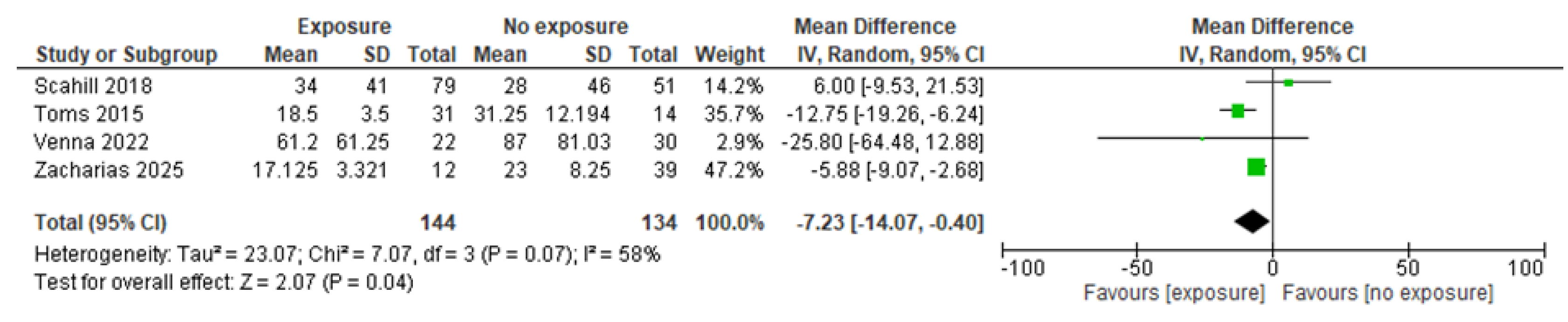
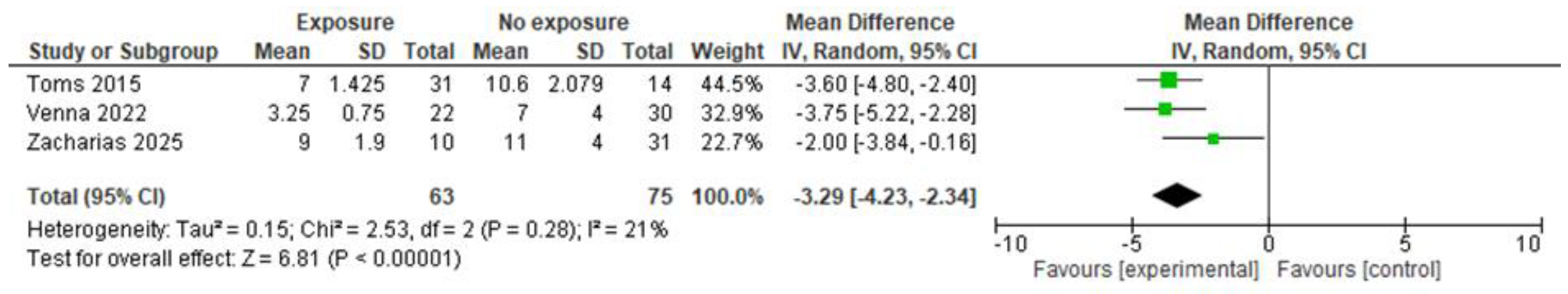
3.4.5.4. Effects of any preoperative feeding vs. no feeding
3.4.5.4.1. Effectiveness
3.4.5.4.2. Safety
3.4.5.5. Effectiveness of Meeting Versus Not Meeting Center-Recommended Preoperative Caloric Intake Requirements (Cohort Study)
3.4.5.6. Preoperative Fortified vs. Unfortified Nutrition
3.4.5.6.1. Effectiveness
3.4.5.6.2. Safety
3.4.6. Preoperative Exclusive Human Milk and Direct Breastfeeding
3.5. Quality of Evidence
4. Discussion
4.1. Summary
4.2. Comparison with Other Systematic Reviews
4.3. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BDG | Bidirectional Glenn |
| BMI | Body mass index |
| CHD | Congenital heart disease |
| CI | Confidence interval |
| CS-AKI | Cardiac surgery–associated acute kidney injury |
| DHA | Docosahexaenoic acid |
| ERAS | Enhanced Recovery After Surgery |
| GER | Gastroesophageal reflux |
| GRADE | Grading of Recommendations, Assessment, Development, and Evaluation |
| HMF | Human milk fortifier |
| HR | Hazard ratio |
| ICU | Intensive care unit |
| ITT | Intention-to-treat |
| IV | Intravenous |
| LCT | Long-chain triglycerides |
| MD | Mean difference |
| MCT | Medium-chain triglycerides |
| MV | Mechanical ventilation |
| n | Number of participants |
| ND | No difference |
| NEC | Necrotizing enterocolitis |
| NeoC3 | Neonatal Cardiac Care Collaborative |
| NOS | Newcastle–Ottawa Scale |
| OR | Odds ratio |
| PP | Per-protocol |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| RCT | Randomized controlled trial |
| RoB 2 | Revised Cochrane risk-of-bias tool for randomized trials |
| RR | Risk ratio |
| SD | Standard deviation |
| STRONG | Screening Tool for Risk on Nutritional Status and Growth |
References
- Diao J, Chen L, Wei J, Shu J, Li Y, Li J, et al. Prevalence of Malnutrition in Children with Congenital Heart Disease: A Systematic Review and Meta-Analysis. The Journal of pediatrics. 2022;242:39-47.e4.
- Abbas Q, Ali H, Ahuja AK, Bhatti OA, Ladak S, Khan I, et al. Preoperative nutrition status in children with congenital heart disease and its impact on postoperative outcomes: a systematic review and meta-analysis. Sci Rep. 2025;15(1):25738.
- Ljungqvist O, Scott M, Fearon KC. Enhanced Recovery After Surgery: A Review. JAMA Surg. 2017;152(3):292-8.
- Lau CS, Chamberlain RS. Enhanced Recovery After Surgery Programs Improve Patient Outcomes and Recovery: A Meta-analysis. World J Surg. 2017;41(4):899-913.
- Hidayah BA, Toh ZA, Cheng LJ, Syahzarin BD, Zhu Y, Pölkki T, et al. Enhanced recovery after surgery in children undergoing abdominal surgery: meta-analysis. BJS Open. 2023;7(1).
- Anesthesiology 2017, 126(3), 376–93.
- Frykholm P, Disma N, Andersson H, Beck C, Bouvet L, Cercueil E, et al. Pre-operative fasting in children: A guideline from the European Society of Anaesthesiology and Intensive Care. Eur J Anaesthesiol. 2022;39(1):4-25.
- Mehta NM, Skillman HE, Irving SY, Coss-Bu JA, Vermilyea S, Farrington EA, et al. Guidelines for the Provision and Assessment of Nutrition Support Therapy in the Pediatric Critically Ill Patient: Society of Critical Care Medicine and American Society for Parenteral and Enteral Nutrition. JPEN J Parenter Enteral Nutr. 2017;41(5):706-42.
- Kołodziej M, Skulimowska J. A Systematic Review of Clinical Practice Guidelines on the Management of Malnutrition in Children with Congenital Heart Disease. Nutrients. 2024;16(16).
- Raval MV, Brockel MA, Kolaček S, Simpson KE, Spoede E, Starr KNP, et al. Key Strategies for Optimizing Pediatric Perioperative Nutrition-Insight from a Multidisciplinary Expert Panel. Nutrients. 2023;15(5).
- Weimann A, Bezmarevic M, Braga M, Correia M, Funk-Debleds P, Gianotti L, et al. ESPEN guideline on clinical nutrition in surgery - Update 2025. Clin Nutr. 2025;53:222-61.
- Page MJ, Moher D, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ. 2021;372:n160.
- Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page M, et al. Cochrane Handbook for Systematic Reviews of Interventions version 6.4 (updated August 2023). Cochrane, 2023. Available from www.training.cochrane.org/handbook. 2023.
- Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. Bmj. 2019;366:l4898.
- Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Medical Research Methodology. 2005;5(1):13.
- El-Ganzoury MM, El-Farrash RA, Ahmed GF, Hassan SI, Barakat NM. Perioperative nutritional prehabilitation in malnourished children with congenital heart disease: a randomized controlled trial. Nutrition (Burbank, Los Angeles County, Calif). 2021;84:111027.
- Lin YF, Lin WH, Lin SH, Zhang QL, Chen Q, Zheng YR. Using Human Milk Fortifiers to Improve the Preoperative Nutritional Status of Infants With Non-restricted Ventricular Septal Defect. Frontiers in Pediatrics. 2022;10.
- Zyblewski SC, Nietert PJ, Graham EM, Taylor SN, Atz AM, Wagner CL. Randomized Clinical Trial of Preoperative Feeding to Evaluate Intestinal Barrier Function in Neonates Requiring Cardiac Surgery. Journal of pediatrics. 2015;167(1):47-51.e1.
- Xu LP, Lin SH, Zhang QL, Zheng Y, Lin G. Short-Term Nutritional Support Improves The Preoperative Nutritional Status of Infants With Non-Restrictive Ventricular Septal Defect: A Prospective Controlled Study. Heart Surg Forum. 2022;25(5):E745-e9.
- In GRADE handbook for grading quality of evidence and strength of recommendations; Schünemann H, Brożek J, Guyatt G, Oxman A, editors. GRADE handbook for grading quality of evidence and strength of recommendations. Updated October 2013. The GRADE Working Group, 2013. Available from guidelinedevelopment.org/handbook.
- Bernabe-Garcia M, Lopez-Alarcon M, Villegas-Silva R, Mancilla-Ramirez J, Rodriguez-Cruz M, Maldonado-Hernandez J, et al. Beneficial Effects of Enteral Docosahexaenoic Acid on the Markers of Inflammation and Clinical Outcomes of Neonates Undergoing Cardiovascular Surgery: an Intervention Study. Annals of nutrition & metabolism. 2016;69(1):15-23.
- Larsen BM, Goonewardene LA, Joffe AR, Van Aerde JE, Field CJ, Olstad DL, et al. Pre-treatment with an intravenous lipid emulsion containing fish oil (eicosapentaenoic and docosahexaenoic acid) decreases inflammatory markers after open-heart surgery in infants: a randomized, controlled trial. Clinical nutrition (Edinburgh, Scotland). 2012;31(3):322-9.
- McNally JD, O’Hearn K, Fergusson DA, Lougheed J, Doherty DR, Maharajh G, et al. Prevention of post-cardiac surgery vitamin D deficiency in children with congenital heart disease: a pilot feasibility dose evaluation randomized controlled trial. Pilot and Feasibility Studies. 2020;6(1).
- Jelveh-Moghaddam H, Fani K, Hekmat M, Azari AA. The effects of Vitamin D3 in pediatric patients undergoing congenital heart surgery. Journal of cellular and molecular anesthesia. 2020;5(2):66-73.
- Sahu MK, Bipin C, Niraghatam HV, Karanjkar A, Singh SP, Rajashekar P, et al. Vitamin D Deficiency and Its Response to Supplementation as Stoss Therapy in Children with Cyanotic Congenital Heart Disease Undergoing Open Heart Surgery. Journal of Cardiac Critical Care. 2019;3(1):17-23.
- Bertrandt RA, Gist K, Hasson D, Zang H, Reichle G, Krawczeski C, et al. Cardiac Surgery-Associated Acute Kidney Injury in Neonates Undergoing the Norwood Operation: Retrospective Analysis of the Multicenter Neonatal and Pediatric Heart and Renal Outcomes Network Dataset, 2015-2018. Pediatr Crit Care Med. 2024;25(5):e246-e57.
- Dabbagh A, Miller S, McCulloch M, Rosenthal G, Conaway M, White S. Preoperative Oral Feeding in Infants with Congenital Heart Disease Within the First Month of Life is Associated with a Higher Likelihood of Freedom From Tube Feeding at Time of Postoperative Discharge. Pediatric cardiology. 2025.
- Elgersma KM, Wolfson J, Fulkerson JA, Georgieff MK, Looman WS, Spatz DL, et al. Human Milk Feeding and Direct Breastfeeding Improve Outcomes for Infants With Single Ventricle Congenital Heart Disease: Propensity Score-Matched Analysis of the NPC-QIC Registry. Journal of the American Heart Association. 2023;12(17):e030756.
- Menon SC, McCandless RT, Mack GK, Lambert LM, McFadden M, Williams RV, et al. Clinical outcomes and resource use for infants with hypoplastic left heart syndrome during bidirectional Glenn: summary from the Joint Council for Congenital Heart Disease National Pediatric Cardiology Quality Improvement Collaborative registry. Pediatr Cardiol. 2013;34(1):143-8.
- Murray SE, Zimmerman D, Patel SS. Preoperative Feeding Fortification Among Infants with Congenital Heart Disease is Associated with Higher Growth Velocity in the First 30 Days Post-repair and Lower BMI Percentile for Age at 10 Years: A Retrospective Cohort Study. Pediatric cardiology. 2025;46(2):394-400.
- Scahill CJ, Graham EM, Atz AM, Bradley SM, Kavarana MN, Zyblewski SC. Preoperative Feeding Neonates With Cardiac Disease. World J Pediatr Congenit Heart Surg. 2017;8(1):62-8.
- Toms R, Jackson KW, Dabal RJ, Reebals CH, Alten JA. Preoperative trophic feeds in neonates with hypoplastic left heart syndrome. Congenit Heart Dis. 2015;10(1):36-42.
- Zacharias P, Blinci J, Shenoy R, Lee J, Singh Y. Impact of the Pre-Operative Standardized Nutritional Protocol in Infants with Congenital Heart Disease (CHD). J Cardiovasc Dev Dis. 2025;12(5).
- Venna A, Reid K, Davis S, Gai J, d’Udekem Y, Clauss S. Preoperative Feeding in Single Ventricle Neonates is Predictive of Shorter Time to Goal Feed. Congenital Heart Disease. 2022;17(5):505-18.
- Iliopoulos I, Burke R, Hannan R, Bolivar J, Cooper DS, Zafar F, et al. Preoperative Intubation and Lack of Enteral Nutrition are Associated with Prolonged Stay After Arterial Switch Operation. Pediatric cardiology. 2016;37(6):1078-84.
- Kataria-Hale J, Osborne SW, Hair A, Hagan J, Pammi M. Preoperative Feeds in Ductal-Dependent Cardiac Disease: A Systematic Review and Meta-analysis. Hosp Pediatr. 2019;9(12):998-1006.
- Bell D, Suna J, Marathe SP, Perumal G, Betts KS, Venugopal P, et al. Feeding Neonates and Infants Prior to Surgery for Congenital Heart Defects: Systematic Review and Meta-Analysis. Children (Basel). 2022;9(12).
- Mills KI, Kim JH, Fogg K, Goldshtrom N, Graham EM, Kataria-Hale J, et al. Nutritional Considerations for the Neonate With Congenital Heart Disease. Pediatrics. 2022;150(Suppl 2).
- Silva-Gburek J, May K, Walvoord B, Lozano C, Coss-Bu JA. Perioperative Nutrition in Pediatric Patients with Congenital Heart Disease and Heart Failure. Nutrients. 2025;17(22).
- Szentirmay LS, Heywood A, James-Nunez K, Trivedi A, Dalrymple H. Preoperative enteral nutrition in neonates with congenital heart disease: A review of literature. Journal of Neonatal Nursing. 2025;31(3):101667.
- Vandenbroucke JP, von Elm E, Altman DG, Gøtzsche PC, Mulrow CD, Pocock SJ, et al. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): explanation and elaboration. Int J Surg. 2014;12(12):1500-24.
- European Medicines Agency (EMA). Guideline on registry-based studies. EMA/426390/2021 (22 October 2021). .

 |
| Intervention/Exposure vs comparator |
Number of studies and participants (n) |
Efficacy/Effectiveness | Safety |
|---|---|---|---|
| Intravenous 50% MCT and 40% LCT and 10% of fish oil vs fully LCT emulsion |
1 RCT, n=32 | ↓ ICU stay ↓ hospital stay ↓ MV |
Sepsis – ND |
| Enteral DHA in sunflower oil vs sunflower oil only |
1 RCT, n=34 | ↓ ICU stay | Intraoperative bleeding – ND( Postoperative sepsis – ND Bleeding – ND Severe sepsis – ND Organ dysfunctions – ND Vomiting at ICU – ND Mortality – ND |
| High-dose vitamin D vs usual care/no intervention |
2 RCTs, n=101 | ICU stay – ND (2 RCTs, n=101) MV – ND (2 RCTs, n=101) ↓ hospital stay (1 RCT, n=41) ↑ MV (1 non-RCT, n=60) |
Adverse events – ND (2 RCT, n=101) Mortality ND (2 RCT, n=101) Need for inotropes ND (1 RCT, n=41) |
| Human milk fortifier vs placebo |
1 RCT, n=58 | ↑ Albumins and Prealbumins ↑ STRONG Kids score Hemoglobin ND |
Preoperative adverse events – ND No NEC No death |
| Preoperative trophic breast milk feeding vs no enteral feeding |
1 RCT, n=27 | Postoperative feeding intolerance ND Nasogastric tube dependence |
Postoperative NEC – ND GER medication – ND Mortality – ND |
| Preoperative nutrition support 2-weeks vs 1-week |
1 RCT, n=40 | ↓ ICU stay ↓ hospital stay ↓ MV ↑ postoperative feeding intake other feeding related outcomes – ND ↑ weight z-score pre- and postoperatively height z-score – inconsistent Albumins – ND Hemoglobulin – ND |
Sepsis – ND Successful extubation – ND Feeding-related adverse events ND |
| 1-month preoperative nutrition support vs no support |
1 RCT, n=80 | ↓ ICU and hospital stay ↓ preoperative STRONG Kids Score ↑ Albumins, Prealbumins and Hemoglobulin |
Not reported |
| Any preoperative feeding vs no feeding |
7 studies, n=917 | ICU stay – ND (3 studies, n=226) ↓ hospital stay (4 studies, n=278) ↓ days to achieve full feeds postoperatively (3 studies, n=138) MV duration – inconsistent results (2 studies, n=175) postoperative weight z-score – ND (2 studies, n=181) ↑ Albumins (1 study, n=45) ↑ children on full feeding (1 study, n=235) |
↓ CS-AKI (1 study, n=347) NEC – ND (3 studies, n=232), postoperative infection rate – ND (1 study, n=45) mortality risk – ND (2 studies, n=97) |
| Achieved vs not achieved center-recommended pre-operative caloric intake requirements |
1 study, n=160 | ↑ postoperative hospital stay ( | Not reported |
| Preoperative fortified nutrition (≥22 kcal/oz) vs unfortified nutrition (20 kcal/oz) |
1 study, n=84 | ↑ ICU stay ↑ hospital stay ↓ postoperative weight MV duration – ND BMI-for-age percentiles at 5 and 10 years |
Surgical site infection risk – ND |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




