Submitted:
15 January 2026
Posted:
21 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Tumor Cases in Axolotls
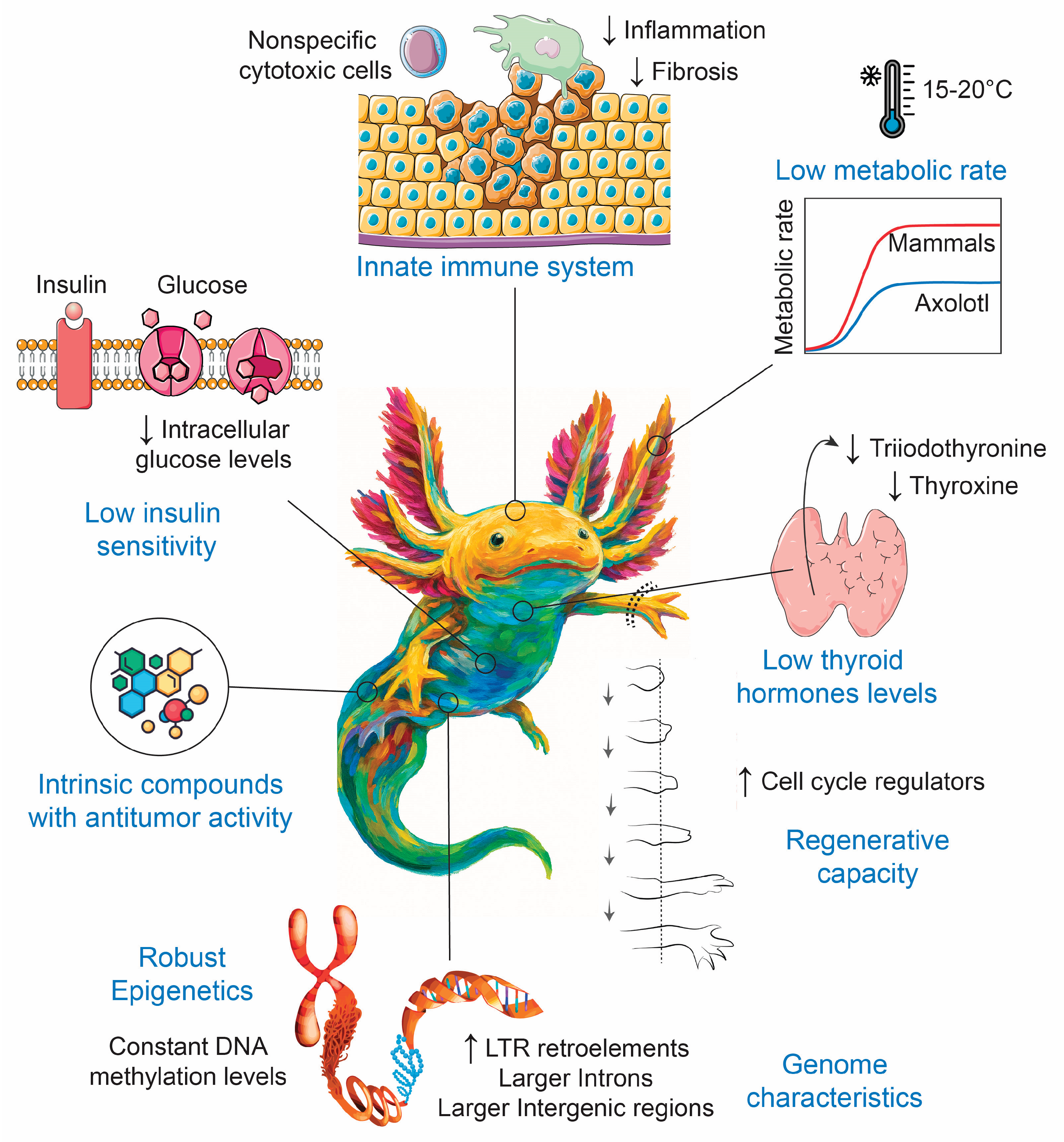
3. Possible Molecular Mechanisms Involved in the Reduction of Cancer Incidence in Axolotls
3.1. Habitat and Low Metabolic Rate
3.2. Low Levels of Thyroid Hormones
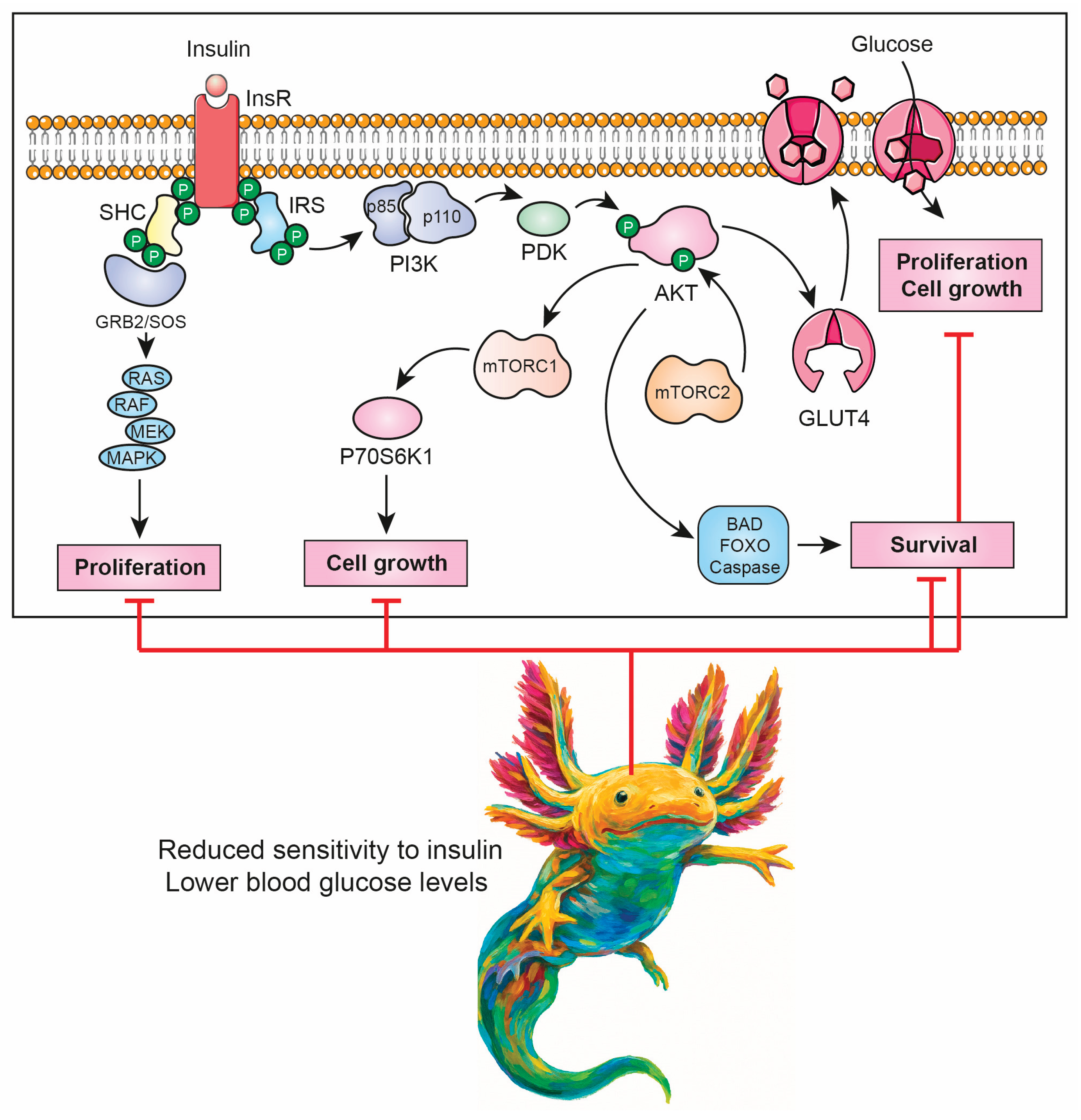
3.3. Low Insulin Sensitivity
3.4. Intrinsic Natural Compounds with Antitumor Activity
3.5. Genome and Epigenetic Characteristics
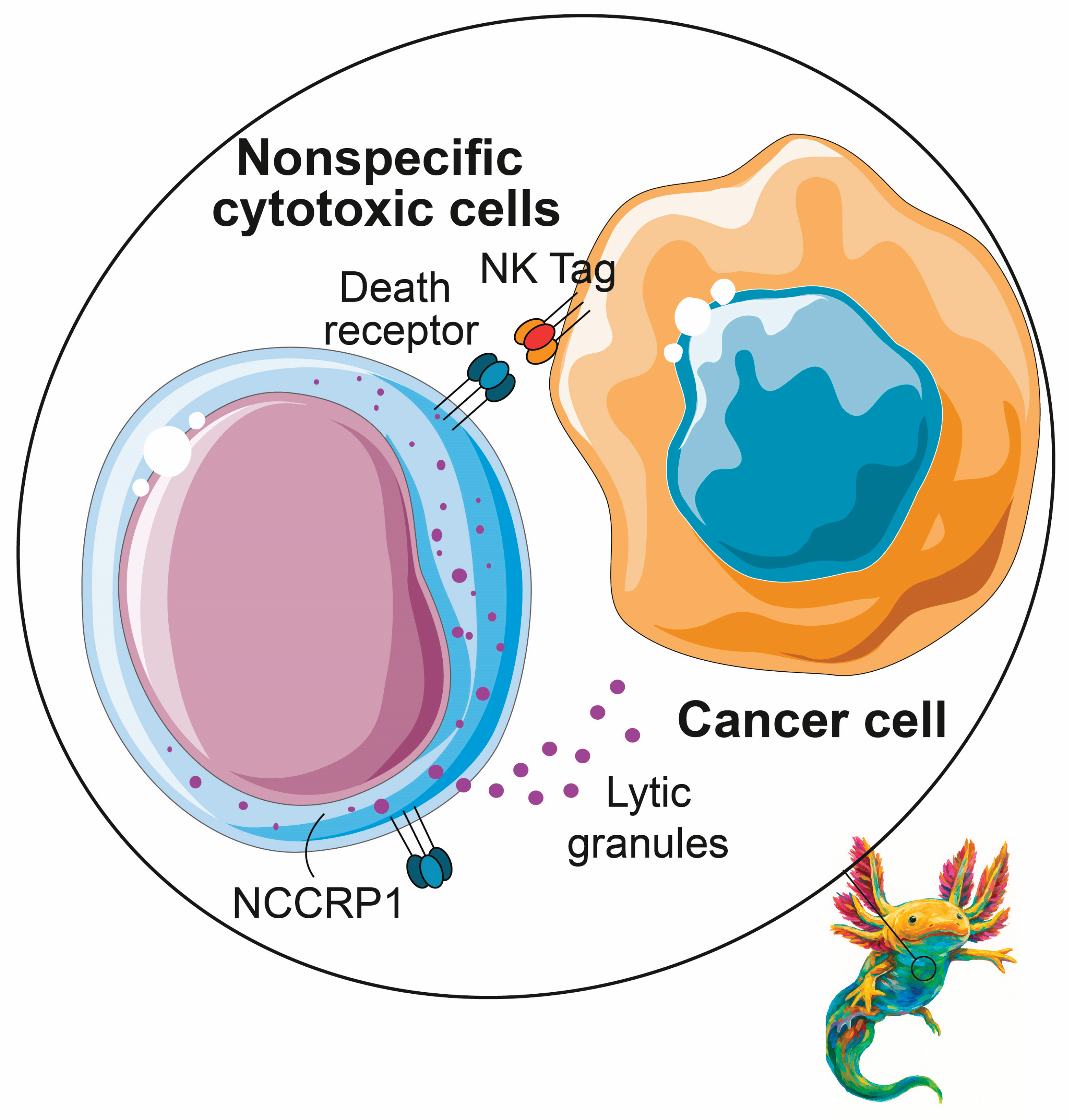
3.6. Innate Immune System
3.7. Regeneration Capacity
4. Conclusions and Future Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| NCDs | Noncommunicable diseases |
| ROS | Reactive oxygen species |
| TP53 | Tumor protein p53 |
| HPT | Hypothalamo-pituitary-thyroid (axis) |
| NCCRP1 | Non-specific cytotoxic cell receptor protein 1 |
| NCCs | Nonspecific cytotoxic cells |
| NK | Natural killer (Cells) |
| NK Tag | Natural killer target antigen |
| MHC | Major histocompatibility complex |
| ECM | Extracellular matrix |
| H2O2 | Hydrogen peroxide |
| SASP | Senescence-associated secretory phenotype |
| IGF-1 | Insulin-like growth factor |
| BLAST | basic local alignment search tool |
References
- T. Sandoval-Guzmán, The axolotl, Nat Methods 20 (2023) 1117–1119. [CrossRef]
- C. McCusker, S. V. Bryant, D.M. Gardiner, The axolotl limb blastema: cellular and molecular mechanisms driving blastema formation and limb regeneration in tetrapods, Regeneration 2 (2015) 54–71. [CrossRef]
- R. Stone, In Mexico, the world’s last wild axolotls face extinction, Science (1979) 380 (2023) 570–570. [CrossRef]
- D.M. Parkin, F. Bray, J. Ferlay, P. Pisani, Global cancer statistics, 2002., CA Cancer J Clin 55 (2005) 74–108. [CrossRef]
- F.A. Lagunas-Rangel, DNA damage accumulation and repair defects in FLT3-ITD acute myeloid leukemia: Implications for clonal evolution and disease progression, Hematol Oncol 41 (2023) 26–38. [CrossRef]
- F.A. Lagunas-Rangel, Role of circular RNAs in DNA repair, RNA Biol 21 (2024) 149–161. [CrossRef]
- D. Hanahan, Hallmarks of Cancer: New Dimensions, Cancer Discov 12 (2022) 31–46. [CrossRef]
- L. Nunney, The real war on cancer: the evolutionary dynamics of cancer suppression, Evol Appl 6 (2013) 11–19. [CrossRef]
- F.A. Lagunas-Rangel, V. Chávez-Valencia, Learning of nature: The curious case of the naked mole rat, Mech Ageing Dev 164 (2017) 76–81. [CrossRef]
- F.A. Lagunas-Rangel, Cancer-free aging: Insights from Spalax ehrenbergi superspecies, Ageing Res Rev 47 (2018) 18–23. [CrossRef]
- F.A. Lagunas-Rangel, Why do bats live so long?—Possible molecular mechanisms, Biogerontology 21 (2020) 1–11. [CrossRef]
- F.A. Lagunas-Rangel, Deciphering the whale’s secrets to have a long life, Exp Gerontol 151 (2021) 111425. [CrossRef]
- A. Seluanov, V.N. Gladyshev, J. Vijg, V. Gorbunova, Mechanisms of cancer resistance in long-lived mammals, Nat Rev Cancer (2018) 1. [CrossRef]
- A.J. Ingram, The reactions to carcinogens in the axolotl ( Ambystoma mexicanum ) in relation to the ‘regeneration field control’ hypothesis, Development 26 (1971) 425–441. [CrossRef]
- R. Tacutu, D. Thornton, E. Johnson, A. Budovsky, Di. Barardo, T. Craig, E. Diana, G. Lehmann, D. Toren, J. Wang, V.E. Fraifeld, J.P. de Magalhães, Human Ageing Genomic Resources: new and updated databases, Nucleic Acids Res 46 (2018) D1083–D1090. [CrossRef]
- W.A. Vieira, K.M. Wells, C.D. McCusker, Advancements to the Axolotl Model for Regeneration and Aging, Gerontology 66 (2020) 212–222. [CrossRef]
- K. Sousounis, A.T. Athippozhy, S.R. Voss, P.A. Tsonis, Plasticity for axolotl lens regeneration is associated with age-related changes in gene expression, Regeneration 1 (2014) 47–57. [CrossRef]
- A. Bölük, M. Yavuz, T. Demircan, Axolotl: A resourceful vertebrate model for regeneration and beyond, Developmental Dynamics 251 (2022) 1914–1933. [CrossRef]
- S. Nowoshilow, S. Schloissnig, J.-F. Fei, A. Dahl, A.W.C.W.C. Pang, M. Pippel, S. Winkler, A.R.R. Hastie, G. Young, J.G.G. Roscito, F. Falcon, D. Knapp, S. Powell, A. Cruz, H. Cao, B. Habermann, M. Hiller, E.M.M. Tanaka, E.W.W. Myers, The axolotl genome and the evolution of key tissue formation regulators, Nature 554 (2018) 50–55. [CrossRef]
- Multiple Melanomas in the Axolot Ambystoma mexicanum, JNCI: Journal of the National Cancer Institute (1979). [CrossRef]
- H.G. Schlumberger, B.H.E.W. Lucke, Tumors of fishes, amphibians, and reptiles., Cancer Res 8 (1948) 657–753. http://www.ncbi.nlm.nih.gov/pubmed/18119893.
- S.K. Frost, L.G. Epp, S.J. Robinson, The pigmentary system of developing axolotls, Development 81 (1984) 105–125. [CrossRef]
- L. Denat, A.L. Kadekaro, L. Marrot, S.A. Leachman, Z.A. Abdel-Malek, Melanocytes as Instigators and Victims of Oxidative Stress, Journal of Investigative Dermatology 134 (2014) 1512–1518. [CrossRef]
- E.A.A. Sheremetieva, Spontaneous melanoma in regenerating tails of axolotls, Journal of Experimental Zoology 158 (1965) 101–121. [CrossRef]
- K.S. Vuong, C. Metral, W. Sheley, M. Liu, A.K. Carlson, M.P. Jones, Chromatophoromas on the tail of a pet axolotl (Ambystoma mexicanum) with presumptive metastasis, J Exot Pet Med 45 (2023) 8–13. [CrossRef]
- V.V. V Brunst, A.L.L. Roque, A spontaneous teratoma in an axolotl (Siredon mexicanum)., Cancer Res 29 (1969) 223–9.
- J.C.C. Harshbarger, S.C.C. Chang, L.E.E. DeLanney, F.L.L. Rose, D.E.E. Green, Cutaneous mastocytomas in the neotenic caudate amphibians Ambystoma mexicanum (axolotl) and Ambystoma tigrinum (tiger salamander), J Cancer Res Clin Oncol 125 (1999) 187–192. [CrossRef]
- M. Lo, P.R. Badial, P.M. DiGeronimo, E.M. Littman, N. Aquilino, T. Stokol, N.I. Stacy, R.J. Ossiboff, Acute myeloid leukemia in a genetically related captive population of axolotls ( Ambystoma mexicanum ), Vet Pathol (2025). [CrossRef]
- B. Menger, P.M. Vogt, I.D. Jacobsen, C. Allmeling, J.W. Kuhbier, F. Mutschmann, K. Reimers, Resection of a large intra-abdominal tumor in the Mexican axolotl: a case report., Vet Surg 39 (2010) 232–3. [CrossRef]
- F. Modesto, A. Nicolier, C. Hurtrel, J. Benoît, Excisional biopsy and radiotherapy for management of an olfactory neuroblastoma in an axolotl (Ambystoma mexicanum), J Am Vet Med Assoc 260 (2022) 436–441. [CrossRef]
- C. Shioda, K. Uchida, H. Nakayama, Pathological features of olfactory neuroblastoma in an axolotl (Ambystoma mexicanum)., J Vet Med Sci 73 (2011) 1109–11. [CrossRef]
- R.R. Humphrey, Tumors of the Testis in the Mexican Axolotl (Ambystoma, or Siredon, mexicanum), in: 1969: pp. 220–228. [CrossRef]
- L.E.E. DeLanney, Lymphosarcoma in the Mexican Axolotl, Ambystoma mexicanum, in: Bibl Haematol, 1970: pp. 642–642. [CrossRef]
- T. Seki, Y. Yang, X. Sun, S. Lim, S. Xie, Z. Guo, W. Xiong, M. Kuroda, H. Sakaue, K. Hosaka, X. Jing, M. Yoshihara, L. Qu, X. Li, Y. Chen, Y. Cao, Brown-fat-mediated tumour suppression by cold-altered global metabolism, Nature 608 (2022) 421–428. [CrossRef]
- J.C.M. Ng, C.M. Schooling, Effect of Basal Metabolic Rate on Cancer: A Mendelian Randomization Study, Front Genet 12 (2021). [CrossRef]
- H. Ma, G. Peng, Y. Hu, B. Lu, Y. Zheng, Y. Wu, W. Feng, Y. Shi, X. Pan, L. Song, I. Stützer, Y. Liu, J. Fei, Revealing the biological features of the axolotl pancreas as a new research model, Front Cell Dev Biol 13 (2025). [CrossRef]
- A. Dittrich, S.A. Andersson, M. Busk, K. Hansen, C.B. Foldager, J. Palmfeldt, A. Andersen, M. Pedersen, M. Vendelbo, K.L. Nielsen, H. Lauridsen, Metabolic changes during cardiac regeneration in the axolotl, Developmental Dynamics (2025). [CrossRef]
- T.P. Grazioso, N. Djouder, The forgotten art of cold therapeutic properties in cancer: A comprehensive historical guide, IScience 26 (2023) 107010. [CrossRef]
- É. Villiard, H. Brinkmann, O. Moiseeva, F.A. Mallette, G. Ferbeyre, S. Roy, Urodele p53 tolerates amino acid changes found in p53 variants linked to human cancer, BMC Evol Biol 7 (2007) 180. [CrossRef]
- Y. Liu, Z. Su, O. Tavana, W. Gu, Understanding the complexity of p53 in a new era of tumor suppression, Cancer Cell 42 (2024) 946–967. [CrossRef]
- K. V. Prahlad, L.E. DeLanney, A study of induced metamorphosis in the Axolotl, Journal of Experimental Zoology 160 (1965) 137–145. [CrossRef]
- B. De Groef, S.V.H. Grommen, V.M. Darras, Forever young: Endocrinology of paedomorphosis in the Mexican axolotl (Ambystoma mexicanum), Gen Comp Endocrinol 266 (2018) 194–201. [CrossRef]
- R. Mullur, Y.-Y. Liu, G.A. Brent, Thyroid Hormone Regulation of Metabolism, Physiol Rev 94 (2014) 355–382. [CrossRef]
- E. Krashin, A. Piekiełko-Witkowska, M. Ellis, O. Ashur-Fabian, Thyroid Hormones and Cancer: A Comprehensive Review of Preclinical and Clinical Studies, Front Endocrinol (Lausanne) 10 (2019). [CrossRef]
- H. Lin, Y. Chin, Y.S.H. Yang, H. Lai, J. Whang-Peng, L.F. Liu, H. Tang, P.J. Davis, Thyroid Hormone, Cancer, and Apoptosis, in: Compr Physiol, Wiley, 2016: pp. 1221–1237. [CrossRef]
- B.R. Gauthier, A. Sola-García, M.Á. Cáliz-Molina, P.I. Lorenzo, N. Cobo-Vuilleumier, V. Capilla-González, A. Martin-Montalvo, Thyroid hormones in diabetes, cancer, and aging, Aging Cell 19 (2020). [CrossRef]
- F.A. Lagunas-Rangel, Naked mole-rat hyaluronan, Biochimie 220 (2024) 58–66. [CrossRef]
- B.P. Leitner, S. Siebel, N.D. Akingbesote, X. Zhang, R.J. Perry, Insulin and cancer: a tangled web, Biochemical Journal 479 (2022) 583–607. [CrossRef]
- D. Guo, Y. Meng, G. Zhao, Q. Wu, Z. Lu, Moonlighting functions of glucose metabolic enzymes and metabolites in cancer, Nat Rev Cancer 25 (2025) 426–446. [CrossRef]
- N. Dastagir, C. Liebsch, J. Kutz, S. Wronski, A. Pich, D. Obed, P.M. Vogt, V. Bucan, S. Strauß, Identification of antimicrobial peptides from the Ambystoma mexicanum displaying antibacterial and antitumor activity, PLoS One 20 (2025) e0316257. [CrossRef]
- S. Suleiman, R. Di Fiore, A. Cassar, M.M. Formosa, P. Schembri-Wismayer, J. Calleja-Agius, Axolotl Ambystoma mexicanum extract induces cell cycle arrest and differentiation in human acute myeloid leukemia HL-60 cells., Tumour Biol 42 (2020) 1010428320954735. [CrossRef]
- C. Allegrucci, M.D. Rushton, J.E. Dixon, V. Sottile, M. Shah, R. Kumari, S. Watson, R. Alberio, A.D. Johnson, Epigenetic reprogramming of breast cancer cells with oocyte extracts, Mol Cancer 10 (2011) 7. [CrossRef]
- R. Stewart, C.A. Rascón, S. Tian, J. Nie, C. Barry, L.-F. Chu, H. Ardalani, R.J. Wagner, M.D. Probasco, J.M. Bolin, N. Leng, S. Sengupta, M. Volkmer, B. Habermann, E.M. Tanaka, J.A. Thomson, C.N. Dewey, Comparative RNA-seq Analysis in the Unsequenced Axolotl: The Oncogene Burst Highlights Early Gene Expression in the Blastema, PLoS Comput Biol 9 (2013) e1002936. [CrossRef]
- S.R. Voss, A. Palumbo, R. Nagarajan, D.M. Gardiner, K. Muneoka, A.J. Stromberg, A.T. Athippozhy, Gene expression during the first 28 days of axolotl limb regeneration I: Experimental design and global analysis of gene expression, Regeneration 2 (2015) 120–136. [CrossRef]
- B. Corradetti, P. Dogra, S. Pisano, Z. Wang, M. Ferrari, S. Chen, R.L. Sidman, R. Pasqualini, W. Arap, V. Cristini, Amphibian regeneration and mammalian cancer: Similarities and contrasts from an evolutionary biology perspective, BioEssays 43 (2021). [CrossRef]
- M. Conrad, The mutation buffering concept of biomolecular structure, J Biosci 8 (1985) 669–679. [CrossRef]
- S. Nowoshilow, E.M. Tanaka, Introducing www.axolotl-omics.org – an integrated -omics data portal for the axolotl research community, Exp Cell Res 394 (2020) 112143. [CrossRef]
- Y. Haluza, J.A. Zoller, A.T. Lu, H.E. Walters, M. Lachnit, R. Lowe, A. Haghani, R.T. Brooke, N. Park, M.H. Yun, S. Horvath, Axolotl epigenetic clocks offer insights into the nature of negligible senescence, BioRxiv (2024). [CrossRef]
- S. Sonar, S. Nyahatkar, K. Kalele, M.D. Adhikari, Role of DNA methylation in cancer development and its clinical applications, Clinical and Translational Discovery 4 (2024). [CrossRef]
- D. Lopez, L. Lin, J.R. Monaghan, C.R. Cogle, F.J. Bova, M. Maden, E.W. Scott, Mapping hematopoiesis in a fully regenerative vertebrate: the axolotl, Blood 124 (2014) 1232–1241. [CrossRef]
- J.S. Fellah, F. Kerfourn, A.-M. Dumay, G. Aubet, J. Charlemagne, Structure and diversity of the T-cell receptor α chain in the Mexican axolotl, Immunogenetics 45 (1997) 235–241. [CrossRef]
- A. Tournefier, V. Laurens, C. Chapusot, P. Ducoroy, M.R. Padros, F. Salvudori, B. Sammut, Structure of MHC class I and class II cDNAs and possible immunodeficiency linked to class II expression in the Mexican axolotl, Immunol Rev 166 (1998) 259–277. [CrossRef]
- J.D. Cotter, A. Storfer, R.B. Page, C.K. Beachy, S.R. Voss, Transcriptional response of Mexican axolotls to Ambystoma tigrinum virus (ATV) infection, BMC Genomics 9 (2008) 493. [CrossRef]
- K. Reimers, M. Abu Qarn, C. Allmeling, V. Bucan, P.M. Vogt, Identification of the non-specific cytotoxic cell receptor protein 1 (NCCRP1) in regenerating axolotl limbs, Journal of Comparative Physiology B 176 (2006) 599–605. [CrossRef]
- M. Huang, Z. Zhang, X. Li, J. Feng, Y.Y. Huang, A. Kwaku, Y.Y. Huang, J. Jian, Molecular characteristics and functional analysis of non-specific cytotoxic cell receptor (NCCRP1) in golden pompano (Trachinotus ovatus), Fish Shellfish Immunol 145 (2024) 109344. [CrossRef]
- D.T. Harris, L. Jaso-Friedmann, R.B. Devlin, H.S. Koren, D.L. Evans, Identification of an evolutionarily conserved, function-associated molecule on human natural killer cells., Proceedings of the National Academy of Sciences 88 (1991) 3009–3013. [CrossRef]
- A. Czarkwiani, M. Lobo, L.A.B. Castro, A. Petzold, F. Rost, R. Maehr, M.H. Yun, Molecular basis for de novo thymus regeneration in a vertebrate, the axolotl, Sci Immunol 10 (2025). [CrossRef]
- F.A. Lagunas-Rangel, Neutrophil-to-lymphocyte ratio in aging: Trends and clinical implications, Exp Gerontol 211 (2025) 112908. [CrossRef]
- A. Mier-Briseño, M.A. Benavides-Huerto, I. Padilla-Ponce, F.A. Lagunas-Rangel, Atypical Carcinoid of the Thymus: Early Diagnosis in a Case Report, Medical Sciences 13 (2025) 96. [CrossRef]
- Y.-C. Ching, R.J. Wedgwood, Immunologic Responses in the Axolotl, Siredon Mexicanum, The Journal of Immunology 99 (1967) 191–200. [CrossRef]
- A. Tournefier, S. Fellah, J. Charlemagne, Monoclonal antibodies to axolotl immunoglobulins specific for different heavy chains isotypes expressed by independent lymphocyte subpopulations, Immunol Lett 18 (1988) 145–148. [CrossRef]
- J. Charlemagne, Thymus independent anti-horse erythrocyte antibody response and suppressor T cells in the Mexican axolotl (Amphibia, Urodela, ambystoma mexicanum)., Immunology 36 (1979) 643–8.
- A.P. Ussing, P. Rosenkilde, Effect of Induced Metamorphosis on the Immune System of the Axolotl, Ambystoma mexicanum, Gen Comp Endocrinol 97 (1995) 308–319. [CrossRef]
- Y. Wen, Y. Zhu, C. Zhang, X. Yang, Y. Gao, M. Li, H. Yang, T. Liu, H. Tang, Chronic inflammation, cancer development and immunotherapy, Front Pharmacol 13 (2022). [CrossRef]
- F.A. Lagunas-Rangel, Neutrophil-to-lymphocyte ratio in aging: Trends and clinical implications, Exp Gerontol 211 (2025) 112908. [CrossRef]
- E.M. Tanaka, P.W. Reddien, The Cellular Basis for Animal Regeneration, Dev Cell 21 (2011) 172–185. [CrossRef]
- A. Espinal-Centeno, M. Dipp-Álvarez, C. Saldaña, L. Bako, A. Cruz-Ramírez, Conservation analysis of core cell cycle regulators and their transcriptional behavior during limb regeneration in Ambystoma mexicanum, Mech Dev 164 (2020) 103651. [CrossRef]
- S. Ruiz-Pérez, N. Alcaraz, K. Torres-Arciga, J.A. Ocampo-Cervantes, A. Cervera, C. Castro-Hernández, C.G. Sámano-Salazar, E. Soto-Reyes, R. González-Barrios, Retrotransposon Expression Is Upregulated in Adulthood and Suppressed during Regeneration of the Limb in the Axolotl ( Ambystoma mexicanum ), Adv Biol (2025). [CrossRef]
- A. Zippo, S. Beyes, Molecular mechanisms altering cell identity in cancer, Oncogene (2025). [CrossRef]
- B. Piersma, M.-K. Hayward, V.M. Weaver, Fibrosis and cancer: A strained relationship, Biochimica et Biophysica Acta (BBA) - Reviews on Cancer 1873 (2020) 188356. [CrossRef]
- T. Endo, S. V Bryant, D.M. Gardiner, A stepwise model system for limb regeneration, Dev Biol 270 (2004) 135–145. [CrossRef]
- A.W. Seifert, J.R. Monaghan, S.R. Voss, M. Maden, Skin Regeneration in Adult Axolotls: A Blueprint for Scar-Free Healing in Vertebrates, PLoS One 7 (2012) e32875. [CrossRef]
- A.K. Rodgers, J.J. Smith, S.R. Voss, Identification of immune and non-immune cells in regenerating axolotl limbs by single-cell sequencing, Exp Cell Res 394 (2020) 112149. [CrossRef]
- J.W. Godwin, R. Debuque, E. Salimova, N.A. Rosenthal, Heart regeneration in the salamander relies on macrophage-mediated control of fibroblast activation and the extracellular landscape, NPJ Regen Med 2 (2017) 22. [CrossRef]
- B. Carbonell-M, J. Zapata Cardona, J.P. Delgado, Post-amputation reactive oxygen species production is necessary for axolotls limb regeneration, Front Cell Dev Biol 10 (2022). [CrossRef]
- B. Carbonell M, J. Zapata Cardona, J.P. Delgado, Hydrogen peroxide is necessary during tail regeneration in juvenile axolotl, Developmental Dynamics 251 (2022) 1054–1076. [CrossRef]
- J.W. Godwin, A.R. Pinto, N.A. Rosenthal, Macrophages are required for adult salamander limb regeneration, Proceedings of the National Academy of Sciences 110 (2013) 9415–9420. [CrossRef]
- F.A. Lagunas-Rangel, J.V. Linnea-Niemi, B. Kudłak, M.J. Williams, J. Jönsson, H.B. Schiöth, Role of the Synergistic Interactions of Environmental Pollutants in the Development of Cancer., Geohealth 6 (2022) e2021GH000552. [CrossRef]
- P.D. Ray, B.-W. Huang, Y. Tsuji, Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling, Cell Signal 24 (2012) 981–990. [CrossRef]
- J.R. Monaghan, A.C. Stier, F. Michonneau, M.D. Smith, B. Pasch, M. Maden, A.W. Seifert, Experimentally induced metamorphosis in axolotls reduces regenerative rate and fidelity, Regeneration 1 (2014) 2–14. [CrossRef]
| Organism | ||||
|---|---|---|---|---|
| Feature | Axolotl | Zebrafish | Mice | Human |
| Maximum longevity [15] | 21 years (captivity) |
5.5 years (captivity) |
4 years (captivity) |
122.5 years |
| Basal metabolic rate [15] | ~0.025 W | ~0.00056 W | 0.2710 W | 82.7800 W |
| Body mass [15] | ~60–150 g (lab-bred individuals) |
~0.3–0.9 g (lab-reared adults) |
18.0 g | 70,000.0 g |
| Typical body temperature [15] | ~16–20 °C (laboratory) |
~28 °C (laboratory) |
36.9 °C | 37.0 °C |
| Cancer incidence | Rare (suggested) |
Moderate | High | Moderate |
| Regenerative capacity | Very high | High (embryos) Moderate (adults) |
Limited | Very limited |
| Genome size [19] | ∼32 Gb | ∼1.5 Gb | ∼2.7 Gb | ∼3.2 Gb |
| Chromosomes [19] | 56 | 50 | 40 | 46 |
| Protein-coding genes [19] | 23,251 | 26,206 | 21,529 | 19,435 |
| Median intron length [19] | 22,759 bp | ~1350 bp | 1469 bp | 1750 bp |
| LTR retroelements [19] | High | Low | Moderate | Moderate |
| Innate immune system | Robust and active | Functional and efficient | Functional and balanced | Functional and balanced |
| Adaptive immune system | Limited | Limited | Diversified and efficient | Highly diversified and efficient |
| Hallmark of Cancer | Hallmark Description | Axolotl Mechanisms That May Counteract the Hallmark Trait |
|---|---|---|
| Sustaining proliferative signaling | Cancer cells maintain chronic growth-promoting signals that drive uncontrolled proliferation. | - Low levels of thyroid hormones - Low insulin sensitivity - Genomic stability |
| Evading growth suppressors | Cancer cells inactivate tumor suppressor pathways that normally restrict cell division. | - Genomic stability - Action of tumor suppressor genes |
| Resisting cell death | Malignant cells circumvent apoptosis and other programmed cell death mechanisms. | - Low levels of thyroid hormones - Low insulin sensitivity |
| Enabling replicative immortality | Telomere maintenance and bypassing senescence mechanisms permit unlimited cellular replication. | - Genomic stability |
| Inducing or accessing vasculature | Tumors promote angiogenesis or exploit existing blood vessels to secure oxygen and nutrients. | - Low levels of thyroid hormones - Low insulin sensitivity |
| Activating invasion and metastasis | Cancer cells acquire abilities to invade tissues and colonize distant organs. | - Low levels of thyroid hormones - Low insulin sensitivity - Genomic stability |
| Reprogramming cellular metabolism | Tumor cells adapt metabolic pathways to support growth, survival, and biosynthesis. | - Cool environment - Low levels of thyroid hormones - Low insulin sensitivity - Genomic stability |
| Avoiding immune destruction | Cancer cells evade recognition and elimination by the host immune system. | - Nonspecific cytotoxic cells - Thymus regeneration |
| Non-mutational epigenetic reprogramming | Epigenetic changes, such as DNA methylation or histone modification, alter gene expression without changing the DNA sequence, allowing cancer cells to adapt and progress more rapidly. | - Robust epigenetic characteristics |
| Polymorphic microbiomes | Variations in host-associated microbiota influence tumor initiation, progression, and therapeutic response. | --- |
| Tumor-promoting inflammation | Inflammatory mediators and cells create a microenvironment that supports tumor progression. | - Cool environment - Attenuated adaptive immune response |
| Senescent cells | Senescent cells have been identified as functional contributors to cancer development and progression | --- |
UNANSWERED QUESTIONS
|
UNANSWERED QUESTIONS
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




