Submitted:
19 January 2026
Posted:
21 January 2026
Read the latest preprint version here
Abstract
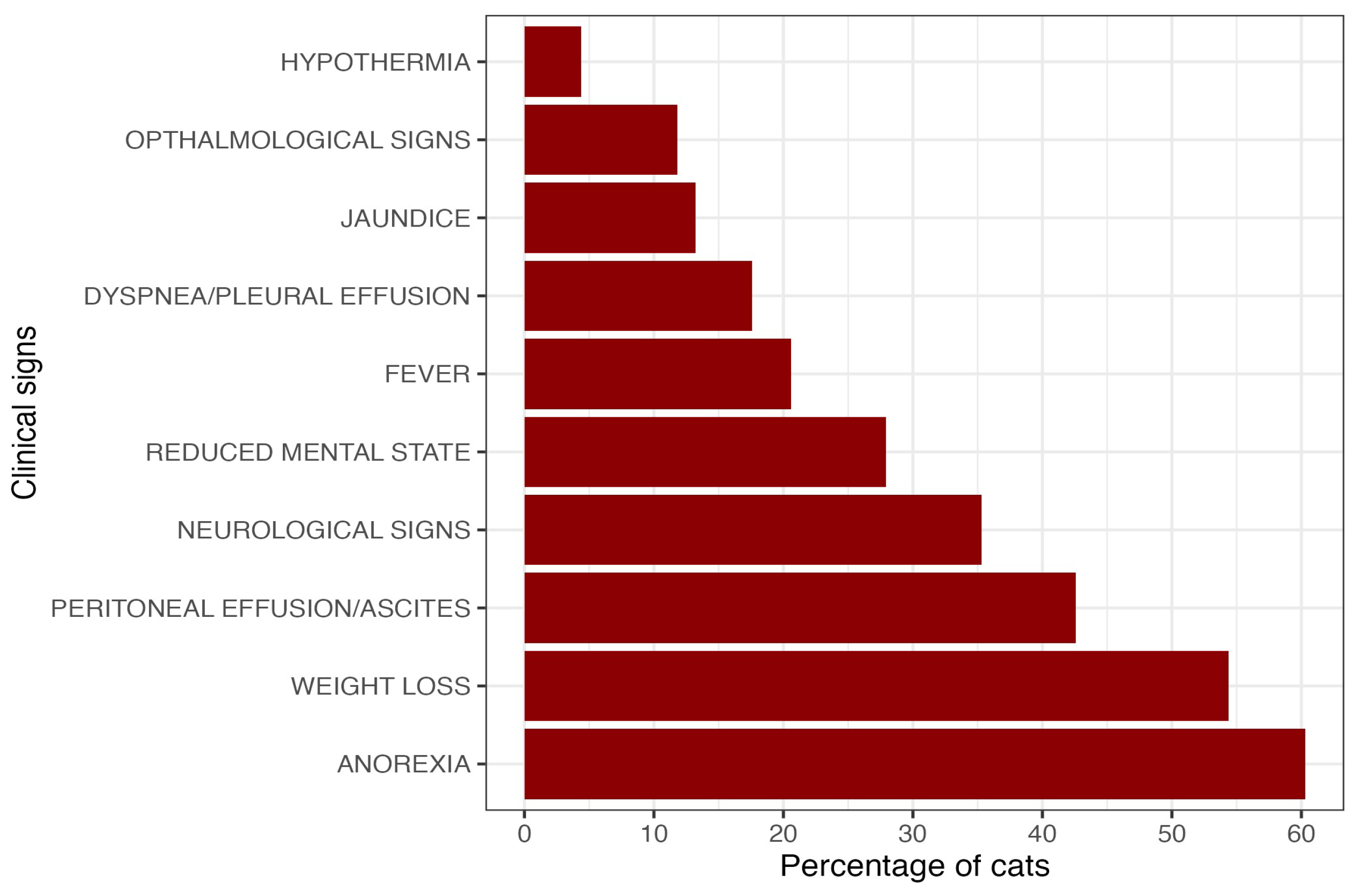
In 2023, Cyprus experienced a large-scale epizootic of feline infectious peritonitis (FIP) associated with a novel feline coronavirus, FCoV-23. While molecular investigations have elucidated its recombinant origin, field-based clinical and epidemiological data remain limited. A prospective study used a structured 30-item questionnaire embedded in veterinary management software to characterize FIP cases. Data were voluntarily submitted by registered veterinarians across Cyprus for cases identified between late 2022 and 2025. Data from 68 cases reported by 22 clinics (response rate 21.0%) were analyzed. Affected cats were older than typically reported for FIP (mean age of 3.9 years; median = 3.0; range 0.4y - 12.9y, SD = 3.41), with most cases documented from Limassol (51.5%) and Nicosia (25.0%). The most frequently reported clinical signs were anorexia (60.3%) and weight loss (54.4%), while neurological involvement was documented in 35.3% of cases. An albumin-to-globulin ratio <0.8 was observed in 86.8% of cats tested. Antiviral (GS-441524 or molnupiravir) therapy was administered in 92.2% of cases, with reported clinical improvement in 88.9%. These findings demonstrate the value of questionnaire-based reporting for documenting outbreak-associated disease patterns and confirm the change in viral tropism seen in FCoV-23 infection as an increased prevalence of neurological cases compared to ‘traditional’ FIP.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Data storage and analysis
3. Results
3.1. Questionnaire response
3.2. Case signalment and clinical history, plus geographical distribution of cases
3.3. Categorization and clinical presentation
3.4. Co-infections and stressors
3.5. Hematology and biochemistry
3.6. Diagnostic imaging
3.7. FIP diagnosis and form
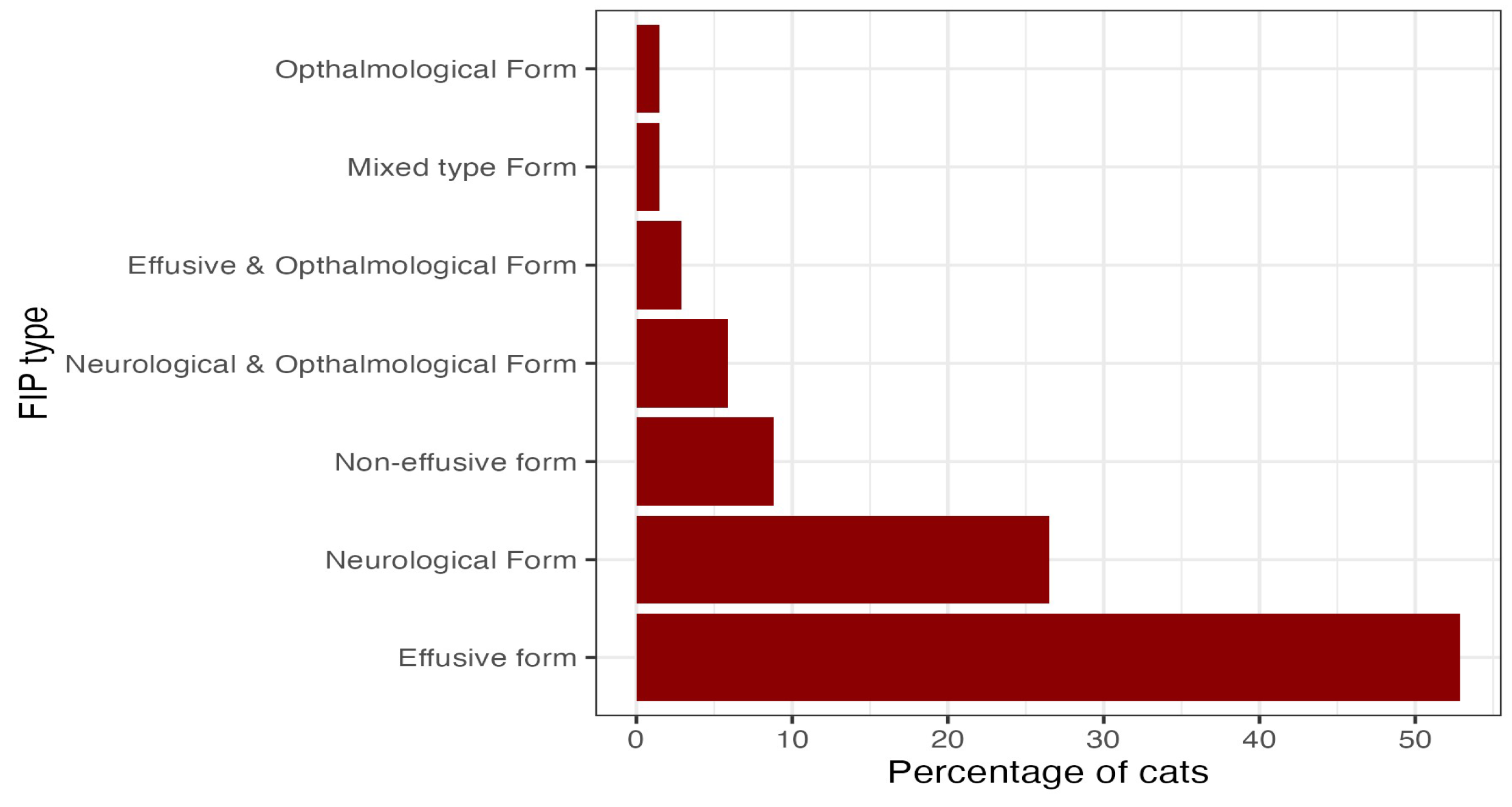
3.8. Treatment and outcome
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| FIP | Feline Infectious Peritonitis |
| FIV | Feline Immunodeficiency Virus |
| FeLV | Feline Leukaemia Virus |
| CCoV | Canine Coronavirus |
| P-CCoV | Pantropic Canine Coronavirus |
| RNA | Ribonucleic acid |
| RT-PCR | Real Time Polymerase Chain Reaction |
| ALT | Alanine Aminotransferase |
| BUN | Blood Urea Nitrogen |
| PVA | Pancyprian Veterinary Association |
| 1 | ICTV, classifies
FCoV in the order of Nidovirales, the family of Coronaviridae, the subfamily of
Orthocoronavirinae, the Genus Alphacoronavirus, the Subgenus of Tegacovirus |
| 2 | D.S. Compass
Solutions LTD |
| 3 | European Advisory Board of Cat Diseases (ABCD) Guidelines on Feline Coronavirus and Feline Infectious Peritonitis. https://www.abcdcatsvets.org/guideline-for-feline-infectious-peritonitis/Preprints.org (www.preprints.org) | NOT PEER-REVIEWED | Posted: 21 January 2026 doi:10.20944/preprints202601.1567.v1 © 2025 |
References
- Holgzinek, M.C.; Ostermaus, A.D.M.E. Archives of Virology The Girolofy and Pathogenesis of Feline Infectious Peritonitis Brief Review; 1979; Vol. 59. [Google Scholar]
- Felten, S.; Hartmann, K. Diagnosis of Feline Infectious Peritonitis: A Review of the Current Literature. Viruses 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, N.C.; Boyle, J.F.; Floyd, K.; Fudge, A.; Barker, J.; Niels Pedersen, B.C. An Enteric Coronavirus Infection of Cats and Its An Enteric Coronavirus Infection of Cats and Its Relationship to Feline Infectious Peritonitis Relationship to Feline Infectious Peritonitis.
- Jaimes, J.A.; Millet, J.K.; Stout, A.E.; André, N.M.; Whittaker, G.R. A Tale of Two Viruses: The Distinct Spike Glycoproteins of Feline Coronaviruses. Viruses 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Lauzi, S.; Stranieri, A.; Giordano, A.; Luzzago, C.; Zehender, G.; Paltrinieri, S.; Ebranati, E. Origin and Transmission of Feline Coronavirus Type I in Domestic Cats from Northern Italy: A Phylogeographic Approach. Vet. Microbiol. 2020, 244. [Google Scholar] [CrossRef]
- Pedersen, N.C. An Update on Feline Infectious Peritonitis: Virology and Immunopathogenesis. Veterinary Journal 2014, 201, 123–132. [Google Scholar] [CrossRef]
- Kipar, A.; Meli, M.L. Feline Infectious Peritonitis: Still an Enigma? Vet. Pathol. 2014, 51, 505–526. [Google Scholar] [CrossRef]
- Solikhah, T.I.; Agustin, Q.A.D.; Damaratri, R.A.; Siwi, D.A.F.; Rafi’uttaqi, G.N.; Hartadi, V.A.; Solikhah, G.P. A Review of Feline Infectious Peritonitis Virus Infection. Vet. World 2024, 17, 2417. [Google Scholar] [CrossRef]
- Attipa, C.; Gunn-Moore, D.; Mazeri, S.; Epaminondas, D.; Lyraki, M.; Hardas, A.; Loukaidou, S.; Gentil, M. Concerning Feline Infectious Peritonitis Outbreak in Cyprus. Veterinary Record 2023, 192, 449–450. [Google Scholar] [CrossRef]
- Attipa, C.; Gunn-Moore, D.; Mazeri, S.; Epaminondas, D.; Lyraki, M.; Hardas, A.; Loukaidou, S.; Gentil, M. Concerning Feline Infectious Peritonitis Outbreak in Cyprus. Veterinary Record 2023, 192, 449–450. [Google Scholar] [CrossRef]
- Attipa, C.; Warr, A.S.; Epaminondas, D.; O’Shea, M.; Hanton, A.J.; Fletcher, S.; Malbon, A.; Lyraki, M.; Hammond, R.; Hardas, A.; et al. Feline Infectious Peritonitis Epizootic Caused by a Recombinant Coronavirus. Nature 2025, 645, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Attipa, C.; Warr, A.S.; Epaminondas, D.; O’Shea, M.; Hanton, A.J.; Fletcher, S.; Malbon, A.; Lyraki, M.; Hammond, R.; Hardas, A.; et al. Emergence and Spread of Feline Infectious Peritonitis Due to a Highly Pathogenic Canine/Feline Recombinant Coronavirus 2023.
- Warr, A.; Attipa, C.; Gunn-Moore, D.; Tait-Burkard, C. FCoV-23 Causing FIP in a Cat Imported to the UK from Cyprus. Veterinary Record 2023, 193, 414–415. [Google Scholar] [CrossRef]
- Sharif, S.; Arshad, S.S.; Hair-Bejo, M.; Omar, A.R.; Zeenathul, N.A.; Hafidz, M.A. Prevalence of Feline Coronavirus in Two Cat Populations in Malaysia. J. Feline Med. Surg. 2009, 11, 1031–1034. [Google Scholar] [CrossRef]
- Felten, S.; Klein-Richers, U.; Hofmann-Lehmann, R.; Bergmann, M.; Unterer, S.; Leutenegger, C.M.; Hartmann, K. Correlation of Feline Coronavirus Shedding in Feces with Coronavirus Antibody Titer. Pathogens 2020, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Herrewegh, A.A.P.M.; De Groot, R.J.; Cepica, A.; Egberink, H.F.; Horzinek, M.C.; Rottier, P.J.M. Detection of Feline Coronavirus RNA in Feces, Tissues, and Body Fluids of Naturally Infected Cats by Reverse Transcriptase PCR; 1995; Vol. 33. [Google Scholar]
- Bell, E.T.; Toribio, J.A.L.M.L.; White, J.D.; Malik, R.; Norris, J.M. Seroprevalence Study of Feline Coronavirus in Owned and Feral Cats in Sydney, Australia. Aust. Vet. J. 2006, 84, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Kokkinaki, K.C.G.; Saridomichelakis, M.N.; Mylonakis, M.E.; Leontides, L.; Xenoulis, P.G. Seroprevalence of and Risk Factors for Feline Coronavirus Infection in Cats from Greece. Comp. Immunol. Microbiol. Infect. Dis. 2023, 94. [Google Scholar] [CrossRef]
- Pedersen, N.C.; Eckstrand, C.; Liu, H.; Leutenegger, C.; Murphy, B. Levels of Feline Infectious Peritonitis Virus in Blood, Effusions, and Various Tissues and the Role of Lymphopenia in Disease Outcome Following Experimental Infection. Vet. Microbiol. 2015, 175, 157–166. [Google Scholar] [CrossRef]
- Stranieri, A.; Probo, M.; Pisu, M.C.; Fioletti, A.; Meazzi, S.; Gelain, M.E.; Bonsembiante, F.; Lauzi, S.; Paltrinieri, S. Preliminary Investigation on Feline Coronavirus Presence in the Reproductive Tract of the Tom Cat as a Potential Route of Viral Transmission. J. Feline Med. Surg. 2020, 22, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.Y.; Wang, Q.; Liang, X.Y.; Zhang, S.; Bao, D.; Zhao, H.; Li, S.B.; Wang, K.; Hu, G.X.; Gao, F.S. An Updated Review of Feline Coronavirus: Mind the Two Biotypes. Virus Res. 2023, 326. [Google Scholar] [CrossRef]
- Pedersen, N.C.; Boyle, J.F.; Floyd, K.; Fudge, A.; Barker, J.; Niels Pedersen, B.C. An Enteric Coronavirus Infection of Cats and Its An Enteric Coronavirus Infection of Cats and Its Relationship to Feline Infectious Peritonitis Relationship to Feline Infectious Peritonitis; 1981. [Google Scholar]
- Addie, DD. J.O. Control of Feline Coronavirus Infection in Kittens. Veterinary Record 1990, 7, 164–164. [Google Scholar]
- Jaimes, J.A.; Millet, J.K.; Stout, A.E.; André, N.M.; Whittaker, G.R. A Tale of Two Viruses: The Distinct Spike Glycoproteins of Feline Coronaviruses. Viruses 2020, 12. [Google Scholar] [CrossRef]
- Pedersen, N.C.; Liu, H.; Dodd, K.A.; Pesavento, P.A. Significance of Coronavirus Mutants in Feces and Diseased Tissues of Cats Suffering from Feline Infectious Peritonitis. Viruses 2009, 1, 166–184. [Google Scholar] [CrossRef]
- Myrrha, L.W.; Silva, F.M.F.; Vidigal, P.M.P.; Resende, M.; Bressan, G.C.; Fietto, J.L.R.; Santos, M.R.; Silva, L.M.N.; Assao, V.S.; Silva-Júnior, A.; et al. Feline Coronavirus Isolates from a Part of Brazil: Insights into Molecular Epidemiology and Phylogeny Inferred from the 7b Gene. J. Vet. Med. Sci. 2019, 81, 1455–1460. [Google Scholar] [CrossRef] [PubMed]
- Kimble, B.; Coggins, S.J.; Norris, J.M.; Thompson, M.F.; Govendir, M. Quantification of GS-441524 Concentration in Feline Plasma Using High Performance Liquid Chromatography with Fluorescence Detection. Veterinary Quarterly 2023, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.S.; Coggins, S.; Barker, E.N.; Gunn-Moore, D.; Jeevaratnam, K.; Norris, J.M.; Hughes, D.; Stacey, E.; MacFarlane, L.; O’Brien, C.; et al. Retrospective Study and Outcome of 307 Cats with Feline Infectious Peritonitis Treated with Legally Sourced Veterinary Compounded Preparations of Remdesivir and GS-441524 (2020–2022). J. Feline Med. Surg. 2023, 25. [Google Scholar] [CrossRef]
- Riemer, F.; Kuehner, K.A.; Ritz, S.; Sauter-Louis, C.; Hartmann, K. Clinical and Laboratory Features of Cats with Feline Infectious Peritonitis – a Retrospective Study of 231 Confirmed Cases (2000–2010). J. Feline Med. Surg. 2016, 18, 348–356. [Google Scholar] [CrossRef]
- Pedersen, N.C. An Update on Feline Infectious Peritonitis: Virology and Immunopathogenesis. Veterinary Journal 2014, 201, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, N.C.; Allen, C.E.; Lyons, L.A. Pathogenesis of Feline Enteric Coronavirus Infection. J. Feline Med. Surg. 2008, 10, 529–541. [Google Scholar] [CrossRef]
- Hardwick, J.J.; Ioannides-Hoey, C.S.F.K.; Finch, N.; Black, V. Bicavitary Effusion in Cats: Retrospective Analysis of Signalment, Clinical Investigations, Diagnosis and Outcome. J. Feline Med. Surg. 2024, 26. [Google Scholar] [CrossRef]
- Dickinson, P.J.; Bannasch, M.; Thomasy, S.M.; Murthy, V.D.; Vernau, K.M.; Liepnieks, M.; Montgomery, E.; Knickelbein, K.E.; Murphy, B.; Pedersen, N.C. Antiviral Treatment Using the Adenosine Nucleoside Analogue GS-441524 in Cats with Clinically Diagnosed Neurological Feline Infectious Peritonitis. J. Vet. Intern. Med. 2020, 34, 1587–1593. [Google Scholar] [CrossRef]
- Riemer, F.; Kuehner, K.A.; Ritz, S.; Sauter-Louis, C.; Hartmann, K. Clinical and Laboratory Features of Cats with Feline Infectious Peritonitis – a Retrospective Study of 231 Confirmed Cases (2000–2010). J. Feline Med. Surg. 2016, 18, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Tasker, S. Diagnosis of Feline Infectious Peritonitis: Update on Evidence Supporting Available Tests. J. Feline Med. Surg. 2018, 20, 228–243. [Google Scholar] [CrossRef]
- Tasker, S.; Addie, D.D.; Egberink, H.; Hofmann-Lehmann, R.; Hosie, M.J.; Truyen, U.; Belák, S.; Boucraut-Baralon, C.; Frymus, T.; Lloret, A.; et al. Feline Infectious Peritonitis: European Advisory Board on Cat Diseases Guidelines. Viruses 2023, 15. [Google Scholar] [CrossRef]
- Taylor, S.S.; Coggins, S.; Barker, E.N.; Gunn-Moore, D.; Jeevaratnam, K.; Norris, J.M.; Hughes, D.; Stacey, E.; MacFarlane, L.; O’Brien, C.; et al. Retrospective Study and Outcome of 307 Cats with Feline Infectious Peritonitis Treated with Legally Sourced Veterinary Compounded Preparations of Remdesivir and GS-441524 (2020–2022). J. Feline Med. Surg. 2023, 25. [Google Scholar] [CrossRef]
- Gao, Y.Y.; Wang, Q.; Liang, X.Y.; Zhang, S.; Bao, D.; Zhao, H.; Li, S.B.; Wang, K.; Hu, G.X.; Gao, F.S. An Updated Review of Feline Coronavirus: Mind the Two Biotypes. Virus Res. 2023, 326. [Google Scholar] [CrossRef]
- Barker, E.N.; Tasker, S.; Gruffydd-Jones, T.J.; Tuplin, C.K.; Burton, K.; Porter, E.; Day, M.J.; Harley, R.; Fews, D.; Helps, C.R.; et al. Phylogenetic Analysis of Feline Coronavirus Strains in an Epizootic Outbreak of Feline Infectious Peritonitis. J. Vet. Intern. Med. 2013, 27, 445–450. [Google Scholar] [CrossRef]
- Wang, Y.T.; Su, B.L.; Hsieh, L.E.; Chueh, L.L. An Outbreak of Feline Infectious Peritonitis in a Taiwanese Shelter: Epidemiologic and Molecular Evidence for Horizontal Transmission of a Novel Type II Feline Coronavirus. Vet. Res. 2013, 44. [Google Scholar] [CrossRef]
- I-cat care Cat Welfare and Population Management: 10-Year Strategic Framework for Cyprus. Available online: https://icatcare.org/resources/icatcare_10-year_strategic_framework_cyprus.pdf (accessed on 9 January 2026).
- Cyprus News Agency Around 8,000 Clinical FIP Cases This Year in Cyprus, Veterinary Association Says. Available online: https://cna.org.cy/en/article/5263722/around-8-000-clinical-fip-cases-this-year-in-cyprus-veterinary-association-says (accessed on 30 December 2025).
- Tortorici, M.A.; Choi, A.; Gibson, C.A.; Lee, J.; Brown, J.T.; Stewart, C.; Joshi, A.; Harari, S.; Willoughby, I.; Treichel, C.; et al. Loss of FCoV-23 Spike Domain 0 Enhances Fusogenicity and Entry Kinetics. Nature 2025, 645, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Paltrinieri, S.; Giordano, A.; Stranieri, A.; Lauzi, S. Feline Infectious Peritonitis (FIP) and Coronavirus Disease 19 (COVID-19): Are They Similar? Transbound. Emerg. Dis. 2021, 68, 1786–1799. [Google Scholar] [CrossRef] [PubMed]
- Attipa, C.; Warr, A.S.; Epaminondas, D.; O’Shea, M.; Hanton, A.J.; Fletcher, S.; Malbon, A.; Lyraki, M.; Hammond, R.; Hardas, A.; et al. Feline Infectious Peritonitis Epizootic Caused by a Recombinant Coronavirus. Nature 2025. [Google Scholar] [CrossRef]
- Tasker, S.; Addie, D.D.; Egberink, H.; Hartmann, K.; Hofmann-Lehmann, R.; Hosie, M.J.; Truyen, U.; Belak, S.; Boucraut-Baralon, C.; Frymus, T. European Advisory Board on Cat Diseases: FIP Diagnostic Tools. Available online: https://www.abcdcatsvets.org/wp-content/uploads/2022/11/TOOL_FIP_Feline_infectious_peritonitis_December_2021_EN.pdf (accessed on 11 January 2026).
- Microsoft Corporation Microsoft Excel 2025.
- R: What Is R? Available online: https://www.r-project.org/about.html (accessed on 26 December 2025).
- Wickham, H.; Averick, M.; Bryan, J.; Chang, W.; D’, L.; Mcgowan, A.; François, R.; Grolemund, G.; Hayes, A.; Henry, L.; et al. Welcome to the Tidyverse. J. Open Source Softw. 2019, 4, 1686. [Google Scholar] [CrossRef]
- Tortorici, M.A.; Choi, A.; Gibson, C.A.; Lee, J.; Brown, J.T.; Stewart, C.; Joshi, A.; Harari, S.; Willoughby, I.; Treichel, C.; et al. Loss of FCoV-23 Spike Domain 0 Enhances Fusogenicity and Entry Kinetics. Nature 2025, 645, 235–243. [Google Scholar] [CrossRef]
- Nielsen, T.D.; Dean, R.S.; Robinson, N.J.; Massey, A.; Brennan, M.L. Survey of the UK Veterinary Profession: Common Species and Conditions Nominated by Veterinarians in Practice. Vet. Rec. 2014, 174, 324. [Google Scholar] [CrossRef] [PubMed]
- Jessen, L.R.; Sørensen, T.M.; Lilja, Z.L.; Kristensen, M.; Hald, T.; Damborg, P. Cross-Sectional Survey on the Use and Impact of the Danish National Antibiotic Use Guidelines for Companion Animal Practice. Acta Vet. Scand. 2017, 59, 81. [Google Scholar] [CrossRef] [PubMed]
- Sousa, A.; de Rago, L.; Pinho, J.O.; Estrela, M.; Coelho, A.C.; Oliveira, P.A.; Figueiras, A.; Roque, F.; Herdeiro, M.T. Understanding How Veterinarians’ Knowledge, Attitudes, and Practices Influence Antibiotic Prescription: A Systematic Review of Survey Studies. BMC Veterinary Research 2025, 21, 543. [Google Scholar] [CrossRef]
- Robin, C.; Bettridge, J.; McMaster, F. Zoonotic Disease Risk Perceptions in the British Veterinary Profession. Prev. Vet. Med. 2017, 136, 39–48. [Google Scholar] [CrossRef]
- Pedersen, N.C. An Update on Feline Infectious Peritonitis: Diagnostics and Therapeutics. Veterinary Journal 2014, 201, 133–141. [Google Scholar] [CrossRef]
- Chan, I.; Dowsey, A.; Lait, P.; Tasker, S.; Blackwell, E.; Helps, C.R.; Barker, E.N. Prevalence and Risk Factors for Common Respiratory Pathogens within a Cohort of Pet Cats in the UK. Journal of Small Animal Practice 2023, 64, 552–560. [Google Scholar] [CrossRef]
- Cats: Lovable Pets with a Big Downside for Nature - BirdLife Cyprus. Available online: https://birdlifecyprus.org/cats-lovable-pets-with-a-big-downside-for-nature/ (accessed on 9 January 2026).
- Addie, D.; Belák, S.; Boucraut-Baralon, C.; Egberink, H.; Frymus, T.; Gruffydd-Jones, T.; Hartmann, K.; Hosie, M.J.; Lloret, A.; Lutz, H.; et al. Feline Infectious Peritonitis ABCD Guidelines on Prevention and Management. J. Feline Med. Surg. 2009, 11, 594–604. [Google Scholar] [CrossRef]
- Hartmann, K. Feline Infectious Peritonitis. Veterinary Clinics of North America - Small Animal Practice 2005, 35, 39–79. [Google Scholar] [CrossRef]
- Hu, T.; Zhang, H.; Zhang, X.; Hong, X.; Zhang, T. Prevalence and Risk Factors Associated with Feline Infectious Peritonitis (FIP) in Mainland China between 2008 and 2023: A Systematic Review and Meta-Analysis. Animals (Basel) 2024, 14. [Google Scholar] [CrossRef]
- Chen, C.H.; Chang, C.C.; Chen, W.C.; Lee, Y.J. Evaluation of Chronic Stress Status and Quality of Life in Cats Suffering from Chronic Kidney Disease and Suspected Feline Infectious Peritonitis Based on Hair Cortisol Concentration Analysis and a Questionnaire. Vet. Q. 2024, 44, 1–9. [Google Scholar] [CrossRef]
- Rissi, D.R. A Retrospective Study of the Neuropathology and Diagnosis of Naturally Occurring Feline Infectious Peritonitis. Journal of Veterinary Diagnostic Investigation 2018, 30, 392–399. [Google Scholar] [CrossRef]
- Zappulli, V.; Ferro, S.; Bonsembiante, F.; Brocca, G.; Calore, A.; Cavicchioli, L.; Centelleghe, C.; Corazzola, G.; De Vreese, S.; Gelain, M.E.; et al. Pathology of Coronavirus Infections: A Review of Lesions in Animals in the One-Health Perspective. Animals 2020, 10, 2377. [Google Scholar] [CrossRef]
- Alfano, F.; Fusco, G.; Mari, V.; Occhiogrosso, L.; Miletti, G.; Brunetti, R.; Galiero, G.; Desario, C.; Cirilli, M.; Decaro, N. Circulation of Pantropic Canine Coronavirus in Autochthonous and Imported Dogs, Italy. Transbound. In Emerg. Dis.; PAGE:STRING:ARTICLE/CHAPTER, 2020; Volume 67, pp. 1991–1999. [Google Scholar] [CrossRef]
- Buonavoglia, A.; Pellegrini, F.; Decaro, N.; Galgano, M.; Pratelli, A. A One Health Perspective on Canine Coronavirus: A Wolf in Sheep’s Clothing? Microorganisms 2023, 11, 921. [Google Scholar] [CrossRef] [PubMed]
- Buonavoglia, C.; Decaro, N.; Martella, V.; Elia, G.; Campolo, M.; Desario, C.; Castagnaro, M.; Tempesta, M. Canine Coronavirus Highly Pathogenic for Dogs. Emerg. Infect. Dis. 2006, 12, 492. [Google Scholar] [CrossRef] [PubMed]
- Ntafis, V.; Mari, V.; Decaro, N.; Papanastassopoulou, M.; Pardali, D.; Rallis, T.S.; Kanellos, T.; Buonavoglia, C.; Xylouri, E. Canine Coronavirus, Greece. Molecular Analysis and Genetic Diversity Characterization. Infection, Genetics and Evolution 2013, 16, 129–136. [Google Scholar] [CrossRef]
- Decaro, N.; Mari, V.; von Reitzenstein, M.; Lucente, M.S.; Cirone, F.; Elia, G.; Martella, V.; King, V.L.; Di Bello, A.; Varello, K.; et al. A Pantropic Canine Coronavirus Genetically Related to the Prototype Isolate CB/05. Vet. Microbiol. 2012, 159, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Pratelli, A. The Evolutionary Processes of Canine Coronaviruses. Adv. Virol. 2011, 2011, 562831. [Google Scholar] [CrossRef]
- Lin, C.M.; Saif, L.J.; Marthaler, D.; Wang, Q. Evolution, Antigenicity and Pathogenicity of Global Porcine Epidemic Diarrhea Virus Strains. Virus Res. 2016, 226, 20. [Google Scholar] [CrossRef]
- Su, S.; Wong, G.; Shi, W.; Liu, J.; Lai, A.C.K.; Zhou, J.; Liu, W.; Bi, Y.; Gao, G.F. Epidemiology, Genetic Recombination, and Pathogenesis of Coronaviruses. Trends Microbiol. 2016, 24, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Felten, S.; Klein-Richers, U.; Hofmann-Lehmann, R.; Bergmann, M.; Unterer, S.; Leutenegger, C.M.; Hartmann, K. Correlation of Feline Coronavirus Shedding in Feces with Coronavirus Antibody Titer. Pathogens 2020, 9, 1–13. [Google Scholar] [CrossRef]
- Pasteur, K.; Diana, A.; Yatcilla, J.K.; Barnard, S.; Croney, C.C. Access to Veterinary Care: Evaluating Working Definitions, Barriers, and Implications for Animal Welfare. Front. Vet. Sci. 2024, 11, 1335410. [Google Scholar] [CrossRef]
- Becker, M.; Volk, H.; Kunzmann, P. Is Pet Health Insurance Able to Improve Veterinary Care? Why Pet Health Insurance for Dogs and Cats Has Limits: An Ethical Consideration on Pet Health Insurance. Animals 2022, 12, 1728. [Google Scholar] [CrossRef]
- Springer, S.; Lund, T.B.; Grimm, H.; Kristensen, A.T.; Corr, S.A.; Sandøe, P. Comparing Veterinarians’ Attitudes to and the Potential Influence of Pet Health Insurance in Austria, Denmark and the UK. In Veterinary Record; ISSUE:ISSUE:DOI, 2022; Volume 190, p. no. [Google Scholar] [CrossRef]
- Taylor, S.S.; Tappin, S.W.; Dodkin, S.J.; Papasouliotis, K.; Casamian-Sorrosal, D.; Tasker, S. Serum Protein Electrophoresis in 155 Cats. J. Feline Med. Surg. 2010, 12, 643–653. [Google Scholar] [CrossRef]
- Stranieri, A.; Giordano, A.; Bo, S.; Braghiroli, C.; Paltrinieri, S. Frequency of Electrophoretic Changes Consistent with Feline Infectious Peritonitis in Two Different Time Periods (2004–2009 vs 2013–2014). J. Feline Med. Surg. WEBSITE:WEBSITE:SAGE;REQUESTEDJOURNAL:JOURNAL:JFMA. 2017, 19, 880–887. [Google Scholar] [CrossRef] [PubMed]
- FECAVA, 2023 Feline Infectious Peritonitis (FIP) Treatment Allowed in Cyprus after FCoV-2023-Outbreak - FECAVA. Available online: https://www.fecava.org/news-and-events/news/feline-infectious-peritonitis-fip-treatment-allowed-in-cyprus/ (accessed on 7 November 2025).
- Pedersen, N.C.; Perron, M.; Bannasch, M.; Montgomery, E.; Murakami, E.; Liepnieks, M.; Liu, H. Efficacy and Safety of the Nucleoside Analog GS-441524 for Treatment of Cats with Naturally Occurring Feline Infectious Peritonitis. J. Feline Med. Surg. 2019, 21, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Černá, P.; Wittenburg, L.; Hawley, J.; Willis, M.; Siegenthaler, B.; Lappin, M.R. Pharmacokinetics of Molnupiravir in Cats with Naturally Occurring Feline Infectious Peritonitis. Pathogens 2025, 14. [Google Scholar] [CrossRef]
- Kimble, B.; Coggins, S.J.; Norris, J.M.; Thompson, M.F.; Govendir, M. Quantification of GS-441524 Concentration in Feline Plasma Using High Performance Liquid Chromatography with Fluorescence Detection. Veterinary Quarterly 2023, 43, 1–9. [Google Scholar] [CrossRef]
- Kent, A.M.; Guan, S.; Jacque, N.; Novicoff, W.; Evans, S.J.M. Unlicensed Antiviral Products Used for the At-Home Treatment of Feline Infectious Peritonitis Contain GS-441524 at Significantly Different Amounts than Advertised. J. Am. Vet. Med. Assoc. 2024, 262, 489–497. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.; Novicoff, W.; Nadeau, J.; Evans, S. Unlicensed Gs-441524-like Antiviral Therapy Can Be Effective for at-Home Treatment of Feline Infectious Peritonitis. Animals 2021, 11. [Google Scholar] [CrossRef]
- Cerna, P.; Dow, S.; Hawley, J.; Willis, M.; Lappin, M.R. EXPRESS: Clinical Trial of Molnupiravir with or without an Oral Immune Stimulant as a First-Line Treatment of Feline Infectious Peritonitis. J. Feline Med. Surg. 2025. [Google Scholar] [CrossRef]
- Roy, M.; Jacque, N.; Novicoff, W.; Li, E.; Negash, R.; Evans, S.J.M. Unlicensed Molnupiravir Is an Effective Rescue Treatment Following Failure of Unlicensed GS-441524-like Therapy for Cats with Suspected Feline Infectious Peritonitis. Pathogens 2022, 11. [Google Scholar] [CrossRef]
- Clark, T.M.; Coggins, S.J.; Korman, R.; King, J.; Malik, R. Treatment of Feline Infectious Peritonitis in Cats with Molnupiravir: Clinical Observations and Outcomes for 54 Cases. Aust. Vet. J. 2025, 103, 339–353. [Google Scholar] [CrossRef] [PubMed]
- Sase, O. Molnupiravir Treatment of 18 Cats with Feline Infectious Peritonitis: A Case Series. J. Vet. Intern. Med. 2023, 37, 1876–1880. [Google Scholar] [CrossRef]
- Reagan, K.L.; Brostoff, T.; Pires, J.; Rose, A.; Castillo, D.; Murphy, B.G. Open Label Clinical Trial of Orally Administered Molnupiravir as a First-Line Treatment for Naturally Occurring Effusive Feline Infectious Peritonitis. J. Vet. Intern. Med. 2024, 38, 3087–3094. [Google Scholar] [CrossRef]
- Sase, O.; Iwami, T.; Sasaki, T.; Sano, T. GS-441524 and Molnupiravir Are Similarly Effective for the Treatment of Cats with Feline Infectious Peritonitis. Front. Vet. Sci. 2024, 11. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



